Summary
Background
Parastomal hernia is observed in every third patient having a stoma. The different methods of repair still have a recurrence rate of 12–35 %. According to increasing literature data, placing a mesh with a preventive intention seems to decrease dramatically the possibility of parastomal hernia formation.
Methods
Between 2003 and 2009 we have placed a 3-dimensional mesh extraperitoneally, at the time of Miles operations (14 open and 3 laparoscopic cases, total 17). This group of patients was compared in a non-randomized, prospective, observational study to a control group consisting of the same number of patients in which open Miles operations were performed in the same observational period. From 2012, based on this experience, a new settled device was introduced to the market and applied for prevention and for repair of parastomal hernia at our institute.
Results
In the first trial, in the mesh group, after a 4.7-year mean follow-up period there were no parastomal hernia formations at all. In the non-mesh group, after 4.6-year mean follow-up period parastomal herniation was found in 55 % of the cases. In the mesh group, two strictures were observed as complications, both of which could be managed conservatively. Second trial’s interim results are also very promising.
Conclusions
Our experiences confirm the literature data that placing a mesh at the time of definitive stoma formation is preferable. The devices used by us unite the advantages of strengthening both sheets of the rectus abdominis muscle. In addition, changing the operative strategy to a laparoscopic approach gives an extra advantage to this procedure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A parastomal hernia (PSH) is by definition an incisional hernia related to an abdominal wall stoma [1–3]. The clinical diagnosis is mainly based on physical examination. Peristomal ultrasound or computed tomography (CT) can either strengthen the physical diagnosis or even reveal subclinical PSH; however, the only method that has been shown to accurately capture the rate of PSH is CT scan in prone position [4]. If available, 3-dimensional intrastomal ultrasonography has the potential to be the investigation of choice to differentiate between a bulge, a hernia, or a protrusion [5]. The true incidence of the PSH is uncertain because of underestimation and lack of reporting [6]. Data show a great variability, depending on the follow-up periods, methods of assessment, and type of stomas. Carne et al. [3] found 4.0–48.1 % incidence of PSH for end, and 0–38.1 % incidence for loop colostomies. The paraileostomy hernias are less frequent, (end: 1.8–28.3 %, loop ileostomy: 0–6.2 %). Some authors suggest that PSH is an inevitable consequence, rather than a complication of stoma formation [7]. In a recent prospective audit the overall rate of PSH was found to be 33 % in non-selected stoma patients [8]. Approximately one-third [9] of the patients with PSH undergo some kind of surgical correction because of related symptoms. Incarceration, perforation, or obstruction require immediate interventions, otherwise problems should range from mild discomfort and pain to intermittent obstructive episodes, or peristomal complications such as fistula. The most common indication for surgery is the difficulty in the stoma device application [8]. A large series of publications argue for mesh repair of PSH, which has the lowest recurrence rate (0–33 %), compared with local fascial repair (46–100 %) and relocation of the stoma (0–76 %) [2, 3, 6, 10]. In recent years, there have been encouraging results concerning preventive mesh implantation at the time of stoma formation [11, 12]. At the time of writing of this paper four randomized controlled studies [13–16] and other observational ones [8, 17–24] suggest that the best solution is to prevent hernia formation at the very beginning. This should be achieved by different methods of mesh implantation (extraperitoneal: onlay and sublay, or intraperitoneal). Open and laparoscopic [17, 19, 22] or both [24] approaches have been described.
In the present study, we describe implantation of Polypropylene hernia system large (PHSL; Ethicon inc. Johnson and Johnson, USA) or ULTRAPRO Hernia System Oval (UHSOV; Ethicon inc. Johnson and Johnson, USA) meshes at the time of stoma formation during Miles operations. These meshes were used in our institute from 2003 to 2009. The original method was developed further and served as the basis for an ongoing trial in which we are implanting newly designed 3-dimensional meshes, the Surgimesh® Parastomal (Aspide Medical). These special 3-dimensional devices have two patches and can be inserted as both onlay and sublay at the same time, thus an extra strengthening of the peristomal tissues can be achieved. Our objective was to assess the tolerance of these types of meshes and the reduction of PSH formation during a long-term follow-up period.
