Abstract
Background
Difficulties in endoscopic operations and therapeutic procedures seem to occur due to the complexity of operating the endoscope dial as well as difficulty in performing synchronized movements with both hands. We developed a prototype robotic-assisted flexible endoscope that can be controlled with a single hand in order to simplify the operation of the endoscope. The aim of this study was to confirm the operability of the robotic-assisted flexible endoscope (RAFE) by performing endoscopic submucosal dissection (ESD).
Methods
Study 1: ESD was performed manually or with RAFE by an expert endoscopist in ex vivo porcine stomachs; six operations manually and six were performed with RAFE. The procedure time per unit circumferential length/area was calculated, and the results were statistically analyzed. Study 2: We evaluated how smoothly a non-endoscopist can move a RAFE compared to a manual endoscope by assessing the designated movement of the endoscope.
Results
Study 1: En bloc resection was achieved by ESD using the RAFE. The procedure time was gradually shortened with increasing experience, and the procedure time of ESD performed with the RAFE was not significantly different from that of ESD performed with a manual endoscope. Study 2: The time for the designated movement of the endoscope was significantly shorter with a RAFE than that with a manual endoscope as for a non-endoscopist.
Conclusions
The RAFE that we developed enabled an expert endoscopist to perform the ESD procedure without any problems and allowed a non-endoscopist to control the endoscope more easily and quickly than a manual endoscope. The RAFE is expected to undergo further development.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The flexible endoscope, which was originally developed for diagnostic purposes, is operated with both hands using two dials for up–down and left–right movement; the scope axis allows for back-and-forth and twisting. The endoscope design and mechanism have remained largely unchanged over the years. Endoscopic submucosal dissection (ESD), which was developed in Japan, represents a highly advanced flexible endoscopic procedure [1]. ESD has now become the gold standard procedure for the en bloc resection of superficial gastrointestinal tumors. Furthermore, the application of flexible endoscopic procedures is spreading to the abdominal cavity, as typified by natural orifice transluminal endoscopic surgery (NOTES) [2, 3]. However, appropriate training is required to master the operation of flexible endoscopes. If flexible endoscopes could be operated more easily, endoscopic procedures in the abdominal cavity would become easier because the flexibility would enable it to reach narrow spaces and to approach from various angles.
One problem associated with the operation of flexible endoscopes is the complexity of operating the scope dials, which prompted us to develop an endoscope with robotic control of the scope dials [4, 5]. However, the operation of the endoscope remains difficult because it is necessary to synchronize movement of both hands when the scope axis is held with the other hand. Although Kume et al. developed a robotic system that enabled the control of both the scope dials and axis with one hand [6], it was too large to use in clinical practice. We therefore developed a prototype robotic-assisted flexible endoscope (RAFE). The RAFE enabled us to operate both the endoscope dials and the axis of the endoscope with a single hand at the bedside.
The aim of this study was to investigate the operability of the RAFE and to confirm whether the RAFE (operated with a single hand) performs as well as a conventional endoscope during ESD.
Materials and methods
The robotic-assisted flexible endoscope
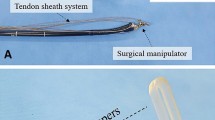
The RAFE system was developed at the Center for Advanced Medical Innovation, Kyushu University, Fukuoka, Japan. This system is designed to be attached to a commercially available endoscope. In the present study, it was attached to a GIF-Q260J (Olympus Medical Systems Corporation, Tokyo, Japan), which is commonly used for ESD. The overall system is shown in Fig. 1. A flexible endoscope has four-axis movement: up–down and right–left (controlled by two angle dials), back–forth (controlled by axial insertion–retraction), and twisting (controlled by axial rotation). The system requires the control of these four axes. The endoscope grip is set into the endoscope holder. Inside the holder are two motorized wheels that fit the angle dials and elastic nails that push two valve buttons for suction, air supply, and water supply. The endoscope rotates on the axis when it is rotated by the endoscope holder itself. The axial insertion–retraction movement is accomplished by a motorized arm. The coupling of the arm to the flexible part of an endoscope is free for axial rotation but fixed for axial insertion–retraction. The master controller is a one-handle type (Fig. 2). The up–down and right–left movement of the handle is linked with that of the endoscope, and the handle rotation and handle insertion–retraction are also linked with those of the endoscope (Fig. 3A–D and Video). The buttons for suction, air supply, and water supply are attached to the base of the handle, and they can be pushed with the forefinger of the hand that is holding the handle.
