Abstract
Background
Introduction of the full-thickness resection device (FTRD) has allowed endoscopic resection of difficult lesions such as those with deep wall origin/infiltration or those located in difficult anatomic locations. The aim of this study is to assess the outcomes of the FTRD among its early users in the USA.
Methods
Patients who underwent endoscopic full-thickness resection (EFTR) for lower gastrointestinal tract lesions using the FTRD at 26 US tertiary care centers between 10/2017 and 12/2018 were included. Primary outcome was R0 resection rate. Secondary outcomes included rate of technical success (en bloc resection), achievement of histologic full-thickness resection (FTR), and adverse events (AE).
Results
A total of 95 patients (mean age 65.5 ± 12.6 year, 38.9% F) were included. The most common indication, for use of FTRD, was resection of difficult adenomas (non-lifting, recurrent, residual, or involving appendiceal orifice/diverticular opening) (66.3%), followed by adenocarcinomas (22.1%), and subepithelial tumors (SET) (11.6%). Lesions were located in the proximal colon (61.1%), distal colon (18.9%), or rectum (20%). Mean lesion diameter was 15.5 ± 6.4 mm and 61.1% had a prior resection attempt. The mean total procedure time was 59.7 ± 31.8 min. R0 resection was achieved in 82.7% while technical success was achieved in 84.2%. Histologically FTR was demonstrated in 88.1% of patients. There were five clinical AE (5.3%) with 2 (2.1%) requiring surgical intervention.
Conclusions
Results from this first US multicenter study suggest that EFTR with the FTRD is a technically feasible, safe, and effective technique for resecting difficult colonic lesions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD) are highly effective methods for resecting large colon polyps or low-risk colorectal neoplasms [1,2,3,4]. A major challenge for these techniques is the “non-lifting” lesion, in which the mucosal layer cannot be separated from the muscularis propria. This can occur when submucosal scarring develops after previous biopsy, attempted resection, or when carcinoma is present. When separation does not occur, performing ESD and EMR is associated with a higher risk of perforation [5, 6]. These techniques are also challenging when lesions are located in difficult anatomic locations, such as within a diverticulum or the appendiceal orifice. As a result, it is not infrequent for polyps that are non-lifting or in difficult anatomic locations to be referred for surgical resection [7,8,9].
More recently, a minimally invasive approach for resecting colorectal lesions has been introduced with the potential of filling the gap between endoscopic and surgical resection techniques. This method may have particular indications for “difficult-to-resect” lesions and in patients who are not considered surgical candidates [10, 11]. It involves performing endoscopic full-thickness resection (EFTR) using a modified over-the-scope-clip (OTSC), FTRD® (Full Thickness Resection Device; Ovesco Endoscopy, Tübingen, Germany).
In addition to the multiple case series and retrospective studies showing positive initial results [12,13,14,15,16,17,18,19,20,21], the first multicenter prospective study was recently published by Schmidt et al. [15] and showed that the FTRD device had 89.5% technical efficacy and R0 resection rate of 76.9% [15]. Although initial studies have been promising, the outcome results of FTRD in the literature remain scarce. In the current study, we report the first multicenter study on FTRD for resecting colorectal lesions in the USA
Materials and methods
This was a multicenter retrospective study on the use of the FTRD for colorectal lesions. Data were collected from 26 tertiary care centers in the USA from 10/2017 to 1/2019. The study was approved by the Institutional Review Board at all participating centers. Inclusion criteria were the use of FTRD to perform FTR of colorectal lesions that were deemed impossible or difficult to resect with conventional endoscopic methods. The indications were grouped into three main categories: difficult adenomas, adenocarcinomas, and subepithelial tumors (SET).
Difficult adenomas consisted of colorectal adenomatous lesions deemed difficult or impossible to resect using other endoscopic techniques. They constituted non-lifting polyps, recurrent or residual lesions, or lesions in difficult anatomic locations, such as within a diverticulum or involving the appendiceal orifice. The adenocarcinoma category included lesions that were previously biopsied and proven to be carcinomas, or lesions suspected to be carcinoma in situ. SET included suspected as well as confirmed tumors. The included cases in this study have not been used in previous abstracts or published studies.
EFTR technique
The FTRD consists of a transparent elongated cap with a modified OTSC and an integrated snare. The device is designed to perform single-step, non-exposure resection such that the tissue is cut above the clipping level avoiding the exposure of peritoneal cavity to colonic contents.
