Abstract
In tropical estuaries, fish diversity varies spatially and temporally due to behavioral processes such as reproductive migrations, predator avoidance, and foraging, which are affected by water quality. Eutrophication is one of the main factors affecting water quality in estuaries. The objective of this study was to determine variation in fish assemblage explained by fluctuating water quality in the Buenaventura Bay. Fish were captured using artisanal trawl nets during the wet, dry, and transitional seasons at four sampling sites. Additionally, alkalinity; phosphate, nitrite, and nitrate concentrations; dissolved oxygen; pH; temperature; and suspended solids were measured. Multivariate analysis was used to assess the effect of water quality on fish assemblage. In Buenaventura Bay, the assemblage composition of Pseudupeneus grandisquamis, Daector dowi, and Citharichthys gilberti was affected by nitrate concentration. Moreover, large fish biomasses were associated with high nitrite concentration, intermediate salinity, and low dissolved oxygen, suggesting that these estuaries are dominated by species tolerant to poor water quality. Species richness was associated with low nitrate and phosphate concentrations, more suitable water quality indicators, and intermediate temperatures. These results suggest that the deteriorating water quality of estuaries as a result of the anthropogenic impact could increase dominance and decrease richness, resulting in structural changes of fish assemblages.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The geographic, biotic, and abiotic factors affect fish richness and abundance in estuaries (Brown et al. 2007). The geographic factors include connectivity, while the biotic factors include reproductive migrations, predator avoidance, and foraging (Sheaves et al. 2015) and the abiotic factors include salinity, temperature, dissolved oxygen, sediments, and nutrients, among others (Menegotto et al. 2019; Rau et al. 2019). The fluctuation of these physicochemical variables determines the water quality, influencing the dynamic of aquatic organisms and regulating the ecological processes (Ji 2008).
Water quality of estuarine ecosystems can be characterized using the concentration ranges of nitrogen, phosphorous, and dissolved oxygen, among other characteristics, which promote appropriate ecosystem functioning and support the generation of ecosystem services (Foley et al. 2015; Pouso et al. 2018). In particular, the Colombian Pacific region is strongly socioeconomically dependent on the ecosystem services for the local fish consumption and the commercialization of fishery resources (Saavedra-Díaz et al. 2016; Salas et al. 2019; Villanueva and Flores-Nava 2019). However, previous studies from this region suggest that pollutant concentrations affect benthic communities (Martínez et al. 2019) and that these pollutants are bioaccumulating in organisms of commercial interest (Duque and Cogua 2016; Gamboa-García et al. 2020) as well as in organisms at higher trophic levels (Gamboa-García et al. 2018b).
Water quality is affected by anthropogenic waste discharge, which in turn affects pollutant concentrations and physicochemical variables and, ultimately, ecological processes such as the nutrient cycles, primary production, trophic relationships, and consumer–species dynamics (Barletta et al. 2019; Jickells et al. 2017; Lemley et al. 2017; Nie et al. 2018; Warry et al. 2016). In particular, the effect of eutrophication of coastal ecosystems caused by nutrients from rivers and discharge from adjacent communities on fish assemblages remains unknown. Eutrophication may positively affect fish assemblage by increasing secondary production through a bottom-up trophic cascade or may negatively affect fish assemblage by subjecting fish to physiological stress or hypoxia (de Mutsert et al. 2016; Fong and Fong 2018; Kenworthy et al. 2016; Nelson et al. 2019; Villafañe et al. 2017; Wilkerson and Dugdale 2016).
Taking into consideration the multiple potential environmental impacts, it is critical to study estuarine biodiversity and its dynamics at different scales to understand their processes and mechanisms (Duque et al. 2018; França et al. 2011; Sheaves and Johnston 2009; Teichert et al. 2017; Vilar et al. 2013), as well as to elucidate the effects of eutrophication in these ecosystems. Species richness, abundance, and fish biomass can be measured to assess the effect of variations in nutrient concentrations in the ecosystem. Fish biomass, in particular, may be a key variable because certain species are sensitive to gaining or losing weight as a result of eutrophication (de Mutsert et al. 2016), and may affect the total of each fish population biomass as well.
We hypothesized that (i) the diversity of fishes varies among sampling seasons and sites, (ii) the abundance of the most representative fish species of the estuary can be explained by changes in water quality, and (iii) the fish species richness and fish biomass are associated with changes in water quality. The main objective of this study was to assess the effect of water quality on estuarine fish diversity, which would enable the evaluation of potential eutrophication in Buenaventura Bay.
Materials and methods
Study area
This study was carried out in the estuary of Buenaventura Bay at the Tropical Eastern Pacific (77° 16′ W to 3° 56′ N). The estuary spans approximately 70 km2 and has a 16-km-long and 5-m-deep central canal. The unique seawater inflow is known as La Bocana and is formed by Punta Bazán in the north and Punta Soldado in the south, which are approximately 1.6 km apart (Castaño 2002).
The rivers Dagua (66 m3s−1) and Anchicayá (98 m3s−1) flow into this bay (Otero 2004). Moreover, this bay has one of the highest levels of humidity and precipitation worldwide, with ~ 6980 mm of average annual rainfall and two wet seasons (from September to November and April to June) with an average monthly rainfall of 567 mm, which represents a significant freshwater source (Cantera and Blanco 2001). The access channel for ships is 9.5 and 11.3 m deep during the low and high tides, respectively; however, as a result of maintenance dredging activities and canal expansion, the depth at the channel may reach more than 16 m (Montenegro and Torres 2016).
In the estuary, there are two well-differentiated zones: the interior and the exterior bays. Within the interior bay, port activities combined with (i) waste from fishing, logging, and mining activities; (ii) discharge from rivers that flow into the bay; and (iii) domestic discharge from the same municipality have contributed to the increasing levels of potential pollutants. These pollutants are mainly wastewater which include, nitrates, nitrites, sulfates, phosphates, and coliforms, increasing the organic matter in both, during the low and high tides (IIAP 2013). In contrast, the exterior bay is influenced by a larger touristic complex and it is more marine influenced (Cantera and Blanco 2001; Palacios and Cantera 2017).
Field sampling
In order to study the different hydroclimatic conditions of the bay, we conducted three sampling trips at different seasons. The first one during the wet season (November 2018, total month precipitation = 753.8 mm), the second one in the dry season (March 2019, total month precipitation = 321.2 mm), and the last one in the transitional season (July 2019, total month precipitation = 469.2 mm) (IDEAM 2020). The four sampling sites represent a wide range of water salinity, water quality, nutrient availability, and fish assemblage dynamics. All samples were taken at sites with water less than 8 m depth.
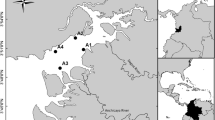
We sampled four sites within the estuary: The first one going from the inside of the bay to the outside was the river estuary (RE, 77° 6′ 33.1″ W and 3° 50′ 51.5″ N), which is the innermost site and is influenced by the Dagua River that flows into it. This site (RE) is the closest sampling site to the urban area of Buenaventura Bay, with around 300,000 inhabitants (DANE 2019). The second site was the inner estuary (IE, 77° 7′ 24.9″ W and 3° 52′ 4.4″ N), which is also located in the internal estuary but is characterized for being a little further from the river discharge and the main urban area. The third site was the outer estuary (OE, 77° 9′ 35.9″ W and 3° 50′ 58.7″ N), which is located in the external estuary and is characterized by having more compacted bottoms and further away from the main urban area. Nevertheless, this site is located near the district of La Bocana, which is inhabited by approximately 3000 people who are highly dependent on marine resources for their own consumption, for tourism (9000 a year approximately), and for supplying the main urban area markets (Escobar-Cárdenas 2009), which is the largest portion of the fish landings (no data); The fourth site was the marine estuary (ME, 77° 12′ 11.4″ W and 3° 49′ 52.44″ N), which is the outermost site and is more influenced by marine conditions and has tourism along the year (15,000 approximately). The average distance between sampling sites was 4 km (Fig. 1).