The overall view of the robotic-assisted flexible endoscope system, which consists of an endoscope holder, a motorized arm, and a master controller. The endoscope axis rotates by the rotation of the endoscope holder itself (A). The angle dials of the endoscope mesh with two motorized wheels inside the holder. Axial insertion–retraction is performed by the anterior–posterior movement of the motorized arm (B)
This figure shows cooperation between the movement of the master controller and the movement of the scope. The up–down (A) and right–left (B) movement of the handle is linked with that of the endoscope. The handle rotation (C) is linked with that of the endoscope by rotating the scope holder directly. The handle insertion–retraction (D) is also linked with that of the endoscope by moving the motorized arm back and forth
Study design
Study 1 was performed by a single experienced endoscopist, defined as an expert endoscopist, who had performed more than 10,000 standard endoscopic examinations and 100 ESDs, and study 2 was performed by a non-medical doctor without any experience in endoscopy, defined as a non-endoscopist.
Study 1: ESD was performed using the RAFE in six cases using ex vivo porcine stomachs obtained from a meat market. As a control, conventional ESD was performed using a manual endoscope in another six cases. We used two porcine stomachs and placed all of the resected sites so that there was no difference between the conditions of the procedures performed using the RAFE and manual endoscope. In both groups, ESD was performed using one electric needle knife, which was inserted into the original forceps channel of the endoscope. In both groups, circumferential marking was performed manually by the conventional method. The ESD procedure consists of two key techniques: circumferential mucosal incision and submucosal dissection. After injecting indigo-dyed saline solution into the submucosal layer using a conventional injection needle, the first (circumferential mucosal incision) and second step (subsequent submucosal dissection) were performed.
Study 2: We evaluated how smoothly a non-endoscopist could control the RAFE compared to a manual endoscope by assessing the designated movement of the endoscope. In general, it is impossible for non-endoscopists to perform ESD. The designated movement of the endoscope was as follows: First, numerical labels from ① to ④ were placed three dimensionally, as shown in Fig. 4. Second, we initially placed the endoscope at label ① on the monitor screen, followed by ②, ③, and ④ in numerical order, and finally returned to place the endoscope at ①. This series of movements was defined as one set, and a non-endoscopist performed eight sets either using the RAFE or a manual endoscope.
Outcome measurement
Study 1: The procedure time for circumferential mucosal incision and submucosal dissection was recorded to evaluate the operability of the RAFE. After the completion of the procedure, the specimens were fixed on a corkboard, and the lengths of the long and short axes of the specimens were measured. The time per unit circumferential length/area was calculated using the formula π (a + b)/2 (circumferential length) and πab/4 (area), respectively, where a is the length of the long axis and b is the length of the short axis, as described by Sakurazawa et al. [7].
Study 2: The procedure time per set was compared between the RAFE and manual endoscope in order to evaluate the operability of the RAFE by a non-endoscopist.
Statistical analyses
The data were expressed as the median (interquartile range) if applicable. The two-sided Mann–Whitney U test was used to compare data. P values of < 0.05 were considered to indicate statistical significance. All of the statistical analyses were performed using the JMP Pro software program (ver. 13.0.0, SAS Institute Inc., Cary, NC, USA).
Results
Study 1: A total of 12 cases of ESD performed with the RAFE or manual endoscope were successfully completed with en bloc resection. There were no cases of muscle layer injury or perforation in either group. The resected specimen size and procedure time for each trial are shown in Table 1, and the procedure time per unit circumferential length/area is shown in Fig. 5. The average sizes of the resected specimen in the operations performed with the manual endoscope and the RAFE were 2.6 × 2.2 cm and 2.8 × 2.2 cm, respectively. The procedure time of ESD with the RAFE was longer than that of ESD with a manual endoscope for both the circumferential mucosal incision (0.382, 0.358–0.423 vs. 0.334, 0.225–0.38 min/cm, respectively) and the submucosal dissection (1.515, 1.19–3.57 vs. 0.96, 0.80–1.31 min/cm2, respectively); however, the difference was not statistically significant (P = 0.20, 0.20, respectively). In a sub-analysis, the procedure time of ESD with the RAFE was found to be significantly longer than that of ESD with a manual endoscope in the first two cases, while no marked difference was observed in the subsequent cases.
