Abstract
Several studies emphasized the function of the thyroid hormones in stem cell biology. These hormones act through the nuclear hormone receptor TRs, which are T3-modulated transcription factors. Pioneer work on T3-dependent amphibian metamorphosis showed that the crosstalk between the epithelium and the underlying mesenchyme is absolutely required for intestinal maturation and stem cell emergence. With the recent advances of powerful animal models and 3D-organoid cultures, similar findings have now begun to be described in mammals, where the action of T3 and TRα1 control physiological and cancer-related stem cell biology. In this review, we have summarized recent findings on the multiple functions of T3 and TRα1 in intestinal epithelium stem cells, cancer stem cells and their niche. In particular, we have highlighted the regulation of metabolic functions directly linked to normal and/or cancer stem cell biology. These findings help explain other possible mechanisms by which TRα1 controls stem cell biology, beyond the more classical Wnt and Notch signaling pathways.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The thyroid hormones (THs) and their nuclear receptors TRs play multiple roles in the development, homeostasis and metabolic processes in several organs and organisms [rev. in 1, 2]. Their function in intestinal development was described about a hundred years ago based on observations of amphibian metamorphosis [3]. During this postnatal maturation program, increased circulating TH levels are responsible for gut remodeling, including the first phase of apoptosis and shortening of the gut length followed by an increase in cell proliferation [4]. Interestingly, it has been reported that these maturation steps in gut tadpoles depend on complex signaling between different cell types, leading to the emergence of stem cells (SCs) and the establishment of the adult epithelium [5, 6]. In the mammalian intestine, it is well established that continuous epithelium renewal and SC maintenance depends on complex cell interactions and on signaling pathways that globally define and contribute to the SC niche [7, 8]. Similar to amphibians, TH signaling is also important for mammalian intestinal postnatal development and SC biology and the establishment of an SC niche [9,10,11]. The focus of this review will be on TH-dependent intestinal SC niche regulation and on how the alteration of this signal can lead to cell transformation and cancer.
The thyroid hormones and their nuclear receptors
THs are synthesized by the thyroid gland in a process finely regulated by the hypothalamus–pituitary–thyroid axis [12]. The hypothalamus synthesizes and secretes thyrotropin-releasing hormone (TRH) which is transported via axons to the pituitary gland where it interacts with its receptor and stimulates the synthesis of thyroid-stimulating hormone (TSH). TSH binds to its receptor located in the follicular cells of the thyroid, the thyrocytes, inducing the production of the hormones l-Thyroxine (T4) and 3,3′,5-Triiodo-l-thyronine (T3). T4 levels tend to be about 40-fold higher compared to T3 [12, 13]. TH synthesis is strictly regulated and subject to a negative feedback loop, resulting in TH-dependent negative regulation of TSH and TRH production [12, 13]. Thyrocytes secrete THs into the bloodstream and are transported to distal organs by serum proteins such as thyroid binding protein (TBP), transthyretin and albumin [14, 15]. It is important to note that, in addition to the central control of TH production, there is a local mechanism for controlling hormone levels. This involves the presence of plasma membrane transporters [16, 17], which ensure TH entry into cells, as well as the presence of deiodinase selenoenzymes, which activate and/or inactivate the THs [18].
Inside the cells THs, in particular T3, act through binding to the nuclear hormone receptors TRs, which are members of a large protein family of transcription factors [19]. T3 is considered the active form of TH because it binds TRs with 10–15 fold higher affinity than T4 [20, 21]. The gene regulation by TRs through direct DNA-binding on target genes is the classical bimodal switch, or type 1 regulation [22]. Other types of TR-mediated regulations, however, have been described. They include TRs participating in multi-protein complexes independent of DNA binding or TRs recruitment to DNA that is not required to regulate transcription [22]. Finally, it is important to note that THs can function independently of TRs and directly bind to other proteins such as integrin αvβ3 [22]. The TRs have a modular organization, typical of the family members (Fig. 1A) [23]. The A/B N-terminal domain allows the binding of protein co-factors and contains a T3-independent activation function 1 (AF1) region. The C domain contains two zinc finger structural units that bind specific DNA elements within target genes. The D domain is a hinge region linking the C and E domains and also includes a nuclear localization signal. The E domain contains the ligand- and protein co-factor-binding regions, which modulate the downstream transcriptional response. The AF-2 region in this domain allows T3-dependent transcriptional control. The TRs are present in numerous tissues and within several vertebrate species including mammals, amphibians, fishes, and birds [24]. Inside of cells, they are mainly located in the nucleus and, unlike other nuclear hormone receptors, TRs do not have an “inactive” form in the cytoplasm.
TRα1 structure and mode of action. A Modular organization of the TRα1 nuclear receptor, including DNA-binding (DBD) and ligand-binding (LBD) domains. A/B, C, D and E indicate the typical structural domains of the nuclear receptors. B Typical organization of thyroid hormone-responsive elements (TREs) on target genes constituted by two repetitions of half-sites organized in different manners, as indicated. C Model of TR action on target genes. For a gene positively regulated by T3, in the absence of T3, co-repressors are recruited resulting in the inhibition of transcription. Upon T3 binding, co-activators bind to TR inducing transcription. The figure was created with BioRender.com (agreement number: QC23PTXLWH)
In mammals, the THRA and THRB genes code for TRα and TRβ protein isoforms. The THRA locus is located on human chromosome 17 and encodes for two major isoforms, TRα1 and TRα2, but only TRα1 is a true T3 nuclear receptor because it has both DNA- and ligand-binding domains, whereas TRα2 only displays the DNA- but not the ligand-binding domain [25, rev. in 26]. In addition to TRα1 and TRα2, the THRA/Thra locus generates two short isoforms, TR∆α1 and TR∆α2, that lack both the DNA- and ligand-binding domains [27]. These isoforms can function as dominant negative proteins vis-à-vis of the TR receptors and their function has been highlighted in the murine intestine [28]. The THRB locus is located on chromosome 3 and encodes TRβ1 and TRβ2 major isoforms, both acting as T3 nuclear receptors [rev. in 26]. The role of TRα1 is of importance during development [rev. in 11, 25] and in certain adult organs such as the heart, brain and intestine [29]. TRβ1 and/or TRβ2 act principally in the liver, retina, developing inner ear and at the level of the hypothalamus-pituitary axis (i.e., TRH neurons and the pituitary gland) [26, 30]. TRs modulate the expression of target genes by binding to specific genomic regulatory regions called thyroid hormone response elements (TREs) [30]. TREs consist of repeated hexameric nucleotide sequences organized in half-sites as direct repeats separated by four nucleotides (DR4) or as palindromes, direct or inverted [30] (Fig. 1B). The type 1 modulation of gene transcription by TRs involves protein complexes acting as co-activators or co-repressors, depending on the presence or the absence of T3 [31] (Fig. 1C).
The mammalian intestinal epithelium and its organization
The intestinal epithelium has a peculiar vertical organization all along the anteroposterior axis, that correlates with specific functional compartmentalization [7, 32] (Fig. 2). Indeed, a proliferation zone is devoted to SC activity and progenitor proliferation, while a differentiation zone defines the region where differentiated cells reside [32, 33].
Functional organization of the small intestine and colon epithelia. Organization of the intestinal mucosa in the small intestine (upper panel) and colon (middle panel), showing the vertical epithelial axis. At the bottom of the axis reside stem/progenitor cells while in the higher part of the vertical axis are located differentiated cells. It is interesting to note that TRα1 has a gradient of expression similar to Wnt and Notch pathway activities, while the BMP pathway presents an opposite gradient. In the bottom panel are depicted the different cell types present in the mucosa, including epithelial and mesenchymal-derived cells. The Figure was created with BioRender.com (agreement number: QC23PTXLWH)
The intestinal SCs have been the focus of active investigations during the last 15 years and we benefited from a strong increase in the knowledge in this field [7, 33, 34]. They are located at the bottom of the small intestine and colon crypts and cycle rapidly to maintain a continuous turnover of epithelial cells. Crypt-base columnar cells (CBCs), originally described by Cheng and Leblond [35], express the cell surface marker LGR5 and are now considered the active SCs of the intestinal epithelium [36]. CBCs can generate both an SC or a progenitor that enters the transit-amplifying zone and differentiates while migrating toward the top of the vertical axis. Differentiated cells finally die by apoptosis and are shed into the lumen [32, 33]. This entire process is completed within 3–5 days in murine tissue depending on the anterior–posterior intestinal region [32].
The intestinal epithelium hosts two major cell lineages generated from absorptive- and secretory-committed progenitors [7, 32]. Absorptive progenitors differentiate into enterocytes/colonocytes which have a brush border that enlarges their cell surface and facilitates efficient nutrient and water absorption [32]. Microfold cells (M cells) sample gut contents and transport, by trans-cytosis, luminal antigens to the underlying immune cells present in the Peyer’s patches, thereby controlling immune responses [7, 32]. The secretory progenitors, on the other hand, give rise to Paneth/Paneth-like, enteroendocrine (EEC), tuft and goblet cells [7, 32]. Paneth (small intestine) and Paneth-like (colon) cells are the only differentiated cells present at the bottom of the crypts and produce antimicrobial compounds and niche factors important for SC maintenance and activity [37, 38]. EECs, the hormone-producing cells of the intestine, secrete hormones that regulate physiological processes in response to food intake [32, 39]. Tuft cells are chemo-sensors and mediate immune responses [40] while goblet cells secrete mucus that helps to protect the epithelium from the aggressive luminal content [32, 41].
Interestingly, several studies have described other cell populations within the intestinal epithelium with stem characteristics and these have been collectively called “reserve stem cells” as opposed to the active Lgr5-expressing SCs [32, 33]. Depending on the reports, they have been referred to as “quiescent, slow-cycling, revival or facultative”, described and debated as candidate SC-like populations localizing within the SC/progenitor zone [42]. These cells express the markers Bmi1 [43], Tert [44], Hopx [45], Lrig [46], Msi1 [47] and Clu [48] and can regenerate the epithelium after injury and loss of active SCs [43,44,45,46,47,48]. These plastic and dynamic regeneration capacities are, however, not only restricted to SCs since there is evidence showing that lineage-specific committed progenitors have the capacity to de-differentiate and acquire SC properties [8, 49, 50].