The type of sedation used varied between the centers. Carbon dioxide was used for insufflation of the colonic lumen. The use of antibiotic prophylaxis varied between the centers. All endoscopists have had previous experience with OTSC clip system and received dedicated training on the proper use of the FTRD device.
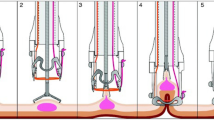
The first step of the EFTR procedure is marking around the target lesion using either FTRD marking probe, included with the kit, argon plasma coagulation probe, or a snare tip. The endoscope is then removed and the FTRD is mounted on the endoscope. The FTRD-fitted colonoscope is then advanced to the target lesion. The lesion is slowly and progressively pulled into the cap using an included tissue grasper inserted through the working channel. Traction is applied until all the marks of the lesion margin are visible within the transparent cap. The OTSC is then deployed by turning the deployment handle. Immediately after clip deployment, the snare is slowly closed on the captured tissue, and snare cautery is applied using Endocut™ setting settings (effect 3, ERBE) while continuing to close the snare. The resected specimen is removed along with the colonoscope. The FTRD is then detached, and the colonoscope is reinserted to the site of resection to carefully examine for signs of perforation, bleeding, or incomplete resection (Fig. 1) (Video 1). Hospital admission post-procedure was at the discretion of the endoscopist.
Endoscopic full-thickness resection of 8 mm non-lifting, residual polyp at the ileocecal valve in 70-year-old female. A Initially, a pediatric colonoscope was advanced to the ileocecal valve for examining the lesion. B, C Using the FTRD marking probe and soft coagulation current, the margins of the polyp were marked. D, E The pediatric colonoscope was removed and replaced with an adult colonoscope fitted with the FTRD system and advanced back to the location of the lesion. Using the FTRD grasper, the polyp was carefully grasped and pulled into the transparent cap using rotation and minimal suctioning in alternation until the markings around the polyp could be clearly seen within the cap. The OTSC was then deployed followed by carefully tightening the snare and resecting the lesion using EndoCut Q current (F). Endoscopic view of the resection site and the OTSC following EFTR
Histologic evaluation
Resected specimens were pinned out and fixed in formalin. Pathology reports used the Vienna staging system for epithelial neoplasms of the GI tract [22]. Details about post-resection specimen size, margins, and specific criteria for malignant polyps were provided.
Follow-up
Clinical follow-ups, which included either clinic visits or phone-call meetings, were completed in the majority of the patients with their main aim being assessment for post-procedure adverse events. Post-procedure colonoscopy follow-ups were not available for all patients.
Study endpoints
The primary outcome of the study was the rate of achieving R0 margin resection, defined as negative lateral and deep margins on post-resection histopathologic evaluation. Secondary outcomes included the following: (1) rate of technical success, defined as the ability to reach the target lesion with FTRD mounted scope, grasp and pull the lesion into the cap, successfully deploy the clip and complete en bloc resection of the lesion; (2) histologic confirmation of full-thickness resection (FTR), defined as resection including muscularis propria, with or without serosa; (3) rate of adverse events; (4) procedure time, (5) need for surgical intervention; and (6) post-procedure follow-up clinical and colonoscopy findings. Adverse events were defined as either technical adverse events such as failure to deploy OTSC, snare malfunction, and iatrogenic stricture as well as clinical adverse events such as bleeding, colorectal wall trauma, appendicitis, perforation/leak, and post-polypectomy syndrome.
Statistical analysis
Descriptive statistics were expressed as mean, median, mode, SD, and range. χ2 test or Pearson correlation and Fisher’s (T-test) exact test was used to compare qualitative data. Independent T-test was used to test the correlation of continuous variables with the outcomes. SPSS (SPSS Statistics for Windows, version 17.0 SPSS Inc., Chicago IL) software was used.
Results
A total of 95 patients (mean age was 65.5 ± 12.6 year, 38.9% Females) underwent EFTR of colorectal lesions using the FTRD® during the study period (Table 1).
