Abstract
In the last decade, an increasing number of studies have reported that soil nitrous oxide (N2O) emissions can be reduced by adding biochar. However, the effect of biochar amendment on soil N2O emissions during freeze-thaw cycle (FTC) is still unknown. In this laboratory study, biochar (0%, 2% and 4%, w/w) was added into a cultivated sandy loam soil and then treated with 15 times of FTC (each FTC consisted of freeze at −5/−10 °C for 24 h and thaw at 5/10 °C for 24 h), to test whether biochar can mitigate soil N2O emissions during FTC, and estimate the relationships between N2O emissions and soil inorganic nitrogen contents/microbial biomass content/enzyme activities. The results showed that biochar amendment suppressed soil N2O emissions by 19.9–69.9% as compared to soils without biochar amendment during FTC. However, N2O emissions were only significantly correlated to soil nitrate nitrogen (NO3−-N) contents, which decreased after biochar amendment, indicating that the decreased soil nitrification by adding biochar played an important role in mitigating N2O emissions during FTC. Further studies are needed to estimate the effectiveness of biochar amendment on reducing freeze-thaw induced N2O emissions from different soils under field conditions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
The potent greenhouse gas nitrous oxide (N2O) plays an important role in the global biogeochemical nitrogen (N) cycle, affecting both ongoing global warming and stratospheric ozone depletion1,2. Its atmospheric concentration has increased dramatically from approximately 270 ppbv in the pre-industrial era to 322.5 ppbv in 20091. Among the sources of atmospheric N2O, agricultural soils have been identified as the most important one, which accounts for approximately 60% of the global anthropogenic N2O emissions1,3. The loss of N from soils will also decrease N availability to crops and affect crop productivity negatively4. Hence, new agricultural management practices are needed to reduce N2O emission as well as maintain N availability of soils.
Biochar, which is produced from the slow pyrolysis (<700 °C) of biomass with partial or complete exclusion of oxygen, has a relatively high carbon (C) content, surface area, and cation exchange capacity compared to unheated biomass5. In the last decade, an increasing number of studies have suggested that adding biochar into soils may have the ability to reduce soil N2O emissions6. However, these impacts vary across soil and biochar types and also strongly depend on soil conditions6. For instance, Bruun et al.7 and Clough et al.8 found that the combined applications of biochar and anaerobically digested slurry or bovine urine could increase soil N2O emissions; while in a subtropical pasture where precipitation is high in summer, Scheer et al.9 reported that biochar amendment had no effect on soil N2O emissions. As a result, the response of soil N2O emissions to biochar amendment may exhibit various behaviors under different soil conditions.
Soil freeze-thaw cycle (FTC), which is caused by the phase transition of soil water, is a common process during the non-growing season in mid-high latitude regions10. Enhanced soil N2O emissions during FTC have been reported under both field and laboratory conditions11,12,13. The mechanisms responsible for the burst of N2O emissions after thawing have been widely discussed. Enhanced microbial metabolism by the accumulated substrates during thaw periods is considered as the most likely reason14, since several studies indicated that FTC might induce significant increases in soil N mineralization15,16. A recent study by Case et al.17 showed that adding biochar into soils could also stimulate soil N mineralization and nitrification, while suppressed cumulative production of N2O by 91%. Therefore, the relationship between soil inorganic N contents and N2O emissions may be complicated after biochar amendment during FTC.
Aside from the availability of substrates, soil microbial biomass and enzyme are two other important factors that influence soil N2O emissions because they involve in catalytic reactions and nutrient mineralization18,19. For instance, Wick et al.20 reported that soil N2O emissions were positively correlated to soil microbial biomass N (SMBN) and β-glucosidase activities during a dry season; Bai et al.21 found that soil urease activity was an indicator of N2O emission because of the close relationship between urease activity and nitrification. Previous studies have demonstrated that soil microbial biomass and enzyme activities can be changed by adding biochar22,23,24. However, the observation periods of these studies all focused on the growing season. During FTC, the dynamics of soil microbial biomass and enzyme activities as well as their relationships with soil N2O emission after adding biochar are poorly understood.
Soils in mid-high latitude regions are projected to experience higher frequencies and larger amplitudes of FTC in the context of climate change, which in turn release more N2O into the atmosphere25. Although biochar amendment is a potential amendment to mitigate soil N2O emissions, very limited information is available on the effect of biochar amendment on soil N2O emission during FTC. The objectives of this laboratory study were to: (1) investigate the effects of biochar amendment on soil N2O emission, inorganic N contents, microbial biomass and enzyme activities during FTC; (2) estimate the relationships between N2O emissions and soil inorganic N contents/soil microbial biomass contents/enzyme activities under the joint effects of FTC and biochar amendment. More specifically, we tested the hypothesis that the N2O bursts during FTC will be suppressed by biochar amendment.