During each season and at each site, an artisanal trawling net was used for sampling with three replicates. Each trawl sampling lasted 10 min and was performed with a net with 2.54-cm mesh size and 8-m width at the mouth.
Water quality variables, including salinity, dissolved oxygen, pH, temperature, and suspended solids, were measured in situ using a multi-parameter probe YSI 556 MPS. Additionally, water samples were collected to determine alkalinity as well as nitrite, nitrate, and phosphate concentrations using a portable photometer YSI 9300.
In order to characterize the community structure, the captured fish were identified to the species level and counted, and their total length, standard length, and weight were measured. Fish identification was performed using published taxonomic keys (Fischer et al. 1995a, b; Froese and Pauly 2017; Marceniuk et al. 2017; Robertson and Allen 2015; Tavera et al. 2018).
Data analysis
Community structure variations were assessed using species richness (i.e., number of species) and biomass (in g m−2) and calculated using all the captured fish. Abundance analysis was performed using only the most representative species, which were selected using the mean of the highest percent frequency (Eq. 1), abundance (Eq. 2), and weight (Eq. 3) (Martins et al. 2015).
The spatiotemporal analysis was addressed by calculating species richness and biomass and checking normality, using square root transformation when required. Analysis of variance was performed using season, site, and their interaction as main factors, and Tukey’s post hoc test was used to examine statistically significant differences (p < 0.05).
In order to assess water quality, the inorganic nitrogen was measured (nitrites and nitrates, mg L−1), inorganic phosphorous (phosphates mg L−1), and dissolved oxygen (mg L−1), as recommended by Lemley et al. (2015). Moreover, the analysis included salinity (PSU, practical salinity units), temperature (°C), and suspended solids (g L−1) measurements. Additionally, the effect of water quality on the abundances of the most important species (defined by their frequency) was calculated by a canonical correspondence analysis (CCA) using the log(x + 1) transformed matrix within the R environment (R Core Team 2013).
On the other hand, the variation in species richness and biomass explained by water quality was calculated by biological descriptors using Bayesian Generalized Additive Models (GAMs). The models were evaluated by using different variable combinations, and these were compared to select the best model using Akaike information criterion (AIC). For each variable, the presented models were selected considering a ∆AIC > 2 between the model and the next lowest AIC (Krause et al. 2019; Martins et al. 2015). All statistical analyses were performed within the R environment (R Core Team 2013).
It is important to mention that GAMs were used because traditional statistical methods are difficult to interpret when the variables have non-linear relationships (Rudy et al. 2016). Moreover, GAMs have been used for a wide range of applications, including medicine as well as fishery and environmental studies, among others areas of research (Amorós et al. 2018; de Souza et al. 2018; Elith et al. 2008; Tang et al. 2017).
Results
Spatiotemporal variation in fish assemblages
It was collected a total of 69 species belonging to 30 families. The highest species richness was observed during the transitional season at the RE (20 ± 2) and OE (20 ± 2) (F = 10.19, Tukey’s p < 0.001) sites (Table 1). However, the highest biomass (F = 8.69, Tukey’s p < 0.001) was observed during the wet season at the ME site (4.4 ± 2.4 g m−2) (Table 1).
The most representative species (defined using the mean of frequency, abundance, and biomass) were Sphoeroides trichocephalus (57.85%), Cathorops multiradiatus (19.73%), Achirus klunzingeri (18.06%), Lile stolifera (16.7%), and Pseudupeneus grandisquamis (16.31%). The highest abundance of Sphoeroides trichocephalus was recorded during the transitional season at the RE site (227 ± 14.9 fish) (F = 2.16, Tukey’s p < 0.05), whereas the highest abundance of Cathorops multiradiatus was recorded during the wet season at the ME site (51.7 ± 35.4 fish) (F = 18.76, Tukey’s p < 0.001). In contrast, the abundance of Achirus klunzingeri did not show significant differences when the season and sampling site interaction was analyzed, and abundance was the highest at the OE site (F = 5.08, Tukey’s p < 0.01). Similarly, the abundance of Lile stolifera was the highest during the wet season at the IE site (F = 3.24, Tukey’s p < 0.05). In addition, the abundance of Achirus klunzingeri was the highest during the dry season at the OE site (F = 15.14, Tukey’s p < 0.001) (Table 2).
Spatiotemporal variation in water quality
In Buenaventura Bay, the highest salinity was recorded during the dry season (22.24 ± 2.05 PSU), followed by the transitional season (21.17 ± 1.38 PSU) and the wet season (15.83 ± 0.87 PSU) (F = 1212.01, Tukey’s p < 0.001). Moreover, spatial analysis revealed a salinity gradient, in which salinity was the lowest in the inner bay (RE and IE) and increased closer to the sea (OE and ME) (F = 139.51, Tukey’s p < 0.001). The highest salinity was recorded during the dry season at the ME site (25.56 ± 0.15 PSU) (F = 31.78, Tukey’s p < 0.001) (Table 3).
The mean water temperature of the Buenaventura Bay was the highest during the transitional season (28.82 ± 0.23 °C), followed by the wet season (28.09 ± 0.10 °C) and the dry season (27.01 ± 0.36 °C) (F = 2252.09, Tukey’s p < 0.001). In addition, a spatial pattern was observed, in which the mean water temperature was the lowest closer to the sea (ME) (F = 96.05, Tukey’s p < 0.001). The highest mean water temperature was recorded during the transitional season at the RE site (29.00 ± 0.11 °C), and the lowest water temperature was verified during the dry season at the ME site (26.42 ± 0.02 °C) (F = 27.10, Tukey’s p < 0.001).
Across all seasons and sampling sites, the highest concentration of dissolved oxygen was recorded during the wet season at the RE (6.92 ± 0.29 mg L−1), IE (7.17 ± 0.39 mg L−1), and OE (7.18 ± 0.37 mg L−1) sites (F = 11.41, Tukey’s p < 0.001). Between seasons, the highest concentration of dissolved oxygen was recorded during the wet season (6.92 ± 0.54 mg L−1), followed by the dry (5.65 ± 0.67 mg L−1) and the transitional (5.35 ± 0.48 mg L−1) (F = 115.06, Tukey’s p < 0.001) seasons. Spatially, the site with the highest dissolved oxygen concentration was at the ME site (6.31 ± 0.67 mg L−1) (F = 6.18, Tukey’s p < 0.001) (Table 3).
