Abstract
Information regarding cellular anti-senescence attributes of probiotic bacteria vis-à-vis modulation of senescence-associated secretory phenotype (SASP) and mTOR signaling is very limited. The present study assessed anti-senescence potential of secretory metabolites of probiotic Lactobacillus fermentum (Lact. fermentum) using H2O2-induced model of senescence in 3T3-L1 preadipocytes. Application of H2O2-induced cellular senescence characterized by increased cell size and SA-β-gal activity, activation of SASP and reactive oxygen species (ROS), DNA damage response and induction of cell cycle inhibitors (p53/p21WAF1/p16INK4a). Further, a robust stimulation of the PI3K/Akt/mTOR pathway and AMPK signaling was also observed in H2O2-treated cells. However, exposure of cells to cell-free supernatant of Lact. fermentum significantly attenuated phosphorylation of PI3K/Akt/mTOR pathway and alleviated senescence markers p53, p21WAF1, SA-β-gal, p38MAPK, iNOS, cox-2, ROS, NF-κB, and DNA damage response. These results provide evidence that secretory metabolites of Lact. fermentum can mitigate the development as well as severity of stress-induced senescence thereby indicating its utility for use as anti-aging or age-delaying agent.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aging is a multi-factorial deleterious process responsible for increased rate of morbidity and mortality in elderly. Recent advances in geroscience have highlighted the significance of cellular senescence in driving the aging process per se. It has been shown that gradual accumulation of senescent cells in various tissues and organs directly contributes to organ dysfunction and the characteristic aged phenotype [1], while selective elimination of senescent cells in vivo could essentially confer anti-aging attributes by delaying tumorigenesis and attenuation of age-related deterioration of several organs including kidney, heart, and fat [2]. Based on nature of contributing factors, cellular senescence is classified into different types such as replicative senescence, oncogene-induced senescence, or stress-induced senescence. Amongst these, ROS-mediated stress-induced senescence is arguably the most persistent yet modifiable factor responsible for inducing and driving the senescence program [3]. Increased systemic oxidative and inflammatory stressors with age are natural contributors to senescence that ultimately results in age-associated tissue and organ dysfunctions. It is thus not surprising that stress-based cellular models of senescence have been extensively used to understand the process of senescence and to identify potential modulators of cellular senescence.
Probiotics are live microorganisms that, when administered in adequate amounts, confer a health benefit on the host [4]. As such, consumption of probiotics has been associated with several health beneficial effects including modulation of gut microbiota, redox homeostasis, immune responses, and the gut-brain axis [5, 6]. This explains the growing emphasis of probiotic-based dietary interventions to treat and prevent different chronic disorders, particularly associated with stress and inflammation. We and others have previously observed that application of probiotic microbes can also successfully alter different aspects of aging such as prevention of diarrheal diseases, inhibition of colon senescence, protection against pathogens, enhancement of the intestinal barrier function, immunomodulatory effects, and prevention of colon cancer [7,8,9,10,11]. However, despite these growing evidences, the underlying molecular mechanisms governing the effects of probiotics are poorly understood. Further, there is very little information on whether and how probiotic bacteria can modulate different aspects of cellular senescence and, in particular, their role in influencing nutrient sensing pathways vis-à-vis senescence and senescence-associated secretory phenotype (SASP) is little explored. The mechanistic target of rapamycin (mTOR) nutrient sensing pathway is essential not only for normal cellular growth but is also emerging as a prominent regulator of various aspects of cell senescence [12]. Indeed, several studies have reported activation of mTOR pathway during aging and that its targeted abrogation can result in pro-longevity effects [13, 14]. Thus, SASP suppression and mTOR inhibition appear promising candidates in the global emphasis to identify molecular targets of aging and prolonging lifespan. Despite several purported health beneficial effects of probiotics, their role in mitigation of cell senescence is relatively unexplored. Therefore, keeping the foregoing discussion in view, the present study hypothesized that cell-free extract of a probiotic Lactobacillus fermentum (Lact. fermentum) could confer cytoprotective effects against hydrogen peroxide (H2O2)-induced premature senescence in murine preadipocytes by altering mTOR signaling and suppressing activation of SASP. To test our hypothesis, a 3T3-L1 cell line-based model of senescence was established and anti-senescence attributes of probiotic Lact. fermentum culture supernatant were determined using a range of biochemical and molecular parameters. Our results indicate that secretory metabolites of Lact. fermentum can effectively attenuate the development and progression of senescence by countering DNA damage, SASP activation, and stress-induced stimulation of PI3K/Akt/mTOR pathway. These findings suggest novel health beneficial attribute of probiotics that may be implicated in the development of pro-longevity strategies.
Materials and Methods
Cell Line and Culture Conditions
Murine preadipocyte cell line, 3T3-L1 (ATCC, Manassas, VA, USA), was used to assess the effects of probiotic treatment on premature senescence induced by acute treatment with H2O2. Cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM, Gibco; 12800-017) supplemented with 10% FBS (HiMedia laboratories, Mumbai, India; RM9970) at 37 °C and 5% CO2 atmosphere. Studies were performed with cells at initial passage number (~ 3–5) based on our previous observations [15].
Preparation of Probiotic-Conditioned Media
The probiotic (Lact. fermentum) used in present study was isolated from fecal matter of adult human [16]. The bacterial species was identified by 16s rRNA gene sequencing and characterized for probiotic attributes as per WHO/FAO guidelines (data unpublished). This strain has been deposited at the national microbial culture collection repository of National center of cell science, Pune, India, via accession number MCC3160. This particular species of probiotic bacteria was chosen as it showed strong cytoprotective effects against inflammatory and oxidative stressors both in vitro and in vivo in our previous work [17]. The bacterial secretory metabolites were collected as probiotic conditioned media (PCM) as previously described [18]. Briefly, bacteria were aerobically cultured in De Man, Rogosa, and Sharpe media (MRS) (HiMedia laboratories, Mumbai, India; GM369) for ~ 16 h at 37 °C, following which the broth was centrifuged, and the bacterial pellet was resuspended in a small volume of phosphate-buffered saline (PBS, pH 7.4). The bacterial suspension was then aseptically inoculated into DMEM (without FBS and antibiotic solution) and concentration adjusted so as to obtain a final optical density of 0.8 (corresponding to 108 CFU/ml). This bacterial suspension in DMEM was then allowed to grow aerobically with periodic shaking for ~ 16 h at 37 °C. Finally, the bacterial culture in DMEM was centrifuged, pellet discarded, and pH of the supernatant was adjusted to 7.4. After sterile filtration, the supernatant was supplemented with 10% FBS and antibiotic solution to prepare the final PCM for cellular treatment.