Materials and Methods
Soil collection and analysis
In May 2015, soil samples (0–20 cm) were collected from a farmland (43°27′ N, 82°54′ E) cultivated with corn in the Ili River Valley, Xinjiang Uygur Autonomous Region, northwest China. The surface soils in this area usually experience seasonal freeze-thaw process during early spring. The soil was classified as Typic Haploboroll (USDA) with a sandy loam texture (4.2%, 23.2% and 72.6% for clay, silt and sand, respectively). Collected soil samples were air-dried, homogenized and grounded to pass through a 2 mm nylon fiber sieve before experimental use.
Soil organic C (SOC) was measured using the H2SO4-K2Cr2O7 oxidation method, while soil total N (TN) was detected using an automatic azotometer (Kjeltec 8400, FOSS, Denmark) according to the Kjeldahl method. Soil ammonium N (NH4+-N) and nitrate N (NO3−-N) were determined using a continuous flow analyzer (AA3, SEAL Analytical, Germany) with 0.01 M CaCl2 extracts (1:10, w/v)26. Soil pH and electric conductivity were measured in a volume ratio (H2O) of 1:5 (w/v) using a pH meter (SevenEasy, Mettler-Toledo, Switzerland) and an electric conductivity meter (DDSJ-308A, Rex, China), respectively. Soil texture was analyzed using a laser diffraction particle analyzer (Mastersizer 2000, Malvern, UK). SMBN was measured using the chloroform fumigation–K2SO4 extraction method (1:4, w/v). The extracts were analyzed at 280 nm using an UV spectrophotometer (Cary 60, Agilent Technologies, USA)27. Activities of urease and protease were determined using the indigo colorimetric method and the ninhydrin colorimetric method with urea and casein as substrates, respectively28,29. Urease and protease activities were expressed as μg NH4-N g−1 h−1 and μg Tyr g−1 h−1, respectively. The physicochemical properties of soil are shown in Table 1.
Biochar analysis
Biochar used for the experiment was made by the Seek Bio-Technology Company, Shanghai, China. It was produced using bamboo under a pyrolysis of 500–600 °C. The biochar was grounded to pass through a 2 mm nylon fiber and mixed thoroughly before experimental use.
Surface structure and elemental analysis of biochar were examined using a scanning electron microscope (Super 55VP, Zesis, Germany) associated with an energy dispersive X-ray spectroscopy (XFlash 5010, Bruker, Germany) (Fig. 1). The pH, electric conductivity, NH4+-N and NO3−-N of biochar were determined using the previously mentioned methods. To estimate ash content, 1.0 g of biochar was heated in a muffle furnace (LC-502, Koyo, Japan) at 500 °C for 8 h. The ash content was calculated from: ash content (%) = mass of ash/mass of biochar × 10030. The elemental (C, H, N and S) contents of biochar were measured using an elemental analyzer (vario MICRO cube, Elementar, Germany). The O content of biochar was determined by calculating the difference between 100% and the sum contents of ash, C, H, N and S. The physicochemical properties of biochar are also shown in Table 1.
Experimental design
In 250 mL Erlenmeyer flasks, 60.0 g (oven-dry basis) of soils were mixed with 0% (BC0), 2% (BC2) and 4% (BC4) (w/w) of biochar, and then wetted with deionized water to reach 60% of water holding capacity. Each flask was covered with parafilms with several small holes to allow gaseous exchange and reduce the loss of soil water. All flasks were pre-incubated at 25 °C in dark condition for seven days. After that, flasks of each application rate were separated into two equal groups to experience different FTC treatments. Taking the field surface temperatures during spring freeze-thaw periods into consideration31, we set up two experiments with one in a small amplitude (−5 °C to 5 °C) of FTC (SFT) and the other in a large amplitude (−10 °C to 10 °C) of FTC (LFT). There were fifteen times of FTC in total and each included freeze at −5 or −10 °C for 24 h and thaw at 5 or 10 °C for 24 h. Six treatments in the present study were established and after the 1st, 3rd, 5th, 10th and 15th FTC, triplicate flasks of each treatment were randomly selected and destructively sampled. Soils were used for determining NO3−-N, NH4+-N and SMBN contents together with urease and protease activities. Furthermore, deionized water was added into each flask at the end of every two FTCs to compensate for the lost soil water. Table 2 shows the details of treatment layout and properties of the mixtures after pre-incubation.
