Abstract
Objective To analyze the diagnostic efficacy of computer aided analysis of relevant coronary artery stenosis using dual source computed tomography (DSCT). Methods In a larger scale study patients scheduled for conventional coronary angiography (CA) were additionally examined with DSCT. Based on a 13-segment model 30 CT scans of this study population were analyzed for significant stenosis using conventional 3D charts (3D) as well as a specialized cardiac analysis tool (CAT). Diagnostic accuracy and time to diagnosis was recorded for each vessel separately as well as the three readers’ confidence. Results With severe coronary artery calcifications, 53 false interpretations of segments were found for the total of 390 coronary segments analyzed. 3D and CAT analysis showed a Sensitivity, Specificity, PPV and NPV of 0.59, 0.91, 0.57, 0.92 and 0.57, 0.92, 0.56, 0.92, respectively. No significant differences in diagnostic accuracy could be found between 3D and CAT (P = 0.1667). 3D took a mean of 5.2 min (3–10 min). With CAT a mean time of 8.2 min (4–12 min) was needed. No significant inter-reader time differences (P = 0.4954) and no significant confidence level differences were found between readers and analyzes. Conclusion CAT of the coronary tree shows comparable accuracy to manual 3D analysis but needs improvements concerning coronary tree segmentation times.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In recent years, cardiac multidetector computed tomography (MDCT) for exclusion of coronary artery stenosis has become a routine clinical tool in daily practice [1–3]. As it is integrated into the daily workflow time efficient analysis of the significant amount of data is mandatory. The most common approach to coronary stenosis detection to date is a manual positioning of the imaging planes to get a near cross sectional or longitudinal view of the vessel of interest. As this can be quite time consuming computer aided reading and specialized software to improve the vascular analysis are offered by the different vendors [3–11]. All of these visualization tools have some kind of vessel segmentation/isolation in common allowing a display of the vessel of interest in fashion designed for optimal stenosis detection. Our scanner software offers a 3D package as well as a specialized cardiac analysis tool (Circulation®, Siemens, Forchheim, Germany) for cardiac computed tomographic (CT) data analysis by segmenting and isolating the coronary tree and presenting the vessels of interest in specified views (curved MPR and true cross sectional views) facilitating stenosis detection. Recently Busch et al. published a performance evaluation study for an automated coronary vessel analysis. They selected perfect image quality scans and found good agreement of the stenosis analysis with the automated approach compared to catheter angiogram stenosis quantification [12]. In the present study the speed and efficacy of manual coronary stenosis detection in cases of varying image quality were compared to the computer aided vascular analysis. The severity of the coronary artery stenosis was determined with conventional catheter angiography as the reference standard.
Materials and methods
Out of a larger study population of 200 patients with known or suspected coronary artery disease scheduled for conventional coronary angiography (CA) that were additionally examined with dual source computed tomographiy (DSCT) 30 patients (22 male, 8 female, mean age 62 years, 50–74) with varying image quality were selected for additional analysis of their DSCT data with the specialized automated cardiac analysis tool (CAT). Image quality of the scans was rated 1–5, with 1 being excellent image quality and 5 being non diagnostic scans. For this study scans with diagnostic image quality (1 or 4) were selected. We chose to exclude the non diagnostic scans as the software tool cannot perform the segmentation in these cases. Having an indication for a conventional catheter angiogram the patients had a higher pretest probability for coronary atherosclerosis than an average patient population, the majority of the patients showed calcifications measured with the Agatston score. All patients gave informed consent to this examination. As a retrospective selection of data was performed no dedicated patient consent was obtained, but the patient gave consent to the DSCT examination and further use of their data. The local ethics committee approved of the study.