Finally, it is important to emphasize that several signaling pathways are involved in the organization of the vertical axis of the intestinal epithelium and in defining the SC zone, by actively participating in the SC niche. This includes Wnt, Notch and epidermal growth factor (EGF) pathways which show a higher gradient of activity in the CBC zone [32] (Fig. 2). The bone morphogenic protein (BMP) and hedgehog (Hh) pathways show an opposite gradient and are highly active at the top of the axis, where they define a no SC/progenitor zone [8, 32]. Collectively, these signaling pathways control the epithelial vertical zonation, the balance between cell proliferation and cell differentiation, as well as between SC self-renewal and commitment [8, 32].
The intestinal epithelium stem cell niche
In 2009, Sato et al. cultured for the first time murine intestinal organoids from isolated Lgr5-expressing crypt SCs [51]. The culture was established in the absence of mesenchymal cells but included growth factors that define “the minimal stromal niche” of necessary signals for the viability and activity of the SCs [51]. Indeed, single Lgr5-expressing cells give rise to 3D multi-budded organoids containing all lineages when supplemented with EGF, R-spondin (a Wnt signaling amplifier) and Noggin (a BMP inhibitor) embedded in an extracellular matrix-like gel [51]. These three niche factors and the complex semi-solid matrix are necessary and sufficient for SC biology and well recapitulated the in vivo situation. In fact, in vivo, the niche surrounds SCs at the bottom of the crypts and sustains their identity, maintenance and activity by supplying Wnt proteins, R-spondins, EGF and BMP inhibitors [8, 32]. The niche is composed of several cell types, including Paneth cells and stromal/sub-epithelial mesenchymal cells (subepithelial myofibroblasts, non-muscle fibroblasts, telocytes, trophocytes, endothelial cells and pericytes) [52,53,54]. Immune cells may also contribute to the intestinal SC niche through action on Paneth and M cells [53, 54]. Moreover, extracellular matrix proteins that form the basal lamina are involved in complex metabolic signaling at the crypt’s bottom and are essential niche elements [55]. Finally, ingested nutrients and diets show a prominent capacity to modulate SCs and this dietary-adaptation capacity has recently been proposed as a hallmark of SC activity [34]. The different cell types and factors described above have been summarized in several reviews [32, 33, 53, 55, 56] and will be discussed here only in relation to THs/TRs.
TH/TR in intestinal physiopathology
As already mentioned, THs and TRs play an important role in intestinal physiology in amphibians, mice and humans, as well as in intestinal tumor biology. Importantly, several studies have attempted to establish a link between THs and intestinal tumor development. However, a consensus has not been reached, and both anti- or pro-tumoral effects of THs have been described [57, 58; rev. in 59,60,61], as summarized in Table 1. Experimental data from models clearly shows a pro-tumoral action of high TH levels [62,63,64,65]. Moreover, studies that have monitored the thyroid axis in patients have reported a clear association between low TSH and high THs with cancers, including colorectal cancers (CRCs) [57, 58, 66]. Finally, some epidemiological studies have been performed in patients treated with either TH-deprivation or TH-supplementation therapies to normalize TH levels and analyzed tumor incidence under these conditions [66,67,68]. However, these treatments do not mimic hypo- or hyperthyroidism and, paradoxically, show highly diverse results, strongly contributing to the lack of consensus in this matter. Altogether, these findings clearly indicate the complexity of the field and would deserve a specific focus.
Regarding the TRs, it has been shown that mutation or altered expression of TRs may be linked to tumorigenesis [69, 70]. Unlike the intestine, TRβ1 has been shown to control cell proliferation in the liver [71], and increased TRβ1 expression is strongly correlated with tumor invasion in hepatic cancer cell lines [72]. In the case of the colon, TRβ1 expression is greatly reduced in cancer due to hypermethylation [73]. Accordingly, TRβ1 is associated with a more differentiated phenotype and its loss of expression is linked to malignant transformation [73, 74]. TRα1 has been reported to be associated with the proliferation of pancreatic cells [75], while another study showed that its expression was associated with a poor prognosis in breast tumors with a mutated BRCA1 gene [76]. Interestingly, the expression of TRβ1 in this same study was inversely associated with a favorable prognosis [76]. Our own functional studies of the TRα1 receptor in mouse models highlighted its role in controlling both intestinal development and homeostasis [rev. in 9, 11]. These properties result from the ability of TRα1 to regulate the Wnt and Notch pathways and to modulate the expression of various cell division and cell cycle regulators [77, 78; rev. in [79]. The importance of these mechanisms was validated by experimentally up-regulating TRα1 expression in intestinal epithelial cells in vivo (vil-TRα1 mice), and we demonstrated the induction of hyperproliferative and hyperplastic crypts [62]. Moreover, according to the assumption of SC-driven tumorigenesis, the overexpression of TRα1 in an Apc-mutated tumor-prone model (vil-TRα1/Apc+/1638N mice) increases tumor incidence, accelerates the intestinal tumorigenic process and induces metastasis dissemination [62]. Conversely, loss of TRα1 expression (TRα0/0/Apc+/1638N mice) drastically reduces tumor incidence [63]. Among the mechanisms at work, the control of [78, 80] or the functional interaction with the Wnt pathway [81] appear to be key processes. Finally, recent findings in human CRCs clearly indicated an up-regulation of THRA gene expression in tumors, a direct correlation with Wnt activity and an association with consensus molecular signature CRC subtypes [63, 82]. Collectively, these observations have clear clinical implications.
In the following paragraphs, we will summarize recent findings in the field of TH/TRs and SC biology, including the SC niche in normal intestines and tumors.
Lessons from amphibian metamorphosis
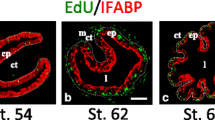
In amphibians, primary culture approaches have shown that the action of THs in non-epithelial cells is required for the appearance of adult epithelium and the emergence of the intestinal SCs [5, 6]. These results underline the importance of TH-dependent epithelial-connective tissue interactions in the establishment of an intestinal SC niche in the developing amphibian intestine [83, 84]. Both TRα and TRβ receptors are important for this developmental program [84,85,86,87] and finely regulate the cell cycle, apoptosis and the remodeling of the extracellular matrix, notably by the induction of metalloproteases [88].
As described earlier, several signaling pathways are important for intestinal development in mammals, such as Hh, WNT, BMP and Notch pathways. Interestingly, some of them are also necessary for the emergence of SCs in amphibians. For example, Shh is a member of the Hh signaling family and is one of the early T3 responsive genes in the metamorphosing amphibian intestine [89]. It is specifically expressed in the epithelium and highly transiently upregulated during the earliest events of metamorphosis. Some downstream components of Shh signaling, such as the receptors Patched, Smoothened and GLIs, are mainly expressed in the connective tissue, thereby responding to the epithelial-derived Shh signal [90]. In addition to Shh, BMP4 is another T3 target gene in the amphibian intestine, but it is specifically expressed in connective tissue and its expression temporally correlates with adult epithelium differentiation [91]. TH-induced BMP4 represses cell proliferation in connective tissue and promotes differentiation of adult epithelial cells through the epithelial-specific presence of its receptor [92]. Importantly, several TH-modulated genes associated with amphibian metamorphosis belong to the WNT pathway and are expressed in both the epithelium and the mesenchyme, directly participating in intestinal remodeling [93,94,95]. Indeed, under the influence of increasing TH levels and in the presence of both TRα and TRβ, the adult epithelium originates from a scattered population of larval absorptive epithelial cells expressing tyrosine kinase-like orphan receptor 2 (Ror2; a receptor for Wnt5a) or Sfrp2 (a Wnt soluble modulator). The non-canonical Wnt5a/Ror2, the canonical sFRP2/Fzd and the hyaluronan/CD44 Wnt signaling, all stimulated by THs, are essential for larval epithelial cells to de-differentiate and generate the adult-type epithelium [93,94,95]. Altogether, the cell–cell interactions that involve WNT, Shh and BMP signaling have an important role in the establishment of the intestinal SC niche that is essential for the neo-generation of SCs of the adult-type epithelium.
Recently, a paper described the importance of TH signaling in tadpole telocytes. These stromal cells in the mammalian intestine surround the bottom of the crypts and are characterized by the marker Foxl1 [96, 97]. Hasebe et al., showed that Foxl1, which is also a marker of telocytes in amphibians, is indirectly upregulated by TH through Shh signaling from the epithelium. These signal exchanges between the epithelium and the Foxl1-expressing niche cells are required for the emergence of SCs in the tadpole intestine [98]. The Notch pathway is activated through ligand-receptor interaction between adjacent cells and is upregulated by TH during metamorphosis [95, 99]. Several genes such as Notch1, Hairy1, Delta-like 1 (Dll1) and Jagged 1 (Jag1) are directly or indirectly TH-responsive [99, 100]. While Dll1 is expressed in the adult epithelium, Jag1 is present in both larval epithelium and stromal fibroblasts beneath adult progenitor/SCs. These findings suggest different roles of the Notch elements in the developing or adult intestinal epithelium and SC niche [95, 99].
What do we know in mammals?
Contrary to amphibians, in mammals the role of TH and TRs in intestinal SCs and their niche is much less characterized. Recently, we showed the role of T3 and of TRα1 in murine intestinal SCs in vivo and in derived organoids. T3, in a TRα1-dependent manner, induced in organoids an increase in cell turnover affecting the commitment of progenitors into goblet cells [10]. In vivo, an increase of crypt cell proliferation upon T3 exposure was also present along with regulation of cell differentiation, but in this case, it was directed towards Paneth cells [10]. This study demonstrated for the first time the role of T3 and TRα1 in increasing the number of SCs and their markers, related to an effect on Paneth cells that, as we described in the previous section, are part of the SC niche [37]. TH/TR-dependent cell–cell interactions and instructive signal exchanges between the epithelium and connective tissue have not been formally demonstrated yet. Nevertheless, TH-dependent stimulation of ligands and receptors participating in Wnt, BMP, Hh and Notch pathways has been shown in several reports [63, 77, 78]. Thus, in the light of these findings, we can hypothesize that THs through TRα1 may also play a role in mesenchymal cells participating to the SC niche. This action might result in direct and indirect modulation of multiple signals in different cell types and tissues, similar to what has been shown in amphibians [96, 98].