Gas sampling and analysis
Triplicate flasks for each treatment were sealed with rubber stoppers to collect gas samples at the end of 1st, 3rd, 5th, 10th and 15th FTC. In the middle of the stopper, a small hole was made and a plastic tube (0.2 cm in inner diameter, 10 cm in length) connected to a three-way stopcock was inserted into the hole. The gaps between stopper and tube were sealed with glue. The three-way stopcock was closed to make a gas-tight environment after covering. During a half hour closure period, a gas sample of approximately 2.5 mL was withdrawn using a gas-tight syringe at 0, 10, 20 and 30 min, respectively. The concentrations of N2O were detected within 3 days using a gas chromatograph (7890B, Agilent Technologies, USA), which was equipped with an electron capture detector. The carrier gas for N2O analysis was high-purity N2. N2O emissions were calculated using formula (1)32:

where ρ is the density of N2O at 0 °C (1.963 g m−3), V (m3) and W (kg) are the head space volume of the flask and the soil weight, respectively, ΔC is the change in N2O concentrations during the measurement period Δt (h), and T is the absolute temperature. Cumulative emissions during the whole incubation were directly computed from the measured emissions and estimated by linear interpolation for days when no measurements were made.
Statistical analysis
Three-way ANOVA was used to examine the differences in soil N2O emissions, NO3−-N, NH4+-N, SMBN contents, urease and protease activities among FTC amplitudes, biochar addition rates and FTC frequencies. Differences in cumulative N2O emissions between FTC amplitudes were tested using independent-samples t test, while differences in cumulative N2O emissions among biochar addition rates were examined using one-way ANOVA. Data sets have gone through the normality and heterogeneity tests and were converted to log-transformation (base 10) when the variances were unequal before analyses. Pearson correlation was employed to examine the correlations among N2O emissions, NO3−-N, NH4+-N, SMBN contents as well as urease and protease activities. Differences and correlations were considered statistically significant if P < 0.05 and highly significant if P < 0.01.
Results
NO3−-N, NH4+-N and SMBN contents
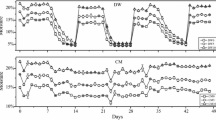
As shown in Fig. 2, soil NO3−-N contents of each treatment all increased during the incubation. As compared to the contents after pre-incubation, soil NO3−-N contents increased by 13.6–23.7% and 24.7–41.0% after 15 times of FTC under SFT and LFT, respectively. The largest increase was detected in LFT-BC0. Results of ANOVA analysis showed that soil NO3−-N content was significantly affected by FTC amplitude, biochar addition rate as well as FTC frequency (Table 3). Soil NH4+-N contents showed decreasing trends for all treatments in the first five FTCs and then slowly increased throughout the rest of FTCs (Fig. 2c,d). After the incubation, NH4+-N contents decreased by 29.1–54.6% and 25.4–49.0% under SFT and LFT compared with the contents after the pre-incubation, respectively. The effects of FTC amplitude and biochar addition rate on soil NH4+-N content were not significant when ignoring their interaction effects with FTC frequency (Table 3).
FTC amplitude and frequency had significant impacts on SMBN content (Table 3). As compared to the contents after the pre-incubation, 15 times of FTC decreased SMBN contents by 0.1–7.7% under SFT, while the range of SMBN content was only 61.2 to 81.3 mg kg−1 during the whole incubation. By contrast, LFT showed stronger effects on decreasing SMBN contents than SFT. SMBN contents of all treatments under LFT generally decreased with the increase of FTC times (Fig. 2e,f). After the 15th FTC, SMBN contents of LFT-BC0, LFT-BC2 and LFT-BC4 decreased to 54.5, 51.9 and 53.1 mg kg−1, respectively, which were the minimums of each treatment. However, biochar addition rate did not show a significant effect on SMBN content when ignoring its interaction effects with FTC amplitude and frequency (Table 3).
Enzyme activities
Similar to soil NO3−-N, soil urease activity was significantly affected by FTC amplitude, biochar addition rate and FTC frequency (Table 3). Under SFT, soil urease activities of each treatment decreased continuously from the 1st FTC to the 5th FTC, but reversed in the remaining FTCs (Fig. 3a). After the 15th FTC, soil urease activities of BC0, BC2 and BC4 increased by 7.5%, 14.3% and 7.5%, respectively, as compared to their activities after the pre-incubation. Soil urease activities under LFT varied from 21.9 to 30.1 μg NH4-N g−1 h−1 and displayed an initial decrease but subsequently increased with the increase of FTC times. Furthermore, soils amended with biochar always showed higher urease activities than BC0 in both FTC conditions. The mean activities decreased in the following order: BC4 > BC2 > BC0.
