Abstract
Little consensus exists on the role of food partitioning in the organization of tadpole assemblages. We studied trophic ecology of syntopic tadpoles through the analysis of gut contents, selectivity, and stable isotopes to assess the occurrence of food partitioning in tadpole assemblages. Tadpoles (n = 194) were collected in three wetlands and corresponded to four species: Elachistocleis bicolor (Eb), Scinax nasicus (Sn), Physalaemus albonotatus (Pa), and Dendropsophus sp. (D); and belonged to four ecomorphological groups (EMGs): suspension feeders (Eb), nektonic (Sn), benthic (Pa) and macrophagous (D). Sn, and Pa showed low selective diet and a wider trophic spectrum than Eb and D, which mainly consumed one or two food categories. Diet overlap was higher between Sn and Pa. Still, Sn and Pa presented some differences in the food resources consumed. Stable isotopes analysis showed that Eb, Sn, and Pa had a lower trophic position than D, explained by the high contribution of animal food oligochaete in D diet, in contrast to the importance of algae in the diet of Eb, Sn, and Pa. Diet specialization of some species, combined with the low dietary and isotopic overlap among the ecomorphological groups, suggests that trophic partitioning facilitates coexistence of syntopic tadpoles.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Anuran tadpoles are found in a variety of freshwater habitats in which different species can coexist (Lajmanovich, 2000; Altig et al., 2007). Tadpoles are transitory consumers and, in the absence of fish, their density is usually high enough to affect primary production, flux of nutrients, and competitive interactions (Seale & Beckvar, 1980; Waringer-Löschenkohl & Schagerl, 2001). Thus, understanding their feeding ecology is essential to disentangling the energy and matter pathways within freshwater systems as well as between water and land (Altig et al., 2007).
Historically, tadpoles have been considered as predominantly non-selective phytophagous (Heyer, 1973; Duellman & Trueb, 1986), exhibiting a narrow differentiation in the trophic niche. However, later studies highlighted the importance of the trophic selectivity of some species (Kupferberg, 1997; Sabagh et al., 2012) and of the animal food in the diet (de Rossa-Feres et al., 2004; Ghioca-Robrecht & Smith, 2010; Asrafuzzaman et al., 2018). Gaps in the knowledge of the trophic ecology of most species still exist because, for example, few studies have analyzed food selection considering food availability (Diaz-Paniagua, 1985; Huckembeck et al., 2014; Vassilieva et al., 2017; Kloh et al., 2019), a limitation that could lead to erroneous interpretations of trophic ecology (Maneyro & da Rosa, 2004; López et al. 2009).
Traditionally, studies on trophic ecology have been based only on the analysis of gut contents, inferring ecological differences among species from a snapshot of their diet (i.e., resources ingested at a given time). Instead, stable isotope analyses allow the study of diet in a wider temporal window than the analysis of gut contents because stable isotope signatures reflect the resources assimilated in the consumer tissue (Post, 2002; Fry, 2007; Schiesari et al., 2009; Schalk et al., 2014; Dalu et al., 2015). Stable isotope analyses have shown that trophic position of tadpoles can vary from primary consumers to relatively high trophic levels (Vander Zanden & Rasmussen, 1999; Schiesari et al., 2009). Ecomorphological analysis also represents a useful approach for studying resource use and niche overlap (e.g., Pianka, 1986; Ricklefs & Miles, 1994; Quiroga et al., 2018; Vassilieva et al., 2017). Tadpoles’ morphology may reflect different feeding habits (Wassersug, 1980; Altig & Johnston, 1989; Vera Candioti, 2007), thereby providing insights on the degree of trophic overlap between species.
At present, little consensus exists regarding the occurrence of food partitioning and its role in the organization of tadpole assemblages (de Rossa-Feres et al., 2004). Some studies have suggested that trophic segregation is not important to tadpole coexistence (Heyer, 1974; Pavignano, 1990; Lajmanovich, 2000), while other more recent and isotopic-based studies have highlighted the role of trophic segregation to explain the coexistence of different species (e.g., Hunte-Brown, 2006; Verburg et al., 2007; Schiesari et al., 2009; Vassilieva et al., 2017). Thus, a more detailed and precise research on trophic ecology of tadpole assemblages is necessary to determine the importance of trophic segregation in organization of tadpole assemblages (Altig et al., 2007).
Here, we study trophic ecology and mechanisms of coexistence of tadpoles of four species, belonging to different ecomorphological groups (Altig & Johnston, 1989), that co-occur in temporal floodplain wetlands of the Paraná River. We combined different analytical approaches: (a) gut content analysis; (b) field surveys of food availability to estimate food selectivity; and (c) nitrogen and carbone stable isotopes ratios to determine trophic position. We expect trophic niche partitioning among species, evidencing that trophic segregation is an important mechanism in the structuring of the assemblages of tadpoles.