The local TH/TR signaling has been investigated in the context of intestinal tumors and cancer stem cells (CSCs). The deiodinases, TH metabolizing enzymes, have been well studied both in normal physiology and in cancer [101]. The stroma of Apc-mutant mice has elevated expression of Dio2, a T3-activating enzyme. When the expression of Dio2 is inhibited in these cells, or when hypothyroidism is chemically induced, the growth of intestinal tumors is blunted. This is indicative of crosstalk between the epithelium and the connective tissue mediated by local or systemic TH levels. Importantly, this effect is accompanied by a reduction in angiogenesis [102]. High expression of Dio3, a T3-inactivating enzyme, has been reported in human CRCs [103]. The depletion of Dio3 in adenocarcinoma cell lines or primary tumor cells indicated a pro-differentiation action of T3, through downregulation of Wnt and increase of BMP pathways [103,104,105]. The contrasting results between these reports may be due to the altered expression of these two enzymes in the stroma (Dio2) or in the epithelial tumor cells (Dio3).
These findings are also discordant with our data showing a positive regulation of TH/TRα1 on the Wnt pathway, cell proliferation, tumorigenicity and on SC activity [10, 62, 63, 78, 80]. While we lack explanations for these discrepancies, it is important to note that TH treatment in vivo and in organoids have different outcomes. Indeed, organoids are an epithelial-only model where T3 induces a “thyroid shock”, which is not the case when treating the animals and perform analyses on the intestinal epithelium in its integrated physiological context [10].
In addition to mesenchymal derivatives, macrophages also participate in the intestinal SC niche and the CRC tumor microenvironment. In these cells, THs regulate the triggering receptor expressed on myeloid cells-2 (TREM2), a cell surface receptor on macrophages and microglia involved in immune suppression of the tumor microenvironment [106]. This finding is in line with previous data showing the influence of THs as immunomodulators with the potential of decreasing the physiological response against tumor cells [107]. We can affirm that TH/TRα1 influences intestinal tumor biology through its interaction with the Wnt/β-catenin and other signaling pathways [63, 108]. However, we should also consider a possible tumor-promoting role via CSC maintenance by an action on the tumor microenvironment (i.e., the CSC niche).
Finally, as previously stated, non-classical TH functions have been described. These are primarily dependent on integrin αvβ3, a transmembrane receptor capable of binding T3 and T4 independently of TRs [109]. Other non-genomic actions are associated with the 30 or 43 kDa TRα isoforms, which are N-terminally truncated proteins located in the plasma membrane and mitochondria, respectively [110]. Interestingly, these non-classical and non-genomic actions may result in SC maintenance via niche factors [111]. These regulations include the control of COX2, hypoxia-inducible factor 1 (HIF1), fibroblast growth factor 2, NOS2, MMP9, and genes related to glucose and lipid metabolism such as sterol regulatory element-binding protein 1 (SREBP-1) [111] and CD47 [109]. Importantly, actions of THs in the tumor stroma via hypoxia induction, tumor microenvironment recruitment and stimulation of angiogenesis have been shown in hepatocellular carcinoma [112, 113].
TH/TR and metabolic regulations in intestinal SC/CSC and their niche
Several reviews have summarized the roles of signaling pathways in relation to TH/TR in the normal intestine and in cancers [9, 11, 70, 81]. Here we have focused on metabolic regulation, given the increasing interest on the role of metabolism in normal tissues and tumors. In the intestine, particularly in the field of gut SCs and their niche, several studies have shown that nutrient availability and metabolic pathways can directly regulate or instruct cellular function [114,115,116]. The various cell types within the intestinal crypt and the cells participating in the niche preferentially use different metabolic pathways and, depending on the cell type or on the cell state, a metabolic switch can occur. For instance, active SCs rely highly upon oxidative phosphorylation (OXPHOS) whereas Paneth cells rely on glycolysis (Fig. 3A) [117, 118].
Proposed models recapitulating the integration of classical signaling and metabolic pathways depending on TH/TRα1 in intestinal crypts and colon cancer cells. A Differential outcome in SCs or in Paneth cells in response to T3, possibly inducing metabolic shifts. Accumulating data suggest that T3 through the TRα1 receptor modulates the stemness in crypts via regulation of the Wnt and Notch pathways. However, recent data also point to the induction of metabolic challenges stimulated by high glucose and high lipid metabolism, acting on the balance between self-renewal and differentiation [8, 32, 37]. Importantly, T3 also induces an increase of Paneth cells in the intestinal crypts [10]. Paneth cells are well known to act on stemness through Wnt and Notch, but also to produce lactate that once up-taken by SCs contributes to the oxidative phosphorylation (OXPHOS) metabolic pathway [115], participating in the maintenance of the stem phenotype. B Overview of the integrated glucose and lipid metabolism in the intestinal SCs under T3 treatment. We speculate that T3 might mimic a high glycolysis and high lipolysis condition increasing OXPHOS. Upon oxidation of fatty acids, stimulated OXPHOS can induce reactive oxygen species (ROS) and p38 expression. The stemness, stimulated by Wnt and Notch, or the differentiation induced by ROS/p38, depend on the balance between these two events. C Integrated view of the action of T3/TRα1 in a cancer context. Adipocytes are strongly mobilized in the cancer environment and considered as part of the cancer stem cell niche [56, 149, 150], releasing high levels of lipids that can be absorbed by cancer cells. The T3-induced transcriptional program includes genes involved in fatty acid oxidation, OXPHOS, and stemness together with mitochondrial turnover. The balance between these major events determines the extent of stem potential versus their differentiation. The Figure was created with BioRender.com (agreement number: PO23PTXM00)
Importantly, THs are critical regulators of metabolic processes and positively or negatively control the transcription of anabolic and catabolic gene subsets that affect energy homeostasis and metabolism [119]. Hyperthyroidism promotes a hypermetabolic state characterized by increased resting energy expenditure, weight loss, reduced cholesterol levels, increased lipolysis and gluconeogenesis. Conversely, hypothyroidism is associated with the opposite effects. THs influence key metabolic pathways that control energy balance by regulating energy storage and expenditure as described in the brain, white fat, brown fat, skeletal muscle, liver and pancreas [119].
In addition to TRs, other nuclear hormone receptors, including peroxisome proliferator-activated receptor alpha (PPARα), PPARγ and liver X receptor (LXR) share a similar structure and mode of action and heterodimerize with the retinoid X receptor (RXR). These similarities have suggested potential functional interactions in the control of metabolic gene expression [120]. PPARs and LXRs are ‘‘permissive’’ RXR partners that bind dietary lipids with low affinity and activate enzymes involved in lipid metabolism [120]. TRs, on the other hand, are "non-permissive" RXR heterodimers, bind to T3 with high affinity and mediate feedback regulation of their ligand [119, 120]. T3-liganded TRs dominate the interaction with RXR and may have a stronger effect in co-regulated genes than nutrient signals acting through PPAR and LXR. Furthermore, THs control the expression of SREBPs [121, 122], key regulators of lipid metabolism and long-chain fatty acids [123]. It has been shown, at least in vitro, that SRPBs can inhibit T3 binding to TRs, suggesting another level of gene regulation through competition between nutrient signaling and T3 binding to TRs [120].
The role of THs and TRs in lipid and carbohydrate metabolism has been extensively studied in the liver, where TRβ1 is the major TR isoform responsible for these actions [2, 120]. In the intestine, TRα1 is the isoform present in the crypt compartment, while TRβ is expressed in differentiated cells of the villi [9]. Within the crypts, TRα1 is enriched in SCs compared with progenitors, whereas Paneth cells express low TRα1 levels [10]. These findings point to a complex T3-mediated metabolic interplay between the various crypt cell types, which eventually influence SC biology (Fig. 3A). In intestinal tumors, the expression of TRα1 is upregulated [63] while the THRB gene is strongly silenced [73, 74], indicating that metabolic controls in this context rely exclusively on TRα1.
Carbohydrate metabolism and mitochondrial activity
TH status influences glucose metabolism. Excess of THs stimulate hepatic glucose production and increases the expression of the glucose transporters GLUT4 and SLGT1 in skeletal muscle and intestine, respectively [119]. Liganded TRα1 impairs glucose-stimulated insulin secretion by acting directly at the level of the pancreatic islets [119]. A further link between glucose metabolism and TH signaling is the T3-dependent induction of the carbohydrate response element-binding protein (ChREBP). This transcription factor stimulates the expression of genes involved in glycolysis and lipogenesis in response to glucose and insulin [120]. In a TRα- and TRβ-dependent manner, THs reduce insulin levels, regulate the expression of genes in the liver and skeletal muscle and stimulate ChREBP expression, which in turn influences glucose response and insulin secretion [120].
In the intestine, THs increase alkaline phosphatase and peptidase activities in a dose-dependent manner in vivo and inhibit γ-glutamyltransferase in vitro [124, 125]. In addition, studies in TRα-knockout animals showed that lactase, sucrase, aminopeptidase and FABP expression and activities are decreased [126], indicating a regulation by the TRα1 receptor of diverse metabolic processes in this organ. More recently, we also showed that T3 impacts the metabolic activity of intestinal organoids by upregulating all enzymes involved in glycolysis, pyruvate metabolism and OXPHOS [10]. This T3-induced metabolic change is parallel to an alteration in SC renewal by favoring secretory progenitor lineages [10]. We can assume that THs via TRα1 control the metabolic state of the SCs and, in consequence, regulate their self-renewal capacity and the fate determination of progenitor cells. The fine-tuned mechanisms at the basis of these complex regulations remain, however, to be fully elucidated, as schematized in Fig. 3.
