Abstract
Actinobacteria is one of the largest phyla within the domain Bacteria. This phylum comprises more than 400 genera heterogeneously distributed in up to 50 families, 20 orders and 6 classes, being composed with very diverse groups of microorganisms. Members included within this phylum were recovered from a wide range of aquatic and terrestrial environments and also from a huge number of higher organisms, including plants. Actinobacteria inhabiting soils and plants are well known as producers of bioactive molecules and as biocontrol agents, possessing antimicrobial activities mostly against pathogenic fungi and/or bacteria. Moreover, some of them have the capacity to exert beneficial effects on plant growth and development via different plant growth-promoting mechanisms, i.e., phytohormones biosynthesis, siderophore production, and phosphate solubilization, among others. The available genomic data revealed that members belonging to this phylum have a huge potential as Plant Probiotic Actinobacteria. A plethora of studies reported the isolation and identification of plant endophytic actinobacteria possessing those features and also their performance under controlled conditions. However, few studies show the effects of the inoculation of these actinobacteria on real field conditions. In this chapter, we will provide an overview of the available data on the Actinobacteria displaying plant growth-promoting features, particularly in the ones that already had applications in agriculture. Together with a correct taxonomic classification, we will present evidence that the Plant Probiotic Actinobacteria should be considered as a source of bacterial candidates that will be important for a future sustainable agriculture.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
15.1 Introduction
The Actinobacteria is a phylum of Gram-positive bacteria and one of the largest taxonomic units within the domain Bacteria (Barka et al. 2015). The majority of the Actinobacteria are free-living organisms, being well known for their ubiquitous presence in soil and aquatic habitats and their contribution to organic material recycling. Between these bacteria, we found indeed some of the most well-known producers of antibiotics, exemplified by the genera Streptomyces, Micromonospora and Actinomadura (Raja and Prabakarana 2011). The Actinobacteria establishes close relationships with their environment and the organisms of their surroundings, with key molecular exchanges that allow their coexistence. Within the phylum, we found pathogenic bacteria for humans (Mycobacteria, Nocardia, or Tropheryma), for plants (Streptomyces scabiei, which cause scab in potatoes), and for animals (Corynebacterium, Mycobacteria). However, beneficial actinobacteria are also found for all these organisms, Bifidobacterium being well known for their implication in human and animal health, Pseudonocardia for the protection of ant’s gardens, or Frankia for their symbiotic relationship with actinorhizal plants. New studies on bacterial communities have shown that Actinobacteria composition is related to plant health (Wang et al. 2017), inducing a new interest in the study of Actinobacteria’s role as plant endophytes. This is clearly remarkable in the number of new species described from plant tissues in the last 10 years (Table 15.1), with more than ten new species published on the International Journal of Systematic and Evolutionary Microbiology only in the last 3 months (August–October 2018). High numbers of actinobacterial taxa found in healthy plant tissues have compelled us to think that these microorganisms have the capacity to improve plant health and could act as plant probiotics.
The FAO/WHO Expert Consultation Report defines Probiotics as “live microorganisms which when administered in adequate amounts confer a health benefit on the host” (Hill et al. 2014). Consequently, plant probiotics should be defined as “live microorganisms which when administered in adequate amounts confer a health benefit on the plant”. Between the actinobacteria, Frankia genus could be considered the first and most studied plant probiotic actinobacteria. This genus has been studied for more than a century due to its ability to fix atmospheric nitrogen, which is exchanged with the plants with which it establishes symbioses (Beijerinck 1901; Carro et al. 2015). Presence of Frankia strains has been also related to improvements in stress tolerance, as high salinity concentration (Ngom et al. 2016) or soil degradation (Diagne et al. 2013). Nevertheless, many other genera have been included in the list of Plant Probiotic Actinobacteria in the last years, exemplified by Streptomyces, which have been shown to improve plant vegetative growth and to induce and contribute to plant defense from pathogen attacks (Conn et al. 2008); by Micromonospora, which are able to improve plant growth and the tripartite symbioses with rhizobia in legumes (Carro 2010; Martínez-Hidalgo et al. 2014); or by Arthrobacter, which are able to increase iron-stress resistance (Sharma et al. 2016). Most of the actinobacteria tested as plant probiotic bacteria have been directly inoculated on plants, in most of the cases to evaluate the protection against some pathogenic microorganisms. However, contrary to other bacteria, many of them have not been tested for the general characteristic evaluated to determine a plant growth-promoting bacteria (PGPB): nitrogen fixation capacity, phosphate solubilization, production of plant hormones (IAA, ACC desaminase), etc. In this chapter, an overview of Actinobacteria known as plant growth promoters will be given, with emphasis on their taxonomic position and their use in agriculture.
15.2 Current Taxonomy of Actinobacteria: Classic and NGS-Based Classification
The taxonomic status of a strain, according to the polyphasic taxonomy, is determined by both phenotypic and genotypic characterization. A combination of chemotaxonomic analysis and other phenotypic features (tolerance tests, enzyme production, ability to metabolize carbon and nitrogen sources) together with other genetic traits of the taxon (16S rRNA phylogeny, GC content, DNA–DNA hybridization) was classically used for new actinobacteria species descriptions (Carro and Nouiuoi 2017). The use of multilocus sequences analyses (MLSA) greatly improved the relationships between these new isolates (Carro et al. 2012) and the upstream taxa, as exemplified by the analysis done by Adekambi et al. (2011). Lately, the new sequencing technologies developed and its availability for the vast majority of researchers have introduced new methods for phylogenomic reconstructions, allowing a better classification regarding higher taxa never seen before. Specifically, the works developed by Sen et al. (2014) for the class Actinobacteria and Nouioui et al. (2018c) for the whole phylum have greatly rearranged their respective status.
