Abstract
The aim of this study was to test the hypothesis that a widespread non-native fish species in Brazil displays opportunistic feeding behavior and changes its diet according to environmental conditions. We compared the diet, feeding selectivity, carbon assimilation, trophic niche, and trophic level of Knodus moenkhausii (a small non-native characid fish species of Upper Paraná River) in streams surrounded by natural riparian vegetation (natural cover streams) and in streams impacted by pasture. We analyzed stomach contents and stable isotopes (carbon and nitrogen), simultaneously. Overall, insects were the most common food items (> 65%). In natural cover streams, K. moenkhausii showed higher selectivity among aquatic macroinvertebrates consumed, while in pasture streams, they fed on the most abundant groups. The proportion of feeding groups assimilated by K. moenkhausii and the proportion of primary sources consumed by each feeding group of macroinvertebrates also varied between natural cover and pasture streams, as indicated by stable isotopes. In natural cover streams, fine and coarse particulate organic matter accounted for approximately 80% of K. moenkhausii’s diet, while in pasture streams, algae and periphyton also contributed greatly. As a result, K. moenkhausii occupied a higher trophic level and exhibited a broader niche width in pasture streams. We conclude that K. moenkhausii presents feeding selectivity with capacity to alter the trophic niche depending on environmental conditions. Such opportunism could be one of the reasons underpinning the abundance and wide distribution of this invasive species.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The introduction of non-native species is a major threat to both aquatic and terrestrial ecosystems (Chapin et al. 2000; Richardson 2011) and a major driver of species loss worldwide (Simberloff et al. 2013; Bellard et al. 2016). Within aquatic ecosystems, freshwaters tend to be the most vulnerable to invasion (Tricarico et al. 2016) and have been extensively altered by invasive species (Ricciardi and MacIsaac 2011). Biotic homogenization, or the replacement of specific native species by generalist non-native species in space and time (McKinney and Lockwood 1999; Tabarelli et al. 2012), results in freshwater systems with lower diversity at both regional and global scales (Rahel 2002; Drake and Lodge 2004). The introduction of non-native species is often related to anthropic actions and can occur accidentally or intentionally, through agriculture, recreation, biological control, fish stocks, aquaculture, sport fishing, and river transposition, among other routes (Welcomme 1988; Courtenay and Williams 1992; Lever 1998).
The potential establishment of non-native species in new geographic locations is closely related to environmental quality (e.g., Herborg et al. 2007; Kilroy et al. 2008; Kulhanek et al. 2011) and may be related to disturbance (Hobbs and Huenneke 1992; Minchinton 2002; Marchetti et al. 2004a). Land-use changes such as the conversion of natural habitats to pastures and croplands constitute a key factor disturbing natural terrestrial and aquatic environments. For instance, the establishment of pastures can increase autochthony in aquatic environments (e.g., Carvalho et al. 2015; De Carvalho et al. 2017), which may facilitate biological invasions due to changes in resource availability (Davis et al. 2000).
In addition to changes in resource availability, habitat alteration facilitates the success of non-native species by reducing native competitors and predators and increasing habitat homogenization that benefits opportunistic invaders (Scott and Helfman 2001; Scott 2006; Johnson et al. 2008). Other characteristics that increase the invasive potential of a non-native species are high reproductive rates, long life spans, long-distance dispersal, high physiological tolerance, generalist habit, and high trophic plasticity (Kolar and Lodge 2001; Marchetti et al. 2004b; Funk 2008; Barrett 2011).
Feeding strategies and trophic plasticity can be evaluated by comparing trophic webs and a species’ niche breadth between environments that experience different levels of disturbance. In particular, combining stomach content analyses with whole-body stable isotopes (e.g., carbon and nitrogen) can yield robust and accurate information. Stomach content analysis provides detailed taxonomic information about the food items consumed and is one of the most common techniques used to evaluate fish feeding habits (e.g., Hyslop 1980; Lima-Junior and Goitein 2001; Buckland et al. 2017). However, stomach content analyses are limited in that they only inform a single point in time (Cortés 1997), and the relative assimilation of some resources is poorly quantified, such as detritus (Keough et al. 1998). Complementary approaches, such as whole-body analysis of stable isotopes, provide an integrated assessment of carbon and nitrogen sources an organism assimilates over time. These analyses have often been used in conjunction to assess the importance of different food items in the diet of fishes and other organisms (e.g., Polito et al. 2011; Carassou et al. 2017).
The objective of this study was to evaluate whether a non-native species displays an opportunistic feeding behavior and exhibits dietary shifts according to environmental conditions. We evaluated the diet and feeding selectivity (through stomach content analysis) and the food sources assimilated, trophic level and trophic niche occupied (through stable isotopes analysis) of Knodus moenkhausii (Eigenmann and Keneddy 1903). To assess whether the invasive species shows trophic plasticity between environments with different levels of disturbance, we compared the diet of fish in streams surrounded by riparian vegetation (natural cover streams) and streams surrounded by pasture (an activity responsible for 71% of deforestation in South America, FAO 2016). We expected different types of resources would be available in streams with different land uses, with a high influence of autochthonous resources in pasture streams and allochthonous resources from riparian forest in natural cover streams. Consequently, the differences in resource availability would have cascading effects on abundance and diversity of aquatic macroinvertebrates leading to changes in K. moenkhausii feeding habits. We tested two hypotheses: (1) As a generalist, K. moenkhausii feeds on the most abundant food items available and (2) variation in local resource availability will result in changes in K. moenkhausii trophic niche.
Methods
Study area and land-use classification
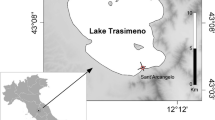
This study was conducted in six second- and third-order streams (Strahler 1957) located in the sub-basin of the Paranaíba River, southeastern Brazil (Fig. 1, Table 1). The Paranaíba River Basin is the second largest hydrographic unit of the Paraná Basin, encompassing 25.4% of its area (222.8 km2). Most of the Paranaíba River Basin is in the Cerrado (neotropical savanna), the second largest Brazilian biome, but much of this area has already been deforested (Strassburg et al. 2017). The hydrological regime of the rivers in this basin is governed by rains from October to March (annual average precipitation 1200 mm) and with episodic rainfall in the remaining months of the year (CBH Paranaíba 2012).
Six streams located in the states of Goiás and Minas Gerais were selected according to the different predominant types of land use and local assessment of the physical habitat of each stream. The length of the section sampled in each stream was 150 m. Each stream was sampled once in the dry season, in September 2012.
