Abstract
Organ-specific adult stem cells are essential for organ homeostasis, tissue repair and regeneration. The formation of such stem cells often takes place during postembryonic development, a period around birth in mammals when plasma thyroid hormone concentration is high. The life-long self-renewal of the intestinal epithelium has made mammalian intestine a valuable model to study the function and regulation and adult stem cells. On the other hand, much less is known about how the adult intestinal stem cells are formed during vertebrate development. Here, we will review some recent progresses on this subject, focusing mainly on the formation of the adult intestine during Xenopus metamorphosis. We will discuss the role of thyroid hormone signaling pathway in the process and potential molecular conservations between amphibians and mammals as well as the implications in organ homeostasis and human diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Adult organ-specific stem cells are critical for organ-homeostasis, tissue-repair and regeneration. The adult mammalian intestine, especially the mouse intestine, has been a valuable model system to study adult organ-specific stem cells largely because of their constant self-renewal of the intestinal epithelium throughout adult life [1–6]. In the intestine, the stem cells residing at the bottom of the crypts, proliferate and their daughter cells differentiate into different epithelial cell types as they migrate along the crypt-villus axis, and eventually undergo apoptosis at the tip of the villus, thus completing the self-renewing cycle once every 1-6 days in adult mammals [2, 7, 8]. Similar processes occur in the intestine in all vertebrates, including amphibians, with self-renewing once every 2 weeks in Xenopus laevis [9]. Such interesting properties together with the development of a number of technologies, such as transgenesis and knockout as well as in vitro cultures, have enabled extensive studies that have revealed important mechanistic insights on the function and properties of the adult stem cells, including many molecular pathways governing stem cells [2, 10]. On the other hand, much less is known about when and how such adult intestinal stem cells are formed during vertebrate development, largely due to the difficulty to manipulate uterus-enclosed mammalian embryos.
Early studies suggests that in mouse, the formation of adult intestinal stem cells takes place shortly after birth when plasma thyroid hormone (T3) level high [3, 11–13], suggesting that T3 plays an important role in the formation of adult intestinal stem cells. Furthermore, TR deficiency leads to defects of the intestinal development, underlining the importance of this hormone. It is, however, difficult to study the role of T3 on intestinal maturation in mammals because of the dependence of the mammalian embryos or even neonates on the maternal supply of nutrients, making it difficult to separate the direct vs. indirect effects of T3 on the embryos or neonates. Interestingly, this postembryonic developmental period in mammals resembles anuran metamorphosis in many aspects [14, 15]. Importantly, amphibian metamorphosis offers a number of advantages to study T3 action in vertebrate development. First, its total dependence on T3 makes it easy to manipulate this process in both intact animals and organ/primary cell cultures [14–16]. Second, extensive earlier studies have provided detailed biochemical, morphological, cytological, and molecular information on the metamorphic transformation of different organs/tissues [14–16]. Finally, the development of transgenic, gene knockout and knockin technologies have made it possible to carry out genetic studies in amphibians, especially the widely used, highly related species Xenopus laevis and tropicalis [17–26].
Intestinal remodeling during amphibian metamorphosis bears many similarities to the maturation of mammalian intestine during the neonatal period, also referred to as the postembryonic development [15]. In the Xenopus laevis and tropicalis, the tadpole intestine is a simple tubular structure made of mainly larval epithelial cells with little connective tissue or muscles, except in the single epithelial fold, the typhlosole, where connective tissue is abundant (Fig. 1) [27, 28]. During metamorphosis, the tadpole epithelium degenerates with the vast majority of the cells undergoing apoptosis. Some larval epithelial cells, however, dedifferentiate into highly proliferative cells that express well-known markers of adult mammalian intestinal stem cells, such as leucine-rich repeat-containing G-protein coupled receptor 5 (Lgr5) and Musashi-1 (Msi-1) [29–31]. These adult stem cells subsequently proliferate and differentiate to form the adult epithelium. Concurrently, the connective tissue and muscles also develop extensively. The resulting frog intestine contains numerous epithelial folds that resemble the crypt-villus structure in mammals and are surrounded by thick layers of connective tissue and muscles [1, 27, 28, 32, 33]. As in adult mammals, throughout adult frog life, the stem cells localized in the trough of the fold proliferate and the daughter cells differentiate into different epithelial cells as they migrate up toward the crest of the fold, where they undergo apoptosis [27].
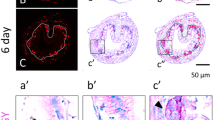
Intestinal metamorphosis involves the formation of clusters of proliferating, undifferentiated epithelial cells at the climax. Tadpoles at premetamorphic stage 54 (a), climax (b, stage 62), and end of metamorphosis (c, stage 66) were injected with EdU one hour before being sacrificed. Cross-sections of the intestine from the resulting tadpoles were double-stained for EdU (5-Ethynyl-2’-deoxyuridine, labeling newly synthesized DNA) and IFABP (intestinal fatty acid binding protein, a marker for differentiated epithelial cells) by immunohistochemistry. The dotted lines depict the epithelium-mesenchyme boundary. Note that the EdU-labeled proliferating cells in the epithelium were few and expressed IFABP at premetamorphosis (a) and increased in form of clustered cells (proliferating adult stem cells) that lacked IFABP at the climax of metamorphosis (b). At the end of metamorphosis, EdU-labeled proliferating cells were localized mainly in the troughs of the epithelial folds where IFABP expression was low (c). ep, epithelium. ct, connective tissue. m, muscles. l, lumen. See [29] for more details
2 T3 regulation of adult intestinal stem cell development during Xenopus metamorphosis
Like during mammalian postembryonic development, T3 levels peak during amphibian metamorphosis with little T3 present in premetamorphic tadpoles [14, 34]. More importantly, blocking the synthesis of endogenous T3 prevents metamorphosis while addition of physiological levels of T3 to the rearing water of premetamorphic tadpoles or even organ cultures derived from premetamorphic tadpoles induces precocious metamorphosis, indicating that T3 plays a causative role on amphibian metamorphosis [14, 34]. Thus, T3-treatment of premetamorphic tadpoles leads to precocious remodeling of the intestine, including the formation of adult intestinal stem cells [27]. Importantly, this process is organ-autonomous as T3 can even induce the formation of adult intestinal stem cells as well as the adult intestinal epithelium when the intestinal organ cultures from premetamorphic tadpoles are treated with physiological levels of T3 [1, 35–38]. Making use of the ability to generate transgenic animals expressing GPF and carrying out recombinant intestinal organ cultures, we have demonstrated that adult epithelial stem cells induced by T3 treatment have their origin in the larval epithelium [32]. Since there has been no evidence for the existence of epithelial stem cells in the larval epithelium [27, 39], these findings suggest that T3 induces some larval cells to develop into adult intestinal stem cells.