Scanning protocol
A dual-source CT scanner (SOMATOM Definition, Siemens Medical Solutions, Forchheim, Germany) was used for all CT examinations with patients in the supine position. Circulation time was determined using a test bolus with 20 ml of contrast media (400 mg iodine/ml; Imeron 400 Altana, Konstanz, Germany) and a 40-ml saline chaser bolus (flow rate 5 ml/s) with a dual-head injector (CT Stellant; Medrad, Indianola, Pennsylvania). For dual-source CT angiography, the following scan protocol was used: 0.6-mm collimation (cardiac mode), 120-kV tube voltage, 330-ms gantry rotation time, and pitch 0.2–0.43 (automatically adapted to the patients’ heart rate) [13]. The mean tube current for both tubes was 560 mA, preset at 320 mAs/rot for each tube. For dose reduction, the tube current was electrocardiographically modulated and reduced during the systolic phases, but maintained at maximum during the diastolic phase of the cardiac cycle. According to the patients’ heart rate, the following ECG-pulsing was adapted automatically (ECG-pulsing, auto) for maximum tube current within the RR interval: 60–70% (<50 beats/min), 55–70% (51–60 beats/min), 35–70% (61–119 beats/min), and 20–75% (>120 beats/min). Contrast media (70 ml; in patients with coronary artery bypass grafts, 90 ml) was injected intravenously with a flow rate of 5 ml/s followed by a saline chaser bolus (50 ml, flow rate 5 ml/s).
The reconstruction interval with the fewest motion artifacts was determined by reconstructing a slice at the level of the middle of the left ventricle in 5% increments from 25 to 75% of the RR interval. CT angiographic images were reconstructed for diagnostic evaluation to the time point with the least motion artifact of the right and left coronary arteries. For CT angiographic images the optimal kernel of B26f for stenosis detection and an effective slice thickness of 0.75 mm with a reconstruction increment of 0.4 mm were chosen. The 180 mm field of view was centered at the heart and images were reconstructed with a high resolution matrix of 512 × 512 pixels.
Image analysis
The DSCT datasets were analyzed for significant stenosis using the original axial slices, multiple planar reconstructions and sliding thin-slab maximum intensity projections (MIP). Significant stenoses were defined as stenoses with luminal narrowing above 50%. The analysis was performed manually using a conventional 3D chart. This allows multiplanar reconstructions and MIP in different orientations, for instance a rough longitudinal slice orientation along the course of the vessel is possible. Stenoses were graded subjectively as is the clinical routine practice in our institution. As in conventional angiography lumen diameter reductions of ≥50% were considered significant stenosis.
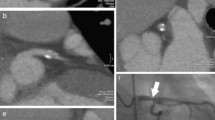
Additionally the scans were assessed using a specialized CAT (Circulation 3®, Siemens, Forchheim, Germany). This software is developed especially for coronary analysis [14]. The computer segments the coronary tree upon a click in the aortic root and displays the vessel of interest in a curved multiplanar reformat view (cMPR) as well as a true axial view (see Fig. 1). The segmentation is based on a geometric model using a region growing algorithm with premasking of the model to exclude interfering structures close to the vessel of interest. After segmentation the coronary tree is digitalized producing a transform based skeleton for distance measurements but the lumen boundaries can also be identified on the source images. Additionally stenosis measurement is also possible along the orthogonal views of the vessel path and in the curved multiplanar reformations [14]. These measurements were used to determine if a stenosis was >50% but the stenosis values given were not recorded for this study.
Using our routine 13-segment system, a modified classification of the American Heart Association, all coronary vessel segments were documented separately (right coronary artery: 1, proximal; 2, middle; 3, distal; and 4, combined posterior descending and posterolateral branches; 5, left main stem; left anterior descending: 6, proximal; 7, middle; 8, distal; 9, first diagonal; 10, second diagonal; left circumflex: 11, proximal; 12, distal; and 13, first marginal branch).
Invasive coronary angiograms were obtained within 1 day after DSCT. All angiograms were evaluated by an independent, experienced interventional cardiologist using quantitative coronary analysis with automated vessel contour detection. The angiography catheter was used for calibration (Quantitative Coronary Analysis, Philips, Best, The Netherlands). Lesions with a diameter reduction of ≥50% were considered significant. All coronary vessel segments were included in the statistical analysis. Coronary angiography was regarded as the reference standard to detect relevant vascular stenosis. In coronary segments with >1 lesion, the lesion with the most severe diameter reduction determined the diagnostic accuracy.
In comparison with invasive coronary angiography, dual-source CT results were analyzed on a segmental basis (each segment in every vessel) and per patient (evaluating the presence of any significant stenosis in a given patient).
Performance evaluation
For both analyses the accuracy compared to the conventional catheter angiography as well as time to diagnosis was recorded. The time to analyze each vessel was also documented separately. Additionally to accuracy and time, reader’s confidence in their findings was reported. Three readers, with varying experience in cardiac CT (reader 1:2 years, reader 2:1.5 years, reader 3:8 months) performed the analyses. Accuracy was analyzed using conventional coronary angiography as the reference standard.