The land use surrounding the sampled streams was evaluated according to the oriented mapping method described in Lima et al. (2010), in eight multispectral RapidEye images of September and October of 2011, with five spectral bands. The percentage cover of natural vegetation and pasture was determined for the six study streams in a 150-m-radius buffer around the upstream limit of the sampled stretch. Orthorectified and atmospherically corrected images were obtained through a partnership between the Federal University of Lavras (UFLA) and the Ministry of Environment (MMA). Acquisition errors, clouds, and shadows were removed in the preprocessing phase (Coppin et al. 2004), which also included visual evaluation of image registration. To validate the classification results, we generated an array of errors measuring the global and kappa accuracy (Shanmugam et al. 2006). The mapping resulted in a high kappa and global accuracy with values from 96% and 98%, respectively. To access the representativeness of the vegetation that is interacting with the stream channel, we also assessed the percentage cover of natural vegetation and pasture within a buffer of 50 m upstream of each site by interpreting a combination of high-resolution satellite images (0.6–5 m spatial resolution, Google Earth data, Google 2010) and Landsat multispectral satellite images (R4G3B2 false color band combination).
Physical habitat at the site scale was characterized using the US EPA field methods (Lazorchak et al. 1998; Peck et al. 2006). At each site, we characterized the physical habitat by channel hydromorphological variables (depth, width, and area); substrate (% of fine substrate—smaller than 16 mm); riparian woody cover (sum of riparian canopy cover, riparian mid-layer and riparian ground layer) and in-stream habitat variables (% of algae and leaf banks). All site variables were calculated according to Kaufmann et al. (1999), who described concepts and analytical procedures for calculating metrics based on data generated from the physical habitat field protocols. These variables were assessed because they consistently reflected the effects of different land uses on physical habitat features related to resource availability (Table 1).
Three streams were located in pasture-dominated areas, and three reference streams were surrounded by natural riparian cover (Fig. 1). The pasture streams were surrounded by a buffer zone containing more than 62% pasture, lacked riparian vegetation (or had only sparse trees), and stream beds were dominated by the prevalence of fine substrate and algae. Natural vegetation, as well as other land uses, occurred upstream of the study reaches (Table 1). The natural cover streams were located in sites surrounded by more than 39% of natural vegetation and were characterized by the presence of riparian forest, few fine substrates, and a higher amount of leaf banks than algae on the stream bed. Although other land uses, especially agriculture, occurred upstream of the natural cover sites, these streams were not influenced by pastures (Table 1).
Characteristics of the invasive fish species and sampling methods
The Upper Paraná is the second largest basin in Brazil and has approximately 310 described fish species, over 20% of which are non-native (Langeani et al. 2007). Knodus moenkhausii is noteworthy among the non-native species mentioned. It is a small characid, originally described based on specimens collected in the drainage of the Paraguay River basin and its presence in the Upper Paraná has uncertain origins (Langeani et al. 2007). This species has been consistently registered in the Upper Paraná in high densities (Casatti et al. 2009; Teresa and Casatti 2013; Fagundes et al. 2015). Due to its remarkable abundance and frequency in the Upper Paraná basin, K. moenkhausii’s habitat use, feeding preferences and reproductive biology have been widely studied (Appendix S1). However, its hypothesized opportunistic feeding behavior and trophic plasticity have not been explored in the context of different land uses.
Individuals of K. moenkhausii were collected using hand nets (80 cm in diameter, 1 mm mesh) and trawls (3 m long, 5 mm mesh). A total of 336 individuals were sampled in the six streams (259 in natural cover streams and 77 in pastures). The average total lengths were similar in natural cover (3.93 ± 0.60 cm) and pasture streams (3.32 ± 0.69 cm). A total of 35 adult individuals (18 sampled in natural cover streams and 17 in pasture streams) was selected for stable isotope analysis and was processed whole (except the digestive track) due to the small size of the sampled individuals. For the stable isotope analysis, the collected fish were immediately killed and stored on ice for further processing in the laboratory.
Sixty-five adult specimens of K. moenkhausii (35 collected in natural cover streams and 30 in pastures) had their stomachs removed and their contents analyzed using a stereomicroscope. Individuals selected for evaluation of stomach contents were anesthetized with eugenol and subsequently fixed in formaldehyde 10% to reduce the chance of rapid digestion of any food item.
For isotopic analysis, fish samples were lyophilized for 24 h and ground to a fine and homogeneous powder using a mortar and pestle and stored in Eppendorf tubes. Approximately 2–5 mg of dry animal tissue was used. The individuals used for stomach content analysis were preserved in 70% alcohol until they were processed in the laboratory. Voucher specimens of K. moenkhausii were deposited in the Ichthyological Collection of the Federal University of Lavras, under the catalogue number CI-UFLA 0839.
Collection and processing of resources
Samples of basal resources and benthic macroinvertebrates were collected in situ, five samples of each resource per stream. Basal resources consisted of filamentous algae, periphyton (or biofilm), coarse particulate organic matter (CPOM), fine particulate organic matter (FPOM), leaves from the riparian vegetation, and grasses. The filamentous algae samples were manually collected, stored in plastic bottles, and immediately frozen. Periphyton was sampled by scraping and washing rocks with distilled water, and then, the collected materials were stored in plastic bottles. The FPOM samples were collected from sediment deposits by revolving the sediment and passing the suspended material through a phytoplankton net (45 µm mesh). After collection, both periphyton and FPOM samples were immediately frozen to preserve the material. In the laboratory, samples were vacuum-filtered through 45-micron Millipore glass fiber filters previously calcined. The CPOM was randomly collected from leaf litter deposits in the streams, and samples of vegetation (riparian forest and grasses) were collected at different points outside the streams. All leaves were packed in paper bags and stored in plant presses until processing in the laboratory.
Benthic macroinvertebrates were collected using a D-frame kick net (30 cm aperture, 500 µm mesh). To obtain the abundance of benthic macroinvertebrates in each stream, eleven subsample units (0.09 m2 each) were taken per site following a systematic zigzag pattern along a segment of 150 m, covering all different substrates and habitats, generating one composite sample for each site and totaling an area of 1 m2 (Castro et al. 2017) (see Appendix S2 to access data of benthic macroinvertebrates abundance). To obtain individuals for isotopic analysis, we carried out an additional collection, following the same procedures described above. The individuals designated for isotopic analysis were immediately frozen and stored in plastic bottles. In the laboratory, the collected organisms were washed in distilled water, taxonomically identified (Merritt et al. 2008; Mugnai et al. 2010; Hamada et al. 2014) and classified into main orders and functional feeding groups (predators, scrapers, shredders, gathering-collectors and filtering-collectors) based on the literature (Cummins et al. 2005; Tomanova et al. 2006; Ramírez and Gutiérrez-Fonseca 2014) (Appendix S3).
