Abstract
The world agricultural scenario has been altered by the increase in temperature, drought, light intensity, variability and rainfall seasonality and chemical modification of the soil solution among other abiotic factors, which have modified the productive bioclimatic zones, hindering the development and reducing the productivity of the plants, causing damage to agribusiness. Understanding the soil-microorganism-plant interaction system allows creating strategies and tools to overcome the challenge of sustainable production. Microbial biotechnology brings together techniques such as inoculation and bioprospecting of microorganisms, intending to provide/induce benefits to plants. This chapter describes the benefits induced by PGPRs in crop production when these are under stress. Predictions of productive gain and possible morphological and biochemical changes can be estimated by the applications of the genomic and proteomic tools, answering gaps left in the understanding of the microorganism-plant interaction. In response to water stress (water deficit and flooding), thermal stress (low and high temperature) and soil chemistry (salinity, soil pH and xenobiotics), various proteins are transcribed and required for the tolerance and resistance process.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Climate change alters abiotic factors, such as periods of drought, precipitation rates, temperatures, evaporation and light intensity, which, associated with the physical and chemical characteristics of the soil, trigger stresses on plants, which consequently can impair development and productivity (Cramer et al. 2011; Ullah et al. 2015; Maxton et al. 2018; Martins et al. 2018). Agricultural production is closely linked to environmental quality, which, when threatened, affects natural resources, of which soil plays an essential role because it is the primary source of the elements necessary for the growth and development of plant species (Akhtar et al. 2020).
2 World Agricultural Sector X Productive Challenges
This productivity is obtained when biotic and abiotic factors, including microbiota, water, light, soil and nutrients, are available and in balance, allowing cultures to express the maximum genetic potential (Cramer et al. 2011; Fan et al. 2018). Genetically improved cultivars are selected to enhance the gene expression of desired characteristics, such as rapid growth; productivity in the volume of grains, fruits, oils, seeds, biomass and wood; stem form; adaptability; quality; and resistance to pests, diseases and abiotic factors, that is, anything that adds commercial value to culture and derived products (Miguel et al. 2016; Fonseca et al. 2017).
The new challenges facing the global agricultural sector are sustainable production, that is, production without harming the environment. This takes into account the change in the global climate panorama that has undergone an intense process of change, according to the reports of the Intergovernmental Panel on Climate Change (IPCC). These reports highlight climate change in the world in recent decades, through rising temperatures and changes in rainfall patterns (IPCC 2007; Ullah et al. 2015). This modification will result in changes in the productive bioclimatic zones (Garcia et al. 2014a, b; Maxton et al. 2018).
3 The Interaction of the Soil System-Microorganism-Plant
The microorganisms and plants’ interaction system has the soil as a common component since it can influence plant and microbial growth, whether in multiplication, survival and even in the metabolic activity present in ecosystems. In the soil, there is a high concentration of microorganisms, especially in the regions close to the roots of the plants. This region is defined as the rhizosphere, which is conceptually considered the portion of soil that is influenced by the roots (Hiltner 1904). Due to its complexity and diversity, the rhizosphere has no measurable shape and size, but a biological and physical-chemical gradient that changes radially and longitudinally around the root (McNear Jr 2013).
The root microbiome enables the presence of microorganisms by being enriched with root exudates, which have a high concentration of components with varying molecular weights, ions, carbon and free oxygen, mucilage and different primary and secondary metabolites. In addition to inhabiting this rich space and benefiting from it, these microorganisms establish ecological relationships with the plant. Among the most important relationships for the agricultural sector are parasitism, commensalism and mutualism (Lebeis 2015; Ullah et al. 2019).
Mutualistic associations allow microbial communities to influence the adaptation, development, health and survival of hosts, with plant-microorganism interactions. This bond may be optional, as is the case of bacteria that promote plant growth, which colonize the rhizosphere, the rhizoplane and even the (endophytic) tissues, which act positively on the plant. These microorganisms can promote plant growth and control pathogens and pests, in addition to serving as anti-stress agents. Also, there are mandatory ones such as mycorrhizae (fungus-root association), whose function is to increase the water and nutrient uptake area (Gray and Smith 2005; Farrar et al. 2014).
Plant growth-promoting rhizobacteria (PGPR) have several physiological mechanisms that contribute to the adaptation and induction of plant species resistance to abiotic stresses caused by edaphoclimatic changes (Kloepper et al. 1989; Kumar et al. 2019; Xia et al. 2020). The PGPR denomination helps to identify bacteria that are beneficial to plants and that consequently are efficient in increasing productivity. There is a range of PGPRs that are tolerant to abiotic stress conditions, which are the most suitable for use and/or applications in agricultural production (Vimal et al. 2017), thus being a sustainable production strategy to reduce the side effects of xenobiotics to the environment (Fig. 2.1).
This productive strategy aims to use the natural relationships between microorganisms and plants, calling this as the study of microbial biotechnology. The techniques employed for this research line have been widely studied, as they are natural, sustainable and economical strategies in the development of production technologies for the industrial and agribusiness sectors (Bianchi et al. 2016; Fukami et al. 2017; Ullah et al. 2019).
The use of microbial biotechnology in the handling of microbial inoculants for plant species has sought the development of new processes and methodologies to quantitatively assess the benefits induced by these microorganisms, such as increased nutritional content, productive gain (production, productivity and mass), tolerance and others. Many of these forms of assessment have been estimated by bioinformatics tools, based on molecular studies, encompassing all “omics” (Genomics, Proteomics, Metabolomics and Transcriptomics), the most used being Genomics and Proteomics (Sarim et al. 2020) (see Sect. 6).
4 PGPRs X Abiotic Stresses: The Main Mechanisms Involved in Resistance
Microbial biotechnology has been studied and applied in the screening of PGPRs that can survive in environments with extreme abiotic conditions, such as water or drought deficit, extreme temperature variations, flooding, salinity, soil acidity and the presence of heavy metals, able to quickly colonize the rhizosphere, mitigate and even induce host resistance (Xia et al. 2020).
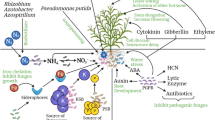
Soil microorganisms, which are capable of directly promoting plant growth (PGP), can provide nutrients and produce phytoregulatory substances and also show how the PGP produced can be evaluated (Fig. 2.2).
The figure summarizes the difference between plants under abiotic stress and inoculated plants, which produce PGPs to combat or induce resistance to abiotic stress. Moreover, it shows the ways to evaluate the efficiency of the inoculation and the benefits of this. On the left is a plant under abiotic stress and the consequences of that stress. In the figure on the right, we can see an inoculated plant, accompanied by the production of PGPs beneficial to the plant, combating abiotic stress and evaluating the efficiency of these PGPs for plants
Plants under prolonged exposure to stress conditions show a significant reduction in AIA (indole-acetic acid) levels. Therefore, inoculation of AIA-producing microorganisms can induce variations in the synthesis of endogenous AIA in the plant (Goswami and Deka 2020). This concentration of AIA, provided by microorganisms, allows roots to develop under stress, as well as photosynthetic production, which is not interrupted by low ethylene production.
The ethylene hormone has its biosynthesis regulated by environmental conditions, which is why it is known as the “stress hormone” (Glick 2014). When in unfavourable conditions, the plant has its ethylene levels altered, and its homeostasis is affected. ACC (aminocyclopropane carboxylic acid) is an immediate precursor to ethylene, which can be degraded by the enzyme ACC deaminase, preventing the production of the hormone. Some bacteria act positively in such situations, as they produce ACC deaminase, thus preventing excessive endogenous ethylene production. Rhizobacteria that produce high rates of ACC deaminase manage to reduce the susceptibility of plants to stressful environments, by stimulating the production of exopolysaccharides (EPS) that are on the surface and bind to cations (such as Na+). This connection results in the formation of films called “biofilm” (Maxton et al. 2018).
There are rhizobacteria capable of producing EPS that help them to survive harsh environments. Exopolysaccharides are formed from long chains of sugar units like glucose, galactose and rhamnose in several portions. They are released by the bacterial cell during its growth and are not bound to the cell, being divided into two groups: homopolysaccharides (formed by monomers of the same species) and heteropolysaccharides (composed of different types of monosaccharides) (Etesami and Maheshwari 2018). EPS have anionic properties due to the acyl group, which also increases its lipophilicity and its interactions with other cations and polysaccharides (Kaushal and Wani 2016). Studies show that EPS production is more pronounced when bacteria are exposed to stress than under normal environmental conditions (Martínez-Gil et al. 2014; Naseem and Bano 2014). These EPS not only help in the survival of bacteria but also play an important role in the tolerance of plants to drought and salinity, as well as in plant growth (Vurukonda et al. 2016).