3 Mechanism of gene regulation by TR and an essential role of TR in Xenopus development and adult stem cell formation
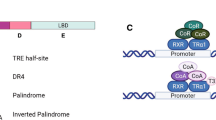
T3 has both genomic and non-genomic effects. The non-genomic effects are mediated by cell surface and cytoplasmic binding proteins, including TRs, although their role in vertebrate development, if any, is unknown [40–45]. At the genomic level, T3 regulates gene transcription through T3 receptors or TRs [14, 40–42, 44, 46]. TRs can both activate and repress gene transcription. For genes that are induced by T3, TR mainly functions as heterodimers formed with 9-cis retinoic acid receptors (RXRs), members of the nuclear hormone receptor superfamily that also include TRs [40, 42, 44, 46, 47]. TR/RXR heterodimers bind to T3-response elements (TREs) in target genes constitutively to regulate target gene expression in a T3-dependent manner [40, 42, 44, 47–50]. In the absence of T3, TR binds to histone deacetylase-containing corepressor complexes to repress transcription [51–67]. When T3 is available, TR binds coactivator complexes, such as those containing histone acetyltransferases SRC (steroid receptor coactivator) 1-3 and histone methyltransferase PRMT1 (protein arginine methyltransferase 1), to facilitate epigenetic modification and gene transcription [42, 56, 68–89]. Molecular studies such as chromatin immunoprecipitation (ChIP) assays have shown that TR and RXR are indeed bound to T3-inducible genes constitutively in pre- and metamorphosing Xenopus laevis and tropicalis tadpoles and recruits corepressor and coactivator complexes in a T3 dependent manner in vivo [67, 81, 86–94]. This leads to corresponding changes in local chromatin structure and histone modifications, accompanying changes in gene transcription [93–98].
To study the role of TRs in Xenopus development, we and others first used transgenic approach to overexpress mutant receptors to show that TR is both necessary and sufficient to mediate the metamorphic effects of T3 [45, 98–112]. To specifically determine the involvement of TR in the formation of adult intestinal stem cells, we generated recombinant organ-cultures of the isolated intestinal epithelium and the non-epithelium (the rest of the intestine) from wild type and transgenic animals expressing a dominant positive TR (dpTR) under the control of a heat shock-inducible promoter [102, 113]. This dpTR functions like constitutively liganded TR except that it does not bind to T3. We observed that when dpTR was induced to express in all tissues of the intestine, intestinal metamorphosis, including larval epithelial cell death and adult stem cell formation, took place even in the absence of T3 [113]. More importantly, we showed that expression of dpTR in the larval epithelium alone is able to induce the dedifferentiation of larval epithelial cells to upregulate sonic hedgehog gene, which is highly expressed in the proliferating adult epithelial progenitor/stem cells. Interestingly, such cells fail to upregulate the expression of well-known adult stem cell markers, such as Msi-1, and the formation of the stem cells expressing such markers also requires the expression of dpTR in the rest of the intestinal tissues (the non-epithelium) in the recombinant organ cultures (Fig. 2) [113]. Theses findings indicate that TR is necessary and sufficient for T3-induction of stem cell formation and that this process requires T3 action in both epithelium and non-epithelium, with the latter likely contribute to the formation of the so-called stem cell niche [35, 107, 113, 114]. They further suggest that T3-regulated genes in both the epithelium and non-epithelium are required for stem cell development. Many such tissue-specific T3-regulated genes have been identified and the analyses of the spatiotemporal expression profiles of some of the epithelial genes indeed support their involvement in adult stem cell formation/proliferation [115–120].
Top. Recombinant intestinal organ culture studies using dpTR-expressing transgenic (Tg) and wild type (Wt) tadpoles indicate that only when both the epithelium (Ep) and non-Ep (the rest of the intestine) are derived from Tg animals, i.e., Tg/Tg, are true stem cells formed. Recombinants made of Tg Ep and Tg non-Ep (Tg/Tg) and Tg Ep and Wt non-EP (Tg/Wt) of premetamorphic intestines were cultured with heat shock treatment for 5 days in vitro. Cross sections were double-immuno-stained with anti-Shh (green, sonic hedgehog, an adult stem cell precursor marker) and anti-CK19 (red, cytokeratin-19, which is expressed in epithelial cells), or anti-Msi-1 (green, Musashi-1, a stem cell marker of the vertebrate adult intestine) and anti-CK19 (red), or anti-Akt (green, a stem cell marker of the vertebrate adult intestine) and anti-CK19 (red) antibodies. In both Tg/Tg (arrowheads) and Tg/Wt intestines, cells positive for Shh and CK19 become detectable among cells expressing, indicating the Shh positive cells can be induced by cell-autonomous action of activated TR in the epithelium. Cells positive for Msi1 and Akt are also detected among CK19- immunoreactive cells in Tg/Tg intestine (arrowheads) but not in Tg/Wt intestine. Thus, activation of TR in the non-epithelial tissues is also required for the stem cell formation. Not shown here is that most of the epithelial cells undergo apoptosis when dpTR is expressed in either the EP or non-EP or both, just like that during metamorphosis when T3 binds to TR. See [113] for details. Bottom: A model for T3 actions during stem cell development in Xenopus laevis intestine. During metamorphosis, T3 acts directly (1) on the larval epithelium as well as (2) on the rest of the intestine (the non-epithelium), mostly the connective tissue. The vast majority of the larval epithelial cells are induced to undergo programmed cell death but a small number of the larval cells within the larval epithelium undergo dedifferentiation upon receiving the T3 signal (1) to dedifferentiate into Shh positive precursor cells. However, T3 action in the non-epithelium (2) is required for these cells to develop into stem cells expressing Msi1 and Akt, with the T3 action in the non-epithelium likely contributes to the establishment of the stem cell niche
4 A role of PRMT1 in thyroid hormone-dependent intestinal stem cell development in Xenopus
Among the T3-regulated genes during intestinal metamorphosis is PRMT1, which has been shown to function as a TR coactivator [79]. PRMT1 binds to SRC1-3 and is capable of methylating histone H4 arginine 3 (H4R3) [121]. Consistently, during intestinal remodeling, PRMT1 is recruited by TR to endogenous target genes and transgenic overexpression of PRMT1 enhances TR target gene expression and accelerates metamorphosis in Xenopus laevis [81].
More importantly, PRMT1 is highly upregulated specifically in the developing/proliferating adult intestinal stem cells during metamorphosis and that its upregulation is one of the earliest events during the dedifferentiation of the larval epithelial cells in their transformation into adult stem cells (Fig. 3), suggesting a role of PRMT1 in this process. In support of this, heat shock treatment of transgenic tadpoles which had transgenic wild type PRMT1 under the control of a heat shock-inducible promoter resulted in an increased number of intestinal stem cells during metamorphosis and knockdown the endogenous PRMT1 with antisense morpholino oligonucleotide reduced the number of such stem cells [13]. It is likely that PRMT1 affects the formation and/or proliferation of adult intestinal stem cells during metamorphosis by enhancing T3 signaling. In addition, PRMT1 may also act as a coactivator for other transcription factors to affect gene expression or influence the function of other proteins through methylation during stem cell development and proliferation.
