Abstract
The freshwater rivers from Shiwalik Himalayas have abundant prawn resources of high economic value and play a major role in the livelihood of local fishermen. The present study aimed to determine the variation in prawn abundance explained by changes in water quality among three different streams (Gho Manhasa stream, Chadwal stream and Nagri stream). The highest abundance of prawns was found in the Gho Manhasa during the premonsoon season and lowest in the Chadwal stream during the monsoon season. Chadwal stream witnessed maximum anthropogenic activities resulting in the decline of the water quality affecting prawn fauna. Cluster analysis based on similarity in terms of prawn abundance revealed that the Chadwal stream is different from the other two streams whereas non-metric multidimensional scaling plot based on species abundance corresponding to different seasons and physiochemical parameters showed the water quality of the monsoon season of the Chadwal stream to be extremely different. Principal component analysis showed clear separation across various sites and seasons based on physicochemical parameters. Karl Pearson correlation coefficient and canonical correspondence analysis indicated that the turbidity, total dissolved solids, nitrate, chloride, calcium, magnesium and dissolved oxygen are significant parameters influencing the abundance of prawns. The population of Macrobrachium dayanum and Macrobrachium kistnense was very less in the Chadwal stream owing to unfavorable physicochemical parameters. Therefore, conservation measures are suggested which should be immediately implemented before the streams witness a further decline in their populations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Large numbers of freshwater rivers born out of Shiwalik Himalayas are the life soul of people living in Northern Indian region and Indo-Gangetic plains. Indus river system with its major rivers and tributaries ensure freshwater resources of Shiwalik Himalayas including Jammu region and support domestic use of water, agriculture, livestock and fish wealth. These rivers are habitat for a number of biota including prawn species having high commercial and aquaculture importance. The quality of water in the natural environment is a measure of the index of its aquatic ecosystem. Assessment of physicochemical parameters governing the water body gives brief information about the quantitative composition of the species (Ji 2008). The physicochemical parameters like water temperature, pH, dissolved oxygen, dissolved carbon dioxide, electrical conductivity, suspended solids, nitrate, ammonium ions, orthophosphates, color, salinity and turbidity affect the quality of water (Arfao et al. 2021). Numerous factors are responsible for variations in the physicochemical properties of water and may exert a positive or negative influence on the survival of the species (Ngodhe et al. 2014). Within a riverine ecosystem, the abundance of a species changes with seasonal variations in physicochemical properties (Sharma et al.2016; Whitfield 1994; Harris and Cyrus 1995; Garcia et al.2003). Many researchers in the past have focused on the importance of physicochemical and ecological parameters for outlining the abundance and diversity of fish, prawns and other crustaceans (Martino and Able 2003; Varadharajan et al. 2012; Shah and Pandit 2013; Lobato et al. 2020; De Sousa et al. 2020; Akongyuure et al. 2021; Fadlaoui et al. 2021; Rathnayake et al. 2021).
The diversity and abundance of prawns are influenced by several factors like dissolved oxygen, temperature, turbidity, total dissolved solids, nitrate and other structural characteristics of the aquatic environment (Berry et al. 1996; Kadye et al. 2008). These factors are associated with one another and change in one factor may lead to change in another which may lead to serious consequences on the water quality as well as the organisms living in it. Dissolved oxygen is one of the most significant factors required for aquatic life, and low levels of dissolved oxygen lead to reduced quality of water which may increase mortality in prawns (Rehman et al. 2020). In the natural environment, the quality of water should be well within the range of tolerance for populations to survive and grow efficiently. Dissolved oxygen concentration in a water body is regulated by the amount of photosynthesis, diffusion of air and exchange of water (Boyd 1998) and if reduced causes stunted growth and bring down the food conversion efficiency ratio in prawns (Allan and Maguise 1991). Temperature is another important factor for the survival of the organism and the best suitable temperature for prawn survival is between 12 and 31 °C, beyond which results in high mortality (Bwadi et al. 2018). Also, runoff from the surroundings including chemicals, dirt, soil and other garbage due to rainfall may be detrimental to the prawn populations (Bwadi et al. 2018).
The deteriorating condition of water quality has a profound effect on the environment as a whole, disturbing the very nature of the ecosystem and degradation of natural habitat is a significant factor resulting in a decrease in the diversity and abundance of the species (Alam et al. 2013). Human activities like the discharge of sewage and runoff of agricultural pollutants are the main contributing factors to reduced water quality index (Hasan et al. 2019; Anderson et al. 2002). An imbalance of nutrients in the ecosystem may cause detrimental effects like algal bloom which further depletes oxygen levels. If the organisms from these polluted waters are consumed may cause mortality in humans (Anderson et al. 2002). Since water quality determines the biological health of a species (Bera et al. 2014; Dhawan and Kaur 2002), therefore water-based studies become essential to evaluate the current data and push forward the management strategies to effectively manage the water bodies in the future. Keeping this in mind, the present study was designed to estimate the physicochemical parameters which would enable the evaluation of water quality among three streams and its effect on prawn abundance. For this study, two hypotheses were tested: (1) Abundance of prawn species differs among collection sites and seasons. (2) Changes in the abundance of prawns can be described by variations in the quality of water.
Material and method
Study site
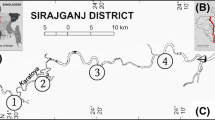
The present study was carried out for 12 months from March 2020 to February 2021 at three different sites—Gho Manhasa stream (GMS), Chadwal stream (CS) and Nagri stream (NS) (Fig. 1). Gho Manhasa stream (32.692°N, 74.768°E) is the tributary of the Chenab river present in the Jammu district, whereas Nagri stream (32.347°N,75.447°E) and Chadwal stream (32.400°N, 75.340°E) are the tributaries of Ravi river found in Kathua district of Jammu division of J&K UT, India. Chenab and Ravi rivers are the most important rivers of the Indus river system that are of Himalayan origin flowing through the Jammu region (lower Shiwalik belt of Himalayas). These two rivers and their tributaries ensure a continuous water supply for agriculture, domestic use and livestock and support the livelihood of people inhabiting the region. These three sites were selected based on accessibility, commercial importance to the local fisheries and anthropogenic pressure with the Chadwal stream witnessing maximum anthropogenic pressure than the other two streams. Months were divided from March to June, July to October and November to February into premonsoon, monsoon and postmonsoon seasons, respectively (Sharma et al. 2016). The map of the sampling location was prepared using QGIS software (version 3.18).