Considering the season and sampling site interaction, the highest concentration of nitrates was recorded during the transitional season at the IE site (2.56 ± 0.03 mg L−1) (F = 13.93, Tukey’s p < 0.001). Among seasons, the highest nitrate concentration was recorded during the transitional season (2.15 ± 0.47 mg L−1) (F = 92.78, Tukey’s p < 0.001). Spatially, the highest nitrate concentrations were recorded in the inner zone (RE = 1.73 ± 0.65 mg L−1, IE = 1.56 ± 0.75 mg L−1) and decreased toward the sites closer to the sea (OE = 1.46 ± 0.18 mg L−1, ME = 1.41 ± 0.5 mg L−1) (F = 5.16, Tukey’s p < 0.01) (Table 3).
Across all seasons and sampling sites, the highest concentration of nitrites was recorded during the transitional season at the IE site (0.17 ± 0.01 mg L−1) (F = 9.23, Tukey’s p < 0.001). Similarly, nitrite concentration was the highest during the transitional season (0.13 ± 0.03 mg L−1) (F = 211.63, Tukey’s p < 0.001) and ranged from high concentrations in the inner estuary (IE = 0.09 ± 0.06 mg L−1, RE = 0.074 ± 0.04 mg L−1) to low concentration toward the sites closer to the sea (F = 10.83, Tukey’s p < 0.001).
Along the estuary, the highest concentration of phosphates was recorded during the wet season at the RE site (0.18 ± 0.06 mg L−1) (F = 4.01, Tukey’s p < 0.01). Although phosphate concentration comparisons were not statistically significant across seasons (F = 1.94), a gradient was detected with the inner sites presenting higher concentrations (IE = 0.13 ± 0.04 mg L−1, RE = 0.12 ± 0.05 mg L−1) than the sites closer to the sea (OE = 0.09 ± 0.06 mg L−1, ME = 0.07 ± 0.01 mg L−1) (F = 5.36, Tukey’s p < 0.01) (Table 3).
Effect of water quality variation on the abundance of the most representative fish species
The canonical correspondence analysis (CCA) suggested that the distribution of the most representative estuarine fish species and the water quality variables nitrites, nitrates, temperature, and dissolved solids were significantly correlated on the first and second ordination axes (r = 0.86 and r = 0.77, respectively), which explained 32% of the variance between fish species and water quality and physicochemical variables (Table 4). The results of the permutational test were significant (p = 0.001), indicating that the relationships between fish species abundance and water quality variables were significant.
The first axis was positively correlated with nitrites and temperature and negatively correlated with total dissolved solids, thus representing the temporal gradient of water quality, with the dry season samples at one end and wet and transitional season samples at the other (Fig. 2 and Table 4). The second axis was negatively correlated with nitrites, temperature, and pH, thus differentiating between the seasons and sampling sites with extreme environmental conditions and the rest of the sampling sites, with the wet season at the ME site together with the dry season at the RE site at one end and the rest of the season–site combinations at the other end.
Canonical correspondence analysis ordination plot illustrating the relationships between the abundance of the most representative species with sampling sites and water quality variables. The arrows indicate water quality variables. Alk = alkalinity, DO = dissolved oxygen, Na = nitrates, Ni = nitrites, P = phosphates, TDS = total dissolved solids, Tem = temperature. Unfilled circles represent the combination between seasons (dry = dry season, rain = wet season, and inter = transitional season) and sampling sites (RE = river estuary, IE = inner estuary, OE = outer estuary, ME = marine estuary)
The water quality variables nitrite concentration, temperature, total dissolved solids, nitrate concentration, and pH significantly affected fish assemblage and habitat distribution comprised by the season–sampling site interactions. Moreover, the habitats comprised by the interaction of the transitional season with the OE and IE sites as well as of the wet season with the IE site displayed the highest nutrient eutrophication, pH, and temperature, although no particular fish assemblage was associated to these environmental conditions. Conversely, the environmental conditions characteristic of the interaction between the dry season and the OE, ME, and RE sites displayed the lowest nutrient eutrophication and temperatures but the highest concentration of dissolved solids, and these conditions were associated with a fish assemblage of three species (Fig. 2 and Table 4).
It was determined a fish assemblage was composed of Sphoeroides trichocephalus, Lile stolifera, Achirus klunzingeri, and Ophioscion typicus, which plotted close to the origin of the axes, suggesting that these fish species are not affected by the water quality gradient (Fig. 2). A second fish assemblage composed of Pseudupeneus grandisquamis, Daector dowi, and Citharichthys gilberti was associated with the dry seasons and low nitrite concentration and temperatures but high total dissolved solid concentrations (Fig. 2). Finally, a third fish assemblage composed of Cathorops multiradiatus and Urotrygon rogersi was associated with low nitrite concentration, temperatures, and pH but high dissolved oxygen concentrations (Fig. 2).
Effects of water quality on fish species richness and biomass variation
The totality of the water quality variables was included in the univariate GAM. The total fish biomass was significantly affected by concentration of nitrates, nitrites, and total dissolved solids; salinity; temperature; and dissolved oxygen (p < 0.05), suggesting that each variable affects fish biomass separately but only accounts for little variation (Table 5). The largest variation was explained by concentrations of nitrates (43.4% (Adj. R2 = 0.25)) and total dissolved solids (31.4% (Adj. R2 = 0.35)), and salinity (28.9% (Adj. R2 = 0.23)), respectively. The model fit for each explanatory variable was low; therefore, a multivariate analysis was performed to assess the overall effect on fish biomass.
On the other hand, fish species richness was significantly and individually affected by temperature; salinity; and concentrations of total solids, nitrites, and dissolved oxygen (p < 0.05) but explained only little variation (Table 5). The largest variation was explained by temperature (39.5% (Adj. R2 = 0.34)) and salinity (30.2% (Adj. R2 = 0.24)). Likewise, the model fit for each explanatory variable was low; therefore, a multivariate analysis was performed to assess the overall effect on fish species richness.
Multivariate analysis showed that the best model for fish biomass included nitrites and dissolved oxygen concentrations and salinity (AIC = 117.97). This model revealed a positive relationship between fish biomass and nitrate concentration and non-linear relationships between salinity (degree = 4) and dissolved oxygen concentration (degree = 2.7) and explained 64.2% of variation (Adj. R2 = 0.54). In contrast, the best multivariate model for fish species richness included nitrates, phosphates, temperature, and pH (AIC = 208.17) and revealed a negative relationship of fish species richness with phosphate and nitrate concentrations as well as a non-linear relationship with temperature (degree = 2.7) and explained 61.2% of variation (Adj. R2 = 0.52) (Table 6).
The total fish biomass showed a positive relationship with nitrite concentration and a non-linear relationship with salinity and dissolved oxygen concentrations, peaking around 17 PSU for salinity and decreasing at 5.5 mg L−1 for dissolved oxygen (Fig. 3).
Effect of water quality predictor variation on fish biomass (multivariate analysis: GAM). Plots represent relationships indicated by the best fitting GAM (Table 6). Smoothed functions are presented as solid lines; dashed lines denote 2 standard errors
In contrast, fish species richness showed a negative relationship with nitrate concentration and was the highest at mean temperatures between 28 and 29 °C but showed no significant relationship with pH (Fig. 4).
Effect of water quality predictor variation on fish species richness (multivariate analysis: GAM). Plots represent relationships indicated by the best fitting GAM (Table 6). Smoothed functions are presented as solid lines; dashed lines denote 2 standard errors
In summary, fish biomass was the largest at higher nitrite concentrations, intermediate salinities between 16 and 18 PSU, and dissolved oxygen between 5 and 5.5 mg L−1, which were the lowest recorded in this study. However, fish species richness was the highest at lower nitrate and phosphate concentrations and temperatures between 28 and 29 °C.
