Abstract
Halopriming is a salt-based, pre-sowing approach that harmonies metabolic processes required for improving seed quality, enhanced emergence rate and healthy seedling vigour by controlling temperature and seed moisture content in the early stages of germination while preventing the seed from transitioning to complete germination. An aerated aqueous solution of different inorganic salts, viz., KNO3, KH2PO4, KCl, NaCl, CaCl2, Ca(NO3)2, CaSO4, CuSO4, Mg(NO3)2, ZnSO4, etc. alone or in combination is applied to the seeds followed by redrying to their actual weight before sowing into seedbeds to achieve halopriming. Halopriming of seeds prevent seed degradation, breaks dormancy and induces systemic resistance to different abiotic stresses in the seedlings of various crops with amended physiological attributes, improved functional biochemistry and metabolic processes with subcellular changes despite some of its major limitations in terms of prolonged storage of imbibed (pre-soaked) seeds. On priming of the seeds, accumulation of osmolytes, and upregulation of antioxidant enzymes and signaling cascades are achieved in the seeds that persist even after germination in the seedlings as well. The technique has been proved beneficial for improved crop establishment and greater uniformity, increased plant performances with higher agricultural productivity and further for sustainable agricultural practices under the regime of abiotic environmental stresses to meet sustainable future food demand. The current study, therefore, is a compilation of literature to explore different dimensions of a simple, cost-effective, eco-friendly and potent halopriming technique, which could help to integrate seed germination, agricultural production and yield attributes in the pertaining changing climatic conditions.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
As the World’s population increases the global food security seems a great challenge ahead in order to sustain food production. In the era of rapid global climate changes, a wide range of environmental stresses severely affects the biochemical and physiological processes in plants which causes a reduction in agricultural production efficiency by 71% (Qin et al. 2010; Kaya et al. 2013; Petrov et al. 2015; Pirasteh-Anosheh and Hashemi 2020). The level of stress stimulation and the growth stage at which the plants are affected have a direct relationship with the impact of stress on plants. Among the various phases, seed germination and seedling establishment are essential aspects of a plant’s life cycle in both natural and stressful environments (Kumar et al. 2016a). Since seed germination is highly sensitive to changing environmental conditions, therefore, treating seeds chemically or physically before sowing in the agriculture fields is a pre-sowing procedure, commonly known as seed priming has been seen as a viable biological approach that focuses on the management and production of crops to survive them under the stressful conditions (Ashraf et al. 2008). The method of seed priming requires pre-exposure to abiotic stress and treating seeds with some physical or chemical agents for a period of time to induce physiological conditions and bring metabolic changes in the seeds making them more likely to germinate further imparting stress tolerance in the seedlings in terms of increased seedling vitality, root length, photosynthetic efficiency and crop yield (Basra et al. 2005b; Bruggink et al. 1999; Hussain et al. 2016; Ibrahim 2016; Kaur et al. 2005; Karim et al. 2020; Kumar and Rajalekshmi 2021; Lal et al. 2018; Taylor and Harman 1990; Wojtyla et al. 2016).
There are various types of seed priming technology, viz., hydropriming, osmopriming, halopriming, biopriming, solid matrix priming, nutripriming, thermopriming and seed priming with plant growth regulators and other organic sources (Ashraf and Foolad 2005; Eskandari et al. 2013; Jisha et al. 2013; Maiti and Pramanik 2013; Nawaz et al. 2013; Paparella et al. 2015; Sher et al. 2019). Among the numerous seed priming techniques, halopriming is a simple and inexpensive agrotechnique that is simple, cost-effective and ideal for recommending to farmers for improved seedling emergence, germination consistency with greater uniformity, improved plant performance, high vitality, crop stand and higher yield in a variety of environmental circumstances (Ashraf and Rauf 2001; Basra et al. 2005a; Bose and Mishra 1999, Sadeghi and Robati 2015, Zhu and Wang 2008). Halopriming is a long-term biological strategy that involves soaking of seeds in the aerated solutions of inorganic salts viz. calcium chloride (CaCl2), calcium sulphate (CaSO4), copper sulphate (CuSO4), monopotassium phosphate (KH2PO4), magnesium sulphate (MgSO4), potassium chloride (KCl), potassium nitrate (KNO3), sodium chloride (NaCl) and zinc sulphate (ZnSO4) followed by drying and sowing into the agrifields (Table 1). Halopriming induces the pre-germination metabolic pathways by preventing radicle protrusion and decreased emergence time for enhanced nutrient uptake, optimization of germination consistency and germination rate with greater uniformity, improved plant performance, high vitality and crop yield in horticulture, floriculture and field crops even in adverse environmental conditions (Biju et al. 2017; Farooq et al. 2007, 2013; Guo et al. 2012; Iseri et al. 2014; Jisha and Puthur 2014; Nawaz et al. 2013; Patade et al. 2012; Pawar and Laware 2018; Sen and Puthur 2020; Srivastava et al. 2010).
Therefore, prospects, possibilities and mechanisms of halo-priming technology in the agroecosystems have been summarized in the present study.
2 Halopriming and Seed Germination
Seed priming improved the plant’s biochemical profile by raising α-amylase function and soluble sugar content during seed germination even at low temperatures (Anaytullah and Bose 2007). Halopriming has been reported to enhance seed efficiency and seedling vitality of watermelon (Demir and Mavi 2004), rice (Farooq et al. 2006), maize (Kumari et al. 2017), and in several other crops. Seed priming with different inorganic salts such as NaCl, KCl and CaCl2 has a beneficial impact on pepper germination under salinity stress by speeding up imbibition, allowing the seeds to faster metabolic function (Aloui et al. 2014). The salt-sensitive (PI94341) and salt-tolerant (Kharchia 65) genotypes of wheat (Triticum aestivum L.), primed with KCl and NaCl increased germination percentage, triggered physiological attributes such as chlorophyll content and decreases mean germination time (Saddiq et al. 2019). Sesame seeds (Sesamum indicum L.) primed with CaCl2 enhanced germination up to 88.75%, improved vigour and productivity (Tizazu et al. 2019). Halopriming with NaCl proved to be superior in maize and tomato for fast emergence, shoot and root length and other attributes such as photosynthetic pigments (Akter et al. 2018; González-Grande et al. 2020). NaCl priming enhanced breaking off hard seed coat which facilitates 20.56% seedling emergence in okra (Jonah et al. 2019). A better effect on crop productivity and a number of tillers per panicle was observed in different rice (Oryza sativa L.) varieties on priming with NaCl (Riadi et al. 2020).
Halopriming with NaCl and CaCl2 improved germination and seedling growth parameters in maize (Zea mays) as compared to nonprimed seeds under salinity (Gebreegziabher and Qufa 2017). Sorghum seeds (Sorghum bicolor L.) treated with different inorganic salts such as NaCl, KCl and CaCl2 provoked healthy germination and early seedling growth (Chen et al. 2021). Cowpea seeds (Vigna unguiculata (L.) Walp.) primed with CaCl2 improve carbohydrate content, photosynthetic pigments and biomass production (Farooq et al. 2020). Priming of peanut seeds variety (Tainan 9) with CaCl2 showed high germination, increase shoot length, root length and reduced mean germination time (Jeammuangpuk et al. 2020).
KNO3 serves as a nutrient and initiator for pre-germination metabolic events such as increased water imbibition, cell division and elongation, essential germination and other growth activity in seedlings of rice (Dhillon et al. 2021). Priming of rice (Oryza sativa L.) cultivars KDML105 and RD15KDML105 with KNO3 increased imbibition pattern, germination percentage and uniform germination of seedlings (Ruttanaruangboworn et al. 2017). Rice seedlings primed with 2.5 and 5% KNO3 improved emergence percentage by 70 and 94% compared with control (54%) in dry conditions (Ali et al. 2021a). Priming of Capsicum frutescens with NaCl and KNO3 enhances germination potential, final germination percentage, plant length and reduce mean germination time (Robledo 2020). Citrullus lanatus (Thunb.) (Watermelon) seeds and China aster seeds (Callistephus chinensis L.) primed with KNO3 enhanced survival percentage of seeds, germination indices, plant height, shoot and root length, root dry matter and vigour index (Barbosa et al. 2016; Wani et al. 2020). Priming of cotton and soybean seeds with KNO3 improved germination and seed vigour under salt stress conditions (Miladinov et al. 2015; Nazir et al. 2014). Priming with ZnSO4 improved germination percentage, vigour, photosynthetic pigment and sugar content in Momordica charantia seeds (Bukhari et al. 2021). On-farm seed priming of maize and pea with ZnSO4 enhanced the length and number of grains/pods and yield attributes in both crops (Sharma et al. 2021).