Physicochemical parameters
Parameters like pH, temperature, turbidity, dissolved oxygen, free carbon dioxide and alkalinity were measured on the site, and total dissolved solids (TDS), calcium, magnesium, nitrate and chloride were estimated in the laboratory on the same day of collection following standard protocols (APHA 1998; Wetzel and Likens 1991). Water temperature, pH and turbidity were measured using a degree centigrade (−10–50° C), thermometer (P-601466), digital pH meter (Testo 206 series) and digital turbidity meter (electronics India), respectively. The water sample was collected in glass biological oxygen demand (BOD) stoppered bottle for the analysis of dissolved oxygen and as required in a borosilicate glass laboratory beaker (2L capacity) for the analysis of other parameters on the sample site. For analysis of parameters to be estimated in the laboratory, water sample was collected in two polyethylene sample bottles (5L capacity) which were properly cleaned and sterilized to avoid any contamination. These bottles were labeled and immediately brought to the laboratory for physiochemical analysis. Six water samples were collected from different regions within a station and estimated for each parameter every month and their mean was calculated. The quality of the data was ensured by careful standardization of the protocol and replicate samples.
Prawn sampling and identification
Samples were collected with the aid of local fishermen early in the morning using a cast net of 5 × 5 mm. The abundance of each species was recorded in terms of the number of individuals of that species collected in 10 nettings each month, and the results were integrated according to seasons. The collected samples were sorted species-wise and counted on the spot. The specimens which were hard to identify were brought to the laboratory and studied. A total of 486 individuals from the Gho Manhasa stream, 140 from the Chadwal stream and 359 from the Nagri stream were collected during the study period. The species belonging to the genus Macrobrachium were identified up to the species level following Paul (1991), Cai and Ng (2002), and Sharma and Subba (2005). Two species belonging to the genus Caridina following Thomas (2011) are named Caridina sp.1 and Caridina sp. 2. The main identifying features of prawn are the rostrum, the number of spines on the dorsal and distal end of the telson and 2nd cheliped.
Statistical analysis
The descriptive statistics were performed using the statistical tool Palaeontological Statistics PAST, version 4.03 for the analysis of multivariate data. Basic data for calculation of mean values, standard deviation and preparation of graphs for data of abundance of each species during different seasons at different stations was done using MS Excel, 2010; two-way ANOVA and Karl Pearson correlation coefficient were estimated using SPSS (version 20.0). Karl Pearson correlation coefficient was performed to study the relationship between the environment variables and prawn abundance. Cluster analysis was performed to analyze the extent of similarity among different stations based on species abundance using the Bray Curtis similarity index in PAST software (version, 4.03). To visualize and verify the results of species abundance at different sites and seasons with respect to different physiochemical parameters, two dimensional, non-metric multidimensional scaling (nMDS) plot was created using PAST (version, 4.03). Principal component analysis was performed to determine the relationship between environmental variables and sampling sites associated with different seasons and to reduce the environmental variables for further analysis. Finally, canonical correspondence analysis was performed to study the influence of different parameters on the abundance of prawns at different sites (PAST version, 4.03). The environmental variables that best define the contribution of PCA axes were used for CCA. The species point on the CCA plot represents the preference of the species for an environmental variable in the particular aquatic habitat.
Result
Physicochemical parameters
Water parameters estimated at different stations and seasons are presented in Table 1 where N is the total number of samples analyzed from each station and the results of the two-way analysis of variance (ANOVA) are presented in Table 2. The maximum water temperature was recorded during the premonsoon season and the minimum during the postmonsoon season for all three sites. Significant variation was observed in the mean water temperature across seasons (p < 0.01), whereas sites did not show any significant differences in water temperature (p > 0.05).
Among all the study sites, the maximum turbidity was recorded in the Chadwal stream (85.62 NTU), followed by the Nagri stream (69.32 NTU) and lowest in the Gho Manhasa stream (63.98 NTU) (p < 0.01). Seasonally, the highest turbidity was recorded in August and the lowest during the postmonsoon season in February (p < 0.01). Similarly, the highest concentration of total dissolved solids was found in the Chadwal stream during the monsoon season (236.62 mg/L) and lowest in the Gho Manhasa stream during the postmonsoon season (70.00 mg/L). There were significant variations in the level of total dissolved solids during different seasons and among different sites (p < 0.01). Maximum pH was found during premonsoon at Gho Manhasa stream and minimum during postmonsoon at Chadwal stream. Significant variation was observed in the seasonal concentration of pH (p < 0.01), but no statistically significant variation was observed among sites (p > 0.05).
Across different sites and seasons, the maximum level of dissolved oxygen was found during the postmonsoon season at the Gho Manhasa stream (6.81 mg/L), Nagri stream (6.75 mg/L) and Chadwal stream (5.89 mg/L) (p < 0.01). Between seasons, the highest concentration was recorded during postmonsoon, followed by premonsoon and monsoon seasons (p < 0.01).
Maximum alkalinity was found during premonsoon season and minimum during the postmonsoon season at the study stations. Significant variation was found between seasons (p < 0.01) as well as stations (p < 0.05). The highest alkalinity was recorded at the Gho Manhasa stream during the premonsoon season (232.21 mg/L) and lowest at the Nagri stream during the postmonsoon season (125.41 mg/L).
Among seasons, the highest concentration of calcium was found during the premonsoon season followed by postmonsoon and monsoon seasons. Across the stations, the highest concentration was found at Gho Manhasa during the premonsoon season (56.92 mg/L) and lowest at the Chadwal stream during the monsoon season (35.46 mg/L). Significant differences in the mean value of calcium were observed both seasonally and stationwise (p < 0.01). The maximum mean value of magnesium was reported at Gho Manhasa during the premonsoon season (35.42 mg/L), while the minimum concentration was observed at Chadwal stream during the monsoon season (19.15 mg/L) with significant seasonal and spatial variation (p < 0.01).
Seasonally, the highest concentration of nitrate was observed during monsoon season and the minimum concentration during the premonsoon season at all the sites (p < 0.01). Stationwise, the highest concentration was recorded at the Chadwal stream (0.29 mg/L) and the lowest at the Gho Manhasa stream (0.04) for different seasons (p < 0.01). Similarly, chloride concentration was observed highest during monsoon season, followed by postmonsoon and premonsoon (p < 0.01). For the monsoon season, the highest level of chloride was reported at the Chadwal stream (47.55 mg/L) and the lowest at the Gho Manhasa stream (34.68 mg/L) (p < 0.01).
Free carbon dioxide was found maximum during monsoon and minimum during the postmonsoon season. The highest concentration was recorded at the Chadwal stream (5.92 mg/L) during the monsoon season and the lowest at the Nagri stream (3.15 mg/L) during the premonsoon season. Significant variations in the mean values of free carbon dioxide were observed between stations (p > 0.05) and seasons (p < 0.01).
