Abstract
Background
Controversy exists regarding the benefit of lymphadenectomy for nonfunctional pancreatic neuroendocrine tumors (NF-PNET).
Patients and Methods
MEDLINE/PubMed, EMBASE, and the Cochrane Library were searched for studies of pancreatic neuroendocrine tumors (PNET) published between 1990 and 2021. Studies of functional PNET were excluded. Reported incidence of lymph node metastasis (LNM) and survival analysis of either disease-free survival (DFS) or overall survival (OS) were required for inclusion.
Results
Overall, 52 studies analyzing 24,608 PNET met the inclusion criteria. The reported LNM rate for NF-PNET ranged from 7 to 64 % (median 24.5%). Reported LNM rates ranged from 7 to 51% (median 11%) for NF-PNET< 2 cm in 14 studies and 29–47% (median 38%) in NF-PNET > 2 cm. In total, 19 studies (66%) reported LNM to have a negative impact on DFS. Additionally, 21 studies (60%) reported LNM to have a negative impact on OS. Two studies investigating the impact of lymphadenectomy (LND) found LND had the greatest impact for large, high-grade tumors. The overall quality of available evidence was low as assessed by the Grading of Recommendations, Assessment, Development, and Evaluation System.
Conclusions
Published literature evaluating the impact of regional LNM and LND in PNET is confounded by heterogeneity in practice patterns and the retrospective nature of these cohort studies. Most studies suggest high rates of LNM in NF-PNET that negatively impact DFS and OS. Given the high rate of LNM in NF-PNET and its potential detrimental effect on DFS and OS, we recommend lymphadenectomy be completed for NF-PNET > 2 cm and strongly considered for NF-PNET < 2 cm.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Pancreatic neuroendocrine tumors (PNET) are increasingly diagnosed with advancements in imaging technology.1 The natural history of PNET is different from the more common type of pancreatic cancer, pancreatic ductal adenocarcinoma, where survival outcomes are poor. PNET biologic behavior can vary greatly, with some patients experiencing slow progression of the disease while others may have rapid progression leading to death.2 Owing to the low incidence of PNET and longer survival rates in most patients, it is challenging to conduct prospective studies to understand their prognosis and survival. Therefore, the current data guiding PNET treatment is largely retrospective and the high variability in practice patterns between centers limits meaningful comparison of the factors driving PNET survival outcomes.
Functional PNET (F-PNET) results when these tumors secrete active hormones, causing a clinical syndrome. In malignant functional PNET, lymph node metastasis (LNM) commonly occurs, and the removal of both the primary tumor and affected lymph nodes is often necessary to address the hormonal imbalance.3,4 However, in nonfunctional PNET (NF-PNET), where no clinically active hormone is produced, the impact of regional LND is more uncertain. Generally, small (< 2 cm) low-grade nonfunctional PNET are thought to have a benign course and a low risk of regional metastasis. This has led some experts to recommend observation or limited tumor removal without sampling the lymph nodes for small nonfunctional PNET in specialized medical centers.5,6 However, other studies report more aggressive biology and LNM in some small NF-PNET, suggesting that lymph node dissection may be beneficial.7,8 Even in larger (≥ 2 cm) low-grade NF-PNET, it is unclear how lymph node metastasis affects survival outcomes. Some studies indicate that patients without lymph node involvement have better survival rates,9,10 while others suggest that distant metastasis and higher tumor grade are more significant prognostic factors, with LMN having limited relevance.2,11 The National Comprehensive Cancer Network (NCCN) guidelines recommend removing both the tumor and the affected lymph nodes for all PNET larger than 2 cm, and considering lymph node removal for tumors between 1 and 2 cm [12].
Therefore, the role of routine LND in low-grade non-functional PNET is still uncertain. For PNET patients where the expected survival often exceeds a decade even in cases of advanced disease at diagnosis, the balance between maximizing treatment benefits and minimizing surgical risks needs to be carefully considered. This systematic review aims to determine whether LNM has prognostic significance in low-grade NF-PNET and, if so, whether LND improves survival.