3 Halopriming and Abiotic Stress Tolerance
Environmental stresses, such as salinity, temperature extremes (heat and cold), drought, osmotic stress, nutrient deficiency and heavy metal toxicity cause adaptation issues to seedlings and challenge seedling emergence of plumules and radicles, and seedling growth and development leading to yield losses (Ghiyasi et al. 2019; Hasanuzzaman et al. 2012). Seed priming treatment mitigates the detrimental consequences of different abiotic stress (salinity, drought, heat, cold, heavy metals, etc.) responses via modifying the antioxidant metabolism and enhancing the accumulation of osmolytes (Basra et al. 2005a; Guan et al. 2009; Kausar and Ashraf 2003; Kumar et al. 2016a; Saha et al. 2010).
Halopriming with NaCl induces a physiological response in maize seeds that interact with plant stress memory causing the plants to respond swiftly and effectively to upcoming adverse environmental conditions (El-Sanatawy et al. 2021). The improved salt tolerance index (STI) based on growth parameters, such as shoot length, root length and relative water content in NaCl primed seeds were reported in sesame seeds and Vigna mungo L. (Biswas and Saha 2020; Mohammadi et al. 2013). NaCl pretreatments also efficiently reduced the harmful effects of salt stress facilitating effective germination due to increased K and Ca content and decreasing Na accumulation in stems and leaves of Cucumis melo (Sivritepe et al. 2005). Fenugreek seeds (Trigonella foenum-greacum) and tomato (Solanum lycopersicum cv.’Río Grande) primed with NaCl mitigated the deleterious effect of salt stress, significantly (González-Grande et al. 2020; Mohammadi et al. 2013). Sorghum seeds (Sorghum bicolor L.) treated with different concentrations of inorganic salts ameliorated the efficacy of saline stress (Chen et al. 2021). Besides, halopriming has also been reported to be beneficial for enhanced seed germination, seedling growth under drought and induced salt resistance in pepper (Amjad et al. 2007), Triticum aestivum L. (Afzal et al. 2008), sugarcane (Patade et al. 2012), Vigna radiata (L.) Wilczek varieties (Jisha and Puthur 2014) and Cajanus cajan L. (Biswas et al. 2018).
Seed priming with calcium salts has proved to be more efficient and cost-effective in enhancing plant stress resistance (Jafar et al. 2012; Tabassum et al. 2017), as the calcium serves as a secondary messenger in signal transduction (White and Broadley 2003) and promotes osmolyte and antioxidant production in stress conditions (Farooq et al. 2017). Halo-conditioning with CaCl2 enhances crop establishment and confers cis-tolerance on salt-affected lands in wheat (Feghhenabi et al. 2020; Tamimi 2016; Yasmeen et al. 2013) and cowpea (Vigna unguiculata (L.) Walp.; Farooq et al. 2020). Seed priming with CaCl2 increased seedling development, seedling establishment, plant length, grain number, grain weight, grain yield and tillers number under drought stress in wheat (Hussian et al. 2013). It was also elucidated when CaCl2 primed seed supplemented by KCl stimulated salt tolerance in rice varieties that is revealed by increased germination performance, seedling emergence and dry weight under saline conditions (Afzal et al. 2012). The seeds of rice and maize primed with NaCl, KCl, CaCl2 and KNO3 solution enhance resistance against salt stress and exhibited improved growth (Gebreegziabher and Qufa 2017; Theerakulpisut et al. 2016). By priming with inorganic salts NaCl and KCl, salt-sensitive wheat (Triticum aestivum L.) genotype PI.94341 mitigated the negative effects of salinity stress through physiological seed enhancement (Saddiq et al. 2019). Halopriming with CaCl2, KNO3 and NaCl show a significant effect in alleviating salt-induced oxidative stress in rice (Oryza sativa L. cv.) (Riadi et al. 2020; Tahjib-Ul-Arif et al. 2019). In addition to this, the devastating effects of salinity stress on sunflower (Bajehbaj 2010; Kaya et al. 2006) and melon (Oliveira et al. 2019) seeds were reduced by treating them with KNO3. Nigella sativa seeds primed with ZnSO4 and KNO3 increased germination percentage, germination rate and seedling growth under severe water stress (Fallah et al. 2018).
Among various abiotic stressors, Chilling (0–15 °C) is one of the most destructive abiotic stress which severely reduces crop yield (Thakur et al. 2010). Cold stress reduces membrane permeability and disturbs ion transport at the cellular level (Ahmad and Prasad 2011). Consequently, cold stress reduces seed emergence rates, causes seedling failure and accelerates the production of free radical ions (Yu and Rengel 1999). Under chilling stress, halo-priming facilitated the crop establishment and growth by enhancing germination rate and physiological parameters in Chickpea (Cicer arietinum) and hairy vetch (Vicia villosa) (Farooq et al. 2017; Yusefi-Tanha et al. 2019). Peanut seeds primed with CaCl2 showed enhanced storability and faster germination under low-temperature conditions (Jeammuangpuk et al. 2020). Priming with KNO3 followed by drying also allowed cotton seeds to be cold tolerant and produced vigorous seedlings (Cokkizgin and Bölek 2015). The priming of maize and eggplant seeds with Ca(NO3)2 promoted germination rate, radicle protrusion rate index and seedling dry mass under low and high-temperature conditions (Gouveia et al. 2017; Salles et al. 2019). Batista et al. (2016) observed that palisade grass (Brachiaria brizantha cv. MG-5) seeds primed with Ca(NO3)2 and KNO3 improved physiological seed quality resulting in seeds with great stress tolerance under high temperature (Batista et al. 2016).
Heavy metals have been reported to reduce the germination parameters of a developing seedling. Halopriming with Mg(NO3)2 and Ca(NO3)2 reduced the severity of heavy metal (HgCl2) stress in wheat var-HUW-468 during the germination process by increasing endosperm α-amylase activity and soluble sugar content resulting in enhanced germination percentage, seedling emergence and growth performance (Kumar et al. 2016a). Black cumin seeds haloprimed with KNO3 also had the highest germination percentage (100%), plumule length, and dry weight mitigating cadmium toxicity (Espanany et al. 2016). Priming of garden cress (Lepidium sativum) with NaCl and KCl promotes seed germination, boosts seedling growth and develops seedling resistance under hazardous metals stress (Nouri and Haddioui 2021). Besides, different responses of conditioning of seeds, i.e. seed priming in several crops under the regime of different abiotic stresses have also been summarized in Table 1.
4 Halopriming and Anti-oxidative Responses
Seed priming allows morphological, physiological and biochemical changes leading to enhanced stress tolerance and amelioration of the adverse effects of abiotic stress in plants by up-regulating the activity of antioxidant enzymes (Ashraf et al. 2008; Basra et al. 2005b; Bussotti et al. 2014; Chiu et al. 1995; Chang and Sung 1998; Chandra Nayaka et al. 2010; Guan et al. 2009; Kausar and Ashraf 2003; Kumar et al. 2016a; Thakur et al. 2019).
Peroxidases probably played a vital role in reducing oxidative damage and hydrogen peroxide content. An enhanced guaiacol peroxidase content was found to play important role in haloprimed pea (Pisum sativum L.) under chilling effect (Yusefi-Tanha et al. 2019). Halopriming with 125 mM NaCl enhanced germination, antioxidant enzymes (CAT, APX, SOD and POD) and biochemical activities in winged bean [Psophocarpus tetragonolobus (L.) DC.; Kumar and Rajalekshmi (2021)]. NaCl priming of fenugreek seeds (Trigonella foenum-graecum) activated the antioxidant machinery that alleviated negative effects of salt stress and maintain ionic balance (Mahmoudi et al. 2020). Antioxidant enzyme activities (CAT, POD, APX) of wheat (Triticum aestivum L.) cultivars, Lu26s (salt-tolerant) and Lasani-06 (salt-sensitive) were found to be significantly increased after halopriming (Islam et al. 2015). Halopriming with KCl significantly increased the activity of antioxidants in chicory (Sadeghi and Robati 2015). Haloprimed Cowpea seeds (Vigna unguiculata (L.) Walp.) with CaCl2 increases total antioxidants activity up to 22.2% under salt stress (Farooq et al. 2020).