5 Types of Abiotic Stress
The authors of this chapter have built it by surveying the types of stress present/recurring in annual and perennial crops (fruit and woody). The most described types of abiotic stress are of a water nature, always portrayed as drought, water deficiency and flood that causes anoxia; of a thermal nature that is described through low and high temperatures, in addition to freezing and frosts; of chemical nature of the soil, the saline environment composed of high concentrations of Na+, Ca+2, Mg+2 and K+; and heavy metals, reaching toxic levels and pH, correlated to the concentration of Al+3 present in the soil.
An example of the multifunctionality of PGPR is Bacillus xiamenensis (PM14), proven to be a bacterium that promoted the tolerance of sugar cane (Saccharum officinarum) to salt stress and heavy metals, thermotolerant up to 45 °C with a production of ACC deaminase and EPS production. It additively promotes the resistance of sugarcane to fungal diseases of the root, by presenting 12 antibiotics (Xia et al. 2020).
5.1 Water Nature
Among the types of abiotic stresses, the most worrying are those of a water nature as they are one of the main causes of growth and productivity restriction of plants around the world (Martins et al. 2018). As a result of this stress, there is a reduction in the availability of animal and human food, in addition to losses in the economy (Kumar and Verma 2018). Therefore, looking for strategies that minimize the impact of drought, so that plant production can be maintained, is of great importance to satisfy the global demand for food production (Goswami and Deka 2020).
The result of an environment with a scarcity of water, with restricted and irregular rainfall distribution, is a dry environment. This has the effect of limiting water in the soil or the excessive loss of water by plants through the transpiration process in relation to the absorption of the roots, affecting vital processes such as photosynthesis, respiration, carbohydrate metabolism and ion absorption (Ullah et al. 2018; Akhtar et al. 2020). Water stress has historically reduced the productive yield of vegetables, especially grains and cereals, with rates of up to 10%. This is the result of changes in bioclimatic zones, as a result of climate change. It is estimated that in approximately 2050, productive land will suffer from drought in more than 50% of the areas, which will consequently affect world production (Jochum et al. 2019).
The mechanisms of reaction to water stress are being understood employing the adoption of traditional technologies that are linked to genetic engineering. The area of interest is microbial biotechnology, which uses the interactions between microorganism-plant cultivation (agricultural and forest) aiming to increase the productivity of plant resistance (Card et al. 2016; Silva et al. 2016; Tang et al. 2017; Bilal et al. 2018).
When using Bacillus subtilis, strain GOT9, as a microbial inoculant in Arabidopsis thaliana and Brassica campestris, we observed responses to correlated saline and hydric stress. This statement is proven by the expressive gene expression of these plants. When the plant interacts with this microorganism in a dry environment or in the field, the RD29 and RAB18 genes encode the dehydrin protein, which forms a protective biomolecule during exposure to stress. RD20 is a kaleosin isoform, which, when expressed in greater quantity, increases the tolerance due to the control of the stomatal opening (Woo et al. 2020). Dehydrins are the first proteins identified in plants with low- or high-water potential (dehydration), in saline environments and even at low temperatures (Banerjee and Roychoudhury 2016).
Other responses to water stress occur by reducing the turgidity of plant cells, which induce stomatal closure and consequently cause restriction of photosynthetic rate and cell elongation, managed by the hormonal balance of plants, mainly through the NCED3 gene, which acts on the biosynthesis of abscisic acid (ABA) (Woo et al. 2020). The second response occurs in the roots, where proteins called aquaporins are activated. These proteins are present in the membranes of root cells that absorb water promptly (Gaspar 2011). However, understanding the processes of signalling the plants’ response to a lack of water is very complex, as there is no single universal route considered (Martins et al. 2018).
Research shows that some PGPR can stimulate the production of phytohormones such as endogenous ABA, AIA (Belimov et al. 2015; Forni et al. 2017), gibberellic acid (AG3) (Maxton et al. 2018) and cytokinin (Peleg and Blumwald 2011). The regulation of ABA stimulates the hydraulic conductivity in the roots and also the regulation of aquaporins, being an ally for plants in tolerance to drought (Goswami and Deka 2020). The combination of the production of AIA and AG3 increases the potential for water intake due to the increase in the number of root hairs (Maxton et al. 2018).
Potato plants grown in a greenhouse in different water potentials with and without PGPR inoculation showed a 50% increase in root biomass and 40% in tuber yield when inoculated (Belimov et al. 2015). In the same study, when evaluated in a field experiment, inoculation increased tuber yield by 27%. Tahir et al. (2019) reported that PGPRs inoculated in corn plants in dry condition produced AIA, ACC deaminase and EPS, increasing the water content and chlorophyll content in the leaves. Also, inoculation caused a gain in grain yield, not only in plants under dry conditions but cultivated in ideal field capacity. Chandra et al. (2019) observed that the inoculation of bacteria producing ACC deaminase in wheat plants under water deficit promoted the accumulation of nutrients and grain productivity gain, both in the variety of wheat resistant to drought and in those typically sensitive.
Another vital hormone during drought tolerance is cytokinin, as it delays senescence and premature death of the leaves, which can increase the plant’s yield (Peleg and Blumwald 2011). The origin of the cytokinin can be endogenous by plants or microbial. Alfalfa plants inoculated with Sinorhizobium meliloti, a cytokinin producer, experienced a delay in drought-induced senescence (Xu et al. 2012). When Methylobacterium oryzae was inoculated in lentil plants (Lens culinaris), changes in morphological and physiological patterns were correlated with water use efficiency and cytokinin levels, deferring drought tolerance (Jorge et al. 2019). Prolonged exposure to drought stimulates the production and regulation of phytohormones, osmolytes, EPS and antioxidants by these bacteria, which induce the plant to a greater tolerance to stress, mainly due to morphological changes in the roots (Yang et al. 2009), as described earlier.
Although less frequent than drought, excess water is an abiotic factor that gives stress to the plant, caused by an anoxic habitat. Flooding also influences the productivity of crops, causing physiological disturbances that result in a deficit in growth and agricultural yield (Grichko and Glick 2001; Sairam et al. 2009).
Excessive rainfall and flooding in poorly drained soils are the main situations that can lead plants to root hypoxia or anoxia (Barnawal et al. 2012). Some plants show tolerance to flooding due to the formation of aerenchyma and adventitious roots, induced by the interaction of AIA and ethylene (Ashraf 2019). Plants sensitive to flooding can suffer several damages with the reduction of photosynthesis, closure of stomata, reduction of growth, epinastia and necrosis, which result in the loss of agricultural productivity (Grichko and Glick 2001; Barnawal et al. 2012).
Excessive endogenous ethylene produced in the roots due to flooding is the main inducer of chemical signals and physiological changes that affect all plant tissues (Barnawal et al. 2012). Ethylene, previously described as “stress hormone”, when in ideal concentrations, helps in root initiation, together with AIA, but in high concentrations, there are a decrease in oxygen and an increase in ACC synthase activity that suppress formation and root elongation (Ahmed et al. 2006).
The reduction of the levels of endogenous ethylene in the plant is one of the ways to mitigate the stress caused by flooding. Therefore, the inoculation of PGPR producing ACC deaminase is an economical and ecological alternative that makes the plant more tolerant of flooding, as has been demonstrated in several studies. The ACC deaminase produced by these bacteria synthesizes ACC (immediate precursor to ethylene) in ammonia and α-ketobutyrate (Glick 2014).
Grichko and Glick (2001) evaluated the inoculation of Pseudomonas putida UW4 and Enterobacter cloacae CAL2 in tomato plants under flooding conditions. The study showed that inoculation increased the tolerance of tomatoes, resulting from the activity of bacterial ACC deaminase, which reduced the content and effect of ethylene in plants. Ocimum sanctum subjected to flooding conditions and inoculated with several PGPRs that present desirable growth promotion mechanisms (ACC deaminase, phosphate solubilization, production of AIA and siderophores) had an increase in root and shoot growth and also a reduction in the ethylene content (Barnawal et al. 2012). Nascimento et al. (2012) tested the inoculation with the Mesorhizobium strain transformed with an exogenous ACC deaminase plasmid in chickpea plants (Cicer arietinum) under flooding. The results showed that there was an increase of 127% in nodulation and 125% in biomass of plants inoculated with the transformed strain, showing that ACC plays an important role in the development of microbial inoculants, especially under stress conditions.