Cell Viability Assay
The effect of PCM on cell viability was determined by measuring the reduction of MTT 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) (HiMedia laboratories, Mumbai, India) to formazan crystals. Briefly, 1 × 106 cells/well were seeded into 96-well culture plate in the presence or absence of PCM at different concentrations (v/v) for 72 h at 37 °C in a humidified CO2 incubator. After visual confirmation of cell morphology, spent media was pipetted out, and 100 μL of acid isopropanol (0.1 N HCl in anhydrous isopropanol) was mixed to dissolve formazan crystals and absorbance was read at 540 nm. Cell viability was calculated as previously reported [19].
Senescence Induction and Probiotic Treatment
3T3-L1 cells were seeded into 6-well plates at 3000 cells/cm2 and allowed to grow until about 80% confluency. The concentration and treatment of H2O2 required for senescence induction in present study was based on our previous work [15]. Briefly, cells were exposed to H2O2 at a final concentration of 150 μM for 3 h at 37 °C following which the cells were washed with PBS twice and resuspended in complete DMEM. This treatment regimen was continued for three consecutive days following which the cells were incubated for additional 3 days in complete media only before analysis of various senescence-associated parameters. For PCM treatment, cells were co-exposed with PCM (5% and 10% v/v) and H2O2 for 3 h at 37 °C, and after washing for removal of H2O2, cells were again kept in PCM (5% and 10% v/v in DMEM) overnight. Next day, H2O2 was directly applied to these cells, and after incubation for 3 h, cells were again washed, and fresh PCM was added to the cells [15]. Similar treatment regimen was followed for the third day, and subsequently, all cells were exposed to complete DMEM only for three additional days following which various cellular and biochemical analyses were performed.
Morphology and Cell Size
The changes in cell morphology against various treatments were observed using EVOS FL Auto 2 Imaging system (Thermo Scientific, USA). Senescence-induced changes in cell size were measured using the “area” feature in brightfield channel of AMNIS ImageStream®X Mark II Imaging Flow Cytometer (Merck Millipore, Germany) as previously described [15].
Senescence Associated β-Galactosidase Staining
Cellular senescence was identified by measuring senescence-associated β-galactosidase (SA-β-gal) activity using commercially available kit (K802; Biovision, USA) as per the manufacturer’s protocol. Briefly, cells were washed with PBS and suspended in a fixative solution for 10–15 min at room temperature. Cells were again washed twice with PBS followed by addition of staining solution and overnight incubation at 37 °C in the absence of CO2. Cells were then observed for the development of blue color indicating β-galactosidase activity with the help of EVOS FL Auto 2 Imaging System (Thermo Scientific, USA). Several images were taken and cells were manually counted for enumeration of percentage senescent cells.
Intracellular ROS Assay
After respective treatments, intracellular ROS were measured by using H2DCFDA dye. Briefly, cells were washed and stained with 10 μM H2DCFDA (D399; Molecular Probes, USA) for 30 min. Oxidation of the probe to 2,7-dichlorofluorescein (DCF) was measured by AMNIS ImageStream®X Mark II Imaging flow cytometer (Merck Millipore, Germany). Median fluorescence intensity was calculated and analyzed by INSPIRE ImageStream system software.
RNA Isolation, Reverse Transcription, and Real-Time PCR
Cellular RNA was isolated by using Qiagen RNeasy mini kit (74104, Qiagen, Germany) according to the manufacturer’s protocol with some modifications. RNA was quantified and checked for organic salt and protein contaminants by Nanodrop. Gene expression was performed by qRT-PCR using Verso SYBR Green 1-Step qRT-PCR ROX Mix kit (Thermo Scientific, #AB-4105/C). The PCR reactions were performed in 96-well plates (Applied Biosystems, USA) using the Step one Plus™ Real-Time PCR system (Applied Biosystems, USA). The ΔΔCt method was used for the relative quantification of mRNA, and GAPDH (glyceraldehyde-3-phosphate dehydrogenase) was used as a house keeping control to normalize the mRNA expression [20]. The primer sequences used in present study were as follows: p53-F TTCTGTAGCTTCAGTTCATTGG, p53-R ATGGCAGTCATCCAGTCTTC; p21-F TGCATCCGTTTCACCCAACC, p21-R TCATTTTTCCAAAGTGCTATTCAGG; and p16-F CCCAACGCCCCGAACT, p16-R GCAGAAGAGCTGCTACGTGAA [15].
Protein Isolation and Western Blotting
For isolation of total cellular proteins, cells were washed twice with cold PBS and then RIPA lysis buffer (Sigma; R0278), containing protease inhibitor cocktail (Sigma; P2714-1BTL), was added onto the cell layer. Cells were kept on ice for 5 min, and then the lysate was scraped and collected into a micro centrifugation tube. Cell lysate was centrifuged at 12,000g for 15 min at 4 °C, and the supernatant was collected for further assays. Protein was quantified in the lysate by Bradford assay [21]. SDS-PAGE was carried out for protein samples using 70 μg of total protein, and then western blotting was performed for various following antibodies: Anti-Akt (#9272; 1:500), anti-phospho-Akt (#9271S; 1:500), anti-COX-2 (#12282; 1:500), anti-iNOS (#13120; 1:500), anti-mTOR (#2983; 1:500), anti-phospho-mTOR (#5536; 1:500), anti-NF-κB (#8242; 1:500), anti-phospho-NF-κB (#3031; 1:500), anti-phospho-ATM/ATR substrate (#2851; 1:500), anti-PI3K(#4257; 1:500), anti-phospho-PI3K (#4228; 1:500), anti-AMPKα (#2603; 1:500), and anti-phospho-AMPKα (#2535; 1:1000) were procured from cell signaling technologies, while anti-p21 (AHZ0422; 1:500), anti-p53 (13-4100; 1:1000), and anti-phospho-p38 (12-9078-41; 1:500) were procured from Thermo Fisher Scientific (USA). Anti-β-tubulin was obtained from Santa Cruz Biotechnology (USA) (sc-58882; 1:500).
Statistical Analyses
Data were analyzed using GraphPad Prism (Version 7) software. Experimental results are presented as mean ± standard error of the mean (SEM). Data were subjected to analysis of variance (ANOVA) and the Tukey test was used to separate the means (p < 0.05), which were considered statistically significant.
Results
Cell Viability Response to PCM
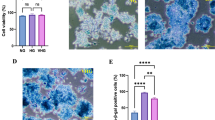
Analysis of PCM cytotoxicity to 3T3-L1 cells revealed no significant effect on cell viability even at concentration as high as 75% (v/v) (Fig. 1). Only the cells cultured in 100% (v/v) complete PCM showed significant (p < 0.05) cell death as compared to control suggesting that secretory metabolites of probiotic Lact. fermentum could be safely applied to cells (Fig. 1). Based on this, we chose the minimal dosage of PCM (5 and 10%, v/v) for further evaluation of its anti-senescence potential.