In most treatments, soil protease activities decreased as FTCs proceeded (Fig. 3c,d). In comparison with the activities after the pre-incubation, soil protease activities decreased by 17.8–25.1% and 10.5–25.5% after 15 times of FTC under SFT and LFT, respectively, with the largest decrease found in LFT-BC0. Soil protease activity was also significantly affected by biochar addition rates (Table 3), and soil protease activities of BC2 and BC4 were higher than those of BC0 during most of the incubation time. However, neither FTC amplitude nor its interaction effects with other factors significantly influenced protease activities (Table 3).
N2O emissions
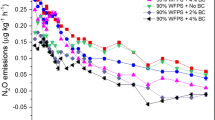
As shown in Table 3, soil N2O emission was significantly affected by FTC amplitude, biochar addition rate, FTC frequency, as well as their interaction effects. Under SFT, soil N2O emissions of all treatments were low with a range of 0.1 to 0.2 μg N2O kg−1 h−1 after the 1st FTC. Thereafter, sharp increases in N2O emissions were observed for all treatments (Fig. 4). The emissions after the 3rd FTC were 3.3, 3.1 and 6.7 times higher than those after the 1st FTC for BC0, BC2 and BC4, respectively. During the rest of FTCs, N2O emissions of each treatment showed decreasing tendencies. The emissions of BC0 were generally higher than those of BC2 and BC4. Soil N2O emissions of each treatment also showed considerable changes with the increase of FTC times under LFT. For BC2 and BC4, soil N2O emissions fluctuated as the incubation continued and the peak emissions were observed after the 3rd FTC. By contrast, soil N2O emissions of BC0 first decreased between the 1st FTC and the 3rd FTC, then increased drastically after the 5th FTC, and finally showed a decreasing trend during the last 10 FTCs. Its peak emission was 2.5 μg N2O kg−1 h−1, which was about 2.0 and 2.1 times higher than peak emissions of BC2 and BC4, respectively.
Soil cumulative N2O emissions during the whole incubation showed significant differences among different biochar addition rates (Fig. 5). In comparison with BC0, biochar amendments decreased soil N2O emissions by 19.9% (BC2) and 37.3% (BC4) under SFT, and by 41.5% (BC2) and 69.9% (BC4) under LFT. Furthermore, LFT induced about 1.7 to 3.4 times higher soil N2O emissions than SFT when soils were treated with same biochar addition rates. The highest and lowest cumulative N2O emissions were observed in LFT-BC0 and SFT-BC4, respectively.
Cumulative N2O emissions of each treatment during the whole incubation.
Bars represent the standard error of the mean (n = 3). Uppercase letters above the bars indicate significant differences among biochar addition rates while under a same FTC amplitude after one-way ANOVA with LSD test (n = 3, P < 0.05). Lowercase letters above the bars indicate significant differences between FTC amplitudes while under a same biochar addition rate after independent-samples t test (n = 3, P < 0.05)
Discussion
Impacts of biochar amendment on soil inorganic N and SMBN contents during FTC
There have been reports of increases in soil NH4+-N and NO3−-N contents after treating successive FTCs33,34. In our study, soil NO3−-N contents of all treatments also increased during FTC. However, soil NH4+-N contents of each treatment showed decreasing tendencies, especially within the first five FTCs. Our results suggested that FTC might inhibit the N mineralization, whereas favor the nitrification, which converted NH4+-N into NO3−-N under aerobic condition. Besides, previous studies have indicated that the volatilization of soil NH4+-N can be stimulated under alkaline condition35,36. Therefore, the decreased soil NH4+-N contents might be also attributed to the volatilization of NH4+-N because the soil pH was high in our study (Table 2). Although the effects of biochar on soil N dynamics have been widely investigated, information on how biochar affects soil inorganic N contents during FTC is still limited. Our results showed that biochar amendments significantly decreased soil NO3−-N content while had little effect on soil NH4+-N content as compared to BC0 during FTC, suggesting that soil nitrification may be inhibited by adding biochar when FTC occurs. In a laboratory study, Zhang et al.37 found that both soil NH4+-N and NO3−-N contents decreased with adding biochar. They suggested that biochar had the ability to adsorb soil inorganic N, and then led to decreases in soil nitrification and net N mineralization. Christenson et al.38 observed a significant negative relationship between net nitrification and soil C/N ratio. They suggested that the low gross NH4+-N production or higher NO3−-N consumption were the possible reasons for this phenomenon. As shown in Table 2, soil C/N ratio increased from 9.1 to 12.4 or 15.5 by biochar amendments. Therefore, the increased soil C/N ratio might be another possible reason for the decreases in soil nitrification.