Methods
From 2003 to 2009, in a prospective, non-randomized, non-blinded, observational study, PHSL and UHSOV meshes were inserted around end sigmoideostomies on patients operated on with elective Miles operations and the results were compared with a group of patients with no mesh implantation. Indication for surgery was a tumor in the middle or lower rectum. Twenty-two cases were preoperatively treated with chemo-radiation. Patients above stage T3, or with evident distant metastases or having any other serious concomitant disease were excluded. The groups were homogenous in terms of their clinical and demographic characteristics. PHSL is constructed of an onlay patch of 4.5 × 10 cm and an underlay patch of 10 × 10 cm, while UHSOV’s onlay patch is 6 × 12 cm and underlay patch is 10 × 12 cm. Both of these specially designed 3-dimensional meshes have the same construction: two patches parallel to each other, being connected with a central tube having a diameter of 1.97 cm and height: 1.27 cm (See Fig. 1). PHSL is a non-absorbable heavyweight mesh, composed entirely of polypropylene. UHSOV is large-pored lightweight partially biodegradable mesh containing non-absorbable and absorbable materials in 50-50 % [10, 14]. Since these devices were originally designed for inguinal hernia repair, the prefabricated central hole of the mesh was sometimes too narrow to admit the intestine used for stoma formation. In case of diameter discrepancy, a V shape incision was made on the mesh’s central hole, which was then reinforced with 3/0 polypropylene running suture. Colostomy was formed in all cases through the rectus abdominis muscle in the left lower quadrant of the abdomen, premarked the day before the operation. The sigma loop prepared for colostomy was closed with a linear or endostapler. A 2.5–3-cm circular skin incision was performed at the premarked site, and with sharp and blunt preparation an approximately 10 × 5 cm plane was dissected above the anterior sheet of the rectus abdominis muscle. Through a longitudinal incision made on the fascia, the muscle fibers were split and an approximately 10 × 10 cm plane was created between the muscle and the posterior fascia (See Fig. 2). The mesh was introduced in the hole in a way that the superior patch of the device was laid in the epifascial layer (onlay), and the inferior one under the rectus muscle and above the posterior fascia and peritoneum (sublay). The mesh was fixed at the four corners with non-absorbable 2/0 monofilament sutures transfascially, in a way that sutures went through both layers of the mesh, the fasciae of the rectus abdominis muscle and the peritoneum. The knots were placed above the superior layer of the mesh. An incision was made on the peritoneum and the sigmoid trunk was gently pulled through the central tube of the mesh (See Fig. 3). The sigmoid was sutured only to the skin with interrupted mucocutan 3/0 absorbable sutures. In case of laparoscopic operations, a 10 mm trocar was introduced through the central hole of the mesh and through the peritoneum and the bowel was pulled out with an endoscopic forceps. The position of the mesh in the abdominal wall is shown in Fig. 4.
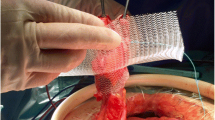
Surgimesh® Parastomal was developed and tested in our institute based on our previous experience with the PHSL and UHSOV meshes. Composed of 100 % non-woven, non-knitted, and non-absorbable polypropylene, it has the same construction of two circular parallel patches and a connecting central tube; the difference is that this central tube is available with three different internal diameters: 3.0, 3.5, and 4 cm. (see Fig. 5).
Repairing parastomal hernias with these kind of meshes is a more demanding procedure then the prevention. In this case, after resolving the parastomal hernia content and resection of the hernia sac 2 cm above the hernia opening, the colon was mobilized and dropped back into the abdomen. The aperture of the stoma was narrowed first by fascial sutures to the diameter of the colon and the inner patch of the mesh was placed between the hernia sac (peritoneum) and posterior fascia of the rectus abdominis muscle, since this plane is more easy to be dissected. Except for this difference, the next steps of the procedure are the same as it is described at the prevention.
Local ethical committee approval was obtained before each study introduction and additional informed consent regarding mesh implantation was obtained from patients in the mesh group.
Results
Between January 2003 and February 2004, PHSL mesh was implanted in 13 cases with preventive intent by the time of Miles operation. Due to financial reasons only four more operations were added between 2005 and 2009. In these cases UHSOV mesh was inserted. There were 14 open and 3 laparoscopic procedures. The mesh insertions were compared with a control group, consisting of 17 patients operated on without mesh implantation by open method in the same observational period. All cases were followed up at 3, 6, and 12 months postoperatively and then annually by two specialist surgeons. Controls were focused on PSH and all other possible complications.