5.2 Thermal Nature
Current estimates of climate change suggest that the increase in temperature is a reality in the coming years (IPCC 2007). This increase in temperature will drastically reduce agricultural production on a global scale, as it alters the distribution of productive bioclimatic zones and growing seasons, leading plants to early maturity and consequently to the anticipation of harvest (Porter 2005; Garcia et al. 2014a, b). The stress caused by high temperatures is one of the main limitations during anthesis and grain filling in cereal crops from temperate regions (Ali et al. 2011). In wheat, for example, which has an ideal temperature after anthesis of 15 °C, each 1 °C above can cause a 3 to 5% reduction in grain weight (Wiegand et al. 1981).
High temperatures also cause denaturation, aggregation and inhibition of protein synthesis and inactivation of enzymes in mitochondria and chloroplasts, affecting the fluidity of membrane lipids causing loss of their integrity (Howarth 2005). All of these factors can lead to reduced growth and production of toxic compounds. Given this, the use of easily accessible, low-cost and environmentally friendly methods would be an alternative to minimize the impact of climate change, with the use of PGPRs being a promising path, as has been shown in some studies. This reflects directly on the search for wild PGPR, which inhabits regions with high temperatures to assess the potential in the production of growth promoters.
The low temperature is another limiting factor of productivity and with geographic distribution for many agricultural species. The stress caused by the cold leads to a series of molecular, biochemical, physiological and morphological changes, which are reflected in the fall in productivity (Barka et al. 2006). Cold-resistant plants tend to increase their tolerance to freezing when exposed, due to a phenomenon called cold acclimation (Thomashow 1999).
As previously reported in this chapter, inoculation with PGPR is a great ally of plants in tolerating adverse conditions, including low temperatures. PGPR species with the potential to reduce damage from this type of stress are generally isolated from regions with constant low temperatures or are found in mountainous regions. Bacillus subtilis and Pseudomonas corrugata strains, from the Himalayan region, inoculated in wheat (Tricicum tivum), proved efficient through the growth of plants in development and height, with greater enzymatic activity and phosphorus concentration (Trivedi and Sa 2008; Trivedi et al. 2012). Vine seedlings (Vitis vinifera L.) inoculated with Burkholderia phytofirmans PsJN, between 4 and 26 °C, showed cold tolerance in addition to the increase in seedling biomass (6 and 2.2 times more biomass than in controls at 26 and 4 °C, respectively) and root growth (11.8 and 10.7 times more than in controls at 26 and 4 °C, respectively), with an increase in the content of starch, proline and phenols, to the non-inoculated, demonstrating the growth and development stimulus of the vine by PGPR to withstand cold stress (Barka et al. 2006). In a second step, the use of the inoculant B. phytofirmans PsJN in grapevines increased the concentration of total soluble sugars, starch and sugars related to cold tolerance (glucose, sucrose and raffinose with its precursor, galactinol) (Fernandez et al. 2012). These results were later explained by the increase in metabolic levels and expression rates of genes related to cold stress (Theocharis et al. 2012).
5.3 Soil Chemical Nature
5.3.1 Salinity
Saline-sodium soils are considered to be those with electrical conductivity greater than 4 dS m−1 in the saturation extract in the rhizospheric region and 15% of exchangeable sodium percentage (Sparks 2003). Saline environments cause ionic and osmotic stress, which results in the suppression of plant growth and consequently loss of crop productivity (Baek et al. 2020). This type of stress influences almost 70% of rainfed land in the world (Goswami and Deka 2020), making it the most important factor for the abandonment of agricultural areas (Maxton et al. 2018).
Bacterial EPS shows remarkable performance for plants in dry conditions. However, they can be very favourable in salinity conditions. In these environments, EPS can bind to Na+ free ions due to their anionic property, thus preventing their absorption by the plant (Upadhyay et al. 2012). In a study that evaluated the effect of Pseudomonas putida (strain Rs-198) under conditions of salt stress in cotton plants, it was possible to verify that there was an increase in the uptake of Ca+2, K+ and Mg+2, in addition to the decrease in uptake of Na+ by plants, reducing the negative impact of stress (Yao et al. 2010). Tewari and Arora (2014) found an increase in the growth of sunflower plants under salinity when inoculated with P. aeruginosa. The authors attributed this effect to the production of EPS by the bacterium.
Several studies demonstrate the benefit of inoculating AIA-producing bacteria in plants under salinity conditions. The inoculation of wheat plants with strains of Pseudomonas spp., which produce AIA, increased root growth by 40% and the number of new shoots in salt stress situations by 52% (100 mmol L−1 of NaCl) (Egamberdieva 2009). Rabhi et al. (2018) found that inoculation of Pseudomonas knackmussii, producer of AIA in Arabidopsis thaliana, promoted growth and decreased oxidative stress caused by salinity compared to uninoculated plants.
Saline stress is also capable of inducing the supra-optimum production of ethylene by the plant, causing damage to its development. For this reason, the production of bacterial ACC deaminase acts directly in maintaining plant growth in saline environments (Bal et al. 2013). The application of Pseudomonas fluorescens TDK1 promoted the growth of peanut plants, as well as a greater tolerance to salinity (Saravanakumar and Samiyappan 2007). In wheat plants inoculated with Bacillus mojavensis k78 at different levels of salinity, a positive effect occurred, such as gain in dry mass of roots and shoots, mainly in the highest concentrations of salts (Pourbabaee et al. 2016). In pepper plants, Wang et al. (2018) found that inoculation with Bacillus sp. AWU5 increased the fresh and dry mass, root length and shoot in salinity conditions, compared to uninoculated plants.
In addition to helping plants under salinity conditions through hormonal regulation, rhizobacteria also produce osmolytes, which are organic molecules responsible for balancing the osmotic difference between the cytosol and the external environment. Rhizobacteria can also produce antioxidant substances, which reduce the damage caused by salinity in plants. Jha et al. (2011) observed that co-inoculation with Pseudomonas pseudoalcaligenes and Bacillus pumilus increased salinity tolerance through the production of osmoprotectors and antioxidants in the early stages of rice plant growth. Abd-Allah et al. (2018) inoculated chickpea plants with a strain of Bacillus subtilis, which significantly increased plant growth by modulating the antioxidant system, eliminating oxidative damage caused by salinity.
Plants, when subjected to salt stress, can express genes such as RD29, RD29A and RD29B, which are part of the dehydrin family, proteins responsible for reducing the water conductivity of the roots and inducing the autophagic degradation of aquaporins. The RD20 and NCED3 genes are expressed during water stress (see Sect. 5.1) and, when in greater quantities, increase salinity tolerance (Woo et al. 2020). Other genes expressed, but in Brassica campestris is the SOS1 gene, essential for the occurrence of Na+ and K+ ions homeostasis; and WRKY8, considered as a positive regulator in salt stress, which stimulates the increase in the levels of endogenous ABA and the ABA responsible for stress. Bacillus oryzicola, strain YC7007, when inoculated in Arabidopsis thaliana, induced the plant’s resistance to salinity through the expression of the SOS1 gene, plant growth and increased number of side roots, in addition to greater fresh weight and chlorophyll rates (Baek et al. 2020).
5.3.2 Soil pH
The pH variation depends on the soil formation factors, that is, source material, relief, climate and organisms as a function of time. Another factor that influences is the use of the soil, being the management adopted crucial to maintain the physical-chemical and mainly biological characteristics, which will directly affect the productive potential of the crops, whatever they may be. Therefore, it is possible to obtain acidic soils with a pH below 5.5 and alkaline soils where the pH is above 6.5. This varies depending on the species and its pH tolerance.
Acid soils limit the growth of PGPRs groups and the root system, consequently the development of the aerial part of crops. The main responsibility for the toxicity in acidic soils is the high concentration of aluminium in the form Al+3, which is soluble in water and readily available for the absorption of plants (Sparks 2003). In alkaline soils, exchangeable aluminium is neutralized, as it is converted into species unavailable for root absorption. In addition to aluminium, other nutritional elements in the soil have reduced availability for the plant, such as micronutrients, iron, manganese, cobalt, zinc, boron and selenium, and also macronutrients, nitrogen, phosphorus and sulfur, causing damage to plant development. Also, molybdenum and chlorine have increased availability, making them potentially toxic to plants.