In the laboratory, all samples of resources (basal resources and benthic macroinvertebrates) were dried at 60° for 48 h prior to homogenization using mortar and pestle and storage in Eppendorf tubes. Approximately 2–5 mg of dry animal tissue was selected for isotopic analysis, while approximately 5–10 mg was required for the basal resources samples.
Isotopic analysis
Stable isotopes for carbon and nitrogen were analyzed at the Center for Nuclear Energy in Agriculture (CENA) at the University of São Paulo. To determine the isotopic ratio, a mass spectrometer system in the continuous-flow (CF-IRMS) mode was used with a Carlo Erba elemental analyzer (CHN 1110) coupled to a Delta Plus mass spectrometer (Thermo Scientific). The results are expressed as the difference of international reference standards, in the delta notation (δ) in parts per mil (‰), and calculated using the following formula:
where X is 13C or 15N and R represents the isotopic ratio 13C/12C or 15N/14N (Barrie and Prosser 1996).
Stomach content analysis
Food items were weighed (0.001 g accuracy/wet weight) and identified under stereomicroscope to the lowest feasible taxonomic unit according to macroinvertebrate identification guides (Costa et al. 2006; Mugnai et al. 2010). To characterize the diet of K. moenkhausii, we used the feeding index (IA) proposed by Kawakami and Vazzoler (1980), combining the frequency of occurrence (Fi = number of times the item i occurred divided by the total number of stomachs with food) and the relative weight (Pi = sum of the weight of the item i divided by the sum of the weight of all items) of each item:
where IAi = feeding index of item i; Fi = frequency of occurrence of item i, and Pi = weight of the item i.
Data analysis
We addressed the hypothesis that K. moenkhausii feeds on the most abundant food items by using aquatic macroinvertebrates (the most common item in stomach contents) as a proxy. A simple linear regression was performed to evaluate whether K. moenkhausii feeds on the most abundant aquatic macroinvertebrates in both natural cover and pasture streams. This analysis was performed to determine whether the frequency of Coleoptera, Diptera, Hemiptera, Ephemeroptera, and Trichoptera in K. moenkhausii stomach contents was proportional to their natural abundances in the streams based on the quantitative samplings.
Differences in carbon (δ13C) and nitrogen (δ15N) signatures of K. moenkhausii between pasture and natural cover areas were tested using a generalized linear mixed model (GLMM) that assumed a normal distribution for variation in carbon (δ13C) and nitrogen (δ15N). In order to avoid pseudoreplication, each fish was nested to the respective stream. The streams were considered as a random factor, while land use was included as a fixed effect. Analyses were conducted in the software R v3.2.2 (R Core Team 2017), using the package lme4 (Bates et al. 2015) for the GLMMs.
Source contributions to K. moenkhausii diet were estimated for both natural cover and pasture streams based on stable isotope data analyzed through Bayesian stable isotope mixing models (Moore and Semmens 2008; Parnell et al. 2010), using the MixSIAR package in R (Stock and Semmens 2016a). Markov chain Monte Carlo sampling was implemented with the following parameters: number of chains = 3; chain length = 100,000; burn in = 50,000; thin = 50 and model 4 (Residual×Process) error structure (Stock and Semmens 2016b). Diagnostic tests (Gelman–Rubin, Heidelberger–Welch and Geweke) and trace plots were examined for model convergence. Individuals of K. moenkhausii were considered consumers and the basal resources (algae, riparian vegetation, grasses and periphyton), and the five feeding groups of benthic macroinvertebrates (predators, shredders, gathering-collectors, filtering-collectors and scrapers) were considered food resources. From this, the partition analysis was developed in two steps. The first step considered the items observed on stomach contents of K. moenkhausii, that is, vegetal remains (fragments of riparian vegetation leaves and grasses) and macroinvertebrates separated into feeding groups. In this step, we chose to use the riparian vegetation and the grasses as resources, as they also represent the isotopic signature of terrestrial insects observed in stomach contents. Algae and periphyton were not observed in the stomachs of K. moenkhausii and therefore were not considered in this step. The second step evaluated the proportion of basal resources (CPOM, FPOM, algae and periphyton) consumed by each feeding group of macroinvertebrates separately to investigate the resources assimilated indirectly by K. moenkhausii. The fractionation values used for fish and for macroinvertebrates were 0.5 ± 0.13‰ for C and 2.3 ± 0.18‰ for N because it is an enrichment appropriate for detritivores or omnivores that consume mixtures of plant material and microbial or animal material (McCutchan et al. 2003).
After the two steps, it was possible to calculate the contribution of primary sources to the isotopic composition of K. moenkhausii in streams with different degrees of conservation. To do so, we multiplied the proportion of each resource consumed by K. moenkhausii (results of step 1) by the proportion of each primary resource consumed by each feeding groups of macroinvertebrates separately (results of step 2).
The isotopic niches of K. moenkhausii in both categories of land use (natural cover and pasture) were quantified based on total area (TA) and standard ellipse areas (SEA and SEAc—expressed in ‰2) through use of the Stable Isotope Bayesian Ellipses package in R (SIBER, Jackson et al. 2011). The standard ellipse area (SEA) represents the core isotopic niche space, and it is a proxy of the richness and evenness of resources consumed by the population (Bearhop et al. 2004). A small sample size correction (indicated by the subscript letter “c”) was applied to SEA to increase the accuracy of the comparisons, enabling the comparison of niches of populations with different sample sizes.
The trophic position (TP) of K. moenkhausii in both categories of land use was estimated through the trophic position model proposed by Vander Zanden et al. (1997): TP = [(15Nfish − 15Nresources) ÷ 2.3] + 1, where 15Nfish= 15N values of each individual of K. moenkhausii, 15Nresources = mean values of 15N of basal resources, 2.3 represents the fractionation per trophic level (McCutchan et al. 2003), and 1 is the position of producers within the food chain. The 15N of basal resources was assessed multiplying the percentage of total contribution of each basal resource (calculated in the partition analysis) by its mean 15N values in each category of land use. The trophic position was calculated for each individual of K. moenkhausii. A t test was performed to determine whether there was variation in the trophic levels occupied by K. moenkhausii in the different land-use categories.
Results
Aquatic and terrestrial insects (also considering insect remains) were the primary food items consumed by K. moenkhausii in natural cover (77%) and in streams influenced by pastures (67%) (Table 2). Detritus was the second most commonly consumed item in pasture-influenced streams (26%), and in natural cover streams it represented 11% as well as plant remains. In pastures, plant remains constituted only 6% of the feeding of K. moenkhausii (Table 2).