Diversity and seasonal abundance of prawn species
Five species of prawns belonging to two genera, i.e., Macrobrachium and Caridina, were recorded from three study stations. The species recorded were M. dayanum, M. kistnense, M. lamarrei, Caridina sp.1 and Caridina sp.2. The Caridina spp. were new records and named viz. Caridina sp.1 and Caridina sp.2. The most abundant genus was Macrobrachium contributing 81.71, 80.76 and 86.43% to the total abundance of Gho Manhasa, Chadwal and Nagri streams, respectively (Fig. 2). Among Macrobrachium, the most abundant species was M. dayanum followed by M. kistnense which were found at all the three sites. Of all the species, the least abundance of total prawn species was constituted by M. lamarrei accounting for 3.07% in Gho Manhasa, 0% in Chadwal and 5.29% in the Nagri stream. Figure 3 represents the stationwise abundance of the species during different seasons.
Cluster analysis and non-metric multidimensional scaling (MDS)
Cluster analysis (Fig. 4) of different stations based on the species abundance revealed that Gho Manhasa and Nagri streams are more similar as they are grouped together, whereas the Chadwal stream formed a separate branch. Results of non-metric MDS analysis (Fig. 5) showed a coincident pattern as revealed by cluster analysis for the observed data. Chadwal stream with all the seasons particularly the monsoon season was found to occupy a distant positive section on the X axis, whereas all the Nagri and Gho Manhasa seasons occupied a negative section on the X axis. Chadwal stream was a distinct site mainly due to the presence of a high concentration of chloride, nitrate, turbidity, total dissolved solids and low concentration of dissolved oxygen whereas the other two streams reported opposite conditions.
Karl Pearson correlation coefficient
Table 3 presents the details of the Karl Pearson correlation coefficient between limnological attributes and the abundance of prawn species. In the present investigation, a significant negative correlation was observed between turbidity and abundance of M. dayanum (r = −0.75; p < 0.05), M. kistnense (r = −0.74; p < 0.05), M. lamarrei (r = −0.62; p < 0.05), Caridina sp.1 (r = −0.87; p < 0.05) and Caridina sp. 2 (r = −0.89; p < 0.05). Total dissolved solids showed a significant negative correlation between the abundance of M. dayanum (r = −0.66; p < 0.05), M. kistnense (r = −0.64; p < 0.05), M. lamarrei (r = −0.67; p < 0.05), Caridina sp.1 (r = −0.85; p < 0.05) and Caridina sp. 2 (r = −0.87; p < 0.05). A significant positive correlation was observed between dissolved oxygen and abundance of M. dayanum (r = 0.67; p < 0.05), M. kistnense (r = 0.64; p < 0.05) and M. lamarrei (r = 0.61; p < 0.05). Calcium and magnesium showed a significant positive correlation with M. dayanum (r = 0.82; p < 0.05) (r = 0.69; p < 0.05), M. kistnense (r = 0.87; p < 0.05) (0.72; p < 0.05), M. lamarrei (r = 0.78; p < 0.05) (r = 0.70; p < 0.05), Caridina sp. 1 (r = 0.76; p < 0.05) (r = 0.84; p < 0.05) and Caridina sp. 2 (r = 0.75; p < 0.05) (r = 0.84; p < 0.05). A significant negative correlation of nitrate was found with M. dayanum (r = −0.88; p < 0.05), M. kistnense (r = −0.86; p < 0.05), M. lamarrei (r = −0.77; p < 0.05), Caridina sp.1 (r = −0.75; p < 0.05) and Caridina sp. 2 (r = −0.77; p < 0.05) and chloride also showed significant negative correlation with M. dayanum (r = −0.85; p < 0.05), M. kistnense (r = −0.86; p < 0.05), M. lamarrei (r = −0.73; p < 0.05), Caridina sp.1 (r = −0.79; p < 0.05) and Caridina sp. 2 (r = −0.80; p < 0.05).
Principal component analysis
PCA was performed for all the eleven environmental variables studied and yielded two principal components with eigenvalues greater than 1 which were extracted and accounted for 85.64% of the total variance (Table 4). The first and the second principal components described 56.20 and 29.44% of the total variance. It is noteworthy that a loading score of 0.30 or above is considered good, 0.40 or above is considered significant and 0.50 or above is considered highly significant (Lomberte et al. 2012). In the present study, loading score greater than 0.6 was extracted for each principal component following Alvarez et al. (2017) which were statistically significant and indicated closely associated variables with PC1 and PC2. The most significant loadings on PC1 were turbidity, TDS, calcium, magnesium, nitrate and chloride, and those on PC2 were dissolved oxygen and alkalinity.
Visual investigation of the scatterplot of PC1 and PC2 showed significant variation in values between the sites due to magnesium, calcium and alkalinity which were associated with Gho Manhasa and Nagri during premonsoon and dissolved oxygen was more linked to Gho Manhasa and Nagri streams during postmonsoon. On the other hand, TDS, turbidity, nitrate and chloride were associated with Chadwal and Nagri stations during monsoon season (Fig. 6).
Canonical correspondence analysis
Canonical correspondence analysis is a direct gradient analysis technique for identifying variation patterns in species data that were best described by environmental parameters (Jongman et al. 1995, Alvarez et al. 2017, Palanivel et al. 2019). Eight statistically significant variables, viz. turbidity, TDS, calcium, magnesium, nitrate, chloride, dissolved oxygen and alkalinity identified with PCA, were selected for CCA. A CCA plot (Fig. 7) was formed to establish the relationship between environmental variables and prawn species found in three streams which showed that eigenvalue for the axis 1 (0.102) and eigenvalue for axis 2 (0.021) explain 68.02 and 38.98% of the correlation, respectively. The vector length indicates the significance of the variables with axis 1 and 2 (Liu et al. 2010). Based on vector length, total dissolved solids, turbidity, nitrate and chloride were significantly negatively correlated, whereas magnesium, calcium and dissolved oxygen were positively correlated with prawn abundance. These water quality parameters significantly affected the habitat features comprised by site-season interaction and hence influenced the overall abundance of prawn species.
Axis 1 showed a positive correlation with magnesium, calcium, dissolved oxygen and alkalinity, therefore discriminating between sites and seasons with Gho Manhasa premonsoon and postmonsoon season and Nagri premonsoon occupying a close position at one end and all the Chadwal season's interaction at the other end. Axis 2 showed a negative correlation with total dissolved solids, turbidity, chloride and nitrate, depicting water quality gradient with monsoon season at an extremity and premonsoon and postmonsoon season on the other side.
The habitat characterized by the interaction of monsoon season was associated with a high concentration of total dissolved solids, turbidity, chloride and nitrate, and no abundance of prawn species was linked to these conditions. On the contrary, interactions of the premonsoon season with the Gho Manhasa and Nagri sites along with the postmonsoon season with the Gho Manhasa site exhibited high concentrations of calcium, magnesium and dissolved oxygen and were associated with a high abundance of prawn species. It was determined that all the species were associated with a low concentration of four variables, i.e., TDS, turbidity, chloride and nitrate. M. dayanum, M. kistnense and M. lamarrei were closely associated with dissolved oxygen, calcium and magnesium, whereas Caridina spp. showed a strong relationship with alkalinity and magnesium. Also, M. dayanum occupied a near centroid position in the plot indicating that it is the most abundant species at all three sites.