Halopriming greatly increased SOD, CAT, APX and GPOX (guaiacol peroxidase) activities in primed seeds compared to non-primed seeds of different Oryza sativa varieties (Sen and Puthur 2021). KNO3 substantially escalated CAT activity within the FARO44 rice seedlings that scavenged ROS indicated increased tolerance to drought (Ali et al. 2021a, b). Under salinity stress, rice seeds (Oryza sativa L. cv. BRRI dhan29) primed with KNO3 mitigated the adverse effects of reactive oxygen species, i.e. H2O2 and O− radical by up-regulating the activity of antioxidant enzymes such as APX at the seedling stage (Tahjib-Ul-Arif et al. 2019). Priming of Chinese cabbage (Brassica rapa subsp. pekinensis) with KNO3 significantly enhanced POD, SOD and CAT activity under drought stress conditions (Yan 2015). Priming with KNO3 showed increased POD activity in Silybum marianum L. and nitrate reductase activity in tomato under salinity stress (Lara et al. 2014; Zavariyan et al. 2015). KNO3 primed sesame seeds (Sesame indium L.) increased the CAT and POD enzyme activities (Kumar et al. 2016b). Ca(NO3)2 treatment enhanced the activity of various antioxidant enzymes such as CAT, APX, SOD, GR and POD in germinating cucumber seeds (Fan et al. 2017). Halo primed tomato seeds with magnesium nitrate [Mg(NO3)2] significantly increased antioxidant enzyme activities such as SOD, POD, APX, and CAT at different temperature regimes (Nafees et al. 2019). Under water stress circumstances, seed priming of black cumin (Nigella sativa L.) with ZnSO4 increased CAT activity in seedlings (Fallah et al. 2018). Priming with ZnSO4 also increased POD activity significantly in fruits and leaves of Momordica charantia (Bukhari et al. 2021).
5 Halopriming and Production of Osmolytes
Halopriming stimulates seed metabolism by producing osmolytes and/or signalling agents, thus facilitating germination and improved growth and yield performances. Osmolytes are compatible solutes that accumulate within plant cells and act as osmoticum to hold water by lowering the osmotic potential, protecting macromolecules and cell membranes from oxidative stress and dehydration (Fahad et al. 2017; Tabassum et al. 2018; Verslues and Juenger 2011).
NaCl priming in tomato seeds resulted in the accumulation of osmoregulating defence molecules (anthocyanin and proline) and activation of the antioxidative enzyme mechanism (Sahin et al. 2011). Among the three varieties of Vigna radiata, haloprimed Pusa Ratna showed a 50% increase in proline content in normal conditions (Jisha and Puthur 2014). Priming of pepper (Capsicum annuum L.) seeds with CaCl2 resulted in a significant increase in proline content under salinity stress to protect from oxidative stress (Aloui et al. 2014). Halopriming significantly increased leaf proline and phenolic contents under salt stress in both salt-sensitive Lasani-06 and resistant Lu26s wheat cultivars (Islam et al. 2015). Sesame (Sesame indium L.) and rice seeds primed with different concentrations of KNO3 enhanced proline accumulation in seedlings to maintain osmoprotection (Kumar et al. 2016b; Tahjib-Ul-Arif et al. 2019).
Tomato seeds primed with magnesium nitrate (Mg(NO3)2 enhanced production of osmolytes, proline and sugar that preserved plant cell structure and metabolism (Nafees et al. 2019). Halopriming treatment of soybean seeds with ZnSO4 and CaCl2 showed increased soluble sugar and proline content under soda saline-alkali stress (Dai et al. 2017). Priming of Nigella sativa and Momordica charantia seeds with ZnSO4 significantly enhanced proline and phenolic compounds to ameliorate the stress effects (Bukhari et al. 2021; Fallah et al. 2018).
6 Mechanism of Halopriming
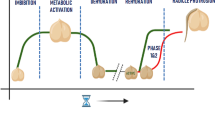
Halopriming is a pre-sowing method that includes exposing the seeds first to a salt solution for a certain period allowing them for partial hydration followed by washing with water and re-drying to original moisture content (Fig. 1). There are two important characteristics of priming such as desiccation tolerance and seed sustainability (Ellis and Hong 1994; Hay and Probert 1995; Gurusinghe and Bradford 2001). Priming enhances the sustainability of low vigour seeds (Varier et al. 2010). The time required for salt treatment is crop-specific and depends upon its variety or cultivar and the priming process activates pre-germinative metabolic activity to start the germination process without radicle emergence (Basra et al. 2003; Bradford 1986; Chen and Arora 2013; Dell’Aquila and Tritto 1991; Giri and Schillinger 2003; Ibrahim 2016; Paparella et al. 2015; Pill 2020). The metabolic activity includes water imbibition, activation of proteins, enzymes, hormones and signal transduction facilitating the breaking of seed dormancy (Ajouri et al. 2004). Under salty alkali stress, halopriming has been linked to a larger osmotic adjustment, more antioxidant defence system activities, more photosynthetic pigment levels, better membrane integrity and more added starch accumulation regulated by biochemical, physiological and molecular mechanisms related to development of embryo, protrusion of plumule and radicle and further with vigorous seedling growth and development (Bewley 1997; Bewley and Black 2013; Lutts et al. 2016; Rajjou et al. 2012).
The process of germination is divided into three stages, viz., imbibition phase (first phase), lag phase (second phase) and growth phase (third phase) (Bewley and Black 1994, 2013; Daszkowska-Golec 2011; Eskandari et al. 2013; Rosental et al. 2014). On priming of the seeds, rapid water absorption facilitates protein synthesis and respiratory activity via mRNA and DNA in the imbibition phase or first phase. Up-regulation of antioxidant enzymes and the accumulation of osmolytes and activation of ROS-mediated signalling pathways have also been documented during the first phase of imbibition (Paparella et al. 2015). The second phase is concerned with controlled water uptake and the start of various physiological processes associated with germination, such as the synthesis of proteins and the availability of soluble sugars (Varier et al. 2010). During the last phase, a marked increase in water intake takes place along with radicle protrusion developed from the seed coat (Bewley and Black 1994; Waqas et al. 2019). Further, the stress resistance of seedlings has been linked to the persistence of the antioxidant mechanism activated and metabolic changes that occurred during the hydration of seeds, even after germination, in the seedlings as well (Paparella et al. 2015). Re-drying is essential for seed storage (Di Girolamo and Barbanti 2012). It prevents quality loss that is caused by severe deterioration of seeds (Di Girolamo and Barbanti 2012; Halmer 2004; Maiti 2011; Parera and Cantliffe 1994; Varier et al. 2010). Priming can also be characterized as a form of preparative germination phase and the lag phase may be extended (Nonogaki et al. 2010). When a primed seed is in the growth phase, it can survive a restoration to its original moisture content without vigour loss thus primed seeds can be stored for a period till the time of sowing (Di Girolamo and Barbanti 2012; Ibrahim 2016; Rajjou et al. 2012; Taylor et al. 1998). However, the primed seeds have a drawback in utilizing the process for commercial purposes with shorter shelf life than seeds that haven’t been primed, so seeds should be processed properly before planting (Ibrahim 2019). Seed priming enhances germination by regulating DNA repair mechanisms, decaying antioxidant scavenging enzymes like catalase, de novo nucleic acid, and protein synthesis, and production of sterols and fatty acids (Afzal et al. 2002; Chen and Arora 2011; Kubala et al. 2015; Paparella et al. 2015; Rajjou et al. 2012). Halopriming can affect seed osmotic balance by increasing K+ and Ca2+ uptake while decreasing Na+ and Cl− absorption thus maintaining cell turgor by balancing membrane potential (Ibrahim 2016; Quintero et al. 2018). Priming triggers ROS scavenging mechanism by enhancing the activity of free radical scavenging enzymes such as catalase (CAT), superoxide dismutase (SOD), peroxidase (POD), ascorbate peroxidase (APX), glutathione reductase (GR) and expression of other stress-responsive proteins that include aquaporins (AQP), dehydrins and late embryogenesis abundant (LEA) proteins (Anaytullah and Bose 2012; Bohnert and Shen 1998; Kumar and Rajalekshmi 2021; Lara et al. 2014; Mittal and Dubey 1995; Sadeghi and Robati 2015; Vander Willigen et al. 2006).