Characteristics of lesions referred for FTRD
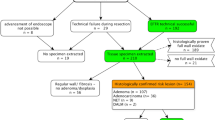
Patients were referred for the resection of three main categories of lesions (Fig. 2). The most common indication was the difficult-to-resect adenomas (n = 63, 66.3%), followed by adenocarcinomas (n = 21, 22.1%) and subepithelial tumors (n = 11, 11.6%). Eleven lesions involved the appendiceal orifice and 1 involved a diverticulum. More than half (61.1%) of the lesions were in the proximal colon, with the remaining 18.9% and 20% in the distal colon and rectum, respectively. The largest measured diameter of the lesions as estimated by the endoscopist prior resection ranged from 3 to 28 mm with a mean of 15.5 ± 6.4 mm. The majority (81%) had either prior intervention: biopsy (n = 18) and prior resection attempts (n = 58), with the most common method (39.4%) being EMR. The most common result of the prior intervention attempts was macroscopic residual lesion post-resection.
EFTR procedures and clinical outcomes
EFTR was performed predominantly under propofol sedation (69.9%), and the majority of the patients (66.3%) did not receive prophylactic antibiotics. The mean total procedure time was 59.7 ± 31.8 min. Maximum resected diameter and depth had a median of 20 ± 7 mm and 5 ± 4.7 mm, respectively (Table 2). Technical success was achieved in 80 patients (84.2%), with the most common reason for technical failure being incomplete lesion resection (n = 9, 9.5%), followed by the inability to deploy the clip due to lesion characteristics (n = 4, 4.2%), or failure to advance the FTRD mounted endoscope to the lesion (n = 2, 2.1%). Technical failure attributed to mechanical failure of the device was reported in 4 patients (4.2%): 3 related to the integrated snare resulting in partial lesion resection and 1 clip related resulting in perforation (Supplementary Table 1). Ten out of the 15 patients who had failed EFTR attempt underwent either same session hot snare resection (n = 6), forceps avulsion (n = 1), or surgical resection (n = 3). The majority of patients (76.3%) were discharged following the procedure. The mean hospital stay was 0.4 ± 1.3 days.
Histologic outcomes
Adenoma was the most common lesion histology (n = 51, 53.7%). Of these, histology was 24 tubular, 15 tubulovillous, 6 sessile serrated, 5 adenoma with high-grade dysplasia, and 1 villous. Adenocarcinoma was present in 18 (19%), followed by 6 subepithelial tumors (6.3%), which included 4 neuroendocrine/carcinoid, 1 gastrointestinal stromal tumor (GIST), and 1 lipoma. The remaining 20 (12%) were found to be non-neoplastic lesions including scar tissue/fibrosis (10), normal tissue (3), hyperplastic polyp (1), and 6 lesions were categorized as “unspecified non-neoplastic” lesions (Fig. 3). R0 histologic margin was achieved in 62 out of the 75 confirmed neoplastic lesions, giving an overall rate of 82.7%. The variable, histologic confirmation of FTR, defined as having the three colonic wall layers with or without the serosa, was available for 42 out of the total 80 cases that underwent successful EFTR, and it was achieved in 37 out of these 42 (88.1%).
Subgroup analysis
Based on pathology, lesions were grouped into three main categories: difficult adenomas, adenocarcinoma, and subepithelial tumors (Fig. 4).
Difficult adenomas
Initially, 63 patients were referred for EFTR for the resection of difficult adenomas. Post-resection pathology revealed that 51 patients had adenomatous lesions. The remainder of lesions were reclassified as either adenocarcinoma (n = 2) or benign non-neoplastic tissue (n = 10). Technical success was achieved in 41/51 (80.4%), resected lesions with R0 histology margin in 40/51 (78.4%), and histologically confirmed FTR in 15/20 (75%).
Adenocarcinomas
Eighteen out of the initial 21 referred lesions were confirmed to be adenocarcinomas, two of which were initially thought to be “difficult adenoma.” The remaining were reclassified as benign lesions. Adenocarcinomas had a mean size of 15.5 mm, ranging from 7 to 25 mm. Technical success was achieved in 18/18 (100%) cases, R0 resection was achieved in 16/18 (88.9%) cases, and histologically confirmed FTR was achieved in 12/14 (85.7%) cases. Two patients who did not achieve R0 margin were referred for surgical resection after EFTR, and post-surgical histopathologic evaluation revealed that one had evidence of deep submucosal infiltration (> 1000 µm) with lymphovascular invasion and was subsequently referred for oncologic management. The second patient had a curative surgical resection as there was no evidence of lymphovascular invasion. Post-EFTR histopathologic evaluation of the 16 adenocarcinomas that were resected with R0 margins revealed no high-risk histologic features and no or only superficial (< 1000 µm) submucosal invasion thus resulting in curative rate 88.9%.