The inoculation of maize (Zea mays) with Bacillus and Burkholderia in soils with pH below 5 increased the length of the roots, between 1.4 and 2 times, respectively, in soils contaminated with aluminium, which reduced by up to 65% the formation of roots in control plants, proving that these microorganisms are efficient in combating the toxic effect of aluminium. However, only plants inoculated with Burkholderia were able to reduce the accumulation of aluminium by 50%, with a 30% increase in phosphorus in the roots, while plants inoculated with Bacillus did not show the same result (Arora et al. 2017). This observation of the inoculation of Bacillus in maize was not observed in wild isolates of the family Bacillaceae (Bacillus and Halobacillus), performed by Banik et al. (2018), which proved the ability of these isolates to tolerate aluminium and acid pH variations (up to 6), to alkaline environments (pH up to 10), enhancing their use in search of the benefits that these microorganisms can provide to plants when used in limited environments.
Phosphorus is one of the main elements whose availability is influenced by soil pH. It is estimated that 70 to 90% of the phosphate applied via mineral fertilization to the soil is immobilized in iron and aluminium oxides in acidic soils and precipitated as calcium phosphate in alkaline soils (Norrish and Rosser 1983; Lindsay et al. 1989).
Insoluble forms of P, such as tricalcium phosphate (Ca3PO4)2, aluminium phosphate (Al3PO4) and iron phosphate (Fe3PO4), can be converted into soluble P by solubilizing organisms that inhabit different soil ecosystems (Song et al. 2008; Sharma et al. 2013). Among the soil bacterial communities, ectorizospheric strains such as Pseudomonas sp., Bacilus sp., Burkholderia sp. and symbionts such as rhizobia have been described as effective phosphate solubilizers (Igual et al. 2001; Song et al. 2008).
The ability of rhizosphere bacteria to solubilize insoluble phosphates is attributed to primary mechanisms such as the excretion of H+ ions, the production of organic acids and the biosynthesis of acid phosphatases (Arcand and Schneider 2006; Richardson and Simpson 2011). Among the organic acids produced, we can mention acetate, lactate, malate, oxalate, succinate, citrate and gluconate, which form complexes with iron or aluminium present in iron and aluminium phosphates, thus making the phosphate present in the soil available for plants (Gyaneshwar et al. 2002). These microorganisms can grow in media with tricalcium phosphate or similar insoluble materials as the only source of phosphate and not only assimilate the element but also solubilize quantities beyond their nutritional requirements, making the surplus available to plants (Chen et al. 2006).
Countless studies have already demonstrated the effect of inoculation of phosphate-solubilizing bacteria on plants. Afzal and Bano (2008) inoculated a rhizobia strain associated with a phosphate solubilizing bacterial strain, in wheat cultivation in a phosphorus-deficient sandy soil. They found an increase in phosphorus absorption, 30 to 40% more than when compared to the treatment that received only fertilizer application. In addition, there was a significant increase in dry root weight, plant height, ear length, grain yield and sugar and leaf protein content.
5.3.3 Heavy Metals
One of the ways to decrease toxicity by heavy metals in plants is through the conversion of these bioavailable elements to inert organisms. Many rhizobacteria can perform this conversion, reducing the toxicity of the element in the rhizospheric region, so that the plant does not absorb them (Etesami and Maheshwari 2018). This is an economical and ecological alternative for the reduction of heavy metals in contaminated environments (Congeevaram et al. 2007; Khan et al. 2012). PGPRs produce organic acids such as citrate, oxalate, gluconic acid, etc., with complexing properties of heavy metals, reducing the potential toxic effect for plants (Archana et al. 2012; Xu et al. 2012). They are also capable of performing bioleaching and/or immobilization by intracellular accumulation and transformation into non-toxic forms by extracellular enzymes (Wani et al. 2009; Khan et al. 2012).
In addition to the direct effects on toxicity, these heavy metal-tolerant microorganisms can promote plant growth through their multiple mechanisms. Organic acids produced by these bacteria can increase the availability of nutrients such as P and K (Patel et al. 2010); in addition to protecting plants from pathogens by synthesizing antimicrobial, cyanogenic and siderophore compounds, they produce AIA, resulting in a better yield of plants grown in contaminated soils (Oves et al. 2013).
Oves et al. (2013) evaluated the inoculation of a chromium-resistant strain (Pseudomonas aeruginosa OSG41) in chickpea plants, grown in the presence of hexavalent chromium. The authors observed that inoculation with the bacteria reduced the uptake of Cr by 36% in the root, 40% in the aerial part and 40% in the grains. Also, there were an increase in dry mass of the aerial part and greater absorption of nutrients (N and P), grain production and nodulation. Moreira et al. (2014) found that inoculation with Ralstonia eutropha reduced cadmium toxicity in corn plants in a greenhouse. The inoculation reduced the accumulation of metal in the plant by immobilization, which led to a low translocation to the aerial part.
A study that evaluated rhizoremediation using the inoculation of Sedum plumbizincicola plants with Bacillus sp. SC2b proved to be efficient in reducing metals such as cadmium and zinc in contaminated soils (Ma et al. 2015). The results of the study showed that the inoculated plants had a significantly higher accumulation of Cd and Zn, demonstrating the effectiveness of inoculating plants with this strain in mobilizing metals for rhizoremediation in contaminated soils. Besides, the bacteria increased the root and shoot biomass and also the leaf chlorophyll content. In the same sense, Shreya et al. (2020) tested the inoculation of Cr-tolerant strains in the cultivation of chickpea plants in a greenhouse, in the presence of Cr. The authors found that inoculation promoted plant growth (increasing root and aerial part biomass and plant length), in the presence of Cr in the soil. It was also reported in the study that inoculation increased phytostabilization, accumulating more Cr both in the root and in the aerial part of the plant, demonstrating the potential as a soil bioremediation agent.
The use of B. subtilis and P. putida in two species of wheat (Triticum aestivum) grown in a greenhouse in two doses, 10 mg kg−1 (low) and 20 mg kg−1 (high) of Cd, presented positive responses with a higher germination rate even in the presence of two heavy metal dosages. It also showed higher growth and lower metal concentration rates in plant tissues, demonstrating the ability of these bacteria to induce resistance to stressful environments, as well as the potential for rhizorremediation (Khatri et al. 2020).
The use of PGPR tends to be successful because they colonize plants subjected to different types of stress. Two bacteria from the Bacillaceae family (Bacillus and Halobacillus) inoculated in peanut seedlings in vitro under saline stress (1% NaCl) and heavy metals (Zn, Al and Pb) showed a positive response to physiological parameters. In addition, plants inoculated with bacteria showed lower concentrations of heavy metals, demonstrating the possible proto-cooperation in supporting resistance to saline and contaminated environments (Banik et al. 2018).
6 Genetic Plant-Microorganism Interaction: An Application of Proteomics in the Detection of Phyto-Beneficial Actions Induced by PGPRs
The use of genomics, transcriptomics and proteomics in the agricultural scenario (according to topic 2) allows the researcher to understand how genes and genetic information of organisms are organized within the genome and how they interact. Through the generation and order of sequences, integration of physical maps, identification of genes and polymorphism, expression of gene products (transcribed) and the structuring of proteins, the researcher can analyze the functional data and predict possible genetic expressions and metabolic routes that are associated with the induction of phyto-beneficial actions of PGPRs in cultivated plants.
The soil-rhizosphere-rhizoplane-endophyte-plant system is a dynamic and coordinated unit that exercises a unique interaction with the components of the environment, generating several phyto-beneficial responses for plant growth (Carvalho et al. 2016). These adaptive responses are correlated with the reciprocal genetic regulation between bacteria and plants during the signaling process, plant colonization and establishment (Dhawi 2020). When we analyze the mutual influence between microorganisms and plants, the understanding of the genetic and biochemical mechanisms that regulate the type of plant-microorganism interaction has not yet been fully elucidated. One of the key points for understanding this interaction is to understand how plants use different signaling pathways to recognize and distinguish beneficial microorganisms from pathogens.
