Abstract
Background
Peritoneal dialysis (PD) is the preferred mode of kidney replacement therapy (KRT) in infants and young children with kidney failure. Hemodialysis (HD) is used less often due to the technical challenges and risk of complications in smaller patients. There are limited data on chronic HD in this patient population.
Methods
This was a retrospective study of children younger than 24 months on HD and PD in the North American Pediatric Renal Trials and Collaborative Studies (NAPRTCS) registry between January 1992 and December 2018. We compared demographic, clinical, and laboratory data and outcomes, including patient survival and kidney transplantation.
Results
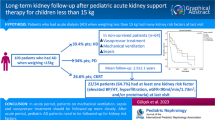
We identified 1125 infants and toddlers younger than 2 years of age who initiated KRT from January 1992 to December 2018. Of those, 1011 (89.8%) initiated peritoneal dialysis and 114 (10.2%) initiated hemodialysis. Median (IQR) age at HD onset was 12 (5.6–18.7) months compared to 4.6 (0.8–11.7) months at PD onset (p < 0.001). The primary cause of kidney failure with replacement therapy was congenital anomalies of the kidney and urinary tract (56.2% of PD versus 39.5% of HD group). Patients on HD had superior growth and nutrition markers than those on PD. Patient survival was similar between the two groups.
Conclusions
While HD may not be the modality of choice for chronic KRT in younger children, 10% of children younger than 24 months of age receive maintenance HD and the numbers have increased over time. Patient survival on dialysis is similar irrespective of dialysis modality.
Graphical abstract
A higher resolution version of the Graphical abstract is available as Supplementary information.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Management of infants and toddlers with kidney failure with replacement therapy (KFRT) can be challenging. Peritoneal dialysis (PD) is often the preferred mode of KRT in infants and young children with KFRT, particularly in those younger than 5 years of age [1, 2]. Maintenance hemodialysis (HD) is technically more challenging in younger age groups and can be associated with high morbidity and mortality [2,3,4,5]. Challenges include relatively higher extracorporeal blood volume of HD circuits necessitating blood priming and the need for long-term vascular access with its associated risk of catheter-related bloodstream infections [6, 7]. Younger patients with KFRT, particularly those with congenital anomalies of the kidneys and urinary tract (CAKUT), may have higher nutrition and volume requirements necessitating more frequent dialysis than the traditional thrice-weekly sessions [1, 8,9,10].
According to the NAPRTCS Dialysis Report, 14.9% of children on maintenance dialysis were younger than 2 years at initiation of dialysis therapy (including both PD and HD) from 2002 to 2011 compared to 13.4% between 1992 and 2001 [11]. The same data demonstrate an overall increase in the number of children receiving HD despite the technical challenges. The proportion of children receiving HD increased from 36% (1992–2001) to 42% (2001–2011) [11].
While there has been an increase in the number of infants and young children receiving HD treatment, there are limited current data on the outcomes of this population in North America. Most data on infants undergoing HD are from small single-center studies [3,4,5, 12]. Feinstein et al. compared outcomes of 20 patients who initiated dialysis at less than 1 year of age with 14 patients who started dialysis at age 1–3 years [3]. Mortality in those less than 1 year old was 50% compared to 7.1% in those in the older group. The younger cohort also had significantly longer hospital stays during the first 3 months of dialysis compared to those in the older group [3]. More patients in the older group were transplanted during the study period (92% versus 45%). Another single-center study examined 18 infants who initiated HD therapy at less than 12 months of age, followed from 1997 to 2013 [4]. Pollack et al. reported a 39% mortality rate (7 of 18) over the 16-year period of the study. Five (28%) eventually received a kidney transplant [4]. Interestingly, Quinlan et al. reported no deaths in their study of 10 patients (< 10 kg, ages ranging from 4 to 22 months at initiation of HD) over a 10-year period, which was attributed to the small cohort size and general lack of co-morbidities [12].
We aimed to use the NAPRTCS dialysis registry to compare clinical characteristics and outcomes of patients younger than 2 years of age on HD and PD. We also compared the HD practice patterns in children younger than 2 years of age with patients between 2 and 5 years of age.