Changes in soil microbial biomass can reflect the process of microbial growth, death, and the degradation of soil organic matter19. Similar to our results, previous studies have also reported that soil microbial biomass contents could be reduced by FTC10,39. Such decreases may be attributed to that FTC has a sterilization function, which kills soil microorganisms during freeze periods40. Moreover, our results indicated that bicohar amendments had little effects on SMBN contents during FTC. Although the effects of biochar on soil microbial biomass have been extensively investigated, the existing results are still disputable. Most of the related studies reported that soil microbial biomass could be increased by adding biochar22,41. Some studies demonstrated that biochar is a porous material, which has many pores, especially macropores (>200 nm) on its surface42,43. These macropores may hold substrates and serve as favorable habitats for soil microorganisms43. However, contrary results were also reported. For example, Dempster et al.23 pointed out that biochar could decrease soil microbial biomass C but not influence SMBN in a pot study. These differences may be explained in part by variations in biochar rate and type (e.g. biochar feedstock, pyrolysis temperature, etc.) along with soil types.
Impacts of biochar amendment on soil enzyme activities during FTC
Soil enzymes play critical roles in maintaining nutrient availability. Their activities are “sensors” of microbial status and soil physicochemical conditions18. In this study, both urease and protease activities decreased during the first five FTCs, suggesting that FTC had a short-term effect on decreasing soil enzyme activities. Previous studies suggested that the decreased enzyme activities during FTC might be attributed to the decreased microbial activities because soil enzymes mainly originate from soil microorganisms10,44. As an example, Wang et al.10 reported that soil enzyme activities were significantly correlated to soil microbial biomass C contents during FTC. Similarly, soil enzyme activities also showed significant correlations with SMBN in this study (Table 4), partially supporting the assertion. However, soil enzyme activities were quite stable or even increased after the 5th FTC, suggesting that soil enzymes or microorganisms had probably been adapted to the FTC conditions.
During most of the incubation period, soil urease and protease activities of BC2 and BC4 were generally higher than those of BC0, indicating that biochar might help retaining soil enzyme activities during FTC. This result was in agreement with previous studies which also found that soil enzyme activities were increased by adding biochar22,45. The potential mechanisms of these increases may be: (1) the macropores of biochar serve as favorable habitats for soil microorganisms and protect them from being killed by FTCs; (2) the increased substrates induced by FTC are fixed on the surface of biochar and can be used by soil microorganisms. However, contrary reports that biochar had no effects or even negative effects on increasing soil enzyme activities also existed24,46. Hence, more studies of biochar amendment on soil enzyme activities are needed to understand its effect as well as its underlying mechanisms.
Impacts of biochar amendment on soil N2O emissions during FTC
It has been demonstrated that soil N2O emissions during freeze-thaw periods are an important part of the annual N2O budget. Our results showed that biochar amendment suppressed N2O emissions by 19.9–69.9% as compared to BC0 during FTC, suggesting that biochar amendment might be a potential way to mitigate soil N2O emissions during FTC. The results of Pearson correlation analysis showed that soil N2O emissions were significantly correlated to soil NO3−-N content (Table 4). As illustrated above, soil nitrification, which converted NH4+-N to NO3−-N, might occur during the incubation. In addition, soil NO3−-N contents of BC2 and BC4 were significantly lower than those of BC0. Therefore, the suppression of N2O emissions might be related to the nitrification, which was inhibited by biochar amendments. Some studies indicated that biochar contains volatile organic compounds such as α-pinene and ethylene, which are known as nitrification inhibitors47,48. Similar to our results, Sarkhot et al.49 pointed out that biochar amendments led to 68–75% and 26% reductions in net nitrification and N2O emission, respectively. They suggested that such reductions were a result of soil inorganic N adsorption. However, Case et al.50 hypothesized that the decreased soil cumulative N2O productions by biochar were related to the biological or physical immobilization of NO3−-N, which removed large amounts of NO3−-N from the extractable pool. Therefore, the adsorption of soil inorganic N as well as the 15N tracer experiments are suggested to be designed in the future to have a better understanding of the mechanisms of N2O suppression by biochar amendment during FTC.