There was a variation among the proportion of each order of insects consumed by K. moenkhausii. In natural cover streams, the most commonly consumed groups were Coleoptera (19%) and terrestrial insects of the order Hymenoptera (16%). In pastures, the most consumed groups were Diptera (15%), Trichoptera (3%), Ephemeroptera (2%) and terrestrial insects of the order Hymenoptera (2%) (Table 2). However, the frequency of occurrence of macroinvertebrates in the stomachs of K. moenkhausii did not reflect the abundance of each order in the substrate sampled in natural cover streams (R2 = 0.09; t = − 0.53; p = 0.63) (Fig. 2a). By contrast, K. moenkhausii in pasture streams fed on aquatic macroinvertebrates that were most abundant (R2 = 0.87; t = 4.58; p = 0.02) (Fig. 2b).
The isotopic composition of K. moenkhausii varied between streams in different land uses for both carbon (t = 4.29; p = 0.041) and nitrogen (t = 4.58; p = 0.031). Stable isotope values of 13C were more enriched in natural cover streams (− 28.11 to − 22.65‰) than in pasture-influenced streams (− 34.09 to − 24.10‰). In contrast, values of 15N were more enriched in pasture-influenced streams (9.20 to 13.09 ‰) than in natural cover streams (7.73–10.42‰) (Fig. 3).
Representation of trophic web (bi-plot space) of the species K. moenkhausii (solid points) sampled in natural cover streams (a) and streams influenced by pasture (b). No enrichment factor was used to construct the bi-plot space graph. Basal resources: AL filamentous algae, RV riparian vegetation, PE periphyton, BM benthic macroinvertebrates, and GR grasses of pasture
Partition analyses revealed that K. moenkhausii resource assimilation differed between natural cover and pasture-influenced streams (Fig. 4). In natural cover streams, K. moenkhausii assimilated more gathering-collectors (32% of their diet), followed by filtering-collectors (20%), predators (17%), shredders (16%) and scrapers (12%). Vegetal remains from riparian vegetation contributed only 3%. Considering indirect assimilation via macroinvertebrates, the basal resources that most contributed to K. moenkhausii stable isotope values in natural cover streams were FPOM (39%) and CPOM (37%), followed by periphyton (13%). Contributions from algae (8%) and riparian vegetation (3%) were relatively minor (Fig. 4a). In pasture streams, K. moenkhausii assimilation consisted primarily of predators (42%), filtering-collectors (21%) and shredders (13%), followed by gathering-collectors and scrapers (9% each). Vegetal remains (including grasses) constituted only 6%. Considering indirect assimilation via macroinvertebrates, the contribution of CPOM and FPOM was also relevant in pastures streams (31% and 26%, respectively). However, the contribution of the autochthonous resources, periphyton (24%) and algae (12%) was double of those in the natural cover streams (Fig. 4b).
Proportion of the resources assimilated by K. moenkhausii in natural cover streams (a) and in streams influenced by pastures (b). Feeding groups of macroinvertebrates: PR predator, SH shredder, FI filtering-collector, CO gathering-collector and SC scraper. Basal resources: AL filamentous algae, FPOM fine particulate matter, CPOM coarse particulate matter, RV riparian vegetation, GR grasses of pasture and PE periphyton
The differences in K. moenkhausii resource utilization between stream types were further reflected by their distributions in carbon–nitrogen bi-plot space (Fig. 3), in the amplitude of the isotopic niche (Fig. 5a) and in its trophic position (Fig. 5b). The isotopic niche was wider in pasture streams (TA = 21.17; SEA = 7.90; SEAc = 8.42), than in natural cover streams (TA = 9.93; SEA = 3.77; SEAc = 4.00) (Fig. 5a). Knodus moenkhausii’s trophic position was also higher in pasture streams than in natural cover streams (p = 0.02) (Fig. 5b).
Representation of amplitude of the isotopic niche (a) and trophic position (b) of K. moenkhausii. The points and triangles in the figure a represent the δ13C and δ15N isotopic signatures of each individual of K. moenkhausii sampled in pasture and natural cover streams, respectively. Standard ellipse areas (SEA, solid sphere) represent the core isotopic niche space and the dashed lines delimit the total area (TA)
Results from carbon assimilation and stomach content analyses were combined to generate the K. moenkhausii food web (Fig. 6). In natural cover streams, the main resources in K. moenkhausii food webs are FPOM and CPOM (indirectly) and macroinvertebrate gathering-collectors (directly). In pasture streams, CPOM and FPOM contributed less to the food webs than in natural cover streams, and autochthonous basal resources (periphyton and filamentous algae) had larger indirect contributions to the food web. In these streams, macroinvertebrate predators were more often consumed by K. moenkhausii. The consumption of plant remains (in natural cover and pasture streams) and terrestrial insects (especially in natural cover streams) was observed through stomach content analysis. However, the isotopic composition of plant remains was not representative in the isotopic composition of K. moenkhausii.
Representation of food web of the species K. moenkhausii sampled in natural cover streams (a) and pasture streams (b) according to the analyses of stomach contents (dashed lines) and stable isotopes (solid lines). PR predator, FI filtering-collector, CO gathering-collector, SH shredder and SC scraper. Basal resources: AL algae, FPOM fine particulate matter, CPOM coarse particulate matter, PE periphyton and Plant remains = riparian vegetation and grasses (in pastures)
Discussion
In the present study, we observed animal and plant material in the stomachs of K. moenkhausii and thus confirmed its omnivorous feeding habit. However, stable isotope analysis revealed that a large proportion of K. moenkhausii’s carbon and nitrogen came from aquatic macroinvertebrates, indicating a strong tendency for insectivory/invertivory. We also observed broad niche and flexible diet in response to different land uses, as well as differences in selectivity of aquatic macroinvertebrates.
Stomach content analysis produced detailed information on items consumed. In pasture streams, the abundance of aquatic macroinvertebrate groups correlated with their occurrence frequency in stomachs of K. moenkhausii. In streams with natural cover, K. moenkhausii consumed more Coleoptera despite it being only the third most abundant order in these streams. However, we were not able to distinguish whether the coleopterans found in gut contents were terrestrial or aquatic. Relative availability of terrestrial insects was not quantified, and terrestrial insects are presumably more abundant in natural streams with intact riparian systems. Therefore, although K. moenkhausii exhibited some selectivity for other taxa, it is not possible to state whether or not K. moenkhausii consumed coleopterans in proportion to their availability or present more selectivity. Such opportunistic feeding may enable K. moenkhausii to allocate energy to reproduction throughout the year even in environments that are physically impacted by human disturbances (Ceneviva-Bastos and Casatti 2007).