The phylum Actinobacteria was first described by Cavalier-Smith (2002) and include six classes: Acidimicrobiia (Norris 2012), Actinobacteria (Stackebrandt et al. 1997), Coriobacteriia (König 2012), Nitriliruptoria (Ludwig et al. 2012), Rubrobacteria (Suzuki 2012) and Thermoleophilia (Suzuki and Whitman 2012). From these classes, 450 genera are unequally distributed, the majority of them (418) being within the class Actinobacteria. Endophytic bacteria have been described only in the class Actinobacteria and in the class Rubrobacteria. This latter class just comprises one plant-associated species, Solirubrobacter phytolaccae (Wei et al. 2014).
After last reclassification based of whole-genome sequences (Nouioui et al. 2018c), the class Actinobacteria comprises 20 orders: Acidothermales, Actinomycetales, Bifidobacteriales, Catenulisporales, Corynebacteriales, Cryptosporangiales, Frankiales, Geodermatophilales, Glycomycetales, Jiangellales, Kineosporiales, Micrococcales, Micromonosporales, Nakamurellales, Nitriliruptorales, Propionibacteriales, Pseudonocardiales, Sporichthyales, Streptomycetales and Streptosporangiales. Most of these orders contain endophytic strains; only in six of them, no plant related strains have been isolated (Acidothermales, Actinomycetales, Bifidobacteriales, Catenulisporales, Nitriliruptorales and Sporichthyales). Strains belonging to those orders are related to human samples or extreme habitats. All the other orders contain genera in which some or most of their species have been described as plant endophytes (isolated from within the plant tissues). Among them, the most important genera of plant pathogens are mainly found in the order Corynebacteriales, including Corynebacterium, Nocardia, and Rhodococcus; in the order Micrococcales, including Clavibacter, Curtobacterium, Leifsonia, and Rathayibacter; and in the order Streptomycetales, including some species of the genus Streptomyces, such as the phytotoxin-producer S. scabies (Lozi 1994). Although several species of these genera have been found to be pathogenic, in most cases other species within the same genus have been described as nonpathogenic endophytes or even plant growth-promoting bacteria, i.e., the strain BMG51109 of Nocardia (Ghodhbane-Gtari et al. 2018) or the strain SK68 of Streptomyces (Damodharan et al. 2018). Although not an exact distribution between pathogen and PGPB could be established between the genera of Actinobacteria, some relationships could be observed mainly due to these double functions of some genera (Fig. 15.1). Most of the pathogens appear in genera from the family Microbacteriaceae of the order Micrococcales, while Frankiales, Jiangellales or Micromonosporales include mainly PGPB or asymptomatic endophytic strains.
15.3 Genomes Data Mining of PGP Traits on Actinobacteria
New technologies have encouraged the research of genes from plant endophytes related to their abilities for plant growth promotion, generating full sets of candidate genes to be further analyzed due to their potential. Trujillo et al. (2014) and Carro et al. (2018a) identified some of these genes in several species of the genus Micromonospora, including genes related to plant hormones production, phosphate solubilization, or siderophores production, among the most common ones, and also genes related to the biosynthesis of trehalases or other degrading enzymes (amylases, cellulases, chitinases, pectinases, and xylanases), among the most interesting ones for biotechnological applications.
Most of the plant endophyte genomes have been shown to harbor a whole set of genes for central carbohydrate metabolism that could be related to the utilization of root exudates as energy source (Kang et al. 2016). Other frequently found genes include the ones related to nutrient deficiencies, oxidative stress, drought tolerance, as well as secretion mechanisms and signaling (Trujillo et al. 2015). Genes related to biosynthesis pathways of plant growth modulators, such as auxins and cytokinins, are generally found in most plant probiotic bacteria, which combined with others related to degradation of ethylene through 1-amino-cyclopropane-1-carboxylic acid deaminase generate further improvements of plant status under stress conditions. Some genes that contribute to efficient colonization and competitiveness are also important in first steps of interactions (Francis et al. 2016).
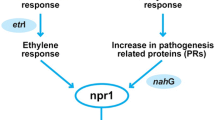
Genome analysis has also put in evidence the importance of actinobacteria as secondary metabolites producers and its possible use in agriculture for biological control. The production of several peptides and antibiotics observed in actinobacteria probiotics could be used to defend the host plant against pathogens (Paterson et al. 2017; Remali et al. 2017). The mechanism of biocontrol also involved induction of plant defense response by, for example, the upregulation of PR10a, NPR1, PAL, and LOX2 genes in colonized plants by Streptomyces (Patel et al. 2018).
15.4 Applications of Plant Probiotic Actinobacteria in Agriculture
The members of the phylum Actinobacteria have a huge and well-appreciated range of biotechnological applications. As we have seen before, the metabolic potential and the biological significance of several groups of actinobacteria are well known, which are of paramount importance in the biotech industries, mostly related to biomedicine (Golinska et al. 2015; Barka et al. 2015; Passari et al. 2017). Actinobacteria associated with plants, namely endophytic actinobacteria, have been studied for its application in agriculture (Palaniyandi et al. 2013), mainly in biocontrol and suppression of plant diseases and, in some cases, in plant growth promotion (Ganapathy and Natesan 2018; Singh and Dubey 2018) (Table 15.2). However, studies showing the effects of Plant Probiotic Actinobacteria on crop yields are still scarce (Viaene et al. 2016; Araujo et al. 2017). Some of those works are enumerated in Table 15.3.
Among Actinobacteria, the streptomycetes are one of the most abundant bacterial groups in soils, accounting for up to 10% of the total microbiome (Janssen 2006). The genus Streptomyces is the most studied genus within the phylum Actinobacteria, not only due to its uncountable properties but also because of the versatility of the species within this genus (Viaene et al. 2016).
The vast majority of the studies about the potential of Streptomyces strains as plant growth promoters and biocontrollers present effects under in vitro controlled conditions due to its innate ability to produce secondary metabolites (including antibiotic and antimicrobial compounds). Strains belonging to different species of the genus Streptomyces isolated from wheat rhizosphere and root endosphere showed several activities, such as chitinase and phytase activities, as well as phosphorous solubilization. These strains are also able to produce different compounds, such as IAA, siderophores, organic acids and antifungal metabolites (Jog et al. 2014). Wheat plants in growth chamber (lab-controlled conditions) inoculated with Streptomyces strains showed higher plant biomass, number of lateral roots and branches, and nutritional content (essential elements) in comparison with uninoculated control plants (Jog et al. 2014).