Conclusion
Results of the present study showed that soil N2O emissions from a cultivated sandy loam soil could be suppressed by adding biochar during FTC. The decreased nitrification indicated by the lower soil NO3−-N contents in the biochar treatments was found to play an important role in such suppressions. Biochar amendments also had a positive effect on retaining soil urease and protease activities, while it did not affect NH4+-N and SMBN contents during FTC. However, soil NH4+-N and SMBN contents, urease and protease activities did not show significant correlations to soil N2O emissions. Our study indicates that biochar amendment can be a potential method to reduce soil N2O emissions during freeze-thaw periods. Although extrapolation of the findings from this short-term laboratory study to long-term field results should be conducted with caution, the results still gave an insight into how biochar affects soil N2O emissions during FTC. Further studies are needed to estimate the effectiveness of biochar amendment on reducing freeze-thaw induced N2O emissions from different soils under field conditions.
Additional Information
How to cite this article: Liu, X. et al. Response of N2O emissions to biochar amendment in a cultivated sandy loam soil during freeze-thaw cycles. Sci. Rep. 6, 35411; doi: 10.1038/srep35411 (2016).
References
Butterbach-Bahl, K., Baggs, E. M., Dannenmann, M., Kiese, R. & Zechmeister-Boltenstern, S. Nitrous oxide emissions from soils: how well do we understand the processes and their controls? Phil. Trans. R. Soc. B 368, 20130122 (2013).
Ravishankara, A., Daniel, J. S. & Portmann, R. W. Nitrous oxide (N2O): the dominant ozone-depleting substance emitted in the 21st century. Science 326, 123–125 (2009).
Forster, P. et al. Changes in atmospheric constituents and in radiative forcing. Chapter 2. In Climate Change 2007. The Physical Science Basis (2007).
Halvorson, A. D., Snyder, C. S., Blaylock, A. D. & Del Grosso, S. J. Enhanced-efficiency nitrogen fertilizers: Potential role in nitrous oxide emission mitigation. Agronomy J. 106, 715–722 (2014).
Joseph, S. & Lehmann, J. Biochar for environmental management: science and technology. (Earthscan, London, 2009).
Clough, T. J., Condron, L. M., Kammann, C. & Müller, C. A review of biochar and soil nitrogen dynamics. Agronomy 3, 275–293 (2013).
Bruun, E., Müller-Stöver, D., Ambus, P. & Hauggaard-Nielsen, H. Application of biochar to soil and N2O emissions: potential effects of blending fast‐pyrolysis biochar with anaerobically digested slurry. Eur. J. Soil Sci. 62, 581–589 (2011).
Clough, T. J. et al. Unweathered wood biochar impact on nitrous oxide emissions from a bovine-urine-amended pasture soil. Soil Sci. Soc. Am. J. 74, 852–860 (2010).
Scheer, C., Grace, P. R., Rowlings, D. W., Kimber, S. & Van Zwieten, L. Effect of biochar amendment on the soil-atmosphere exchange of greenhouse gases from an intensive subtropical pasture in northern New South Wales, Australia. Plant Soil. 345, 47–58 (2011).
Wang, J. et al. Effects of freezing-thawing cycle on peatland active organic carbon fractions and enzyme activities in the Da Xing’anling Mountains, Northeast China. Environ. Earth Sci. 72, 1853–1860 (2014).
Priemé, A. & Christensen, S. Natural perturbations, drying-wetting and freezing-thawing cycles, and the emission of nitrous oxide, carbon dioxide and methane from farmed organic soils. Soil Biol. Biochem. 33, 2083–2091 (2001).
Wolf, B. et al. Grazing-induced reduction of natural nitrous oxide release from continental steppe. Nature 464, 881–884 (2010).
Wu, X., Brüggemann, N., Butterbach-Bahl, K., Fu, B. & Liu, G. Snow cover and soil moisture controls of freeze-thaw-related soil gas fluxes from a typical semi-arid grassland soil: a laboratory experiment. Biol. Fert. Soils. 50, 295–306 (2014).
Kim, D. G., Vargas, R., Bond-Lamberty, B. & Turetsky, M. Effects of soil rewetting and thawing on soil gas fluxes: a review of current literature and suggestions for future research. Biogeosciences 9, 2459–2483 (2012).
DeLuca, T., Keeney, D. & McCarty, G. Effect of freeze-thaw events on mineralization of soil nitrogen. Biol. Fert. Soils. 14, 116–120 (1992).