PRMT1 has conserved spatiotemporal expression patterns during postembryonic intestinal development in Xenopus laevis, fish, and mouse. PRMT1 mRNA was analyzed by in situ hybridization in the intestines at three different developmental stages: larval/neonatal when plasma T3 level is low; larval to adult transition when T3 level is high, and end of the transition when T3 is low, in three different animal species. The stages were stage 54 (premetamorphosis), stage 62 (metamorphic climax), and stage 66 (end of metamorphosis for Xenopus laevis; 15 days post fertilization (dpf), 20 dpf and 1 year post fertilization (ypf) for zebrafish; and postnatal day 7 (P7), P21, and postnatal week 14 (P14w) for mouse, respectively. Arrows indicate PRMT1 positive cells in the intestinal epithelium of zebrafish and mouse. Note that there was little PRMT1 expression in the larval/neonatal stage prior to the transformation in all three species. As T3 level rose during intestinal transformation to the adult type, high levels of PRMT1 expression was detected only in the proliferating/stem cells located in the crypts in both mouse and zebrafish and in the clusters of cells in the epithelium at climax of metamorphosis in Xenopus laevis. ep, epithelium. ct, connective tissue. m, muscles. l, lumen. Bars, 50 μm for zebrafish and mouse, 100 μm for Xenopus. See [13] for details
5 Conservation of T3-dependent intestinal stem cell development in vertebrates
As indicated in the introduction, the maturation of the intestine occurs around the time when plasma T3 levels are high in other vertebrates such as mammals, a period resembling amphibian metamorphosis. Furthermore, T3 or TR deficiency in mouse leads to abnormal intestinal morphology, a decrease in the number of epithelial cells along the crypt-villus axis and in proliferating crypt cells [122–125]. It has also been shown that TRα1 controls intestinal development during maturation at weaning as well as intestinal homeostasis in adulthood by activating the proliferation of intestinal progenitors in the crypt [126]. Thus, T3 and TR may have conserved roles in regulating the formation of vertebrate adult intestinal stem cells. Studies on PRMT1 expression during mouse and zebrafish development support this conservation. Little PRMT1 or no expression is present in the larval/neonatal intestine in zebrafish or mouse when plasma T3 levels were low. During the transition to the adult intestine when T3 levels are high [127, 128], PRMT1 mRNA is upregulated specifically in the bottom of the developing epithelial fold or crypt in the intestine of zebrafish or mouse, respectively, suggesting that T3 regulates the development of the adult epithelial stem cells in zebrafish and mouse intestine in a process that requires high levels of PRMT1, similar to that during Xenopus metamorphosis. The findings also argue that the embryonic/neonatal mouse intestinal stem cells are molecularly distinct from those in the adult mouse intestine.
Two subsequent mouse genetic studies on the transcriptional repressor, B lymphocyte-induced maturation protein 1 (Blimp1) have also provided evidence to support that mouse adult intestinal stem cells are distinct from the embryonic/neonatal epithelial or stem cells [11, 12]. Blimp1 is strongly expressed throughout the intestinal epithelium of embryonic and newborn mice when there are no crypts. Shortly after birth as the intestine matures into the adult form with crypt-villus axis, Blimp1 expression is down-regulated in the intervillus pockets where crypts begin to develop, while its expression in the rest of the epithelial cells persists. As the crypts develop, all cells in the newly formed crypts lack Blimp1 expression and eventually, Blimp1 expression is absent throughout the epithelium in the adult intestine. Thus, the loss of Blimp1 expression in the developing crypt is likely one of the early events for the embryonic/neonatal epithelial cells to develop into the adult stem cells, whose offspring subsequently populate the epithelium in the adult intestine. Subsequently, it has been shown that Blimp1 helps to maintain neonatal tolerance during postembryonic intestinal maturation [129]. These findings suggest that Blimp1 is important for maintaining the natal stage of the intestine and the delay of the formation of adult stem cells while PRMT1 is important for the development of the adult intestinal stem cells and that the two genes may function to keep a balance during transition from the neonatal to adult stage.
6 T3 and adult intestinal physiology and diseases
Aside from its roles in development, T3 is also critical for normal physiological functions of most, if not all, organs in the adult vertebrates and T3 levels regulates metabolic rate [130–132]. T3 and/or T3 metabolism is known to affect stem cell function and regeneration in different tissues including muscle and neurons [133–137]. Similarly, a number of studies suggest that T3 signaling is also critical for adult intestinal physiology. First, recent discoveries of human patients with mutations in TRα revealed that disrupting liganded TRα function causes distinct pathological problems in human compared to similar mutations in the human TRβ [138–141]. In particular, such patients have constipations, suggesting intestinal defects due to the TRα mutations [139–141]. Second, altered T3 levels are associated with intestinal abnormalities and diseases. For example, increased rate of thyroid disorders has been observed in patients with inflammatory bowl diseases (IBD), such as ulcerative colitis and Crohn’s disease [142]. Third, studies in mouse have shown that T3 deficiency or TRα knockout results in abnormal intestinal morphology and a decrease in stem cell proliferation in the adult [122–126]. Finally, in thyroid patients with either hypothyroidism or hyperthyroidism, gastrointestinal manifestations are common signs of the disease. These include reduced motility in hypothyroidism vs. increased motility in hyperthyroidism, autoimmune gastritis, or esophageal compression [143]. Patients with hyperthyroidism can experience frequent bowel movements, diarrhea, nausea and vomiting, while those with hypothyroidism have overall decreased metabolic function accompanied by slow intestinal motility and constipation [144].
T3 and TRs have also been implicated to play a role in intestinal tumorigenesis. Transgenic overexpression of TRα in the intestinal epithelium leads to abnormal intestine that has increased cell proliferation and adenoma in wild type mice; TRα overexpression also synergizes with over-activation of WNT signaling caused by mutations in the WNT pathway to induce intestinal tumorigenesis [5, 145, 146]. Additionally, TRβ mutations and altered TR expression have been reported in intestinal tumors [147–149]. Since intestinal tumor development is likely due to neoplastic transformation of intestinal stem cells [150–152], T3 and TR presumably affect intestinal tumorigenesis by regulating adult intestinal stem cells, as suggested by the adult intestinal phenotypes caused by T3 or TR deficiency in mouse [122–126].
Many T3-regulated genes have also been isolated in the mouse intestinal crypts [124]. T3 may affect intestinal physiology and pathogenesis through these target genes. Of particular interests are genes in the Wnt and Notch signaling pathways, which are known to be important for adult intestinal stem cell function [124, 153, 154]. One such gene is the secreted frizzled-related protein 2 (sFRP2), which is strongly upregulated by T3 in the intestinal crypt and modulates cell fate by regulating Wnt signaling [153]. Similarly, TRα has been shown to directly regulate the transcription of Jag1 gene, a member of the Notch pathway [154]. Additionally, as indicated above, the TR coactivator PRMT1 is also highly upregulated in the developing adult intestinal stem cells during mouse postembryonic intestinal maturation [13]. It remains to be investigated whether PRMT1 plays role in T3-regulation of the Wnt and Notch pathways. On the other hand, our recent unpublished studies suggest that epithelial expression of PRMT1 is required for the proper maturation of the mouse intestine as well as intestinal repair (Roediger, J. and Shi, Y.-B., unpublished observation), supporting a role of PRMT1 in adult intestinal stem cell development and function. Additionally, PRMT1 is overexpressed in colon cancers as well as may other cancers and silencing PRMT1 expression suppresses cancer cell growth, suggesting that PRMT1 also play a role in intestinal cancers [155, 156]. Furthermore, PRMT1 has been associated with other intestinal diseases such as Hischsprung disease, also known as congenital megacolon [157], and lipopolysaccharide-induced intestine tissue inflammation [158]. Thus, proper spatiotemporal expression of PRMT1 is critical for ensuring normal intestinal physiology and preventing diseases, possibly through regulating adult intestinal stem cell function.