In tomato plants, Palaniyandi et al. (2014) isolated a Streptomyces strain, called PGPA39, from an agricultural soil, which possess ACC deaminase, biosynthesize IAA and solubilize phosphate. This strain was also halotolerant. Spores of this strain were mixed in sterilized soil and sown with tomato plants, alleviating stress in those plants and showing higher plant biomass and root development than that of noninoculated salt-stressed tomato control plants.
As shown, there are several species and strains belonging to this genus that have plant growth potential, but there are few reports regarding studies showing improvements in crop yields under real field conditions (Viaene et al. 2016; Araujo et al. 2017) (Table 15.3).
Alekhya and Gopalakrishnan (2017) performed a screening of actinobacteria isolated from chickpea rhizosphere to find strains with antagonistic potential. Seven strains belonging to different species of the genus Streptomyces and displaying several PGP traits (broad spectrum antifungal activity, hydrolytic enzymes, IAA and HCN biosynthesis and siderophore production) were selected and tested under greenhouse conditions and also, in a field assay. Under greenhouse conditions, inoculated chickpea plants exhibit an increase in shoot weight (up to 84%), root weight (up to 57%), pod number (up to 102%) and pod weight (up to 84%). At harvest time, field assays also showed better performance of chickpea plants inoculated with the selected Streptomyces strains: seed number (up to 22%), stover yield (up to 86%), grain yield (up to 17%) and total dry matter (up to 51%).
Studies on grafted Vitis vinifera plants showed also the beneficial effects of Streptomyces strains under field real conditions (Alvarez-Pérez et al. 2017). In this work, several actinobacterial strains were isolated from young grapevine plants rhizosphere and endosphere. The isolates displayed in vitro antifungal activity, which was confirmed in field assays conducted in three experimental open-root field nurseries of grafted plants. The presence of phytopathogenic fungi affecting grafted Vitis plants was dramatically reduced (Alvarez-Pérez et al. 2017).
In cereals, there are also some examples of studies confirming the PGP potential of Streptomyces strains under field conditions. Yandigeri et al. (2012) isolated several Streptomyces strains from roots of 5 different native plants from India. Those isolates produce IAA, ammonia and siderophores. Three of these strains were tested in wheat plants in a field assay under drought conditions. Their findings revealed that the strains were drought-tolerant and improved seedling vigor after inoculation. At harvest time, wheat plants had higher biomass and there was a significative increase in grain yields.
With the aim of identifying good biocontrol agents, Palazzini and colleagues isolated several strains from wheat anthers and later identified one of them as a good biocontrol strain, Streptomyces sp RC87B (Palazzini et al. 2007). This strain presented antifungal activities, particularly against Fusarium graminearum sensu stricto under in vitro and in a greenhouse assay using a wheat cultivar that is susceptible to Fusarium infections. Ten years later, a study using the same strains confirmed that this potential can also be translated to field conditions. Wheat susceptible to Fusarium infection experienced a reduction of disease incidence (Palazzini et al. 2017).
Not only Streptomyces but also other actinobacterial genera, i.e., Micromonospora, Microbispora, Microbacterium, Actinoplanes, or Arthrobacter, were also tested alone or in combination with other bacterial members such as rhizobia or other actinobacteria, mostly under lab-controlled conditions or greenhouse assays, even that there are some of these studies that involved field trials.
Co-inoculation of leguminous plants with actinobacteria and rhizobial strains produced beneficial effects in those plants, increasing the nodule number, symbiotic efficiency and the plant biomass in most of the cases. Micromonospora strains, able to produce hydrolytic enzymes and IAA, alone and in combination with Ensifer (Sinorhizobium) strains produced significative increases in shoot and root dry weights and shoot C, N, P and K elements in Medicago sativa plants under in vitro and greenhouse conditions (Martínez-Hidalgo et al. 2014).
A study involving a set of field trials with soybean plants showed that the co-inoculation of Bradyrhizobium japonicum USDA110 with a strain of Streptomyces leads to an enhancement of nitrogen fixation and the production of a higher plant biomass and grain yield (Soe et al. 2012).
Misk and Franco (2011) co-inoculated two strains of Mesorhizobium ciceri and different biocontrol-tested Streptomyces spp. on chickpea plants under greenhouse conditions. Some of those Streptomyces strains suppressed the incidence of Phytophthora root rot disease and, in combination with both mesorhizobial strains, also enhanced vegetative growth. Interestingly, these authors also identified a non-streptomycete strain belonging to the genus Microbispora, which showed biocontrol and PGP traits; sadly, this strain was not tested in the greenhouse assays.
Interestingly, there is a study reporting the beneficial effects of a triple inoculation of three actinobacterial strains, closely related to the species Actinoplanes campanulatus, Micromonospora chalcea and Streptomyces spiralis, on cucumber plants affected with damping-off disease produced by the phytopathogenic oomycete Pythium. The three isolates produced the highest level of growth promotion when together (El-Tarabily et al. 2009). Moreover, all three actinomycete strains, alone and in combination, significantly increased root and shoot production in the presence or absence of Pythium aphanidermatum in comparison with the untreated control.
Arthrobacter is another genus that is cited frequently as potential plant growth promoter and as bioremediation agent in agriculture. Khan et al. (2018) identified a rhizospheric strain of Arthrobacter woluwensis, strain AK1, which showed ABA and IAA production under saline conditions. This halotolerant strain mitigated salt stress and promoted rice growth under in vitro conditions and also promoted soybean growth under greenhouse conditions.
In a search for phosphate solubilizers, Valetti et al. (2018) isolated an Arthobacter strain that significatively increased the yield of rapeseed crops when compared with the yield produced by the negative control plots (no fertilized and non-inoculated). Interestingly, the harvest index derived from the Arthrobacter sp. LRCP-11 is superior to the one derived from the negative control and fertilized uninoculated treatment.