Methods
Patients and methods
NAPRTCS is a voluntary registry database that collects information from participating centers throughout North America on patients younger than 21 years of age at the time of enrollment. The details of NAPRTCS registry have been previously described [11, 13]. The registry has three arms: chronic kidney disease, dialysis, and kidney transplant. Local institutional review board approval is obtained at each center for data entry and the data collected from each center are sent on a voluntary basis to a data coordinating center. Information is collected upon initial registration, with follow-up status forms submitted 30 days and 6 months after initiation of dialysis and then every 6 months thereafter.
For this analysis, we queried the dialysis registry for children who were less than 2 years of age at the time of their first HD or PD course after enrolling in NAPRTCS, and those who initiated HD at 2–5 years of age. We included patients from January 1992 to December 2018 in the NAPRTCS dialysis registry. Those who underwent dialysis for < 3 consecutive months were excluded. We divided the study period into three eras, 1992–2000, 2001–2009, and 2010–2018, to examine the proportion of patients initiating HD over time.
Statistical analysis
Categorical variables were summarized with counts and proportions and compared using Pearson’s chi-squared test or Fisher’s exact test (mid-p) in cases where at least 20% of expected cell counts were under 5. Continuous variables were summarized with median and interquartile range (IQR) and compared using the Wilcoxon rank-sum test. Clinical outcomes were reported by visit (30 days, 6 months, and 12 months) and independent comparisons of outcomes by modality (PD or HD) were made. Kaplan–Meier survival curves were generated by modality for the outcome variables of death and transplant. Differences in the distributions of time to the outcome variable between modalities were evaluated using the log-rank test. For time-to-event analyses, data were censored at 24 months of follow-up or dialysis modality termination if termination occurred within 24 months of dialysis initiation. The Kaplan–Meier estimates for probability of survival and probability of transplant at 1 year were reported with 95% CI using the log–log method. Multivariate Cox proportional hazards models were fit, adjusting for age, sex, non-White race, weight z-score, primary kidney disease, and era. In addition, cumulative incidence curves were generated by modality, treating transplant, modality change, and death as competing events.
Results
Patient characteristics
We identified 1125 infants and toddlers younger than 2 years of age who initiated KRT from January 1992 to December 2018. Of those, 1011 (89.8%) initiated peritoneal dialysis and 114 (10.2%) initiated hemodialysis. Baseline characteristics by initial dialysis modality are shown in Table 1. Some patients in both cohorts had a history of dialysis prior to being registered in NAPRTCS; however, additional details of previous dialysis were not available (61.6% in HD cohort and 54.5% in PD cohort). The median (IQR) age of patients starting HD was 12 (5.6–18.7) months compared to 4.6 (0.8–11.7) months for those starting PD (p < 0.001). There was no significant difference in the sex distribution between the cohorts (36% females in HD, 33% in PD; p = 0.544). There were more non-White patients who initiated HD compared to PD (50.9% vs. 38.4%, p = 0.013). The most common underlying etiology for KFRT for the entire cohort was CAKUT. However, CAKUT was less common in the HD cohort compared to PD (39.5% vs. 56.2%, p = 0.005). One hundred and twenty-two NAPRTCS centers contributed to this study. There was considerable variability in the size of the centers, with patient enrollment ranging from 1 to 70 patients. Twenty-eight centers (25%) had more than 25 patients in this study.
Dialysis initiation era
We examined the proportion of children younger than 2 years of age initiating HD over the study period which was divided into three eras: 1992–2000, 2001–2009, and 2010–2018. The proportion of patients initiating HD increased over the three eras: 6.8%, 11.1%, and 16.2%, respectively (Cochran–Armitage p-value < 0.001).
Clinical outcomes of Infants (< 2 years) on hemodialysis versus peritoneal dialysis
Hemoglobin and anemia management
Median hematocrit values for the HD cohort were significantly lower at 30 days and 6 months post dialysis initiation compared to the PD cohort (Table 2). Similarly, more patients in the HD cohort had a hematocrit < 30% at 30 days (68.9% for HD vs. 42.4% for PD, p < 0.001) and 6 months (55.2% for HD vs. 34% for PD, p < 0.05). Use of erythropoiesis-stimulating agents (ESA) was similar between the cohorts. At each follow-up point, a higher proportion of patients undergoing HD were documented as having received a blood transfusion. Data on the dose of ESA were not available.