Statistical analysis
T-test calculation was used to calculate sensitivity and specificity, negative and positive predictive value. Wilcoxon tests for non numeric data were performed comparing the subgroups. A value of P < 0.05 was considered to be significant. Kappa statistics (Cohen’s kappa with Landis–Koch extension and combined Fleiss–Nee–Landis test) were used to analyze interreader agreement. Categorical data were presented with absolute frequencies and percentages. Continuous variables are shown as means ± SDs. All analyses were performed using JMP 5.1 SAS Institute Inc. (Cary, NC) and Graph Pad Prism4 Software (San Diego, CA).
Results
All examinations were performed without complications. No bleeding or allergic reactions occurred. As our patient collective was recruited out of patients with an indication for an invasive catheter angiogram, a preselection of more severe arthrosclerosis was present in our collective. The mean Agatston score of our collective was 707.9 (range: 0–1,598). All segments showed sufficient intraluminal contrast with a mean aortic root attenuation of 387.3HU (range: 290–410). The mean heart rate of the patients was 62.7 (range: 41–85), no prescan ß-blockers were administered. No segment had to be excluded from the analysis as the cases were retrospectively selected for diagnostic image quality all segments of the coronary tree were depicted well enough for evaluation. In total, 390 coronary segments (30 × 13 segments) were analyzed. Manual 3D chart analysis presented with 53 false (27 false negative and 26 false positive) interpretations of segments compared to conventional catheter angiography. These results correspond to a sensitivity of 0.59, specificity of 0.91, PPV 0.57, and NPV 0.92. Specialized CAT also revealed 53 misreadings. 26 false negative and 27 false positive interpretations led to a sensitivity of 0.57, specificity of 0.92, PPV 0.56, and NPV 0.92. As most false readings corresponded in the 3D and CAT analysis (25 analogous false negative findings, 20 corresponding false positive findings), no significant differences in diagnostic accuracy could be found (P = 0.1667) (Figs. 2 , 3). With kappa values of 0.58 for 3D and 0.59 for CAT a good interreader agreement was seen.
On the left a paraxial view of the left anterior descending artery created with the manual analysis program (3D) is shown. A high grade stenosis of the proximal first diagonal branch, Segment 9 is suspected (white arrow). The image plane truly axial to the vessel (right) generated by the specialized CAT shows no stenosis in Segment 9, which was confirmed by conventional angiography
The average manual analysis with the 3D chart took 5.2 min (3–10 min). With CAT a mean time of 8.2 min (4–12 min) was needed. The average 3 min plus needed to reach the diagnosis included mean segmentation time of 4.2 min (1–7 min) (Fig. 4). The Wilcoxon test showed no significant inter-reader time differences (P = 0.1250). The level of confidence shows slight interreader differences, reader 1 and 2 felt more confident using the automated approach whereas reader 3 showed no preference for either method. Over all no significant confidence level differences were found between readers (P = 0.4954) and analyzes (P = 0.1525), see Figs. 5 and 6.
Comparison of average time needed to reach the diagnosis for each reader and averaged over all readers (total). 3D is the manual analysis, CAT represents the time needed with the specialized CAT and CAT-seg is analysis time only, with exclusion of the time needed by the computer to segment out the coronary tree. All three radiologists needed less time to reach a diagnosis with the automatic approach but the time gain was lost in segmentation time needed by the software
Discussion
The results showed that the specialized cardiac analysis of the coronary tree is slightly more time consuming than the manual approach but may raise the level of confidence in the readers. The accuracy compared to conventional coronary catheter does not differ significantly between the two methods or different readers.
Analyzing the time differences between the two approaches we found that the usual time to analyze the data in CAT is an average 3.1 min longer than in the manual approach. The mean time needed to segment the coronary tree within the CAT takes 4.2 min, so the analysis itself is actually faster than in the manual approach. The average time to diagnosis with CAT is 8.2 min minus the average time for segmentation 4.2 min is an average 4 min computer aided analysis to diagnosis compared to an average 5.2 min time needed to diagnosis in the manual approach. Segmentation times are software related so increasing speed in the segmentation algorithm should decrease the time needed to reach the diagnosis using the computer aided approach. Dewey et al. found a software aided, modified automated 3D approach (Vitrea 2, Version 3.3, Vital Images) equivalent to and significantly less time consuming than the traditional manual 2D method for evaluation of ≥50%-stenoses by 16 × 0.5-MDCTA in native coronary arteries of patients with high calcium scores [12, 15].