Patients and Methods
Design and Literature Search Strategy
A systematic literature search of studies published between 1990 and 2021 was performed using PubMed, EMBASE, and the Cochran Library. The following Medical Subject Heading (MeSH) terms were used: pancreatic neuroendocrine tumor, islet cell carcinoma, pancreas neuroendocrine neoplasm, lymph node, metastasis, and surgery. This systemic review was considered exempt on the basis of the Medical College of Wisconsin institutional review board policies and followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for systemic reviews.13
Selection of Studies
Non-English text, nonhuman, and pediatric studies were excluded from analysis. Studies of patients with widely metastatic PNET, studies with greater > 50% representation of F-PNET, and studies of other neuroendocrine primary tumors (e.g., gastric, small bowel, or duodenal) were also excluded. Literature reviews without unique content, case reports, and studies where lymph node metastasis and/or survival outcomes were not reported were excluded. The remaining studies that evaluated the impact of positive lymph nodes or LND on recurrence-free survival (RFS) or overall survival (OS) for patients with PNET were selected for analysis.
Data Extraction
Study variables, including patient demographics, representation of F-PNET and NF-PNET, rate of LNM, disease-free-survival, and overall survival were extracted. Extracted data were independently reviewed and verified by two authors (C.C. and E.W.) for accuracy.
Results
Overview of Literature Search
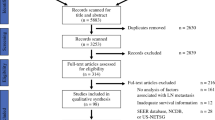
A total of 426 unique articles were identified using the following search terms: pancreatic neuroendocrine tumor, islet cell carcinoma, pancreas neuroendocrine neoplasm, lymph node, metastasis, and/or surgery (Fig. 1). In total, 62 of the original 426 manuscripts were removed, including: not written in English (n = 36), not involving human adults (n = 15), or published before 1990 (n = 11). The remaining 364 articles were then screened on the basis of the title and abstract content. An additional 270 articles were excluded owing to inapplicability of neuroendocrine tumor subtype (duodenal, small bowel, or F-PNET only), metastasis in NF-PNET, and classification as a literature review with no unique content or case report. The 94 remaining manuscripts were then included in a full review of the entire manuscript and reapplication of inclusion criteria. Thereafter, 42 articles were further excluded owing to overrepresentation of F-PNET in the study cohort, lack of lymph node status, or lack of LND. A final total of 52 manuscripts were identified as meeting the full inclusion criteria for this study. According to the Grades of Recommendation, Assessment, Development, and Evaluation (GRADE) system, each study was scored for quality of evidence.14 Findings are reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).13
Reported Rates of Positive Lymph Nodes
Overall, 50 studies reported the rate of positive lymph nodes for NF-PNET (Fig. 2). The median rate of LNM was 24.5% (range 7.1–63.5%). Several studies stratified the rate of positive LNM on the basis of size. For NF-PNET > 2 cm, the median rate of positive LNM was 37.5% (range 29–47%, n = seven studies). For tumors < 2 cm, the median rate of positive LNM was 11.3% (range 5.9–51%, n = 14 studies). Overall, several studies reported increased rates of positive lymph nodes with higher grade and larger tumors, and some reported more LNM for tumors in the head of the pancreas.
Impact of Lymph Node Status on Recurrence-Free Survival
In total, 29 studies retrospectively analyzed the impact of LNM or LND on recurrence-free survival (RFS) for NF-PNET (Table 1); 66% (n = 19) of studies found an association between LNM and worse RFS after curative resection.
In summary, most studies reported that LNM was associated with shorter RFS and resection with regional LND may mitigate the need for additional surgery compared with margin-negative resection without LND. The benefit is most significant in tumors > 2cm and with G2 and G3 tumors.
Impact of Lymph Node Status on Overall Survival
Of the 35 studies that retrospectively analyzed the impact of LNM or LND on OS (Table 2), 60% (n = 21) of studies observed an association between LNM and worse OS after curative resection of PNET.