Given the involvement of T3 in intestinal diseases and physiology, one may expect that genes involved in T3 synthesis and metabolism also play a role. In particular, T3 is synthesized as through the deiodination of thyroxin (T4) and can be metabolized through further deiodination. There are three deiodinases, D1, D2, and D3, in vertebrates, with D1 and D2 capable of converting T4 to T3 while D3 inactivates T3. It has been shown that the expression of deiodinases is altered in several types of human cancers, including the overexpression of D3 in human colorectal cancers [159–161]. D3 is a direct downstream target of the Wnt/ β-catenin pathway and thus represents an interface between the β-catenin and T3 signaling pathways [160]. β-catenin stimulates D3 and reduces D2, the T4 activating deiodinase, leading to a decrease in intracellular T3. The reduction in T3 in turn promotes cell proliferation while inhibiting E-cadherin expression and cell differentiation. In colon cancer cells the activity of the Wnt/ β-catenin pathway is elevated and the expression of D3 is high, suggesting that hormone activation and inactivation pathways are critical in tumorigenesis [160, 162]. Furthermore, T3 treatment of colorectal cancer spheres represses Wnt pathway and inhibits tumorigenic potential, indicating that T3 signaling is a strong determinant in tumorigenesis [162].
7 Conclusion
The external development and total dependence of amphibian metamorphosis on T3 and TR has enabled easy manipulation of this process for molecular and genetic studies of postembryonic organ development in vertebrates [45, 93, 94, 97, 98, 100, 163]. In particular, the analyses of intestinal metamorphosis in Xenopus laevis and Xenopus tropicalis have revealed important mechanistic insights on how T3 induces the formation of adult intestinal stem cells and identified many candidate adult stem cell genes. These studies as well as those in other vertebrates, especially mouse, have revealed conserved roles and mechanisms in the intestinal development and also implicated a role of T3 in regulating adult intestinal stem cell functions during normal physiology and pathogenesis, especially tumorigenesis. Clearly, functional studies of the candidate stem cells genes in mouse and frogs are needed to determine their roles in these processes. The recent advancements in knockout and knockin technologies in Xenopus [19–26] further enhances the value of the amphibian model for studying the role of adult organ-specific stem cells in human intestinal homeostasis and diseases.
References
Shi YB, Hasebe T, Fu L, Fujimoto K, Ishizuya-Oka A. The development of the adult intestinal stem cells: insights from studies on thyroid hormone-dependent amphibian metamorphosis. cell biosci. 2011;1(1):30.
van der Flier LG, Clevers H. Stem cells, self-renewal, and differentiation in the intestinal epithelium. Annu Rev Physiol. 2009;71:241–60.
Sun G, Shi Y-B. Thyroid hormone regulation of adult intestinal stem cell development: mechanisms and evolutionary conservations. Int J Biol Sci. 2012;8:1217–24.
Sun G, Fu L, Shi Y-B. Epigenetic regulation of thyroid hormone-induced adult intestinal stem cell development during anuran metamorphosis. Cell Biosci. 2014;4:73.
Sirakov M, Kress E, Nadjar J, Plateroti M. Thyroid hormones and their nuclear receptors: new players in intestinal epithelium stem cell biology? Cell Mol Life Sci. 2014;71(15):2897–907.
Clevers H. The intestinal crypt, a prototype stem cell compartment. Cell. 2013;154:274–84.
MacDonald WC, Trier JS, Everett NB. Cell proliferation and migration in the stomach, duodenum, and rectum of man: radioautographic studies. Gastroenterology. 1964;46:405–17.
Toner PG, Carr KE, Wyburn GM. The digestive system: an ultrastructural atlas and review. London: Butterworth; 1971.
McAvoy JW, Dixon KE. Cell proliferation and renewal in the small intestinal epithelium of metamorphosing and adult Xenopus laevis. J Exp Zool. 1977;202:129–38.
Sancho E, Eduard Batlle E, Clevers H. Signaling pathways in intestinal development and cancer. Ann Rev Cell Dev Biol. 2004;20:695–723.
Muncan V, Heijmans J, Krasinski SD, Buller NV, Wildenberg ME, Meisner S, et al. Blimp1 regulates the transition of neonatal to adult intestinal epithelium. Nat Commun. 2011;2:452.
Harper J, Mould A, Andrews RM, Bikoff EK, Robertson EJ. The transcriptional repressor Blimp1/Prdm1 regulates postnatal reprogramming of intestinal enterocytes. Proc Natl Acad Sci U S A. 2011;108(26):10585–90.
Matsuda H, Shi YB. An essential and evolutionarily conserved role of protein arginine methyltransferase 1 for adult intestinal stem cells during postembryonic development. Stem Cells. 2010;28(11):2073–83.
Shi Y-B. Amphibian metamorphosis: from morphology to molecular biology. New York: Wiley; 1999.
Tata JR. Gene expression during metamorphosis: an ideal model for post-embryonic development. Bioessays. 1993;15(4):239–48.
Dodd MHI, Dodd JM. The biology of metamorphosis. In: Lofts B, editor. Physiology of the amphibia. New York: Academic; 1976. p. 467–599.
Kroll KL, Amaya E. Transgenic Xenopus embryos from sperm nuclear transplantations reveal FGF signaling requirements during gastrulation. Development. 1996;122(10):3173–83.
Fu L, Buchholz D, Shi YB. Novel double promoter approach for identification of transgenic animals: a tool for in vivo analysis of gene function and development of gene-based therapies. Mol Reprod Dev. 2002;62(4):470–6.
Young JJ, Cherone JM, Doyon Y, Ankoudinova I, Faraji FM, Lee AH, et al. Efficient targeted gene disruption in the soma and germ line of the frog Xenopus tropicalis using engineered zinc-finger nucleases. Proc Natl Acad Sci U S A. 2011;108(17):7052–7.
Lei Y, Guo X, Liu Y, Cao Y, Deng Y, Chen X, et al. Efficient targeted gene disruption in Xenopus embryos using engineered transcription activator-like effector nucleases (TALENs). Proc Natl Acad Sci U S A. 2012;109:17484–9.
Lei Y, Guo X, Deng Y, Chen Y, Zhao H. Generation of gene disruptions by transcription activator-like effector nucleases (TALENs) in Xenopus tropicalis embryos. Cell Biosci. 2013;3(1):21.
Blitz IL, Biesinger J, Xie X, Cho KW. Biallelic genome modification in F(0) Xenopus tropicalis embryos using the CRISPR/Cas system. Genesis. 2013;51(12):827–34.
Nakayama T, Fish MB, Fisher M, Oomen-Hajagos J, Thomsen GH, Grainger RM. Simple and efficient CRISPR/Cas9-mediated targeted mutagenesis in Xenopus tropicalis. Genesis. 2013;51(12):835–43.
Nakade S, Tsubota T, Sakane Y, Kume S, Sakamoto N, Obara M, et al. Microhomology-mediated end-joining-dependent integration of donor DNA in cells and animals using TALENs and CRISPR/Cas9. Nat Commun. 2014;5:5560.
Shi Z, Wang F, Cui Y, Liu Z, Guo X, Zhang Y, et al. Heritable CRISPR/Cas9-mediated targeted integration in Xenopus tropicalis. FASEB J. 2015;29:4914–23.
Wang F, Shi Z, Cui Y, Guo X, Shi YB, Chen Y. Targeted gene disruption in Xenopus laevis using CRISPR/Cas9. Cell Biosci. 2015;5:15.
Shi Y-B, Ishizuya-Oka A. Biphasic intestinal development in amphibians: embryogensis and remodeling during metamorphosis. Curr Topics Dev Biol. 1996;32:205–35.