Materials and methods
Field sampling
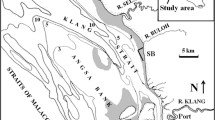
We selected three temporary marshes of the floodplain of the Middle Paraná River, near Santa Fe city, Argentina (site 1: 31° 37′ 28″ S–60° 37′ 23″ W; site 2: 31° 37′ 35″ S–60° 36′ 51″ W; site 3: 31° 38′ 29″ S–60° 40′ 22″ W). Sites area varied between 2000 and 7300 m2 and they were covered by emergent macrophytes (Ludwigia sp., Panicum sp., Echinochloa sp. and Paspalum sp.). The climate of the area is temperate-subtropical, the mean air temperature is 19 °C, and the annual rainfall is approximately 1000 mm, occurring mainly from October to April (Rojas & Saluso, 1987).
Sites were sampled during November 2011 and November 2014 collecting tadpoles in stages 38 to 40 sensu Gosner (1960) at 25 to 45 cm depth using a dip net. Collected tadpoles (n = 194) were kept in ice to later analysis in laboratory.
Availability of food resources was sampled simultaneously to collection of tadpoles. In each site, food resources were sampled by triplicate. Phytoplankton was sampled using a 100-ml bottle. Adhered algae was sampled with the scraping of a pool of three submerged leaves of Ludwigia sp. (Marker & Bolas, 1984, leaves of similar sizes were selected), a plant species present in the three sites. Zooplankton was sampled by filtering 6 L of wetland water through a net 50-µm mesh (Paggi et al., 2001). Samples of sedimentary organic matter were collected with a Core-type tubular sampler 8 cm in diameter (Anderson et al., 2013).
Samples for isotopic analysis were also taken simultaneously to tadpoles’ collection. Measuring the trophic position of consumers requires the estimation of an isotopic baseline; thus, we sampled basal organisms according to the most consumed food categories for each tadpole species. For the adhered algae, we used artificial substrate samplers (green Christmas garlands), which were placed one month before the collection of tadpoles. Sedimentary organic matter and zooplankton were sampled such during food resource sampling as described above. Benthonic invertebrates were collected with a Core sampler (8 cm diameter) that was driven about 15 cm into the bottom sediment (Batzer & Resh, 1992).
Laboratory
Collected tadpoles (n = 194) were identified and assigned to ecomorphological groups (EMGs; Fig. 1) following Altig and Johnston (1989): Elachistocleis bicolor (Guérin-Méneville, 1838) (Microhylidae, suspension feeder EMG, n = 47 tadpoles), Scinax nasicus (Cope, 1862) (Hylidae, nektonic EMG, n = 49), Physalaemus albonotatus (Steindachner, 1864) (Leptodactylidae, benthic EMG, n = 52 tadpoles), and Dendropsophus sp. (Hylidae, macrophagous EMG, n = 46 tadpoles).
Assignation of talpoles to ecomorphological groups (EMGs) following Altig and Johnston (1989). Species oral disk and body side view: aE. bicolor (suspension feeder EMG); bS. nasicus (nektonic EMG); cP. albonotatus (benthic EMG); dDendropsophus sp. (macrophagous EMG)
We analyzed between 45 and 50 tadpoles per species (14–17 tadpoles per site) to study diet through gut contents. For diet analysis, we extracted the digestive tract of each tadpole and the contents were analyzed and identified under an inverted microscope at 400 × (Nikon TS100 Eclipse). Phytoplankton and adhered algae were classified according to seven morphologically based functional groups (MBFG; Kruk et al., 2010; see Table 1 of Appendix for details): Small algae (MBFG1; up to 134 μm2); Algae with silica (MBFG2); Filamentous algae (MBFG3); Medium-size algae (MBFG4; up 791 μm2); Flagellated unicellular algae (MBFG5); Diatomean (MBFG6); and Colonial with mucilage (MBFG7; up to 791 μm2). Food items with an advanced degree of digestion, but still with identifiable structures, such as pieces of chitinous exoskeleton and locomotor appendages, were classified as animal remains. Fragments of vegetation were classified as plant remains (Huckembeck et al., 2014). Unidentified items, which formed a mass of organic material, were classified as remaining sedimentary organic matter (i.e., detritus) (Asrafuzzaman et al., 2018).
For food availability, we estimated relative abundance of each food category in the environment. Phytoplankton and adhered algae were classified according to seven morphologically based functional groups as in diet analysis and were quantified using an inverted microscope following the Utermöhl (1958) technique. Zooplankton and sedimentary organic matter were quantified under a binocular microscope (Molina et al., 2010).
For stable isotopes analysis, we extracted tail muscle tissue from tadpoles collected in sites 1 and 2 (Verburg et al., 2007; Dalu et al., 2015). Ten 1 mg dry weight samples per species were prepared, coming each sample from the tale of one to three tadpoles, depending on amount of tissue in specimens. Among the food resources available in the environment, six 1 mg dry weight samples of each food item (phytoplankton, adhered algae, sedimentary organic matter, cladocerans and oligochaetes) were prepared. We considered only cladocerans of zooplankton and only oligochaetes of benthonic invertebrates because these were the only consumed items within these communities.