Among the 35 studies that retrospectively analyzed the impact of LNM or LND on OS, 40% (n = 14) reported no association between LNM and OS after curative pancreatectomy for PNET. With the exception of the previously discussed SEER data analysis by Li et al.,15 it should be noted that most of these studies consist of smaller cohort studies.8,9,16,17,18,19,20,21,22,23,24,25,26
Discussion
Although LND is widely supported for malignant F-PNET, significant controversy remains regarding the benefit of LND for patients with NF-PNET, especially small NF-PNET. Current literature concerning this topic are all retrospective reviews of single institutions, multi-institution collaborations, or large national cancer databases. In our systematic review, only two studies (n = 2) were found that directly evaluated if LND provides a survival benefit for NF-PNET. One study found that LND improved RFS, while the other study failed to find any difference in RFS with the addition of LND. Wu et al. found the therapeutic index for LND to be greatest in patients with tumors ≥ 2 cm, moderately or poorly differentiated, Ki-67 ≥ 3%, and located in the head of the pancreas.27 Likely because of the very limited data that exist on LND for NF-PNET, most studies (n = 54) assess the prognostic impact of LNM and its effect on survival to then suggest if LND could improve survival outcomes. Thus, they evaluated the incidence and impact of LNM on RFS and OS.
Most of the currently available literature supports the idea that LNM rates can be significant, even for small NF-PNET. Most studies found LNM to be associated with worse survival outcomes. The reported rates of LNM varied significantly between cohorts; generally, rates of LNM were lower for tumors < 2 cm.28,29,30 While the median rate of LNM for the entire cohort was 25%, reported rates of LNM were as high as 65%. While only seven studies stratified LNM rates by tumor size, the median rate of LNM in these studies for tumors > 2 cm was three times the median rate of LNM for tumors of all sizes. Reported rates of LNM for patients with tumors < 2 cm ranged from 5.1% to 51% [28,31]. Several studies also found that higher Ki-67, larger tumor size, and higher grade are associated with a greater risk of LNM.18,32,33 Overall, 66% of studies (n = 19), found LNM to be associated with worse RFS, and 60% of studies (n = 21) found an association with worse OS. In addition to LNM, tumors that were >2 cm, symptomatic from disease burden, and had Ki-67 > 3% were found to correlate with worse RFS.34 In conclusion, current available literature supports that LNM is prognostic of worse survival; pancreatic resection with LND may improve outcomes compared with margin-negative resection without LND, especially for NF-PNET > 2 cm.
NCCN guidelines recommend completion of LND for all NF-PNET > 2 cm and consideration of LND for tumors between 1 and 2 cm;35 North American Neuroendocrine Tumor Society (NANETS) guidelines agree that LND should be considered for all NF-PNET over 2 cm.36,37
Currently, standard resection for patients with tumors > 2 cm requires a pancreatectomy in which a LND is integrated as part of the underlying operation. Given that current consensus statements recommend a formal pancreatectomy for all patients with > 2 cm lesions, the question regarding the need to complete LND for all NF-PNET has the greatest potential impact on patients with small tumors where enucleation without LND remains an option. Owing to the risk of morbidity associated with pancreatic resections, enucleation has become a viable option that is offered in attempt to reduce the risks of exocrine and endocrine insufficiency.8,38 Enucleation is often cited to have a shorter operative time, less blood loss, lower morbidity, and shorter hospital stays compared with a standard pancreatectomy.39 However, some studies have cited similar complication rates and increased rates of clinically significant pancreatic fistulas.5,39,40 Given the relatively high rates of LNM reported for tumors < 2 cm and the potential marginal improvement in operative morbidity with enucleation, the risks of leaving LNM behind must be strongly considered, even for small NF-PNET.
Failure to complete an LND while operating on a NF-PNET risks leaving residual disease behind, which may prove to be difficult to monitor for progression and challenging to address later. Unlike other disease sites, such as melanoma, the location of these lymph nodes makes them impossible to monitor based on a physical exam alone and challenging to monitor on imaging. Thus, the option of close surveillance for missed LNM and progressive disease is limited. In addition, any significant progression of residual LNM not removed at initial resection has the potential to challenge removal of symptomatic local compression in a previously operated field. Although formal pancreatectomy has its own risks, the risks of leaving LNM behind are not insignificant. These risks must be weighed against the benefits of more limited upfront surgery.