Growth and nutrition
Overall, markers of growth and nutrition were superior for children on HD compared to PD. Weight and Z-scores at dialysis initiation and at 30 days post initiation were significantly higher in patients who initiated HD compared to those starting PD (Table 2). While there was improvement in both groups over time, children on PD continued to have lower weight Z-scores. Similar trends were seen in linear growth. There was no significant difference in the proportion of patients prescribed growth hormone in the two cohorts. Serum albumin remained significantly higher in patients on HD at all follow-up points.
Hypertension
Hypertension was defined as having either systolic blood pressure (BP) or diastolic BP greater than or equal to the 95th percentile for age and sex. There was no significant difference in the incidence of hypertension between the HD and PD cohorts. There were limited data on blood pressure control or antihypertensive drug use.
Hemodialysis prescription
We compared the HD prescription between children younger than 2 years of age and those between 2 and 5 years of age (Table 3). At 30 days post dialysis initiation, a higher proportion of children younger than 2 years of age had dialysis sessions that lasted less than 3 h (63.3% vs. 53.8%, p > 0.05) and underwent dialysis 4 or more times per week (25% vs. 2.8%, p < 0.05). In the younger patients, 75% achieved a single pool Kt/V > 1.2 compared to 91% in the older cohort (p < 0.001).
Long-term outcomes
The most common reason for termination of dialysis was transplantation. The proportion (95% CI) terminating for transplant was 28% (19–40) for the HD cohort and 16% (14–19) in the PD cohort. Time to transplantation was similar for patients on PD or HD between 0 and 24 months after dialysis initiation (19.5 months for HD and 23.8 months for PD, p = 0.1; Fig. 1). For time to transplantation, no significant difference between infants on HD vs. PD was found in Cox proportional hazards model stratified by age (< 1 year vs > = 1 year) and adjusted for non-White race, sex, weight Z-score, primary kidney disease (CAKUT vs. non-CAKUT), and era. The hazard ratio for HD versus PD was 1.20 (95% CI: 0.83, 1.74).
Patient survival over 24 months post dialysis initiation was also similar for the 2 cohorts (96% for HD vs. 91% for PD, p = 0.057; Fig. 2). No significant difference in survival between infants on HD vs. PD was found in a Cox proportional hazards model adjusted for age, non-White race, sex, weight Z-score, primary kidney disease (CAKUT vs. non-CAKUT), and era. The hazard ratio for HD versus PD was 1.34 (95% CI: 0.23, 2.37).
HD patients were more likely to terminate their dialysis course with a switch in modality in the first 11 months after dialysis initiation compared to other reasons for termination, after which transplantation became the predominant reason for termination (Table 4; Figure S1). In contrast, PD patients were less likely to switch modality (Figure S2). The cumulative incidence of transplantation was similar between PD and HD patients.
Discussion
We present data from the NAPRTCS dialysis registry comparing clinical characteristics and outcomes of patients less than 2 years of age on HD and PD. In addition, we describe the HD practice patterns in children less than 2 years of age and those between 2 and 5 years of age.
Overall, 10% of infants and children in this age group on maintenance dialysis received HD, and the proportion of patients younger than 2 years of age on HD has increased over time. Infants and children younger than 2 years starting HD were more likely to be older and non-White compared to those starting PD. Those on HD had superior markers of nutrition such as weight and height Z-scores and albumin, but lower hematocrit values compared to those on PD, despite similar rates of ESA use. The time to transplantation was slightly shorter for patients on HD compared to PD, and at each time point HD patients had a higher probability of transplantation, but this was not statistically significant. Probability of patient survival was independent of modality, similar to what has been described previously.