Discussion
Spatiotemporal variation in fish biomass and species richness
In Buenaventura Bay, the fish assemblages varied across seasons and sampling sites. Among the 69 fish species collected, 25 (36%) accounted for 90% of the biomass, 16 (23%) were present in at least 30% of the trawling events, and 15 (22%) accounted for 90% of the abundance. The presence of dominant species has been previously reported in other studies in this region (Molina et al. 2020) as well as in other tropical estuaries (Castillo-Rivera et al. 2010) and might be explained by the tolerance of these species to a wide range of environmental conditions characteristic of these ecosystems (Sheaves et al. 2015).
In this study, the lowest species richness was recorded during the wet season in the inner estuary (n = 6, 8.7%) and during the dry season in the outer estuary (n = 7, 10.14%). The extreme salinities might explain the lower species richness, as few organisms tolerate these extremes (González-Sansón et al. 2016). This trend has been previously reported in other estuaries from the same region, where the lowest fish species richness was recorded during the seasons with the lowest salinity (n = 4, 6.3%) (Páez et al. 2018).
Nevertheless, the dominance of some species and the lower species richness during certain seasons and at some sampling sites could also be explained by multiple anthropogenic impacts (Fausch et al. 1990; Harrison and Whitfield 2004). In fact, other anthropogenically impacted estuaries followed similar trends. For example, in an Ecuadorian estuary characterized by high population density and mangroves disturbed by shrimp farming, four fish species (12%) accounted for 90% of the fish abundance (Shervette et al. 2007). Similarly, in a Mexican coastal lagoon characterized by high population density and tourist activities, only eight fish species (12.5%) accounted for 90% of the fish abundance (Páez et al. 2018). In the Buenaventura Bay, 25 fish species (36%) accounted for 90% of the fish abundance, suggesting that this ecosystem is resilient to the multiple anthropogenic disturbances. Nonetheless, dominant species might thrive in highly disturbed estuarine ecosystems, thus threatening biodiversity within these ecosystems.
On the other hand, the most abundant fish species was Sphoeroides trichocephalus (Tetraodontidae), in particular during the transitional season and at sites with contrasting characteristics: river discharge (RE) and compacted bottoms (OE). The transitional season corresponds to the July month, which is one of the periods of highest flux of tourism in the Dagua basin and Bocana sand beaches (Herrera et al. 2007; Ospina Niño 2017). The Dagua River drains into the RE site, and Bocana is close to the OE site, which may suggest that during this time there is an increase in anthropogenic discharges over these areas, which, paradoxically, is related to higher abundances of S. trichocephalus. Moreover, the wide environmental distribution of this species may be explained by the differential niche use of juvenile and adult fish (Velasco and Wolff 2000). Juveniles might benefit from murkier waters for predator avoidance, while adults might exploit multiple bottom types for foraging. Finally, S. trichocephalus has been reported to tolerate extreme environmental conditions, which allows it to occupy most of the available habitats within the estuary throughout the year (Molina et al. 2020).
Spatiotemporal variation in water quality
In the studied site, it was recorded a temporal gradient in which salinity was the highest during the dry season, followed by the transitional and wet seasons, as well as a spatial gradient in which salinity was the lowest in the inner sites and highest in the outer sites. This pattern has been previously reported in studies in the same bay and was characterized by salinities below 26 PSU due to high rainfall and runoff from the Dagua and Anchicayá Rivers (Cantera et al. 1999; Gamboa-García et al. 2018a, 2020; Molina et al. 2020).
Nitrate and nitrite concentrations were the highest during the transitional season and at the inner sites, whereas phosphate concentration was the highest during the wet season and at the inner sites. During seasons with the highest rainfall, erosion and runoff increase the discharge of nutrients of the organic matter from mangroves, as well as the anthropogenic runoff that flows into river basins (Nie et al. 2018), in this case the Dagua and Anchicayá Rivers, which might explain the observed patterns. The Dagua River basin, which includes the municipalities of Dagua and Buenaventura, is characterized by anthropogenic pressures including human settlements, tourism activities, farming, and the consequent use of fertilizers and mining, among others. Moreover, the inner areas of the estuary are directly affected by domestic wastewater runoff from the Buenaventura Bay. Additionally, pollution and mangrove logging impacts have been reported upstream of the mouth of the Dagua River (Cantera et al. 1999; Romero et al. 2006), which could have an effect on nutrient cycling in these ecosystems. This agrees with reports from northern Brazil (Goiana River estuary), where the highest phosphorus concentration was reported during the season with the highest rainfall and within the inner estuary (Costa et al. 2017).
In this study, most of the sites and seasons presented a moderate to low water quality. For example, the lowest dissolved oxygen concentrations were recorded during the transitional season and at the IE (5.00 ± 0.14 mg L−1) and OE (4.93 ± 0.11) sites, which were classified as moderate. However, these sites were classified as having low water quality due to their nutrient and inorganic phosphorus concentrations. These results highlight the susceptibility to low water quality along the Buenaventura Bay estuary and during the year. Nevertheless, a study in the Tumaco Bay (Colombian Pacific, closer to Ecuador) reported that phosphate concentration had a range of 0.2 ± 0.1 mg L−1 and nitrite plus nitrate concentrations of 1.9 ± 1.8 mg L−1 (Guzmán et al. 2014), which were similar to the ranges found in Buenaventura Bay. In that study, a phytoplankton characterization was performed, and an oceanographic analysis, which suggested that despite the susceptibility of the Tumaco Bay, water quality was improved by the hydrodynamics of the system, which may flow away the pollutants. Therefore, the variation in the water quality and the resilience of the fish community in Buenaventura Bay may be explained by its hydrodynamic regime, the prominent tide range, and the high seasonal variation of river flow, which, similar to Tumaco bay, may improve the ecosystem services.
Effect of water quality on variations in assemblage of the most representative fish species
In the estuary of Buenaventura Bay, the most representative fish species were distributed across three assemblages according to water quality variables. The dry season on one side, and wet and transitional seasons on the other, strongly affected fish assemblage structure. The environmental variables that were correlated most strongly with the fish assemblages were mean nitrite concentration and temperature. One assemblage was composed of Sphoeroides trichocephalus, Lile stolifera, Achirus klunzingeri, and Ophioscion typicus. These species were recorded across seasons and sampling sites in more than 45% of the trawling events, suggesting that these fish species are tolerant of water quality variation. This trend has been previously reported for the estuarine resident species in Buenaventura Bay (Molina et al. 2020), as well as in other estuaries around the world (Cabral et al. 2011; Franco et al. 2006; Martinho et al. 2007). In general, species with a wide physiological tolerance breadth tend to predominate in these ecosystems (Potter et al. 2015). For example, Sphoeroides annulatus is classified as euryhaline and tends to dominate a great proportion of assemblages it is part of (Chávez Sánchez et al. 2008). Interestingly, this trend has also observed in the Buenaventura Bay as this species represented the highest abundance during the wet and transitional seasons and was tolerant to nutrient concentration variation, dissolved oxygen, and temperature changes.