The Notch/FOXO/mitochondria axis regulates goblet and Paneth cell lineages, with mitochondrial fission as a requisite for inducing differentiation [127]. Mitochondrial dysfunctions are central in the development of inflammatory bowel diseases and cancer, and affect intestinal SCs and Paneth cells by modifying the cellular phenotypes and lineage commitment [128]. Interestingly, THs and TRα1 have been implicated in mitochondrial biogenesis and activity in mammals [129, 130] and the mitochondrial fission is also a key process for the amphibian TH-dependent intestinal maturation and the emergence of SCs [131]. The pyruvate dehydrogenase kinase PDK4 gene expression had been shown to be modulated in the rat liver by T3 [132]. This finding suggests that T3, by modulating the mitochondrial metabolic pathways, may direct the differentiation of SCs towards secretory lineages, as it has been shown in mouse organoids [10] (Fig. 3B). Thus, it is possible that in both mammals and amphibians an interplay occurs between classical cell signaling and metabolic pathways and both share the mitochondria as an essential regulator of SC maintenance and differentiation potentialities.
Aside from direct regulation of target genes, THs can also modulate the expression of several genes by non-genomic actions. TH binding to integrin αVβ3 in colon adenocarcinoma cell lines has been shown to favor an increase of AMPK and the inhibition of mTOR resulting in increased tumor cell aggressivity [133]. In the mitochondria, the truncated TRα isoforms or the hormones T3 or T2, generated from the deiodination of T3, modulate the activity of several components of the respiratory chain leading to an overall increase in OXPHOS [134]. Moreover, THs can regulate the expression of several enzymes and transporters, like the intestinal fructose transporter GLUT5 [135] and PKM2 enzyme, involved in the generation of acetyl-CoA that favors OXPHOS metabolism [136]. Future research will need to focus on characterizing the cell types in which these regulations take place for a better understanding of the integrated processes.
Lipid metabolism
Mouse models with TRα and TRβ gene point mutations show a range of metabolic phenotypes, including impairment of cholesterol metabolism, fatty acid oxidation, lipolysis and increased cholesterol and triglyceride serum levels [120].
THs stimulate lipolysis, lipogenesis and cholesterol reduction by regulating the expression of SREBPs. The SREBP-1c isoform, involved in the fatty acid synthesis and glucose metabolism, is repressed by THs, while the SREBP-2 isoform, which is a lipid sensor important in cholesterol metabolism, is induced by THs. In addition, both TREs and sterol regulatory elements (SREs) are required for the activation of human acetyl–CoA carboxylase [35] and low-density lipoprotein receptor genes [36]. Thus, THs directly regulate SREBPs but also control genes harboring close TREs and SREs in their regulatory regions [137, 138]. Interestingly, SREBPs have SC- and tumorigenic- promoting activities in the intestine [139, 140], indicating another possible functional link between TRα1 and SREBPs. TRs can also bind to LXR-responsive elements in the intestinal ABCA1 gene, affecting the cholesterol efflux through suppression of the LXR-dependent ABCA1 expression [119]. In addition, a crosstalk between TRs and PPARγ has been shown in the regulation of lipid metabolism, in particular the induction of carnitine palmitoyltransferase 1 (CPT1a) enzyme, through peroxisome proliferator-activated receptor gamma co-activator (PGC-1α), a TH-target gene [141]. CPT1a is the key enzyme involved in fatty acid oxidation (FAO) as it exchanges the acyl group of the fatty acid for carnitine, so it can enter the mitochondria to be oxidated [142] (Fig. 3B).
T3 treatment of intestinal organoids causes downregulation of the lipid metabolism pathway, including most of the enzymes involved in FAO, compared to non-treated organoids, with the only exception of CPT1a [9]. AMPK, another major regulator of FAO, is regulated by THs [143] (Fig. 3B). In addition, the mitochondrial trifunction protein (MTP), which catalyzes three FAO reactions, is stabilized and modulated by the TRα p43 isoform [144]. MTP modulation was also observed in intestinal SCs during calorie restriction and fasting [116, 145], leading to an increase of SCs, Paneth cells and progenitors, which is similar to what happens in mice when treated with T3 [10]. During calorie restriction, it was shown that mTORC1 inhibition is important for intestinal regeneration and preservation of “reserve SCs” from DNA damage [146]. T3 treatment may have a similar role in this context, as it inhibits mTOR [10]. Moreover, TRα1 protects the epithelium from DNA damage [147], strongly supporting its role in epithelium regeneration upon injury, through action on “reserve SCs”.
In the case of the high-fat diet, PPARδ activates the Wnt pathway and increases the intestinal SC number by modulation of metabolic responses and provoking SC self-renewal independently from Paneth cells, through the secretion of Jag1 [114]. A high-fat diet also has a tumor-inducing role via increased CPT1a [148], suggesting that THs and TRα1 may have a similar inducing role via CPT1a and FAO induction through Wnt or Jag1 modulation [10, 77, 78].
Altogether, the combination of classical cell signaling with metabolism and diet, nuclear receptors and hormones (like THs) help to maintain SC self-renewal and regulate differentiation through modification of mitochondrial metabolism both in SCs and the cells of their niche. It is worth highlighting that most of the studies to date have focused on the role of metabolism in SCs and Paneth cells. Therefore, additional studies on other cell types within the intestinal SC niche are necessary to fully understand the interrelation between different cell types in this complex metabolic interplay. Towards this end, newer studies have begun to investigate the role of adipocytes in CSCs and intestinal tumorigenesis. As mentioned above, THs have an important role in FAO and SC/CSC metabolism, resulting in increased stemness and tumorigenesis. They are also important in adipose tissue lipidic mobilization [119]. During calorie restriction, SCs and CSCs uptake fatty acids from the nearby adipocytes and promote the expression of CPT1a through PPARγ [149]. This same phenomenon was observed in organoids supplemented with fatty acids [114]. A similar role can be hypothesized for TRs, as both nuclear receptors engage in crosstalk in the regulation of lipid metabolism, adipogenesis, and tumorigenesis [150]. Colon tumors typically grow in an adipose tissue-enriched microenvironment, making adipocytes a key niche component for regulating cancer metabolism (Fig. 3C). The uptake of fatty acids from adipocytes renders colon cancer cells resistant to nutrient deprivation and favors a high level of Wnt signaling by acetylation of β-catenin [149]. It is therefore likely that THs and TRα1 contribute to the maintenance of CSCs in CRC by a positive induction of the Wnt signaling in CSCs and their niche by upregulating CPT1a and increasing the metabolism of fatty acids in adipocytes [10, 63, 78].
Chromatin remodelling
As described earlier, changes in nutrient availability can result in the reprogramming of cellular metabolism. Core metabolite pools such as ATP, S-adenosyl methionine, acetyl-CoA, NAD/NADP and α-ketoglutarate underlie a variety of essential metabolic reactions and are used by chromatin modifiers in the establishment of epigenetic signatures [151].
Chromatin remodeling is a requirement for the transcription of genes associated with anuran metamorphosis. Intestinal SCs are generally characterized by having open chromatin that allows them to change quickly from self-renewal to differentiation. In amphibians, the presence of different co-activators, which act as histone modifiers, remodel the chromatin in the presence of THs and allow the transcription of TR-responsive genes [152]. Many data on TR-mediated chromatin remodeling came from observations on TRβ and its interactions with various protein complexes. The SWI/SNF chromatin remodeling complex, for example, which is associated with NCOR, a well-known TR co-repressor [31], has been shown to be required for TRβ-mediated gene repression in human thyroid epithelial cell lines [153]. In these same cells, the recruitment of the SWI/SNF complex in T3-activated genes is dependent on interactions between TRβ and the p300/CBP coactivator complex [153], with T3 increasing the chromatin accessibility in proximal rather than in distal binding sites [153]. Furthermore, steroid receptor coactivators (SRCs) and p300/CBP function synergistically to stimulate TRβ-dependent transcription [154]. SRC facilitates the recruitment of p300/CBP to T3-regulated promoters via histone acetylation [154], allowing the recruitment of SWI/SNF and Mediator complexes [155]. It is important to underline that the TRs involved in these chromatin modifications may play a critical role in cellular reprogramming. As discussed earlier, TRα1 is expressed in the intestinal crypts in mammals, whereas TRβ1 is expressed in the villi. This specific expression pattern suggests that, as in amphibians, each TR is associated with a distinct profile of epigenetic regulation in the various epithelial cells [156].
Large-scale studies showed that regulation of gene expression by TRs is not only due to the binding of TREs but also to chromatin remodeling in regions close to TREs [138]. This changed the prevailing view that the transition from a repressed to an activated state might not simply work through a switch from co-repressor to co-activator interaction with pre-occupied TRs [138]. In addition, co-activators and chromatin remodelers may also be recruited through ligand-dependent TR occupancy of chromatin, a mechanism already observed for other nuclear hormone receptors [138]. Most T3-activated genes associate with hormone-facilitated TR recruitment to chromatin [138]. For example, some TH target genes fall under epigenomic regulation, which modifies their transcription [152, 156, 157]. Interestingly, sirtuin-mediated epigenetic modifications may regulate the metabolic state of various adult SCs, including intestinal SCs as well as the cells participating in the niche [rev. in 158]. SIRT1 is a NAD-dependent deacetylase that removes acetyl groups from proteins like histones inducing NAD+/SIRT1-mediated gene repression [157]. The NAD+/NADH ratio is important for SIRT1 activity and reflects the metabolic status of the cells. Indeed, SIRT1 has been connected to the beneficial effects of calorie restriction in mice. Physiological adaptation to starvation results in higher SIRT1 activity in liver and brown adipose tissue parallel to decreased TH serum levels, to achieve energy conservation [159]. SIRT1 was also found to cooperate with PGC-1α to enhance the response to T3 in the liver, but it also stimulated TRβ1 activity independently of PGC-1α [160]. This last effect depends on the interaction between SIRT1 and TRβ1, promoting T3-dependent TRβ1 deacetylation and increasing ubiquitin-mediated TRβ1 turnover [160]. Surprisingly, only a subset of TRβ1 target genes, including glucose 6 phosphatase, were strongly inhibited by SIRT1 knockdown in the liver [160], suggesting a complex SIRT1-dependent and independent function of TRβ1. The hormone T2 can also activate SIRT1 and induce PGC-1α deacetylation, resulting in the activation of genes required for FAO [2]. Thus, it can be speculated that SIRT1 might cooperate with PGC-1α and stimulate TRα1 to modulate the metabolic status of the intestinal SCs and their niche cells.