Sterling J, Fu L, Matsuura K, Shi Y-B. Cytological and morphological analyses reveal distinct features of intestinal development during Xenopus tropicalis metamorphosis. PLoS ONE. 2012;7:e47407. 47401-47410.
Okada M, Wen L, Miller TC, Su D, Shi YB. Molecular and cytological analyses reveal distinct transformations of intestinal epithelial cells during Xenopus metamorphosis. Cell Biosci. 2015;5:74.
Wen L, Hasebe T, Miller TC, Ishizuya-Oka A, Shi YB. A requirement for hedgehog signaling in thyroid hormone-induced postembryonic intestinal remodeling. Cell Biosci. 2015;5:13.
Ishizuya-Oka A, Shimizu K, Sakakibara S, Okano H, Ueda S. Thyroid hormone-upregulated expression of Musashi-1 is specific for progenitor cells of the adult epithelium during amphibian gastrointestinal remodeling. J Cell Sci. 2003;116(Pt 15):3157–64.
Ishizuya-Oka A, Hasebe T, Buchholz DR, Kajita M, Fu L, Shi YB. Origin of the adult intestinal stem cells induced by thyroid hormone in Xenopus laevis. Faseb J. 2009;23:2568–75.
Schreiber AM, Cai L, Brown DD. Remodeling of the intestine during metamorphosis of Xenopus laevis. Proc Natl Acad Sci U S A. 2005;102(10):3720–5.
Gilbert LI, Tata JR, Atkinson BG. Metamorphosis: post-embryonic reprogramming of gene expression in amphibian and insect cells. New York: Academic; 1996.
Ishizuya-Oka A, Hasebe T. Establishment of intestinal stem cell niche during amphibian metamorphosis. Curr Top Dev Biol. 2013;103:305–27.
Ishizuya-Oka A, Shi YB. Evolutionary insights into postembryonic development of adult intestinal stem cells. Cell Biosci. 2011;1(1):37.
Hasebe T, Fu L, Miller TC, Zhang Y, Shi YB, Ishizuya-Oka A. Thyroid hormone-induced cell-cell interactions are required for the development of adult intestinal stem cells. Cell Biosci. 2013;3(1):18.
Ishizuya-Oka A, Shimozawa A. Induction of metamorphosis by thyroid hormone in anuran small intestine cultured organotypically in vitro. In Vitro Cell Dev Biol. 1991;27A(11):853–7.
Ishizuya-Oka A, Shi YB. Thyroid hormone regulation of stem cell development during intestinal remodeling. Mol Cell Endocrinol. 2008;288(1-2):71–8.
Lazar MA. Thyroid hormone receptors: multiple forms, multiple possibilities. Endocr Rev. 1993;14(2):184–93.
Evans RM. The steroid and thyroid hormone receptor superfamily. Science. 1988;240:889–95.
Yen PM. Physiological and molecular basis of thyroid hormone action. Physiol Rev. 2001;81(3):1097–142.
Davis PJ, Davis FB. Nongenomic actions of thyroid hormone. Thyroid. 1996;6:497–504.
Tsai MJ, O’Malley BW. Molecular mechanisms of action of steroid/thyroid receptor superfamily members. Ann Rev Biochem. 1994;63:451–86.
Buchholz DR, Paul BD, Fu L, Shi YB. Molecular and developmental analyses of thyroid hormone receptor function in Xenopus laevis, the African clawed frog. Gen Comp Endocrinol. 2006;145(1):1–19.
Laudet V, Gronemeyer H. The nuclear receptor FactsBook. San Diego: Academic; 2002.
Mangelsdorf DJ, Thummel C, Beato M, Herrlich P, Schutz G, Umesono K, et al. The nuclear receptor superfamily: the second decade. Cell. 1995;83(6):835–9.
Wong J, Shi YB, Wolffe AP. A role for nucleosome assembly in both silencing and activation of the Xenopus TR beta A gene by the thyroid hormone receptor. Genes Dev. 1995;9(21):2696–711.
Wong J, Shi Y-B, Wolffe AP. Determinants of chromatin disruption and transcriptional regulation instigated by the thyroid hormone receptor: hormone-regulated chromatin disruption is not sufficient for transcriptinal activation. EMBO J. 1997;16:3158–71.
Wong J, Patterton D, Imhof D, Guschin D, Shi Y-B, Wolffe AP. Distinct requirements for chromatin assembly in transcriptional repression by thyroid hormone receptor and histone deacetylase. EMBO J. 1998;17:520–34.
Horlein AJ, Naar AM, Heinzel T, Torchia J, Gloss B, Kurokawa R, et al. Ligand-independent repression by the thyroid hormone receptor mediated by a nuclear receptor co-repressor. Nature. 1995;377(6548):397–404.
Chen JD, Evans RM. A transcriptional co-repressor that interacts with nuclear hormone receptors. Nature. 1995;377:454–7.
Burke LJ, Baniahmad A. Co-repressors 2000. FASEB J. 2000;14(13):1876–88.
Jones PL, Shi Y-B. N-CoR-HDAC corepressor complexes: roles in transcriptional regulation by nuclear hormone receptors. In: Workman JL, editor. Current topics in microbiology and immunology: protein complexes that modify chromatin, vol. 274. Berlin: Springer; 2003. p. 237–68.
Glass CK, Rosenfeld MG. The coregulator exchange in transcriptional functions of nuclear receptors. Genes Dev. 2000;14(2):121–41.
Zhang J, Lazar MA. The mechanism of action of thyroid hormones. Annu Rev Physiol. 2000;62:439–66.
Yoon H-G, Chan DW, Huang ZQ, Li J, Fondell JD, Qin J, et al. Purification and functional characterization of the human N-CoR complex: the roles of HDAC3, TBL1 and TBLR1. Embo J. 2003;22:1336–46.
Zhang J, Kalkum M, Chait BT, Roeder RG. The N-CoR-HDAC3 nuclear receptor corepressor complex inhibits the JNK pathway through the integral subunit GPS2. Mol Cell. 2002;9:611–23.
Ishizuka T, Lazar MA. The N-CoR/histone deacetylase 3 complex is required for repression by thyroid hormone receptor. Mol Cell Biol. 2003;23:5122–31.
Guenther MG, Lane WS, Fischle W, Verdin E, Lazar MA, Shiekhattar R. A core SMRT corepressor complex containing HDAC3 and TBL1, a WD40-repeat protein linked to deafness. Genes & Devel. 2000;14:1048–57.
Li J, Wang J, Wang J, Nawaz Z, Liu JM, Qin J, et al. Both corepressor proteins SMRT and N-CoR exist in large protein complexes containing HDAC3. Embo J. 2000;19:4342–50.
Perissi V, Jepsen K, Glass CK, Rosenfeld MG. Deconstructing repression: evolving models of co-repressor action. Nat Rev Genet. 2010;11:109–23.
Stewart D, Tomita A, Shi YB, Wong J. Chromatin immunoprecipitation for studying transcriptional regulation in Xenopus oocytes and tadpoles. Methods Mol Biol. 2006;322:165–81.
Stewart MD, Li J, Wong J. Relationship between histone H3 lysine 9 methylation, transcription repression, and heterochromatin protein 1 recruitment. Mol Cell Biol. 2005;25(7):2525–38.
Li J, Lin Q, Yoon HG, Huang ZQ, Strahl BD, Allis CD, et al. Involvement of histone methylation and phosphorylation in regulation of transcription by thyroid hormone receptor. Mol Cell Biol. 2002;22:5688–97.