7 Factors Affecting Halopriming
The efficacy of seed priming has been strongly linked with various physical factors such as temperature, aeration, salt concentration, light, priming process, duration of treatment, dehydration after priming, oxygen availability, storage conditions, etc. and certain biological factors like species and its cultivar and variety, age of the seeds, seed vitality and quality (Corbineau and Come 2006; Farooq et al. 2012; Maiti and Pramanik 2013; Parera and Cantliffe 1994). Aeration is the most effective and critical factor that influences seed respiration, seed sustainability and seedling protrusion (Bujalski and Nienow 1991; Heydecker et al. 1973; Fig. 2).
Temperature is another critical factor affecting seed germination. For priming and germination, the optimal temperature varies from 15 to 30 °C (Wahid et al. 2008), however, haloprimed seeds can endure in low and high-temperature regimes. Magnesium nitrate (Mg(NO3)2 primed seeds at 40 °C showed the optimum germination ratio, fresh and dry weight of seedlings, shoot and root length, and overall seedling length up to 10 days (Nafees et al. 2019). Haloprimed seeds of a tomato hybrid variety (Lycopersicon esculentum Mill. NUN 5024) performed well at 40 °C and room temperature (35/28 °C, day/night), but poorly at 10 and 25 °C. Potassium primed seeds of Papaver rhoeas (55.01%) and Papaver dubium (49.61%) showed maximum germination rate on 25 and 20 °C temperatures, respectively, and the minimum germination (7.55%) was observed at 10 °C in either case (Golmohammadzadeh et al. 2020).
Duration of treatment solution also influenced the seed emergence, significantly (Wahid et al. 2008). By increasing the duration of ZnSO4 treatment from 4 to 12 h, the greatest influence on yield attributes was recorded in 12-h duration in maize and pea crops (Sharma et al. 2021). Papaver rhoeas and P. dubium seeds primed for 24 h with 0.5 g/L KNO3 solutions showed maximum seed germination and lowest germination when primed with 4g/L g/L KNO3 for 96 h (Golmohammadzadeh et al. 2020). Soaking of perennial rye seeds at 125 mM NaCl for 24 h enhanced germination percentage (Araghi Shahri et al. 2015). Soaking of three wheat varieties (Sherodi, Fajer, and Taram) for 12, 24, or 36 h in 0.5 or 1% calcium chloride (CaCl2) solutions, 24 h exposure was found to be effective in terms of increased germination percentage in all three varieties in either case (Yari et al. 2012). Priming of China aster seeds (Callistephus chinensis (L.) with KNO3 at 12 and 18 h, though priming duration for 12 h proved to be more favourable for seedling establishment in field conditions (Wani et al. 2020).
The concentration of priming agents is also a crucial factor for enhanced growth attributes in different crops. The optimum concentration of CaCl2 for gerbera was 25 mM and for Zinnia elegans, it was 50 mM CaCl2 (Ahmad et al. 2017). Maize seeds primed with 4000 ppm NaCl had a higher germination index and a shorter mean germination time than seeds primed with 8000 ppm NaCl (El-Sanatawy et al. 2021). Priming with 1.0% KNO3 was more effective to enhance seed emergence and uniform germination of rice cultivars, but priming with 2.0% KNO3 showed a lesser effect on seedlings (Ruttanaruangboworn et al. 2017). Halopriming with different concentrations of KNO3 (10–30 ppm) of Capsicum frutescens seeds, showed the highest influence on radicle and plumule length as well as fresh and dry biomass was observed at 25 ppm KNO3 (Robledo 2020). Among the various concentrations of ZnSO4, priming with 1% ZnSO4 has been shown to be more efficient than priming with 2 and 3% ZnSO4 in maize and pea crops (Sharma et al. 2021).
The type of halopriming agents and cultivar/variety of the seeds are also some other determinants for better growth and development. In various inorganic salts, priming with CaCl2 proved to be stronger than other halopriming agents in mitigating salinity-induced suppression of germination in rice (Oryza sativa L. cv. BRRI dhan29) (Tahjib-Ul-Arif et al. 2019). Haloprimed seeds of two wheat (Triticum aestivum L.) cultivars, Inqlab-91 and SARC-1 with NaCl, CaCl2 and CaSO4 in saline environment showed cultivar dependent differential responses and CaCl2 or CaSO4 proved to be more effective than NaCl as a priming agent in mitigating the adverse effects of salinity on wheat growth (Afzal et al. 2008). Likewise, haloprimed seeds of two rice cultivars, Shaheen Basmati and Basmati 2000 with 2.2% CaCl2 and KCl for 36 h showed optimum results with CaCl2 in terms of increased germination potential, germination rate, seedling growth and salinity resistance in both cultivars (Afzal et al. 2012). Contrary to this, in the case of black seeds (Nigella sativa), of the various priming agents, viz., NaCl, CaCl2, KNO3, CuSO4 and ZnSO4 tested NaCl proved to be the most efficient to combat salt stress in terms of higher germination efficacy and further growth and development (Gholami et al. 2015). The effects of the two halopriming agents, CaCl2 and NaCl on maize physiology were distinct. CaCl2 priming increased the germination process, while NaCl priming enhanced seed maturity and yield (Gebreegziabher and Qufa 2017). In the case of tomato seeds, KNO3 was more successful than the other halopriming agents in increasing final germination, germination rate and seedling vigour (Farooq et al. 2005). Of the different inorganic salts such as NaCl and KCl, CaCl2 proved to be the most efficient halopriming agent for improving Sorghum crop establishment (Chen et al. 2021). Halopriming of Capsicum frutescens with varying concentrations of two inorganic salts, i.e. NaCl and KNO3, KNO3 was found to be more effective than NaCl (Riadi et al. 2020). Pigeon pea seeds were primed with different salts, viz. ZnSO4, CaCl2 and KCl and ZnSO4 showed maximum germination up to 92% (Vanitha and Kathiravan 2019).
8 Conclusion and Future Prospects
Since managing seeds is much easier for farmers in comparison to managing crops in the field, therefore, halo priming has been regarded as a viable remedy for germination issues and in the reduction of the risk of poor stand establishment when seeds are cultivated in adverse environments. The goal of the potent seed priming technique is to synchronize the emergence and protection of seeds against abiotic influences during the crucial seedling establishing phase, resulting in a uniform stand and increased production in the near future. Halopriming technique has proved promising in breaking seed dormancy, improving germination and yield potential of crops and rendering systemic resistance to abiotic stresses in a range of environmental conditions. As a result, halopriming would be a promising technique in the time ahead for sustainable food security and to sustain green agricultural practices with much fewer threats to the environment.