Discussion
Physicochemical parameters
Monitoring of limnological parameters is significant for determining the quality of water (Bhateria and Jain 2016) which in turn is important for studying the distribution and abundance of biota in the water body (Sharma et al. 2007). In the present study, the maximum level of turbidity and total dissolved solids were recorded at Chadwal during the monsoon season. Sutlej river flowing through Himachal Pradesh shows a similar trend with a high level of turbidity during monsoon season (Jindal and Sharma 2011). All the soluble material either organic or inorganic dissolved in water constitutes total dissolved solids and is positively related to the pollution level in the water body (Butler and Ford 2018; Islam et al. 2017). High turbidity and total dissolved solids in monsoon are due to heavy soil erosion and suspension of particles like clay, silt, plankton and garbage runoff from surroundings (Choudhary et al. 2014).
pH plays a vital role in the biological mechanism of all water-dwelling organisms (Welch 1952). Most of the freshwater bodies are alkaline in nature since they contain some amount of carbonates and bicarbonates (Ishaq and Khan 2013). Lower pH makes the water more caustic, and all three studied water bodies were found to be alkaline in nature with no significant variation between stations. Dissolved oxygen is the most influential parameter affecting aquatic life as is required for respiration and adjudging the productivity level of the aquatic ecosystem and estimating the condition of the natural water ecosystems (Yang et al. 2008). All three streams showed the highest level of dissolved oxygen in the postmonsoon season, followed by the premonsoon and monsoon seasons. Our findings were consistent with the studies conducted on many rivers of the Gangetic plain of India (Rani et al. 2011). The minimum level of dissolved oxygen in monsoon season may be on account of the large influx of organic matter in the water which results in a high rate of organic decomposition in these months, consuming a large amount of dissolved oxygen (Matta et al. 2018). Also, the high level of dissolved oxygen in premonsoon and postmonsoon may be due to the increased photosynthetic rate of phytoplankton during these seasons (Ravinder et al. 2003). Calcium and magnesium represent the hardness of water (Rao 2001), and their low value was found in monsoon due to the dilution of water. Singh et al. (2010) also found calcium and magnesium to be low during this season. Mg2+ is an essential core element of chlorophyll which absorbs light during the process of photosynthesis, therefore determining the phytoplankton population, which directly and indirectly serves food for prawns. Ca2+ is important for the growth process and development of the exoskeleton of prawns.
High concentration of nitrate in the river is toxic which leads to many environmental problems like eutrophication, thus causing harm to aquatic organisms (Eddy and Williams 1987). Freshwater organisms particularly freshwater invertebrates and fish are more sensitive to nitrate than marine animals where salinity reduces the effect of nitrate toxicity (Camargo et al. 2005). Also, high nitrate concentration has been shown to influence the survival and growth of aquatic fauna (Duque et al. 2020). High levels of chloride along with other ions like Mg++, Ca++, CO3−−, HCO3− lead to the salinization of freshwater bodies and are destructive to freshwater organisms especially small invertebrates (Elphich et al. 2010). Even a small increase in the concentration of these ions can have an amplified effect on the organism interfering with osmoregulation, growth and reproduction. In the present study, the maximum concentration of chloride and nitrate was found at the Chadwal stream during the monsoon season which may be considered an indicator of pollution status (Xue 2016). High human activities were observed in this stream as it receives high amounts of municipal sewage, domestic waste and industrial effluents. Also, the influx of pesticides, fertilizers and other agricultural waste as well as open defecation from nearby fields add to the elevated levels of nitrate and chloride at this site. The effect is amplified during the rainy season as the stream is loaded with a high amount of waste. The highest concentration of free carbon dioxide was recorded at the Chadwal stream during monsoon season. Increased concentration during monsoon season may be due to the addition and decomposition of a huge load of sewage and decreased rate of photosynthesis during this season (Surana et al. 2010).
The study of limnological parameters revealed that the Chadwal stream was the most degraded site among the three streams because of maximum anthropogenic activities including cattle movement. Also, the construction of new roads in the recent past in this area has further worsened the condition by dumping waste material in the stream as well as destroying the marginal aquatic vegetation. The water quality of average to low was detected among seasons and different stations. For instance, the minimum value of dissolved oxygen was reported from the Chadwal site during monsoon season, which is considered average water quality. But due to the presence of a high concentration of nutrients like nitrate and chloride along with turbidity and total dissolved solids, the water of this site is categorized as low quality. A study conducted by Sharma et al. (2016) reported a similar result in the Baldi stream of the Garhwal Himalayas.
Variation in abundance of prawn species
In the present study, variation in the abundance of prawn species was observed among different sites and seasons. Out of five species, M. dayanum showed its dominance of 48.55, 48.7 and 74.13 percent at the Gho Manhasa, Chadwal and Nagri streams. M. dayanum has been reported to be the most dominant species of the region in previous studies as well (Sharma 2015). The high abundance of the species in a particular habitat may be related to the tolerance range of the species to various environmental parameters (Sheaves et al. 2015). The species showed maximum abundance during the premonsoon season followed by postmonsoon season and minimum in the monsoon season. A similar pattern was observed at all three sites (Fig. 3). A similar study conducted by Collocott et al. (2014) and De Sousa et al. (2020) showed summer months favor a high abundance of freshwater prawns and marine prawns, e.g., Farfantepenaeus brasiliensis, respectively.
Generally, abundance of prawn species significantly declined during monsoon season, especially in the Chadwal stream. Besides the reduced water quality, these months receive heavy rainfall and the habitat cover of prawns is destroyed as vegetation is uprooted and washed off which could be one of the probable reasons for the declined population. Also, the fast flowing nature of streams lowers the efficiency of catching the prawn species. A similar reason was reported by Galib et al. (2018) in the case of reduced fish abundance during this season. Akhi et al. (2020) reported that fast flow of water reduces the availability of food making the conditions unfavorable.
Cluster analysis and nMDS revealed that the Chadwal stream reported to have a low abundance of species, especially during the monsoon and M. lamarrei was found to be absent in this stream, indicating its narrow tolerance range to different parameters of the stream. Variations in the abundance of species may be attributed due to changes in hydrological properties and anthropogenic influences which further influence the structure of aquatic habitat with respect to distribution, abundance and survival of species, as also reported by Fausch et al. (1990); Harrison and Whitfield (2004); Hossain et al. (2012); Cendejas et al. (2013).