Hemodialysis is not the preferred first-line modality of KRT in young children. Peritoneal dialysis has several advantages over HD, including no need for vascular access, ability to perform dialysis at home, and fewer restrictions on diet and daily fluid intake. However, PD may not always be feasible due to relative or absolute contraindications, such as a complex abdominal anatomy, and in those situations, HD is the primary modality [1]. Hemodialysis may also be a secondary modality in patients who were previously on PD after modality failure or due to other psychosocial reasons [2, 3, 10]. Additionally, HD may also be the initial modality in those who need to initiate dialysis acutely due to their underlying disease. As stated previously, there are challenges associated with HD in younger age groups, including hemodynamic instability at initiation due to higher extracorporeal volumes, and need for more frequent dialysis to achieve appropriate fluid balance [1, 6, 7]. Our data show that 10% of children less than 2 years of age receive maintenance KRT with HD. This is less than what was reported in the European Society for Pediatric Nephrology (ESPN)/European Renal Association-European Dialysis and Transplant Association (ERA–EDTA) Registry study which is the largest study of long-term outcomes among infants on HD [2]. Vidal et al. found that 13.5% of infants younger than 12 months of age initiated HD therapy [2]. However, our data show that the proportion of patients initiating HD is increasing over time from 6.8% (1992–2000) to 16.2% (2010–2018). This may be in part due to improvement in HD technology over the recent years, including development of pediatric tubing, smaller, high flux filters with more biocompatible synthetic membranes, and improved methods of assessing dry weight with hematocrit monitoring [14].
We observed a difference in the demographic characteristics of children younger than 2 years on dialysis. There is a significant difference in race between the two dialysis modalities with more non-White patients receiving HD than PD. This is similar to what has been shown previously that White children are more likely to receive PD [15, 16]. In the Medicare-ESRD registry, Furth et al. reported that Black children were significantly more likely than White children to receive hemodialysis (OR, 2.4; 95% CI: 1.7, 3.5), even after controlling for other patient and facility characteristics in multivariate analysis [15, 16]. They suggested possible explanations including differences in socioeconomic status, cultural differences and perspectives on home modalities of KRT, access to healthcare, and also systematic racial bias [16]. We were unable to do additional analyses as data on other social determinants of health were incomplete, especially in the earlier years of the registry.
In addition to the technical challenges noted with the HD process itself, younger children may also need more frequent dialysis. Occasionally, younger patients may require a proportionately greater fluid intake to provide the appropriate nutrition for optimal growth. Studies have also shown that intensified dialysis with shorter, more frequent sessions may improve growth in some patients [8, 17]. In our study, a significantly higher proportion of children less than 2 years of age (25%) underwent hemodialysis 4 or more times per week when compared to children aged 2–5 years (2.8%). In the younger patients, only 75% achieved a single pool Kt/V > 1.2 compared to 91% in the older cohort. This was likely secondary to shorter, more frequent dialysis sessions in the younger cohort. Paglialonga et al. described in the Italian Pediatric Dialysis Registry that of 21 infants aged < 2 years of age, 64.7% had 4 or more HD sessions per week at the start of therapy [18]. While our proportion of patients receiving more frequent dialysis was smaller, it further affirms that infants and children < 2 years have a higher burden of care on HD compared to those in older groups [18]. This may contribute to the “physical, psychological and financial toll” that families of children face while on dialysis and is an important aspect when choosing KRT modality [19].
Children with KFRT may continue to have challenges with growth even after initiation of KRT. In our study, children receiving HD had superior markers of growth and nutrition at initiation of dialysis and at follow-up compared to those on PD. This is in contrast to previous data from Kaiser et al. which showed that continuous ambulatory peritoneal dialysis (CAPD) may be superior to cycler/intermittent PD (CCPD) and HD [20]. Turenne et al. looked at growth rates between PD, HD, and transplant and found that in younger children (6 months–4 years of age), growth rates were higher in those receiving a transplant compared to dialysis but were similar among the different dialysis modalities (CAPD, CCPD, and HD) [21]. Various factors may affect growth on dialysis, including provision of intradialytic parenteral nutrition (IDPN) during HD, absorption of glucose during PD, and infections for both modalities. Vidal et al. have previously reported that infants younger than 1 year of age on PD who did not experience peritonitis had better growth [2]. We had limited data on IDPN or peritonitis or catheter-associated blood stream infections to study this.