Abbreviations
- %:
-
Percent
- °C:
-
Degree celsius
- APX:
-
Ascorbate peroxidase
- AQP:
-
Aquaporins
- Ca(NO3)2:
-
Calcium nitrate
- Ca++:
-
Calcium ion
- CaCl2:
-
Calcium chloride
- CaSO4:
-
Calcium sulphate
- CAT:
-
Catalase
- Cl−:
-
Chloride ion
- CuSO4:
-
Copper sulphate
- GPOX:
-
Guaiacol peroxidase
- GR:
-
Glutathione reductase
- H2O2:
-
Hydrogen peroxide
- K+:
-
Potassium ion
- KCl:
-
Potassium chloride
- KH2PO4:
-
Potassium dihydrogen phosphate
- KNO3:
-
Potassium nitrate
- LEA:
-
Late embryogenesis abundant
- Mg(NO3)2:
-
Magnesium nitrate
- Na+:
-
Sodium ion
- NaCl:
-
Sodium chloride
- POD:
-
Peroxidase
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- ZnSO4:
-
Zinc nitrate
References
Afzal I, Basra SMA, Ahmad N et al (2002) Effect of priming and growth regulator treatments on emergence and seedling growth of hybrid maize (Zea mays L.). Int J Agric Biol 4:303–306
Afzal I, Rauf S, Basra SMA et al (2008) Halopriming improves vigor, metabolism of reserves and ionic contents in wheat seedlings under salt stress. Plant Soil Environ 54:382–388
Afzal I, Butt A, Ur Rehman H et al (2012) Alleviation of salt stress in fine aromatic rice by seed priming. Aust J Crop Sci 6:1401–1407
Ahmad I, Saleem AM, Mustafa G et al (2017) Seed halopriming enhances germination performance and seedling vigor of Gerbera jamesonii and Zinnia elegans. Sarhad J Agric 33:199–205
Ahmad P, Prasad MNV (2011) Abiotic stress responses in plants: metabolism, productivity and sustainability. Springer Science & Business Media, pp 1–386
Ajouri A, Asgedom H, Becker M (2004) Seed priming enhances germination and seedling growth of barley under conditions of P and Zn deficiency. J Plant Nutr Soil Sci 167:630–636
Akter L, Fakir OA, Alam MK et al (2018) Amelioration of salinity stress in maize seed germination and seedling growth attributes through seed priming. Open J Soil Sci 8:137
Ali LG, Nulit R, Ibrahim MH, Yien CYS (2021a) Efficacy of KNO3, SiO2 and SA priming for improving emergence, seedling growth and antioxidant enzymes of rice (Oryza sativa), under drought. Sci Rep 11:1–11. https://doi.org/10.1038/s41598-021-83434-3
Ali LG, Nulit R, Ibrahim MH, Yien CYS (2021b) Potassium nitrate and silicon dioxide priming improve germination, seedling growth and protective enzymes of rice var. FARO44 under drought. J Plant Nutr 1–14
Aloui H, Souguir M, Latique S et al (2014) Germination and growth in control and primed seeds of pepper as affected by salt stress. Cercet Agronin Mold 47:83–95
Amjad M, Ziaf K, Iqbal Q et al (2007) Effect of seed priming on seed vigour and salt tolerance in hot pepper. Pak J Agric Sci 44:408–416
Anaytullah BB, Bose B (2007) Nitrate-hardened seeds increase germination, amylase activity and proline content in wheat seedlings at low temperature. Physiol Mol Biol Plants 13:199–207
Anaytullah SAK, Bose B (2012) Impact of seed hardening treatment with nitrate salts on nitrogen and antioxidant defense metabolisms in Triticum aestivum L. under different sowing conditions. Vegetos 25:292–299
Araghi Shahri SM, Dianati Tilaki GA, Behtari B, Alizadeh MA (2015) Growth responses of Secale cereale and S. ceremont to priming treatments under salinity stress. J Rangel Sci 5:202–211
Ashraf M, Foolad MR (2005) Pre-sowing seed treatment—a shotgun approach to improve germination, plant growth, and crop yield under saline and non-saline conditions. Adv Agron 88:223–271
Ashraf M, Rauf H (2001) Inducing salt tolerance in maize (Zea mays L.) through seed priming with chloride salts: growth and ion transport at early growth stages. Acta Physiol Plant 23:407–414
Ashraf M, Athar HR, Harris PJC, Kwon TR (2008) Some prospective strategies for improving crop salt tolerance. Adv Agron 97:45–110
Bajehbaj AA (2010) The effects of NaCl priming on salt tolerance in sunflower germination and seedling grown under salinity conditions. Afr J Biotechnol 9:1764–1770
Barbosa WFS, Steiner F, de Oliveira LCM et al (2016) Comparison of seed priming techniques with regards to germination and growth of watermelon seedlings in laboratory condition. Afr J Biotechnol 15:2596–2602
Basra SMA, Farooq M, Khaliq A (2003) Comparative study of pre-sowing seed enhancement treatments in fine rice (Oryza sativa L.). Pak J Life Soc Sci 1:21–25
Basra SMA, Afzal I, Anwar S et al (2005a) Effect of different seed invigoration techniques on wheat (Triticum aestivum L.) seeds sown under saline and non-saline conditions. J Seed Technol 28:36–45
Basra SMA, Farooq M, Tabassam R, Ahmad N (2005b) Physiological and biochemical aspects of pre-sowing seed treatments in fine rice (Oryza sativa L.). Seed Sci Technol 33:623–628. https://doi.org/10.15258/sst.2005.33.3.09
Batista TB, Cardoso ED, Binotti FFDS et al (2016) Priming and stress under high humidity and temperature on the physiological quality of Brachiaria brizantha cv. MG-5 seeds. Acta Sci Agron 38:123–127
Bewley JD (1997) Seed germination and dormancy. Plant Cell 9:1055
Bewley JD, Black M (1994) Cellular events during germination and seedling growth. In: Seeds. Springer, pp 147–197
Bewley JD, Black M (2013) Seeds: physiology of development and germination. Springer Science & Business Media, pp 356–356
Biju S, Fuentes S, Gupta D (2017) Silicon improves seed germination and alleviates drought stress in lentil crops by regulating osmolytes, hydrolytic enzymes and antioxidant defense system. Plant Physiol Biochem 119:250–264
Biswas A, Saha JP (2020) On minimal complements in groups. Ramanujan J 1–25
Biswas S, Biswas AK, De B (2018) Metabolomics analysis of Cajanus cajan L. seedlings unravelled amelioration of stress induced responses to salinity after halopriming of seeds. Plant Signal Behav 13:e1489670
Bohnert HJ, Shen B (1998) Transformation and compatible solutes. Sci Hortic (Amsterdam) 78:237–260
Bose B, Mishra T (1999) Influence of pre-sowing soaking treatment in Brassica juncea seeds with Mg-salts on growth, nitrate reductase activity, total protein content and yield responses. Physiol Mol Biol Plants 5:83–88
Bradford KJ (1986) Manipulation of seed water relations via osmotic priming to improve germination under stress. HortScience 21:1105–1112
Bruggink GT, Ooms JJJ, der Toorn P (1999) Induction of longevity in primed seeds. Seed Sci Res 9:49–53
Bujalski W, Nienow AW (1991) Large-scale osmotic priming of onion seeds: a comparison of different strategies for oxygenation. Sci Hortic (Amsterdam) 46:13–24
Bukhari SA, Farah N, Mahmood S, et al (2021) Effects of Seed priming with zinc sulfate on nutritional enrichment and biochemical fingerprints of Momordica charantia. J Food Qual 2021
Bussotti F, Ferrini F, Pollastrini M, Fini A (2014) The challenge of Mediterranean sclerophyllous vegetation under climate change: from acclimation to adaptation. Environ Exp Bot 103:80–98
Chandra Nayaka S, Niranjana SR, Uday Shankar AC et al (2010) Seed biopriming with novel strain of Trichoderma harzianum for the control of toxigenic Fusarium verticillioides and fumonisins in maize. Arch Phytopathol Plant Prot 43:264–282
Chang SM, Sung JM (1998) Deteriorative changes in primed sweet corn seeds during storage. Seed Sci Technol 26:613–625
Chen K, Arora R (2011) Dynamics of the antioxidant system during seed osmopriming, post-priming germination, and seedling establishment in spinach (Spinacia oleracea). Plant Sci 180:212–220
Chen K, Arora R (2013) Priming memory invokes seed stress-tolerance. Environ Exp Bot 94:33–45
Chen X, Zhang R, Xing Y, et al (2021) The efficacy of different seed priming agents for promoting sorghum germination under salt stress. PLoS One 16:e0245505