Influence of water quality on the abundance of prawn species
Particularly in tropical and subtropical regions where variations in environmental factors are mostly influenced by seasons, environmental variables are crucial in regulating species abundance (Blabber 2000). During the present study, monsoon season, on the one hand, and premonsoon and postmonsoon season, on the other, profoundly affected prawn abundance pattern as low abundance of prawn species was associated with low water quality. Rashed-un-Nabi et al. (2011) and Duque et al. (2020) reported a similar trend in the case of the abundance of Macrobrachium villosimanus and fish species, where the least abundance was associated with degraded water quality. M. dayanum and M. kistnense were reported across all seasons and sites, indicating their wide range of tolerance to variations in water quality parameters. Potter et al. (2015) stated that the species having a broad range of tolerance outweigh species with a narrow tolerance range in general. According to Karl Pearson correlation matrix, calcium, magnesium, dissolved oxygen, total dissolved solids, turbidity, nitrate and chloride were the factors highly correlated variables influencing abundance. For detecting the variation in communities particularly for estimating the effect of environmental parameters on the biotic group at the spatiotemporal level and understanding the variation at different gradients of environmental conditions, multivariate approaches appear to be more efficient compared to univariate analysis (Xu et al. 2011). Based on different water parameters, the results of PCA showed variation patterns in the three sites associated with different seasons and these variations further govern the habitat characterization of respective sites. This analysis successfully excluded the redundant information by identifying the variables that differentiated the sites and seasons. These variables reflected the site-specific conditions that could influence the species' abundance. Based on the CCA plot, dissolved oxygen, alkalinity, calcium and magnesium were positively correlated with abundance and turbidity, and total dissolved solids, nitrate and chloride showed a negative correlation with the abundance of prawn species. Higher turbidity and total dissolved solids are undesirable to aquatic organisms (Weber Scannell and Duffy 2007), and prawns are no exception. High concentrations of nitrate and chloride is an indicator of pollution levels (Royer et al. 2004) and are thus toxic to aquatic organisms. Low levels of dissolved oxygen in the water induce a hypoxic environmental state, making the survival of aquatic species difficult. Alkalinity makes the environment productive and nutrient-rich (Munawar 1970) and magnesium and calcium ions are important for development and required for exoskeleton formation as prawns undergo moulting and often shed their carapace.
Water quality promotes appropriate ecosystem functioning and supports the generation of ecosystem services which is determined by both biotic and abiotic measures (Foley et al. 2015; Pouso et al. 2018) and therefore can be characterized by using the concentration ranges of nitrate, chloride, and dissolved oxygen, among other characteristics (Bhateria and Jain 2016; Duque et al. 2022). Increased concentrations of productive nutrients like nitrate have been previously linked to anthropogenic practices (Smith 2003; Camargo and Alonso 2006 and Wilkerson and Dugdale 2016), such as urban settlements, excess nutrient flow from municipal and agricultural waste which has added pressure on freshwater ecosystem contributing to its deterioration. Previously, researchers have reported that aquatic systems enriched with nutrients discharged from anthropogenic wastes have influenced the presence and abundance of macroinvertebrate species by subjecting them to low levels of dissolved oxygen and physiological stress (Kenworthy et al. 2016; Hale et al. 2018; Nelson et al. 2019; Ren et al. 2019; Duque et al. 2022). A low abundance of species was evident in waters with high nitrate, chloride, total dissolved solids and turbidity. The high concentration of these parameters is a reflection of low-grade water quality and disturbed habitat condition due to wastewater from different sources that finds its way into the stream, which contain organic as well as inorganic pollutants, leading to toxicity. As a result, it could deteriorate the structure and function of the biotic community. The present study could verify the influence of fluctuating water quality on the abundance of prawns, especially with regards to nitrate, chloride, total dissolved solids and turbidity and therefore abundance patterns can be used as an ecological indicator.
Threat factors and conservation implications
The population abundance of a species is one of the indicators of the health of the ecosystem (Bender et al. 1998). Unfavorable changes in the aquatic ecosystem lead to reduced water quality, influencing species abundance (Moyleand Leidy 1992). Macrobrachium species are consumable with high economic value particularly M. dayanum and Caridina spp. are important from an ornamental point of view. Keeping in view the socioeconomic importance of the water bodies and the results of the present study, conservation measures to improve the water quality, protect the prawn population and the river as a whole ecosystem should be implemented so that future decline of the biota in general and prawn population, in particular, could be mitigated. These measures should be taken keeping in mind the life cycle and tolerable range of environmental physiology. First, the flow of sewage waste and other municipal waste should be restricted from flowing into the aquatic system to keep it from further deteriorating the water quality. Second, keeping cattle away from the edge of the water body and planting grass, shrubs or trees between the river and agricultural field to prevent agricultural runoff directly into the river that is mainly responsible for the increase in the nitrate concentration. Third, the destruction of the habitat (vegetation) along the marginal banks of the river should be prevented during the construction of roads and other developmental projects. Fourth, over-capturing of the prawn species also causes a decline in the population (Le Pape et al. 2017), so catch value should be specified and restrictions should be strictly implemented until the population revives. Lastly, creating awareness among local masses residing in the nearby area to protect the river ecosystem by not discharging domestic waste can make a major difference. The above measures should be well formulated and firmly imposed in the aquatic ecosystems harboring an enormous wealth of biodiversity, especially those that have witnessed shrinkage in the past.
Conclusion
The present study describes the influence of various limnological parameters on the abundance of different prawn species found in three important streams of the Jammu division. Based on the season, maximum abundance was found in premonsoon at all three sites due to suitable values of physicochemical parameters. Stationwise, maximum and minimum abundance was recorded from the Gho Manhasa stream and Chadwal stream, respectively. Chadwal stream showed low water quality levels as revealed through low dissolved oxygen levels and high values of nitrate, chloride, turbidity, total dissolved solids and less abundance of species. If the water quality at this site is degraded further, it will have a potential impact on the population in the nearby future. Therefore, the integrated management framework for the protection of the water body as well as the conservation of this particular population becomes significant and should be applied effectively.
Data availability
All data and materials mentioned in the manuscript are cited in the reference section with DOI links.
