Abstract
Objective
Sleep-disordered breathing (SDB) has been suggested to be associated with chronic kidney disease (CKD). Positive airway pressure (PAP) is an effective treatment for SDB, but the impact of PAP therapy on glomerular filtration rate (GFR) in patients with SDB remains unclear. The present meta-analysis was performed to determine whether PAP therapy could increase GFR.
Design
A systematic search of PubMed, Embase, Web of Science, and Cochrane library was performed for literature published up to January 2016. Standardized mean difference (SMD) was calculated to estimate the treatment effects of pre- and post-PAP therapy.
Results
A total of eight studies with 240 patients were pooled into a meta-analysis. The meta-analysis showed that there was no change of GFR before and after PAP treatment in SDB patients (SMD = 0.010, 95 % confidence interval (CI) = −0.331 to 0.350, z = 0.06, p = 0.956), Subgroup analyses indicated that GFR was significantly increased after PAP treatment in elder patients (≥55 years) (SMD = −0.283, 95 % CI = −0.518 to −0.047, z = 2.35, p = 0.019) and patients with therapeutic duration ≥ 3 months (SMD = −0.276, 95 % CI = −0.522 to −0.031, z = 2.20, p = 0.027).
Conclusion
The present meta-analysis suggested that PAP treatment had no impact on GFR in SDB patients. However, longer PAP usage for SDB patients significantly improved GFR. In elder SDB subjects, PAP was also associated with a statistically significant increase in GFR.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sleep-disordered breathing (SDB) is a common disorder characterized by repetitive apneas and hypopneas during sleep. SDB, including obstructive sleep apnea (OSA), and Cheyne–Stokes respiration (CSR) and central sleep apnea (CSA), is common in patients with chronic kidney disease (CKD) [1]. CKD describes patients with a chronically decreased glomerular filtration rate (GFR) or other evidence of kidney damage. The prevalence of OSA in CKD population has been reported to range from 27 to 54 % [1–3], which is considerably higher than the general population. The prevalence of CKD is up to 18 % in severe OSA patients without hypertension or diabetes [4]. The overlap of both conditions has been receiving increasing attention in the medical literature.
GFR is widely accepted as the best overall measure of kidney function [5]. The urinary or plasma clearance of an ideal filtration marker such as inulin or of alternative exogenous markers such as iothalamate, EDTA, diethylene triamine pentaacetic acid, and iohexol are used to determine GFR. Some equations based on serum creatinine, age, race, and sex are used to obtain an estimation of GFR (eGFR). Positive airway pressure (PAP) is the primary treatment of SDB including OSA, CSR, and CSA. Continuous positive airway pressure (CPAP) is the most common form to treat SDB. Other forms that provide noninvasive positive pressure ventilation include bi-level positive airway pressure (BiPAP), and adaptive servo-ventilation (ASV). Some studies demonstrated the favorable effect of PAP on GFR [6–8], while others did not [9, 10].
The purpose of the present meta-analysis was to quantitatively evaluate the efficacy of PAP ventilation on GFR in patients with SDB.
Methods
Search strategy
We looked for relevant articles in the following databases from inception to January 5, 2016: PubMed, Embase, Web of Science, and Cochrane library. The reference lists of selected articles were manually searched. The search terms used were sleep apnea or sleep-disordered breathing and positive airway pressure or adaptive servo ventilation or noninvasive ventilation combined with glomerular filtration rate or GFR. Two researchers independently identified the eligible studies.
Study selection
Studies were considered eligible if the following criteria were met: (1) The study populations were limited to adults (age > 18) with SDB (OSA, CSA, or CSR); (2) The intervention was an application of PAP (CPAP, BiPAP, or ASV); (3) Mean and SD (or SE) of GFR needed to be reported in before and after PAP therapy. Reviews, abstracts, case reports, editorials, non-human studies, and non-English studies were excluded. Unpublished studies for which we could not obtain data from the authors were also excluded.
Data extraction
The following information was abstracted and evaluated independently by two investigators: first author, publication year, country of the study, sample size, patient inclusion criteria, participant characteristics, study design, PAP type, mean daily PAP usage time, duration of PAP therapy, GFR measurement, and GFR values before and after PAP treatment.
Statistical analysis
Statistical analyses were performed by using STATA version 12.0. The pooled estimate of the standardized mean difference (SMD) and 95 % confidence interval (CI) were calculated. The fixed or random effects model was used for non-heterogeneous or heterogeneous data, respectively, as appropriate. Statistical heterogeneity among individual was assessed on the basis of Q and I 2 statistics. To explore the possible sources of heterogeneity in PAP treatment effects, sensitive analysis and subgroup analyses were conducted. Publication bias was assessed by Begg’s correlation and Egger’s regression.