The clinical diagnosis of parastomal hernia was made after removing the appliance by physical examination considering any palpable defect or bulge in close vicinity to the stoma while the patient was performing a Valsalva manaeuvre in both supine and standing position.
Colonoscopy, peristomal wall ultrasound, and CT were performed in all cases of PSH and in the cases when physical diagnosis was not certain. Patients clinical and demographic characteristics are shown in Table 1.
Mean follow-up period was 56.4 months (4.7 years) and 55.2 months (4.6 years), respectively. There was no postoperative mortality and at the end of the first postoperative year all patients were alive. After 6 years only 11 patients in the mesh group and 9 in the control group were available for control. From the remaining, 3 patients had been lost to the survey and 11 died of cancer. There was no mesh-related mortality.
Among the 11 patients of the mesh group available at the 5-year follow-up, no PSH have been found. From the nine patients of the control group five (55 %) PSH were diagnosed. The incidences of PSH and other complications in the two groups at the end of the first and fifth postoperative year are presented in Tables 2 and 3.
Three of the recurrences underwent mesh repair with the same type of mesh we used in the study and had no recurrence in the first 2 postoperative years. Each of the two strictures in the mesh group (18.1 %) were successfully treated by only one dilatation manoeuvre with Hegar series. There was no reoperation, necrosis, rejection of the mesh prolapse, fistula, infection, or any other complication in connection with mesh implantation. In the control group, one superficial postoperative necrosis was corrected locally. There was no reoperation in this group either, except the three PSH reconstructions mentioned above.
From 2012 to date, Surgimesh® Parastomal was implanted in seven cases for prevention in Miles procedure and in three cases for reconstruction of parastomal hernias. These cases are involved in a single institute randomized controlled trial, in which only 3 and 6 months results are available so far. These results are also very promising, but can’t be published until longer follow-up period results are not available.
Discussion
The literature clearly states that mesh repair of PSH is superior in terms of recurrence and morbidity compared with local fascial reconstruction and transposition of the stoma. In case of large parastomal eventerations or when concomitant incisional hernia is present there is still a choice to relocate the stoma and reinforce both the former and the new site of the stoma with mesh [10, 25]. The great incidence of PSH and comorbidities associated with their repair led to the logical idea of preventing them by inserting a mesh at the time of stoma formation. Creating a hole in the abdominal wall’s structure means by itself a constant enlargement tendency of the trephine opening [2] and causes disturbance in the wound healing pathways at the aperture [26]. According to the law of Laplace, forces working on the edge of the trephine opening are related to the radius of the opening. Confirming De Ruiter and Bijnen’s experimental study [27], the tangential force activating on the edge of the aperture (Ftang) is related to the radial force on the abdominal wall (Frad) and the radius of the opening (R): Ftang = Frad × R2. In turn, the radial force is related to the intra-abdominal pressure and the radius of the abdominal cavity. Consequently, tangential forces are greater in patients with large waist circumferences, which is an independent risk factor for hernia formation [28]. Obese patients (large waist circumference) with large openings (loop stomas rather than end stomas, colostomies rather than ileostomies) should theoretically be at the highest risk of hernia formation [3]. Recent prospective studies confirmed that age [8], and subcutaneous fat thickness ≥ 23 mm [16] are also independent risk factors for PSH formation. Other patient related factors such as smoking, malnutrition, diabetes, as well as the so-called disease process related factors (raised intra-abdominal pressure caused by chronic obstructive pulmonary disease (COPD), constipation, prostate hypertrophy, ascites, postoperative sepsis, corticosteroid therapy, and malignancy) are accepted risk factors of PSH formation [6, 8], despite the uncertainty caused by the relatively small number of investigated patients [29]. Considering the technical factors, only the aperture size was proven to be an independent predictor in a multivariate analysis [8]. There is controversy about the consequences of hernia formation by pulling out the intestine through or lateral to the rectus abdominis muscle [3, 8, 30, 31]. Lateral fixation or suturing the bowel to the fascia, as well as the emergency situation were not proven to significantly influence PSH formation [3, 30]. Many surgeons prefer to construct an extraperitoneal path when creating a colostomy [32, 33], a technique which seems to decrease the PSH formation. Comparative controlled studies cross-checking this technique to other demonstrated techniques like mesh prevention, however are still missing.