Furthermore, there is a recent study discussing the potential role of the genus Arthrobacter in burned forests (Fernández-González et al. 2017). These authors performed a metagenomic analysis of the holm oak rhizosphere of undisturbed and burned oak forests. Actinobacteria was the most abundant phyla in both cases but is more abundant in the burned one. The genus Arthrobacter was one of the genera in burned rhizospheres, showing a significant increase in abundance with respect to other genera of Actinobacteria. Isolates from this genus displayed hydrolytic enzyme activities and IAA production and some of them lead to the significant increase of alfalfa and pepper vegetative growth under greenhouse conditions.
15.5 Conclusions and Futures Perspectives
The use of Actinobacteria as plant probiotics is still in a very early stage compared with the use and application of other PGP bacteria. However, the high number of new species described having a close relationship with plants, including endophytic and rhizosphere actinobacteria, as well as the importance of these microorganisms revealed by plant microbiomes, make them a very interesting alternative to solve agricultural problems. These microorganisms have an excellent potential for plant protection due to its ability to produce inhibitory compounds that will not allow the development of plant pathogens, as well as inducing the natural defense systems of the plants, even from an early stage of development. The sequencing and further analysis of complete or nearly complete genomes have also evidenced the potential of the Actinobacteria. Future studies will help in the discovery of new molecules implicated in plant–endophyte symbiotic interactions. The actinobacteria are also soil microorganisms, a feature that will help in their permanence for a long period of time in this unpleasant environment. Until now, the application of these microorganisms in real agricultural conditions has been limited; however, the limitations of the use of pesticides and chemical fertilizers in several worldwide countries and the global acceptance of the use of Plant Probiotic Bacteria as a “Green” alternative will encourage the use of these Plant Probiotic Actinobacteria in real crop production.
References
Adekambi BRW, Hanrahan F, Delcher AL et al (2011) Core gene set as the basis of multilocus sequence analysis of the subclass Actinobacteridae. PLoS One 6(3):e14792. https://doi.org/10.1371/journal.pone.0014792
Alekhya G, Gopalakrishnan S (2017) Biological control and plant growth-promotion traits of Streptomyces species under greenhouse and field conditions in chickpea. Agric Res 6(4):410–420
Alvarez-Pérez JM, González-García S, Cobos R et al (2017) Using endophytic and rhizospheric actinobacteria from grapevine plants to reduce fungal graft infections in nurseries that lead to young grapevine decline. App Environ Microbiol 83:24. https://doi.org/10.1128/AEM.01564-17
Anwar S, Ali B, Sajid I (2016) Screening of rhizospheric Actinomycetes for various in-vitro and in-vivo plant growth promoting (PGP) traits and for agroactive compounds. Front Microbiol 7:1334
Araujo R, Kaewkla O, Franco CM (2017) Endophytic actinobacteria: beneficial partners for sustainable agriculture. In: Maheshwari D (ed) Endophytes: biology and biotechnology, Sustainable development and biodiversity, vol 15. Springer, Cham, pp 171–192
Bal HB, Das S, Dangar TK et al (2013) ACC deaminase and IAA producing growth promoting bacteria from the rhizosphere soil of tropical rice plants. J Basic Microbiol 53:972–984
Banik A, Mukhopadhaya SK, Dangar TK (2016) Characterization of N2-fixing plant growth promoting endophytic and epiphytic bacterial community of Indian cultivated and wild rice (Oryza spp.) genotypes. Planta 243:799–812
Barka EA, Vatsa P, Sanchez L, Gaveau-Vaillant N, Jacquard C, Klenk H-P, Clément C, Ouhdouch Y, van Wezel GP (2015) Taxonomy, physiology, and natural products of Actinobacteria. Microbiol Mol Biol Rev 80(1):1–43
Beijerinck MW (1901) Über oligonitrophile Mikroben. Zentr Bakt Parasitenk Infektionskrank Hyg, Abt II 7:561–582
Bertani I, Abbruscato P, Piffanelli P et al (2016) Rice bacterial endophytes: isolation of a collection, identification of beneficial strains and microbiome analysis. Environ Microbiol Rep 8:388–398
Cardinale M, Ratering S, Suarez C et al (2015) Paradox of plant growth promotion potential of rhizobacteria and their actual promotion effect on growth of barley (Hordeum vulgare L.) under salt stress. Microbiol Res 181:22–32
Carro L (2010) Avances en la sistemática del genéro Micromonospora: estudio de cepas aisladas de la rizosfera y nódulos de Pisum sativum. PhD Thesis
Carro L, Nouiuoi I (2017) Taxonomy and systematics of plant probiotic bacteria in the genomic era. AIMS Microbiol 3(3):383–412
Carro L, Sproer C, Alonso P et al (2012) Diversity of Micromonospora strains isolated from nitrogen fixing nodules and rhizosphere of Pisum sativum analyzed by multilocus sequence analysis. Syst Appl Microbiol 35:73–80
Carro L, Pujic P, Alloisio N et al (2015) Alnus peptides modify membrane porosity and induce the release of nitrogen rich metabolites from nitrogen-fixing Frankia. ISME J 9:1723–1733