PCM Attenuates the Development of H2O2-Induced Senescent Cells
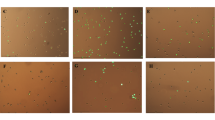
For inducing senescence, the concentration of H2O2 used in the present study was based on our previous observation [15]. Cells treated with H2O2 became irregular, flattened, and enlarged in shape as opposed to normal cells. The mean cell size showed a significant (p < 0.05) and stark increase in H2O2-treated cells as compared to normal cells, while PCM treated cells appeared to counter this effect, albeit in a statistically non-significant manner (Fig. 2). Further, as indicated by SA-β-gal activity, H2O2 application significantly (p < 0.05) enhanced the development of senescent cells, with over 65% cells appearing senescent as compared to control (Fig. 3). On the other hand, application of PCM appeared to counter the development of senescent cells, as a significant (p < 0.05) decrease in SA-β-gal active cells was observed as compared to H2O2 alone-treated cells (Fig. 3).
PCM treatment alleviates H2O2-induced morphological changes in preadipocytes. Premature senescence was induced in cells after treatment with H2O2 for 3 h (150 μM) and for three consecutive days in the presence or absence of PCM. a Control cells. b Cells treated with H2O2 alone. c, d Cells supplemented with 5 and 10% PCM respectively in addition to H2O2. e Median cell area. Values are mean ± SEM. Means that do not share a common letter indicate statistical difference at p < 0.05
PCM treatment ameliorates SA-β-gal activity in H2O2-treated preadipocytes. Premature senescence was induced in cells after treatment with H2O2 for 3 h (150 μM) and for three consecutive days in the presence or absence of PCM. a Control cells. b Cells treated with H2O2 alone. c, d Cells supplemented with 5 and 10% PCM respectively in addition to H2O2. e Percentage of cells positive for SA-β-gal activity. Values are mean ± SEM. Means that do not share a common letter indicate statistical difference at p < 0.05
PCM Downregulates the Expression of Cell Cycle Inhibitors Associated with Senescence
Abrogation of cell cycle progression due to overexpression of cell cycle inhibitors p53/p21WAF1/p16INK4a is an essential feature of senescent cells. It was observed that H2O2 treatment significantly (p < 0.05) enhanced the gene as well as protein expression of p53, which on the other hand, was significantly (p < 0.05) inhibited by the application of PCM at both the tested concentrations (Fig. 4a–d). Similarly, expression of p21WAF1 showed a striking and significant (p < 0.05) upregulation at both gene as well as protein levels in H2O2-treated cells which was also significantly (p < 0.05) attenuated by application of PCM (Fig. 4a, c, d). In addition, gene expression of p16INK4a also showed a significant (p < 0.05) upregulation in H2O2-treated cells as compared to control, which was significantly (p < 0.05) down-regulated on account of PCM treatment (Fig. 4a).
PCM treatment inhibits the accumulation of cell cycle inhibitors associated with H2O2-induced premature senescence. Cells were treated with H2O2 for 3 h (150 μM) and for three consecutive days in the presence or absence of PCM. a Relative gene expression of p53, p21, and p16. Relative protein expression of b p53, c p21, and d representative western blot images. Values are mean ± SEM. Significant difference in comparison to H2O2-treated group at *p < 0.05 and ***p < 0.01. Means that do not share a common letter indicate statistical difference at p < 0.05
PCM Treatment Protects Against H2O2-Induced Oxi-inflammatory Phenotype and DNA Damage
It was observed that treatment with H2O2 significantly (p < 0.05) interrupted cellular redox homeostasis, as a near 3-fold increase in intracellular ROS production was observed in H2O2-treated cells as compared to control (Fig. 5a). Application of PCM appeared to counter this effect, albeit in a non-significant manner. Further analysis revealed robust elevation (p < 0.05) in the protein expression of iNOS and cox-2 signifying the presence of acute oxidative stress in H2O2-treated cells (Fig. 5b–d). On the contrary, PCM treated cells, especially at 10% (v/v), showed a significant (p < 0.05) and robust inhibition in expression of both iNOS and cox-2 thereby suggesting lower levels of oxidative stress in PCM treated cells (Fig. 5b–d). Analysis of inflammatory marker p38MAPK revealed a strong (p < 0.05) upregulation of its phosphorylated form in H2O2-treated cells which was significantly (p < 0.05) abrogated in the presence of PCM (Fig. 6a, c). Corroborating this, phosphorylated NF-κB levels also showed a robust upregulation (p < 0.05) in H2O2-treated cells as compared to control, while a strong inhibition (p < 0.05) of NF-κB activation was observed in PCM treated cells suggesting inflammatory homeostasis on account of PCM treatment (Fig. 6b, c). The improved oxi-inflammatory cellular environment was further confirmed by the inhibition of phosphorylated substrates of ATM/ATR in PCM treated cells, indicating minimal DNA damage response as compared to H2O2-treated cells (Fig. 5d).
Application of PCM prevents oxidative damage associated with H2O2-induced premature senescence. Cells were treated with H2O2 for 3 h (150 μM) and for three consecutive days in the presence or absence of PCM. a Intracellular ROS production. Relative protein expression of b iNOS, c Cox2, and d representative western blot images. Values are mean ± SEM. Means that do not share a common letter indicate statistical difference at p < 0.05
PCM treatment attenuates the development of SASP. Cells were treated with H2O2 for 3 h (150 μM) and for three consecutive days in the presence or absence of PCM. Relative protein expression of a p-p38MAPK, b p-NF-κB/NF-κB ratio, and c representative western blot images. Values are mean ± SEM. Means that do not share a common letter indicate statistical difference at p < 0.05
PCM Treatment Inhibits H2O2-Induced PI3K/Akt/mTOR Signaling Pathway
Analysis of mTOR pathway revealed a robust upregulation on account of H2O2 treatment in senescent cells. The phosphorylated forms of PI3K, Akt, and mTOR were strongly elevated (p < 0.05) in H2O2-treated cells as compared to control (Fig. 7a–d). On the other hand, PCM treatment categorically and dose-dependently showed significant (p < 0.05) inhibition of the activation of PI3K/Akt/mTOR pathway suggesting the modulation of this nutrient sensing pathway for apparent anti-senescence effects of probiotic secretory metabolites (Fig. 7a–d).