Patients on HD had lower hematocrits than those on PD, despite similar ESA use between the two groups. This is similar to what was seen in the ESPN/ERA–EDTA registry. There can be multiple reasons for this. Patients undergoing HD may have greater blood loss with the HD circuit or may have fluid overload around timing of blood draw [2]. Studies have also shown that patients undergoing hemodialysis, particularly those using central catheters and with history of line infections, may have chronic inflammation and endothelial dysfunction, which may lead to a lower hematocrit [22, 23].
Despite these differences between the patients on HD and PD, the transplantation and mortality rates were similar. In our study, patient survival on dialysis was similar irrespective of dialysis modality 24 months after dialysis initiation. Vidal et al. found similar findings in their European cohort as well [2]. In contrast, Mitsnefes et al. reported a potential protective effect of PD over HD in children less than 5 years of age in the United States Renal Data System [24]. It is possible that the effect of technology and development and management of HD is seen in examining these studies over time.
Our study has some limitations. NAPRTCS is a voluntary registry and dependent upon the reporting of information from individual centers, with inherent limitations such as sampling bias and reporter bias. There was significant variability in the size of the centers, with patient enrollment ranging from 1 to 70 patients, and most centers contributing fewer than 25 patients. Some of the practice patterns we see could be from variations in institutional practice and not necessarily standard of care. In addition, there are missing data on laboratory markers at follow-up time points. The timeframe of our cohort spans 26 years, over which technology and practice patterns have changed. A large proportion of patients in both groups had some dialysis history prior to enrollment in NAPRTCS. The registry does not collect additional information on the previous therapy, and we were unable to determine if the prior dialysis was acute KRT, or another form of maintenance dialysis. We also do not have information on whether there was a change in modality prior to entry into the registry and the reason for that change. Finally, we were unable to control for confounders such as socioeconomic status and comorbid conditions in this analysis, since those data elements were only added to the NAPRTCS registry in 2018.
This NAPRTCS analysis demonstrates that while HD may not be the modality of choice for chronic KRT in younger children, 10% of children younger than 24 months of age receive maintenance HD and the numbers have increased over time. Patient survival on dialysis is similar irrespective of dialysis modality. More data are needed on HD dose and prescription in this age group, so that optimal dialysis may be provided without increasing burden on families.
Availability of data and material
The datasets generated during and/or analyzed during the current study are not publicly available but may be available from NAPRTCS with IRB approval.
Code availability
Not applicable.
References
Chua AN, Warady BA (2017) Care of the pediatric patient on chronic dialysis. Adv Chronic Kidney Dis 24:388–397
Vidal E, van Stralen KJ, Chesnaye NC, Bonthuis M, Holmberg C, Zurowska A, Trivelli A, Da Silva JEE, Herthelius M, Adams B, Bjerre A, Jankauskiene A, Miteva P, Emirova K, Bayazit AK, Mache CJ, Sanchez-Moreno A, Harambat J, Groothoff JW, Jager KJ, Schaefer F, Verrina E, European Society for Pediatric Nephrology (ESPN)/European Renal Association-European Dialysis and Transplant Association Registry (ERA-EDTA) (2017) Infants requiring maintenance dialysis: outcomes of hemodialysis and peritoneal dialysis. Am J Kidney Dis 69(617):625
Feinstein S, Rinat C, Becker-Cohen R, Ben-Shalom E, Schwartz SB, Frishberg Y (2008) The outcome of chronic dialysis in infants and toddlers–advantages and drawbacks of haemodialysis. Nephrol Dial Transplant 23:1336–1345
Pollack S, Eisenstein I, Tarabeih M, Shasha-Lavski H, Magen D, Zelikovic I (2016) Long-term hemodialysis therapy in neonates and infants with end-stage renal disease: a 16-year experience and outcome. Pediatr Nephrol 31:305–313