On the other hand, a second fish assemblage composed of Pseudupeneus grandisquamis, Daector dowi, and Citharichthys gilberti was the most susceptible to nitrite concentration and temperature, and was only reported during the dry season. Previous studies have reported the effect of inorganic nitrogen concentration (Wilkerson and Dugdale 2016) as well as temperature on fish assemblages (Harrison and Whitfield 2006; Molina et al. 2020; Rau et al. 2019). Even though these species were only recorded during the season with the highest salinity, they were also classified as being highly dependent on bottom characteristics considering their movement and foraging behavior (Ramírez-Luna et al. 2008; Rau et al. 2019). Therefore, the increasing nitrite concentration in combination with increasing temperatures due to solar radiation could result in a bottom-up nutrient control, which could increase the eutrophication conditions and negatively affect the fish assemblages. Moreover, increased nitrite concentration (Camargo and Alonso 2006; Schlacher et al. 2007) and water temperature (Jeffries et al. 2016; Madeira et al. 2016) could represent physiologically stressful surroundings for fishes.
Furthermore, a third fish assemblage of fishes composed of Cathorops multiradiatus and Urotrygon rogersi was recorded during the wet season in the outer bay and was associated with low pH and high dissolved oxygen concentrations, which is characteristic of the runoff of the rivers Dagua and Anchicayá (Cantera and Blanco 2001). The distribution of species forming this assemblage is consistent with that reported in previous studies from this region (Castellanos-Galindo et al. 2006; Molina et al. 2020). Moreover, the highest abundance of Cathorops multiradiatus and Urotrygon rogersi recorded in the outer estuary region might be explained by the runoff of the rivers that creates environmental conditions to adjacent waters, facilitating resource provisioning to the most marine species (Elliott et al. 2007; Potter et al. 2015; Molina et al. 2020). Finally, fish assemblages varied mostly temporally as an effect of nitrite concentration and temperature, suggesting that water quality and estuarine ecosystem services in the Buenaventura Bay are susceptible to eutrophication and highlighting the complexity and ecological relevance of the processes.
Effect of water quality on fish species richness and biomass variation
In the estuary of Buenaventura Bay, fish species richness and biomass were affected by water quality. Higher biomasses were recorded in low-quality waters, enriched with nitrites and with a low dissolved oxygen. This trend was reported during a hypoxia event in a Mexican estuary, where the fish species representing the highest biomass benefited from the bottom-up effect as a result of primary and secondary production and could also tolerate low dissolved oxygen concentrations, which allowed them to avoid predators (de Mutsert et al. 2016). In summary, in the Buenaventura Bay, the increased fish biomass and dominance of certain species could be an indicator of the effect of low water quality.
In contrast, fish species richness was the highest at intermediate salinities, which is consistent with results from the Málaga Bay, an adjacent estuary to the Buenaventura Bay, where the highest species richness was recorded at intermediate salinities (Castellanos-Galindo and Krumme 2015). These open estuaries are characterized by a wide salinity range: low-salinity habitats (under 10 PSU) that are unsuitable for marine fish in some reported estuaries (Martino and Able 2003), as well as intermediate-salinity habitats. Thus, these relatively intermediate salinities could provide a salinity ecotone, which could be tolerated by resident estuarine species as well as marine species that also depend on the estuary ontogenetically.
Furthermore, a low species richness was reported in low-quality waters, characterized by high nitrate and phosphate concentrations. Increased nitrate and phosphate concentrations have been previously linked to anthropogenic activities (Camargo and Alonso 2006; Smith 2003; Wilkerson and Dugdale 2016), such as wastewater runoff from urban settlements into estuaries, which in turn affects ecological cycles (Berbel et al. 2015). This suggests that the presence of these nutrients indicates a disturbed and low-quality habitat as a result of urban wastewater runoff, which contains nutrients as well as other pollutants. Consequently, fish could suffer from pathologies of different organs, such as the gills, liver, and kidney, or become more susceptible to parasites (Schlacher et al. 2007). Moreover, this could affect their vitality, affecting community structure and ecosystem functioning. Thus, the effect of anthropogenic nutrient runoff on fish assemblages may be evident in Buenaventura Bay, particularly in relation to nitrates, as this nutrient presented the highest concentrations in most of the estuary.
Considering the socioeconomic importance of the Buenaventura Bay estuary for the region for tourism and fishing for livelihood and commercial purposes, it is critical to monitor, control, and treat anthropogenic runoff that might flow into the estuary. In addition, the relevant authorities should develop initiatives to monitor and assess septic tanks from rural communities and tourist centers adjacent to the sea. This study highlights the importance of assessing inorganic pollution within estuaries, and future studies should complement this with histopathology of fish and its potential effect on human health, in addition, chlorophyll-a and microbiological analyses. Moreover, fish assemblages could be used as ecosystem functioning indicators, and certain fish populations should be permanent monitored.
Data availability
The data in this study are available upon reasonable request to the corresponding author.
References
Amorós R, Murcia M, Ballester F, Broberg K, Iñiguez C, Rebagliato M, Skröder H, González L, Lopez-Espinosa MJ, Llop S (2018) Selenium status during pregnancy: influential factors and effects on neuropsychological development among Spanish infants. Sci Total Environ 610–611:741–749. https://doi.org/10.1016/j.scitotenv.2017.08.042
Barletta M, Lima ARA, Costa MF (2019) Distribution, sources and consequences of nutrients, persistent organic pollutants, metals and microplastics in South American estuaries. Sci Total Environ 651:1199–1218. https://doi.org/10.1016/j.scitotenv.2018.09.276
Berbel GBB, Favaro DIT, Braga ES (2015) Impact of harbour, industry and sewage on the phosphorus geochemistry of a subtropical estuary in Brazil. Mar Pollut Bull 93(1–2):44–52. https://doi.org/10.1016/j.marpolbul.2015.02.016