Application of PCM inhibits H2O2-induced activation of mTOR pathway. Cells were treated with H2O2 for 3 h (150 μM) and for three consecutive days in the presence or absence of PCM. a p-PI3K/PI3K ratio. b p-Akt/Akt ratio. c p-mTOR/mTOR ratio. d Representative western blot images. Values are mean ± SEM. Means that do not share a common letter indicate statistical difference at p < 0.05
PCM Modulates Senescence-Induced Activation of AMPK Pathway
Analysis of protein levels of AMPK and p-AMPK revealed robust and significant (p < 0.05) upregulation of AMPK/p-AMPK ratio in H2O2-treated cells, suggesting imbalance in energetic homeostasis of senescent cells (Fig. 8a, b). On the other hand, presence of PCM appeared to alleviate AMPK activation as evidenced by a slight, yet non-significant inhibition of AMPK/p-AMPK ratio in PCM treated cells (Fig. 8a, b).
PCM treatment attenuates senescence-induced activation of AMPK pathway. Cells were treated with H2O2 for 3 h (150 μM) and for three consecutive days in the presence or absence of PCM. a p-AMPK/AMPK ratio. b Representative western blot images. Values are mean ± SEM. Means that do not share a common letter indicate statistical difference at p < 0.05
Discussion
Several lines of evidence indicate that probiotic bacteria play a critical role in regulating different aspects of human health including immune responses, suppression of tumorigenesis, hypercholesterolemia, diabetes, or intestinal putrefaction. However, there appears to be a distinct dearth of studies pertaining to understanding the impact of probiotic bacteria or their secretory metabolites in modulating cellular senescence per se. This is relevant since emerging evidences have shown that age-associated accumulation of senescent cells in tissues and organs is the causative link of organ dysfunction and inflammatory disorders during aging [1]. In the present study, we reveal that secretory metabolites of probiotic Lact. fermentum can modulate the development as well as severity of stress-induced senescence in preadipocytes by suppressing SASP and mTOR signaling.
Oxi-inflamm-aging theory proposes that gradual accumulation of ROS is a key mediator of the aging process [22]. Hydrogen peroxide is an important source of ROS within the cells, and at acute sub-lethal concentrations, H2O2 has been shown to induce senescence and features resembling those of replicative senescence [23]. Indeed, application of H2O2 in the present study resulted in enlarged and flattened cells with strong upregulation of SA-β-gal activity thereby indicating the onset of senescence program. PCM treatment to cells attenuated SA-β-gal activity and thus appeared to counter the development of senescent cells. Senescence is also associated with cell hypertrophy which occurs due to prevalent cell cycle arrest despite sustained cell growth. In the present study, PCM treatment could slightly, yet non-significantly, decrease enlarged cell size in preadipocytes suggesting that true reversal of morphological changes induced by senescence is elusive. We argue that increase in exposure as well as using higher PCM concentration could induce morphological changes in senescent preadipocytes or in their subsequent generations at par with control. Notwithstanding, further analysis clearly showed that PCM treatment effectively blocked the activation of p53/p21WAF1 cell cycle inhibitors indicating preserved proliferative capacity in PCM treated cells. Studies pertaining to assessment of anti-senescence attributes of probiotic bacteria or their metabolites are very limited. Working on Lactobacillus pentosus var. plantarum C29, research group of Jeong et al. [10, 24] has shown the suppression of senescence marker p16 in the colon and hippocampus of aged rats. The present study provides evidence that probiotic secretory metabolites can inhibit the onset of senescence in adipose tissue mediated by regulation of cell cycle inhibitors.
Application of acute H2O2 to cells results in rapid production of ROS which could activate oxidative damage and antioxidant machinery. It was observed that 3 days post H2O2 treatment, cells exhibited increased levels of intracellular ROS as well as protein expressions of iNOS and cox-2. This scenario is indicative of prevalent and overwhelming oxidative stress in H2O2-treated cells which could be implicated in the development of senescence program. Indeed, such type of stress would have rendered imminent DNA damage as confirmed by enhanced accumulation of ATM/ATR substrates. On the other hand, PCM appeared to counter these effects, as attenuated levels of ROS, iNOS, cox-2, and ATM/ATR substrate were observed in PCM treated cells. There are several documented evidences suggesting cytoprotective effects of probiotics mediated by modulation of antioxidative responses [25, 26]. A recent report by Finamore et al. [27] have shown that Lactobacillus casei Shirota can protect against 2,2′-azobis (2-amidinopropane) dihydrochloride-induced oxidative and inflammatory stress in enterocytes-like epithelial cells by inhibiting ROS generation and modulation of antioxidant enzymes and NF-κB expression. It has also been observed that cell-free supernatant of probiotic bacteria exhibits anti-inflammatory and antioxidant activity on human gut epithelial cells and macrophages stimulated with LPS [28]. The present study suggests that secretory metabolites of probiotic bacteria could also influence redox stimulation during stress-induced premature senescence. The prevalent ROS in senescent cells can trigger inflammatory pathways thereby aiding in the development of SASP which is a milieu of pro-inflammatory molecules and growth signals. SASP represents a primary driving force of senescence, as in wake of age-associated immune dysfunctions, its pro-inflammatory signals can induce oxi-inflammatory stress in neighboring cells that can accelerate senescence and tumorigenic environment [29]. Indeed, as observed in the present study, H2O2-treated senescent cells showed enhanced expression of phosphorylated p38MAPK and NF-κB, suggesting robust activation of pro-inflammatory pathways. NF-κB is considered as the master regulator of SASP, and its activation during senescence has been shown to promote the transcription of several SASP-related components [30,31,32], while its inhibition is associated with improved lifespan and health span during aging [33,34,35,36]. Therefore, it is evident that NF-κB inhibition is a potential molecular target for suppression of SASP as well as prevention of senescence. Similarly, the p38MAPK senses stress signals and is responsible for the downstream activation of transcription factors such as NF-κB. It has also been shown that p38MAPK is directly involved in the development of both replicative senescence and stress-induced premature senescence and further plays a critical role in regulating mTOR-mediated senescence [37,38,39,40]. In present study, it was observed that PCM treated cells showed striking downregulation in expression of both phosphorylated p38MAPK and NF-κB. Previously, Lactobacillus casei OLL2768 has been shown to attenuate ETEC-induced pro-inflammatory responses by inhibiting NF-κB and p38MAPK signaling pathways in bovine intestinal epithelial cells [41]. Similarly, Lactobacillus rhamnosus GG has been shown to inhibit p38MAPK activation in response to inflammatory cytokines in intestinal epithelial cells [42]. The present work indicates that secretory metabolites of probiotic bacteria can also attenuate stress-induced activation of oxidative and inflammatory pathways in cells resulting in suppression of SASP and associated cellular damage ultimately culminating in the inhibition of senescence program.