Jones PL, Sachs LM, Rouse N, Wade PA, Shi YB. Multiple N-CoR complexes contain distinct histone deacetylases. J Biol Chem. 2001;276(12):8807–11.
Tomita A, Buchholz DR, Shi Y-B. Recruitment of N-CoR/SMRT-TBLR1 corepressor complex by unliganded thyroid hormone receptor for gene repression during frog development. Mol Cell Biol. 2004;24:3337–46.
Heimeier RA, Hsia VS-C, Shi Y-B. Participation of BAF57 and BRG1-containing chromatin remodeling complexes in thyroid hormone-dependent gene activation during vertebrate development. Mol Endocrinol. 2008;22:1065–77.
Huang Z-Q, Li J, Sachs LM, Cole PA, Wong J. A role for cofactor–cofactor and cofactor–histone interactions in targeting p300, SWI/SNF and Mediator for transcription. EMBO J. 2003;22:2146–55.
McKenna NJ, O’Malley BW. Nuclear receptors, coregulators, ligands, and selective receptor modulators: making sense of the patchwork quilt. Ann N Y Acad Sci. 2001;949:3–5.
Rachez C, Freedman LP. Mediator complexes and transcription. Curr Opin Cell Biol. 2001;13(3):274–80.
Ito M, Roeder RG. The TRAP/SMCC/Mediator complex and thyroid hormone receptor function. Trends Endocrinol Metab. 2001;12(3):127–34.
Sheppard HM, Harries JC, Hussain S, Bevan C, Heery DM. Analysis of the steroid receptor coactivator 1 (SRC1)-CREB binding protein interaction interface and its importance for the function of SRC1. Mol Cell Biol. 2001;21:39–50.
Chen H, Lin RJ, Schiltz RL, Chakravarti D, Nash A, Nagy L, et al. Nuclear receptor coactivator ACTR is a novel histone acetyltransferase and forms a multimeric activation complex with P/CAF and CBP/p300. Cell. 1997;90:569–80.
Demarest SJ, Martinez-Yamout M, Chung J, Chen H, Xu W, Dyson HJ, et al. Mutual synergistic folding in recruitment of CBP/p300 by p160 nuclear receptor coactivators. Nature. 2002;415:549–53.
Onate SA, Tsai SY, Tsai MJ, O’Malley BW. Sequence and characterization of a coactivator for the steroid hormone receptor superfamily. Science. 1995;270(5240):1354–7.
Li J, O’Malley BW, Wong J. p300 requires its histone acetyltransferase activity and SRC-1 interaction domain to facilitate thyroid hormone receptor activation in chromatin. Mol And Cell Biol. 2000;20(6):2031–42.
Torchia J, Rose DW, Inostroza J, Kamei Y, Westin S, Glass CK, et al. The transcriptional co-activator p/CIP binds CBP and mediates nuclear-receptor function. Nature. 1997;387:677–84.
Chen D, Ma H, Hong H, Koh SS, Huang SM, Schurter BT, et al. Regulation of transcription by a protein methyltransferase. Science. 1999;284:2174–7.
Koh SS, Chen DG, Lee YH, Stallcup MR. Synergistic enhancement of nuclear receptor function by p160 coactivators and two coactivators with protein methyltransferase activities. J Biol Chem. 2001;276:1089–98.
Matsuda H, Paul BD, Choi CY, Hasebe T, Shi Y-B. Novel functions of protein arginine methyltransferase 1 in thyroid hormone receptor-mediated transcription and in the regulation of metamorphic rate in Xenopus laevis. Mol Cell Biol. 2009;29:745–57.
Matsuda H, Paul BD, Choi CY, Shi Y-B. Contrasting effects of two alternative splicing forms of coactivator-associated arginine methyltransferase 1 on thyroid hormone receptor-mediated transcription in Xenopus laevis. Mol Endocrinol. 2007;21(5):1082–94.
O’Malley BW, Malovannaya A, Qin J. Minireview: nuclear receptor and coregulator proteomics--2012 and beyond. Mol Endocrinol. 2012;26(10):1646–50.
Bulynko YA, O’Malley BW. Nuclear receptor coactivators: structural and functional biochemistry. Biochemistry. 2011;50(3):313–28.
McKenna NJ, Cooney AJ, DeMayo FJ, Downes M, Glass CK, Lanz RB, et al. Minireview: evolution of NURSA, the nuclear receptor signaling atlas. Mol Endocrinol. 2009;23(6):740–6.
Havis E, Sachs LM, Demeneix BA. Metamorphic T3-response genes have specific co-regulator requirements. EMBO Rep. 2003;4:883–8.
Paul BD, Fu L, Buchholz DR, Shi Y-B. Coactivator recruitment is essential for liganded thyroid hormone receptor to initiate amphibian metamorphosis. Mol Cell Biol. 2005;25:5712–24.
Paul BD, Buchholz DR, Fu L, Shi Y-B. Tissue- and gene-specific recruitment of steroid receptor coactivator-3 by thyroid hormone receptor during development. J Biol Chem. 2005;280:27165–72.
Paul BD, Buchholz DR, Fu L, Shi Y-B. SRC-p300 coactivator complex is required for thyroid hormone induced amphibian metamorphosis. J Biol Chem. 2007;282:7472–81.
Sachs LM, Shi Y-B. Targeted chromatin binding and histone acetylation in vivo by thyroid hormone receptor during amphibian development. PNAS. 2000;97:13138–43.
Wang X, Matsuda H, Shi Y-B. Developmental regulation and function of thyroid hormone receptors and 9-cis retinoic acid receptors during Xenopus tropicalis metamorphosis. Endocrinology. 2008;149:5610–8.
Sachs LM, Jones PL, Havis E, Rouse N, Demeneix BA, Shi Y-B. N-CoR recruitment by unliganded thyroid hormone receptor in gene repression during Xenopus laevis development. Mol Cell Biol. 2002;22:8527–38.
Bilesimo P, Jolivet P, Alfama G, Buisine N, Le Mevel S, Havis E, et al. Specific histone lysine 4 methylation patterns define TR-binding capacity and differentiate direct T3 responses. Mol Endocrinol. 2011;25:225–37.
Matsuura K, Fujimoto K, Fu L, Shi Y-B. Liganded thyroid hormone receptor induces nucleosome removal and histone modifications to activate transcription during larval intestinal cell death and adult stem cell development. Endocrinology. 2012;153:961–72.
Sachs LM, Amano T, Shi YB. An essential role of histone deacetylases in postembryonic organ transformations in Xenopus laevis. Int J Mol Med. 2001;8(6):595–601.
Sachs LM, Amano T, Rouse N, Shi YB. Involvement of histone deacetylase at two distinct steps in gene regulation during intestinal development in Xenopus laevis. Dev Dyn. 2001;222(2):280–91.
Grimaldi A, Buisine N, Miller T, Shi YB, Sachs LM. Mechanisms of thyroid hormone receptor action during development: lessons from amphibian studies. Biochim Biophys Acta. 2013;1830(7):3882–92.
Shi YB, Matsuura K, Fujimoto K, Wen L, Fu L. Thyroid hormone receptor actions on transcription in amphibia: the roles of histone modification and chromatin disruption. Cell Biosci. 2012;2(1):42.
Schreiber AM, Das B, Huang H, Marsh-Armstrong N, Brown DD. Diverse developmental programs of Xenopus laevis metamorphosis are inhibited by a dominant negative thyroid hormone receptor. PNAS. 2001;98:10739–44.