Chiu KY, Wang CS, Sung JM (1995) Lipid peroxidation and peroxide-scavenging enzymes associated with accelerated aging and hydration of watermelon seeds differing in ploidy. Physiol Plant 94:441–446
Cokkizgin H, Bölek Y (2015) Priming treatments for improvement of germination and emergence of cotton seeds at low temperature. Plant Breed Seed Sci 71:121–134
Corbineau F, Come D (2006) Priming: a technique for improving seed quality. Seed Test Int 132:38–40
Dai LY, De Zhu H, De Yin K et al (2017) Seed priming mitigates the effects of saline-alkali stress in soybean seedlings. Chil J Agric Res 77:118–125
Daszkowska-Golec A (2011) Arabidopsis seed germination under abiotic stress as a concert of action of phytohormones. Omics J Integr Biol 15:763–774
Dell’Aquila A, Tritto V (1991) Germination and biochemical activities in wheat seeds following delayed harvesting, ageing and osmotic priming. Seed Sci Technol 19:73–82
Demir I, Mavi K (2004) The effect of priming on seedling emergence of differentially matured watermelon (Citrullus lanatus (Thunb.) Matsum and Nakai) seeds. Sci Hortic (Amsterdam) 102:467–473
Dhillon BS, Kumar V, Sagwal P, et al (2021) Seed priming with potassium nitrate and gibberellic acid enhances the performance of dry direct seeded rice (Oryza sativa L.) in North-Western India. Agronomy 11:849
Di Girolamo G, Barbanti L (2012) Treatment conditions and biochemical processes influencing seed priming effectiveness. Ital J Agron 7:e25–e25
Ellis RH, Hong TD (1994) Desiccation tolerance and potential longevity of developing seeds of rice (Oryza sativa L.). Ann Bot 73:501–506
El-Sanatawy AM, Ash-Shormillesy SMAI, Qabil N et al (2021) Seed halo-priming improves seedling vigor, grain yield, and water use efficiency of maize under varying irrigation regimes. Water 13:2115
Eskandari H et al (2013) Effects of priming technique on seed germination properties, emergence and field performance of crops: a review. Int J Agron Plant Prod 4:454–458
Espanany A, Fallah S, Tadayyon A (2016) Seed priming improves seed germination and reduces oxidative stress in black cumin (Nigella sativa) in presence of cadmium. Ind Crops Prod 79:195–204
Fahad S, Bajwa AA, Nazir U et al (2017) Crop production under drought and heat stress: plant responses and management options. Front Plant Sci 8:1147
Fallah S, Malekzadeh S, Pessarakli M (2018) Seed priming improves seedling emergence and reduces oxidative stress in Nigella sativa under soil moisture stress. J Plant Nutr 41:29–40
Fan H, Ding L, Xu Y, Du C (2017) Seed germination, seedling growth and antioxidant system responses in cucumber exposed to Ca(NO3)2. Hortic Environ Biotechnol 58:548–559
Farooq M, Basra SMA, Saleem BA et al (2005) Enhancement of tomato seed germination and seedling vigor by osmopriming. Pak J Agric Sci 42:3–4
Farooq M, Basra SMA, Khalid M et al (2006) Nutrient homeostasis, metabolism of reserves, and seedling vigor as affected by seed priming in coarse rice. Botany 84:1196–1202
Farooq M, Basra SMA, Ahmad N (2007) Improving the performance of transplanted rice by seed priming. Plant Growth Regul 51:129–137
Farooq M, Wahid A, Siddique KHM (2012) Micronutrient application through seed treatments: a review. J Soil Sci Plant Nutr 12:125–142
Farooq M, Irfan M, Aziz T et al (2013) Seed priming with ascorbic acid improves drought resistance of wheat. J Agron Crop Sci 199:12–22
Farooq M, Hussain M, Nawaz A et al (2017) Seed priming improves chilling tolerance in chickpea by modulating germination metabolism, trehalose accumulation and carbon assimilation. Plant Physiol Biochem 111:274–283
Farooq M, Rehman A, Al-Alawi AKM, et al (2020) Integrated use of seed priming and biochar improves salt tolerance in cowpea. Sci Hortic (Amsterdam) 272:109507
Feghhenabi F, Hadi H, Khodaverdiloo H, van Genuchten MT (2020) Seed priming alleviated salinity stress during germination and emergence of wheat (Triticum aestivum L.). Agric Water Manag 231:106022
Gebreegziabher BG, Qufa CA (2017) Plant physiological stimulation by seeds salt priming in maize (Zea mays): Prospect for salt tolerance. Afr J Biotechnol 16:209–223
Ghiyasi M, Moghaddam SS, Amirnia R, Damalas CA (2019) Chemical priming with salt and urea improves germination and seedling growth of black cumin (Nigella sativa L.) under osmotic stress. J Plant Growth Regul 38:1170–1178
Gholami M, Mokhtarian F, Baninasab B (2015) Seed halopriming improves the germination performance of black seed (Nigella sativa) under salinity stress conditions. J Crop Sci Biotechnol 18:21–26
Giri GS, Schillinger WF (2003) Seed priming winter wheat for germination, emergence, and yield. Crop Sci 43:2135–2141
Golmohammadzadeh S, Zaefarian F, Rezvani M (2020) Priming techniques, germination and seedling emergence in two Papaver species (P. rhoeas L. and P. dubium L., Papaveraceae). Brazilian J Bot 43:503–512
González-Grande P, Suárez N, Marín O (2020) Effect of salinity and seed salt priming on the physiology of adult plants of Solanum lycopersicum cv.‘Rio Grande.’ Brazilian J Bot 43:775–787
Gouveia GCC, Binotti FFDS, Costa E (2017) Priming effect on the physiological potential of maize seeds under abiotic stress1. Pesqui Agropecuária Trop 47:328–335
Guan Y, Hu J, Wang X, Shao C (2009) Seed priming with chitosan improves maize germination and seedling growth in relation to physiological changes under low temperature stress. J Zhejiang Univ Sci B 10:427–433
Guo SJ, Wang YC, Wang WS (2012) Effects of priming treatments on germination and biochemical characteristics of Pinus bungeana seeds. For Stud China 14:200–204. https://doi.org/10.1007/s11632-012-0302-3
Gurusinghe S, Bradford KJ (2001) Galactosyl-sucrose oligosaccharides and potential longevity of primed seeds. Seed Sci Res 11:121–134
Halmer P (2004) Methods to improve seed performance in the field. In: Handbook of seed physiology. pp 125–165
Hasanuzzaman M, Hossain MA, da Silva JAT, Fujita M (2012) Plant response and tolerance to abiotic oxidative stress: antioxidant defense is a key factor. In: Crop stress and its management: perspectives and strategies. Springer, pp 261–315
Hay FR, Probert RJ (1995) Seed maturity and the effects of different drying conditions on desiccation tolerance and seed longevity in foxglove (Digitalis purpurea L.). Ann Bot 76:639–647
Heydecker W, Higgins J, Gulliver RL (1973) Accelerated germination by osmotic seed treatment. Nature 246:42–44
Hussain S, Khan F, Cao W et al (2016) Seed priming alters the production and detoxification of reactive oxygen intermediates in rice seedlings grown under sub-optimal temperature and nutrient supply. Front Plant Sci 7:439
Hussian I, Riaz A, Farooq M, Wahid A (2013) Seed priming improves the performance of poor quality wheat seed. Int J Agric Biol 15:1343–1348
Ibrahim EA (2016) Seed priming to alleviate salinity stress in germinating seeds. J Plant Physiol 192:38–46
Ibrahim EA-A (2019) Fundamental processes involved in seed priming. In: Priming and pretreatment of seeds and seedlings. Springer, pp 63–115
Iseri OD, Sahin FI, Haberal M (2014) Sodium chloride priming improves salinity response of tomato at seedling stage. J Plant Nutr 37:374–392
Islam F, Yasmeen T, Ali S et al (2015) Priming-induced antioxidative responses in two wheat cultivars under saline stress. Acta Physiol Plant 37:1–12
Jafar MZ, Farooq M, Cheema MA et al (2012) Improving the performance of wheat by seed priming under saline conditions. J Agron Crop Sci 198:38–45