Results
Searching results
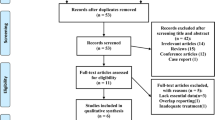
Our search yielded a total of 68 studies of potential interest after excluding duplicates. Of the 68 studies, 12 studies were obtained after careful review of the titles and abstracts. Of these, four studies were excluded. Two studies did not report data as mean and SD (or SE) [11, 12], while the other two studies in which the diagnostic criteria of SDB was not reported [13, 14]. Finally, eight studies were included in the meta-analysis. The detailed steps of the literature search were presented in Fig. 1.
Characteristics of the studies
A total of eight studies with 240 patients met the inclusion criteria and were included in this meta-analysis. All studies were observational. Patients with heart failure in three studies received ASV therapy [7, 8, 15], and patients in the remaining five studies received CPAP therapy [6, 9, 10, 16, 17]. The characteristics of the eight included studies and the patients’ characteristics were summarized in Tables 1 and 2, respectively.
Impact of PAP on GFR
Significant heterogeneity was detected among these studies (chi-squared = 23.69, p = 0.001; I 2 = 70.5 %). Consequently, a random-effect model was used for the pooled analysis. Pooling the data with a random-effect model meta-analysis showed that PAP therapy had no impact on GFR in SDB patients (SMD = 0.010, 95 % confidence interval (CI) = −0.331 to 0.350, z = 0.06, p = 0.956) (Fig. 2). A similar result (SMD = −0.055, 95 % CI = −0.237 to 0.126, z = 0.60, p = 0.549) was observed from a fixed-effect model.
Sensitivity and subgroup analyses
In addition, the sensitivity analysis showed that the overall risk estimates were stable (Fig. 3). Since the effectiveness of PAP could be influenced by many factors, subgroup analyses were performed (Table 3). The results showed that GFR was significantly increased after PAP treatment in elder patients (≥55 years) (SMD = −0.283, 95 % CI = −0.518 to −0.047, z = 2.35, p = 0.019) and patients with therapeutic duration ≥3 months (SMD = −0.276, 95 % CI = −0.522 to −0.031, z = 2.20, p = 0.027). While the differences in body mass index (BMI), apnea-hypopnea index (AHI), and sample size hardly influenced effectiveness of PAP. ASV therapy might have a trend in increase of GFR in comparison with pre-ASV treatment (SMD = −0.250, 95 % CI = −0.512 to 0.012, z = 1.87, p = 0.061).
Publication bias
There was no statistical significance of publication bias in the present meta-analysis (Begg’s test, p = 0.174; Egger’s test, p = 0.108) (Fig. 4). In addition, the trim-and-fill method showed that no study needed to be statistically corrected for funnel plot asymmetry.
Discussion
The present meta-analysis, comprised of 240 patients, suggested that PAP had no impact on GFR in SDB patients. However, subgroup analyses indicated that PAP was associated with a statistically significant increase on GFR in elder SDB patients (≥55 years) or patients with therapeutic duration ≥3 months. ASV therapy might have a trend in increase of GFR in SDB patients compared with baseline.
The impact of SDB on the development of CKD has been demonstrated by accumulating evidence [4, 18–20]. A retrospective cohort study including 4674 adult sleep apnea (SA) patients and 23,370 non-SA patients as the comparison group found that patients with SA experienced a 1.94-fold and 2.2-fold increase in the incidence of CKD and ESRD, respectively, after adjustment for sex, age, and comorbid medical conditions [18]. A cross-sectional study that enrolled 40 severe OSA patients without diabetes or hypertension showed that the prevalence of CKD in severe OSA subjects was 18 %. Further stepwise multivariate linear regression analysis revealed that AHI and desaturation index were the only independent predictor of urine albumin-to-creatinine ratio and eGFR [4]. Sakaguchi et al. [20] followed 161 patients with stage 3 to 4 CKD for over 1 year and found that the eGFR declined three- to fourfold faster in patients with moderate-to-severe nocturnal hypoxemia than patients with no or mild nocturnal hypoxemia (the mean values were −2.14, −3.02, and −8.59 ml/min/1.73 m2 per year in the no, mild, and moderate-to-severe nocturnal hypoxemia groups, respectively). After controlling for various baseline confounding factors, nocturnal hypoxemia remained a significant predictor of decline in eGFR. The pathogenesis of CKD in SDB is likely to be a multifactorial process involving several mechanisms: direct effects of hypoxia on the kidney [21, 22], or oxidative stress, endothelial dysfunction, inflammatory cytokine levels, sympathetic nervous system activity, and systemic blood pressure dysfunction [23, 24], all of which have been proposed to reduce kidney function.