Altogether, THs and TRs can increase the pool of regulated target genes by controlling metabolic fluxes and chromatin remodeling. This, in turn, may affect the metabolic activities of different cell types in both the normal intestine and in tumors. Last, but not least, TH/TR-dependent alterations in the metabolic status influence cell identity (i.e., stem vs non-stem) and behavior (i.e., normal vs tumor).
Conclusions
In this review, we have highlighted the importance of TH-dependent signaling on intestinal SC physiology, including a direct action on SCs as well as in the SC niche. In addition, since alteration of TH signaling can lead to tumor development, we also discussed new insights into CSC biology and human CRCs.
The THRA gene is frequently upregulated in CRC patients [63], in particular in molecular subtypes linked to high metabolic features and elevated Wnt signaling [63]. These results are clearly of high clinical relevance and raise the question of the overall impact of an altered TH/TRα1 axis in CRC biology from tumor initiation to progression and metastasis.
Data availability
Not applicable.
References
Tata JR (2006) Amphibian metamorphosis as a model for the developmental actions of thyroid hormone. Mol Cell Endocrinol 246:10–20
Sinha RA, Singh BK, Yen PM (2014) Thyroid hormone regulation of hepatic lipid and carbohydrate metabolism. Trends Endocrinol Metab 25:538–545
Galton VA (1992) The role of thyroid hormone in amphibian metamorphosis. Trends Endocrinol Metab 3:96–100
Brown DD, Cai L (2007) Amphibian metamorphosis. Dev Biol 306:20–33
Schreiber AM, Cai L, Brown DD (2005) Remodeling of the intestine during metamorphosis of Xenopus laevis. Proc Natl Acad Sci 102:3720–3725
Ishizuya-Oka A, Shi YB (2008) Thyroid hormone regulation of stem cell development during intestinal remodeling. Mol Cell Endocrinol 288:71–78
Clevers H (2013) The intestinal crypt, a prototype stem cell compartment. Cell 154:274–284
Beumer J, Clevers H (2016) Regulation and plasticity of intestinal stem cells during homeostasis and regeneration. Dev 143:3639–3649
Sirakov M, Kress E, Nadjar J, Plateroti M (2014) Thyroid hormones and their nuclear receptors: new players in intestinal epithelium stem cell biology? Cell Mol Life Sci 71:2897–2907
Godart M, Frau C, Farhat D, Giolito MV, Jamard C, Le Nevé C, Freund J-N, Penalva LO, Sirakov M, Plateroti M (2021) Murine intestinal stem cells are highly sensitive to modulation of the T3/TRα1-dependent pathway. Development 148:dev194357
Frau C, Godart M, Plateroti M (2017) Thyroid hormone regulation of intestinal epithelial stem cell biology. Mol Cell Endocrinol 459:90–97
Ortiga-Carvalho TM, Chiamolera MI, Pazos-Moura CC, Wondisford FE (2016) Hypothalamus-pituitary-thyroid axis. Compr Physiol 6:1387–1428
Feldt-Rasmussen U, Effraimidis G, Klose M (2021) The hypothalamus-pituitary-thyroid (HPT)-axis and its role in physiology and pathophysiology of other hypothalamus-pituitary functions. Mol Cell Endocrinol 525:111173
Bartalena L, Robbins J (1993) Thyroid hormone transport proteins. Clin Lab Med 13:583–598
Refetoff S (2000) Thyroid hormone serum transport proteins. Endotext. South Dartmouth (MA): MDText.com. PMID: 25905421
Groeneweg S, Van Geest FS, Peeters RP, Heuer H, Visser WE (2020) Thyroid hormone transporters. Endocr Rev 41:146–201
Kinne A, Schülein R, Krause G (2011) Primary and secondary thyroid hormone transporters. Thyroid Res 4:S7
Bianco AC, Kim BW (2006) Deiodinases: implications of the local control of thyroid hormone action. J Clin Invest 116:2571–2579
Weikum ER, Liu X, Ortlund EA (2018) The nuclear receptor superfamily: a structural perspective. Protein Sci 27:1876–1892
Apriletti JW, Eberhardt NL, Latham KR, Baxter JD (1981) Affinity chromatography of thyroid hormone receptors. Biospecific elution from support matrices, characterization of the partially purified receptor. J Biol Chem 256:12094–12101
Samuels HH, Tsai JS, Casanova J, Stanley F (1974) Thyroid hormone action. In vitro characterization of solubilized nuclear receptors from rat liver and cultured GH1 cells. J Clin Invest 54:853–865
Flamant F, Cheng SY, Hollenberg AN, Moeller LC, Samarut J, Wondisford FE, Yen PM, Refetoff S (2017) Thyroid hormone signaling pathways: Time for a more precise nomenclature. Endocrinology 158:2052–2057
Germain P, Staels B, Dacquet C, Spedding M, Laudet V (2006) Overview of nomenclature of nuclear receptors. Pharmacol Rev 58:685–704
Pascual A, Aranda A (2013) Thyroid hormone receptors, cell growth and differentiation. Biochim Biophys Acta Gen Subj 1830:3908–3916
Koenig RJ, Lazar MA, Hodin RA, Brent GA, Larsen PR, Chin WW, Moore DD (1989) Inhibition of thyroid hormone action by a non-hormone binding c-erbA protein generated by alternative mRNA splicing. Nature 337:659–661
Flamant F, Samarut J (2003) Thyroid hormone receptors: lessons from knockout and knock-in mutant mice. Trends Endocrinol Metab 14:85–90
Chassande O, Fraichard A, Gauthier K, Flamant F, Legrand C, Savatier P, Laudet V, Samarut J (1997) Identification of transcripts initiated from an internal promoter in the c-erbAα locus that encode inhibitors of retinoic acid receptor-α and triiodothyronine receptor activities. Mol Endocrinol 11:1278–1290
Plateroti M, Gauthier K, Domon-Dell C, Freund J-N, Samarut J, Chassande O (2001) Functional interference between thyroid hormone receptor α (TRα) and natural truncated TRΔα isoforms in the control of intestine development. Mol Cell Biol 21:4761–4772
Jones I, Srinivas M, Ng L, Forrest D (2004) The thyroid hormone receptor β gene: structure and functions in the brain and sensory systems. Thyroid 13:1057–1068
Yen PM (2001) Physiological and molecular basis of Thyroid hormone action. Physiol Rev 81:1097–1142
Brent GA (2012) Mechanisms of thyroid hormone action. J Clin Invest 122:3035–3043
Beumer J, Clevers H (2020) Cell fate specification and differentiation in the adult mammalian intestine. Nat Rev Mol Cell Biol 22:1–39
Gehart H, Clevers H (2019) Tales from the crypt: new insights into intestinal stem cells. Nat Rev Gastroenterol Hepatol 16:19–34
Baulies A, Angelis N, Li VSW (2020) Hallmarks of intestinal stem cells. Development 147:1–12
Cheng H, Leblond CP (1974) Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine I Columnar cell. Am J Anat 141:461–479
Barker N, Van Es JH, Kuipers J, Kujala P, Van Den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H, Peters PJ, Clevers H (2007) Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449:1003–1007
Sato T, Van Es JH, Snippert HJ, Stange DE, Vries RG, Van Den Born M, Barker N, Shroyer NF, Van De Wetering M, Clevers H (2011) Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature 469:415–418
Rothenberg ME, Nusse Y, Kalisky T, Lee JJ, Dalerba P, Scheeren F, Lobo N, Kulkarni S, Sim S, Qian D, Beachy PA, Pasricha PJ, Quake SR, Clarke MF (2012) Identification of a cKit+ colonic crypt base secretory cell that supports Lgr5+ stem cells in mice. Gastroenterology 142:1195-1205.e6
Beumer J, Gehart H, Clevers H (2020) Enteroendocrine dynamics—new tools reveal hormonal plasticity in the gut. Endocr Rev 41:695
Banerjee A, McKinley ET, Von Moltke J, Coffey RJ, Lau KS (2018) Interpreting heterogeneity in intestinal tuft cell structure and function. J Clin Invest 128:1711–1719
McCauley HA, Guasch G (2015) Three cheers for the goblet cell: maintaining homeostasis in mucosal epithelia. Trends Mol Med 21:492–503
Muñoz J, Stange DE, Schepers AG, Van De Wetering M, Koo BK, Itzkovitz S, Volckmann R, Kung KS, Koster J, Radulescu S, Myant K, Versteeg R, Sansom OJ, Van Es JH, Barker N, Van Oudenaarden A, Mohammed S, Heck AJR, Clevers H (2012) The Lgr5 intestinal stem cell signature: robust expression of proposed quiescent ′ +4′ cell markers. EMBO J 31:3079–3091
Sangiorgi E, Capecchi MR (2008) Bmi1 is expressed in vivo in intestinal stem cells. Nat Genet 40:915–920
Montgomery RK, Carlone DL, Richmond CA, Farilla L, Kranendonk MEG, Henderson DE, Baffour-Awuah NY, Ambruzs DM, Fogli LK, Algra S, Breault DT (2011) Mouse telomerase reverse transcriptase (mTert) expression marks slowly cycling intestinal stem cells. Proc Natl Acad Sci USA 108:179–184
Takeda N, Jain R, LeBoeuf MR, Wang Q, Lu MM, Epstein JA (2011) Interconversion between intestinal stem cell populations in distinct niches. Science 334:1420–1424