Jeammuangpuk P, Promchote P, Duangpatra J, et al (2020) Enhancement of Tainan 9 peanut seed storability and germination under low temperature. Int J Agron 2020
Jisha KC, Puthur JT (2014) Halopriming of seeds imparts tolerance to NaCl and PEG induced stress in Vigna radiata (L.) Wilczek varieties. Physiol Mol Biol Plants 20:303–312
Jisha KC, Vijayakumari K, Puthur JT (2013) Seed priming for abiotic stress tolerance: an overview. Acta Physiol Plant 35:1381–1396
Jonah PM, Farauta GK, Kwaga YM et al (2019) Emergence rate of seedlings from hard okra seed coats and seedling growth of some genotypes of West African okra (Abelmoschus caillei). Agric Sci Technol 11:118–122
Karim M, Sani M, Hossain N et al (2020) Stimulatory effect of seed priming as pretreatment factors on germination and yield performance of yard long bean (Vigna unguiculata). Horticulturae 6:104
Kaur S, Gupta AK, Kaur N (2005) Seed priming increases crop yield possibly by modulating enzymes of sucrose metabolism in chickpea. J Agron Crop Sci 191:81–87
Kausar A, Ashraf M (2003) Alleviation of salt stress in pearl millet (Pennisetum glaucum (L.) R. Br.) through seed treatments. Agronomie 23:227–234
Kaya MD, Okçu G, Atak M et al (2006) Seed treatments to overcome salt and drought stress during germination in sunflower (Helianthus annuus L.). Eur J Agron 24:291–295
Kaya C, Sönmez O, Aydemir S, Dikilitaş M (2013) Mitigation effects of glycinebetaine on oxidative stress and some key growth parameters of maize exposed to salt stress. Turkish J Agric For 37:188–194
Kubala S, Garnczarska M, Wojtyla Ł et al (2015) Deciphering priming-induced improvement of rapeseed (Brassica napus L.) germination through an integrated transcriptomic and proteomic approach. Plant Sci 231:94–113
Kumar S, Hemantaranjan A, Mondal S, Bose B (2016) Impact of KNO3 primed seeds on the performance of late sown sesame (Sesamum indicum L.). Int J Bio-Resour Stress Manag 7:950–954
Kumar M, Pant B, Mondal S, Bose B (2016) Hydro and halo priming: influenced germination responses in wheat Var-HUW-468 under heavy metal stress. Acta Physiol Plant 38:1–7
Kumar VK, Rajalekshmi R (2021) Effect of hydro-, halo- and osmopriming on seed germination and seedling performance of Psophocarpus tetragonolobus (L.) DC.(winged bean). J Crop Sci Biotechnol 1–18
Kumari N, Rai PK, Bara BM et al (2017) Effect of halo priming and hormonal priming on seed germination and seedling vigour in maize (Zea mays L.) seeds. J Pharmacogn Phytochem 6:27–30
Lal SK, Kumar S, Sheri V, et al (2018) Seed priming: an emerging technology to impart abiotic stress tolerance in crop plants. In: Advances in seed priming. Springer, pp 41–50
Lara TS, Lira JMS, Rodrigues AC et al (2014) Potassium nitrate priming affects the activity of nitrate reductase and antioxidant enzymes in tomato germination. J Agric Sci 6:72
Lutts S, Benincasa P, Wojtyla L, et al (2016) Seed priming: new comprehensive approaches for an old empirical technique. In: New challenges in seed biology—basic and translational research driving seed technology. pp 1–46
Mahmoudi H, Ben Salah I, Zaouali W et al (2020) Priming-induced changes in germination, morpho-physiological and leaf biochemical responses of fenugreek (Trigonella foenum-graecum) under salt stress. Plant Biosyst Int J Deal with All Asp Plant Biol 154:601–614
Maiti R (2011) Seed priming: an efficient farmers’ technology to improve seedling vigour, seedling establishment and crop productivity. Int J Bio-Resour Stress Manag 2:1997–2000
Maiti R, Pramanik K (2013) Vegetable seed priming: a low cost, simple and powerful techniques for farmers’ livelihood. Int J Bio-Resour Stress Manag 4:475–481
Miladinov ZJ, Balešević-Tubić SN, Djordjevic VB et al (2015) Optimal time of soybean seed priming and primer effect under salt stress conditions. J Agric Sci 60:109–117
Mittal R, Dubey RS (1995) Influence of sodium chloride salinity on polyphenol oxidase, indole 3-acetic acid oxidase and catalase activities in rice seedlings differing in salt tolerance. Trop Sci (United Kingdom) 35:141–149
Mohammadi H et al (2013) Seed germination, seedling growth and enzyme activity of sesame (Sesamum indicum) seed primed under salinity and different temperature conditions. Int J Agron Plant Prod 4:2537–2542
Nafees K, Kumar M, Bose B (2019) Effect of different temperatures on germination and seedling growth of primed seeds of tomato. Russ J Plant Physiol 66:778–784
Nawaz A, Amjad M, Pervez MA, Afzal I (2011) Effect of halopriming on germination and seedling vigor of tomato. Afr J Agri Res 6:3551–3559
Nawaz J, Hussain M, Jabbar A et al (2013) Seed priming a technique. Int J Agric Crop Sci 6:1373
Nazir MS, Saad A, Anjum Y, Ahmad W (2014) Possibility of seed priming for good germination of cotton seed under salinity stress. J Biol Agric Healthc 4:66–68
Nonogaki H, Bassel GW, Bewley JD (2010) Germination—still a mystery. Plant Sci 179:574–581
Nouri M, Haddioui A (2021) Improving seed germination and seedling growth of Lepidium sativum with different priming methods under arsenic stress. Acta Ecol Sin 41:64–71
Oliveira CE da S, Steiner F, Zuffo AM, et al (2019) Seed priming improves the germination and growth rate of melon seedlings under saline stress. Ciência Rural 49
Paparella S, Araújo SS, Rossi G et al (2015) Seed priming: state of the art and new perspectives. Plant Cell Rep 34:1281–1293
Parera CA, Cantliffe DJ (1994) Presowing Seed Priming. Hortic Rev (Am Soc Hortic Sci) 16:109–141
Patade VY, Bhargava S, Suprasanna P (2012) Halopriming mediated salt and iso-osmotic PEG stress tolerance and gene expression profiling in sugarcane (Saccharum officinarum L.). Mol Biol Rep 39:9563–9572
Pawar VA, Laware SL (2018) Seed priming a critical review. Int J Sci Res Biol Sci 5:94–101
Petrov V, Hille J, Mueller-Roeber B, Gechev TS (2015) ROS-mediated abiotic stress-induced programmed cell death in plants. Front Plant Sci 6:69
Pill WG (2020) Low water potential and presowing germination treatments to improve seed quality. In: Seed quality. CRC Press, pp 319–359
Pirasteh-Anosheh H, Hashemi S-E (2020) Priming, a promising practical approach to improve seed germination and plant growth in saline conditions. Asian J Agric Food Sci 8(1)
Qin J, Dong WY, He KN et al (2010) NaCl salinity-induced changes in water status, ion contents and photosynthetic properties of Shepherdia argentea (Pursh) Nutt. seedlings. Plant Soil Environ 56:325–332
Quintero CMF, Castillo OG, Sánchez PD, et al (2018) Relieving dormancy and improving germination of Piquin Chili Pepper (Capsicum annuum var. glabriusculum) by priming techniques. Cogent Food Agric 4:1550275
Rajjou L, Duval M, Gallardo K et al (2012) Seed germination and vigor. Annu Rev Plant Biol 63:507–533
Riadi M, Sjahril R, Syam’un E, et al (2020) Growth and production of three rice varieties (Oryza sativa L.) in saline stress condition following halopriming and hydropriming treatment. In: IOP conference series: earth and environmental science. p 12117
Robledo DAR (2020) Effects of Halopriming on seed germination and seedling emergence of Capsicum frutescens. J Bot Res 3:114–118
Rosental L, Nonogaki H, Fait A (2014) Activation and regulation of primary metabolism during seed germination. Seed Sci Res 24:1–15
Ruttanaruangboworn A, Chanprasert W, Tobunluepop P, Onwimol D (2017) Effect of seed priming with different concentrations of potassium nitrate on the pattern of seed imbibition and germination of rice (Oryza sativa L.). J Integr Agric 16:605–613