PAP, as first choice for SDB treatment, has been reported to reverse SDB-associated hypoxia, sympathetic activation [25], inflammatory cytokine levels [26], blood pressure [27], and ameliorate glomerular hyperfiltration [16], which contributed to CKD in SDB. Our meta-analysis indicated that PAP treatment appeared to have no impact on GFR in SDB patients. As effectiveness of PAP treatment was influenced by multiple factors, we performed subgroup analyses in terms of age, baseline BMI, baseline AHI, treatment duration, sample size, and PAP type. The results of subgroup analyses supported that as little as 3 months of PAP was required to have a change on GFR. Besides treatment duration, our results suggested that GFR in elder patients might respond better to PAP therapy.
Our meta-analysis has several strengths. Firstly, this was the first meta-analysis that examined the effect of PAP on GFR in SDB patients. Secondly, pooling of information from all eligible studies yielded more precise and reliable results than that of individual studies. Thirdly, most of the included studies had an effective PAP use of 4 h/night or more, suggesting a good PAP compliance. Lastly, Begg’s tests and Egger’s tests showed there was no evidence to support publication bias in our study.
Nevertheless, the meta-analysis also has several limitations. Firstly, the sample sizes of the included studies were small. Secondly, all of the included studies were self-control, not RCT. Thirdly, in our meta-analysis, different studies utilized a variety of measurement techniques for GFR. In addition, the use of only English language papers only may cause some publication biases.
In conclusion, the present meta-analysis suggested that PAP treatment had no impact on GFR in SDB patients. However, longer PAP usage for SDB patients significantly improved GFR. In elder SDB subjects, PAP was also associated with a statistically significant increase in GFR.
References
Markou N, Kanakaki M, Myrianthefs P, Hadjiyanakos D, Vlassopoulos D, Damianos A, Siamopoulos K, Vasiliou M, Konstantopoulos S (2006) Sleep-disordered breathing in nondialyzed patients with chronic renal failure. Lung 184:43–49
Canales MT, Lui LY, Taylor BC, Ishani A, Mehra R, Stone KL, Redline S, Ensrud KE (2008) Renal function and sleep-disordered breathing in older men. Nephrol Dial Transplant: Off Publ Eur Dial Transplant Assoc Eur Ren Assoc 23:3908–3914
Nicholl DD, Ahmed SB, Loewen AH, Hemmelgarn BR, Sola DY, Beecroft JM, Turin TC, Hanly PJ (2012) Declining kidney function increases the prevalence of sleep apnea and nocturnal hypoxia. Chest 141:1422–1430
Chou YT, Lee PH, Yang CT, Lin CL, Veasey S, Chuang LP, Lin SW, Lin YS, Chen NH (2011) Obstructive sleep apnea: a stand-alone risk factor for chronic kidney disease. Nephrol Dial Transplant: Off Publ Eur Dial Transplant Assoc Eur Ren Assoc 26:2244–2250
Stevens LA, Coresh J, Greene T, Levey AS (2006) Assessing kidney function—measured and estimated glomerular filtration rate. N Engl J Med 354:2473–2483
Koga S, Ikeda S, Yasunaga T, Nakata T, Maemura K (2013) Effects of nasal continuous positive airway pressure on the glomerular filtration rate in patients with obstructive sleep apnea syndrome. Intern Med 52:345–349
Yoshihisa A, Suzuki S, Owada T, Iwaya S, Yamauchi H, Miyata M, Yamaki T, Sugimoto K, Kunii H, Nakazato K, Suzuki H, Saitoh S, Takeishi Y (2013) Short-term use of adaptive servo ventilation improves renal function in heart failure patients with sleep-disordered breathing. Heart Vessel 28:728–734
Koyama T, Watanabe H, Terada S, Makabe S, Igarashi G, Nobori K, Ito H (2011) Adaptive servo-ventilation improves renal function in patients with heart failure. Respir Med 105:1946–1953
Zhang L, Huang X, Li X, Wu Q (1997) Alterations in renal function in patients with obstructive sleep apnea syndrome and effects of continuous positive airway pressure. Chin Med J 110:915–918
Nicholl DD, Hanly PJ, Poulin MJ, Handley GB, Hemmelgarn BR, Sola DY, Ahmed SB (2014) Evaluation of continuous positive airway pressure therapy on renin-angiotensin system activity in obstructive sleep apnea. Am J Respir Crit Care Med 190:572–580