Subepithelial tumors
There were six confirmed subepithelial tumors. Four were neuroendocrine/carcinoid tumors, and two mesenchymal neoplasms with 1 GIST and 1 lipoma. Technical success and R0 resection were achieved in 6/6 (100%) cases.
Multivariable analysis
Technical success was more likely to be achieved with adenocarcinomas and subepithelial tumors as compared to difficult adenomas (p = 0.02). Lesions with no prior interventions, biopsy, or resection attempt were more likely to have technically successful EFTR and histologic FTR (p value 0.003 and 0.04, respectively). When post-resection lesion sizes were categorized into two groups: ≤ 20 mm (n = 54) and > 20 mm (n = 41), there was a non-significant correlation of technical failure and non-R0 histology margin with lesions measured size > 20 mm.
Technical and clinical adverse events
There were five (5.3%) reported clinical adverse events. Based on the lexicon for severity classification for endoscopic procedures [23], three out of the five adverse events were classified as mild (3.6%). One of these cases included immediate mild bleeding in a patient being bridged with heparin, which was managed conservatively with thermal coagulation. The other two mild adverse events included one immediate and one delayed iatrogenic luminal stricture. There were two severe adverse events (2.1%), both requiring surgical intervention: one intra-procedural perforation and one case of appendicitis 10 days post-procedure. No significant association was found between any of the collected variables and adverse event occurrence. The two reported cases of immediate and delayed luminal stricture post-EFTR were followed clinically with no symptoms reported by either patient. This lack of symptoms was in the setting of a 60% reduction in luminal diameter for the case with an immediate procedural stricture.
The one case of immediate perforation occurred during attempted resection of a recurrent adenoma involving the appendiceal orifice that was previously resected with a hot snare. There was a technical failure of clip deployment, resulting in a perforation. The patient was managed surgically and fully recovered. Despite 3 days of post-procedural oral antibiotics, one patient developed appendicitis 10 days post-resection of a cecal adenomatous lesion that involved the appendiceal orifice.
There were four (4.2%) instances of mechanical/operator failure of the device. In three cases, there was a problem with the integrated snare resulting in an incomplete resection with a macroscopic residual lesion. Additional hot snare resection was performed in two of these cases, and forceps avulsion in the third. There was one failure related to the clip deployment which resulted in a perforation (Supplementary Table 2).
Follow-up
During a mean follow-up of 61.7 ± 82.3 days, no other late-onset adverse events were reported. One patient was deceased at the time of scheduled clinical follow-up due to medical comorbidity. Only 20/95 patients underwent follow-up colonoscopies at a mean of 114.4 ± 59 days post-procedure. The spontaneous detachment of the clip was noted in 14 out of the 20 patients. Two patients had macroscopic and histologic evidence of residual/recurrent adenomatous tissue. Both cases were difficult adenomas and had technically successful EFTR with R0 resection. Hot snare resection was performed successfully for the two cases and both patients were scheduled for further follow-up colonoscopy.
Discussion
In general, EFTR technique has evolved from exposed EFTR approach, which involves tumor resection initially followed by the closure of the wall defect, to non-exposed EFTR approach, where a serosa-to-serosa apposition is created prior to resection,. Two devices are currently available to perform non-exposed EFTR, endoscopic plication with suturing devices GERDx™ (G-SURG GmbH, Seeon-Seebruck, Germany), and FTRD (Full Thickness Resection Device; Ovesco Endoscopy, Tübingen, Germany). Literature comparing the different approaches of EFTR is scarce and limited to a few retrospective studies, revealing no superiority of any approach [24].
The purpose of this study was to provide a review of the indications, outcomes, efficacy, and safety of the FTRD among early users in the USA. In our cohort, the most common indication for EFTR with FTRD was resection of recurrent or residual adenomas, with the most common method of previous resection attempt being EMR. Several studies [25,26,27] have demonstrated a significant rate of incomplete resection and a higher risk of perforation with EMR and ESD for recurrent or residual lesions, which is essentially attributed to the presence of submucosal fibrosis. FTRD is less technically challenging than ESD and as such has the potential for more widespread use among endoscopists. Prior to the introduction of FTRD, adenomas located within or involving the appendiceal orifice or a diverticulum were resected surgically, as using endoscopic methods is either not possible or associated with a higher risk of perforation [28]. As previously shown in a prospective observational study [29], FTRD can safely resect such lesions, eliminating the need for surgery. However, the use of FTRD for lesions involving the appendiceal orifice can lead to acute appendicitis. Previous studies reported that EFTR resection for adenomas involving the appendix was associated with a 10% risk of appendicitis [14, 15]. In our cohort, one out of the 11 (9%) cecal lesions with appendiceal orifice involvement developed appendicitis and had a subsequent surgical intervention.