Learning the lesson from various descriptive studies of PSH mesh repair, when extraperitoneal mesh was inserted with different techniques either onlay or sublay [3, 34–36], the aim of our study was to develop a technique where both surfaces of the rectus abdominis muscle could be reinforced. Our concept was supported by anatomical and biomechanical considerations. Rath et al. [37] found that the anterior rectus sheeth is more resistant to traction below the arcuate line than above it, whereas the resistance to the simulated intra-abdominal pressure shows an opposite pattern, and it is greater than in the posterior layer at all levels. The resistance to traction of the posterior rectus sheet is virtually uniform over both areas. The resistance to pressure is slightly less in the region of the arcuate line. The authors suggested caution in placing prostheses in weaker zones. In our opinion, the weaker portions of the rectus abdominis muscle sheets must also be strengthened. As the posterior layer is much less resistant to intra-abdominal pressure and the anterior sheet is less resistant to tractions above the arcuate line, there is a need to reinforce both layers. In addition to this, the two plates of the mesh must move together in the same direction while the muscle is contracting, otherwise there is a chance to erode into the bowel. The buildup of the meshes used in our series allows both covering the two sides of the rectus abdominis muscle and the codirectional movements of the two plates. We believed that this buildup, which is additionally strengthened by the transfascial sutures, is essential to avoid possible bowel erosion caused by the mesh during the normal abdominal wall movements. Beyond that, the transfacial sutures serve not only against seroma formation by keeping the layers of the abdominal wall together, but theoretically they have role in avoiding any asymmetrical shrinking of the device in the later phases. The inner surface of the polypropylene tube, having a very strong adhesive feature to the visceral peritoneum of the bowel, prevents the formation of a sliding hernia in the early postoperative period.
We started preventive mesh insertion early in 2003 and preliminary results of that study were published in 2004 [38]. A total of 13 cases of open approach PHSL mesh insertion at the time of the Miles operation were reported. From 2005 to 2009 another four cases were operated on, three of them by laparoscopic approach. They were compared with 17 control patients without mesh implantation exclusively operated on through the open approach. Although there are contradictory results concerning the use of meshes exclusively made by polypropylene [20, 21, 34, 35, 39, 40], in our experience PHSL and UHSOV caused no complication and proved to be safe in preventing PSH formation. There was no perforation, penetration, or any other bowel damage observed in the group of patients with mesh during the follow-up period. The two strictures noticed might have occurred because of the inappropriate diameters. These cases were successfully treated by one consecutive Hegar dilatation in each case. We investigated and found no mesh penetration with colonoscopy in these cases either, but these unusual complications drew the attention to adapt properly the size of the hole in the mesh to the individual patient.
From nine available patients at the 6-year follow-up (median 4.6 years) five (55 %) PSH were observed in the control group. In the mesh group’s 11 available patients no PSH was observed in the same observational period. These results are comparable or even better than other results regarding PSH formation [13–16]. However, the small number of patients included in our series could limit the interpretation value.
Admitting the favorable buildup of the device, it was our desire to possess prefabricated meshes with two or three fixed diameters of the central tube. This goal was achieved by the new series of Surgimesh® Parastomal meshes, developed and tested in close cooperation between our team and the manufacturer. A prospective randomized trial with promising interim results is under way in our unit to evaluate the use of these products for not only prevention, but also for repair of PSH.
In the course of practicing the demonstrated technique we came to realize how to simplify the procedure even more. If the inner side of the inferior patch of the device would be coated with silicone or other antiadherent material that could allow putting this patch in intra-abdominal position, there would be no need for the somehow demanding step dissecting the sublay plane. This modification is going to be tested in the near future.
It is still to be answered whether we need preventive mesh insertion in all cases of stoma formation. There is still reluctance among surgeons to put a mesh in a potentially contaminated field like a stoma. The results show that this problem can be avoided with the appropriate technique [23]. Other important questions such as the choice of the mesh material, mesh design, location of the mesh must be analyzed in large double blinded and randomized clinical trials.
Conclusion
Patients at high-risk of PSH formation and those with potential long-term survival will surely benefit from mesh prevention at the time of stoma formation. We found very positive results in long-term follow-up regarding PSH formation and mesh-related complications in patients who received a special designed 3-dimensional extraperitoneal mesh with preventive intent. This device unites the advantages of strengthening both sheets of the rectus abdominis muscle, and changing the operative strategy to laparoscopic approach gives an extra advantage to this procedure. Evaluating the efficacy of newer prefabricated products with different internal diameters is under way at our institute.