LND has multiple potential benefits for patients with NF-PNET, including potential complete resection of all cancer and accurate staging/prognosis based on the presence of LNM. Given the significant impact LNM has on both DFS and OS in most of the reports, accurate staging may help to inform decisions on the frequency of surveillance and may help to guide decisions regarding additional future therapies. Although LND may provide important prognostic information for some patients, it is also important to note that major pancreatectomy with LND may overtreat small, low-grade PNET without benefitting the patient.41 Unfortunately, at this time, we do not have a great way to delineate who will have positive nodes except LND. Unlike in melanoma and breast cancer, the variability of lymph drainage has limited the efficacy of a sentinel lymph node mapping approach for pancreas tumors.42 Thus, given the relatively high rates of LNM at this time and the prognosis, even in small NF-PNET, LND should be considered for all patients when comorbidities allow for formal pancreatectomy. There are several limitations to this study; primarily, as the entirety of the literature included in this systematic review is retrospective, each study likely has associated biases. Selection bias was likely common among the studies as patients were not randomized to LND. In addition, although each paper focused on NF-PNET, heterogeneity within the cohorts existed. Cohorts with an over representation of F-PNET were excluded. These exclusions were in the case of studies only including specific subtypes of NF-PNET, including tumors < 2 cm or cystic tumors < 2 cm. In addition, as some of the large studies were based on national cancer databases, they included overlapping cohorts. Lastly, very few (only two) of the studies specifically looked at the benefit of LND. Thus, our conclusions that LND may provide benefits to patients with LNM are based on assumptions that surgical removal of LNM may mitigate the worse RFS and OS associated with LNM. Unfortunately, until improved techniques are developed to predict which patients will have LNM, the only way to identify these patients will be through LND.
The definition of adequate lymphadenectomy in patients with NF-PNET is still being debated. In a National Cancer Database (NCDB) study of 999 patients who underwent surgical resection for PNET, 72.8% of patients had regional lymphadenectomy performed with a median of eight LNs examined.43 Zhang et al. also sought to determine the appropriate number of lymph nodes necessary to accurately stage PNET using Surveillance, Epidemiology, and End Results (SEER) data.44 They found that LMN was associated with worse RFS and that discriminatory power was highest when more than eight lymph nodes were examined. As such, we believe the best threshold for the number of LNs to be examined for PNET appears to be ≥ eight.
In summary, we believe that oncological resection with regional LND should be offered to all patients with NF-PNET that are > 2 cm. Significant consideration of the risks and benefits of LND for small NF-PNET should be made as rates of LNM for small NF-PNET are not insignificant and are associated with worse survival outcomes. Additional studies are warranted to validate that removal of LNM through LND mitigates the survival outcomes associated with LNM for NF-PNET.
Conclusions
On the basis of the available evidence, we strongly recommended that patients diagnosed with low-grade NF-PNET tumors larger than 2 cm should undergo surgical resection with routine regional lymphadenectomy. For low-grade NF-PNET smaller than 2 cm, careful consideration should be given to preoperative imaging, patient symptoms, and Ki-67 levels when deciding between suitability for observation versus surgical resection. However, it is important to acknowledge that the quality of evidence supporting these recommendations is currently low. Further research and prospective studies are needed to provide higher-quality evidence and a more comprehensive understanding of the role of lymphadenectomy in the management of NF-PNET.
References
Halfdanarson TR, Rabe KG, Rubin J, Petersen GM. Pancreatic neuroendocrine tumors (PNETs): incidence, prognosis and recent trend toward improved survival. Ann Surg Oncol. 2008;19(10):1727–33.
Bilimoria KY, et al. Prognostic score predicting survival after resection of pancreatic neuroendocrine tumors: analysis of 3851 patients. Ann Surg. 2008;247(3):490–500.
Grant CS. Surgical management of malignant islet cell tumors. World J Surg. 1993;17(4):498–503.
Tsutsumi K, et al. Analysis of lymph node metastasis in pancreatic neuroendocrine tumors (PNETs) based on the tumor size and hormonal production. J Gastroenterol. 2012;47(6):678–85.
Chua TC, Yang TX, Gill AJ, Samra JS. Systematic review and meta-analysis of enucleation versus standardized resection for small pancreatic lesions. Ann Surg Oncol. 2016;23(2):592–9.
Partelli S, et al. Management of small asymptomatic nonfunctioning pancreatic neuroendocrine tumors: limitations to apply guidelines into real life. Surgery. 2019;166(2):157–63.