Herrmann, A. & Witter, E. Sources of C and N contributing to the flush in mineralization upon freeze-thaw cycles in soils. Soil Biology and Biochemistry 34, 1495–1505 (2002).
Case, S. D. et al. Biochar suppresses N2O emissions while maintaining N availability in a sandy loam soil. Soil Biol. Biochem. 81, 178–185 (2015).
Acosta-Martinez, V. & Tabatabai, M. Enzyme activities in a limed agricultural soil. Biol. Fert. Soils. 31, 85–91 (2000).
Balota, E. L., Colozzi-Filho, A., Andrade, D. S. & Dick, R. P. Microbial biomass in soils under different tillage and crop rotation systems. Biol. Fert. Soils. 38, 15–20 (2003).
Wick, B., Veldkamp, E., De Mello, W., Keller, M. & Crill, P. Nitrous oxide fluxes and nitrogen cycling along a pasture chronosequence in Central Amazonia, Brazil. Biogeosciences Discussions 2, 499–535 (2005).
Bai, H., Zhang, Y., Han, J. & Li, C. Nitrous oxide emission and urease activity in wheat. B. Environ. Contam Tox. 71, 1282–1288 (2003).
Demisie, W., Liu, Z. & Zhang, M. Effect of biochar on carbon fractions and enzyme activity of red soil. Catena 121, 214–221 (2014).
Dempster, D., Gleeson, D., Solaiman, Z., Jones, D. & Murphy, D. Decreased soil microbial biomass and nitrogen mineralisation with Eucalyptus biochar addition to a coarse textured soil. Plant Soil. 354, 311–324 (2012).
Wu, F., Jia, Z., Wang, S., Chang, S. X. & Startsev, A. Contrasting effects of wheat straw and its biochar on greenhouse gas emissions and enzyme activities in a Chernozemic soil. Biol. Fert. Soils. 49, 555–565 (2013).
Schaefer, K., Zhang, T., Bruhwiler, L. & Barrett, A. P. Amount and timing of permafrost carbon release in response to climate warming. Tellus B 63, 16–180 (2011).
Yao, Z. et al. Tillage and crop residue management significantly affects N-trace gas emissions during the non-rice season of a subtropical rice-wheat rotation. Soil Biol. Biochem. 41, 2131–2140 (2009).
Nunan, N., Morgan, M. & Herlihy, M. Ultraviolet absorbance (280nm) of compounds released from soil during chloroform fumigation as an estimate of the microbial biomass. Soil Biol. Biochem. 30, 1599–1603 (1998).
Wang, C. et al. Fertilization and litter effects on the functional group biomass, species diversity of plants, microbial biomass, and enzyme activity of two alpine meadow communities. Plant Soil. 331, 377–389 (2010).
Wang, Q. Y., Zhou, D. M. & Cang, L. Microbial and enzyme properties of apple orchard soil as affected by long-term application of copper fungicide. Soil Biol. Biochem. 41, 1504–1509 (2009).
Muhammad, N. et al. Changes in microbial community structure due to biochars generated from different feedstocks and their relationships with soil chemical properties. Geoderma 226, 270–278 (2014).
Guo, L. P., Li, L. H., Xu, J. R. & Bao, A. M. Responses of snow depth and seasonal frozen ground temperature to enhanced air temperature in Kunges Valley, Tianshan Mountains. Resour. Sci. 34, 636–643 (in Chinese) (2012).
Lim, S. S. & Choi, W. J. Changes in microbial biomass, CH4 and CO2 emissions, and soil carbon content by fly ash co-applied with organic inputs with contrasting substrate quality under changing water regimes. Soil Biol. Biochem. 68, 494–502 (2014).
Freppaz, M., Williams, B. L., Edwards, A. C., Scalenghe, R. & Zanini, E. Simulating soil freeze/thaw cycles typical of winter alpine conditions: implications for N and P availability. Appl. Soil Ecol. 35, 247–255 (2007).
Zhou, W. et al. Effect of freezing-thawing on nitrogen mineralization in vegetation soils of four landscape zones of Changbai Mountain. Ann. Forest Sci. 68, 943–951 (2011).
Ernst, J. W. & Massey, H. F. The effects of several factors on volatilization of ammonia formed from urea in the soil. Soil Sci. Soc. Am. J. 24, 87–90 (1960).
Soares, J. R., Cantarella, H. & de Campos Menegale, M. L. Ammonia volatilization losses from surface-applied urea with urease and nitrification inhibitors. Soil Biol. Biochem. 52, 82–89 (2012).