Brown R, Jacobs L, Peet R (2007) Species richness: small scale. In: Encyclopedia of life science. Wiley, New Jersey
Cabral HN, Fonseca VF, Gamito R, Gonalves CI, Costa JL, Erzini K et al (2011) Ecological quality assessment of transitional waters based on fish assemblages in Portuguese estuaries: the Estuarine Fish Assessment Index (EFAI). Ecol Indic 19:144–153. https://doi.org/10.1016/j.ecolind.2011.08.005
Camargo JA, Alonso Á (2006) Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: a global assessment. Environ Int 32(6):831–849. https://doi.org/10.1016/j.envint.2006.05.002
Cantera JR, Blanco JF (2001) The estuary ecosystem of Buenaventura Bay, Colombia. In: Seeliger U, Kjerfve B (eds) Coastal marine ecosystems of Latin America. Ecological studies (analysis and synthesis), vol 144. Springer, Berlin, pp 265–280
Cantera JR, Thomassin BA, Arnaud PM (1999) Faunal zonation and assemblages in the Pacific Colombian mangroves. Hydrobiologia 413:17–33. https://doi.org/10.1023/A:1003890826741
Castaño C (2002) Golfos y bahías del Pacífico colombiano. In: Golfos y bahías de Colombia. Banco de Occidente, Cali, p 530
Castellanos-Galindo GA, Krumme U (2015) Tides, salinity, and biogeography affect fish assemblage structure and function in macrotidal mangroves of the Neotropics. Ecosystems 18(7):1165–1178. https://doi.org/10.1007/s10021-015-9887-4
Castellanos-Galindo GA, Caicedo-Pantoja JA, Mejía-Ladino LM, Rubio E (2006) Peces marinos y estuarinos de Bahía Málaga, Valle del Cauca, Pacífico colombiano. Biota Colombiana 7(2):263–282
Castillo-Rivera M, Zárate-Hernández R, Ortiz-Burgos S, Zavala-Hurtado J (2010) Diel and seasonal variability in the fish community structure of a mud-bottom estuarine habitat in the Gulf of Mexico. Mar Ecol 31(4):633–642. https://doi.org/10.1111/j.1439-0485.2010.00394.x
Chávez Sánchez MC, Álvarez-Lajonchère L, Abdo De La Parra MI, García Aguilar N (2008) Advances in the culture of the Mexican bullseye puffer fish Sphoeroides annulatus, Jenyns (1842). Aquac Res 39(7):718–730. https://doi.org/10.1111/j.1365-2109.2008.01924.x
Costa CR, da Costa MF, Barletta M, Alves LHB (2017) Interannual water quality changes at the head of a tropical estuary. Environ Monit Assess 189(12):628. https://doi.org/10.1007/s10661-017-6343-2
DANE (2019) Resultados Preliminares: CENSO NACIONAL DE POBLACIÓN Y VIVIENDA 2018
de Mutsert K, Steenbeek J, Lewis K, Buszowski J, Cowan JH, Christensen V (2016) Exploring effects of hypoxia on fish and fisheries in the northern Gulf of Mexico using a dynamic spatially explicit ecosystem model. Ecol Model 331:142–150. https://doi.org/10.1016/j.ecolmodel.2015.10.013
de Souza JB, Reisen VA, Franco GC, Ispány M, Bondon P, Santos JM (2018) Generalized additive models with principal component analysis: an application to time series of respiratory disease and air pollution data. J R Stat Soc Ser C Appl Stat 67(2):453–480. https://doi.org/10.1111/rssc.12239
Duque G, Cogua P (2016) Mercurio en peces de la bahía de Buenaventura. Ingenium 10(29):11–17 Retrieved from https://repository.usc.edu.co/bitstream/20.500.12421/794/1/MERCUR~1.PDF
Duque G, Panesso-Guevara M, Cogua P (2018) Áreas marinas protegidas: ¿son efectivas? Caso bahía Málaga. In P. Lavelle & J. Leon (Eds.), Ingeniería Ambiental: manejo de ecosistemas, concepción de políticas publicas y reciclaje de materiales (pp. 159–179). Retrieved from https://www.uneditorial.com/ingenieria-ambiental-manejo-de-ecosistemas-concepcion-de-politicas-publicas-y-reciclaje-de-materiales-ingenieria-ambiental.html
Elith J, Leathwick JR, Hastie T (2008, July) A working guide to boosted regression trees. J Anim Ecol 77:802–813. https://doi.org/10.1111/j.1365-2656.2008.01390.x
Elliott M, Whitfield AK, Potter IC, Blaber SJM, Cyrus DP, Nordlie FG, Harrison TD (2007) The guild approach to categorizing estuarine fish assemblages: a global review. Fish Fish 8(3):241–268. https://doi.org/10.1111/j.1467-2679.2007.00253.x
Escobar-Cárdenas AM (2009) PUERTO PACÍFICO: TERMINAL TURÍSTICO Y PRODUCTIVO PARA EL DESARROLLO SOSTENIBLE EN LA BOCANA/VALLE DEL C. Pontificia Universidad Javeriana
Fausch K, Lyons J, Karr J, Angermeier P (1990) Fish communities as indicators of environmental degradation. Am Fish Soc Symp 8:123–144
Fischer W, Krupp F, Schneider W, Sommer C, Carpenter KE, Niem VH (1995a) Guía FAO para la identificación de especies para los fines de la pesca. Pacífico centro-oriental. Volumen II. Vertebrados - Parte 1. FAO, Roma
Fischer W, Krupp F, Schneider W, Sommer C, Carpenter KE, Niem VH (1995b) Guia FAO para la identificación de especies para los fines de la pesca. Pacífico centro-oriental. Volumen III. Vertebrados - Parte 2. FAO, Roma
Foley MM, Duda JJ, Beirne MM, Paradis R, Ritchie A, Warrick JA (2015) Rapid water quality change in the Elwha River estuary complex during dam removal. Limnol Oceanogr 60(5):1719–1732. https://doi.org/10.1002/lno.10129
Fong CR, Fong P (2018) Nutrient fluctuations in marine systems: press versus pulse nutrient subsidies affect producer competition and diversity in estuaries and coral reefs. Estuar Coasts 41(2):421–429. https://doi.org/10.1007/s12237-017-0291-5
França S, Costa MJ, Cabral HN (2011) Inter- and intra-estuarine fish assemblage variability patterns along the Portuguese coast. Estuar Coast Shelf Sci 91(2):262–271. https://doi.org/10.1016/j.ecss.2010.10.035
Franco A, Franzoi P, Malavasi S, Riccato F, Torricelli P, Mainardi D (2006) Use of shallow water habitats by fish assemblages in a Mediterranean coastal lagoon. Estuar Coast Shelf Sci 66(1–2):67–83. https://doi.org/10.1016/j.ecss.2005.07.020
Froese R, Pauly D (2017) Fishbase. World Wide Web electronic publication
Gamboa-García DE, Duque G, Cogua P (2018a) Structural and compositional dynamics of macroinvertebrates and their relation to environmental variables in Buenaventura Bay. Boletin de Investigaciones Marinas y Costeras 47(1). https://doi.org/10.25268/bimc.invemar.2018.47.1.738
Gamboa-García DE, Duque G, Cogua P, Freire MF (2018b) Mercurio total en plumas de Pelecanus occidentalis en el Pacífico vallecaucano. In: Cogua P, Ordóñez E, Chávez M, Nieto L, Molina S, Fernández S et al (eds) Comunidades epistemológicas. Investigando la actualidad desde diversas disciplinas. Tomo II. Universidad Santiago de Cali, Cali, p 184
Gamboa-García DE, Duque G, Cogua P, Marrugo-Negrete JL (2020) Mercury dynamics in macroinvertebrates in relation to environmental factors in a highly impacted tropical estuary: Buenaventura Bay, Colombian Pacific. Environ Sci Pollut Res 27(4):4044–4057. https://doi.org/10.1007/s11356-019-06970-6
González-Sansón G, Aguilar-Betancourt C, Kosonoy-Aceves D, Lucano-Ramírez G, Ruiz-Ramírez S, Flores-Ortega JR (2016) Variaciones espaciales y temporales de la abundancia de peces juveniles en la laguna costera Barra de Navidad, Jalisco, México: Efectos del huracán Jova. Rev Biol Mar Oceanogr 51(1):123–136. https://doi.org/10.4067/s0718-19572016000100012