Analyses of stable isotopes were performed in a mass spectrometer (IRMS Finnigan MATDelta S) coupled to an elemental analyser (CATNAS, Facultad de Agronomía, Universidad de la República, Uruguay). The ratio of stable isotopes was expressed by convention in delta (δ) notation: δX = {(Rsample/Rstandard) − 1} × 1000, where δX is the isotope ratio of the sample relative to a standard, Rsample and Rstandard are the fractions of heavy to light isotopes in the sample and the standard, respectively. The standard values for δ15N were atmospheric Nitrogen and for δ13C was Pee Dee Belemnite. As high lipid levels (indicated by a high C:N ratio) may drive δ13C values in a negative direction (Matthews & Mazumder, 2005), when C:N ratios were higher than 3.5, the δ13C values were normalized using the following equation: δ13Ccorrected = δ13C − 3.32 + 0.99 × C:N (Post et al., 2007).
Data analyses
Collected tadpoles among the three sampled sites were combined for data analysis. The Index of Relative Importance (IRI%) proposed by Pinkas et al. (1971) was used to determine the importance of each food category in the diet of each species. This index combines abundance, frequency of occurrence and volume in a single estimator of relative importance of each food category in the diet (Hart & Christensen, 2002). Volume of ingested items was calculated by approximation to regular geometric shapes. When we found chaeta of oligochaetes in tadpoles’ guts, we considered these as equivalent to only one individual for data analysis.
An overlap index (Pianka, 1973) was calculated to assess differences (and similarities) of diet among species. We tested whether overlap indices were different from those expected by chance using the RA2 Overlap Randomization Algorithm through the EcoSimR package in R (R Core Team, 2016) with IRI% values.
A Permutational Multivariate Analysis (PERMANOVA) was performed to assess whether the composition of the diet of the tadpoles (i.e., relative abundance of each food category) differed among the species. To assess these differences (and similarities) among species graphically, we used a Non-metric Multidimensional Scaling (NMDS). The NMDS ordination was evaluated by the coefficient of determination based on stress (R2; Oksanen, 2011). The matrix of food relative abundances to PERMANOVA and NMDS was double standardized through the Wisconsin method, where food categories are first standardized by maxima category and then by total categories (Oksanen, 2011). The trophic dissimilarity matrix was based on the Bray–Curtis index. The PERMANOVA and the NMDS ordination were created using the ‘adonis’ and ‘metaMDS’ functions, respectively, of the vegan package for R (Oksanen et al., 2015; R Core Team, 2016). In addition, we performed an Indicator Species Analysis (ISA) to determine which food categories were particularly characteristic (indicative) of each tadpole species. This analysis was performed using the indicspecies package in R (Cáceres & Legendre, 2009; R Core Team, 2016).
We tested for selectivity of tadpoles on the more important food categories by comparing their relative abundances between guts contents and the environment using a two-sample permutation test through the perm package in R (Fay, 2010; R Core Team, 2016). Feeding selectivity was calculated as the linear index of food selection (Li) proposed by Strauss (1979). Li ranges from − 1 to +1. Li = 0 indicates a lack of selection (i.e., consumption of the category in the same proportion as it occurs in the environment); Li < 0 indicates negative selection, avoidance or inaccessibility (i.e., less of food category i occurs in the diet than expected from random feeding); and Li > 0 indicates positive selection or preference (more of food category i occurs in the diet than expected from its environmental abundance).
We assessed the differences in isotopic signatures among the tadpoles by a Kruskall-Wallis test. The baseline for each species was selected according to their most important food categories: phytoplankton and sedimentary organic matter for E. bicolor, phytoplankton and algae adhered for S. nasicus and P. albonotatus, and oligochaete for Dendropsophus sp. To estimate the trophic position of Dendropsophus sp. we used a single-end member equation: TPconsumer = {(δ15Nconsumer − δ15Nbase) − TPbase}/TEF, where TPconsumer is the trophic position of the tadpole and TPbase is the trophic position of the baseline (in this case, 1). For the other species, we applied a two-end member model proposed by Post (2002): FCL = (δ15Nconsumer − (δ15Nbase1 × α + \({{\delta }}^{15}{\text N_{\text{base2}}}\) × (1 − α)))/TEF, and α = (δ13Cconsumer − δ13Cbase1)/(δ13Cbase1 − δ13Cbase2), where TEF is the trophic enrichment factor for δ15N. We set the TEF as 2.22, which is the mean value reported for tadpoles (Schiesari et al., 2009; Caut et al., 2013; San Sebastián et al., 2015).
Results
Diet of E. bicolor was composed mainly of unicellular algae (Table 1), mostly represented by Chlorella sp. (IRI = 47.25%), and Cladocera, represented by individuals of the family Chydoridae (IRI = 25.78%). Diet of S. nasicus was composed of filamentous algae of the genus Oedogonium sp. (IRI = 39.56%), Cladocera (IRI = 23.56%), unicellular algae (IRI = 11.80%) and medium-size algae (IRI = 10.64%). Diet of P. albonotatus was composed mainly of Cladocera (IRI = 33.19%), sedimentary organic matter (IRI = 19.33%), diatoms (IRI = 16.01%) and filamentous algae (IRI = 14.03%). Diet of Dendropsophus sp. was mainly composed of Oligochaeta (IRI = 60.97%).