Adenocarcinoma resection was the second most common indication for EFTR in our cohort. It is important to acknowledge that for high-risk carcinoma, surgical resection remains the preferred treatment option as it allows curative and complete resection of colonic segments in addition to lymph nodes that may harbor cancer [4, 30]. However, FTRD can be a less invasive method of resecting low-risk adenocarcinomas [13], particularly among patients who are considered high-risk for surgery. In the only prospective study to date, the R0 resection was achieved in 21/29 (72.4%) cases; however, the curative resection, meaning R0 histologic margin as well as having low-risk histology and with no or only superficial submucosal invasion (< 1000 µm) on post-EFTR histology, was achieved only in 13/29 (45%) cases [15]. Although in our cohort, the rate of R0 resection for adenocarcinoma was 89%, the sample size is limited. A recent multicenter study that was aimed to further clarify the role of EFTR in malignant colorectal lesions [31] reported R0 resection for 112 of 156 histologically confirmed adenocarcinoma (71.8%) with a curative rate of 66%. Another important finding was that 99.3% of the resected lesions were properly evaluated for high-risk features, thus identifying the lesions that require further surgical intervention and highlighting an important feature of EFTR [32]. Further prospective trials comparing EFTR vs. surgical resection of low-risk adenocarcinoma are required.
The effectiveness of achieving a full-thickness resection using FTRD depends on the ability to incorporate the entire lesion within the device cap, which requires a firm grasp of the tissue to sufficiently pull/retract the lesion. We postulate that two factors can contribute to technical failure: the size of the lesion and the mobility of the wall. Wall mobility impacts the degree of traction generated by pulling on the tissue, which in turn might be influenced by the location in the GI tract and to a higher degree, the presence of fibrosis. In this regard, the majority of the resected lesions (81%) in our cohort had a prior intervention. Nonetheless, the overall technical success and R0 resection rates were 84.2% and 82.7%, respectively. Although difficult adenoma resection was possibly correlated with a lower rate of technical success and R0 resection, the outcomes were promising. The total rate of clinical adverse events in our sample was 5.3% with only 2 (2.1%) severe requiring surgical intervention. This is similar to data reported by Schmidt et al. [15] with a 9.9% rate of adverse events with 2.2% requiring surgical intervention. We reported 4 cases of mechanical failure resulting in incomplete resection and perforation. Assessing the causality whether they occurred due to device or operator failure was not feasible. However, it is important to note that clip misdeployment can result in perforation. Therefore, it is highly recommended for the endoscopist to undergo proper preclinical training for the proper and safe use of FTRD. This is the first US multicenter study examining the efficacy and safety of endoscopic full-thickness resection using the FTRD. The study was a retrospective chart review with a limited sample size; therefore, there were limitations in terms of assessing correlations and presenting long-term outcomes in terms of both safety and efficacy. Another limitation is that we were unable to provide complete data on the recurrence rate due to the limited follow-up colonoscopy results. This was greatly due to the preference of endoscopists not to commit patients who had technically successful resection with R0 histologic margin EFTRs of benign lesions for a follow-up colonoscopy. Determining the rate of recurrence, in particular for SETs and adenocarcinomas, is of great clinical relevance and should be addressed in future prospective trials.
Histologic confirmation of full-thickness resection was not reported in more than half of the cases. This may be due to the unfamiliarity of the pathologists on the nature of the resection, thus commenting on whether full-thickness resection is achieved or not was not part of the standard histopathology report of the endoscopically resected specimens.
Outcomes from this first US multicenter FTRD experience for resecting lesions in the lower GI tract demonstrated satisfactory technical success, safety, and R0 resection rates. It is well-suited to the management of “difficult adenomas” and appropriately sized subepithelial tumors. It is still premature to recommend endoscopic full-thickness resection using FTRD for low-risk adenocarcinomas, which requires further studies. However, FTRD can be considered a reasonable option for patients who are not surgical candidates.