References
Nagy A, Jánó Z. Parastomal hernias (Parastomalis sérvek). MaSeb. 2010;63(5):335–9.
Kasperk R, Willis S, Klinge U, Schumpelick V. Update on incisional hernia. Parastomal hernia. Chirurg. 2002;73(9):895–8.
Carne PWG, Robertson GM, Frizelle FA. Parastomal hernia. Br J Surg. 2003;90(9):784–93.
Jänes A, Weisby L, Israelsson LA. Parastomal hernia: clinical and radiological definitions. Hernia. 2011;15(2):189–92.
Strigård K, Gurmu A, Näsvall P, Påhlman P, Gunnarson U. Intrastomal 3D ultrasound; an inter-and intra-observer evaluation. Int J Colorectal Dis. 2012. doi:10.1007/s00384-012-1526-3.
Dykes SL. Ostomies and stomal therapy. American Society of Colon & Rectal Surgeons, Core Subjects. 2010. Available from: http://www.fascrs.org/physicians/education/core_subjects/.
Berger D, Bientzle M. Laparoscopic repair of parastomal hernias—a single surgeon’s experience in 66 patients. Dis Colon Rectum. 2007;50(10):1668–73.
Pilgrim CH, McIntyre R, Bailey M. Prospective audit of parastomal hernia: prevalence and associated comorbidities. Dis Colon Rectum. 2010;53(1):71–6.
Israelsson LA. Preventing and treating parastomal hernia. World J Surg. 2005;29(8):1086–9.
Voeller GR. Innovations in ventral hernia repair. Surg Technol Int. 2007;16:117–22.
Wijeyekoon SP, Gurusamy K, El-Gendy K, Chan CL. Prevention of parastomal herniation with biologic/composite prosthetic mesh: a systematic review and meta-analysis of randomized controlled trials. J Am Coll Surg. 2010;211(5):637–45.
Tam KW, Wei PL, Kuo LJ, Wu CH. Systematic review of the use of a mesh to prevent parastomal hernia. World J Surg. 2010;34:2723–29.
Janes A, Cengiz Y, Israelsson LA. Preventing parastomal hernia with a prosthetic mesh: a 5-year follow-up of a randomized study. World J Surg. 2009;33(1):118–21; discussion 122–3.
Serra-Aracil X, Bombardo-Junca J, Moreno-Matias J, et al. Randomized, controlled, prospecive trial of the use of a mesh to prevent parastomal hernia. Ann Surg. 2009;249(4):583–7.
Hammond TM, Huang A, Prosser K, Frye JN, Williams NS. Parastomal hernia prevention using a novel collagen implant: a randomised controlled phase 1 study. Hernia. 2008;12(5):475–81.
López-Cano M, Lozoya-Trujillo R, Quiroga S, et al. Use of a prosthetic mesh to prevent parastomal hernia during laparoscopic abdominoperineal resection: a randomized controlled trial. Hernia. 2012. doi:10.1007/s/10029-012-0952-z.
López-Cano M, Lozoya-Trujillo R, Espin-Basany E. Prosthetic mesh in parastomal hernia prevention. Laparoscopic approach. Dis Colon Rectum. 2009;52(5):1006–7.
Bayer I, Kyzer S, Chaimoff C. A new approach to primary strengthening of colostomy with Marlex® mesh to prevent paracolostomy hernia. Surg Gynecol Obstet. 1986;163:579–80.
Berger D. Prevention of parastomal hernias by prophylactic use of a specially designed intraperitoneal onlay mesh (Dynamesh IPST®). Hernia. 2008;12(3):243–6.
Gögenur I, Mortensen J, Harvald T, Rosenberg J, Fischer A. Prevention of parastomal hernia by placement of a polypropylene mesh at the primary operation. Dis Colon Rectum. 2006;49(8):1131–5.
Vijayasekar C, Marimuthu K, Jadhav V, Mathewm G. Parastomal hernia: is prevention better than cure? Use of preperitoneal polypropylene mesh at the time of stoma formation. Tech Coloproctol. 2008;12(4):309–13.