Haynes AB, et al. Implications of incidentally discovered, nonfunctioning pancreatic endocrine tumors: short-term and long-term patient outcomes. Arch Surg. 2011;146(5):534–8.
Cherenfant J, et al. Predicting aggressive behavior in nonfunctioning pancreatic neuroendocrine tumors. Surgery. 2013;154(4):785–91.
Hashim YM, et al. Regional lymphadenectomy is indicated in the surgical treatment of pancreatic neuroendocrine tumors (PNETs). Ann Surg. 2014;259(2):197–203.
Tomassetti P, et al. Endocrine pancreatic tumors: factors correlated with survival. Ann Oncol. 2005;16(11):1806–10.
Ellison TA, et al. A single institution’s 26-year experience with nonfunctional pancreatic neuroendocrine tumors: a validation of current staging systems and a new prognostic nomogram. Ann Surg. 2014;259(2):204–12.
NCCN Guidelines Neuroendocrine and Adrenal Tumors Version 1.2023 2023 6/1/2023]; Available from: https://www.nccn.org/professionals/physician_gls/pdf/neuroendocrine.pdf.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Br Med J. 2009;339:b2535.
Goldet G, Howick J. Understanding GRADE: an introduction. J Evid-Based Med. 2013;6(1):50–4.
Li G, et al. Clinicopathological features and prognosis factors for survival in elderly patients with pancreatic neuroendocrine tumor: a STROBE-compliant article. Med (Baltimore). 2019;98(11):e14576.
Harimoto N, et al. Significance of lymph node metastasis in resectable well-differentiated pancreatic neuroendocrine tumor. Pancreas. 2019;48(7):943–7.
Dima SO, et al. Prognostic factors in patients with surgical resection of pancreatic neuroendocrine tumours. Acta Endocrinol (Buchar). 2018;14(3):389–93.
Masui T, et al. Predictive value of the Ki67 index for lymph node metastasis of small non-functioning pancreatic neuroendocrine neoplasms. Surg Today. 2019;49(7):593–600.
Genc CG, et al. A new scoring system to predict recurrent disease in grade 1 and 2 nonfunctional pancreatic neuroendocrine tumors. Ann Surg. 2018;267(6):1148–54.
Ge W, et al. Surveillance and comparison of surgical prognosis for asymptomatic and symptomatic non-functioning pancreatic neuroendocrine tumors. Int J Surg. 2017;39:127–34.
Wong J, et al. Predictors of lymph node metastases and impact on survival in resected pancreatic neuroendocrine tumors: a single-center experience. Am J Surg. 2014;208(5):775–80.
Demir R, et al. Necrosis and angioinvasion predict adverse outcome in pancreatic neuroendocrine tumors after curative surgical resection: results of a single-center series. World J Surg. 2011;35(12):2764–72.
Chung JC, et al. Malignant nonfunctioning endocrine tumors of the pancreas: predictive factors for survival after surgical treatment. World J Surg. 2007;31(3):579–85.
Schurr PG, et al. Aggressive surgery improves long-term survival in neuroendocrine pancreatic tumors: an institutional experience. Ann Surg. 2007;245(2):273–81.
Kazanjian KK, Reber HA, Hines OJ. Resection of pancreatic neuroendocrine tumors: results of 70 cases. Arch Surg. 2006;141(8):765–9.
Madeira I, et al. Prognostic factors in patients with endocrine tumours of the duodenopancreatic area. Gut. 1998;43(3):422–7.
Wu L, et al. Therapeutic index of lymphadenectomy among patients with pancreatic neuroendocrine tumors: a multi-institutional analysis. J Surg Oncol. 2019;120(7):1080–6.
Toste PA, et al. Nonfunctional pancreatic neuroendocrine tumors < 2 cm on preoperative imaging are associated with a low incidence of nodal metastasis and an excellent overall survival. J Gastrointest Surg. 2013;17(12):2105–13.
Sarmiento JM, Farnell MB, Que FG, Nagorney DM. Pancreaticoduodenectomy for islet cell tumors of the head of the pancreas: long-term survival analysis. World J Surg. 2002;26(10):1267–71.