Diet composition differed among species (PERMANOVA: F3, 189 = 101.01, P < 0.001, R2 = 0. 62). NMDS showed a greater separation in the diet between Dendropsophus sp. from the rest of the species along the first dimension, related to Dendropsophus sp. predation upon Oligochaeta (Fig. 2). A different proportion of fine-particulate sedimentary organic matter and unicellular algae in the diet of E. bicolor in relation to S. nasicus and P. albonotatus accounted for the separation between these species in the second dimension (Fig. 2). Oligochaeta were more abundant and/or frequent in the diet of Dendropsophus sp. (ISA: P = 0.005); sedimentary organic matter and algae with silica were more important in the diet of P. albonotatus (ISA: P < 0.01); sedimentary organic matter was more abundant and/or frequent in the diet of E. bicolor (ISA: P < 0.05); and plant remains and Nematoda were more common in the diet of P. albonotatus and S. nasicus in relation to Dendropsophus sp. and E. bicolor (ISA: P < 0.05). Moreover, all categories of food (unicellular algae, medium-size algae, diatomean, colonial with mucilage Protozoa and Rotifera) were more abundant and frequent in P. albonotatus, E. bicolor and S. nasicus than in of Dendropsophus sp. (ISA: P < 0.001).
Trophic overlap was less than expected by chance among Dendropsophus sp. and the rest of the species and between S. nasicus and P. albonotatus, and was not different than expected by chance between E. bicolor and S. nasicus and between E. bicolor and P. albonotatus (Table 2).
Elachistocleis bicolor selectively fed on unicellular algae, while the consumption of Cladocera was proportional to the environmental availability (Fig. 3A). Scinax nasicus avoided the medium-size algae and P. albonotatus selectively fed on diatomean (Fig. 3B and 3C). The selection of Oligochaeta by Dendropsophus sp. was not measured because of the difficulty mentioned in estimating the number of worms ingested (see methods section).
Strauss’s linear index of selectivity (Li) for E. bicolor (A), S. nasicus (B), P. albonotatus (C) and Dendropsophus sp (D), applied to the relative abundance of trophic items. Li > 0: positive selection, Li < 0: negative selection, Li = 0 absence of selectivity. Asterisk (*) indicates statistical difference (P < 0.05) from zero (two-sample permutation tests, two-tailed)
The isotopic signature of δ13C varied among species (KW = 27.5; P < 0.001), with differences between E. bicolor and Dendropsophus sp. (Dunn: P = 0.006), P. albonotatus and Dendropsophus sp. (Dunn: P = 0.021), P. albonotatus and E. bicolor (Dunn: P < 0.001), S. nasicus and Dendropsophus sp. (Dunn: P = 0.021), S. nasicus and E. bicolor (Dunn: P < 0.001). The isotopic signature of δ15N also varied among species (KW = 8.45, P < 0.037), with differences between P. albonotatus and Dendropsophus sp. (Dunn: P < 0.036), S. nasicus and Dendropsophus sp. (Dunn: P < 0.002) (Table 3). E. bicolor, S. nasicus and P. albonotatus had a lower trophic position than Dendropsophus sp. (Fig. 4).
Discussion
Our results show that co-occurring tadpoles of E. bicolor, S. nasicus, P. albonotatus, and Dendropsophus sp. exploit different food resources in the floodplain wetlands of the Paraná River, selecting and feeding upon different aquatic communities. These results reinforce the conclusions of other studies that have also found differences in the diet among tadpoles of syntopic species (e.g., de Rossa-Feres et al., 2004; Pollo et al., 2015; Santos et al., 2015; Vassilieva et al., 2017; Schmidt et al., 2017), suggesting that trophic segregation may represent a mechanism of coexistence in tadpole assemblages.
Tadpoles of the four species differed in diet composition, and these differences reflected the ecomorphological differences among them. Dendropsophus sp. showed the greatest diet differences. The elevated trophic segregation of Dendropsophus sp. seems to be a characteristic extended throughout its wide distribution (de Rossa-Feres et al., 2004). Dendropsophus sp. can ingest larger food items than any other syntopic species because of its macrophagous buccal structure, allowing ingestion of large preys by suction, such as benthos oligochaetes as long as tadpoles size (Vera Candioti, 2007). The branchial basket is relatively reduced in some genus Dendropsophus (Vera Candioti, 2007), limiting the ingestion of very small items by filtration. In contrast, the highly developed branchial basket of the suspension feeder tadpoles of E. bicolor (Vera Candioti, 2007), likely allows for filter feeding on very small items such as the microalgae (≈ 20μm). Similarly, Echeverría et al., (2007) reported the consumption of relatively small items such as desmid planktonic algae (medium-size 50 μm) in tadpoles of E. bicolor. However, the highly developed branchial basket of E. bicolor not necessarily precludes from ingesting large prey, since we also found cladocerans of ≈ 250 μm in their gut contents, as also reported Vera Candioti (2007). Similarly, the consume of relatively large prey such as zooplankton, insect larvae, and tadpoles was observed in other species with suspension feeder tadpoles as in genus Rhinophrynus (Starrett, 1960; Wassersug, 1972).