mTOR signaling pathway is emerging as a molecular target for enhancing longevity and curbing senescence. This pathway is essential for growth and development in early life, but as a case of antagonistic pleiotropy; its activation in later part of life is regarded as a primary driver of aging and related diseases [43]. Although in vivo experiments have shown that inhibition of Akt/mTOR signaling prolongs lifespan [13], however, causes and consequences of persistent mTOR signaling during senescence are still not clear [44]. It is speculated that dysregulated ROS production during aging considerably influences growth factor-independent activation of mTOR pathway [45]. Indeed, corroborating our previous work, the present study observed that H2O2-induced ROS dysregulation was accompanied by activation of the mTOR signaling pathway. Unabated ROS in cells can stimulate PI3K which results in activation of Akt and subsequently of mTOR. The activated Akt has been shown to directly enhance ROS production by increasing oxygen consumption thereby indicating a vicious circle of ROS generation and Akt activation that could sensitize cells to ROS-dependent premature senescence [46]. Further, the activated mTOR has been implicated in p53 protein translation and stabilization, as well as enhanced SA-β-gal activity [47], along with the development of SASP factors by enhancing NF-κB transcription [48]. Thus, it is reasonable to suggest that the transient H2O2 treatment to cells in present study would have instigated acute aggravation in ROS levels resulting in overwhelmed antioxidant defenses and subsequent activation of the mTOR pathway which would have directly contributed to the development of SASP as well as stabilization of p53-mediated cell cycle arrest (Fig. 9). On the other hand, a dose-dependent inhibition of PI3K, Akt, and mTOR activation was observed in PCM treated groups suggesting that suppression of mTOR pathway may have contributed to the apparent attenuation of p53/p21, SASP, and ROS-related pathways thereby mitigating senescence (Fig. 9). Previously, it has been shown that oral administration of Lactobacillus pentosus var. plantarum C29 to aged rats can inhibit mTOR activation in hippocampus and enhance memory thereby indicating its potency in ameliorating age-related degenerative dementia [24]. Another report has observed that culture supernatants Lactobacillus crispatus and Lactobacillus rhamnosus can differentially modulate mTOR and Wnt/ β-catenin pathways in different cancer cell lines [49]. In another study, it was observed that oral administration of Bacillus coagulans suppressed mTOR activation and phosphorylation of its downstream factors in a mouse model of food allergy to shrimp tropomyosin [50]. In addition, Lactobacillus casei extract has been shown to induce apoptosis in gastric cancer cells by inhibiting NF-κB expression as well as decreased phosphorylation of mTOR signaling components, such as PI3K, Akt, and p70S6 kinase [51]. However, to the best of our knowledge, the present study is the first to implicate that secretory metabolites of probiotic Lact. fermentum can suppress stress-associated induction of cell senescence mediated by inhibition of mTOR signaling thereby leading to abrogation of cellular senescence. Senescent cells are metabolically active but are characterized by an increased ADP/ATP and AMP/ATP ratios that result in energetic stress ultimately activating the AMPK pathway. In the presence of persistent cell stress, the AMP kinase has been shown to directly activate cell cycle inhibitors such as p53/p21 and thus help establish senescence. Considering the apparent impact of Lact. fermentum metabolites in mitigating senescence, we next analyzed their effect on AMPK activation. It was observed that the presence of Lact. fermentum metabolites aided in improving the cellular energetic homeostasis, as a minor suppression of AMPK activation was observed in Lact. fermentum treated cells.
Schematic diagram of the observed anti-senescence effects of PCM in the present study. Transient H2O2 treatment resulted in severe oxidative damage that stimulated DNA damage response and activated PI3K/Akt/mTOR pathway. The stimulated mTOR resulted in multiple effects including ROS generation, SASP development, and accumulation of cell cycle inhibitors thereby developing senescent phenotype. PCM treatment attenuated the activation of PI3K/Akt/mTOR pathway resulting in suppression of ROS, SASP, and cell cycle inhibitors thereby preventing senescence
Emerging evidences have suggested a link between gut dysbiosis and the development of inflammation, insulin resistance, and type 2 diabetes [52], and that some effects of the drug metformin may also be mediated by changes in the gut microbiota [53]. Further, the age-associated onset of obesity and co-morbidities in humans are associated with increase in adipocytokines and pro-inflammatory cytokines as well as induction of chronic oxidative stress. In fact, both these conditions were also observed in normal weight obese people as compared to non-obese people, suggesting that these are early hallmarks of metabolic changes [54]. Together, these observations indicate the crucial role of gut microbiome in the development and progression of obesity and type 2 diabetes which are often aggravated during aging. In this perspective, the present study suggests that application of Lact. fermentum can promote the preservation of preadipocyte cell functions during aging which may result in delay/inhibition of age-associated onset of inflammatory disorders such as type 2 diabetes. Metabolic pathways in human body are influenced by activities of both human genome and gut microbiome, and this crosstalk between metabolites has been shown to influence several aspects of human metabolism, enteric nervous system, immune responses, obesity, and cancer [55]. In response to environmental signals, the commensal bacteria in the gut interact with epithelial cells via secretory metabolites that have often shown health beneficial attributes. These metabolic products often include polyphosphates, peptides, lactic acid, tryptophan metabolites, histamine, and quorum sensing molecules [56]. Metabolites produced by probiotic bacteria in the intestinal lumen can pass the intestinal barrier and get absorbed into the circulation by passive or active mechanisms, thereby potentially influencing different cellular processes at sites distant to gut microenvironment [55, 57]. It is thus plausible that the apparent beneficial secretory factors of Lact. fermentum in present study may have modulated different cellular pathways, including the vitagenes network [58], thereby resulting in the mitigation of some of the deleterious aspects of senescence. As such, previously, cell-free extracts of probiotic bacteria have been identified for anti-obesity and adipogenesis modulatory attributes using in vitro model of 3T3-L1 preadipocytes [59, 60]. Further, it has been shown that senescence in preadipocytes not only impairs its functional attributes but also hampers neighboring non-senescent preadipocytes [61, 62], making this cell line a suitable model for senescence modulatory studies. Together, this clarifies our choice of using probiotic culture supernatant and 3T3-L1 cells for investigation of anti-senescence attributes in the present study.