Guzmán ÁI, Zambrano-Ortiz MM, Casanova-Rosero R, Selvaraj JJ, Martínez A (2014) La condición ecológica de la bahía de Tumaco (Pacífico colombiano): evaluación de la calidad del agua y del fitoplancton. The ecological condition of Tumaco Bay (colombian Pacific): an assessment of water quality and phytoplankton. Bol Cient CIOH 32:3–16
Harrison T, Whitfield AK (2004) A multi-metric fish index to assess the environmental condition of estuaries. J Fish Biol 65:683–710. https://doi.org/10.1111/j.0022-1112.2004.00477.x
Harrison T, Whitfield AK (2006) Estuarine typology and the structuring of fish communities in South Africa. Environ Biol Fish 75(3):269–293. https://doi.org/10.1007/s10641-006-0028-y
Herrera J, Flórez L, Ávila I, Falk P, Capella J, Tobón I (2007) Efecto de las embarcaciones de turismo en el comportamiento de grupos de cría de ballena jorobada (Megaptera novaeangliae) en Bahía Málaga, Colombia. In: Félix F (ed) Memorias del Taller de Trabajo sobre el Impacto de las Actividades Antropogénicas en Mamíferos Marinos en el Pacífico Sudeste, pp 88–93 Retrieved from http://www.unep.org/regionalseas/
IDEAM (2020) Consulta y Descarga de Datos Hidrometeorológicos. Retrieved March 18, 2020, from http://dhime.ideam.gov.co/atencionciudadano/
IIAP (2013) Evaluación fisicoquímica y ecológica de aguas costeras en la Bahía de Buenaventura como instrumento de análisis de los aportes contaminantes del río Dagua. Municipio de Buenaventura-Valle, Colombia
Jeffries KM, Connon RE, Davis BE, Komoroske LM, Britton MT, Sommer T et al (2016) Effects of high temperatures on threatened estuarine fishes during periods of extreme drought. https://doi.org/10.1242/jeb.134528
Ji Z-G (2008) Hydrodynamics and water quality. In: Modeling rivers, lakes and estuaries. Wiley
Jickells TD, Buitenhuis E, Altieri K, Baker AR, Capone D, Duce RA, Dentener F, Fennel K, Kanakidou M, LaRoche J, Lee K, Liss P, Middelburg JJ, Moore JK, Okin G, Oschlies A, Sarin M, Seitzinger S, Sharples J, Singh A, Suntharalingam P, Uematsu M, Zamora LM (2017) A reevaluation of the magnitude and impacts of anthropogenic atmospheric nitrogen inputs on the ocean. Glob Biogeochem Cycles 31(2):289–305. https://doi.org/10.1002/2016GB005586
Kenworthy JM, Paterson DM, Bishop MJ (2016) Response of benthic assemblages to multiple stressors: comparative effects of nutrient enrichment and physical disturbance. Mar Ecol Prog Ser 562:37–51. https://doi.org/10.3354/meps11935
Krause KP, Wu C-L, Chu ML, Knouft JH, Krause P (2019) Fish assemblage-environment relationships suggest differential trophic responses to heavy metal contamination. https://doi.org/10.1111/fwb.13248
Lemley DA, Adams JB, Taljaard S, Strydom NA (2015) Towards the classification of eutrophic condition in estuaries. Estuar Coast Shelf Sci 164:221–232. https://doi.org/10.1016/j.ecss.2015.07.033
Lemley DA, Adams JB, Strydom NA, Lemley DA, Adams JB (2017) Testing the efficacy of an estuarine eutrophic condition index: does it account for shifts in flow conditions? https://doi.org/10.1016/j.ecolind.2016.11.034
Madeira D, Vinagre C, Diniz MS (2016) Are fish in hot water? Effects of warming on oxidative stress metabolism in the commercial species Sparus aurata. Ecol Indic 63:324–331. https://doi.org/10.1016/j.ecolind.2015.12.008
Marceniuk AP, Acero AP, Cooke R, Betancur R (2017) Taxonomic revision of the New World genus Ariopsis gill (Siluriformes: Ariidae) , with description of two new species. Zootaxa 4290:1–42. https://doi.org/10.11646/zootaxa.4290.1.1
Martínez O, Molina AE, Duque G (2019) Influencia de la materia orgánica en la abundancia de poliquetos (Annelida:Polychaeta) de la Bahía de Buenaventura, Pacífico Colombiano. In: Campos-Campos NH, Acero A (eds) Ciencias del mar. Una mirada desde la Universidad Nacional de Colombia. Universidad Nacional de Colombia
Martinho F, Leit Ao R, Viegas I, Dolbeth M, Neto JM, Cabral HN, Pardal MA (2007) The influence of an extreme drought event in the fish community of a southern Europe temperate estuary. https://doi.org/10.1016/j.ecss.2007.05.040
Martino EJ, Able KW (2003) Fish assemblages across the marine to low salinity transition zone of a temperate estuary. Estuar Coast Shelf Sci 56(5–6):969–987. https://doi.org/10.1016/S0272-7714(02)00305-0
Martins ACB, Kinas PG, Marangoni JC, Moraes LE, Vieira JP (2015) Medium- and long-term temporal trends in the fish assemblage inhabiting a surf zone, analyzed by Bayesian generalized additive models. Aquat Ecol 49(1):57–69. https://doi.org/10.1007/s10452-015-9504-9
Menegotto A, Dambros CS, Netto SA (2019) The scale-dependent effect of environmental filters on species turnover and nestedness in an estuarine benthic community. Ecology 100(7):e02721. https://doi.org/10.1002/ecy.2721
Molina A, Duque G, Cogua P (2020) Influences of environmental conditions in the fish assemblage structure of a tropical estuary. Mar Biodivers 50(1):1–13. https://doi.org/10.1007/s12526-019-01023-0
Montenegro D, Torres W (2016). LA INCIDENCIA DEL DRAGADO DEL CANAL DE ACCESO DE LOS BUQUES A LOS PUERTOS MARÍTIMOS DE BUENAVENTURA, EN LA PRÁCTICA PESQUERA ARTESANAL DESARROLLADA EN LA COMUNIDAD DE BAZÁN BOCANA. Universidad del Valle
Nelson JA, Johnson DS, Deegan LA, Spivak AC, Sommer NR (2019) Feedbacks between nutrient enrichment and geomorphology alter bottom-up control on food webs. Ecosystems 22(2):229–242. https://doi.org/10.1007/s10021-018-0265-x
Nie J, Feng H, Witherell BB, Alebus M, Mahajan MD, Zhang W, Yu L (2018) Causes, assessment, and treatment of nutrient (N and P) pollution in rivers, estuaries, and coastal waters. Curr Pollut Rep 4(2):154–161. https://doi.org/10.1007/s40726-018-0083-y
Ospina Niño JA (2017) Paisaje y territorio en la playa Juan de Dios, Bahía Málaga, Pacífico colombiano (2005-2016): una aproximación desde la nueva geografía del turismo*. Cuadernos de Geografía: Revista Colombiana de Geografía 26(2):31–52. https://doi.org/10.15446/rcdg.v26n2.59192
Otero JL (2004) Determinación del régimen medio y extremal del nivel del mar para la bahía de Buenaventura. Boletín Científico CCCP 11:30–41
Páez YC, Aguilar-Betancourt CM, González-Sansón G, Rodríguez FN, Gray M (2018) Sediment granulometry and salinity drive spatial and seasonal variability of an estuarine demersal fish assemblage dominated by juvenile fish. Estuar Coast Shelf Sci 212(April):241–252. https://doi.org/10.1016/j.ecss.2018.07.019
Palacios ML, Cantera JR (2017) Mangrove timber use as an ecosystem service in the Colombian Pacific. Hydrobiologia 803(1):345–358. https://doi.org/10.1007/s10750-017-3309-x