Puckrin R, Iqbal S, Zidulka A, Vasilevsky M, Barre P (2015) Renoprotective effects of continuous positive airway pressure in chronic kidney disease patients with sleep apnea. Int Urol Nephrol 47:1839–1845
Yasar ZA, Ucar ZZ, Demir AU, Kirakli C, Kalenci D, Tibet G (2014) Does CPAP therapy alter urinary albumin level in adult patients with moderate to severe obstructive sleep apnea syndrome? Sleep Breath Schlaf Atmung 18:525–532
Krieger J, Imbs JL, Schmidt M, Kurtz D (1988) Renal function in patients with obstructive sleep apnea effects of nasal continuous positive airway pressure. Arch Intern Med 148:1337–1340
Krieger J, Schmidt M, Sforza E, Lehr L, Imbs JL, Coumaros G, Kurtz D (1989) Urinary excretion of guanosine 3′:5′-cyclic monophosphate during sleep in obstructive sleep apnoea patients with and without nasal continuous positive airway pressure treatment. Clin Sci (Lond) 76:31–37
Owada T, Yoshihisa A, Yamauchi H, Iwaya S, Suzuki S, Yamaki T, Sugimoto K, Kunii H, Nakazato K, Suzuki H, Saitoh S, Takeishi Y (2013) Adaptive servoventilation improves cardiorenal function and prognosis in heart failure patients with chronic kidney disease and sleep-disordered breathing. J Card Fail 19:225–232
Kinebuchi S, Kazama JJ, Satoh M, Sakai K, Nakayama H, Yoshizawa H, Narita I, Suzuki E, Gejyo F (2004) Short-term use of continuous positive airway pressure ameliorates glomerular hyperfiltration in patients with obstructive sleep apnoea syndrome. Clin Sci (Lond) 107:317–322
Zhang XB, Jiang XT, Lin QC, Chen X, Zeng HQ (2014) Effect of continuous positive airway pressure on serum cystatin C among obstructive sleep apnea syndrome patients. Int Urol Nephrol 46:1997–2002
Lee YC, Hung SY, Wang HK, Lin CW, Wang HH, Chen SW, Chang MY, Ho LC, Chen YT, Liou HH, Tsai TC, Tseng SH, Wang WM, Lin SH, Chiou YY (2015) Sleep apnea and the risk of chronic kidney disease: a nationwide population-based cohort study. Sleep 38:213–221
Sim JJ, Rasgon SA, Kujubu DA, Kumar VA, Liu IL, Shi JM, Pham TT, Derose SF (2009) Sleep apnea in early and advanced chronic kidney disease: Kaiser Permanente Southern California cohort. Chest 135:710–716
Sakaguchi Y, Hatta T, Hayashi T, Shoji T, Suzuki A, Tomida K, Okada N, Rakugi H, Isaka Y, Tsubakihara Y (2013) Association of nocturnal hypoxemia with progression of CKD. Clin J Am Soc Nephrol: CJASN 8:1502–1507
Fine LG, Orphanides C, Norman JT (1998) Progressive renal disease: the chronic hypoxia hypothesis. Kidney Int Suppl 65:S74–S78
Fine LG, Norman JT (2008) Chronic hypoxia as a mechanism of progression of chronic kidney diseases: from hypothesis to novel therapeutics. Kidney Int 74:867–872
Adeseun GA, Rosas SE (2010) The impact of obstructive sleep apnea on chronic kidney disease. Curr Hypertens Rep 12:378–383
Hanly PJ, Ahmed SB (2014) Sleep apnea and the kidney: is sleep apnea a risk factor for chronic kidney disease? Chest 146:1114–1122
Shimizu K, Chin K, Nakamura T, Masuzaki H, Ogawa Y, Hosokawa R, Niimi A, Hattori N, Nohara R, Sasayama S, Nakao K, Mishima M, Ohi M (2002) Plasma leptin levels and cardiac sympathetic function in patients with obstructive sleep apnoea-hypopnoea syndrome. Thorax 57:429–434
Xie X, Pan L, Ren D, Du C, Guo Y (2013) Effects of continuous positive airway pressure therapy on systemic inflammation in obstructive sleep apnea: a meta-analysis. Sleep Med 14:1139–1150
Fava C, Dorigoni S, Dalle Vedove F, Danese E, Montagnana M, Guidi GC, Narkiewicz K, Minuz P (2014) Effect of CPAP on blood pressure in patients with OSA/hypopnea a systematic review and meta-analysis. Chest 145:762–771
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants performed by any of the authors.
Funding
The Fujian Provincial Health Bureau provided financial support in the form of Youth Research Fund (grant 2015–1-98). The sponsor had no role in the design or conduct of this research.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Chen, LD., Lin, L., Ou, YW. et al. Effect of positive airway pressure on glomerular filtration rate in patients with sleep-disordered breathing: a meta-analysis. Sleep Breath 21, 53–59 (2017). https://doi.org/10.1007/s11325-016-1364-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-016-1364-6