Stable isotope analyses provided information about the food resources assimilated over time. Higher assimilation of gathering-collector macroinvertebrates in preserved streams and predators in pasture-influenced streams may be due to the relative abundance and availability of different feeding guilds under different land uses. This is not surprising, given that land-use changes can lead to shifts in resource availability and abiotic conditions and may alter the presence of different functional feeding groups of macroinvertebrates (García et al. 2017). Greater abundance of shredders and gathering-collectors is expected under natural conditions due to the greater supply of CPOM in forested covered streams than pasture streams (Graça et al. 2015; Linares et al. 2017). An increase in the number of scrapers and filtering-collectors is expected in pasture-influenced streams because reduced canopy cover frequently leads to higher periphyton production (Winkelmann et al. 2014; Neres-Lima et al. 2016, 2017).
We also expected different basal resources to contribute to the food web of K. moenkhausii, since land-use changes alter nutrient cycling, which can lead to differences in the quantity and quality of available food resources (Richardson et al. 2010). Aquatic macroinvertebrates may also present more generalist feeding habits in human-impacted sites, whereas more specialization may occur in macroinvertebrate assemblages under less disturbed conditions (Castro et al. 2016). Greater assimilation of CPOM and FPOM is expected under more natural environments, since the abundance of these resources is closely related to the presence of riparian forest. Pastures may also provide sources of CPOM and FPOM, especially coming from grass and manure associated with cattle grazing. The C4 grasses from pastures usually present enriched δ13C values relative to C3 plants from riparian vegetation (see Fig. 3), which occurs in accordance with the decarboxylation processes employed by each plant (Manetta and Benedito-Cecílio 2003). Therefore, the depleted δ13C values from aquatic macroinvertebrates and K. moenkhausii in pasture streams indicate the main source of FPOM and CPOM in these streams is from the riparian vegetation, which occurs upstream of the sampled reaches (see land use in Table 1). Grass leaves are also less nutritious and difficult to digest, which can explain the low assimilation by K. moenkhausii and other fish species (Carvalho et al. 2015). Increased dependence on autochthony in pasture streams may also have contributed to the depleted values of consumers at these streams (see Fig. 3). All these changes to food web dynamics demonstrate the consequences of replacing native vegetation with pastures, which is one of the main drivers of deforestation in tropical and subtropical regions (FAO 2016).
Flexible feeding habit has been documented in other invasive fish species (Pettitt-Wade et al. 2015; Tran et al. 2015; Busst and Britton 2017) and is a key trait associated with successful colonization, establishment, and spread (Peterson and Vieglais 2001; Pettitt-Wade et al. 2015). The greater niche amplitude of K. moenkhausii recorded in pasture streams indicates a more generalist feeding habit, which is characteristic of species adapted to unstable environments (Levins 1968). Knodus moenkhausii displays this ability, as individuals fed at different trophic levels and presented broad and plastic isotopic niche in environments with different degrees of conservation. The higher assimilation of macroinvertebrate predators explained the higher trophic level occupied by K. moenkhausii in pasture streams. However, it remains unclear why K. moenkhausii relied heavily on macroinvertebrate predators in pasture streams, since predators (as Odonata, Naucoridae and Perlidae) were also abundant in natural cover streams (see Appendix S3). One hypothesis is a likely easiest capture of macroinvertebrates predators in pasture streams, especially when grasses from adjacent pasture enter the stream channel. In this case, macroinvertebrates predators with epiphytic habits (as Odonata) are easily consumed for fishes that forage in macrophyte banks.
Several classical studies (e.g., Hutchinson 1957; Van Valen 1965) have indicated that an absence of interspecific competition can lead to larger trophic niches. If this hypothesis is correct, we can assume that resources are not being fully exploited in pasture streams, and K. moenkhausii occupies previously vacant dietary niches, which has facilitated their colonization (Shea and Chesson 2002). However, if K. moenkhausii is actually feeding opportunistically in all streams, the differences in diet and isotopic signatures would depict differences in the underlying basal food webs (proportions of autochthonous sources) and availability of terrestrial insects.
It is necessary to emphasize that dietary interactions with resident species can strongly influence and impact native communities through a variety of ways, including altering predator–prey relationships (Alexander et al. 2014; Guo et al. 2017) and resource competition (Cucherousset et al. 2012; Busst and Britton 2017). Because predicting the ecological consequences of invasions by non-native species is a fundamental aspect of their risk-based management, it is crucial to understand the traits associated with invasive potential (Britton et al. 2007; 2011; Busst and Britton 2017). We also highlight that each invasive species presents its own particularities that allow them to be widespread or rare in the invaded environment, and in our study, we evaluated only the trophic plasticity of a single fish species.
Our study identified trophic characteristics that could be related to the success of Knodus moenkhausii as an invasive species. Through our results, it was also possible to evaluate how habitat degradation can change the trophic responses of such species, which reflected the aspects of ecosystem functioning. This information is important especially in developing countries, where rapid agricultural expansion leads to rapid changes in land use.