Powell AE, Wang Y, Li Y, Poulin EJ, Means AL, Washington MK, Higginbotham JN, Juchheim A, Prasad N, Levy SE, Guo Y, Shyr Y, Aronow BJ, Haigis KM, Franklin JL, Coffey RJ (2012) The pan-ErbB negative regulator lrig1 is an intestinal stem cell marker that functions as a tumor suppressor. Cell 149:146–158
Yousefi M, Li N, Nakauka-Ddamba A, Wang S, Davidow K, Schoenberger J, Yu Z, Jensen ST, Kharas MG, Lengner CJ (2016) Msi RNA-binding proteins control reserve intestinal stem cell quiescence. J Cell Biol 215:401–413
Ayyaz A, Kumar S, Sangiorgi B, Ghoshal B, Gosio J, Ouladan S, Fink M, Barutcu S, Trcka D, Shen J, Chan K, Wrana JL, Gregorieff A (2019) Single-cell transcriptomes of the regenerating intestine reveal a revival stem cell. Nature 569:121–125
Seishima R, Barker N (2019) A contemporary snapshot of intestinal stem cells and their regulation. Differentiation 108:3–7
Sheahan BJ, Freeman AN, Keeley TM, Samuelson LC, Roper J, Hasapis S, Lee CL, Dekaney CM (2021) Epithelial regeneration after doxorubicin arises primarily from early progeny of active intestinal stem cells. Cmgh 12:119–140
Sato T, Vries RG, Snippert HJ, van de Wetering M, Barker N, Stange DE, van Es JH, Abo A, Kujala P, Peters PJ, Clevers H (2009) Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459:262–265
Powell DW, Pinchuk IV, Saada JI, Chen X, Mifflin RC (2011) Mesenchymal cells of the intestinal lamina propria. Annu Rev Physiol 73:213–237
McCarthy N, Kraiczy J, Shivdasani RA (2020) Cellular and molecular architecture of the intestinal stem cell niche. Nat Cell Biol 22:1033–1041
Pinchuk IV, Mifflin RC, Saada JI, Powell DW (2010) Intestinal mesenchymal cells. Curr Gastroenterol Rep 12:310–318
Spit M, Koo BK, Maurice MM (2018) Tales from the crypt: intestinal niche signals in tissue renewal, plasticity and cancer. Open Biol 8:180120
Sphyris N, Hodder MC, Sansom OJ (2021) Subversion of niche-signalling pathways in colorectal cancer: What makes and breaks the intestinal stem cell. Cancers (Basel) 13:1–55
L’Heureux A, Wieland DR, Weng CH, Chen YH, Lin CH, Lin TH, Weng CH (2019) Association between thyroid disorders and colorectal cancer risk in adult patients in Taiwan. JAMA Netw Open 2:e193755
Hellevik AI, Åsvold BO, Bjøro T, Romundstad PR, Nilsen TIL, Vatten LJ (2009) Thyroid function and cancer risk: a prospective population study. Cancer Epidemiol Biomarkers Prev 18:570–574
Brown AR, Simmen RCM, Simmen FA (2013) The role of thyroid hormone signaling in the prevention of digestive system cancers. Int J Mol Sci 14:16240–16257
Moeller LC, Führer D (2013) Thyroid hormone, thyroid hormone receptors, and cancer: A clinical perspective. Endocr Relat Cancer 20:R19–R29
Goemann IM, Romitti M, Meyer ELS, Wajner SM, Maia AL (2017) Role of thyroid hormones in the neoplastic process: an overview. Endocr Relat Cancer 24:R367–R385
Kress E, Skah S, Sirakov M, Nadjar J, Gadot N, Scoazec JY, Samarut J, Plateroti M (2010) Cooperation between the thyroid hormone receptor TRα1 and the WNT pathway in the induction of intestinal tumorigenesis. Gastroenterology 138:1863–1874
Uchuya-Castillo J, Aznar N, Frau C, Martinez P, Le Nevé C, Marisa L, Penalva LOF, Laurent-Puig P, Puisieux A, Scoazec JY, Samarut J, Ansieau S, Plateroti M (2018) Increased expression of the thyroid hormone nuclear receptor TRa1 characterizes intestinal tumors with high Wnt activity. Oncotarget 9:30979–30996
Lee YS, Chin YT, Yang YCSH, Wei PL, Wu HC, Shih A, Lu YT, Pedersen JZ, Incerpi S, Liu LF, Lin HY, Davis PJ (2016) The combination of tetraiodothyroacetic acid and cetuximab inhibits cell proliferation in colorectal cancers with different K-ras status. Steroids 111:63–70
Iishi H, Tatsuta M, Baba M, Okuda S, Taniguchi H (1992) Enhancement by thyroxine of experimental carcinogenesis induced in rat colon by azoxymethane. Int J Cancer 50:974–976
Rennert G, Rennert HS, Pinchev M, Gruber SB (2010) A case-control study of levothyroxine and the risk of colorectal cancer. JNCI J Natl Cancer Inst 102:568–572
Wändell P, Carlsson AC, Li X, Sundquist J, Sundquist K (2020) Levothyroxine treatment is associated with an increased relative risk of overall and organ specific incident cancers—a cohort study of the Swedish population. Cancer Epidemiol 66:101707
Boursi B, Haynes K, Mamtani R, Yang YX (2015) Thyroid dysfunction, thyroid hormone replacement and colorectal cancer risk. JNCI J Natl Cancer Inst 107:5
Sirakov M, Plateroti M (2011) The thyroid hormones and their nuclear receptors in the gut: from developmental biology to cancer. Biochim Biophys Acta Mol Basis Dis 1812:938–946
Kim WG, Cheng SY (2013) Thyroid hormone receptors and cancer. Biochim Biophys Acta Gen Subj 1830:3928–3936
Kowalik MA, Perra A, Pibiri M, Cocco MT, Samarut J, Plateroti M, Ledda-Columbano GM, Columbano A (2010) TRβ is the critical thyroid hormone receptor isoform in T3-induced proliferation of hepatocytes and pancreatic acinar cells. J Hepatol 53:686–692
Lin KH, Lin YW, Lee HF, Liu WL, Chen ST, Chang KSS, Cheng SY (1995) Increased invasive activity of human hepatocellular carcinoma cells is associated with an overexpression of thyroid hormone β1 nuclear receptor and low expression of the anti-metastatic nm23 gene. Cancer Lett 98:89–95
Markowitz S, Haut M, Stellato T, Gerbic C, Molkentin K (1989) Expression of the ErbA-beta class of thyroid hormone receptors is selectively lost in human colon carcinoma. J Clin Invest 84:1683–1687
Hörkkö TT, Tuppurainen K, George SM, Jernvall P, Karttunen TJ, Mäkinen MJ (2006) Thyroid hormone receptor β1 in normal colon and colorectal cancer–association with differentiation, polypoid growth type and K-ras mutations. Int J Cancer 118:1653–1659
Furuya F, Shimura H, Yamashita S, Endo T, Kobayashi T (2010) Liganded thyroid hormone receptor-α enhances proliferation of pancreatic β-cells. J Biol Chem 285:24477–24486
Heublein S, Mayr D, Meindl A, Angele M, Gallwas J, Jeschke U, Ditsch N (2015) Thyroid hormone receptors predict prognosis in BRCA1 associated breast cancer in opposing ways. PLoS ONE 10:e0127072
Sirakov M, Boussouar AA, Kress E, Frau C, Lone IN, Nadjar J, Angelov D, Plateroti M (2015) The thyroid hormone nuclear receptor TRα1 controls the Notch signaling pathway and cell fate in murine intestine. Dev 142:2764–2774
Kress E, Rezza A, Nadjar J, Samarut J, Plateroti M (2009) The frizzled-related sFRP2 gene is a target of thyroid hormone receptor α1 and activates β-catenin signaling in mouse intestine. J Biol Chem 284:1234–1241
Skah S, Uchuya-Castillo J, Sirakov M, Plateroti M (2017) The thyroid hormone nuclear receptors and the Wnt/β-catenin pathway: an intriguing liaison. Dev Biol 422:71–82
Plateroti M, Kress E, Mori JI, Samarut J (2006) Thyroid hormone receptor α1 directly controls transcription of the β-catenin gene in intestinal epithelial cells. Mol Cell Biol 26:3204–3214
Sirakov M, Claret L, Plateroti M (2021) Thyroid hormone nuclear receptor TRα1 and canonical WNT pathway cross-regulation in normal intestine and cancer. Front Endocrinol (Lausanne) 12:1653
Guinney J, Dienstmann R, Wang X, De Reyniès A, Schlicker A, Soneson C, Marisa L, Roepman P, Nyamundanda G, Angelino P, Bot BM, Morris JS, Simon IM, Gerster S, Fessler E, De Sousa E, Melo F, Missiaglia E, Ramay H, Barras D, Homicsko K, Maru D, Manyam GC, Broom B, Boige V, Perez-Villamil B, Laderas T, Salazar R, Gray JW, Hanahan D, Tabernero J, Bernards R, Friend SH, Laurent-Puig P, Medema JP, Sadanandam A, Wessels L, Delorenzi M, Kopetz S, Vermeulen L, Tejpar S (2015) The consensus molecular subtypes of colorectal cancer. Nat Med 21:1350–1356
Hasebe T, Buchholz DR, Shi YB, Ishizuya-Oka A (2011) Epithelial-connective tissue interactions induced by thyroid hormone receptor are essential for adult stem cell development in the Xenopus laevis intestine. Stem Cells 29:154–161
Shi YB (2021) Life without thyroid hormone receptor. Endocrinology 162:1–12
Wen L, Shi YB (2016) Regulation of growth rate and developmental timing by Xenopus thyroid hormone receptor α. Dev Growth Differ 58:106–115
Shibata Y, Tanizaki Y, Shi YB (2020) Thyroid hormone receptor beta is critical for intestinal remodeling during Xenopus tropicalis metamorphosis. Cell Biosci 10:1–15