The sequence of steps that occurs during the associative process has demonstrated the existence of the formation of different amounts of transcriptomes in a single individual, which makes it difficult to read sequentially all transcripts; moreover, mRNA abundance is not always well correlated with protein abundance, since the activity of proteins encoded by mRNA is regulated at various levels after expression (Salvato and de Carvalho 2010). The development of proteomic tools has greatly facilitated the application of protein analysis to investigate plant-rhizobacterial interaction (Singh et al. 2017).
Proteins and their functional interactions form a network of connectivity that needs to be considered for a complete understanding of biological phenomena. The STRING platform (https://string-db.org/) is the main database that involves and integrates all publicly available sources on protein-protein interaction information (Szklarczyk et al. 2019). Thus, the BLAST (Basic Local Alignment Search Tool) matrix determines the average percentage similarity between the proteomes, measuring the proportion of gene families conserved and shared between the strains and the total number of gene families within each strain. The absolute number of families of genes shared and combined for each microorganism is displayed at the output of the matrix, determining the number of proteins shared between each proteome.
Transcriptional factor-type proteins function as repressors or gene activators (Poupin et al. 2016) in stressful situations such as anoxia, high temperatures, freezing, water deficit, pH and salinity, among others. PGPR inoculation improves the expression of sLTPs (non-specific lipid transfer protein), which are small basic proteins present in abundance in plants and which are involved in key processes of plant cytology, such as membrane stabilization, cell wall organization and signal transduction (Singh et al. 2017).
The expression of several proteins associated with biotic stress such as ACC deaminase, APx, ArsR, Aux_AIA, kaleosin, carbohydrate kinases (FGGY_C), cafeoil-CoA O-methyltransferase (CCoAOMT), cold shock, dehydrin, FTsZ, phosphodiesterase, glutathione S-transferase (GST), heat shock protein 70 (HSP70), MreB_Mbl, nsLTPs4, sulfatase, thioredoxin H, thaumatin, ubiquitin, ribulose carboxylase large chain, hama-glutamyl-hydrolase (GGH) and vegetative storage glycoprotein (VSP) among others (Tables 2.1 and 2.2), are increased 2 to 10 times more in the plant cell system.
Studies with Pseudomonas sp. inoculated in sorghum seedlings revealed the increased presence of reciprocal stimulation of proteins FtsZ, MreB, Mbl, AcrR and ArsR (Table 2.1) during and after colonization (Dhawi 2020). The use of Enterobacter cloacae in response to salt stress (with NaCl) resulted in the regulation of several proteins such as tubulin, profilin, retinoblastoma, CASP (casparian membrane protein) and xyloglucan endotransglycosylase (Singh et al. 2017). These act in the lignin biosynthesis and acceleration of protein synthesis, strengthening of the cell wall and in the maintenance of the cell structure to prevent damage during the salt stress condition, leading to the synthesis of storage proteins and osmoprotectors. Paenibacillus polymyxa inoculated in watermelon roots induced the expression of caffeoyl-CoAO-methyltransferase (CCoAOMT), glutathione S-transferase (GST) and ubiquitin, which act in the regulation of unwanted proteins, increase the plant’s hardiness and resistance to compression, while it resists damage, metabolic, biotic and abiotic stress involved in growth, photosynthesis and other metabolic and physiological activities (Yaoyao et al. 2017). The use of Bacillus sp. and Arthrobacter pascens also under salt stress resulted in the production of ACC deaminase, bacteriocin and siderophores (Ullah and Bano 2015). Pseudomonas simiae inoculated in soybeans produces positive regulation of the large subunit of RuBisCo, showing an increased abundance of photosynthesis and proteins related to abiotic stress in the expression of VSP and GGH (Vaishnav et al. 2015).
The mRNA expressions of ACS gene transcripts are commonly found in plants located in typically stressful environments, such as nutritional imbalance, heavy metal toxicity, drought and salinity, potentiating the production of ACC oxidase (ACO). The inoculation of PGPRs in several cultures reduces the accumulation of ACO in the roots and increases the expression of ACC deaminase (typically of microbial origin) reducing the deleterious action of ethylene (ACO) (Saikia et al. 2018), inducing morphological changes and biochemical, resulting in systemic tolerance induced to abiotic stresses (Etesami and Maheshwari 2018).
In the ethylene biosynthetic pathway, S-adenosylmethionine is converted by ACC synthase into ACC, the immediate precursor of ethylene, which immediately after the onset of stress produces small peaks of ethylene, consuming the ACC present in plant tissues and probably activating the synthesis of defensive genes within the plant, initiating the inhibitory processes of plant growth and survival, as an example of chlorosis, abscission and senescence, caused by secondary peaks of harmful ethylene. The ACC deaminase produced by PGPRs degrades the plant’s ACC under stress to provide nitrogen and energy, which reduces the harmful effect of ethylene and consequently improves the plant’s stress tolerance (Etesami and Maheshwari 2018).
The bioprospecting of new proteins has revealed major discoveries such as chlorophyll a/b binding protein 40 (cab-BO3–1), known to increase in quantity during development and exposure to light, increased about twice with inoculation of Bacillus subtilis JS in plants of Lactuca sativa and Nicotiana tabacum (Kim et al. 2018). The combined inoculation of Pseudomonas brassicacearum, Bacillus thuringiensis, Bacillus cereus W6 and Bacillus subtilis induced thermotolerance through the production of heat-tolerant proteins of high molecular weight, in addition to increasing the activity of antioxidant enzymes in the plants of Zea mays L. (Ashraf et al. 2019). The identified proteins can be useful for genetic transformation and thus improve the mechanism of tolerance to adverse culture conditions. Corroborating the application of PGPR-based bioinoculant benefits the plant, inducing the expression of several proteins involved in growth, photosynthesis and other metabolic and physiological activities.
7 Conclusions and Future Perspectives
World agriculture is undergoing major changes due to climate change and human pressure. These are capable of modifying agricultural soils, depleting it by the intense exploitation of mineral nutrients that allow for the productive supply required by global food demand. These changes cause a series of abiotic stresses, which are limiting production, such as availability of water (drought, flood), temperature (heat, cold and freezing), salinity, soil acidity and heavy metals, creating an alert about the management of crops. Thus, there is a need to explore more sustainable production systems, in which the exploration of the environment must be balanced and/or more similar to the natural ecological functions, mainly with the reduction of the use of resilient synthetic xenobionts.
In this chapter, we present the success of microbial biotechnology, which through its techniques, can be used as an alternative means by rural producers to overcome the challenges of today’s agriculture. The adoption of the use of biotechnological products of microbial origin in production systems has proven the potential of their use through the processes of bioprospecting of microorganisms in plants and inoculation of PGPRs that benefit plants. The review shows how it is possible to evaluate and quantify the benefits promoted by microorganisms in association with plants, such as increased production, development and plant growth and expressiveness in secondary metabolism. Numerous quali-quantitative tools can be used to evaluate the gains achieved by the use of this technology, such as analysis of gene expression and proteomics, production determination by scale and estimates.
Understanding the beneficial effects of PGPs on the interaction processes that make up the soil-microorganism-plant system points to the need for further research on biotechnological development for microorganisms present in soil and inside plants, to leverage and improve technologies developed over the last few decades. This is necessary since the adaptability of organisms to climate and soil conditions is a key point for the positive effect of microbial inoculants.
The authors believe that the new directions of microbial biotechnology will occur on the bioprospecting of new microorganisms in the production system in locu, which will make it more efficient, production of formulations and techniques for applying mixtures of strains that have distinct and complementary secondary action/metabolisms of action. The prospects for this branch of science are a reality, as technical-scientific and productive advances have proven to be efficient when applied to the most diverse agricultural environments, under different types of management and cultivated agricultural species, with economic gains. These results have leveraged the public and private sectors, stimulating the creation of public policies aimed at promoting and encouraging the research and commercial sectors to create, develop and produce biotechnological products aimed at sustainable agricultural production.