Conclusions
It has been affirmed that probiotics should be explored either prophylactically or as biotherapeutics to manage symptoms associated with aging, immunosenescence, fatigue, and autism thereby providing a nutrient-mediated prospect of “healthy aging” and attenuating age-associated disorders [4, 63]. Observations in the present study indicate that although H2O2-induced cell hypertrophy is not significantly reversed by PCM treatment, yet several other parameters associated with cell cycle, oxi-inflammatory stress, and nutrient sensing pathways during the senescence program are effectively influenced by secretory probiotic metabolites. These results justify our hypothesis and provide a novel perspective of the beneficial effects of probiotics as anti-senescence agents. However, future research pertaining to assessment of probiotics for further anti-senescence attributes, such as senolytic potential, along with in vivo validation are required to fully comprehend the senescence modulatory characteristics of probiotics.
References
Bhatia-Dey N, Kanherkar RR, Stair SE, Makarev EO, Csoka AB (2016) Cellular senescence as the causal nexus of aging. Front Genet 7:13. https://doi.org/10.3389/fgene.2016.00013
Baker DJ, Childs BG, Durik M, Wijers ME, Sieben CJ, Zhong JA, Saltness R, Jeganathan KB, Verzosa GC, Pezeshki A, Khazaie K, Miller JD, van Deursen JM (2016) Naturally occurring p16Ink4a-positive cells shorten healthy lifespan. Nature 530(7589):184–189. https://doi.org/10.1038/nature16932
Davalli P, Mitic T, Caporali A, Lauriola A, D’Arca D (2016) ROS, cell senescence, and novel molecular mechanisms in aging and age-related diseases. Oxidative Med Cell Longev 2016:1–18. https://doi.org/10.1155/2016/3565127
Sharma R, Kapila R, Kapila S (2013) Probiotics as anti-immunosenescence agents. Food Rev Int 29(2):201–216. https://doi.org/10.1080/87559129.2012.751547
Markowiak P, Slizewska K (2017) Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients 9(9):1021. https://doi.org/10.3390/nu9091021
Kerry RG, Patra JK, Gouda S, Park Y, Shin H-S, Das G (2018) Benefaction of probiotics for human health: a review. J Food Drug Anal 26(3):927–939. https://doi.org/10.1016/j.jfda.2018.01.002
Ershler WB, Keller ET (2000) Age-associated increased interleukin-6 gene expression, late-life diseases, and frailty. Annu Rev Med 51(1):245–270. https://doi.org/10.1146/annurev.med.51.1.245
Malaguarnera G, Leggio F, Vacante M, Motta M, Giordano M, Biondi A, Basile F, Mastrojeni S, Mistretta A, Malaguarnera M, Toscano MA, Salmeri M (2011) Probiotics in the gastrointestinal diseases of the elderly. J Nutr Health Aging 16(4):402–410. https://doi.org/10.1007/s12603-011-0357-1
Sharma R, Kapila R, Kapasiya M, Saliganti V, Dass G, Kapila S (2014) Dietary supplementation of milk fermented with probiotic Lactobacillus fermentum enhances systemic immune response and antioxidant capacity in aging mice. Nutr Res 34(11):968–981. https://doi.org/10.1016/j.nutres.2014.09.006
Jeong J-J, Kim K-A, Jang S-E, Woo J-Y, Han MJ, Kim D-H (2015) Orally administrated Lactobacillus pentosus var. plantarum C29 ameliorates age-dependent colitis by inhibiting the nuclear factor-kappa B signaling pathway via the regulation of lipopolysaccharide production by gut microbiota. PLoS One 10(2):e0116533. https://doi.org/10.1371/journal.pone.0116533
Landete JM, Gaya P, Rodríguez E, Langa S, Peirotén Á, Medina M, Arqués JL (2017) Probiotic bacteria for healthier aging: immunomodulation and metabolism of phytoestrogens. Biomed Res Int 2017:1–10. https://doi.org/10.1155/2017/5939818
Blagosklonny MV (2008) Aging: ROS or TOR. Cell Cycle 7(21):3344–3354. https://doi.org/10.4161/cc.7.21.6965
Johnson SC, Rabinovitch PS, Kaeberlein M (2013) mTOR is a key modulator of ageing and age-related disease. Nature 493(7432):338–345. https://doi.org/10.1038/nature11861
Kennedy BK, Lamming DW (2016) The mechanistic target of rapamycin: the grand conducTOR of metabolism and aging. Cell Metab 23(6):990–1003. https://doi.org/10.1016/j.cmet.2016.05.009
Kumar R, Sharma A, Kumari A, Gulati A, Padwad Y, Sharma R (2018) Epigallocatechin gallate suppresses premature senescence of preadipocytes by inhibition of PI3K/Akt/mTOR pathway and induces senescent cell death by regulation of Bax/Bcl-2 pathway. Biogerontology 20:171–189. https://doi.org/10.1007/s10522-018-9785-1
Kumari M, Swarnkar MK, Kumar S, Singh AK, Gupta M (2015) Genome sequence of a potential probiotic strain, Lactobacillus fermentum HFB3, isolated from a human gut. Genome Announc 3(6). https://doi.org/10.1128/genomea.01296-15
Sharma R, Kumari M, Kumari A, Sharma A, Gulati A, Gupta M, Padwad Y (2019) Diet supplemented with phytochemical epigallocatechin gallate and probiotic Lactobacillus fermentum confers second generation synbiotic effects by modulating cellular immune responses and antioxidant capacity in aging mice. Eur J Nutr. https://doi.org/10.1007/s00394-018-01890-6
Nanjundaiah YS, Wright DA, Baydoun AR, O’Hare WT, Ali Z, Khaled Z, Sarker MH (2016) Lactobacillus rhamnosus GG conditioned media modulates acute reactive oxygen species and nitric oxide in J774 murine macrophages. Biochem Biophys Rep 6:68–75. https://doi.org/10.1016/j.bbrep.2016.03.003
Sharma A, Joshi R, Kumar S, Sharma R, Rajneesh, Padwad Y, Gupta M (2018) Prunus cerasoides fruit extract ameliorates inflammatory stress by modulation of iNOS pathway and Th1/Th2 immune homeostasis in activated murine macrophages and lymphocytes. Inflammopharmacology 26(6):1483–1495. https://doi.org/10.1007/s10787-018-0448-2