Manual analysis of CT angiography data often is limited by intra- and inter-observer variability [16]. Fully automatic tools enable not only fast quantification without manual interaction and also a reproducible measurement without user dependence [17, 18]. We could show a good interreader agreement in the 3D as well as the CAT approach and the intraindividual agreement with both methods was also very high. Thus the reliability of the preprocessing with the CAT system does not impair diagnostics.
Cordeiro et al. [19] stated that if automatic coronary vessel detection is feasible it has the potential to reduce the time required to create reformations by a factor of approximately two without deteriorating the diagnostic accuracy. On the basis of optimal contrast-enhanced dataset automated segmentation algorithms have the potential to be more accurate and more time-effective [20, 21]. However, a major bottleneck of cardiac CT imaging of the coronary arteries is the potential lack of image quality due to limitations in the spatial and temporal resolution, severe coronary calcifications, irregular or high heart beat, respiratory effects, and variations of the distribution of the contrast agent all limiting the automated quantitative analysis of the coronary arteries, for which good image quality is required [22]. A human brain can piece together a coronary vessel chopped up by motion artifacts. Computer algorithms, however, can experience great difficulties in tracking a vessel with motion artifacts. This leads to more time consuming manual alterations of either the segmentation or the source images. Different methods are available to automatically segment vessels in medical images, but they are either not fully automated (initial vessel points are required) or they are very sensitive to noise in the image. Unfortunately, the presence of image noise, the variability of the background, and the low and varying contrast of vessels, makes it quite difficult to obtain reliable fully automatic or even semi-automatic vessel detection procedures [23].
In this trial the level of confidence was scaled subjectively from sure to unsure. It is crucial especially in computer assisted analysis to yield a high level of confidence as automation always requires a level of trust towards the data alteration performed by the computer before presentation of data for diagnostics. On the other hand preprocessing and management of the ever growing amount of data acquired is becoming more and more important for effective radiology work. A typical CT coronary angiography will have an average of 400–1,000 images in each volume dataset. This is an extreme example but CT angiographies are becoming a key component of state-of-the-art imaging making three-dimensional postprocessing crucial for time effective and sensible visualization [24].
Soto et al. [25]. analyzed the impact of a picture archiving and communication systems (PACS)-based software package on the requests for 3D reconstructions of multidetector CT (MDCT) datasets in the emergency radiology department of a level 1 trauma center. In the polytrauma patient, implementation of the program increased utilization of 3D reconstructions of MDCT datasets [25]. Multiple image display techniques are available to the radiologist at standard workstations. They are multiplanar reformation (MPR), oblique MPR, curved MPR, MIP, shaded-surface display, and direct volume rendering. Each of these techniques has its advantages and disadvantages for the visualization of the coronary artery tree. Several additions to the basic techniques have been developed to overcome some of their shortcomings. Different clinical examinations, such as stent evaluation, stenosis evaluation, and bypass evaluation, require different visualization techniques. The choice of preferred technique for each clinical study depends on the advantages and disadvantages of the various techniques as described in the literature [16, 20, 26–28]. In general, for optimal acceptance a computer assisted analysis should enable, flexibility with tolerance for non optimal image quality, automatic parameter estimation pre choosing the most accurate display forms, high sensitivity in detection of pathologic findings and a reasonable processing time.
This may provide an enhanced accuracy of the CAT system without increase in the overall diagnosis time [29, 30]. Because of the large number of possible settings and projection angles, it is important for users to interactively manipulate the images and review the whole vessel volume rather than just looking at static reformatted images [28]. Misinterpretations such as false positive vascular stenoses may be reduced by means of accurate and appropriate use of software features. This requires training of users both with regard to the capabilities of the software and the background of the different techniques and their possible pitfalls [28]. Postprocessing techniques have different advantages and disadvantages when used in clinical practice, and it is important that radiologists understand when and how each technique should be used [24].
As we have a relative small study population small but maybe significant differences between the manual and automated approach might have not been apparent. Further studies with larger patient collectives are needed. But with the ever increasing amount of data created by each scan time effective and reliable software tools facilitating data analysis are of significant importance in all day practice.