The diets of the tadpoles of S. nasicus and P. albonotatus were more diverse in comparison with that of Dendropsophus sp. and E. bicolor. Diet overlap between S. nasicus and P. albonotatus was the higher within the assemblage. Tadpoles of these two species have a similar configuration of the hyobranchial apparatus, with around 50% of the total area occupied by the branchial basket (Vera Candioti, 2007), and a similar buccal apparatus adapted to scrape surfaces. This allows them to consume adhered algae (mainly filamentous algae), as it has also been found in other species included within the benthic and nektonic ecomorphological groups (de Rossa-Feres et al., 2004; Vera Candioti et al., 2004; de Sousa Filho et al., 2007). We found that tadpoles of both species prey on microcrustaceans (cladocerans and rotiferans), as it has been reported in previous studies for tadpoles of these species and other congeneric species (de Rossa-Feres et al., 2004, de Sousa Filho et al., 2007, Vera Candioti (2007). Beyond these similarities, S. nasicus and P. albonotatus also presented some differences in their diets. Physalaemus albonotatus consumed a higher proportion of sedimentary organic matter and a lower proportion of filamentous algae than S. nasicus. The higher importance of sedimentary organic matter in the diet of P. albonotatus could be related to benthic habits of their tadpoles, in contrast to more nektonic habits of tadpoles of S. nasicus (Peltzer & Lajmanovich, 2004). Pollo et al. (2015) also studied diet of nektonic and benthic tadpoles and suggested that differences in diet where related to behavioral differences for food acquisition in the water column between species. An alternative – or complementary – explanation to the lower consumption of filamentous algae by P. albonotatus, is the lower number of labial teeth than in S. nasicus (Kehr & Duré, 1995; Kehr et al., 2004; Vera Candioti 2007). Studies have shown that the efficiency in the consume of adhered algae is lower in tadpoles with a lower number of labial teeth (Venesky et al., 2010). Thus, subtle differences in morphology and microhabitat exploitation contributes diet segregation (Kehr & Duré, 1995; Kehr et al., 2004; Peltzer & Lajmanovich, 2004; Vera Candioti 2007), even for tadpoles inhabiting the same shallow pond where the water column is less than half a meter deep.
The degree to which tadpoles of different species select their food or not remains little known. Both, selectivity (Johnson 1991; Kupferberg, 1997) and non-selectivity (Seale & Beckvar, 1980; Chen et al., 2008), have been reported for feeding of tadpoles of different species, although the number of studies is limited, in part because of the difficulty of assessing in detail environmental food availability. We found a selectivity gradient among studied species. Tadpoles of the nektonic and benthic ecomorphological groups, S. nasicus and P. albonotatus, showed a wider trophic spectrum and did not select their most important food categories. In contrast tadpoles of the suspension feeder and macrophagous ecomorphological groups, E. bicolor and Dendropsophus sp. respectively, could be considered specialized consumers, because they largely and selectively consumed one or two food categories, which accounted for more than 80% of their diet: small unicellular algae in the case of E. bicolor and oligochaetes in Dendropsophus sp.
A less studied approach to understanding the trophic ecology of tadpoles’ assemblages is the analysis of stable carbon and nitrogen isotopes. In recent studies, authors have suggested different positions in food webs for tadpoles (Verburg et al., 2007; Huckembeck et al., 2014; Schmidt et al., 2017). In accordance with these suggestions, we found a low trophic position for E. bicolor, S. nasicus and P. albonotatus tadpoles and a higher trophic position for Dendropsophus sp. Thus, our results show that suspension feeder, nektonic and benthic tadpoles, are consumers of the first levels of the food web, while microphagous tadpoles have a higher trophic position, one level avobe in the food web.
Schalk et al. (2017) found differences between the trophic position of S. nasicus and P. albonotatus, assigning a higher trophic position to S. nasicus (TP = 5.7) than to P. albonotatus (TP = 3). Differences with our results could be due to differences in food resources availability together with the trophic plasticity of tadpoles (López et al., 2015). The selection of different sources of carbon and nitrogen for isotopic analysis by Schalk et al. (2017) could also account for the differences with our study. In both species, we found a broad trophic spectrum composed by algae and, in a lower proportion by zooplankton. The low consumption of animal prey would not be enough to increase their trophic position. In relation to E. bicolor, its narrow trophic spectrum focused in the consumption of microalgae explain this species low trophic position in the food web. Although S. nasicus, P. albonotatus and E. bicolor are at the lowest trophic level within consumers of the food web, our multi-approaches strategy to the study of the trophic ecology of the assembly allowed us to recognize trophic segregation among these species, indicating that the three fulfil different functions in the ecosystem. Lastly, the predation on oligochaete by Dendropsophus sp., the largest prey registered for the hole tadpoles assemblage, allows to the macrophagous tadpoles the incorporation of a great amount of energy per prey and the access to nutrients not available to other syntopic tadpoles, and explain its higher trophic position in the food web.
Conclusion
Our integrative approach provided a more in-depth view of the trophic ecology of neotropical tadpoles assemblages. Using a combination of techniques and a detailed analysis of food categories, we found a relatively high specialization of the diet among tadpoles of different species, showing the importance of using an integrative approach to study the trophic ecology. Our results suggest that the partitioning of the trophic niche is an important mechanism for the segregation and coexistence of tadpoles in subtropical floodplain wetlands. Finally, the absence of ecological redundancy among neotropical tadpoles assemblage highlights the importance of conserving the diversity of species to maintain the identity and balance of aquatic ecosystems they inhabit.