Carro L, Riesco R, Spröer C et al (2016a) Micromonospora luteifusca sp. nov. isolated from cultivated Pisum sativum. Syst Appl Microbiol 39:237–242
Carro L, Riesco R, Sproër C et al (2016b) Micromonospora ureilytica sp. nov., Micromonospora noduli sp. nov. and Micromonospora vinacea sp. nov., isolated from Pisum sativum nodules. Int J Syst Evol Microbiol 66:3509–3514
Carro L, Nouoioui I, Sangal V et al (2018a) Genome-based classification of micromonosporae with a focus on their biotechnological and ecological potential. Sci Rep 8:525
Carro L, Veyisoglu A, Riesco R et al (2018b) Micromonospora phytophila sp. nov. and Micromonospora luteiviridis sp. nov., isolated as natural inhabitants of plant nodules. Int J Syst Evol Microbiol 68:248–253
Cavalier-Smith T (2002) The neomuran origin of archaebacteria, the negibacterial root of the universal tree and bacterial megaclassification. Int J Syst Evol Microbiol 52:7–76
Conn VM, Walker AR, Franco CM (2008) Endophytic actinobacteria induce defense pathways in Arabidopsis thaliana. Mol Plant Microbe Interact 21(2):208–218. https://doi.org/10.1094/MPMI-21-2-0208
Curtis SM, Norton I, Everest GJ et al (2018) Kribbella podocarpi sp. nov., isolated from the leaves of a yellowwood tree (Podocarpus latifolius). Antonie Van Leeuwenhoek 111(6):875–882. https://doi.org/10.1007/s10482-017-0984-6
Damodharan K, Palaniyandi SA, Le B et al (2018) Streptomyces sp. strain SK68, isolated from peanut rhizosphere, promotes growth and alleviates salt stress in tomato (Solanum lycopersicum cv. Micro-Tom). J Microbiol 56:753–759. https://doi.org/10.1007/s12275-018-8120-5
Diagne N, Arumugam K, Ngom M et al (2013) Use of Frankia and actinorhizal plants for degraded lands reclamation. Biomed Res Int 2013:948258. https://doi.org/10.1155/2013/948258
El-Tarabily KA, Nassar AH, Hardy GSJ, Sivasithamparam K (2009) Plant growth promotion and biological control of Pythium aphanidermatum, a pathogen of cucumber, by endophytic actinomycetes. J App Microbiol 106(1):13–26
Fabri S, Caucas V, Abril A (1996) Infectivity and effectiveness of different strains of Frankia spp. on Atriplex cordobensis plants. Rev Argent Microbiol 28:31–38
Fernández-González AJ, Martínez-Hidalgo P, Cobo-Díaz JF et al (2017) The rhizosphere microbiome of burned holm-oak: potential role of the genus Arthrobacter in the recovery of burned soils. Sci Rep 7(1):6008
Francis IM, Stes E, Zhang Y, Rangel D, Audenaert K, Vereecke D (2016) Mining the genome of Rhodococcus fascians, a plant growth-promoting bacterium gone astray. N Biotechnol 33(5):706–717. https://doi.org/10.1016/j.nbt.2016.01.009
Ganapathy A, Natesan S (2018) Metabolic potential and biotechnological importance of plant associated endophytic Actinobacteria. In: Gupta V, Rodriguz-Couto S (eds) New and future developments in microbial biotechnology and bioengineering. Elsevier, Amsterdam, pp 207–224
Garcia LC, Martínez-Molina E, Trujillo ME (2010) Micromonospora pisi sp. nov., isolated from root nodules of Pisum sativum. Int J Syst Evol Microbiol 60:331–337
Ghodhbane-Gtari F, Nouioui I, Hezbri K et al (2018) The plant-growth-promoting actinobacteria of the genus Nocardia induces root nodule formation in Casuarina glauca. Antonie Van Leeuwenhoek 112(1):75–90. https://doi.org/10.1007/s10482-018-1147-0
Golinska P, Wypij M, Agarkar G et al (2015) Endophytic actinobacteria of medicinal plants: diversity and bioactivity. Antonie Van Leeuwenhoek 108(2):267–289
Goudjal Y, Toumatia O, Yekkour A et al (2014) Biocontrol of Rhizoctonia solani damping-off and promotion of tomato plant growth by endophytic actinomycetes isolated from native plants of Algerian Sahara. Microbiol Res 169:59–65
Hamdali H, Hafidi M, Virolle MJ, Ouhdouch Y (2008a) Rock phosphate-solubilizing Actinomycetes: screening for plant growth-promoting activities. World J Microbiol Biotechnol 24(11):2565–2575
Hamdali H, Hafidi M, Virolle MJ, Ouhdouch Y (2008b) Growth promotion and protection against damping-off of wheat by two rock phosphate solubilizing actinomycetes in a P-deficient soil under greenhouse conditions. App Soil Ecol 40(3):510–517
Hill C, Guarner F, Reid G et al (2014) Expert consensus document: the international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol 11:506–514
Janssen PH (2006) Identifying the dominant soil bacteria taxa in libraries of 16S rRNA and 16S rRNA genes. Appl Environ Microbiol 72:1719–1728
Jiang ZK, Pan Z, Li FN et al (2017) Marmoricola endophyticus sp. nov., an endophytic actinobacterium isolated from Thespesia populnea. Int J Syst Evol Microbiol 67:4379–4384
Jog R, Pandya M, Nareshkumar G, Rajkumar S (2014) Mechanism of phosphate solubilization and antifungal activity of Streptomyces spp. isolated from wheat roots and rhizosphere and their application in improving plant growth. Microbiology 160(4):778–788
Kaewkla O, Franco CMM (2018) Actinomycetospora callitridis sp. nov., an endophytic actinobacterium isolated from the surface-sterilised root of an Australian native pine tree. Antonie Van Leeuwenhoek 112(3):331–337. https://doi.org/10.1007/s10482-018-1162-1