In conclusion the manual approach using the 3D option is appropriate for a first, fast overview over the scan data. Currently this is the most time effective way to a rough coronary status estimate of the patient with an idea of the disease burden. Especially for problem solving the specialized CAT is a helpful tool to yield an accurate and reliable status of coronary artery disease for the patient. At this point in time the automated approach is still more time consuming, and as such is not recommendable as a first line approach. But with the rapid software and computer development segmentations times may soon be reduced significantly making computer assisted analysis a valuable alternative to the manual approach.
References
Lawler LP, Pannu HK, Fishman EK (2005) MDCT evaluation of the coronary arteries, 2004: how we do it—data acquisition, postprocessing, display, and interpretation. AJR Am J Roentgenol 184(5):1402–1412
Budoff MJ, Gopal A, Gopalakrishnan D (2006) Cardiac computed tomography: diagnostic utility and integration in clinical practice. Clin Cardiol 29(9):I4–I14
Schoenhagen P, Halliburton SS, Stillman AE, Kuzmiak SA, Nissen SE, Tuzcu EM et al (2004) Noninvasive imaging of coronary arteries: current and future role of multi-detector row CT. Radiology 232(1):7–17. doi:10.1148/radiol.2321021803
Brown MS, McNitt-Gray MF, Pais R, Shah SK, Qing P, Da Coasta I et al (2007) CAD in clinical trials: current role and architectural requirements. Comput Med Imaging Graph 31(4–5):332–337. doi:10.1016/j.compmedimag.2007.02.014
Doi K (2007) Computer-aided diagnosis in medical imaging: historical review, current status and future potential. Comput Med Imaging Graph 31(4–5):198–211. doi:10.1016/j.compmedimag.2007.02.002
Katsuragawa S, Doi K (2007) Computer-aided diagnosis in chest radiography. Comput Med Imaging Graph 31(4–5):212–223. doi:10.1016/j.compmedimag.2007.02.003
Marten K, Engelke C (2007) Computer-aided detection and automated CT volumetry of pulmonary nodules. Eur Radiol 17(4):888–901. doi:10.1007/s00330-006-0410-3
Mougiakakou SG, Golemati S, Gousias I, Nicolaides AN, Nikita KS (2007) Computer-aided diagnosis of carotid atherosclerosis based on ultrasound image statistics, laws’ texture and neural networks. Ultrasound Med Biol 33(1):26–36. doi:10.1016/j.ultrasmedbio.2006.07.032
Yoshida H, Nappi J (2007) CAD in CT colonography without and with oral contrast agents: progress and challenges. Comput Med Imaging Graph 31(4–5):267–284. doi:10.1016/j.compmedimag.2007.02.011
Bogoni L, Cathier P, Dundar M, Jerebko A, Lakare S, Liang J et al (2005) Computer-aided detection (CAD) for CT colonography: a tool to address a growing need. Br J Radiol 78(Spec No 1):S57–S62
Hirai T, Korogi Y, Arimura H, Katsuragawa S, Kitajima M, Yamura M et al (2005) Intracranial aneurysms at MR angiography: effect of computer-aided diagnosis on radiologists’ detection performance. Radiology 237(2):605–610. doi:10.1148/radiol.2372041734
Busch S, Johnson TR, Nikolaou K, von Ziegler F, Knez A, Reiser MF et al (2007) Visual and automatic grading of coronary artery stenoses with 64-slice CT angiography in reference to invasive angiography. Eur Radiol 17(6):1445–1451. doi:10.1007/s00330-006-0512-y
Flohr TG, McCollough CH, Bruder H, Petersilka M, Gruber K, Suss C et al (2006) First performance evaluation of a dual-source CT (DSCT) system. Eur Radiol 16(2):256–268. doi:10.1007/s00330-005-2919-2
Boskamp T, Rinck D, Link F, Kummerlen B, Stamm G, Mildenberger P (2004) New vessel analysis tool for morphometric quantification and visualization of vessels in CT and MR imaging data sets. Radiographics 24(1):287–297. doi:10.1148/rg.241035073
Dewey M, Schnapauff D, Laule M, Lembcke A, Borges AC, Rutsch W et al (2004) Multislice CT coronary angiography: evaluation of an automatic vessel detection tool. Rofo 176(4):478–483