References
Anderson, J. T., F. Zilli, L. Montalto, M. Marchese, M. McKinney & Y. Lak Park, 2013. Sampling of aquatic and terrestrial invertebrates in wetlands. In Anderson, J. T. & C. A. Davis (eds), Wetland Techniques. Organisms. Springer Science+Business Media, Dordrecht: 143–195.
Altig, R. & G. F. Johnston, 1989. Guilds of anuran larvae: relationships among developmental modes, morphologies, and habitats. Herpetological Monographs 3: 81.
Altig, R., M. R. Whiles & C. L. Taylor, 2007. What do tadpoles really eat? Assessing the trophic status of an understudied and imperiled group of consumers in freshwater habitats. Freshwater Biology 52: 386–395.
Asrafuzzaman, S., S. Mahapatra, J. Rout & G. Sahoo, 2018. Dietary assessment of five species of anuran tadpoles from northern Odisha, India. Journal of Threatened Taxa 10: 12382–12388.
Batzer, D. P., & V. H. Resh, 1992. Macroinvertebrates of a California seasonal wetland and responses to experimental habitat manipulation. Wetlands 12: 1–7.
Cáceres, M. D. & P. Legendre, 2009. Associations between species and groups of sites: indices and statistical inference. Ecology 90: 3566–3574.
Caut, S., E. Angulo, C. Díaz-Paniagua & I. Gomez-Mestre, 2013. Plastic changes in tadpole trophic ecology revealed by stable isotope analysis. Oecologia 173: 95–105.
Chen, H. C., B. C. Lai, G. M. Fellers, W. L. Wang & Y. C. Kam, 2008. Diet and foraging of Rana sauteri and Bufo bankorensis tadpoles in subtropical Taiwanese streams. Zoological Studies 47: 685–696.
Dalu, T., O. L. F. Weyl, P. W. Froneman & R. J. Wasserman, 2015. Trophic interactions in an austral temperate ephemeral pond inferred using stable isotope analysis. Hydrobiologia 768: 81–94.
de Rossa-Feres, D., C. J. Jim & M. G. Fonseca, 2004. Diets of tadpoles from a temporary pond in southeastern Brazil (Amphibia, Anura). Revista Brasileira de Zoologia 21: 745–754.
de Sousa Filho, I. F., C. C. Branco, A. M. Carvalho-e-Silva, G. R. da Silva & L. T. Sabagh, 2007. The diet of Scinax angrensis (Lutz) tadpoles in an area of the Atlantic Forest (Mangaratiba, Rio de Janeiro) (Amphibia, Anura, Hylidae). Revista Brasileira de Zoologia 24: 965–970.
Diaz-Paniagua, C., 1985. Larval diets related to morphological characters of five anuran species in the Biological Reserve of Doñana (Huelva, Spain). Amphibia-Reptilia 6: 307–321.
Duellman, W. E., & L. Trueb, 1986. Biology of Amphibians. New York.
Echeverría, D. D., A. V., Volpedo, & V. I., Mascitti, 2007. Diet of tadpoles from a pond in Iguazu National Park, Argentina. Gayana 71: 8–14.
Fay, M. P., 2010. Two-sided exact tests and matching confidence intervals for discrete data. R Journal 2: 53–58.
Fry, B., 2007. Stable isotope ecology. Springer Science & Business Media, New York.
Ghioca-Robrecht, D. M. & L. M. Smith, 2010. The role of Spadefoot Toad tadpoles in wetland trophic structure as influenced by environmental and morphological factors. Canadian Journal of Zoology 89: 47–59.
Gosner, K. L., 1960. A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16: 183–190.
Hart, S. L. & C. M. Christensen, 2002. The great leap: driving innovation from the base of the pyramid. MIT Sloan Management Review 44: 51–56.
Heyer, W. R., 1973. Ecological interactions of frog larvae at a seasonal tropical location in Thailand. Journal of Herpetology 7: 337.
Heyer, W. R., 1974. Niche measurements of frog larvae from a seasonal tropical location in Thailand. Ecology 55: 651–656.
Huckembeck, S., D. Loebmann, E. F. Albertoni, S. M. Hefler, M. C. Oliveira & A. M. Garcia, 2014. Feeding ecology and basal food sources that sustain the Paradoxal frog Pseudis minuta: a multiple approach combining stomach content, prey availability, and stable isotopes. Hydrobiologia 740: 253–264.
Hunte-Brown, M. E., 2006. The effects of extirpation of frogs on the trophic structure in tropical montane streams in Panama. Ph dissertation. ©Copyright Meshagae E. Hunte-Brown. 175pp.
Johnson, L. M., 1991. Growth and development of larval northern cricket frogs (Acris crepitans) in relation to phytoplankton abundance. Freshwater Biology 25: 51–59.
Kehr, A. I. & M. I. Duré, 1995. Descripción de la larva de Scinax nasica (Cope, 1862) (Anura, Hylidae). Facena 11: 99–103.
Kehr, A. I., E. F. Schaefer & M. I. Duré, 2004. The tadpole of Physalaemus albonotatus (Anura: Leptodactylidae). Journal of Herpetology 38: 145–148.