Kaewkla O, Thamchaipenet A, Franco CM (2017) Micromonospora terminaliae sp. nov., an endo phytic actinobacterium isolated from the surface-sterilized stem of the medicinal plant Terminalia mucronata. Int J Syst Evol Microbiol 67:225–230
Kämpfer P, Glaeser SP, McInroy JA et al (2016) Nocardioides zeicaulis sp. nov., an endophyte actinobacterium of maize. Int J Syst Evol Microbiol 67:225–230
Kang SM, Asaf S, Kim SJ et al (2016) Complete genome sequence of plant growth-promoting bacterium Leifsonia xyli SE134, a possible gibberellin and auxin producer. J Biotechnol 239:34–38. https://doi.org/10.1016/j.jbiotec.2016.10.004
Khan MA, Ullah I, Waqas M et al (2018) Halo-tolerant rhizospheric Arthrobacter woluwensis AK1 mitigates salt stress and induces physio-hormonal changes and expression of GmST1 and GmLAX3 in soybean. Symbiosis 77(1):9–21. https://doi.org/10.1007/s13199-018-0562-3
Kim WI, Cho WK, Kim SN et al (2011) Genetic diversity of cultivable plant growth-promoting rhizobacteria in Korea. J Microbiol Biotechnol 21:777–790
Kirby BM, Meyers PR (2010) Micromonospora tulbaghiae sp. nov., isolated from the leaves of wild garlic, Tulbaghia violacea. Int J Syst Evol Microbiol 60:1328–1333
Kittiwongwattana C, Thanaboripat D, Laosinwattana C et al (2015) Micromonospora oryzae sp. nov., isolated from roots of upland rice. Int J Syst Evol Microbiol 65:3818–3823
König H (2012) Class III. Coriobacteriia class. nov. In: Goodfellow M, Kämpfer P, Busse HJ, Trujillo ME, Suzuki KI, Ludwig W, Whitman WB (eds) Bergey’s manual of systematic bacteriology, The Actinobacteria, part B, vol 5, 2nd edn. Springer, New York, p 1968
Kuncharoen N, Pittayakhajonwut P, Tanasupawat S (2018) Micromonospora globbae sp. nov., an endophytic actinomycete isolated from roots of Globba winitii C. H. Wright. Int J Syst Evol Microbiol 68:1073–1077
Li L, Tang YL, Wei B et al (2013) Micromonospora sonneratiae sp. nov., isolated from a root of Sonneratia apetala. Int J Syst Evol Microbiol 63:2383–2388
Li L, Ma JB, Abdalla Mohamad O et al (2015) Phytoactinopolyspora endophytica gen. nov., sp. nov., a halotolerant filamentous actinomycete isolated from the roots of Glycyrrhiza uralensis F. Int J Syst Evol Microbiol 65:2671–2677
Li L, Li YQ, Fu YS et al (2018a) Nesterenkonia endophytica sp. nov., isolated from roots of Glycyrrhiza uralensis. Int J Syst Evol Microbiol 68(8):2659–2663. https://doi.org/10.1099/ijsem.0.002905
Li X, Lai X, Gan L et al (2018b) Streptomyces geranii sp. nov., a novel endophytic actinobacterium isolated from root of Geranium carolinianum L. Int J Syst Evol Microbiol 68(8):2562–2567. https://doi.org/10.1099/ijsem.0.002876
Li X, Wang Z, Lu F et al (2018c) Actinocorallia populi sp. nov., an endophytic actinomycete isolated from a root of Populus adenopoda (Maxim.). Int J Syst Evol Microbiol. 68(7):2325–2330. https://doi.org/10.1099/ijsem.0.002840
Li FN, Tuo L, Lee SM et al (2018d) Amnibacterium endophyticum sp. nov., an endophytic actino bacterium isolated from Aegiceras corniculatum. Int J Syst Evol Microbiol 68(4):1327–1332. https://doi.org/10.1099/ijsem.0.002676
Lin L, Guo W, Xing Y, Zhang X, Li Z, Hu C, Li S, Li Y, An Q (2012) The actinobacterium Microbacterium sp. 16SH accepts pBBR1-based pPROBE vectors, forms biofilms, invades roots, and fixes N2 associated with micropropagated sugarcane plants. Appl Microbiol Biotechnol 93(3):1185–1195
Liu N, Wang H, Liu M et al (2009) Streptomyces alni sp. nov., a daidzein-producing endophyte isolated from a root of Alnus nepalensis D. Don. Int J Syst Evol Microbiol 59:254–258
Lozi R (1994) Actinomycetes as plant pathogens. Eur J Plant Path 100:179–200
Ludwig W, Euzéby J, Whitman WB (2012) Class IV. Nitriliruptoria class. nov. In: Goodfellow M, Kämpfer P, Busse HJ, Trujillo ME, Suzuki KI, Ludwig W, Whitman WB (eds) Bergey’s manual of systematic bacteriology, The Actinobacteria, part B, vol 5, 2nd edn. Springer, New York, p 1968
Martínez-Hidalgo P, Galindo-Villardón P, Trujillo ME et al (2014) Micromonospora from nitrogen fixing nodules of alfalfa (Medicago sativa L.). A new promising plant probiotic bacteria. Sci Rep 4:6389
Misk A, Franco C (2011) Biocontrol of chickpea root rot using endophytic actinobacteria. BioControl 56(5):811–822
Ngom M, Gray K, Diagne N et al (2016) Symbiotic performance of diverse Frankia strains on salt-stressed Casuarina glauca and Casuarina equisetifolia plants. Front Plant Sci 7:1331. https://doi.org/10.3389/fpls.2016.01331
Normand P, Nouioui I, Pujic P et al (2018) Frankia canadensis sp. nov., isolated from root nodules of Alnus incana subspecies rugosa. Int J Syst Evol Microbiol 68(9):3001–3011. https://doi.org/10.1099/ijsem.0.002939
Norris PR (2012) Class II. Acidimicrobiia class. nov. In: Goodfellow M, Kämpfer P, Busse HJ, Trujillo ME, Suzuki KI, Ludwig W, Whitman WB (eds) Bergey’s manual of systematic bac teriology, The Actinobacteria, part B, vol 5, 2nd edn. Springer, New York, p 1968
Nouioui I, Ghodhbane-Gtari F, Jando M et al (2018a) Frankia torreyi sp. nov., the first actinobacterium of the genus Frankia Brunchorst 1886, 174AL isolated in axenic culture. Antonie Van Leeuwenhoek. https://doi.org/10.1007/s10482-018-1131-8