References
Akhi MM, Jewel MAS, Haque MA, Sarker BK, Khatun MS, Paul AK, Islam MS, Das SK (2020) Multivariate approaches to determine the relationship between fish assemblage structure and environmental variables in Karatoya River, Bangladesh. Community Ecol 21:171–181. https://doi.org/10.1007/s42974-020-00015-6
Akongyuure DN, Alhassan EH (2021) Variation of water quality parameters and correlation among them and fish catch per unit effort of the Tono reservoir in Northern Ghana. J Freshw Ecol 36(1):253–269. https://doi.org/10.1080/02705060.2021.1969295
Alam MS, Hossain MS, Monwar MM, Hoque ME (2013) Assessment of fish distribution and biodiversity status in upper Halda River, Chittagong. Int J Biodiversity Conserv 5:349–357. https://doi.org/10.5897/IJBC2013.0555
Allan GL, Maguire GB (1991) Lethal levels of low dissolved oxygen and effects of short-term oxygen stress on subsequent growth of juvenile Penaeus monodon. Aquaculture 94:27–37. https://doi.org/10.1016/0044-8486(91)90126-R
Alvarez FS, Matamoros WA, Chicas FA (2017) The contribution of environmental factors to fish assemblages in the Rio Acahuapa, a small drainage in Central America. Neotrop Ichthyol 15:e170023. https://doi.org/10.1590/1982-0224-20170023
Anderson DM, Glibert PM, Burkholder JM (2002) Harmful algal blooms and eutrophication nutrient sources, composition, and consequences. Estuaries 25:704–726. https://doi.org/10.1007/BF02804901
APHA (1998) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association, Washington, DC
Arfao AT, Onana MF, Koji E, Moungang LM, Ewoti OVN, Emadjeu JBT, Njoya STAM, Sime-Ngando T, Moise N (2021) Using principal component analysis to assess water quality from the landing stages in coastal region. Am J Water Resour 9(1):23–31. https://doi.org/10.12691/ajwr-9-1-4
Barry JP, Yoklavich MM, Cailliet GM, Ambrose DA, Antrim BS (1996) Trophic ecology of the dominant fishes in Elkhorn Slough, California, 1974–1980. Estuaries 19:115–118. https://doi.org/10.2307/1352657
Bender DJ, Contreras TA, Fahrig L (1998) Habitat loss and population decline: a meta-analysis of the patch size effect. Ecology 79:517–533. https://doi.org/10.2307/176950
Bera A, Bhattacharya M, Patra BC, Sar UK (2014) Ichthyofaunal diversity and water quality in the Kangsabati Reservoir, West Bengal, India. Adv Zool 1:1–8. https://doi.org/10.1155/2014/674313
Berbel GBB, Favaro DIT, Braga ES (2015) Impact of harbour, industry and sewage on the phosphorus geochemistry of a subtropical estuary in Brazil. Mar Pollut Bull 93:44–52. https://doi.org/10.1016/j.marpolbul.2015.02.016
Bhateria R, Jain D (2016) Water quality assessment of lake water: a review. Sustain Water Resour Manag 2:161–173. https://doi.org/10.1007/s40899-015-0014-7
Blaber SJM (2000) Tropical estuarine fishes: ecology. Exploitation and Conservation, Oxford, Blackwell Science
Boyd CE (1998) Water quality for pond aquaculture. Alabama Agricultural Experiment Station, Auburn University, Alabama, International Center for Aquaculture and Aquatic Environments
Butler BA, Ford RG (2018) Evaluating relationships between total dissolved solids (TDS) and total suspended solids (TSS) in a mining-influenced watershed. Mine Water Environ 37:18–30. https://doi.org/10.1007/s10230-017-0484-y
Bwadi BE, Mustafa FB, Ali ML, Bhassu S (2018) Spatial analysis of water quality and its suitability in farming giant freshwater prawn (Macrobrachiumrosenbergii) in Negeri Sembilan region, Peninsular Malaysia. Singap J Trop Geogr 40(1):71–91. https://doi.org/10.1111/sjtg.12250
Cai Y, Ng PKL (2002) The freshwater palaemonid prawns (Crustacea: Decapoda: Caridea) of Myanmar. Hydrobiologia 487:59–83. https://doi.org/10.1023/A:1022991224381
Camargo JA, Alonso Á (2006) Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: a global assessment. Environ Int 32(6):831–849. https://doi.org/10.1016/j.envint.2006.05.002
Camargo JA, Alonso A, Salamanca A (2005) Nitrate toxicity to aquatic animals: a review with new data for freshwater invertebrates. Chemosphere 58:1255–1267. https://doi.org/10.1016/j.chemosphere.2004.10.044
Cendejas MEV, Santillana MHD, Norris S (2013) Habitat characteristics and environmental parameters influencing fish assemblages of karstic pools in southern Mexico. Neotropical Ichthyology 11(4):859–870. https://doi.org/10.1590/S1679-62252013000400014
Choudhary J, Singh SN, Singh S (2014) Physico-chemical and biological parameters of the three rural ponds of Sasaram of Bihar. Int J Appl Sci Biotech 2:206–210. https://doi.org/10.3126/ijasbt.v2i2.10371
Collocott SJ, Vivier L, Cyrus DP (2014) Prawn community structure in the subtropical Mfolozi-Msunduzi estuarine system, KwaZulu-Natal, South Africa. Afr J Aquat Sci 39:127–140. https://doi.org/10.2989/16085914.2014.925419
De Sousa AN, De Almeida AC, Hiroki KA, Bernardo CH, Bernardes VP, Fransozo A (2020) Responses of pink shrimp Farfantepenaeusbrasiliensis (Latreille, 1817) (Penaeoidea) to physicochemical parameters in a marine protected area: changes in abundance and distribution after 20 years. J Nat Hist 54(7–8):419–433. https://doi.org/10.1080/00222933.2020.1765036
Dhawan A, Kaur S (2002) Effect of pig dung on water quality and polyculture of carp species during winter and summer. Aquacult Int 10:297–307. https://doi.org/10.1023/A:1022406800631
Duque G, Garcia DEG, Molina A, Cogua P (2020) Effect of water quality variation on fish assemblages in an anthropogenically impacted tropical estuary, Colombian Pacific. Environ Sci Pollut Res 27:25740–25753. https://doi.org/10.1007/s11356-020-08971-2
Duque G, Gamboa-García DE, Molina A, Cogua P (2022) Influence of water quality on the macroinvertebrate community in a tropical estuary (Buenaventura Bay). Integr Environ Assess Manag 18:796–812. https://doi.org/10.1002/ieam.4521
Eddy FB, Williams EM (1987) Nitrite and freshwater Fish. Chem Ecol 3:1–38. https://doi.org/10.1080/02757548708070832
Elphick JRF, Bergh KD, Bailey HC (2010) Chronic toxicity of chloride to freshwater species: effects of hardness and implications for water quality guidelines. Environ Toxicol Chem 30:239–246. https://doi.org/10.1002/etc.365
Fadlaoui S, Asri QE, Bouterfas M, Melhaoui M (2021) Effects of physicochemical variables of superficial waters on the abundance of the North African freshwater crab Potamonalgeriense (Bott, 1967). J Toxicol 1:1–13. https://doi.org/10.1155/2021/6669919
Fausch K, Lyons J, Karr J, Angermeier P (1990) Fish communities as indicators of environmental degradation. Am Fish Soc Symp 8:123–144