Saddiq MS, Iqbal S, Afzal I et al (2019) Mitigation of salinity stress in wheat (Triticum aestivum L.) seedlings through physiological seed enhancements. J Plant Nutr 42:1192–1204
Sadeghi H, Robati Z (2015) Response of Cichorium intybus L. to eight seed priming methods under osmotic stress conditions. Biocatal Agric Biotechnol 4:443–448
Saha P, Chatterjee P, Biswas AK (2010) NaCl pretreatment alleviates salt stress by enhancement of antioxidant defense system and osmolyte accumulation in mungbean (Vigna radiata L. Wilczek). Indian J Exp Biol 48:593–600
Sahin FI, Iseri ÖD, Haberal M (2011) NaCl priming improves salinity response of tomato (Lycopersium esculentum Mill.) at seedling stage. Curr Opin Biotechnol S46–S47
Salles JS, de Lima AH, Binotti F et al (2019) Calcium nitrate priming increases the germination rate of eggplant seeds. J Agric Sci 11:181–186
Sen A, Puthur JT (2020) Influence of different seed priming techniques on oxidative and antioxidative responses during the germination of Oryza sativa varieties. Physiol Mol Biol Plants 26:551–565
Sen A, Puthur JT (2021) Halo and UV-B priming influences various physiological and importantly yield parameters of Oryza sativa var. Vyttila 6. New Zeal J Crop Hortic Sci 49:1–16
Sharma AD, Rathore SVS, Srinivasan K, Tyagi RK (2014) Comparison of various seed priming methods for seed germination, seedling vigour and fruit yield in okra (Abelmoschus esculentus L. Moench). Sci Hort 165:75–81
Sharma M, Parmar DK, Sharma SK (2021) On-farm seed priming with zinc nutrition: a cost effective way to increase the yield of resource poor farmers. J Plant Nutr 1–14
Sher A, Sarwar T, Nawaz A, et al (2019) Methods of seed priming. In: Priming and pretreatment of seeds and seedlings. Springer, pp 1–10
Sivritepe HÖ, Sivritepe N, Erics A, et al (2005) The effects of NaCl pre-treatments on salt tolerance of melons grown under long-term salinity. Sci Hortic (Amsterdam) 106:568–581
Srivastava AK, Lokhande VH, Patade VY et al (2010) Comparative evaluation of hydro-, chemo-, and hormonal-priming methods for imparting salt and PEG stress tolerance in Indian mustard (Brassica juncea L.). Acta Physiol Plant 32:1135–1144
Tabassum T, Farooq M, Ahmad R et al (2017) Seed priming and transgenerational drought memory improves tolerance against salt stress in bread wheat. Plant Physiol Biochem 118:362–369
Tabassum T, Farooq M, Ahmad R et al (2018) Terminal drought and seed priming improves drought tolerance in wheat. Physiol Mol Biol Plants 24:845–856
Tahjib-Ul-Arif M, Afrin S, Polash MAS et al (2019) Role of exogenous signaling molecules in alleviating salt-induced oxidative stress in rice (Oryza sativa L.): a comparative study. Acta Physiol Plant 41:1–14
Tamimi SM (2016) Effect of seed priming on growth and physiological traits of five Jordanian wheat (Triticum aestivum L.) landraces under salt stress. J Biosci Agric Res 11:906–922
Tania SS, Rhaman MS, Hossain MM (2020) Hydro-priming and halo-priming improve seed germination, yield and yield contributing characters of okra (Abelmoschus esculentus L.). Trop Plant Res 7:86–93
Taylor AG, Harman GE (1990) Concepts and technologies of selected seed treatments. Annu Rev Phytopathol 28:321–339
Taylor AG, Allen PS, Bennett MA et al (1998) Seed enhancements. Seed Sci Res 8:245–256
Thakur P, Kumar S, Malik JA et al (2010) Cold stress effects on reproductive development in grain crops: an overview. Environ Exp Bot 67:429–443
Thakur M, Sharma P, Anand A (2019) Seed priming-induced early vigor in crops: an alternate strategy for abiotic stress tolerance. In: Priming and pretreatment of seeds and seedlings. Springer, pp 163–180
Thakur M, Sharma P, Anand A, Pandita VK, Bhatia A, Pushkar S (2020) Raffinose and hexose sugar content during germination are related to infrared thermal fingerprints of primed onion (Allium cepa L.) seeds. Front Plant Sci 11:1502
Theerakulpisut P, Kanawapee N, Panwong B (2016) Seed priming alleviated salt stress effects on rice seedlings by improving Na+/K+ and maintaining membrane integrity. Int J Plant Biol 7:53–58
Tizazu Y, Ayalew D, Terefe G, Assefa F (2019) Evaluation of seed priming and coating on germination and early seedling growth of sesame (Sesamum indicum L.) under laboratory condition at Gondar, Ethiopia. Cogent Food Agric 5:1609252
Vander Willigen C, Postaire O, Tournaire-Roux C et al (2006) Expression and inhibition of aquaporins in germinating Arabidopsis seeds. Plant Cell Physiol 47:1241–1250
Vanitha C, Kathiravan M (2019) Response of pigeonpea to season, halopriming and plant bioregulators intervention in relation to plant physiology and yield potential. Indian J Agric Res 53:190–195
Varier A, Vari AK, Dadlani M (2010) The subcellular basis of seed priming. Curr Sci 450–456
Verslues PE, Juenger TE (2011) Drought, metabolites, and Arabidopsis natural variation: a promising combination for understanding adaptation to water-limited environments. Curr Opin Plant Biol 14:240–245
Wahid A, Noreen A, Basra SMA et al (2008) Priming-induced metabolic changes in sunflower (Helianthus annuus) achenes improve germination and seedling growth. Bot Stud 49:343–350
Wani MA, Khan F, Din A, et al (2020) Elucidating the impact of priming substrates on seedling survival and seed quality of China Aster. In: Biology and life sciences forum. p 34
Waqas M, Korres NE, Khan MD, et al (2019) Advances in the concept and methods of seed priming. In: Priming and pretreatment of seeds and seedlings. Springer, pp 11–41
White PJ, Broadley MR (2003) Calcium in plants. Ann Bot 92:487–511
Wojtyla Ł, Lechowska K, Kubala S, Garnczarska M (2016) Molecular processes induced in primed seeds—increasing the potential to stabilize crop yields under drought conditions. J Plant Physiol 203:116–126
Yan M (2015) Seed priming stimulate germination and early seedling growth of Chinese cabbage under drought stress. S Afr J Bot 99:88–92
Yari L, Sheidaie S, Sadeghi H et al (2012) Evaluation of temperature and seed priming duration on seed germination behavior of rice (Oryza sativa L.). Int J Agric Res Rev 2:7–11
Yasmeen A, Basra SMA, Wahid A et al (2013) Exploring the potential of Moringa oleifera leaf extract (MLE) as a seed priming agent in improving wheat performance. Turk J Bot 37:512–520
Yu Q, Rengel Z (1999) Drought and salinity differentially influence activities of superoxide dismutases in narrow-leafed lupins. Plant Sci 142:1–11
Yusefi-Tanha E, Fallah S, Pessarakli M (2019) Effects of seed priming on growth and antioxidant components of hairy vetch (Vicia villosa) seedlings under chilling stress. J Plant Nutr 42:428–443
Zavariyan AM, Rad MY, Asghari M (2015) Effect of seed priming by potassium nitrate on germination and biochemical indices in Silybum marianum L. under salinity stress. Int J Life Sci 9:23–29
Zhu J, Wang Q (2008) Effects and timeliness of priming on Welsh onion (Allium fistulosum L.) Seeds. Seed 27:32–34
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Gour, T. et al. (2022). Halopriming: Sustainable Approach for Abiotic Stress Management in Crops. In: Roy, S., Mathur, P., Chakraborty, A.P., Saha, S.P. (eds) Plant Stress: Challenges and Management in the New Decade. Advances in Science, Technology & Innovation. Springer, Cham. https://doi.org/10.1007/978-3-030-95365-2_9
Download citation
DOI: https://doi.org/10.1007/978-3-030-95365-2_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-95364-5
Online ISBN: 978-3-030-95365-2
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)