References
Alexander ME, Dick JT, Weyl OL, Robinson TB, Richardson DM (2014) Existing and emerging high impact invasive species are characterized by higher functional responses than natives. Biol Lett 10:20130946. https://doi.org/10.1098/rsbl.2013.0946
Barrett SCH (2011) Why reproductive systems matter for the invasion biology of plants. In: Richardson DM (ed) Fifty years of invasion ecology: the legacy of Charles Elton. Wiley, Oxford, pp 195–210
Barrie A, Prosser SJ (1996) Automated analysis of light-element stable isotopes by isotope ratio mass spectrometry. In: Boutton TW, Yamaski S (eds) Mass spectrometry of soils. Marcel Decker, New York, pp 1–46
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bearhop S, Adams CE, Waldron S (2004) Determining trophic niche width: a novel approach using stable isotope analysis. J Anim Ecol 73:1007–1012
Bellard C, Cassey P, Blackburn TM (2016) Alien species as a driver of recent extinctions. Biol Lett 12:20150623. https://doi.org/10.1098/rsbl.2015.0623
Britton JR, Davies GD, Brazier M, Pinder AC (2007) A case study on the population ecology of a top mouth gudgeon Pseudorasbora parva population in the UK and the implications for native fish communities. Aquat Conserv Mar Freshw Ecosyst 17:749–759
Britton JR, Gozlan RE, Copp GH (2011) Managing non-native fish in the environment. Fish Fish 12:256–274
Buckland A, Baker R, Loneragan N, Sheaves M (2017) Standardising fish stomach content analysis: the importance of prey condition. Fish Res 196:126–140. https://doi.org/10.1016/j.fishres.2017.08.003
Busst GMA, Britton JR (2017) Comparative trophic impacts of two globally invasive cyprinid fishes reveal species-specific invasion consequences for a threatened native fish. Freshw Biol 62:1587–1595. https://doi.org/10.1111/fwb.12970
Carassou L, Whitfield AK, Moyo S, Richoux NB (2017) Dietary tracers and stomach contents reveal pronounced alimentary flexibility in the freshwater mullet (Myxus capensis, Mugilidae) concomitant with ontogenetic shifts in habitat use and seasonal food availability. Hydrobiologia 799:327–348. https://doi.org/10.1007/s10750-017-3230-3
Carvalho DR, Castro DMP, Callisto M, Moreira MZ, Pompeu PS (2015) Isotopic variation in five species of stream fish under the influence of different land uses. J Fish Biol 87:559–578
Casatti L, Ferreira CP, Carvalho FR (2009) Grass-dominated stream sites exhibit low fish species diversity and dominance by guppies: an assessment of two tropical pasture river basins. Hydrobiologia 632:273–283
Castro DMP, De Carvalho DR, Pompeu PS, Moreira MZ, Nardoto GB, Callisto M (2016) Land use influences niche size and the assimilation of resources by benthic macroinvertebrates in tropical headwater streams. PLoS ONE 11:e0150527
Castro DMP, Dolédec S, Callisto M (2017) Landscape variables influence taxonomic and trait composition of insect assemblages in neotropical savanna streams. Freshw Biol 62:1472–1486. https://doi.org/10.1111/fwb.12961
CBH Paranaíba: Comitê da bacia hidrográfica do Rio Paranaíba (2012). http://www.paranaiba.cbh.gov.br. Accessed 12 July 2016
Ceneviva-Bastos M, Casatti L (2007) Oportunismo alimentar de Knodus moenkhausii (Teleostei, Characidae): uma espécie abundante em riachos do noroeste do Estado de São Paulo, Brasil. Iheringia Ser Zool 97:7–15
Ceneviva-Bastos M, Casatti L, Rossa-Feres DC (2010) Meso and microhabitat analysis and feeding habits of small nektonic characins (Teleostei: Characiformes) in neotropical streams. Zoologia (Curitiba) 27:191–200
Ceneviva-Bastos M, Taboga SR, Casatti L (2015) Microscopic evidence of the opportunistic reproductive strategy and early sexual maturation of the small-sized characin Knodus moenkhausii (Characidae, Pisces). Anat Histol Embryol 44:72–80. https://doi.org/10.1111/ahe.12112
Chapin FS, Zavaleta ES, Eviner VT, Naylor RL, Vitousek PM, Reynolds HL, Hooper DU, Lavorel S, Sala OE, Hobbie SE, Mack MC, Diaz S (2000) Consequences of changing biodiversity. Nature 405:234–242
Coppin P, Jonckheere I, Nackaerts K, Muys B, Lambin E (2004) Digital change detection methods in ecosystem monitoring; a review. Remote Sens 25:1565–1596
Cortés E (1997) A critical review of methods of studying fish feeding based on analysis of stomach contents: application to elasmobranch fishes. J Fish Aquat Sci 54:726–738
Costa C, Ide S, Simonka CE (2006) Insetos Imaturos. Metamorfose e identificação. Holos, Editora, Ribeirão Preto
Courtenay WR Jr, Williams JD (1992) Dispersal of exotic species from aquaculture sources, with emphasis on freshwater fishes. In: Rosenfield A, Mann R (eds) Dispersal of living organisms into aquatic ecosystems. Maryland Sea Grant Program, College Park, pp 49–82
Cucherousset J, Bouletreau S, Martino A, Roussel JM, Santoul F (2012) Using stable isotope analyses to determine the ecological effects of non-native fishes. Fish Manag Ecol 19:111–119
Cummins KW, Merritt RW, Andrade PC (2005) The use of invertebrate functional groups to characterize ecosystem attributes in selected streams and rivers in south Brazil. Stud Neotrop Fauna Environ 40:69–89. https://doi.org/10.1080/01650520400025720
Davis MA, Grime JP, Thompson J (2000) Fluctuating resources in plant communities: a general theory of invasibility. J Ecol 88:528–534
De Carvalho DR, Castro D, Callisto M, Moreira M, Pompeu PS (2017) The trophic structure of fish communities from streams in the Brazilian cerrado under different land uses: an approach using stable isotopes. Hydrobiologia 795:199–217. https://doi.org/10.1007/s10750-017-3130-6
Drake JM, Lodge DM (2004) Global hot spots of biological invasions: evaluating options for ballast-water management. Proc R Soc Lond B Biol Sci 271:575–580
Fagundes DC, Leal CG, Carvalho DR, Junqueira NT, Langeani F, Pompeu PS (2015) The stream fish fauna from three regions of the Upper Paraná River basin. Biota Neotrop 15:1–8. https://doi.org/10.1590/1676-06032015018714
FAO - Food and Agriculture Organization of the United Nations (2016) El Estado de los bosques del mundo 2016. Los bosques y la agricultura: desafíos y oportunidades en relación con el uso de la tierra. http://www.fao.org/publications/sofo/2016/es/. Accessed 02 Oct 2017
Funk JL (2008) Differences in plasticity between invasive and native plants from a low resource environment. J Ecol 96:1162–1173. https://doi.org/10.1111/j.1365-2745.2008.01435.x
García L, Cross WF, Pardo I, Richardson JS (2017) Effects of land use intensification on stream basal resources and invertebrate communities. Fresh Sci 36:609–625. https://doi.org/10.1086/693457
Google (2010) Google earth. Google Inc., Mountain View