Shroff R, Wright E, Ledermann S, Hutchinson C, Rees L (2003) Chronic hemodialysis in infants and children under 2 years of age. Pediatr Nephrol 18:378–383
Raina R, Vijayaraghavan P, Kapur G, Sethi SK, Krishnappa V, Kumar D, Bunchman TE, Bolen SD, Chand D (2018) Hemodialysis in neonates and infants: a systematic review. Semin Dial 31:289–299
Kaur A, Davenport A (2014) Hemodialysis for infants, children, and adolescents. Hemodial Int 18:573–582
Haffner D (2020) Strategies for optimizing growth in children with chronic kidney disease. Front Pediatr 8:399
Gotta V, Marsenic O, Pfister M (2018) Age- and weight-based differences in haemodialysis prescription and delivery in children, adolescents and young adults. Nephrol Dial Transplant 33:1649–1660
Müller D, Goldstein SL (2011) Hemodialysis in children with end-stage renal disease. Nat Rev Nephrol 7:650–658
Weaver DJ Jr, Somers MJG, Martz K, Mitsnefes MM (2017) Clinical outcomes and survival in pediatric patients initiating chronic dialysis: a report of the North American Pediatric Renal Transplant Cooperative Study registry. Pediatr Nephrol 32:2319–2330
Quinlan C, Bates M, Sheils A, Dolan N, Riordan M, Awan A (2012) Chronic hemodialysis in children weighing less than 10 kg. Pediatr Nephrol 28:803–809
Warady BA, Hebert D, Sullivan EK, Alexander SR, Tejani A (1997) Renal transplantation, chronic dialysis, and chronic renal insufficiency in children and adolescents. The 1995 Annual Report of the North American Pediatric Renal Transplant Cooperative Study. Pediatr Nephrol 11:49–64
Fischbach M, Edefonti A, Schroder C, Watson A, European Pediatric Dialysis Working Group (2005) Hemodialysis in children: general practical guidelines. Pediatr Nephrol 20:1054–1066
Minnick ML, Boynton S, Ndirangu J, Furth S (2010) Sex, race, and socioeconomic disparities in kidney disease in children. Semin Nephrol 30:26–32
Furth SL, Powe NR, Hwang W, Neu AM, Fivush BA (1997) Racial differences in choice of dialysis modality for children with end-stage renal disease. Pediatrics 99:E6
Leonard MB, Donaldson LA, Ho M, Geary DF (2003) A prospective cohort study of incident maintenance dialysis in children: an North American Pediatric Renal Transplant Cooperative Study. Kidney Int 63:744–755
Paglialonga F, Consolo S, Pecoraro C, Vidal E, Gianoglio B, Puteo F, Picca S, Saravo MT, Edefonti A, Verrina E (2016) Chronic haemodialysis in small children: a retrospective study of the Italian Pediatric Dialysis Registry. Pediatr Nephrol 31:833–841
Wightman A, Zimmerman C, Neul S, Lepere K, Cedars K, Opel D (2019) Caregiver experience in pediatric dialysis. Pediatrics 143:e20182102
Kaiser BA, Polinsky MS, Stover J, Morgenstern BZ, Baluarte HJ (1994) Growth of children following the initiation of dialysis: a comparison of three dialysis modalities. Pediatr Nephrol 8:733–738
Turenne MN, Port FK, Strawderman RL, Ettenger RB, Alexander SR, Lewy JE, Jones CA, Agodoa LY, Held PJ (1997) Growth rates in pediatric dialysis patients and renal transplant recipients. Am J Kidney Dis 30:193–203
Achinger SG, Ayus JC (2019) When the source of inflammation is hiding in plain sight: failed kidney transplants, clotted arteriovenous grafts, and central venous catheters. Semin Dial 32:15–21
Ağbaş A, Canpolat N, Çalışkan S, Yılmaz A, Ekmekçi H, Mayes M, Aitkenhead H, Schaefer F, Sever L, Shroff R (2018) Hemodiafiltration is associated with reduced inflammation, oxidative stress and improved endothelial risk profile compared to high-flux hemodialysis in children. PLoS One 13:e0198320
Mitsnefes MM, Laskin BL, Dahhou M, Zhang X, Foster BJ (2013) Mortality risk among children initially treated with dialysis for end-stage kidney disease, 1990–2010. JAMA 309:1921–1929
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yu, E.D., Galbiati, S., Munshi, R. et al. Practice patterns and outcomes of maintenance dialysis in children < 2 years of age: a report of the North American Pediatric Renal Trials and Collaborative Studies (NAPRTCS). Pediatr Nephrol 37, 1117–1124 (2022). https://doi.org/10.1007/s00467-021-05287-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-021-05287-2