Shibata Y, Tanizaki Y, Zhang H, Lee H, Dasso M, Shi YB (2021) Thyroid hormone receptor is essential for larval epithelial apoptosis and adult epithelial stem cell development but not adult intestinal morphogenesis during xenopus tropicalis metamorphosis. Cells 536(10):536
Tanizaki Y, Shibata Y, Zhang H, Shi Y-B (2021) Analysis of thyroid hormone receptor α-knockout tadpoles reveals that the activation of cell cycle program is involved in thyroid hormone-induced larval epithelial cell death and adult intestinal stem cell development during xenopus tropicalis. Metamorphosis 31:128–142
Shi YB, Brown DD (1993) The earliest changes in gene expression in tadpole intestine induced by thyroid hormone. J Biol Chem 268:20312–20317
Hasebe T, Kajita M, Fu L, Shi YB, Ishizuya-Oka A (2012) Thyroid hormone-induced sonic hedgehog signal up-regulates its own pathway in a paracrine manner in the Xenopus laevis intestine during metamorphosis. Dev Dyn 241:403–414
Ishizuya-Oka A, Ueda S, Amano T, Shimizu K, Suzuki K, Ueno N, Yoshizato K (2001) Thyroid-hormone-dependent and fibroblast-specific expression of BMP-4 correlates with adult epithelial development during amphibian intestinal remodeling. Cell Tissue Res 303:187–195
Ishizuya-Oka A, Hasebe T, Shimizu K, Suzuki K, Ueda S (2006) Shh/BMP-4 signaling pathway is essential for intestinal epithelial development during Xenopus larval-to-adult remodeling. Dev Dyn 235:3240–3249
Ishizuya-Oka A, Kajita M, Hasebe T (2014) Thyroid hormone-regulated wnt5a/ror2 signaling is essential for dedifferentiation of larval epithelial cells into adult stem cells in the xenopus laevis intestine. PLoS ONE 9:9
Hasebe T, Fujimoto K, Kajita M, Ishizuya-Oka A (2016) Thyroid hormone activates Wnt/β-catenin signaling involved in adult epithelial development during intestinal remodeling in Xenopus laevis. Cell Tissue Res 365:309–318
Ishizuya-Oka A (2017) How thyroid hormone regulates transformation of larval epithelial cells into adult stem cells in the amphibian intestine. Mol Cell Endocrinol 459:98–103
Kaestner KH (2019) The intestinal stem cell niche: a central role for Foxl1-expressing subepithelial telocytes. Cmgh 8:111–117
Kondo A, Kaestner KH (2019) Emerging diverse roles of telocytes. Development 146:14
Hasebe T, Fujimoto K, Ishizuya-Oka A (2020) Thyroid hormone-induced expression of Foxl1 in subepithelial fibroblasts correlates with adult stem cell development during Xenopus intestinal remodeling. Sci Rep 10:1–11
Hasebe T, Fujimoto K, Kajita M, Fu L, Shi YB, Ishizuya-Oka A (2017) Thyroid hormone-induced activation of notch signaling is required for adult intestinal stem cell development during xenopus laevis metamorphosis. Stem Cells 35:1028–1039
Ishizuya-Oka A, Shimizu K, Sakakibara SI, Okano H, Ueda S (2003) Thyroid hormone-upregulated expression of Musashi-1 is specific for progenitor cells of the adult epithelium during amphibian gastrointestinal remodeling. J Cell Sci 116:3157–3164
Salvatore D (2018) Deiodinases and stem cells: an intimate relationship. J Endocrinol Invest 41:59–66
Kojima Y, Kondo Y, Fujishita T, Mishiro-Sato E, Kajino-Sakamoto R, Taketo MM, Aoki M (2019) Stromal iodothyronine deiodinase 2 (DIO2) promotes the growth of intestinal tumors in ApcΔ716 mutant mice. Cancer Sci 110:2520–2528
Dentice M, Luongo C, Ambrosio R, Sibilio A, Casillo A, Iaccarino A, Troncone G, Fenzi G, Larsen PR, Salvatore D (2012) β-Catenin regulates deiodinase levels and thyroid hormone signaling in colon cancer cells. Gastroenterology 143:1037–1047
Catalano V, Dentice M, Ambrosio R, Luongo C, Carollo R, Benfante A, Todaro M, Stassi G, Salvatore D (2016) Activated thyroid hormone promotes differentiation and chemotherapeutic sensitization of colorectal cancer stem cells by regulating Wnt and BMP4 signaling. Cancer Res 76:137–1244
Dentice M, Marsili A, Zavacki A, Larsen PR, Salvatore D (2013) The deiodinases and the control of intracellular thyroid hormone signaling during cellular differentiation. Biochim Biophys Acta Gen Subj 1830:3937–3945
Ferrara SJ, Chaudhary P, DeBell MJ, Marracci G, Miller H, Calkins E, Pocius E, Napier BA, Emery B, Bourdette D, Scanlan TS (2021) TREM2 is thyroid hormone regulated making the TREM2 pathway druggable with ligands for thyroid hormone receptor. Cell Chem Biol 81:1097
De Luca R, Davis PJ, Lin HY, Gionfra F, Percario ZA, Affabris E, Pedersen JZ, Marchese C, Trivedi P, Anastasiadou E, Negro R, Incerpi S (2021) Thyroid hormones interaction with immune response, inflammation and non-thyroidal illness syndrome. Front Cell Dev Biol 8:1775
Lee YS, Chin YT, Shih YJ, Nana AW, Chen YR, Wu HC, Yang YCSH, Lin HY, Davis PJ (2018) Thyroid hormone promotes β-catenin activation and cell proliferation in colorectal cancer. Horm Cancer 9:156–165
Davis PJ, Leonard JL, Lin H-Y, Leinung M, Mousa SA (2018) Molecular basis of nongenomic actions of thyroid hormone. Vitamins and hormones. Academic Pres, Hoboken, pp 67–96
Anyetei-Anum CS, Roggero VR, Allison LA (2018) Thyroid hormone receptor localization in target tissues. J Endocrinol 237(19):34
Davis PJ, Goglia F, Leonard JL (2016) Nongenomic actions of thyroid hormone. Nat Rev Endocrinol 12:111–121
Schmohl KA, Mueller AM, Dohmann M, Spellerberg R, Urnauer S, Schwenk N, Ziegler SI, Bartenstein P, Nelson PJ, Spitzweg C (2019) Integrin αvβ3-mediated effects of thyroid hormones on mesenchymal stem cells in tumor angiogenesis. Thyroid 29:1843–1857
Schmohl KA, Müller AM, Nelson PJ, Spitzweg C (2020) Thyroid hormone effects on mesenchymal stem cell biology in the tumour microenvironment. Exp Clin Endocrinol Diabetes 128:462–468
Beyaz S, Mana MD, Roper J, Kedrin D, Saadatpour A, Hong SJ, Bauer-Rowe KE, Xifaras ME, Akkad A, Arias E, Pinello L, Katz Y, Shinagare S, Abu-Remaileh M, Mihaylova MM, Lamming DW, Dogum R, Guo G, Bell GW, Selig M, Nielsen GP, Gupta N, Ferrone CR, Deshpande V, Yuan GC, Orkin SH, Sabatini DM, Yilmaz ÖH (2016) High-fat diet enhances stemness and tumorigenicity of intestinal progenitors. Nature 531:53–58
Rodríguez-Colman MJ, Schewe M, Meerlo M, Stigter E, Gerrits J, Pras-Raves M, Sacchetti A, Hornsveld M, Oost KC, Snippert HJ, Verhoeven-Duif N, Fodde R, Burgering BMT (2017) Interplay between metabolic identities in the intestinal crypt supports stem cell function. Nature 543:424–427
Yilmaz ÖH, Katajisto P, Lamming DW, Gültekin Y, Bauer-Rowe KE, Sengupta S, Birsoy K, Dursun A, Onur Yilmaz V, Selig M, Nielsen GP, Mino-Kenudson M, Zukerberg LR, Bhan AK, Deshpande V, Sabatini DM (2012) MTORC1 in the paneth cell niche couples intestinal stem-cell function to calorie intake. Nature 486:490–495
Alonso S, Yilmaz ÖH (2018) Nutritional regulation of intestinal stem cells. Annu Rev Nutr 38:273–301
Calibasi-Kocal G, Mashinchian O, Basbinar Y, Ellidokuz E, Cheng CW, Yilmaz ÖH (2021) Nutritional control of intestinal stem cells in homeostasis and tumorigenesis. Trends Endocrinol Metab 32:20–35
Mullur R, Liu YY, Brent GA (2014) Thyroid hormone regulation of metabolism. Physiol Rev 94:355–382
Liu YY, Brent GA (2010) Thyroid hormone crosstalk with nuclear receptor signaling in metabolic regulation. Trends Endocrinol Metab 21:166–173
Yin L, Zhang Y, Bradley Hillgartner F (2002) Sterol regulatory element-binding protein-1 interacts with the nuclear thyroid hormone receptor to enhance acetyl-CoA carboxylase-α transcription in hepatocytes *. J Biol Chem 277:19554–19565
Shin DJ, Plateroti M, Samarut J, Osborne TF (2006) Two uniquely arranged thyroid hormone response elements in the far upstream 5′ flanking region confer direct thyroid hormone regulation to the murine cholesterol 7α hydroxylase gene. Nucleic Acids Res 34:3853–3861
Bertolio R, Napoletano F, Mano M, Maurer-Stroh S, Fantuz M, Zannini A, Bicciato S, Sorrentino G, Del Sal G (2019) Sterol regulatory element binding protein 1 couples mechanical cues and lipid metabolism. Nat Commun 101(10):1–11
Jumarie C, Malo C (1994) Alkaline phosphatase and peptidase activities in Caco-2 cells: differential response to triiodothyronine. Vitr Cell Dev Biol Anim 30:753–760
Hodin RA, Chamberlain SM, Upton MP (1992) Thyroid hormone differentially regulates rat intestinal brush border enzyme gene expression. Gastroenterology 103:1529–1536
Plateroti M, Chassande O, Fraichard A, Gauthier K, Freund JN, Samarut J, Kedinger M (1999) Involvement of T3Rα- and β-receptor subtypes in mediation of T3 functions during postnatal murine intestinal development. Gastroenterology 116:1367–1378