The circumferential incision time (A) and submucosal dissection time (B) are shown as the procedure time per unit. The robotic-assisted flexible endoscope took more time than the manual endoscope for both circumferential incision and submucosal dissection; however, the procedure times did not differ to a statistically significant extent
Study 2: The procedure time per set for both groups is shown in Table 2, and a summary of each procedure time is shown in Fig. 6. The time to use the RAFE (19.5, 16.8–21.3) was significantly shorter than that to use the manual endoscope (47, 42–58.8 s) (P = 0.0008).
Discussion
Endoscopic treatment is currently indispensable for gastrointestinal tract operations, and advanced endoscopic procedures such as ESD require considerable skill. Endoscopists must undergo several years of training to be able to perform such advanced techniques, and even experienced endoscopists often feel pressure to complete their procedures. A novel method allowing for the easier and more perfect control of an endoscope has been awaited, and the RAFE may be a good candidate tool, as it enables us to perform single-handed procedures.
In Study 1, the circumferential mucosal incision and submucosal dissection initially took longer with the RAFE than with a manual endoscope. However, after the third case, there was no marked difference in the procedure time between the RAFE and the manual endoscope, despite the fact that the expert endoscopist in this study had never performed ESD using the RAFE before and used the RAFE only a couple of times briefly to ascertain the operating condition before starting the study. This indicated that the RAFE can be manipulated by expert endoscopists without any trouble within a short while and that the RAFE is easy to master, since it is considered the procedure time for ESD performed by Japanese expert endoscopists is faster than that by the endoscopists in the rest of the world. In Study 2, the RAFE enabled the non-endoscopist to control the scope more quickly than a manual endoscope. We consider that the single-hand controller of the RAFE can be operated more easily than a manual endoscope because it is unnecessary to operate the difficult dial controls, which are divided into up–down and right–left movements and because the operator does not need to synchronize the movement of both of their hands. Furthermore, the fact that the endoscope can be controlled with one hand naturally means that the other hand is free, which is one of the useful points of the RAFE. In this study 1, therapeutic devices could be operated simultaneously with the other hand while using the RAFE (Fig. 7). In the clinical setting, the fine tuning of the device position from a forceps channel while moving an endoscope is often required and the single-handed RAFE makes this technique much easier.
While the RAFE is useful in its own right, further advantages can be achieved with the attachment of two articulating arms. Recently, flexible endoscopic robots with two articulating arms (one arm is typically used for a grasping forceps, while the other is used for an electronic knife) have been developed [8,9,10,11]. However, the endoscope itself is often operated by another endoscopist (aside from the operator of the robot), because the robotic operation is only for the two articulating arms and the operation of an endoscope remains manual in many endoscopic robots that are currently under development. If these articulating arms and the RAFE can be combined into a single manipulation interface, it will become possible for one person to seamlessly operate the endoscope and therapeutic devices. This will allow ESD by the RAFE with two articulating arms to be performed more easily. Furthermore, the RAFE has the potential to be applied beyond the abdominal cavity to various intraluminal procedures. We have developed our own manual version of the two articulating arms [12] and plan to use these arms with our RAFE.
A RAFE combined with two articulating arms would bring additional advantages when performing endoscopic procedures. With a RAFE, the endoscope dials and axis do not return to the neutral position, even if the operator disengages their hands from the endoscope controller. In other words, after moving the endoscope to the optimal position, the two articulating arms can then be operated with both hands without having to touch the endoscope controller.
In conclusion, the RAFE enabled an expert endoscopist to perform the ESD procedures without any problems, similar to a manual endoscope, and it also allowed a non-endoscopist to control the scope more easily and quickly than a manual endoscope. We will continue to make improvements as the RAFE undergoes further development.