Abbreviations
- AE:
-
Adverse event
- APC:
-
Argon plasma coagulation
- EFTR:
-
Endoscopic full-thickness resection
- EMR:
-
Endoscopic mucosal resection
- ESD:
-
Endoscopic submucosal dissection
- FTRD:
-
Full-thickness resection device
- HF:
-
High-frequency
- OTSC:
-
Over-the-scope clip
References
Raju GS, Lum PJ, Ross WA, Thirumurthi S, Miller E, Lynch PM, Lee JH, Bhutani MS, Shafi MA, Weston BR, Pande M, Bresalier RS, Rashid A, Mishra L, Davila ML, Stroehlein JR (2016) Outcome of EMR as an alternative to surgery in patients with complex colon polyps. Gastrointest Endosc 84:315–325
Moss A, Williams SJ, Hourigan LF, Brown G, Tam W, Singh R, Zanati S, Burgess NG, Sonson R, Byth K, Bourke MJ (2015) Long-term adenoma recurrence following wide-field endoscopic mucosal resection (WF-EMR) for advanced colonic mucosal neoplasia is infrequent: results and risk factors in 1000 cases from the Australian Colonic EMR (ACE) study. Gut 64:57–65
Oka S, Tanaka S, Saito Y, Iishi H, Kudo SE, Ikematsu H, Igarashi M, Saitoh Y, Inoue Y, Kobayashi K, Hisabe T, Tsuruta O, Sano Y, Yamano H, Shimizu S, Yahagi N, Watanabe T, Nakamura H, Fujii T, Ishikawa H, Sugihara K (2015) Local recurrence after endoscopic resection for large colorectal neoplasia: a multicenter prospective study in Japan. Am J Gastroenterol 110:697–707
Burgess NG, Bourke MJ (2016) Endoscopic resection of colorectal lesions: the narrowing divide between East and West. Dig Endosc 28:296–305
Agapov M, Dvoinikova E (2014) Factors predicting clinical outcomes of endoscopic submucosal dissection in the rectum and sigmoid colon during the learning curve. Endosc Int Open 2:E235–240
He YQ, Wang X, Li AQ, Yang L, Zhang J, Kang Q, Tang S, Jin P, Sheng JQ (2015) Factors for Endoscopic submucosal dissection in early colorectal neoplasms: a single center clinical experience in China. Clin Endosc 48:405–410
Lee SP, Kim JH, Sung IK, Lee SY, Park HS, Shim CS, Han HS (2015) Effect of submucosal fibrosis on endoscopic submucosal dissection of colorectal tumors: pathologic review of 173 cases. J Gastroenterol Hepatol 30:872–878
Mizushima T, Kato M, Iwanaga I, Sato F, Kubo K, Ehira N, Uebayashi M, Ono S, Nakagawa M, Mabe K, Shimizu Y, Sakamoto N (2015) Technical difficulty according to location, and risk factors for perforation, in endoscopic submucosal dissection of colorectal tumors. Surg Endosc 29:133–139
Hong SN, Byeon JS, Lee BI, Yang DH, Kim J, Cho KB, Cho JW, Jang HJ, Jeon SW, Jung SA, Chang DK (2016) Prediction model and risk score for perforation in patients undergoing colorectal endoscopic submucosal dissection. Gastrointest Endosc 84:98–108
Fahndrich M, Sandmann M (2015) Endoscopic full-thickness resection for gastrointestinal lesions using the over-the-scope clip system: a case series. Endoscopy 47:76–79
Monkemuller K, Peter S, Toshniwal J, Popa D, Zabielski M, Stahl RD, Ramesh J, Wilcox CM (2014) Multipurpose use of the 'bear claw' (over-the-scope-clip system) to treat endoluminal gastrointestinal disorders. Dig Endosc 26:350–357
Schmidt A, Bauerfeind P, Gubler C, Damm M, Bauder M, Caca K (2015) Endoscopic full-thickness resection in the colorectum with a novel over-the-scope device: first experience. Endoscopy 47:719–725
Andrisani G, Pizzicannella M, Martino M, Rea R, Pandolfi M, Taffon C, Caricato M, Coppola R, Crescenzi A, Costamagna G, Di Matteo FM (2017) Endoscopic full-thickness resection of superficial colorectal neoplasms using a new over-the-scope clip system: a single-centre study. Dig Liver Dis 49:1009–1013
Valli PV, Mertens J, Bauerfeind P (2018) Safe and successful resection of difficult GI lesions using a novel single-step full-thickness resection device (FTRD((R))). Surg Endosc 32:289–299