Kloh, J. S., C. C. Figueredo & P. C. Eterovick, 2019. How close is microhabitat and diet association in aquatic ecomorphotypes? A test with tadpoles of syntopic species. Hydrobiologia 828: 271–285.
Kruk, C., V. L. M. Huszar, E. T. H. M. Peeters, S. Bonilla, L. Costa, M. LüRling, C. S. Reynolds & M. Scheffer, 2010. A morphological classification capturing functional variation in phytoplankton. Freshwater Biology 55: 614–627.
Kupferberg, S. J., 1997. The role of larval diet in anuran metamorphosis. American Zoologist 37: 146–159.
Lajmanovich, R. C., 2000. Interpretación ecológica de una comunidad larvaria de anfibios anuros. Interciencia 25: 71–79.
López, J. A., P. A. Scarabotti, M. C. Medrano & R. Ghirardi, 2009. Is red spotted green frog (Hypsiboas punctatus, Anura: Hylidae) selecting its preys? Prey availability importance when analyzing trophic selectivity. Revista de Biología Tropical 57: 847–857.
López, J. A., P. A. Scarabotti & R. Ghirardi, 2015. Amphibian trophic ecology in increasingly human-altered wetlands. Herpetological Conservation and Biology 10: 819–832.
Maneyro, R. & I. da Rosa, 2004. Temporal and spatial changes in the diet of Hyla pulchella (Anura, Hylidae) in southern Uruguay. Phyllomedusa: Journal of Herpetology 3: 101–103.
Marker, A. F. H. & P. M. Bolas, 1984. Sampling of Non-planktonic Algae (Benthic Algae or Periphyton). HM Stationery Office, Richmond.
Matthews, B. & A. Mazumder, 2005. Temporal variation in body composition (C: N) helps explain seasonal patterns of zooplankton δ13C. Freshwater Biology 50: 502–515.
Molina, F. R., J. C. Paggi & M. Devercelli, 2010. Zooplanktophagy in the natural diet and selectivity of the invasive mollusk Limnoperna fortunei. Biological Invasions 12: 1647–1659.
Oksanen, J., 2011. Multivariate Analysis of Ecological Communities in R: vegan tutorial. http://cran.r-project.org.
Oksanen, J., R. Blanchet, P. Kindt, B. Legendre, Minchin, O’Hara, Simpson, Solymos, Stevens, & Wagner, 2015. The vegan package. Community ecology package 10. http://cran.r-project.org/.
Paggi, A. C., H. R. Fernández & E. Domínguez, 2001. Díptera: Chironomidae. In Hernández, H. R. & E. Domínguez (eds), Guía Para la Determinación de los Artrópodos Bentónicos Sudamericanos. Investigaciones de la UNT, Serie: 167–193.
Pavignano, I., 1990. Niche overlap in tadpole populations of Pelobates fuscus insubricus and Hyla arborea at a pond in north western Italy. Italian Journal of Zoology 57: 83–87.
Peltzer, P. M. & R. C. Lajmanovich, 2004. Anuran tadpole assemblages in riparian areas of the Middle Paraná River, Argentina. Biodiversity & Conservation 13: 1833–1842.
Pianka, E. R., 1973. The structure of lizard communities. Annual Review of Ecology and Systematics 4: 53–74.
Pianka, E. R., 1986. Ecology and Natural History of Desert Lizards: Analyses of the Ecological Niche and Community Structure. Princeton University Press, Princeton.
Pinkas, L. M., M. S. Oliphant & Z. L. Iverson, 1971. Food habits of albacore, bluefin tuna and bonito in California waters. California Department of Fish and Game Bulletin, Fishery Bulletin U.S. 152: 1–105.
Pollo, F. E., L. C. Martina, C. L. Bionda, N. E. Salas & A. L. Martino, 2015. Trophic ecology of syntopic anuran larvae, Rhinella arenarum (Anura: Bufonidae) and Hypsiboas cordobae (Anura: Hylidae): its relation to the structure of periphyton. EDP Sciences 51: 211–217.
Post, D. M., 2002. Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83: 703–718.
Post, D. M., C. A. Layman, D. A. Arrington, G. Takimoto, J. Quattrochi & C. G. Montana, 2007. Getting to the fat of the matter: models, methods and assumptions for dealing with lipids in stable isotope analyses. Oecologia 152: 179–189.
Quiroga, V., R. E. Lorenzón, G. Maglier & A. L. Ronchi-Virgolini, 2018. Relationship between morphology and trophic ecology in an assemblage of passerine birds in riparian forests of the Paraná River (Argentina). Avian Biology Research 11: 44–53.
R Core Team, 2016. R: A Language and Environment for Statistical Computing. Version 3.3.0. R Foundation for Statistical Computing. Vienna, Austria. http://www.R-project.org/.
Ricklefs, R. E. & D. B. Miles, 1994. Ecological and evolutionary inferences from morphology: an ecological perspective. Ecological Morphology: Integrative Organismal Biology 1: 13–41.
Rojas, A. E & J. H. Saluso, 1987. Informe climático de la provincia de Entre Ríos. Publicación Técnica Nº 14. INTA-EEA Paraná (E.R). 20pp.