Brown DD, Cai L. Amphibian metamorphosis. Dev Biol. 2007;306(1):20–33.
Buchholz DR, Hsia VS-C, Fu L, Shi Y-B. A dominant negative thyroid hormone receptor blocks amphibian metamorphosis by retaining corepressors at target genes. Mol Cell Biol. 2003;23:6750–8.
Buchholz DR, Tomita A, Fu L, Paul BD, Shi Y-B. Transgenic analysis reveals that thyroid hormone receptor is sufficient to mediate the thyroid hormone signal in frog metamorphosis. Mol Cell Biol. 2004;24:9026–37.
Shi Y-B. Dual functions of thyroid hormone receptors in vertebrate development: the roles of histone-modifying cofactor complexes. Thyroid. 2009;19:987–99.
Nakajima K, Yaoita Y. Dual mechanisms governing muscle cell death in tadpole tail during amphibian metamorphosis. Dev Dyn. 2003;227:246–55.
Denver RJ, Hu F, Scanlan TS, Furlow JD. Thyroid hormone receptor subtype specificity for hormone-dependent neurogenesis in Xenopus laevis. Dev Biol. 2009;326(1):155–68.
Bagamasbad P, Howdeshell KL, Sachs LM, Demeneix BA, Denver RJ. A role for basic transcription element-binding protein 1 (BTEB1) in the autoinduction of thyroid hormone receptor beta. J Biol Chem. 2008;283:2275–85.
Schreiber AM, Mukhi S, Brown DD. Cell-cell interactions during remodeling of the intestine at metamorphosis in Xenopus laevis. Dev Biol. 2009;331(1):89–98.
Sachs LM, Damjanovski S, Jones PL, Li Q, Amano T, Ueda S, et al. Dual functions of thyroid hormone receptors during Xenopus development. Comp Biochem Physiol B: Biochem Mol Biol. 2000;126(2):199–211.
Shi Y-B. Molecular biology of amphibian metamorphosis: a new approach to an old problem. Trends Endocrinol Metab. 1994;5:14–20.
Yen PM. Unliganded TRs regulate growth and developmental timing during early embryogenesis: evidence for a dual function mechanism of TR action. Cell Biosci. 2015;5:8.
Wen L, Shi YB. Unliganded thyroid hormone receptor alpha controls developmental timing in Xenopus tropicalis. Endocrinology. 2015;156:721–34.
Choi J, Suzuki KI, Sakuma T, Shewade L, Yamamoto T, Buchholz DR. Unliganded thyroid hormone receptor alpha regulates developmental timing via gene repression as revealed by gene disruption in Xenopus tropicalis. Endocrinology. 2015;156:735–44.
Hasebe T, Buchholz DR, Shi YB, Ishizuya-Oka A. Epithelial-connective tissue interactions induced by thyroid hormone receptor are essential for adult stem cell development in the Xenopus laevis intestine. Stem Cells. 2011;29(1):154–61.
Ishizuya-Oka A, Shimozawa A. Connective tissue is involved in adult epithelial development of the small intestine during anuran metamorphosis in vitro. Roux’s Arch Dev Biol. 1992;201:322–9.
Luu N, Wen L, Fu L, Fujimoto K, Shi YB, Sun G. Differential regulation of two histidine ammonia-lyase genes during Xenopus development implicates distinct functions during thyroid hormone-induced formation of adult stem cells. Cell Biosci. 2013;3(1):43.
Sun G, Heimeier RA, Fu L, Hasebe T, Das B, Ishizuya-Oka A, et al. Expression profiling of intestinal tissues implicates tissue-specific genes and pathways essential for thyroid hormone-induced adult stem cell development. Endocrinology. 2013;154(11):4396–407.
Miller TC, Sun G, Hasebe T, Fu L, Heimeier RA, Das B, et al. Tissue-specific upregulation of MDS/EVI gene transcripts in the intestine by thyroid hormone during Xenopus metamorphosis. PLoS ONE. 2013;8(1):e55585.
Sun G, Fu L, Wen L, Shi YB. Activation of sox3 gene by thyroid hormone in the developing adult intestinal stem cell during Xenopus metamorphosis. Endocrinology. 2014;155(12):5024–32.
Ishizuya-Oka A, Ueda S, Inokuchi T, Amano T, Damjanovski S, Stolow M, et al. Thyroid hormone-induced expression of Sonic hedgehog correlates with adult epithelial development during remodeling of the Xenopus stomach and intestine. Differentiation. 2001;69:27–37.
Hasebe T, Kajita M, Fu L, Shi YB, Ishizuya-Oka A. Thyroid hormone-induced sonic hedgehog signal up-regulates its own pathway in a paracrine manner in the Xenopus laevis intestine during metamorphosis. Dev Dyn. 2012;241(2):403–14.
Strahl BD, Briggs SD, Brame CJ, Caldwell JA, Koh SS, Ma H, et al. Methylation of histone H4 at arginine 3 occurs in vivo and is mediated by the nuclear receptor coactivator PRMT1. Curr Biol. 2001;11(12):996–1000.
Plateroti M, Gauthier K, Domon-Dell C, Freund JN, Samarut J, Chassande O. Functional interference between thyroid hormone receptor alpha (TRalpha) and natural truncated TRDeltaalpha isoforms in the control of intestine development. Mol Cell Biol. 2001;21(14):4761–72.
Flamant F, Poguet AL, Plateroti M, Chassande O, Gauthier K, Streichenberger N, et al. Congenital hypothyroid Pax8(-/-) mutant mice can be rescued by inactivating the TRalpha gene. Mol Endocrinol. 2002;16(1):24–32.
Kress E, Rezza A, Nadjar J, Samarut J, Plateroti M. The frizzled-related sFRP2 gene is a target of thyroid hormone receptor alpha1 and activates beta-catenin signaling in mouse intestine. J Biol Chem. 2009;284(2):1234–41.
Plateroti M, Chassande O, Fraichard A, Gauthier K, Freund JN, Samarut J, et al. Involvement of T3Ralpha- and beta-receptor subtypes in mediation of T3 functions during postnatal murine intestinal development. Gastroenterology. 1999;116(6):1367–78.
Plateroti M, Kress E, Mori JI, Samarut J. Thyroid hormone receptor alpha1 directly controls transcription of the beta-catenin gene in intestinal epithelial cells. Mol Cell Biol. 2006;26(8):3204–14.
Brown DD. The role of thyroid hormone in zebrafish and axoloft development. Proc Natl Acad Sci U S A. 1997;94:13011–6.
Friedrichsen S, Christ S, Heuer H, Schäfer MKH, Mansouri A, Bauer K, et al. Regulation of iodothyronine deiodinases in the Pax8-/- mouse model of congenital hypothyroidism. Endocrinology. 2003;144:777–84.
Mould AW, Morgan MA, Nelson AC, Bikoff EK, Robertson EJ. Blimp1/Prdm1 functions in opposition to Irf1 to maintain neonatal tolerance during postnatal intestinal maturation. PLoS Genet. 2015;11(7):e1005375.
Franklyn JA, Gammage MD. Thyroid disease: effects on cardiovascular function. TEM. 1996;7:50–4.
Silva JE. Thyroid hormone control of thermogenesis and energy balance. Thyroid. 1995;5(6):481–92.