Keller D, Wildermuth S, Boehm T, Boskamp T, Mayer D, Schuster HL et al (2006) CT angiography of peripheral arterial bypass grafts: accuracy and time-effectiveness of quantitative image analysis with an automated software tool. Acad Radiol 13(5):610–620. doi:10.1016/j.acra.2006.01.008
Burling D, Halligan S, Roddie ME, McQuillan J, Honeyfield L, Amin H et al (2005) Computed tomography colonography: automated diameter and volume measurement of colonic polyps compared with a manual technique–in vitro study. J Comput Assist Tomogr 29(3):387–393. doi:10.1097/01.rct.0000160985.66259.96
Heussel CP, Achenbach T, Buschsieweke C, Kuhnigk J, Weinheimer O, Hammer G et al (2006) Quantification of pulmonary emphysema in multislice-CT using different software tools. Rofo 178(10):987–998
Cordeiro MA, Lardo AC, Brito MS, Rosario Neto MA, Siqueira MH, Parga JR et al (2006) CT angiography in highly calcified arteries: 2D manual vs. modified automated 3D approach to identify coronary stenoses. Int J Cardiovasc Imaging 22(3-4):507–516. doi:10.1007/s10554-005-9044-9
Mühlenbruch G, Das M, Hohl C, Wildberger JE, Rinck D, Flohr TG et al (2006) Global left ventricular function in cardiac CT. Evaluation of an automated 3D region-growing segmentation algorithm. Eur Radiol 5(16):1117–1123. doi:10.1007/s00330-005-0079-z
Taylor SA, Halligan S, Slater A, Goh V, Burling DN, Roddie ME et al (2006) Polyp detection with CT colonography: primary 3D endoluminal analysis versus primary 2D transverse analysis with computer-assisted reader software. Radiology 239(3):759–767. doi:10.1148/radiol.2392050483
Marquering HA, Dijkstra J, de Koning PJ, Stoel BC, Reiber JH (2005) Towards quantitative analysis of coronary CTA. Int J Cardiovasc Imaging 21(1):73–84. doi:10.1007/s10554-004-5341-y
Valverde FL, Guil N, Munoz J (2004) Segmentation of vessels from mammograms using a deformable model. Comput Methods Programs Biomed 73(3):233–247. doi:10.1016/S0169-2607(03)00043-9
Fishman EK, Ney DR, Heath DG, Corl FM, Horton KM, Johnson PT (2006) Volume rendering versus maximum intensity projection in CT angiography: what works best, when, and why. Radiographics 26(3):905–922. doi:10.1148/rg.263055186
Soto JA, Lucey BC, Stuhlfaut JW, Varghese JC (2005) Use of 3D imaging in CT of the acute trauma patient: impact of a PACS-based software package. Emerg Radiol 11(3):173–176. doi:10.1007/s10140-004-0384-x
Lado MJ, Cadarso-Suarez C, Roca-Pardinas J, Tahoces PG (2006) Using generalized additive models for construction of nonlinear classifiers in computer-aided diagnosis systems. IEEE Trans Inf Technol Biomed 10(2):246–253. doi:10.1109/TITB.2005.859892
Achenbach S (2005) Current and future status on cardiac computed tomography imaging for diagnosis and risk stratification. J Nucl Cardiol 12(6):703–713. doi:10.1016/j.nuclcard.2005.09.001
van Ooijen PM, Ho KY, Dorgelo J, Oudkerk M (2003) Coronary artery imaging with multidetector CT: visualization issues. Radiographics 23(6):e16. doi:10.1148/rg.e16
Farag AA, El Baz A, Gimelfarb G, El Ghar MA, Eldiasty T (2005) Quantitative nodule detection in low dose chest CT scans: new template modeling and evaluation for CAD system design. Med Image Comput Comput Assist Interv Int Conf Med Image Comput Comput Assist Interv 8(Pt 1):720–728
Juchems MS, Fleiter TR, Pauls S, Schmidt SA, Brambs HJ, Aschoff AJ (2006) CT colonography: comparison of a colon dissection display versus 3D endoluminal view for the detection of polyps. Eur Radiol 16(1):68–72. doi:10.1007/s00330-005-2805-y
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reimann, A.J., Tsiflikas, I., Brodoefel, H. et al. Efficacy of computer aided analysis in detection of significant coronary artery stenosis in cardiac using dual source computed tomography. Int J Cardiovasc Imaging 25, 195–203 (2009). https://doi.org/10.1007/s10554-008-9372-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-008-9372-7