Schmidt A, Beyna T, Schumacher B, Meining A, Richter-Schrag HJ, Messmann H, Neuhaus H, Albers D, Birk M, Thimme R, Probst A, Faehndrich M, Frieling T, Goetz M, Riecken B, Caca K (2018) Colonoscopic full-thickness resection using an over-the-scope device: a prospective multicentre study in various indications. Gut 67:1280–1289
Schmidt A, Damm M, Caca K (2014) Endoscopic full-thickness resection using a novel over-the-scope device. Gastroenterology 147:740–742.e742
Richter-Schrag HJ, Walker C, Thimme R, Fischer A (2016) Full thickness resection device (FTRD). Experience and outcome for benign neoplasms of the rectum and colon. Chirurg 87:316–325
Sarker S, Gutierrez JP, Council L, Brazelton JD, Kyanam Kabir Baig KR, Monkemuller K (2014) Over-the-scope clip-assisted method for resection of full-thickness submucosal lesions of the gastrointestinal tract. Endoscopy 46:758–761
Lagoussis P, Soriani P, Tontini GE, Neumann H, Pastorelli L, de Nucci G, Vecchi M (2016) Over-the-scope clip-assisted endoscopic full-thickness resection after incomplete resection of rectal adenocarcinoma. Endoscopy 48(Suppl 1):E59–E60
Soriani P, Tontini GE, Neumann H, de Nucci G, De Toma D, Bruni B, Vavassori S, Pastorelli L, Vecchi M, Lagoussis P (2017) Endoscopic full-thickness resection for T1 early rectal cancer: a case series and video report. Endosc Int Open 5:E1081–1086
Aepli P, Criblez D, Baumeler S, Borovicka J, Frei R (2018) Endoscopic full thickness resection (EFTR) of colorectal neoplasms with the Full Thickness Resection Device (FTRD): Clinical experience from two tertiary referral centers in Switzerland. United Eur Gastroenterol J 6:463–470
Quirke P, Risio M, Lambert R, von Karsa L, Vieth M (2011) Quality assurance in pathology in colorectal cancer screening and diagnosis-European recommendations. Virchows Arch 458:1–19
Cotton PB, Eisen GM, Aabakken L, Baron TH, Hutter MM, Jacobson BC, Mergener K, Nemcek A Jr, Petersen BT, Petrini JL, Pike IM, Rabeneck L, Romagnuolo J, Vargo JJ (2010) A lexicon for endoscopic adverse events: report of an ASGE workshop. Gastrointest Endosc 71:446–454
Aslanian HR, Sethi A, Bhutani MS, Goodman AJ, Krishnan K, Lichtenstein DR, Melson J, Navaneethan U, Pannala R, Parsi MA, Schulman AR, Sullivan SA, Thosani N, Trikudanathan G, Trindade AJ, Watson RR, Maple JT (2019) ASGE guideline for endoscopic full-thickness resection and submucosal tunnel endoscopic resection. VideoGIE 4:343–350
Isomoto H, Nishiyama H, Yamaguchi N, Fukuda E, Ishii H, Ikeda K, Ohnita K, Nakao K, Kohno S, Shikuwa S (2009) Clinicopathological factors associated with clinical outcomes of endoscopic submucosal dissection for colorectal epithelial neoplasms. Endoscopy 41:679–683
Hori K, Uraoka T, Harada K, Higashi R, Kawahara Y, Okada H, Ramberan H, Yahagi N, Yamamoto K (2014) Predictive factors for technically difficult endoscopic submucosal dissection in the colorectum. Endoscopy 46:862–870
Saito Y, Uraoka T, Yamaguchi Y, Hotta K, Sakamoto N, Ikematsu H, Fukuzawa M, Kobayashi N, Nasu J, Michida T, Yoshida S, Ikehara H, Otake Y, Nakajima T, Matsuda T, Saito D (2010) A prospective, multicenter study of 1111 colorectal endoscopic submucosal dissections (with video). Gastrointest Endosc 72:1217–1225
Bucalau AM, Lemmers A, Arvanitakis M, Blero D, Neuhaus H (2018) Endoscopic Full-thickness resection of a colonic lateral spreading tumor. Dig Dis 36:252–256
Bronzwaer ME, Bastiaansen BA, Koens L, Dekker E, Fockens P (2018) Endoscopic full-thickness resection of polyps involving the appendiceal orifice: a prospective observational case study. Endosc Int Open 6:E1112–1119