Graça MAS, Ferreira WR, Firmiano K, França J, Callisto M (2015) Macroinvertebrate identity, not diversity, differed across patches differing in substrate particle size and leaf litter packs in low order, tropical Atlantic forest streams. Limnetica 34:29–40
Guo Z, Sheath D, Amat-Trigo F, Britton JR (2017) Comparative functional responses of native and high impacting invasive fishes: impact predictions for native prey populations. Ecol Freshw Fish 26:533–540. https://doi.org/10.1111/eff.12297
Hamada N, Nessimian JL, Querino RB (2014) Insetos aquáticos na Amazônia brasileira: taxonomia, biologia e ecologia. Editora do INPA, Manaus
Herborg LM, Jerde CL, Lodge DM, Ruiz GM, MacIsaac HJ (2007) Predicting invasion risk using measures of introduction effort and environmental niche models. Ecol Appl 17:663–674
Hobbs RJ, Huenneke LF (1992) Disturbance, diversity, and invasion: implications for conservation. Conserv Biol 6:324–337
Hutchinson GE (1957) The multivariate niche. Cold Spring Harb Symp Quant Biol 22:415–421
Hyslop EJ (1980) Stomach contents analysis—a review of methods and their application. J Fish Biol 17:411–429. https://doi.org/10.1111/j.1095-8649.1980.tb02775.x
Jackson AL, Inger R, Parnell AC, Bearhop S (2011) Comparing isotopic niche widths among and within communities: SIBER—stable isotope Bayesian ellipses in R. J Anim Ecol 80:595–602
Johnson PT, Olden JD, Vander Zanden MJ (2008) Dam invaders: impoundments facilitate biological invasions in freshwaters. Front Ecol Environ 6:357–363
Kaufmann PR, Levine P, Robison EG, Seeliger C, Peck DV (1999) Quantifying physical habitat in wadeable streams. U.S. Environmental Protection Agency EPA/620/R-99/003. USEPA, Washington, DC
Kawakami E, Vazzoler G (1980) Método gráfico e estimativa de Índice Alimentar aplicado no estudo de alimentação de peixes. Bolm Inst Oceanogr 29:205–207
Keough JR, Hagley CA, Ruzycki E, Sierszen M (1998) 13C composition of primary producers and role of detritus in a freshwater coastal ecosystem. Limnol Oceanogr 43:734–740
Kilroy C, Snelder TH, Floerl O, Vieglais CC, Dey KL (2008) A rapid technique for assessing the suitability of areas for invasive species applied to New Zealand’s rivers. Divers Distrib 14:262–272
Kolar CS, Lodge DM (2001) Progress in invasion biology: predicting invaders. Trends Ecol Evol 16:199–204
Kulhanek SA, Brian L, Ricciardi A (2011) Using ecological niche models to predict the abundance and impact of invasive species: application to the common carp. Ecol Appl 21:203–213
Langeani F, Castro RMC, Oyakawa OT, Shibatta OA, Pavanelli CS, Casatti L (2007) Ichthyofauna diversity of the upper rio Paraná: present composition and future perspectives. Biota Neotrop 7:181–197. https://doi.org/10.1590/S1676-06032007000300020
Lazorchak JM, Klemm DJ, Peck DV (1998) Environmental monitoring and assessment program-surface waters: field operations and methods for measuring the ecological condition of wadeable streams. U.S. Environmental Protection Agency EPA 620/R-94/004F. Cincinnati, OH, USEPA
Lever C (1998) Introduced fishes: an overview. In: Cowx IG (ed) Stocking and Introduction of Fish. Fishing News Books, Oxford, pp 143–152
Levins R (1968) Evolution in changing environments, some theoretical explorations. Monographs in population biology. Princeton University Press, Princeton
Lima LPZ, Pompeu PS, Suzuki FM, Carvalho LMT (2010) Dinâmica espacial de lagoas marginais presentes no rio Aiuruoca, MG, em períodos de cheia e seca. Rev Bras Biocienc 8:253–256
Lima-Junior SE, Goitein RA (2001) A new method for the analysis of fish stomach contents. Acta Sci Biol Sci 23:421–424
Linares MS, Callisto M, Marques JC (2017) Compliance of secondary production and eco-exergy as indicators of benthic macroinvertebrates assemblages’ response to canopy cover conditions in Neotropical headwater streams. Sci Total Environ 613–614:1543–1550. https://doi.org/10.1016/j.scitotenv.2017.08.282
Manetta GI, Benedito-Cecílio E (2003) Aplicação da técnica de isótopos estáveis na estimativa da taxa de turnover em estudos ecológicos: uma síntese. Acta Sci Biol Sci 25:121–129
Marchetti MP, Light T, Moyle PB, Viers J (2004a) Fish invasions in California watersheds: testing hypotheses using landscape patterns. Ecol Appl 14:1507–1525
Marchetti MP, Moyle PB, Levine R (2004b) Invasive species profiling? Exploring the characteristics of non-native fishes across invasion stages in California. Freshw Biol 49:646–661
McCutchan JH, Lewis WM, Kendall C, McGrath CC (2003) Variation in trophic shift for stable isotope ratios of carbon, nitrogen, and sulfur. Oikos 102:378–390. https://doi.org/10.1034/j.1600-0706.2003.12098.x
McKinney ML, Lockwood JL (1999) Biotic homogenization: a few winners replacing many losers in the next mass extinction. Trends Ecol Evol 14:450–453
Merritt RW, Cummins K, Berg M (2008) An introduction to the aquatic insects of North America. Kendall/Hunt Publishing, Dubuque
Minchinton TE (2002) Disturbance by wrack facilitates spread of Phragmites australis in a coastal marsh. J Exp Mar Biol Ecol 281:89–107
Moore JW, Semmens BX (2008) Incorporating uncertainty and prior information into stable isotope mixing models. Ecol Lett 11:470–480. https://doi.org/10.1111/j.1461-0248.2008.01163.xPMID:18294213
Mugnai R, Nessimian JL, Baptista DF (2010) Manual de Identificação de Macroinvertebrados Aquáticos do Estado do Rio de Janeiro. Technical Books, Rio de Janeiro
Neres-Lima V, Brito EF, Krsulović FA, Detweiler AM, Hershey AE, Moulton TP (2016) High importance of autochthonous basal food source for the food web of a Brazilian tropical stream regardless of shading. Int Rev Hydrobiol 101:132–142. https://doi.org/10.1002/iroh.201601851
Neres-Lima V, Machado-Silva F, Baptista DF, Oliveira RBS, Andrade PM, Oliveira AF, Sasada-Sato CY, Silva-Junior EF, Feijó-Lima R, Angelini R, Camargo PB, Moulton TP (2017) Allochthonous and autochthonous carbon flows in food webs of tropical forest streams. Freshw Biol 62:1012–1023. https://doi.org/10.1111/fwb.12921
Parnell AC, Inger R, Bearhop S, Jackson AL (2010) Source partitioning using stable isotopes: coping with too much variation. PLoS ONE 5:e09672. https://doi.org/10.1371/journal.pone.0009672
Peck DV, Herlihy AT, Hill BH, Hughes RM, Kaufmann PR, Klemm DJ, Lazorchak JM, Mccormick FH, Peterson SA, Ringold PL, Magee T, Cappaert MR (2006) Environmental monitoring and assessment program: surface waters western pilot study—field operations manual for wadeable streams. EPA 620/R-06/003. US Environmental Protection Agency, Washington, DC
Peterson AT, Vieglais DA (2001) Predicting species invasions using ecological niche modeling: new approaches from bioinformatics attack a pressing problem. Bioscience 51:363–371