References
Abd-Allah EF, Alqarawi AA, Hashem A, Radhakrishnan R, Al-Huqail AA, Al-Otibi FON, Egamberdieva D (2018) Endophytic bacterium Bacillus subtilis (BERA 71) improves salt tolerance in chickpea plants by regulating the plant defense mechanisms. J Plant Interact 13(1):37–44
Afzal A, Bano A (2008) Rhizobium and phosphate solubilizing Bacteria improve the yield and phosphorus uptake in wheat (Triticum aestivum). Int J Agric Biol 10:85–88
Ahmed S, Nawata E, Sakuratani T (2006) Changes of endogenous ABA and ACC, and their correlations to photosynthesis and water relations in mungbean (Vigna radiata (L.) Wilczak cv. KPS1) during waterlogging. Environ Exp Bot 57(3):278–284
Akhtar SS, Amby DB, Hegelund JN, Fimognari L, Großkinsky DK, Westergaard JC, Roitsch T (2020) Bacillus licheniformis FMCH001 increases water use efficiency via growth stimulation in both normal and drought conditions. Front Plant Sci 11:297
Ali SZ, Sandhya V, Grover M, Linga VR, Bandi V (2011) Effect of inoculation with a thermotolerant plant growth promoting Pseudomonas putida strain AKMP7 on growth of wheat (Triticum spp.) under heat stress. J Plant Interact 6(4):239–246
Arcand MM, Schneider KIMD (2006) Plant- and microbial-based mechanisms to improve the agronomic effectiveness of phosphate rock: a review. Ann Braz Acad Sci 78(4):791–807
Archana G, Buch A, Kumar GN (2012) Pivotal role of organic acid secretion by rhizobacteria in plant growth promotion. In: Microorganisms in sustainable agriculture and biotechnology. Springer, Dordrecht, pp 35–53
Arora P, Singh G, Tiwari A (2017) Effect of microbial inoculation in combating the aluminium toxicity effect on growth of Zea mays. Cell Mol Biol 63(6):79–82
Ashraf A, Bano A, Ali SA (2019) Characterisation of plant growth-promoting rhizobacteria from rhizosphere soil of heat-stressed and unstressed wheat and their use as bio-inoculant. Plant Biol 21(4):762–769
Baek D, Rokibuzzaman M, Khan A, Kim MC, Park HJ, Yun DJ, Chung YR (2020) Plant-growth promoting Bacillus oryzicola YC7007 modulates stress-response gene expression and provides protection from salt stress. Front Plant Sci 10(1646):1–13
Bal HB, Nayak L, Das S, Adhya TK (2013) Isolation of ACC deaminase producing PGPR from rice rhizosphere and evaluating their plant growth promoting activity under salt stress. Plant and Soil 366(1–2):93–105
Banerjee A, Roychoudhury A (2016) Group II late embryogenesis abundant (LEA) proteins: structural and functional aspects in plant abiotic stress. Plant Growth Regul 79(1):1–17
Banik A, Pandya P, Patel B, Rathod C, Dangar M (2018) Characterization of halotolerant, pigmented, plant growth promoting bacteria of groundnut rhizosphere and its in-vitro evaluation of plant-microbe protocooperation to withstand salinity and metal stress. Sci Total Environ 630:231–242
Barka EA, Nowak J, Clément C (2006) Enhancement of chilling resistance of inoculated grapevine plantlets with a plant growth-promoting rhizobacterium, Burkholderia phytofirmans strain PsJN. Appl Environ Microbiol 72(11):7246–7252
Barnawal D, Bharti N, Maji D, Chanotiya CS, Kalra A (2012) 1-Aminocyclopropane-1-carboxylic acid (ACC) deaminase-containing rhizobacteria protect Ocimum sanctum plants during waterlogging stress via reduced ethylene generation. Plant Physiol Biochem 58:227–235
Belimov AA, Dodd IC, Safronova VI, Shaposhnikov AI, Azarova TS, Makarova NM, Tikhonovich IA (2015) Rhizobacteria that produce auxins and contain 1-amino-cyclopropane-1-carboxylic acid deaminase decrease amino acid concentrations in the rhizosphere and improve growth and yield of well-watered and water-limited potato (Solanum tuberosum). Ann Appl Biol 167(1):11–25
Bianchi L, Germino GH, de Almeida SM (2016) Adaptação das plantas ao déficit hídrico. Acta Iguazu 5(4):15–32
Bilal S, Shahzad R, Khan AL, Kang SK, Imran QM, Al-Harrasi A, Yun BW, Lee J (2018) Endophytic microbial consortia of phytohormones-producing fungus Paecilomyces formosus LHL10 and bacteria Sphingomonas sp.LK11 to Glycine max L. regulates physio-hormonal changes to attenuate aluminum and zinc stresses. Front Plant Sci 9:1–18
Card S, Johnson L, Teasdale S, Caradus J (2016) Deciphering endophyte behaviour: the link between endophyte biology and efficacious biological control agents. FEMS Microbiol Ecol 92:1–19
Carvalho TLG, Ballesteros HGF, Thiebaut F, Ferreira PCG, Hemerly AS (2016) Nice to meet you: genetic, epigenetic and metabolic controls of plant perception of beneficial associative and endophytic diazotrophic bacteria in non-leguminous plants. Plant Mol Biol 90(6):561–574
Chandra D, Srivastava R, Gupta VV, Franco CM, Paasricha N, Saifi SK, Sharma AK (2019) Field performance of bacterial inoculants to alleviate water stress effects in wheat (Triticum aestivum L.). Plant and Soil 441(1–2):261–281
Chen YP, Rekha PD, Arun AB, Shen FT, Lai WA, Young CC (2006) Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. Appl Soil Ecol 34(1):33–41
Congeevaram S, Dhanarani S, Park J, Dexilin M, Thamaraiselvi K (2007) Biosorption of chromium and nickel by heavy metal resistant fungal and bacterial isolates. J Hazard Mater 146(1–2):270–277
Cramer GR, Urano K, Delrot S, Pezzotti M, Shinozaki K (2011) Effects of abiotic stress on plants: a systems biology perspective. BMC Plant Biol 11(1):1–14
Dhawi F (2020) Plant growth promoting Rhizobacteria (PGPR) regulated Phyto and microbial beneficial protein interactions. Open Life Sci 15(1):68–78
Egamberdieva D (2009) Alleviation of salt stress by plant growth regulators and IAA producing bacteria in wheat. Acta Physiol Plant 31(4):861–864
Etesami H, Maheshwari DK (2018) Use of plant growth promoting rhizobacteria (PGPRs) with multiple plant growth promoting traits in stress agriculture: action mechanisms and future prospects. Ecotoxicol Environ Saf 156:225–246
Fan C, Yao H, Qiu Z, Ma H, Zeng B (2018) Genome-wide analysis of Eucalyptus grandis WRKY genes family and their expression profiling in response to hormone and abiotic stress treatment. Gene 678:38–48
Farrar K, Bryant D, Cope-Selby N (2014) Understanding and engineering beneficial plant–microbe interactions: plant growth promotion in energy crops. Plant Biotechnol J 12(9):1193–1206
Fernandez O, Theocharis A, Bordiec S, Feil R, Jacquens L, Clément C, Fontaine F, Barka EA (2012) Burkholderia phytofirmans PsJN acclimates grapevine to cold by modulating carbohydrate metabolism. Mol Plant Microbe Interact 25:496–504
Fonseca ES, Peixoto RS, Rosado AS, Balieiro SC, Tiedje JM, Rachid CTCC (2017) The microbiome of Eucalyptus roots under different management conditions and its potential for biological nitrogen fixation. Microb Ecol 75(1):183–191
Forni C, Duca D, Glick BR (2017) Mechanisms of plant response to salt and drought stress and their alteration by rhizobacteria. Plant and Soil 410(1–2):335–356
Fukami J, Ollero FJ, Megías M, Hungria M (2017) Phytohormones and induction of plant-stress tolerance and defense genes by seed and foliar inoculation with Azospirillum brasilense cells and metabolites promote maize growth. AMB Express 7(1):153
Garcia LG, Ferraz, SDB, Alvares CA, Ferraz KDB, Higa RCV (2014a) Modeling the climatic aptitude of Eucalyptus grandis in the face of climate change scenarios in Brazil. Embrapa Florestas-Artigo em periódico indexado (ALICE)
Garcia RA, Cabeza M, Rahbek C, Araújo MB (2014b) Multiple dimensions of climate change and their implications for biodiversity. Science 344(6183):1247579