References
Gotoda T, Kondo H, Ono H, Saito Y, Yamaguchi H, Saito D, Yokota T (1999) A new endoscopic mucosal resection procedure using an insulation-tipped electrosurgical knife for rectal flat lesions: report of two cases. Gastrointest Endosc 50:560–563
Rattner D, Kalloo A (2006) ASGE/SAGES Working Group on Natural Orifice Translumenal Endoscopic Surgery. October 2005. Surg Endosc 20:329–333
Hazey JW, Narula VK, Renton DB, Reavis KM, Paul CM, Hinshaw KE, Muscarella P, Ellison EC, Melvin WS (2008) Natural-orifice transgastric endoscopic peritoneoscopy in humans: initial clinical trial. Surg Endosc 22:16–20
Pullens HJ, van der Stap N, Rozeboom ED, Schwartz MP, van der Heijden F, van Oijen MG, Siersema PD, Broeders IA (2016) Colonoscopy with robotic steering and automated lumen centralization: a feasibility study in a colon model. Endoscopy 48:286–290
Rozeboom ED, Bastiaansen BA, de Vries ES, Dekker E, Fockens PA, Broeders IA (2016) Robotic-assisted flexible colonoscopy: preliminary safety and efficiency in humans. Gastrointest Endosc 83:1267–1271
Kume K, Sakai N, Goto T (2015) Development of a novel endoscopic manipulation system: the Endoscopic Operation Robot ver.3. Endoscopy 47:815–819
Sakurazawa N, Kato S, Miyashita M, Kiyama T, Fujita I, Yamashita N, Saitou Y, Tajiri T, Uchida E (2009) An innovative technique for endoscopic submucosal dissection of early gastric cancer using a new spring device. Endoscopy 41:929–933
Ho KY, Phee SJ, Shabbir A, Low SC, Huynh VA, Kencana AP, Yang K, Lomanto D, So BY, Wong YY, Chung SC (2010) Endoscopic submucosal dissection of gastric lesions by using a Master and Slave Transluminal Endoscopic Robot (MASTER). Gastrointest Endosc 72:593–599
Ikeda K, Sumiyama K, Tajiri H, Yasuda K, Kitano S (2011) Evaluation of a new multitasking platform for endoscopic full-thickness resection. Gastrointest Endosc 73:117–122
Phee SJ, Reddy N, Chiu PW, Rebala P, Rao GV, Wang Z, Sun Z, Wong JY, Ho KY (2012) Robot-assisted endoscopic submucosal dissection is effective in treating patients with early-stage gastric neoplasia. Clin Gastroenterol Hepatol 10:1117–1121
Diana M, Chung H, Liu KH, Dallemagne B, Demartines N, Mutter D, Marescaux J (2013) Endoluminal surgical triangulation: overcoming challenges of colonic endoscopic submucosal dissections using a novel flexible endoscopic surgical platform: feasibility study in a porcine model. Surg Endosc 27:4130–4135
Nakadate R, Nakamura S, Moriyama T, Kenmotsu H, Oguri S, Arata J, Uemura M, Ohuchida K, Akahoshi T, Ikeda T, Hashizume M (2015) Gastric endoscopic submucosal dissection using novel 2.6-mm articulating devices: an ex vivo comparative and in vivo feasibility study. Endoscopy 47:820–824
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Tsutomu Iwasa, Ryu Nakadate, Shinya Onogi, Yasuharu Okamoto, Jumpei Arata, Susumu Oguri, Haruei Ogino, Eikichi Ihara, Kenoki Ohuchida, Tomohiko Akahoshi, Tetsuo Ikeda, Yoshihiro Ogawa, and Makoto Hashizume have no conflicts of interest or financial ties to disclose.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (MP4 20981 KB)
Rights and permissions
About this article
Cite this article
Iwasa, T., Nakadate, R., Onogi, S. et al. A new robotic-assisted flexible endoscope with single-hand control: endoscopic submucosal dissection in the ex vivo porcine stomach. Surg Endosc 32, 3386–3392 (2018). https://doi.org/10.1007/s00464-018-6188-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-018-6188-y