Sharpe SM, et al. Surgical resection provides an overall survival benefit for patients with small pancreatic neuroendocrine tumors. J Gastrointest Surg. 2015;19(1):117–23.
Ha S, et al. The clinicopathologic and operative characteristics of patients with small nonfunctioning pancreatic neuroendocrine tumors. ANZ J Surg. 2021;91(7–8):E484-e492.
Genç CG, et al. Recurrence of pancreatic neuroendocrine tumors and survival predicted by Ki67. Ann Surg Oncol. 2018;25(8):2467–74.
Lopez-Aguiar AG, et al. Redefining the Ki-67 index stratification for low-grade pancreatic neuroendocrine tumors: improving its prognostic value for recurrence of disease. Ann Surg Oncol. 2018;25(1):290–8.
Zaidi MY, et al. A novel validated recurrence risk score to guide a pragmatic surveillance strategy after resection of pancreatic neuroendocrine tumors: an international study of 1006 patients. Ann Surg. 2019;270(3):422–33.
NCCN: National Comprehensive Cancer Network. NCCN guidelines version 1.2015 neuroendocrine tumors.
Amin MB, et al. The Eighth Edition AJCC Cancer Staging Manual continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67(2):93–9.
Kulke MH, et al. NANETS treatment guidelines: well-differentiated neuroendocrine tumors of the stomach and pancreas. Pancreas. 2010;39(6):735–52.
Falconi M, et al. Parenchyma-preserving resections for small nonfunctioning pancreatic endocrine tumors. Ann Surg Oncol. 2010;17(6):1621–7.
Yang Z, et al. Comparison of clinical outcomes between enucleation and regular pancreatectomy in patients with non-functional pancreatic neuroendocrine tumors: a retrospective multicenter and propensity score-matched study. Jpn J Clin Oncol. 2020;51(4):595–603.
Pitt SC, et al. Small pancreatic and periampullary neuroendocrine tumors: resect or enucleate? J Gastro Intest Surg. 2009;13(9):1692–8.
Tanaka M, et al. Systematic review and metaanalysis of lymph node metastases of resected pancreatic neuroendocrine tumors. Ann Surg Oncol. 2021;28(3):1614–24.
Durczyński A, et al. Sentinel lymph node mapping in tumors of the pancreatic body: preliminary report. Contemp Oncol (Pozn). 2012;16(3):206–9.
Gratian L, et al. Impact of extent of surgery on survival in patients with small nonfunctional pancreatic neuroendocrine tumors in the United States. Ann Surg Oncol. 2014;21(11):3515–21.
Zhang X-F, et al. New nodal staging for primary pancreatic neuroendocrine tumors: a multi-institutional and national data analysis. Ann Surg. 2021;274(1):e28–35.
Dong DH, et al. Impact of tumor size and nodal status on recurrence of nonfunctional pancreatic neuroendocrine tumors </=2 cm after curative resection: a multi-institutional study of 392 cases. J Surg Oncol. 2019;120(7):1071–9.
Harimoto N, et al. Prognostic significance of neutrophil-lymphocyte ratio in resectable pancreatic neuroendocrine tumors with special reference to tumor-associated macrophages. Pancreatology. 2019;19(6):897–902.
Kim H, et al. Time-trend and recurrence analysis of pancreatic neuroendocrine tumors. Endocr Connect. 2019;8(7):1052–60.
Lopez-Aguiar AG, et al. The conundrum of < 2-cm pancreatic neuroendocrine tumors: a preoperative risk score to predict lymph node metastases and guide surgical management. Surgery. 2019;166(1):15–21.
Lopez-Aguiar AG, et al. Defining the role of lymphadenectomy for pancreatic neuroendocrine tumors: an eight-institution study of 695 patients from the US neuroendocrine tumor study group. Ann Surg Oncol. 2019;26(8):2517–24.
Sho S, et al. A prognostic scoring system for the prediction of metastatic recurrence following curative resection of pancreatic neuroendocrine tumors. J Gastrointest Surg. 2019;23(7):1392–400.
Sallinen VJ, et al. Prognosis of sporadic resected small (</=2 cm) nonfunctional pancreatic neuroendocrine tumors—A multi-institutional study. HPB (Oxford). 2018;20(3):251–9.