Ludikhuize MC, Meerlo M, Gallego MP, Xanthakis D, Burgaya Julià M, Nguyen NTB, Brombacher EC, Liv N, Maurice MM, Paik JH, Burgering BMT, Rodriguez-Colman MJ (2020) Mitochondria define intestinal stem cell differentiation downstream of a FOXO/Notch axis. Cell Metab 32:889-900.e7
Urbauer E, Rath E, Haller D (2021) Mitochondrial metabolism in the intestinal stem cell niche—sensing and signaling in health and disease. Front Cell Dev Biol 8:1520
Lesmana R, Sinha RA, Singh BK, Zhou J, Ohba K, Wu Y, Yau WW, Bay BH, Yen PM (2016) Thyroid hormone stimulation of autophagy is essential for mitochondrial biogenesis and activity in skeletal muscle. Endocrinology 157:23–38
Zhou J, Gauthier K, Ho JP, Lim A, Zhu XG, Han CR, Sinha RA, Cheng SY, Yen PM (2021) Thyroid hormone receptor α regulates autophagy, mitochondrial biogenesis, and fatty acid use in skeletal muscle. Endocrinol (US) 162:1–11
Na W, Fu L, Luu N, Shi YB (2020) Thyroid hormone directly activates mitochondrial fission process 1 (Mtfp1) gene transcription during adult intestinal stem cell development and proliferation in Xenopus tropicalis. Gen Comp Endocrinol 299:113590
Attia RR, Connnaughton S, Boone LR, Wang F, Elam MB, Ness GC, Cook GA, Park EA (2010) Regulation of pyruvate dehydrogenase kinase 4 (PDK4) by thyroid hormone role of the peroxisome proliferator-activated receptor γ coactivator (PGC-1α). J Biol Chem 285:2375–2385
Lin HY, Chin YT, Yang YCSH, Lai HY, Wang-Peng J, Liu LF, Tang HY, Davis PJ (2016) Thyroid hormone, cancer, and apoptosis. Compr Physiol 6:1221–1237
Bassett JHD, Harvey CB, Williams GR (2003) Mechanisms of thyroid hormone receptor-specific nuclear and extra nuclear actions. Mol Cell Endocrinol 2003:1–11
Matosin-Matekalo M, Mesonero JE, Laroche TJ, Lacasa M, Brot-Laroche E (1999) Glucose and thyroid hormone co-regulate the expression of the intestinal fructose transporter GLUT5. Biochem J 339:233–239
Choi J, Moskalik CL, Ng A, Matter SF, Buchholz DR (2015) Regulation of thyroid hormone-induced development in vivo by thyroid hormone transporters and cytosolic binding proteins. Gen Comp Endocrinol 222:69–80
Weinhofer I, Kunze M, Rampler H, Forss-Petter S, Samarut J, Plateroti M, Berger J (2008) Distinct modulatory roles for thyroid hormone receptors TRα and TRβ in SREBP1-activated ABCD2 expression. Eur J Cell Biol 87:933–945
Grøntved L, Waterfall JJ, Kim DW, Baek S, Sung MH, Zhao L, Won Park J, Nielsen R, Walker RL, Zhu YJ, Meltzer PS, Hager GL, Cheng SY (2015) Transcriptional activation by the thyroid hormone receptor through ligand-dependent receptor recruitment and chromatin remodelling. Nat Commun 6:1–11
Wang B, Rong X, Palladino END, Wang J, Fogelman AM, Martín MG, Alrefai WA, Ford DA, Tontonoz P (2018) Phospholipid remodeling and cholesterol availability regulate intestinal stemness and tumorigenesis. Cell Stem Cell 22:206-220.e4
Wen YA, Xiong X, Zaytseva YY, Napier DL, Vallee E, Li AT, Wang C, Weiss HL, Evers BM, Gao T (2018) Downregulation of SREBP inhibits tumor growth and initiation by altering cellular metabolism in colon cancer article. Cell Death Dis 9:3
Zhang Y, Ma K, Song S, Elam MB, Cook GA, Park EA (2004) Peroxisomal proliferator-activated receptor-γ coactivator-1α (PGC-1α) enhances the thyroid hormone induction of carnitine palmitoyltransferase I (CPT-Iα). J Biol Chem 279:53963–53971
Jackson-Hayes L, Song S, Lavrentyev EN, Jansen MS, Hillgartner FB, Tian L, Wood PA, Cook GA, Park EA (2003) A thyroid hormone response unit formed between the promoter and first intron of the carnitine palmitoyltransferase-Iα gene mediates the liver-specific induction by thyroid hormone. J Biol Chem 278:7964–7972
Sayre NL, Lechleiter JD (2012) Fatty acid metabolism and thyroid hormones. Curr trends Endocrinol 6:65–76
Sandra Chocron E, Sayre NL, Holstein D, Saelim N, Ibdah JA, Dong LQ, Zhu X, Cheng SY, Lechleiter JD (2012) The trifunctional protein mediates thyroid hormone receptor-dependent stimulation of mitochondria metabolism. Mol Endocrinol 26:1117–1128
Mihaylova MM, Cheng CW, Cao AQ, Tripathi S, Mana MD, Bauer-Rowe KE, Abu-Remaileh M, Clavain L, Erdemir A, Lewis CA, Freinkman E, Dickey AS, La Spada AR, Huang Y, Bell GW, Deshpande V, Carmeliet P, Katajisto P, Sabatini DM, Yilmaz ÖH (2018) Fasting activates fatty acid oxidation to enhance intestinal stem cell function during homeostasis and aging. Cell Stem Cell 22:769-778.e4
Yousefi M, Nakauka-Ddamba A, Berry CT, Li N, Schoenberger J, Simeonov KP, Cedeno RJ, Yu Z, Lengner CJ (2018) Calorie restriction governs intestinal epithelial regeneration through cell-autonomous regulation of mTORC1 in reserve stem cells. Stem Cell Rep 10:703–711
Kress E, Rezza A, Nadjar J, Samarut J, Plateroti M (2008) The thyroid hormone receptor-α (TRα) gene encoding TRα1 controls deoxyribonucleic acid damage-induced tissue repair. Mol Endocrinol 22:47–55
Mana MD, Hussey AM, Tzouanas CN, Imada S, Barrera Millan Y, Bahceci D, Saiz DR, Webb AT, Lewis CA, Carmeliet P, Mihaylova MM, Shalek AK, Yilmaz ÖH (2021) High-fat diet-activated fatty acid oxidation mediates intestinal stemness and tumorigenicity. Cell Rep 35:109211
Xiong X, Wen Y-AA, Fairchild R, Zaytseva YY, Weiss HL, Evers BM, Gao T (2020) Upregulation of CPT1A is essential for the tumor-promoting effect of adipocytes in colon cáncer. Cell Death Dis 11:9
Lu C, Cheng SY (2010) Thyroid hormone receptors regulate adipogenesis and carcinogenesis via crosstalk signaling with peroxisome proliferator-activated receptors. J Mol Endocrinol 44:143–154
Boon R, Silveira GG, Mostoslavsky R (2020) Nuclear metabolism and the regulation of the epigenome. Nat Metab 2:1190–1203
Fu L, Yin J, Shi YB (2019) Involvement of epigenetic modifications in thyroid hormone-dependent formation of adult intestinal stem cells during amphibian metamorphosis. Gen Comp Endocrinol 271:91–96
Gillis NE, Boyd JR, Tomczak JA, Frietze S, Carr FE (2022) Thyroid hormone dependent transcriptional programming by TRβ requires SWI/SNF chromatin remodelers. Nucleic Acids Res 50:1382–1395
Lee KC, Li J, Cole PA, Wong J, Kraus WL (2003) Transcriptional activation by thyroid hormone receptor-beta involves chromatin remodeling, histone acetylation, and synergistic stimulation by p300 and steroid receptor coactivators. Mol Endocrinol 17:908–922
Huang ZQ, Li J, Sachs LM, Cole PA, Wong J (2003) A role for cofactor-cofactor and cofactor-histone interactions in targeting p300, SWI/SNF and Mediator for transcription. EMBO J 22:2146–2155
Hasebe T, Fujimoto K, Buchholz DR, Ishizuya-Oka A (2020) Stem cell development involves divergent thyroid hormone receptor subtype expression and epigenetic modifications in the Xenopus metamorphosing intestine. Gen Comp Endocrinol 292:113441
Anselmo J, Chaves CM (2020) Physiologic significance of epigenetic regulation of thyroid hormone target gene expression. Eur Thyroid J 9:114–123
Fang Y, Tang S, Li X (2019) Sirtuins in metabolic and epigenetic regulation of stem cells. Trends Endocrinol Metab 30:177–188
Cordeiro A, Lopes de Souza L, Soares Oliveira L, Costa Faustino L, Aragão Santiago L, Fonseca Bloise F, Ortiga-Carvalho TM, Dos Santos Almeida NA, Cabanelas Pazos-Moura C (2013) Thyroid hormone regulation of Sirtuin 1 expression and implications to integrated responses in fasted mice. J Endocrinol 216:181–193
Suh JH, Sieglaff DH, Zhang A, Xia X, Cvoro A, Winnier GE, Webb P (2013) SIRT1 is a direct coactivator of thyroid hormone receptor β1 with gene-specific actions. PLoS ONE 8:e70097
Funding
MP lab is supported by the FRM (Equipes FRM 2018, DEQ20181039598), by the Inca (PLBIO19-289) and by the Ligue Contre le Cancer, Département Grand Est (01X.2020 and 01AE.2021). MVG received support from the FRM.
Author information
Authors and Affiliations
Contributions
MVG and MP shared the responsibility to write the paper.
Corresponding author
Ethics declarations
Conflict of interests
The authors have no competing interests.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Giolito, M.V., Plateroti, M. Thyroid hormone signaling in the intestinal stem cells and their niche. Cell. Mol. Life Sci. 79, 476 (2022). https://doi.org/10.1007/s00018-022-04503-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04503-y