Foley MM, Duda JJ, Beirne MM, Paradis R, Ritchie A, Warrick JA (2015) Rapid water quality change in the Elwha River estuary complex during dam removal. Limnol Oceanogr 60:1719–1732. https://doi.org/10.1002/lno.10129
Galib SM, Mohsin ABM, Parvez MT, Lucas MC, Chaki N, Arnob SS, Hossain MI, Islam MN (2018) Municipal wastewater can result in a dramatic decline in freshwater fishes: a lesson from a developing country. Knowl Manag Aquat Ecosyst 419:37. https://doi.org/10.1051/kmae/2018025
Garcia AM, Vieira JP, Winemiller KO (2003) Effects of 1997–1998 El Nino on the dynamics of the shallow-water fish assemblage of the Patos Lagoon estuary (Brazil). Estuar Coast Shelf Sci 57:489–500. https://doi.org/10.1016/s0272-7714(02)00382-7
Hale SS, HughesMM BHW (2018) Historical trends of benthic invertebrate biodiversity spanning 182 years in a southern New England Estuary. Estuar Coasts 41:1525–1538. https://doi.org/10.1007/s12237-018-0378-7
Harris SA, Cyrus DP (1995) Occurrence of larval fishes in the St. Lucia estuary, KwaZulu-Natal, South Africa. S Afr J Mar Sci 16:333–350. https://doi.org/10.2989/025776195784156601
Harrison T, Whitfield AK (2004) A multi-metric fish index to assess the environmental condition of estuaries. J Fish Biol 65:683–710. https://doi.org/10.1111/j.0022-1112.2004.00477.x
Hasan MK, Shahriar A, Jim KU (2019) Water pollution in Bangladesh and its impact on public health. Heliyon 5:e02145. https://doi.org/10.1016/j.heliyon.2019.e02145
Hossain MS, Das NG, Sarker S, Rahaman MZ (2012) Fish diversity and habitat relationship with environmental variables at Meghna river estuary Bangladesh. Egypt J Aquat Res 38(3):213–226. https://doi.org/10.1016/j.ejar.2012.12.006
Ishaq F, Khan A (2013) Aquatic biodiversity as an ecological indicator for water quality criteria of river Yamuna in Doon Valley, Uttarakhand, India. World J Fish Maine Sci 5:322–334. https://doi.org/10.5829/idosi.wjfms.2013.05.03.72126
Islam R, Faysal SM, Amin MR, Juliana FM, Islam MJ, Alam MJ, Hossain MN, Asaduzzaman M (2017) Assessment of pH and total dissolved substances (TDS) in the commercially available bottled drinking water. IOSR J Nurs Health Sci 6:35–40. https://doi.org/10.9790/1959-0605093540
Ji ZG (2008) Hydrodynamics and water quality. In: Modeling rivers, lakes and estuaries, Wiley
Jindal R, Sharma C (2011) Studies on water quality of Sutlej River around Ludhiana with reference to physicochemical parameters. Environ Monit Assess 174:417–425. https://doi.org/10.1007/s10661-010-1466-8
Jonghman RHG, Ter Braak CJF, Van Tongeren OFR (1995) Data analysis in community and landscape ecology. Cambridge University Press, Cambridge, UK
Kadye WT, Magadza CHD, Moyo NAG, Kativu S (2008) Stream fish assemblages in relation to environmental factors on a Montane Plateau (Nyika Plateau, Malawi). Environ Biol Fishes 83:417–428. https://doi.org/10.1007/s10641-008-9364-4
Kenworthy JM, Paterson DM, Bishop MJ (2016) Response of benthic assemblages to multiple stressors: comparative effects of nutrient enrichment and physical disturbance. Mar Ecol Prog Ser 562:37–51. https://doi.org/10.3354/meps11935
Le Pape O, Bonhommeau S, Nieblas AE, Fromentin JM (2017) Overfishing causes frequent fish population collapses but rare extinctions. Proc Natl Acad Sci 114:E6274–E6274. https://doi.org/10.1073/pnas.1706893114
Liu C, Liu L, Shen H (2010) Seasonal variations of phytoplankton community structure in relation to physico-chemical factors in Lake Baiyangdian, China. Procedia Environ Sci 2:1622–1631. https://doi.org/10.1016/j.proenv.2010.10.173
Lobato RDO, Cruz LC, Tavares PB, Wasielesky W, Maciel FE, Lima JV (2020) Influence of physicochemical parameters on Litopenaeus vannamei survival. Pan-Am J Aquatic Sci 15(2):74–80
Lombarte A, Gordoa A, Whitfield AK, James NC, Tuset VM (2012) Eco morphological analysis as a complementary tool to detect changes in fish communities following major perturbations in two South African estuarine systems. Environ Biol Fishes 94:601–614
Martino EJ, Able KW (2003) Fish assemblages across the marine to low salinity transition zone of a temperate estuary. Estuar Coast Shelf Sci 56:969–987. https://doi.org/10.1016/S0272-7714(02)00305-0
Matta G, Naik PK, Machell J, Kumar A, Gjyli L, Tiwari AK, Kumar A (2018) Comparative study on seasonal variation in hydro-chemical parameters of Ganga River water using comprehensive pollution index (CPI) at Rishikesh (Uttarakhand) India. Desalin Water Treat 118:87–95. https://doi.org/10.5004/dwt.2018.22487
Moyle PB, Leidy RA (1992) Conservation Biology. In: Fiedler PL, Jain SK (eds) Loss of biodiversity in Aquatic ecosystems: Evidences from fish faunas. Springer, Boston, pp 129–169
Munawar M (1970) Limnological studies on freshwater pond of Hyderabad-India. Hydrobiologia 35:127–162. https://doi.org/10.1007/BF00751286
Nelson JA, Johnson DS, Deegan LA, Spivak AC, Sommer NR (2019) Feedbacks between nutrient enrichment and geomorphology alter bottom-up control on food webs. Ecosystems 22:229–242. https://doi.org/10.1007/s10021-018-0265-x
Ngodhe SO, Raburu PO, Achieng A (2014) The impact of water quality on species diversity and richness of macroinvertebrates in small water bodies in Lake Victoria, Kenya. J Ecol Nat Environ 6(1):32–41
Palanivel PS, Veeraiyan B, Palingam G, Perumal M (2019) Influence of physico-chemical parameters and pCO2 concentration on mangroves-associated polychaetes at Pichavaram, southeast coast of India. SN Appl Sci. https://doi.org/10.1007/s42452-019-1581-2
Paul AL (1991) Distribution, ecology and biology of fresh water prawns (Macrobrachium spp) of North Eastern region. Dissertation, North- Eastern Hill University.
Potter IC, Tweedley JR, Elliott M, Whitfield AK (2015) The ways in which fish use estuaries: a refinement and expansion of the guild approach. Fish Fish 16(2):230–239. https://doi.org/10.1111/faf.12050
Pouso S, Uyarra MC, Borja A (2018) The recovery of estuarine quality and the perceived increase of cultural ecosystem services by beach users: a case study from northern Spain. J Environ Manage 212:450–461. https://doi.org/10.1016/j.jenvman.2018.02.033
Rani N, Sinha RK, Prasad K, Kedia DK (2011) Assessment of temporal variation in water quality of some important rivers in middle Gangetic plains, India. Environ Monit Assess 174:401–415. https://doi.org/10.1007/s10661-010-1465-9
Rao AM (2001) An environmental assessment on SIPCOT industrial complex, Cuddalore, Tamilnadu in relation to water pollution and its ethical implications. Dissertation, Annamalai University.