Sharma R, Sharma A, Kumari A, Kulurkar PM, Raj R, Gulati A, Padwad YS (2017) Consumption of green tea epigallocatechin-3-gallate enhances systemic immune response, antioxidative capacity and HPA axis functions in aged male swiss albino mice. Biogerontology 18(3):367–382. https://doi.org/10.1007/s10522-017-9696-6
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1006/abio.1976.9999
Fuente M, Miquel J (2009) An update of the oxidation-inflammation theory of aging: the involvement of the immune system in oxi-inflamm-aging. Curr Pharm Des 15(26):3003–3026. https://doi.org/10.2174/138161209789058110
Wang Z, Wei D, Xiao H (2013) Methods of cellular senescence induction using oxidative stress. In: Biological aging. Humana Press, Totowa, NJ, pp 135–144. https://doi.org/10.1007/978-1-62703-556-9_11
Jeong JJ, Woo JY, Kim KA, Han MJ, Kim DH (2015) Lactobacillus pentosus var. plantarum C29 ameliorates age-dependent memory impairment in Fischer 344 rats. Lett Appl Microbiol 60(4):307–314. https://doi.org/10.1111/lam.12393
Mishra V, Shah C, Mokashe N, Chavan R, Yadav H, Prajapati J (2015) Probiotics as potential antioxidants: a systematic review. J Agric Food Chem 63(14):3615–3626. https://doi.org/10.1021/jf506326t
Wang Y, Wu Y, Wang Y, Xu H, Mei X, Yu D, Wang Y, Li W (2017) Antioxidant properties of probiotic bacteria. Nutrients 9(5):521. https://doi.org/10.3390/nu9050521
Finamore A, Ambra R, Nobili F, Garaguso I, Raguzzini A, Serafini M (2018) Redox role of Lactobacillus casei Shirota against the cellular damage induced by 2,2′-Azobis (2-Amidinopropane) dihydrochloride-induced oxidative and inflammatory stress in enterocytes-like epithelial cells. Front Immunol 9:1131. https://doi.org/10.3389/fimmu.2018.01131
De Marco S, Sichetti M, Muradyan D, Piccioni M, Traina G, Pagiotti R, Pietrella D (2018) Probiotic cell-free supernatants exhibited anti-inflammatory and antioxidant activity on human gut epithelial cells and macrophages stimulated with LPS. Evid Based Complement Alternat Med 2018:1–12. https://doi.org/10.1155/2018/1756308
Borodkina AV, Deryabin PI, Giukova AA, Nikolsky NN (2018) “Social Life” of senescent cells: what is SASP and why study it? Acta Nat 10(1):4–14
Chien Y, Scuoppo C, Wang X, Fang X, Balgley B, Bolden JE, Premsrirut P, Luo W, Chicas A, Lee CS, Kogan SC, Lowe SW (2011) Control of the senescence-associated secretory phenotype by NF-κB promotes senescence and enhances chemosensitivity. Genes Dev 25(20):2125–2136. https://doi.org/10.1101/gad.17276711
Salminen A, Kauppinen A, Kaarniranta K (2012) Emerging role of NF-κB signaling in the induction of senescence-associated secretory phenotype (SASP). Cell Signal 24(4):835–845. https://doi.org/10.1016/j.cellsig.2011.12.006
Birch J, Passos JF (2017) Targeting the SASP to combat ageing: mitochondria as possible intracellular allies? Bioessays 39(5):1600235. https://doi.org/10.1002/bies.201600235
Osorio FG, Barcena C, Soria-Valles C, Ramsay AJ, de Carlos F, Cobo J, Fueyo A, Freije JMP, Lopez-Otin C (2012) Nuclear lamina defects cause ATM-dependent NF-κB activation and link accelerated aging to a systemic inflammatory response. Genes Dev 26(20):2311–2324. https://doi.org/10.1101/gad.197954.112
Jurk D, Wilson C, Passos JF, Oakley F, Correia-Melo C, Greaves L, Saretzki G, Fox C, Lawless C, Anderson R, Hewitt G, Pender SLF, Fullard N, Nelson G, Mann J, van de Sluis B, Mann DA, von Zglinicki T (2014) Chronic inflammation induces telomere dysfunction and accelerates ageing in mice. Nat Commun 5(1). https://doi.org/10.1038/ncomms5172
Laberge RM, Sun Y, Orjalo AV, Patil CK, Freund A, Zhou L, Curran Samuel C, Davalos AR, Wilson-Edell KA, Liu S, Limbad C, Demaria M, Li P, Hubbard GB, Ikeno Y, Javors M, Desprez P-Y, Benz CC, Kapahi P, Nelson PS, Campisi J (2015) MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation. Nat Cell Biol 17(8):1049–1061. https://doi.org/10.1038/ncb3195
Stout MB, Steyn FJ, Jurczak MJ, Camporez J-PG, Zhu Y, Hawse JR, Jurk D, Palmer AK, Xu M, Pirtskhalava T, Evans GL, de Souza SR, Frank AP, White TA, Monroe DG, Singh RJ, Casaclang-Verzosa G, Miller JD, Clegg DJ, LeBrasseur NK, von Zglinicki T, Shulman GI, Tchkonia T, Kirkland JL (2016) 17α-Estradiol alleviates age-related metabolic and inflammatory dysfunction in male mice without inducing feminization. J Gerontol A Biol Sci Med Sci 72(1):3–15. https://doi.org/10.1093/gerona/glv309
Dasari A, Bartholomew JN, Volonte D, Galbiati F (2006) Oxidative stress induces premature senescence by stimulating caveolin-1 gene transcription through p38 mitogen-activated protein kinase/Sp1-mediated activation of two GC-rich promoter elements. Cancer Res 66(22):10805–10814. https://doi.org/10.1158/0008-5472.can-06-1236
Harada G, Neng Q, Fujiki T, Katakura Y (2014) Molecular mechanisms for the p38-induced cellular senescence in normal human fibroblast. J Biochem 156(5):283–290. https://doi.org/10.1093/jb/mvu040
Xu Y, Li N, Xiang R, Sun P (2014) Emerging roles of the p38 MAPK and PI3K/AKT/mTOR pathways in oncogene-induced senescence. Trends Biochem Sci 39(6):268–276. https://doi.org/10.1016/j.tibs.2014.04.004
Borodkina AV, Shatrova AN, Nikolsky NN, Burova EB (2016) The role of p38 MAP-kinase in stress-induced senescence of human endometrium-derived mesenchymal stem cells. Cell Tissue Biol 10(5):365–371. https://doi.org/10.1134/s1990519x16050023
Takanashi N, Tomosada Y, Villena J, Murata K, Takahashi T, Chiba E, Tohno M, Shimazu T, Aso H, Suda Y, Ikegami S, Itoh H, Kawai Y, Saito T, Alvarez S, Kitazawa H (2013) Advanced application of bovine intestinal epithelial cell line for evaluating regulatory effect of lactobacilli against heat-killed enterotoxigenic Escherichia coli-mediated inflammation. BMC Microbiol 13(1):54. https://doi.org/10.1186/1471-2180-13-54