Potter IC, Tweedley JR, Elliott M, Whitfield AK (2015) The ways in which fish use estuaries: a refinement and expansion of the guild approach. Fish Fish 16(2):230–239. https://doi.org/10.1111/faf.12050
Pouso S, Uyarra MC, Borja Á (2018) The recovery of estuarine quality and the perceived increase of cultural ecosystem services by beach users: a case study from northern Spain. J Environ Manag 212:450–461. https://doi.org/10.1016/j.jenvman.2018.02.033
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing. Retrieved from http://www.r-project.org/
Ramírez-Luna V, Navia AF, Rubio EA (2008) Food habits and feeding ecology of an estuarine fish assemblage of northern Pacific coast of Ecuador. Pan-Am J Aquat Sci 3(3):361–372
Rau A, Lewin WC, Zettler ML, Gogina M, von Dorrien C (2019) Abiotic and biotic drivers of flatfish abundance within distinct demersal fish assemblages in a brackish ecosystem (western Baltic Sea). Estuar Coast Shelf Sci 220:38–47. https://doi.org/10.1016/j.ecss.2019.02.035
Robertson, D. R., & Allen, G. R. (2015). Shorefishes of the tropical eastern Pacific: online information system
Romero IC, Cantera JR, Peña EJ (2006) Consumo de hojas por herbívoros en manglares del estuario del Río Dagua, Costa Pacífica Colombiana. Rev Biol Trop 54(4):1205–1214. https://doi.org/10.15517/rbt.v54i4.14103
Rudy ACA, Lamoureux SF, Treitz P, van Ewijk KY (2016) Transferability of regional permafrost disturbance susceptibility modelling using generalized linear and generalized additive models. Geomorphology 264(February 2018):95–108. https://doi.org/10.1016/j.geomorph.2016.04.011
Saavedra-Díaz LM, Pomeroy R, Rosenberg AA (2016) Managing small-scale fisheries in Colombia. Mar Stud 15(1):6. https://doi.org/10.1186/s40152-016-0047-z
Salas S, Huchim-Lara O, Guevara-Cruz C, Chin W (2019) Viability and sustainability of small-scale fisheries in Latin America and the Caribbean. https://doi.org/10.1007/978-3-319-76078-0_5
Schlacher TA, Mondon JA, Connolly RM (2007) Estuarine fish health assessment: evidence of wastewater impacts based on nitrogen isotopes and histopathology. Mar Pollut Bull 54(11):1762–1776. https://doi.org/10.1016/j.marpolbul.2007.07.014
Sheaves M, Johnston R (2009) Ecological drivers of spatial variability among fish fauna of 21 tropical Australian estuaries. Mar Ecol Prog Ser 385(2005):245–260. https://doi.org/10.3354/meps08040
Sheaves M, Baker R, Nagelkerken I, Connolly RM (2015) True value of estuarine and coastal nurseries for fish: incorporating complexity and dynamics. Estuar Coasts 38(2):401–414. https://doi.org/10.1007/s12237-014-9846-x
Shervette VR, Aguirre WE, Blacio E, Cevallos R, Gonzalez M, Pozo F, Gelwick F (2007) Fish communities of a disturbed mangrove wetland and an adjacent tidal river in Palmar, Ecuador. Estuar Coast Shelf Sci 72:115–128. https://doi.org/10.1016/j.ecss.2006.10.010
Smith VH (2003) Eutrophication of freshwater and coastal marine ecosystems: a global problem. Environ Sci Pollut Res 10(2):126–139. https://doi.org/10.1065/espr2002.12.142
Tang H, Xu L, Zhou C, Wang X, Zhu G, Hu F (2017) The effect of environmental variables, gear design and operational parameters on sinking performance of tuna purse seine setting on free-swimming schools. Fish Res 196(December):151–159. https://doi.org/10.1016/j.fishres.2017.08.006
Tavera J, Acero A, Wainwright PC (2018) Multilocus phylogeny, divergence times, and a major role for the benthic-to-pelagic axis in the diversification of grunts (Haemulidae). Mol Phylogenet Evol 121(December 2017):212–223. https://doi.org/10.1016/j.ympev.2017.12.032
Teichert N, Pasquaud S, Borja A, Chust G, Uriarte A, Lepage M (2017) Living under stressful conditions: fish life history strategies across environmental gradients in estuaries. Estuar Coast Shelf Sci 188:18–26. https://doi.org/10.1016/j.ecss.2017.02.006
Velasco A, Wolff M (2000) Ictiofauna juvenil de fondos blandos durante la transición de “secas” a lluvias en la costa pacífica de Colombia. Rev Biol Trop 48(1):215–228. https://doi.org/10.15517/rbt.v48i1.29355
Vilar CC, Joyeux JC, Giarrizzo T, Spach HL, Vieira JP, Vaske-Junior T (2013) Local and regional ecological drivers of fish assemblages in Brazilian estuaries. Mar Ecol Prog Ser 485(June 2013):181–197. https://doi.org/10.3354/meps10343
Villafañe VE, Cabrerizo MJ, Erzinger GS, Bermejo P, Strauch SM, Valiñas MS, Helbling EW (2017) Photosynthesis and growth of temperate and sub-tropical estuarine phytoplankton in a scenario of nutrient enrichment under solar ultraviolet radiation exposure. Estuar Coasts 40(3):842–855. https://doi.org/10.1007/s12237-016-0176-z
Villanueva J, Flores-Nava A (2019) The contribution of small-scale fisheries to food security and family income in Chile, Colombia ,and Perú. In: Salas S, Barragán-Paladines M, Chuenpagdee R (eds) Viability and sustainability of small-scale fisheries in Latin America and the Caribbean. MARE Publi, pp 329–352. https://doi.org/10.1007/978-3-319-76078-0_14
Warry FY, Reich P, Cook PLM, Mac Nally R, Thomson JR, Woodland RJ (2016) Nitrogen loads influence trophic organization of estuarine fish assemblages. Funct Ecol 30(10):1723–1733. https://doi.org/10.1111/1365-2435.12647
Wilkerson F, Dugdale R (2016) The ammonium paradox of an urban high-nutrient low-growth estuary. In: Aquatic microbial ecology and biogeochemistry: a dual perspective, pp 117–126. https://doi.org/10.1007/978-3-319-30259-1_10
Acknowledgments
We thank the Universidad Nacional de Colombia for their administrative support. We also thank the Ecología y Contaminación Acuática research group for their support in the field, sample processing, and data analysis.
Funding
We received from the Universidad Nacional de Colombia financial support of the project: “Efectos de los cambios en la calidad del agua en las comunidades de macroinvertebrados y peces del estuario Bahía de Buenaventura” código Hermes 42118. We also received from the Universidad Santiago de Cali financial support of the project: “Fortalecimiento de grupos de investigación” código: 934-621118-204.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The methods in this study were approved by the ethics committee of the Environmental Studies Institute (Spanish acronym: IDEA) of the Universidad Nacional de Colombia and follow international, national, and institutional animal use and care guidelines.
Additional information
Responsible editor: Vedula VSS Sarma
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Duque, G., Gamboa-García, D.E., Molina, A. et al. Effect of water quality variation on fish assemblages in an anthropogenically impacted tropical estuary, Colombian Pacific. Environ Sci Pollut Res 27, 25740–25753 (2020). https://doi.org/10.1007/s11356-020-08971-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08971-2