Rashed-un-Nabi M, Al-Mamun MA, Ullah MH, Mustafa MG (2011) Temporal and spatial distribution of fish and shrimp assemblage in the Bakkhali river estuary of Bangladesh in relation to some water quality parameters. Mar Biol Res 7:436–452. https://doi.org/10.1080/17451000.2010.527988
Rathnayake SW, Sirimanna SR, Dissanayake CT (2021) Variation in fish abundance in different habitats of a tropical Lagoon with an attempt to model the effects of physicochemical parameters on their abundance. Ocean Sci J 57:91–103. https://doi.org/10.1007/s12601-021-00048-6
Ravindra K, Ameena M, Monika R, Kaushik A (2003) Seasonal variations in physicochemical characteristics of river Yamuna in Haryana and its ecological best designated- use. J Environ Monit 5:419–426. https://doi.org/10.1039/B301723K
Rehman A, Dabrowski J, McCulloch J (2020) Dissolved oxygen prediction in prawn ponds from a group of one step predictors. Inform Process Agric 7:307–317. https://doi.org/10.1016/j.inpa.2019.08.002
RenQ XianW, LiuC LiW (2019) Spring-time nektonic invertebrate assemblages of and adjacent to the Yangtze Estuary. Estuar Coast Shelf Sci 227:106338–113077. https://doi.org/10.1016/j.ecss.2019.106338
Royer TV, Tank JL, David MB (2004) Transport and fate of nitrate in headwater agricultural streams in Illinois. J Environ Qual 33:1296–1304. https://doi.org/10.2134/jeq2004.1296
Shah JA, Pandit AK (2013) Relation between physicochemical limnology and crustacean community in Wular Lake of Kashmir Himalaya. Pakistan J Biol Sci 16:976–983. https://doi.org/10.3923/pjbs.2013.976.983
Sharma A, Subba BR (2005) General biology of freshwater prawn Macrobrachium lamarrei (H. Milne Edwards) of Biratnagar. Nepal Our Nature 3:31–41. https://doi.org/10.3126/on.v3i1.332
Sharma RC, Singh N, Chauhan A (2016) The influence of physicochemical parameters on phytoplankton distribution in a headwater stream of Garhwal Himalays: a case study. Egypt J Aquat Res 42:11–21. https://doi.org/10.1016/j.ejar.2015.11.004
Sharma A, Sharma RC, Anthwal A (2007) Monitoring phytoplankton diversity in the hill stream Chandrabhaga in Garhwal Himalayas. Life Sci J 4:80–84
Sharma N (2015) Taxonomy and population dynamics of freshwater prawns inhabiting some Jammu waters. Dissertation, University of Jammu.
Sheaves M, Baker R, Nagelkerken I, Connolly RM (2015) True value of estuarine and coastal nurseries for fish: incorporating complexity and dynamics. Estuar Coasts 38(2):401–414. https://doi.org/10.1007/s12237-014-9846-x
Singh MR, Gupta A, Beeteswari KH (2010) Physico-chemical properties of water samples from Manipur river system, India. J Appl Sci Environ Manag 14:85–89. https://doi.org/10.4314/jasem.v14i4.63263
Smith VH (2003) Eutrophication of freshwater and coastal marine ecosystems: a global problem. Environ Sci Pollut Res 10(2):126–139. https://doi.org/10.1065/espr2002.12.142
Surana R, Subba BR, Limbu KP (2010) Physico-chemical studies on Chimdi lake of Sunsari district during its restoration stage. Our Nature 8:258–269. https://doi.org/10.3126/on.v8i1.4337
Thomas T (2011) Freshwater shrimps of the family Atyidae de Haan of Kerala and culture of a selected species for forage/food in aquaculture. Dissertation, Mahatma Gandhi University
Varadharajan D, Soundarapandian P, Pushparajan N (2012) Effect of physicochemical parameters on crabs biodiversity. J Marine Sci Res Develop 3:116. https://doi.org/10.4172/2155-9910.1000116
Weber Scannell P, Duffy LK (2007) Effects of total dissolved solids on aquatic organisms: a review of literature and recommendation for salmonid species. Am J Environ Sci 3:1–6. https://doi.org/10.3844/ajessp.2007.1.6
Welch PS (1952) Limnology, 2nd edn. Mc Graw-Hill Book Company Inc, New York
Wetzel RG, Likens GE (1991) Limnological Analyses, 2nd edn. Springer, New York
Whitfield AK (1994) An estuary-association classification for the fishes of southern Africa. South African Journal of Science 90:411–17 https://hdl.handle.net/10520/AJA00382353_5904
Wilkerson F, Dugdale R (2016) Aquatic microbial ecology and biogeochemistry: a dual perspective. In: Gilbert P, Kana T (eds) The ammonium paradox of an urban high-nutrient low-growth estuary. Springer, Cham, pp 117–126
Xu H, Jiang Y, Al-Rasheid KAS, Al-Faraj SA, Song W (2011) Application of an indicator based taxonomic relatedness of ciliated protozoan assemblages for marine environmental assessment. Environ Sci Poll Res 18:1213–1221. https://doi.org/10.1007/s11356-011-0476-6
Xue Y, Song J, Zhang Y, Kong F, Wen M, Zhang G (2016) Nitrate pollution and preliminary source identification of surface water in a semi-arid river basin. Using Isot Hydrochem Approaches Water 8:328. https://doi.org/10.3390/w8080328
Yang XE, Wu X, Hao HL, He ZL (2008) Mechanisms and assessment of water eutrophication. J Zhejiang Univ Sci B 9(3):197–209. https://doi.org/10.1631/jzus.B0710626
Acknowledgements
The authors are grateful to the University Grants Commission, New Delhi, for providing financial support in the form of a Junior research fellowship to Ms. Nidhi Slathia, the corresponding author. We also thank the Head of the Department, Zoology, for providing the necessary facilities to carry out the research work. Also, the help and cooperation of local fishermen are greatly acknowledged for carrying out this study. We are grateful to Prof. Jayachandran KV (Honorary professor, University of Kerala) for his guidance and assistance in consolidating the manuscript in its present form.
Funding
The present research was funded by University Grants Commission, New Delhi, through NET-JRF. One of the authors (Nidhi Slathia) received a fellowship through NET-JRF from University Grants Commission. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Code availability
Not applicable.
Ethical approval
All applicable international, national and institutional guidelines for the care and use of animals were followed.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Slathia, N., Langer, S. & Jasrotia, R. Assessment of water quality and its effect on prawn abundance in three tributaries of Shiwalik rivers: Chenab and Ravi of Jammu, India—a case study. Appl Water Sci 13, 77 (2023). https://doi.org/10.1007/s13201-023-01882-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-023-01882-w