Gaspar M (2011) Aquaporins: from water channels to multifunctional transporters in plants. Rev Bras Bot 34(4):481–491
Glick BR (2014) Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol Res 169(1):30–39
Goswami M, Deka S (2020) Plant growth-promoting rhizobacteria—alleviators of abiotic stresses in soil: a review. Pedosphere 30(1):40–61
Gray EJ, Smith DL (2005) Intracellular and extracellular PGPR: commonalities and distinctions in the plant–bacterium signaling processes. Soil Biol Biochem 37:395–412
Grichko VP, Glick BR (2001) Amelioration of flooding stress by ACC deaminase-containingplant growth-promoting bacteria. Plant Physiol Biochem 39(1):11–17
Gyaneshwar P, Kumar GN, Parekh LJ, Poole PS (2002) Role of soil microorganisms in improving P nutrition of plants. Plant and Soil 245(1):83–93
Hiltner L (1904) Ueber neuere Erfahrungen und Probleme auf dem Gebiete der Bodenbakteriologie und unter besonderer BerUcksichtigung der Grundungung und Brache. Arb Deut Landw Gesell 98:59–78
Howarth CJ (2005) Genetic improvements of tolerance to high temperature. In: Ashraf M, Harris PJC (eds) Abiotic stresses: plant resistance through breeding and molecular approaches. Howarth Press, New York, pp 277–300
Igual J, Valverde A, Cervantes E, Velázquez E (2001) Phosphate-solubilizing bacteria as inoculants for agriculture: use of updated molecular techniques in their study. Agronomie 21:561–568
IPCC – Intergovernamental Panel on Climate Change (2007) Climate change 2007: mitigation—contribution of working group III to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Jha Y, Subramanian RB, Patel S (2011) Combination of endophytic and rhizospheric plant growth promoting rhizobacteria in Oryza sativa shows higher accumulation of osmoprotectant against saline stress. Acta Physiol Plant 33(3):797–802
Jochum M, McWilliams KM, Borrego E, Kolomiets M, Niu G, Pierson E, Jo YK (2019) Bioprospecting plant growth-promoting rhizobacteria that mitigate drought stress in grasses. Front Microbiol 10:2106
Jorge GL, Kisiala A, Morrison E, Aoki M, Nogueira APO, Emery RN (2019) Endosymbiotic Methylobacterium oryzae mitigates the impact of limited water availability in lentil (Lens culinaris Medik.) by increasing plant cytokinin levels. Environ Exp Bot 162:525–540
Kaushal M, Wani SP (2016) Rhizobacterial-plant interactions: strategies ensuring plant growth promotion under drought and salinity stress. Agric Ecosyst Environ 231:68–78
Khan N, Mishra A, Chauhan PS, Sharma YK, Nautiyal CS (2012) Paenibacillus lentimorbus enhances growth of chickpea (Cicer arietinum L.) in chromium-amended soil. Antonie Van Leeuwenhoek 101(2):453–459
Khatri S, Sharma RK, Shridhar V (2020) Influence of cadmium-tolerant and plant growth-promoting Rhizobacteria on cadmium accumulation and growth response of wheat seedlings under mountain ecosystem. Agric Res 9(1):56–65
Kim JS, Lee JE, Nie H, Lee YJ, Kim ST, Kim SH (2018) Physiological and proteomic analysis of plant growth enhancement by the rhizobacteria Bacillus sp. JS. Genes Genomics 40(2):129–136
Kloepper JW, Lifshitz R, Zablotowicz RM (1989) Free-living bacterial inocula for enhancing crop productivity. Trends Biotechnol 7:39–44
Kumar A, Verma JP (2018) Does plant - microbe interaction confer stresstolerance in plants: a review? Microbiol Res 207:41–52
Kumar A, Patel JS, Meena VS, Ramteke PW (2019) Plant growth-promoting rhizobacteria: strategies to improve abiotic stresses under sustainable agriculture. J Plant Nutr 42(11–12):1402–1415
Lebeis SL (2015) Greater than the sum of their parts: characterizing plant microbiomes at the community-level. Curr Opin Plant Biol 24:82–86
Lindsay WL, Vlek PLG, Chien SH (1989) Phosphate minerals. In: Dixon JB, Weed SB (eds) Minerals in soil environment, 2nd edn. Soil Science Society of America, Madison, WI, pp 1089–1130
Ma Y, Oliveira RS, Nai F, Rajkumar M, Luo Y, Rocha I, Freitas H (2015) The hyper accumulator Sedum plumbizincicola harbors metal-resistant endophytic bacteria that improve its phytoextraction capacity in multi-metal contaminated soil. J Environ Manage 156:62–69
Martínez-Gil M, Ramos-González MI, Espinosa-Urgel M (2014) Roles of cyclic Di-GMP and the Gac system in transcriptional control of the genes coding for the Pseudomonas putida adhesins LapA and LapF. J Bacteriol 196(8):1484–1495
Martins GS, Freitas NC, Máximo WPF, Paiva LV (2018) Gene expression in two contrasting hybrid clones of Eucalyptus camaldulensis x Eucalyptus urophylla grown under water deficit conditions. J Plant Physiol 229:122–131
Maxton A, Singh P, Masih SA (2018) ACC deaminase-producing bacteria mediated drought and salt tolerance in Capsicum annuum. J Plant Nutr 41(5):574–583
McNear DH Jr (2013) The rhizosphere—roots, soil and everything in between. Nat Educ Knowl 4(3):1
Miguel PSB, de Oliveira MNV, Delvaux JC, de Jesus GL, Borges AC, Tótola MR, Costa MD (2016) Diversity and distribution of the endophytic bacterial community at different stages of Eucalyptus growth. Antonie Van Leeuwenhoek 109(6):755–771
Mukhtar T, Smith D, Sultan T, Seleiman MF, Alsadon AA, Ali S, Chaudhary HJ, Solieman THI, Ibrahim AA, Saad MA (2020). Mitigation of heat stress in Solanum lycopersicum L. by ACC-deaminase and exopolysaccharide producing Bacillus cereus: Effects on biochemical profiling. Sustainability, 12(6); 2159
Moreira H, Marques AP, Franco AR, Rangel AO, Castro PM (2014) Phytomanagement of cd-contaminated soils using maize (Zea mays L.) assisted by plant growth-promoting rhizobacteria. Environ Sci Pollut Res 21(16):9742–9753
Nascimento F, Brígido C, Alho L, Glick BR, Oliveira S (2012) Enhanced chickpea growth-promotion ability of a Mesorhizobium strain expressing an exogenous ACC deaminase gene. Plant and Soil 353(1–2):221–230
Naseem H, Bano A (2014) Role of plant growth-promoting rhizobacteria and their exopolysaccharide in drought tolerance of maize. J Plant Interact 9(1):689–701
Norrish K, Rosser H (1983) Mineral phosphate. In: Soils: an Australian viewpoint. Academic Press, CSIRO, Melbourne, London, pp 335–361
Oves M, Khan MS, Zaidi A (2013) Chromium reducing and plant growth promoting novel strain Pseudomonas aeruginosa OSG41 enhance chickpea growth in chromium amended soils. Eur J Soil Biol 56:72–83
Patel KJ, Singh AK, Nareshkumar G, Archana G (2010) Organic-acid-producing, phytate-mineralizing rhizobacteria and their effect on growth of pigeon pea (Cajanus cajan). Appl Soil Ecol 44(3):252–261
Peleg Z, Blumwald E (2011) Hormone balance and abiotic stress tolerance in crop plants. Curr Opin Plant Biol 14(3):290–295
Porter JR (2005) Rising temperatures are likely to reduce crop yields. Nature 436(7048):174–174
Poupin MJ, Greve M, Carmona V, Pinedo I (2016) A complex molecular interplay of auxin and ethylene signaling pathways is involved in Arabidopsis growth promotion by Burkholderia phytofirmans PsJN. Front Plant Sci 7:492
Pourbabaee AA, Bahmani E, Alikhani HA, Emami S (2016) Promotion of wheat growth under salt stress by halotolerant bacteria containing ACC deaminase. J Agric Sci Technol 18(3):855–864
Rabhi NEH, Silini A, Cherif-Silin H, Yahiaoui B, Lekired A, Robineau M, Aït Barka E (2018) Pseudomonas knackmussii MLR6, a rhizospheric strain isolated from halophyte, enhances salt tolerance in Arabidopsis thaliana. J Appl Microbiol 125(6):1836–1851
Richardson AE, Simpson RJ (2011) Soil microorganisms mediating phosphorus availability. Plant Physiol 156:989–996