Taki K, et al. Significance of lymph node metastasis in pancreatic neuroendocrine tumor. Surg Today. 2017;47(9):1104–10.
Postlewait LM, et al. Pancreatic neuroendocrine tumors: preoperative factors that predict lymph node metastases to guide operative strategy. J Surg Oncol. 2016;114(4):440–5.
Kaltenborn A, et al. Prediction of survival and tumor recurrence in patients undergoing surgery for pancreatic neuroendocrine neoplasms. J Surg Oncol. 2016;113(2):194–202.
Jiang Y, et al. Impact and clinical predictors of lymph node metastases in nonfunctional pancreatic neuroendocrine tumors. Chin Med J (Engl). 2015;128(24):3335–44.
Yoo YJ, et al. Overestimated oncologic significance of lymph node metastasis in G1 nonfunctioning neuroendocrine tumor in the left side of the pancreas. Medicine (Baltimore). 2015;94(36):e1404.
Furukori M, et al. Clinicopathological features of small nonfunctioning pancreatic neuroendocrine tumors. World J Gastroenterol. 2014;20(47):17949–54.
Tsutsumi K, et al. Analysis of risk factors for recurrence after curative resection of well-differentiated pancreatic neuroendocrine tumors based on the new grading classification. J Hepatobiliary Pancreat Sci. 2014;21(6):418–25.
Partelli S, et al. Pattern and clinical predictors of lymph node involvement in nonfunctioning pancreatic neuroendocrine tumors (NF-PanNETs). JAMA Surg. 2013;148(10):932–9.
Lee LC, et al. Small, nonfunctioning, asymptomatic pancreatic neuroendocrine tumors (PNETs): role for nonoperative management. Surgery. 2012;152(6):965–74.
Wang SE, et al. Comparison of functional and nonfunctional neuroendocrine tumors in the pancreas and peripancreatic region. Pancreas. 2011;40(2):253–9.
Watzka FM, et al. Prognostic assessment of non-functioning neuroendocrine pancreatic neoplasms as a basis for risk-adapted resection strategies. World J Surg. 2020;44(2):594–603.
Liu P, et al. Lymph node ratio, but not the total number of examined lymph nodes or lymph node metastasis, is a predictor of overall survival for pancreatic neuroendocrine neoplasms after surgical resection. Oncotarget. 2017;8(51):89245–55.
Jutric Z, et al. Regional metastatic behavior of nonfunctional pancreatic neuroendocrine tumors: impact of lymph node positivity on survival. Pancreas. 2017;46(7):898–903.
Jin K, et al. Clinical outcomes and prognostic factors of resected pancreatic neuroendocrine neoplasms: a single-center experience in China. Oncol Lett. 2017;13(5):3163–8.
Conrad C, et al. Prognostic value of lymph node status and extent of lymphadenectomy in pancreatic neuroendocrine tumors confined to and extending beyond the pancreas. J Gastrointest Surg. 2016;20(12):1966–74.
Fitzgerald TL, et al. Indications for surgical resection in low-grade pancreatic neuroendocrine tumors. Am Surg. 2016;82(8):737–42.
Curran T, et al. Importance of lymph node involvement in pancreatic neuroendocrine tumors: impact on survival and implications for surgical resection. J Gastrointest Surg. 2015;19(1):152–60.
Song KB, et al. Prognostic factors in 151 patients with surgically resected non-functioning pancreatic neuroendocrine tumours. ANZ J Surg. 2016;86(7–8):563–7.
Han X, et al. Clinicopathological characteristics and prognosis-related factors of resectable pancreatic neuroendocrine tumors: a retrospective study of 104 cases in a single Chinese center. Pancreas. 2014;43(4):526–31.
Matthews BD, et al. Surgical experience with nonfunctioning neuroendocrine tumors of the pancreas. Am Surg. 2000;66(12):1116–22.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Clarke, C.N., Ward, E., Henry, V. et al. Impact of Regional Metastasis on Survival for Patients with Nonfunctional Pancreatic Neuroendocrine Tumors: A Systematic Review. Ann Surg Oncol 31, 4976–4985 (2024). https://doi.org/10.1245/s10434-024-15249-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-024-15249-1