Ahlenstiel G, Hourigan LF, Brown G, Zanati S, Williams SJ, Singh R, Moss A, Sonson R, Bourke MJ (2014) Actual endoscopic versus predicted surgical mortality for treatment of advanced mucosal neoplasia of the colon. Gastrointest Endosc 80:668–676
Kuellmer A, Mueller J, Caca K, Aepli P, Albers D, Schumacher B, Glitsch A, Schafer C, Wallstabe I, Hofmann C, Erhardt A, Meier B, Bettinger D, Thimme R, Schmidt A (2019) Endoscopic full-thickness resection for early colorectal cancer. Gastrointest Endosc 89:1180–1189.e1181
Schreiner P, Valli P, Marques Maggio E, Bauerfeind P (2018) Simultaneous endoscopic full-thickness resection of two synchronous colonic granular cell tumours. BMJ Case Rep. https://doi.org/10.1136/bcr-2017-222223
Author information
Authors and Affiliations
Contributions
MK, YI, and KV were involved in the study planning, interpretation of the data, and development of the manuscript. DD, GI, JT, and TA were involved in critically revising the manuscript for important intellectual content. All remaining authors involved in performing the procedure and data extraction. All authors approved the final submitted draft of this manuscript.
Corresponding author
Ethics declarations
Disclosures
Dr. Khashab is consultant for Boston Scientific, Medtronic, and Olympus. Dr. Ian Grimm is a consultant for Boston Scientific. Dr. Irani is a consultant for Boston Scientific. Dr. Kumbhari is a consultant for ReShape Life Sciences, Apollo Endosurgery, Medtronic, and Boston Scientific. Dr. Nikhil is a consultant for Apollo Endosurgery, Boston scientific, and Olympus. Dr. Amateau is a consultant for Merit Endoscopy, Boston Scientific, US Endoscopy, and Neurotronic and the recipient of research support from Cook Medical. Dr. Smallfield has research funding from CSA medical and C2 therapeutics. Dr. Aadam AA is a consultant for Boston Scientific. Dr. Diehl Md Consultant for Boston Scientific, Olympus, Pentax, Cook Medical, Merit, ConMed, US Endoscopy, Medtronic, Lumendi. Dr. Chang consultant for Apollo, Boston Scientific, Cook, Covidien, Erbe, Endogastric Solutions, Mauna Kea Mederi, Medtronic, Olympus, Ovesco, Pentax, Torax. Dr. Samarasena has educational grant from Cook and consultant for Mauna Kea, Medtronic, Olympus, Pentax, US Endoscopy. Dr. Al-Haddad received research and teaching support from Boston Scientific. Dr. Pohl received grants from Boston Scientific, US Endoscopy, and Aries/Cosmo Pharamceuticals. Dr. Templeton is a consultant for Boston Scientific and Medtronic. Dr. Ginsberg is a consultant for Olympus Inc. and Boston Scientific. Dr. Fukami is consultant for Boston Scientific and Olympus. Dr. Sharaiha is consultant for Boston Scientific, Olympus, Apollo, and Medtronic. Dr. Ichkhanian, Dr. Vosoughi, Dr. James, Dr. Hajifathalian, Dr. Tokar, Dr. Lee, Dr. Mizrahi, Dr. Barawi, Dr. Friedland, Dr. Korc, Dr. Kowalski, Dr. Novikov, Dr. Oza, Dr. Panuu, Dr. Lajin, Dr. Kumta, Dr. Tang, Dr. Naga, and Dr. Brewer have no conflicts of interest or financial ties to disclose
Ethical approval
IRB approval for this study across the multiple centers performed in accordance with the principles of the Declaration of Helsinki. The formal informed consent was waived.
Informed consent
The formal informed consent was waived.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary file1 (MP4 91407 kb)
Rights and permissions
About this article
Cite this article
Ichkhanian, Y., Vosoughi, K., Diehl, D.L. et al. A large multicenter cohort on the use of full-thickness resection device for difficult colonic lesions. Surg Endosc 35, 1296–1306 (2021). https://doi.org/10.1007/s00464-020-07504-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-020-07504-9