Nouioui I, Ghodhbane-Gtari F, Rhode M et al (2018b) Frankia irregularis sp. nov., an actinobacterium unable to nodulate its original host, Casuarina equisetifolia, but effectively nodulates members of the actinorhizal Rhamnales. Int J Syst Evol Microbiol 68(9):2883–2914. https://doi.org/10.1099/ijsem.0.002914
Nouioui I, Carro L, García-López M, Meier-Kolthoff JP, Woyke T, Kyrpides NC, Pukall R, Klenk H-P, Goodfellow M, Göker M (2018c) Genome-based taxonomic classification of the phylum Actinobacteria. Front Microbiol 9:2007
Palaniyandi SA, Yang SH, Zhang L, Suh JW (2013) Effects of actinobacteria on plant disease suppression and growth promotion. App Microbiol Biotech 97(22):9621–9636
Palaniyandi SA, Damodharan K, Yang SH, Suh JW (2014) Streptomyces sp. strain PGPA39 alleviates salt stress and promotes growth of ‘Micro Tom’ tomato plants. J Appl Microbiol 117(3):766–773
Palazzini JM, Ramirez ML, Torres AM, Chulze SN (2007) Potential biocontrol agents for Fusarium head blight and deoxynivalenol production in wheat. Crop Prot 26(11):1702–1710
Palazzini JM, Yerkovich N, Alberione E et al (2017) An integrated dual strategy to control Fusarium graminearum sensu stricto by the biocontrol agent Streptomyces sp. RC 87B under field conditions. Plant Gene 9:13–18
Passari AK, Mishra VK, Singh G et al (2017) Insights into the functionality of endophytic actinobacteria with a focus on their biosynthetic potential and secondary metabolites production. Sci Rep 7(1):11809
Patel JK, Madaan S, Archana G (2018) Antibiotic producing endophytic Streptomyces spp. colonize above-ground plant parts and promote shoot growth in multiple healthy and pathogen-challenged cereal crops. Microbiol Res 215:36–45. https://doi.org/10.1016/j.micres.2018.06.003
Paterson J, Jahanshah G, Li Y et al (2017) The contribution of genome mining strategies to the understanding of active principles of PGPR strains. FEMS Microbiol Ecol 93(3):fiw249. https://doi.org/10.1093/femsec/fiw249
Qin S, Zhao GZ, Li J et al (2009) Jiangella alba sp. nov., an endophytic actinomycete isolated from the stem of Maytenus austroyunnanensis. Int J Syst Evol Microbiol 59:2162–2165
Rachniyom H, Matsumoto A, Inahashi Y et al (2018) Actinomadura barringtoniae sp. nov., an endophytic actinomycete isolated from the roots of Barringtonia acutangula (L.) Gaertn. Int J Syst Evol Microbiol 68(5):1584–1590. https://doi.org/10.1099/ijsem.0.002714
Raja A, Prabakarana P (2011) Actinomycetes and drug-an overview. Am J Drug Discov Dev 1:75–84. https://doi.org/10.3923/ajdd.2011.75.84
Remali J, Sarmin NM, Ng CL et al (2017) Genomic characterization of a new endophytic Streptomyces kebangsaanensis identifies biosynthetic pathway gene clusters for novel phenazine antibiotic production. Peer J 5:e3738. https://doi.org/10.7717/peerj.3738
Roman-Ponce B, Wang D, Vasquez-Murrieta MS et al (2016) Kocuria arsenatis sp. nov., an arsenic-resistant endophytic actinobacterium associated with Prosopis laegivata grown on high-arsenic-polluted mine tailing. Int J Syst Evol Microbiol 66:1027–1033
Sakdapetsiri C, Ngaemthao W, Suriyachadkun C et al (2018) Actinomycetospora endophytica sp. nov., isolated from wild orchid (Podochilus microphyllus Lindl.) in Thailand. Int J Syst Evol Microbiol 68(9):3017–3021. https://doi.org/10.1099/ijsem.0.002938
Salomon MV, Purpora R, Bottini R et al (2016) Rhizosphere associated bacteria trigger accumulation of terpenes in leaves of Vitis vinifera L. cv. Malbec that protect cells against reactive oxygen species. Plant Physiol Biochem 106:295–304
Schwachtje J, Karojet S, Kunz S et al (2012) Plant-growth promoting effect of newly isolated rhizobacteria varies between two Arabidopsis ecotypes. Plant Signal Behav 7:623–627
Sen A, Daubin V, Abrouk D et al (2014) Phylogeny of the class Actinobacteria revisited in the light of complete genomes. The orders ‘Frankiales’ and Micrococcales should be split into coherent entities: proposal of Frankiales ord. nov., Geodermatophilales ord. nov., Acidothermales ord. nov. and Nakamurellales ord. nov. Int J Syst Evol Microbiol 64:3821–3832
Sharma M, Mishra V, Rau N, Sharma RS (2016) Increased iron-stress resilience of maize through inoculation of siderophore-producing Arthrobacter globiformis from mine. J Basic Microbiol 56(7):719–735. https://doi.org/10.1002/jobm.201500450
Shen Y, Zhang Y, Liu C et al (2014) Micromonospora zeae sp. nov., a novel endophytic actinomycete isolated from corn root (Zea mays L.). J Antibiot (Tokyo) 67(11):739–743. https://doi.org/10.1038/ja.2014.54
Singh R, Dubey AK (2018) Diversity and applications of endophytic actinobacteria of plants in special and other ecological niches. Front Microbiol 9:1767
Soe KM, Bhromsiri A, Karladee D, Yamakawa T (2012) Effects of endophytic actinomycetes and Bradyrhizobium japonicum strains on growth, nodulation, nitrogen fixation and seed weight of different soybean varieties. Soil Sci Plant Nutr 58:319–325
Solans M (2007) Discaria trinervis-Frankia symbiosis promotion by saprophytic actinomycetes. J Basic Microbiol 47:243–250