Sabagh, L. T., G. L. Ferreira, C. W. Branco, C. F. D. Rocha & N. Y. Dias, 2012. Larval diet in bromeliad pools: a case study of tadpoles of two species in the genus Scinax (Hylidae). Copeia 2012: 683–689.
San Sebastián, O., J. Navarro, G. A. Llorente & Á. Richter-Boix, 2015. Trophic strategies of a non-native and a native amphibian species in shared ponds. PLoS ONE 10: 1–17.
Santos, F. J. M., A. S. Protázio, C. W. N. Moura & F. A. Juncá, 2015. Diet and food resource partition among benthic tadpoles of three anuran species in Atlantic Forest tropical streams. Journal of Freshwater Ecology 31: 53–60.
Schalk, C. M., C. G. Montaña, J. L. Klemish & E. R. Wild, 2014. On the Diet of the Frogs of the Ceratophryidae: Synopsis and New Contributions. South American Journal of Herpetology 9: 90–105.
Schalk, C. M., C. G. Montaña, K. O. Winemiller & L. A. Fitzgerald, 2017. Trophic plasticity, environmental gradients and food-web structure of tropical pond communities. Freshwater Biology 62: 519–529.
Schiesari, L., E. E. Werner & G. W. Kling, 2009. Carnivory and resource-based niche differentiation in anuran larvae: implications for food web and experimental ecology. Freshwater Biology 54: 572–586.
Seale, D. B. & N. Beckvar, 1980. The comparative ability of anuran larvae (Genera: Hyla, Bufo and Rana) to Ingest suspended blue-green algae. Copeia 1980: 495.
Schmidt, K., M. L. Blanchette, R. G. Pearson, R. A. Alford & A. M. Davis, 2017. Trophic roles of tadpoles in tropical Australian streams. Freshwater Biology 62: 1929–1941.
Starrett, P. H., 1960. Description of tadpoles of Middle America frogs. Miscellaneous Publications of the Museum of Zoology 110: 1–37.
Strauss, R. E., 1979. Reliability estimates for Ivlev’s electivity index, the forage ratio, and proposed linear index of food selection. Transactions of the American Fisheries Society 108: 344–352.
Utermöhl, H., 1958. Zur Vervollkommnung der quantitativen Phytoplankton-Methodik. Internationalen Vereinigung für Theoretische und Angewandte Limnologie 9: 1–38.
Vander Zanden, M. J., & J. B. Rasmussen, 1999. Primary consumer δ 13 C and δ 15 N and the trophic position of aquatic consumers. Ecology 80: 1395–1404.
Vassilieva, A. B., A. Y. Sinev & A. V. Tiunov, 2017. Trophic segregation of anuran larvae in two temporary tropical ponds in southern Vietnam. Herpetological Journal 27: 217–229.
Venesky, M., D. R. J. Wassersug & M. J. Parris, 2010. The impact of variation in labial tooth number on the feeding kinematics of tadpoles of southern leopard frog (Lithobates sphenocephalus). Copeia 3: 481–486.
Vera Candioti, M. F., 2007. Anatomy of anuran tadpoles from lentic water bodies: systematic relevance and correlation with feeding habits. Zootaxa 1600: 1–175.
Vera Candioti, M. F., E. O. Lavilla & D. D. Echeverría, 2004. Feeding mechanisms in two treefrogs, Hyla nana and Scinax nasicus (Anura: Hylidae): Feeding Mechanisms in two treefrogs. Journal of Morphology 261: 206–224.
Verburg, P., S. S. Kilham, C. M. Pringle, K. R. Lips & D. L. Drake, 2007. A stable isotope study of a neotropical stream food web prior to the extirpation of its large amphibian community. Journal of Tropical Ecology 23: 643–651.
Waringer-Löschenkohl, A. & M. Schagerl, 2001. Algal exploitation by tadpoles—an experimental approach. International Review of Hydrobiology 86: 105–125.
Wassersug, R. J., 1972. The mechanism of ultraplanktonic entrapment in anuran larvae. Journal of Morphology 137: 279–288.
Wassersug, R. J., 1980. Internal oral features of larvae from eight anuran families. Functional, systematics, evolutionary and ecological considerations. Miscellaneous Publications of the Museum of Natural History 65: 1–146.
Acknowledgements
This study was supported by Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET, Argentina) with a PhD fellowship to CE Antoniazzi. This study was financed by PIP-CONICET 2013-2017, PICT 2016-0916, PIP 112-201301-00790-CO and CAI+D-UNL PJoven-50020150100009LI. Collection of animals was authorized (Administrative Resolution 152-2016) by the Environmental Agency of Santa Fe Province, Argentine (Ministerio de Medio Ambiente de la Provincia de Santa Fe, Argentina) and use of specimens are in accordance with animal care guidelines of Regional and National Fauna Administrators. We are grateful to an anonymous reviewer and Associate Editor Lee Kats for the suggestions that contributed to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Lee B. Kats
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Antoniazzi, C.E., López, J.A., Lorenzón, R.E. et al. Trophic ecology of tadpoles in floodplain wetlands: combining gut contents, selectivity, and stable isotopes to study feeding segregation of syntopic species. Hydrobiologia 847, 3013–3024 (2020). https://doi.org/10.1007/s10750-020-04303-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-020-04303-0