Janson AR, Jänes A, Israelsson LA. Laparoscopic stoma formation with a prophylactic prosthetic mesh. Hernia. 2012;14. doi:10.1007/s10029-010-0673-0.
Jänes A, Cengiz Y, Israelsson LA. Experiences with prophylactic mesh in 93 consecutive ostomies. World J Surg. 2010;34:1637–40.
Hauters P, Cardin JL, Lepere M, Valverde A, Cossa JP, Auvray S. Prevention of parastomal hernia by intraperitoneal onlay mesh reinforcement at the time of stoma formation. Hernia. 2012. doi:10.1007/s/10029-012-0947-9.
García-Vallejo L, Concheiro P, Mena E, Baltar J, Baamonde I, Folgar L. Parastomal hernia repair: laparoscopic ventral hernia meshplasty with stoma relocation. The current state and a clinical case presentation. Hernia. 2010;14. doi:10.1007/s10029-009-0617-8.
Hammond TM, Chin-Aleong J, Navsaria H, Williams NS. Human in vivo cellular response to a cross-linked acellular collagen implant. Br J Surg. 2008;95(4):438–46.
De Ruiter P, Bijnen AB. Succesful local repair of paracolostomy hernia with a newly developed prosthetic device. Int J Colorectal Dis. 1992;7:132–4.
De Raet JD, Haentjens P, et al. Waist circumference is an independent risk factor for the development of parastomal hernia after permanent colostomy. Dis Colon Rectum. 2008;51(12):1806–9.
Helgstrand F, Gögenur I, Rosenberg J. Prevention of parastomal hernia by the placement of a mesh at the primary operation. Hernia. 2008;12(6):577–82.
Londono-Schimmer EE, Leong AP, Phillips RK. Life table analysis of stomal complications following colostomy. Dis Colon Rectum. 1994;37:916–20.
Cingi A, Cakir T, Sever A, Aktan AO. Enterostomy site hernias—a clinical and computerized tomographic evaluation. Dis Colon Rectum. 2006;49(10):1559–63.
Hamada M, Ozaki K, Muraoka G, Kawakita N, Nishioka Y. Permanent end-sigmoid colostomy through the extraperitoneal route prevents parastomal hernia after laparoscopic abdominoperineal resection. Dis Colon Rectum. 2012;55(9):963–9.
Leroy J, Diana M, Callari C, et al. Laparoscopic extraperitoneal colostomy in elective abdominoperineal resection for cancer: a single surgeon experience. Colorectal Dis. 2012;14(9):e618–22. doi:10.1111/j.1463-1318.2012.03015.x.
Aldridge AJ, Simson JN. Erosion and perforation of colon by synthetic mesh in a recurrent paracolostomy hernia. Hernia. 2001;5(2):110–2.
Steele SR, Lee P, Martin MJ, Mullenix PS, Sullivan ES. Is parastomal hernia repair with polypropylene mesh safe? Am J Surg. 2003;185(5):436–40.
Martinez-Munive A, Quijano-Orvananos OF, Padilla-Longoria R, Zavala-Ruiz JS, Hesiquio-Silva R. Intraparietal mesh repair for parastomal hernias. Hernia. 2000;4(4):272–4.
Rath AM, Zhang J, Chevrel JP. The sheath of the rectus abdominis muscle: an anatomical and biomechanical study. Hernia. 1997;1(3):139–42.
Nagy A, Kovács T, Bognár J, Mohos E, Lóderer Z. Parastomal hernia repair and prevention with PHSL type mesh after abdomino-perineal rectum exstirpation. Zentralbl Chir. 2004;129(2):149–52.
Longman RJ, Thomson WH. Mesh repair of parastomal hernias—a safety modification. Colorectal Dis. 2005;7(3):292–4.
Morris-Stiff G, Hughes LE. The continuing challenge of parastomal hernia: failure of a novel polypropylene mesh repair. Ann R Coll Surg Engl. 1998;80:184–7.
Conflict of interest
The authors declare that there are no actual or potential conflicts of interest in relation to this article. Surgimesh® Parastomal meshes were provided free of charge by Aspide Medical.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jánó, Z., Nagy, A. Results of 3-dimensional mesh implantations at the time of Miles operation to prevent parastomal hernia. Eur Surg 46, 25–31 (2014). https://doi.org/10.1007/s10353-013-0245-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10353-013-0245-5