Saikia J, Sarma RK, Dhandia R, Yadav A, Bharali R, Gupta VK, Saikia R (2018) Alleviation of drought stress in pulse crops with ACC deaminase producing rhizobacteria isolated from acidic soil of Northeast India. Sci Rep 8(1):1–16
Sairam RK, Kumutha D, Ezhilmathi K, Chinnusamy V, Meena RC (2009) Waterlogging induced oxidative stress and antioxidant enzyme activities in pigeon pea. Biol Plant 53(3):493–504
Salvato F, de Carvalho MCCG (2010) Methods and strategies in proteomics and their applications in plants. Cienc Rural 40(3):727–734
Saravanakumar D, Samiyappan R (2007) ACC deaminase from Pseudomonas fluorescens mediated saline resistance in groundnut (Arachis hypogea) plants. J Appl Microbiol 102(5):1283–1292
Sarim KM, Srivastava R, Ramteke PW (2020) Next-generation omics technologies for exploring complex metabolic regulation during plant-microbe interaction. Elsevier, Amsterdam
Sharma SB, Sayyed RZ, Trivedi MH, Gobi TA (2013) Phosphate solubilizing microbes: sustainable approach for managing phosphorus deficiency in agricultural soils. Springerplus 2:587
Shreya D, Jinal HN, Kartik VP, Amaresan N (2020) Amelioration effect of chromium-tolerant bacteria on growth, physiological properties and chromium mobilization in chickpea (Cicer arietinum) under chromium stress. Arch Microbiol:1–8
Silva MCS, Mendes IR, Paula TA, Dias RS, Paula SO, Silva CC, Kasuya MCM (2016) Expression of the nifH gene in diazotrophic bacteria in Eucalyptus urograndis plantations. Can J For Res 46:190–199
Singh RP, Runthala A, Khan S, Jha PN (2017) Quantitative proteomics analysis reveals the tolerance of wheat to salt stress in response to Enterobacter cloacae SBP-8. PLoS One 12(9):e0183513
Song OR, Lee SJ, Lee YS, Lee SC, Kim KK, Choi YL (2008) Solubilization of insoluble inorganic phosphate by Burkholderia cepacia DA23 isolated from cultivated soil. Braz J Microbiol 39(1):151–156
Sparks DL (2003) Environmental soil chemistry. Academic Press, San Diego, p 352
Szklarczyk D, Gable AL, Lyon D, Junge A, Wyder S, Huerta-Cepas J, Jensen LJ (2019) STRING v11: protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res 47(D1):D607–D613
Tahir M, Khalid U, Khan MB, Shahid M, Ahmad I, Akram M, Ahmad N (2019) Auxin and 1-Aminocyclopropane-1-carboxylate deaminase activity exhibiting rhizobacteria improved maize quality and productivity under drought conditions. Int J Agric Biol 21:943–954
Tang Q, Puri A, Padda KP, Chanway CP (2017) Biological nitrogen fixation and plant growth promotion of lodgepole pine by an endophytic diazotroph Paenibacillus polymyxa and its GFP-tagged derivative. Botany 95:611–619
Tewari S, Arora NK (2014) Multifunctional exopolysaccharides from Pseudomonas aeruginosa PF23 involved in plant growth stimulation, biocontrol and stress amelioration in sunflower under saline conditions. Curr Microbiol 69(4):484–494
Theocharis A, Bordiec S, Fernandez O, Paquis S, Dhondt-Cordelier S, Baillieul F, Clément C, Barka EA (2012) Burkholderia phytofirmans PsJN primes Vitis vinifera L. and confers a better tolerance to low nonfreezing temperatures. Mol. Plant Microbe Interact 25:241–249
Thomashow MF (1999) Plant cold acclimation: freezing tolerance genes and regulatory mechanisms. Annu Rev Plant Biol 50(1):571–599
Trivedi P, Sa T (2008) Pseudomonas corrugata (NRRL B-30409) mutants increased phosphate solubilization, organic acid production, and plant growth at lower temperatures. Curr Microbiol 56(2):140–144
Trivedi P, Pandey A, Palni LMS (2012) Bacterial inoculants for field applications under mountain ecosystem: present initiatives and future prospects. In: Maheshwari DK (ed) Bacteria in agrobiology: plant probiotics. Springer, Berlin, pp 15–44
Ullah S, Bano A (2015) Isolation of PGPRS from rhizospheric soil of halophytes and its impact on maize (Zea mays L.) under induced soil salinity. Can J Microbiol 61(4):307–313
Ullah A, Heng S, Munis MFH, Fahad S, Yang X (2015) Phytoremediation of heavy metals assisted by plant growth promoting (PGP) bacteria: a review. Environ Exp Bot, v. 117: 28–40.
Ullah A, Sun H, Yang X, Zhang X (2018) A novel cotton WRKY gene, GhWRKY6-like, improves salt tolerance by activating the ABA signaling pathway and scavenging of reactive oxygen species. Physiol Plant 162:439–454
Ullah A, Nisar M, Ali H, Hazrat A, Hayat K, Keerio AA, Khan A (2019) Drought tolerance improvement in plants: an endophytic bacterial approach. Appl Microbiol Biotechnol 103(18):7385–7397
Upadhyay SK, Singh JS, Saxena AK, Singh DP (2012) Impact of PGPR inoculation on growth and antioxidant status of wheat under saline conditions. Plant Biol 4(4):605–611
Vaishnav A, Kumari S, Jain S, Varma A, Choudhary DK (2015) Putative bacterial volatile-mediated growth in soybean (Glycine max L. Merrill) and expression of induced proteins under salt stress. J Appl Microbiol 119(2):539–551
Vimal SR, Singh JS, Arora NK, Singh S (2017) Soil-plant-microbe interactions in stressed agriculture management: a review. Pedosphere 27(2):177–192
Vurukonda SSKP, Vardharajula S, Shrivastava M, SkZ A (2016) Enhancement of drought stress tolerance in crops by plant growth promoting rhizobacteria. Microbiol Res 184:13–24
Wang W, Wu Z, He Y, Huang Y, Li X, Ye BC (2018) Plant growth promotion and alleviation of salinity stress in Capsicum annuum L. by Bacillus isolated from saline soil in Xinjiang. Ecotoxicol Environ Saf 164:520–529
Wani PA, Zaidi A, Khan MS (2009) Chromium reducing and plant growth promoting potential of Mesorhizobium species under chromium stress. Biorem J 13(3):121–129
Wiegand CL, Gerbermann AH, Cuellar JA (1981) Development and yield of hard red winter Wheats under semitropical conditions 1. Agron J 73(1):29–37
Woo OG, Kim H, Kim JS, Keum HL, Lee KC, Sul WJ, Lee JH (2020) Bacillus subtilis strain GOT9 confers enhanced tolerance to drought and salt stresses in Arabidopsis thaliana and Brassica campestris. Plant Physiol Biochem 148:359–367
Xia Y, Farooq MA, Javed MT, Kamran MA, Mukhtar T, Ali J, Chaudhary HJ (2020) Multi-stress tolerant PGPR Bacillus xiamenensis PM14 activating sugarcane (Saccharum officinarum L.) red rot disease resistance. Plant Physiol Biochem 151:640–649
Xu J, Li XL, Luo L (2012) Effects of engineered Sinorhizobium meliloti on cytokinin synthesis and tolerance of alfalfa to extreme drought stress. Appl Environ Microbiol 78(22):8056–8061
Yang J, Kloepper JW, Ryu CM (2009) Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci 14(1):1–4
Yao L, Wu Z, Zheng Y, Kaleem I, Li C (2010) Growth promotion and protection against salt stress by Pseudomonas putida Rs-198 on cotton. Eur J Soil Biol 46(1):49–54
Yaoyao E, Yuan J, Yang F, Wang L, Ma J, Li J, Shen Q (2017) PGPR strain Paenibacillus polymyxa SQR-21 potentially benefits watermelon growth by re-shaping root protein expression. AMB Express 7(1):1–12
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Monteiro, P.H.R., da Silva, F.B., de Abreu, C.M., da Silva, G.J. (2021). Plant Growth Promoting Rhizobacteria in Amelioration of Abiotic Stresses: A Functional Interplay and Prospective. In: Mohamed, H.I., El-Beltagi, H.ED.S., Abd-Elsalam, K.A. (eds) Plant Growth-Promoting Microbes for Sustainable Biotic and Abiotic Stress Management. Springer, Cham. https://doi.org/10.1007/978-3-030-66587-6_2
Download citation
DOI: https://doi.org/10.1007/978-3-030-66587-6_2
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-66586-9
Online ISBN: 978-3-030-66587-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)