Yan F, Polk DB (2002) Probiotic bacterium prevents cytokine-induced apoptosis in intestinal epithelial cells. J Biol Chem 277(52):50959–50965. https://doi.org/10.1074/jbc.m207050200
Blagosklonny MV (2010) Revisiting the antagonistic pleiotropy theory of aging: TOR-driven program and quasi-program. Cell Cycle 9(16):3171–3176. https://doi.org/10.4161/cc.9.16.13120
Carroll B, Korolchuk VI (2017) Dysregulation of mTORC1/autophagy axis in senescence. Aging (Albany NY) 9(8):1851–1852. https://doi.org/10.18632/aging.101277
Nacarelli T, Azar A, Sell C (2015) Aberrant mTOR activation in senescence and aging: a mitochondrial stress response? Exp Gerontol 68:66–70. https://doi.org/10.1016/j.exger.2014.11.004
Nogueira V, Park Y, Chen C-C, Xu P-Z, Chen M-L, Tonic I, Unterman T, Hay N (2008) Akt determines replicative senescence and oxidative or oncogenic premature senescence and sensitizes cells to oxidative apoptosis. Cancer Cell 14(6):458–470. https://doi.org/10.1016/j.ccr.2008.11.003
Swami M (2008) Akt: a double-edged sword. Nat Rev Cancer 9(2):76–77. https://doi.org/10.1038/nrc2586
Bent EH, Gilbert LA, Hemann MT (2016) A senescence secretory switch mediated by PI3K/AKT/mTOR activation controls chemoprotective endothelial secretory responses. Genes Dev 30(16):1811–1821. https://doi.org/10.1101/gad.284851.116
Taherian-Esfahani Z, Abedin-Do A, Nouri Z, Mirfakhraie R, Ghafouri-Fard S, Motevaseli E (2016) Lactobacilli differentially modulate mTOR and Wnt/ β-catenin pathways in different cancer cell lines. Iran J Cancer Prev 9(3). https://doi.org/10.17795/ijcp-5369
Fu L, Peng J, Zhao S, Zhang Y, Su X, Wang Y (2017) Lactic acid bacteria-specific induction of CD4+Foxp3+T cells ameliorates shrimp tropomyosin-induced allergic response in mice via suppression of mTOR signaling. Sci Rep 7(1):1987. https://doi.org/10.1038/s41598-017-02260-8
Hwang JW, Baek Y-M, Yang KE, Yoo H-S, Cho C-K, Lee Y-W, Park J, Eom C-Y, Lee Z-W, Choi J-S, Jang I-S (2012) Lactobacillus casei extract induces apoptosis in gastric cancer by inhibiting NF-κB and mTOR-mediated signaling. Integr Cancer Ther 12(2):165–173. https://doi.org/10.1177/1534735412442380
Aw W, Fukuda S (2018) Understanding the role of the gut ecosystem in diabetes mellitus. J Diabetes Investig 9(1):5–12. https://doi.org/10.1111/jdi.12673
de la Cuesta-Zuluaga J, Mueller NT, Corrales-Agudelo V, Velásquez-Mejía EP, Carmona JA, Abad JM, Escobar JS (2017) Metformin is associated with higher relative abundance of mucin-degrading Akkermansia muciniphila and several short-chain fatty acid-producing microbiota in the gut. Diabetes Care 40(1):54–62. https://doi.org/10.2337/dc16-1324
Tomasello B, Malfa G, Galvano F, Renis M (2011) DNA damage in normal-weight obese syndrome measured by comet assay. Mediterr J Nutr Metab 4:99–104. https://doi.org/10.1007/s12349-010-0035-6
Sharon G, Garg N, Debelius J, Knight R, Dorrestein Pieter C, Mazmanian Sarkis K (2014) Specialized metabolites from the microbiome in health and disease. Cell Metab 20(5):719–730. https://doi.org/10.1016/j.cmet.2014.10.016
Wu R, Jeffrey M, Johnson-Henry K, Green-Johnson J, Sherman P (2016) Impact of prebiotics, probiotics and gut derived metabolites on host immunity. LymphoSign J 4(1):1–24. https://doi.org/10.14785/lymphosign-2016-0012
Hemarajata P, Versalovic J (2012) Effects of probiotics on gut microbiota: mechanisms of intestinal immunomodulation and neuromodulation. Ther Adv Gastroenterol 6(1):39–51. https://doi.org/10.1177/1756283x12459294
Calabrese V, Dattilo S, Petralia A, Parenti R, Pennisi M, Koverech G, Calabrese V, Graziano A, Monte I, Maiolino L, Ferreri T, Calabrese EJ (2015) Analytical approaches to the diagnosis and treatment of aging and aging-related disease: redox status and proteomics. Free Radic Res 49(5):511–524. https://doi.org/10.3109/10715762.2015
Park J-E, Oh S-H, Cha Y-S (2013) Lactobacillus plantarum LG42 isolated from gajami sik-hae inhibits adipogenesis in 3T3-L1 adipocyte. Biomed Res Int 2013:1–7. https://doi.org/10.1155/2013/460927
Lee E, Jung S-R, Lee S-Y, Lee N-K, Paik H-D, Lim S-I (2018) Lactobacillus plantarum strain ln4 attenuates diet-induced obesity, insulin resistance, and changes in hepatic mRNA levels associated with glucose and lipid metabolism. Nutrients 10(5):643. https://doi.org/10.3390/nu10050643
Zoico E, Di Francesco V, Olioso D, Fratta Pasini AM, Sepe A, Bosello O, Cinti S, Cominacini L, Zamboni M (2009) In vitro aging of 3T3-L1 mouse adipocytes leads to altered metabolism and response to inflammation. Biogerontology 11(1):111–122. https://doi.org/10.1007/s10522-009-9236-0
Trabucco Sally E, Zhang H (2016) Finding Shangri-La: limiting the impact of senescence on aging. Cell Stem Cell 18(3):305–306. https://doi.org/10.1016/j.stem.2016.02.002
Kaur IP, Kuhad A, Garg A, Chopra K (2009) Probiotics: delineation of prophylactic and therapeutic benefits. J Med Food 12(2):219–235. https://doi.org/10.1089/jmf.2007.0544
Funding
This work was supported by grants from Department of Science and Technology, Government of India, under the INSPIRE Faculty scheme (IFA17-LSPA79). The CSIR-IHBT publication number of this manuscript is 4350.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
This article does not contain any studies with human or animal subjects.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kumar, R., Sharma, A., Gupta, M. et al. Cell-Free Culture Supernatant of Probiotic Lactobacillus fermentum Protects Against H2O2-Induced Premature Senescence by Suppressing ROS-Akt-mTOR Axis in Murine Preadipocytes. Probiotics & Antimicro. Prot. 12, 563–576 (2020). https://doi.org/10.1007/s12602-019-09576-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-019-09576-z