Stackebrandt E, Rainey FA, Ward-Rainey NL (1997) Proposal for a new hierarchic classification system, Actinobacteria classis nov. Int J Syst Bacteriol 47:479–491
Sun Y, Chen HH, Sun HM et al (2017) Naumannella huperziae sp. nov., an endophytic actinobacterium isolated from Huperzia serrata (Thunb.). Int J Syst Evol Microbiol 67:1867–1872
Suzuki KI (2012) Class V. Rubrobacteria class. nov. In: Goodfellow M, Kämpfer P, Busse HJ, Trujillo ME, Suzuki KI, Ludwig W, Whitman WB (eds) Bergey’s manual of systematic bacteriology, The Actinobacteria, part B, vol 5, 2nd edn. Springer, New York, p 1968
Suzuki KI, Whitman WB (2012) Class VI. Thermoleophilia class. nov. In: Goodfellow M, Kämpfer P, Busse HJ, Trujillo ME, Suzuki KI, Ludwig W, Whitman WB (eds) Bergey’s manual of systematic bacteriology, The Actinobacteria, part B, vol 5, 2nd edn. Springer, New York, p 1968
Thawai C (2015) Micromonospora costi sp. nov., isolated from a leaf of Costus speciosus. Int J Syst Evol Microbiol 65:1456–1461
Trujillo ME, Alonso-Vega P, Rodriguez R et al (2010) The genus Micromonospora is widespread in legume root nodules: the example of Lupinus angustifolius. ISME J 4:1265–1281
Trujillo ME, Bacigalupe R, Pujic P et al (2014) Genome features of the endophytic actinobacterium Micromonospora lupini strain Lupac 08: on the process of adaptation to an endophytic life style? PLoS One 9(9):e108522. https://doi.org/10.1371/journal.pone.0108522
Trujillo ME, Riesco R, Benito P et al (2015) Endophytic Actinobacteria and the interaction of Micromonospora and Nitrogen fixing plants. Front Microbiol 6:1341. https://doi.org/10.3389/fmicb.2015.01341
Tuo L, Yan XR, Li FN et al (2018) Brachybacterium endophyticum sp. nov., a novel endophytic actinobacterium isolated from bark of Scutellaria baicalensis Georgi. Int J Syst Evol Microbiol 68(11):3563–3568. https://doi.org/10.1099/ijsem.0.003032
Upadhyay SK, Singh JS, Saxena AK et al (2012) Impact of PGPR inoculation on growth and antioxidant status of wheat under saline conditions. Plant Biol 14:605–611
Valetti L, Iriarte L, Fabra A (2018) Growth promotion of rapeseed (Brassica napus) associated with the inoculation of phosphate solubilizing bacteria. App Soil Ecol 132:1–10
Viaene T, Langendries S, Beirinck S et al (2016) Streptomyces as a plant’s best friend? FEMS Microbiol Ecol 92(8):fiw119. https://doi.org/10.1093/femsec/fiw119
Wang HF, Li L, Zhang YG et al (2015) Arthrobacter endophyticus sp. nov., an endophytic actino bacterium isolated from root of Salsola affinis C. A. Mey. Int J Syst Evol Microbiol 65:2154–2160
Wang R, Zhang H, Sun L et al (2017) Microbial community composition is related to soil biological and chemical properties and bacterial wilt outbreak. Sci Rep 7:343. https://doi.org/10.1038/s41598-017-00472-6
Wang Z, Tian J, Li X et al (2018a) Streptomyces dioscori sp. nov., a novel endophytic actinobacterium isolated from bulbil of Dioscorea bulbifera L. Curr Microbiol 75(10):1384–1390. https://doi.org/10.1007/s00284-018-1534-9
Wang Z, Jiang B, Li X et al (2018b) Streptomyces populi sp. nov., a novel endophytic actinobacterium isolated from stem of Populus adenopoda Maxim. Int J Syst Evol Microbiol 68(8):2568–2573. https://doi.org/10.1099/ijsem.0.002877
Wei L, Ouyang S, Wang Y et al (2014) Solirubrobacter phytolaccae sp. nov., an endophytic bac terium isolated from roots of Phytolacca acinosa Roxb. Int J Syst Evol Microbiol 64:858–862
Yan X, Li Y, Wang N, Chen Y et al (2018) Streptomyces ginkgonis sp. nov., an endophyte from Ginkgo biloba. Antonie Van Leeuwenhoek 111(6):891–896. https://doi.org/10.1007/s10482-017-0987-3
Yandigeri MS, Meena KK, Singh D et al (2012) Drought-tolerant endophytic actinobacteria promote growth of wheat (Triticum aestivum) under water stress conditions. Plant Growth Regul 68(3):411–420
Zhang Y, Liu H, Zhang X et al (2014) Micromonospora violae sp. nov., isolated from a root of Viola philippica Car. Antonie van Leeuwenhoek 106:219–225
Zhang YG, Wang HF, Alkhalifah DHM (2018) Glycomyces anabasis sp. nov., a novel endophytic actinobacterium isolated from roots of Anabasis aphylla L. Int J Syst Evol Microbiol 68(4):1285–1290. https://doi.org/10.1099/ijsem.0.002668
Zhao J, Guo L, He H et al (2014) Micromonospora taraxaci sp. nov., a novel endophytic actinomycete isolated from dandelion root (Taraxacum mongolicum Hand-Mazz.). Antonie van Leeuwenhoek 106(4):667–674
Zhao S, Liu C, Zheng W et al (2017) Micromonospora parathelypteridis sp. nov., an endophytic actinomycete with antifungal activity isolated from the root of Parathelypteris beddomei (Bak.) Ching. Int J Syst Evol Microbiol 67:268–274
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Menendez, E., Carro, L. (2019). Actinobacteria and Their Role as Plant Probiotics. In: Giri, B., Prasad, R., Wu, QS., Varma, A. (eds) Biofertilizers for Sustainable Agriculture and Environment . Soil Biology, vol 55. Springer, Cham. https://doi.org/10.1007/978-3-030-18933-4_15
Download citation
DOI: https://doi.org/10.1007/978-3-030-18933-4_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-18932-7
Online ISBN: 978-3-030-18933-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)