Freake HC, Oppenheimer JH. Thermogenesis and thyroid function. Annu Rev Nutr. 1995;15:263–91.
Dentice M, Marsili A, Ambrosio R, Guardiola O, Sibilio A, Paik JH, et al. The FoxO3/type 2 deiodinase pathway is required for normal mouse myogenesis and muscle regeneration. J Clin Invest. 2010;120(11):4021–30.
Dentice M, Ambrosio R, Damiano V, Sibilio A, Luongo C, Guardiola O, et al. Intracellular inactivation of thyroid hormone is a survival mechanism for muscle stem cell proliferation and lineage progression. Cell Metab. 2014;20(6):1038–48.
Milanesi A, Lee JW, Kim NH, Liu YY, Yang A, Sedrakyan S, et al. Thyroid hormone receptor alpha plays an essential role in male skeletal muscle myoblast proliferation, differentiation, and response to injury. Endocrinology. 2016;157(1):4–15.
Lemkine GF, Raj A, Alfama G, Turque N, Hassani Z, Alegria-Prevot O, et al. Adult neural stem cell cycling in vivo requires thyroid hormone and its alpha receptor. FASEB J. 2005;19(7):863–5.
Lopez-Juarez A, Remaud S, Hassani Z, Jolivet P, Pierre Simons J, Sontag T, et al. Thyroid hormone signaling acts as a neurogenic switch by repressing Sox2 in the adult neural stem cell niche. Cell Stem Cell. 2012;10(5):531–43.
Refetoff S, Weiss RE, Usala SJ. The syndromes of resistance to thyroid hormone. Endocrinol Rev. 1993;14:348–99.
van Mullem A, van Heerebeek R, Chrysis D, Visser E, Medici M, Andrikoula M, et al. Clinical phenotype and mutant TRalpha1. N Engl J Med. 2012;366(15):1451–3.
Bochukova E, Schoenmakers N, Agostini M, Schoenmakers E, Rajanayagam O, Keogh JM, et al. A mutation in the thyroid hormone receptor alpha gene. N Engl J Med. 2012;366(3):243–9.
Moran C, Chatterjee K. Resistance to thyroid hormone due to defective thyroid receptor alpha. Best Pract Res Clin Endocrinol Metab. 2015;29(4):647–57.
Yakut M, Üstün Y, Kabacan G, Soykan I. Thyroid disorders in patients with inflammatory bowel diseases. Int J Clin Med. 2011;2(89–92).
Ebert C. The thyroid and the gut. J Clin Gastroenterol. 2010;44:402–6.
Maser C, Toset A, Roman S. Gastrointestinal manifestations of endocrine disease. World J Gastroenterol. 2006;12:3174–9.
Kress E, Skah S, Sirakov M, Nadjar J, Gadot N, Scoazec JY, et al. Cooperation between the thyroid hormone receptor TRalpha1 and the WNT pathway in the induction of intestinal tumorigenesis. Gastroenterology. 2010;138(5):1863–74.
Sirakov M, Plateroti M. The thyroid hormones and their nuclear receptors in the gut: from developmental biology to cancer. Biochim Biophys Acta. 2011;1812(8):938–46.
Modica S, Gofflot F, Murzilli S, D’Orazio A, Salvatore L, Pellegrini F, et al. The intestinal nuclear receptor signature with epithelial localization patterns and expression modulation in tumors. Gastroenterology. 2010;138(2):636–48. 648 e631-612.
Markowitz S, Haut M, Stellato T, Gerbic C, Molkentin K. Expression of the ErbA-beta class of thyroid hormone receptors is selectively lost in human colon carcinoma. J Clin Invest. 1989;84(5):1683–7.
Horkko TT, Tuppurainen K, George SM, Jernvall P, Karttunen TJ, Makinen MJ. Thyroid hormone receptor beta1 in normal colon and colorectal cancer-association with differentiation, polypoid growth type and K-ras mutations. Int J Cancer. 2006;118(7):1653–9.
Zeuner A, Todaro M, Stassi G, De Maria R. Colorectal cancer stem cells: from the crypt to the clinic. Cell Stem Cell. 2014;15(6):692–705.
Zhu L, Gibson P, Currle DS, Tong Y, Richardson RJ, Bayazitov IT, et al. Prominin 1 marks intestinal stem cells that are susceptible to neoplastic transformation. Nature. 2009;457(7229):603–7.
Barker N, Ridgway RA, van Es JH, van de Wetering M, Begthel H, van den Born M, et al. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature. 2009;457(7229):608–11.
Skah S, Nadjar J, Sirakov M, Plateroti M. The secreted Frizzled-related protein 2 modulates cell fate and the Wnt pathway in the murine intestinal epithelium. Exp Cell Res. 2015;330(1):56–65.
Sirakov M, Boussouar A, Kress E, Frau C, Lone IN, Nadjar J, et al. The thyroid hormone nuclear receptor TRalpha1 controls the Notch signaling pathway and cell fate in murine intestine. Development. 2015;142(16):2764–74.
Yang Y, Bedford MT. Protein arginine methyltransferases and cancer. Nat Rev Cancer. 2013;13(1):37–50.
Yoshimatsu M, Toyokawa G, Hayami S, Unoki M, Tsunoda T, Field HI, et al. Dysregulation of PRMT1 and PRMT6, Type I arginine methyltransferases, is involved in various types of human cancers. Int J Cancer. 2011;128(3):562–73.
Wu TT, Tsai TW, Shen YT, Hsu JD, Yang LC, Li C. Analyses of PRMT1 proteins in human colon tissues from Hirschsprung disease patients. Neurogastroenterol Motil. 2010;2:984–90.
Wu Y, Wang Y, Zhang Y, Chen LP, Wang JY. [Effect of matrine on NO and ADMA metabolism pathways in serum and tissues of mice with lipopolysaccharide-induced intestine tissue inflammation]. Zhongguo Zhong Yao Za Zhi. 2014;39(12):2318–21.
Casula S, Bianco A. Thyroid hormone deiodinases and cancer. Front Endocrinol. 2012; 3(74).
Dentice M, Luongo C, Ambrosio R, Sibilio A, Casillo A, Iaccarino A, et al. Salvatore D: beta-Catenin regulates deiodinase levels and thyroid hormone signaling in colon cancer cells. Gastroenterology. 2012;143(4):1037–47.
Gereben B, Zeold A, Dentice M, Salvatore D, Bianco AC. Activation and inactivation of thyroid hormone by deiodinases: local action with general consequences. Cell Mol Life Sci. 2008;65(4):570–90.
Catalano V, Dentice M, Ambrosio R, Luongo C, Carollo R, Benfante A, et al. Activated thyroid hormone promotes differentiation and chemotherapeutic sensitization of colorectal cancer stem cells by regulating Wnt and BMP4 signaling. Cancer Res. 2016;76(5):1237–44.
Shi YB. Unliganded thyroid hormone receptor regulates metamorphic timing via the recruitment of histone deacetylase complexes. Curr Top Dev Biol. 2013;105:275–97.
Acknowledgments
This work was supported by the intramural Research Program of NICHD, NIH and National Natural Science Foundation of China (Grant No. 31370187 and 81572447).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sun, G., Roediger, J. & Shi, YB. Thyroid hormone regulation of adult intestinal stem cells: Implications on intestinal development and homeostasis. Rev Endocr Metab Disord 17, 559–569 (2016). https://doi.org/10.1007/s11154-016-9380-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11154-016-9380-1