Zhang, H., Voroney, R. P. & Price, G. W. Effects of temperature and processing conditions on biochar chemical properties and their influence on soil C and N transformations. Soil Biol. Biochem. 83, 19–28 (2015).
Christenson, L. M., Lovett, G. M., Weathers, K. C. & Arthur, M. A. The influence of tree species, nitrogen fertilization, and soil C to N ratio on gross soil nitrogen transformations. Soil Sci. Soc. Am. J. 73, 638–646 (2009).
Grogan, P., Michelsen, A., Ambus, P. & Jonasson, S. Freeze-thaw regime effects on carbon and nitrogen dynamics in sub-arctic heath tundra mesocosms. Soil Biol. Biochem. 36, 641–654 (2004).
Larsen, K. S., Jonasson, S. & Michelsen, A. Repeated freeze-thaw cycles and their effects on biological processes in two arctic ecosystem types. Appl. Soil Ecol. 21, 187–195 (2002).
Zhang, Q. et al. Effects of biochar on soil microbial biomass after four years of consecutive application in the north China plain. PloS one 9, e102062 (2014).
Downie, A., Crosky, A. & Munroe, P. Physical properties of biochar. Biochar for environmental management: Science and technology, 13–32 (2009).
Gul, S., Whalen, J. K., Thomas B. W., Sachdeva V. & Deng H. Physico-chemical properties and microbial responses in biochar-amended soils: Mechanisms and future directions, Agr. Ecosys. Environ. 206, 46–59 (2015).
Vallejo, V. E., Roldan, F. & Dick, R. P. Soil enzymatic activities and microbial biomass in an integrated agroforestry chronosequence compared to monoculture and a native forest of Colombia. Biol. Fert. Soils. 46, 577–587 (2010).
Paz-Ferreiro, J., Fu, S., Méndez, A. & Gascó, G. Interactive effects of biochar and the earthworm Pontoscolex corethrurus on plant productivity and soil enzyme activities. J. Soil. Sediment. 14, 483–494 (2014).
Bailey, V. L., Fansler, S. J., Smith, J. L. & Bolton, H. Reconciling apparent variability in effects of biochar amendment on soil enzyme activities by assay optimization. Soil Biol. Biochem. 43, 296–301 (2011).
Spokas, K. A. et al. Qualitative analysis of volatile organic compounds on biochar. Chemosphere 85, 869–882 (2011).
Taghizadeh-Toosi A. et al. Biochar incorporation into pasture soil suppresses in situ nitrous oxide emissions from ruminant urine patches. J. Environ. Qual. 40, 468–476 (2011).
Sarkhot, D. V., Berhe, A. A. & Ghezzehei, T. A. Impact of biochar enriched with dairy manure effluent on carbon and nitrogen dynamics. J. Environ.Qual. 41, 1107–1114 (2012).
Case, S. D., McNamara, N. P., Reay, D. S. & Whitaker, J. The effect of biochar addition on N2O and CO2 emissions from a sandy loam soil-the role of soil aeration. Soil Biol. Biochem. 51, 125–134 (2012).
Acknowledgements
This work was funded by the National Natural Science Foundation of China (41375149) and the open project of Xinjiang Key Laboratory of Water Cycle and Utilization in Arid Zone (XJYS0907-2013-01).
Author information
Authors and Affiliations
Contributions
X.L., L.L. and Q.W. designed the experiments, X.L. performed the experiments, X.L. and L.L. analyzed the data, X.L. wrote the first draft of the manuscript with the help of L.L. and Q.W., L.L., Z.Q., Q.W. and J.H. contributed substantially to revisions.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Liu, X., Wang, Q., Qi, Z. et al. Response of N2O emissions to biochar amendment in a cultivated sandy loam soil during freeze-thaw cycles. Sci Rep 6, 35411 (2016). https://doi.org/10.1038/srep35411
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep35411
- Springer Nature Limited
This article is cited by
-
A pore-scale investigation of soil aggregate structure responding to freeze-thaw cycles using X-ray computed microtomography
Journal of Soils and Sediments (2023)
-
How Do Freeze–Thaw Cycles Affect the Soil Pore Structure in Alpine Meadows Considering Soil Aggregate and Soil Column Scales?
Journal of Soil Science and Plant Nutrition (2022)
-
Nitrogen loading effects on nitrification and denitrification with functional gene quantity/transcription analysis in biochar packed reactors at 5 °C
Scientific Reports (2018)
-
Biochar carrier application for nitrogen removal of domestic WWTPs in winter: challenges and opportunities
Applied Microbiology and Biotechnology (2018)