Pettitt-Wade H, Wellband KW, Heath DD, Fisk AT (2015) Niche plasticity in invasive fishes in the Great Lakes. Biol Invasions 17:2565. https://doi.org/10.1007/s10530-015-0894-3
Polito MJ, Trivelpiece WZ, Karnovsky NJ, Ng E, Patterson WP, Emslie SD (2011) Integrating stomach content and stable isotope analyses to quantify the diets of pygoscelid penguins. PLoS ONE 6:e26642. https://doi.org/10.1371/journal.pone.0026642
R Core Team (2017) A language and environment for statistical computing. 55:275–286. http://www.r-project.org. Accessed 28 Sept 2017
Rahel FJ (2002) Homogenization of freshwater faunas. Ann Rev Ecol Syst 33:291–315
Ramírez A, Gutiérrez-Fonseca PE (2014) Functional feeding groups of aquatic insect families in Latin America: a critical analysis and review of existing literature. Rev Biol Trop 62:155–167
Ricciardi A, MacIsaac HJ (2011) Impacts of biological invasions on freshwater ecosystems. In: Richardson DM (ed) Fifty years of invasion ecology: the legacy of Charles Elton. Wiley, Oxford, pp 211–224
Richardson DM (2011) Fifty years of invasion ecology: the legacy of Charles Elton. Wiley, Oxford
Richardson JS, Zhang Y, Marczak LB (2010) Resource subsidies across the land–freshwater interface and responses in recipient communities. River Res Appl 26:55–66
Scott MC (2006) Winners and losers among stream fishes in relation to land use legacies and urban development in the southeastern US. Biol Conserv 127:301–309
Scott MC, Helfman GS (2001) Native invasions, homogenization, and the mismeasure of integrity of fish assemblages. Fisheries 26:6–15
Shanmugam P, Ahn Y, Sanjeevi SA (2006) A comparison of the classification of wetland characteristics by linear spectral mixture modelling and traditional hard classifiers on multispectral remotely sensed imagery in southern India. Ecol Model 194:379–394
Shea K, Chesson P (2002) Community ecology theory as a framework for biological invasions. Trends Ecol Evol 17:170–176
Simberloff D, Martin J, Genovesi P, Maris V, Wardle DA, Aronson J, Courchamp F, Galil B, García-Berthou E, Pascal M, Pyšek P, Sousa R, Tabacchi E, Vila M (2013) Impacts of biological invasions: what’s what and the way forward. Trends Ecol Evol 28:58–66
Stock BC, Semmens BX (2016a) MixSIAR GUI user manual (version 3.1). https://github.com/brianstock/MixSIAR/. Accessed 19 Feb 2018
Stock BC, Semmens BX (2016b) Unifying error structures in commonly used biotracer mixing models. Ecology 97:2562–2569. https://doi.org/10.1002/ecy.1517
Strahler AN (1957) Quantitative analysis of watershed geomorphology. Trans Am Geophys Union 38:913–920. https://doi.org/10.1029/tr038i006p00913
Strassburg BBN, Brooks T, Feltran-Barbieri R, Iribarrem A, Crouzeilles R, Loyola R, Latawiec AE, Oliveira Filho FJB, de Mattos Scaramuzza CA, Scarano FR, Soares-Filho B, Balmford A (2017) Moment of truth for the Cerrado hotspot. Nat Ecol Evol 99:1–3. https://doi.org/10.1038/s41559-017-0099
Tabarelli M, Peres CA, Melo FPL (2012) The ‘few winners and many losers’ paradigm revisited: emerging prospects for tropical forest biodiversity. Biol Conserv 155:136–140. https://doi.org/10.1016/j.biocon.2012.06.020
Teresa FB, Casatti L (2013) Development of habitat suitability criteria for neotropical stream fishes and an assessment of their transferability to streams with different conservation status. Neotrop Ichthyol 11:395–402
Tomanova S, Goitia E, Helešic J (2006) Trophic levels and functional feeding groups of macroinvertebrates in neotropical streams. Hydrobiologia 556:251–264. https://doi.org/10.1007/s10750-005-1255-5
Tran TNQ, Jackson MC, Sheath D, Verreycken H, Britton JR (2015) Patterns of trophic niche divergence between invasive and native fishes in wild communities are predictable from mesocosm studies. J Anim Ecol 84:1071–1080
Tricarico E, Junqueira AOR, Dudgeon D (2016) Alien species in aquatic environments: a selective comparison of coastal and inland waters in tropical and temperate latitudes. Aquat Conserv 26:872–891
Van Valen L (1965) Morphological variation and width of ecological niche. Am Nat 99:377
Vander Zanden MJ, Cabana G, Rasmussen JB (1997) Comparing trophic position of freshwater fish calculated using stable nitrogen isotope ratios (15 N) and literature dietary data. Can J Fish Aquat Sci 54:1142–1158
Welcomme RL (1988) International introductions of inland aquatic species. FAO fisheries technical paper 294
Winkelmann C, Schneider J, Mewes D, Schmidt SI, Worischka S, Hellmann C, Benndorf J (2014) Top-down and bottom-up control of periphyton by benthivorous fish and light supply in two streams. Freshw Biol 59:803–818. https://doi.org/10.1111/fwb.12305
Acknowledgements
We thank the National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico, CNPq), the Coordination for the Improvement of Higher Education Personnel (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, CAPES), Minas Gerais Energy Company (Companhia Energética de Minas Gerais, CEMIG) and P&D ANEEL/CEMIG GT-487 for the financial support for the project and for the Master’s and Doctoral scholarships granted. Thanks to Ludmilla Portela Zambaldi Lima (Federal University of Lavras/UFLA) and Diego Rodrigues Macedo (Federal University of Minas Gerais/UFMG) for analyses of land use and to UFLA and the Ministry of Environment (MMA) to grant the RapidEye images. Thanks to Amelia K. Weiss (Cornell University) for the English revision. Thanks also to the Benthos Ecology Laboratory (UFMG) and the Laboratory of Fish Ecology (UFLA) who assisted in the collection and processing of samples, to the Centre for Nuclear Energy in Agriculture (CENA) for their support and partnership in the isotopic analysis. DRC was partialy funded by CAPES – Finance Code 001. DMPC received a PDJ scholarship (150307/2018-7) from CNPq. MC was awarded a research productivity CNPq (303380/2015-2, 446155/2014-4) and from the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG No. PPM 00104-18). PSP received a research fellowship from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq No. 306325/2011-0) and from the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG No. PPM-00237/13).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Handling Editor: Télesphore Sime-Ngando.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Carvalho, D.R., de Castro, D.M.P., Callisto, M. et al. Stable isotopes and stomach content analyses indicate omnivorous habits and opportunistic feeding behavior of an invasive fish. Aquat Ecol 53, 365–381 (2019). https://doi.org/10.1007/s10452-019-09695-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-019-09695-3