Abstract
Purpose
To determine the association between lymphovascular invasion (LVI) and upper tract urothelial carcinoma (UTUC) among patients who underwent radical nephroureterectomy (RNU).
Materials and methods
From 2003 and 2018, retrospective data of 453 patients treated for UTUC with open, laparoscopic, or robotic RNU were collected. Pathological specimens were assessed for LVI through hematoxylin and eosin staining. According to presence of LVI, patients were stratified into two groups and compared for perioperative characteristics. Kaplan–Meier analysis was used to assess progression-free (PFS), cancer-specific (CSS), and overall survival (OS). Uni- and multivariate Cox regression models were used to find significance of LVI to survival.
Results
LVI was present in 132 (29.1%) of patients and was associated with higher age and lower preoperative GFR. Pathological outcomes included significantly higher tumor grade, higher rates of lymph node invasion and more positive surgical margins. During median 23.2 months follow-up (mean 37.1 months), 59.2% (n = 268) of total patients had tumor recurrence, with highest incidences in lymph nodes (51.5%). 5-year PFS, CSS, and OS were estimated at 35.4%, 94.6%, and 91.1% in LVI-negative patients and 17.2%, 75.1%, and 70.8% in LVI-positive patients, respectively (all p < 0.001). Multivariate analysis showed LVI to be an independent predictor of PFS (HR = 1.480; p = 0.018).
Conclusion
LVI is an independent predictor of adverse PFS and is associated with poor CSS and OS in patients undergoing RNU for UTUC. These results may guide clinicians in selecting patients for adjuvant chemotherapy. Future prospective trials are necessary to further validate our results.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Upper urinary tract urothelial carcinoma (UTUC), defined as a tumor originating from the surface epithelium of the urothelial lining, is a relatively rare tumor representing only 10% of all renal and 5% of all urothelial malignancies(Godfrey et al. 2012; Liu et al. 2019). Despite advances in endoscopic and nephron-sparing surgery, radical nephroureterectomy (RNU) with bladder cuff excision remains the mainstay of treatment for UTUC. UTUC is associated with relatively poor outcome, with previous literature suggesting difficulty in early detection, anatomically thinner muscular and submucosal layers, and the lack of serosa compared to bladder cancer to be associated with a higher rate of advanced disease, accounting for nearly 60% of total cases (Liu et al. 2019; Van der Poel et al. 2005). To date, several studies have identified predictors for adverse prognosis, including age, tumor grade, tumor stage, lymph node metastasis, and concurrent carcinoma in situ (CIS) (Godfrey et al. 2012; Kikuchi et al. 2005; Ku et al. 2013).
Lymphovascular invasion (LVI), or invasion of tumor cells into the vascular or lymphatic systems, has been established as a factor for poor prognosis in other solid tumors including liver, colorectal, prostate, bladder, and testicular cancer (Ku et al. 2013; Lee et al. 2015). Entry into the angiolymphatic channels allows for initiation of tumor dissemination, and while several studies have reported poor prognosis in UTUC (Godfrey et al. 2012; Kikuchi et al. 2005), others have found the association to be insignificant (Colin et al. 2012; Yoshida et al. 2017), making the role of LVI still controversial. In this study, we aimed to further elucidate independent prognostic factors and identify the relationship of LVI with survival outcome in UTUC patients undergoing RNU at a single institution.
Materials and methods
Study population
This was an institutional review board-approved (no. SNUBH-B-1907-552-110), retrospective single-institution study comprised of 453 patients diagnosed with UTUC and treated with open, laparoscopic, or robotic RNU between 2003 and 2018. Of the 453 patients, LVI was present in 132 patients and absent in 321 patients. We excluded patients with a history of muscle-invasive urothelial carcinoma of the bladder, history of neoadjuvant therapy, distant metastasis prior to surgery, or incomplete data. Surgical technique and the extent of lymphadenectomy were decided based on the individual surgeon’s discretion. Regional lymphadenectomy was performed when suspicious lesions were identified in preoperative imaging or enlarged in intraoperative examination.
Pathologic evaluation
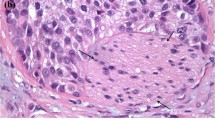
All surgical specimens after RNU were assessed in detail by a single uro-pathologist according to standard pathologic procedures. Tumor stage and grading were evaluated according to the 2002 American Joint Committee on Cancer (AJCC)/Union Internationale Contre le Cancer (UICC) TNM classification and 1998 World Health Organization (WHO)/International Society of Urologic Pathology (ISUP) consensus classification, respectively (Epstein et al. 1998; Greene et al. 2002). Clinicopathologic data included age, patient co-morbidity, presence of concurrent bladder cancer, laterality and location of the primary tumor, tumor stage, tumor grade, presence of LVI, surgical margin, venous or perineural invasion, squamous or glandular metaplasia, and presence of carcinoma in situ (CIS). LVI was defined as the presence of tumor cells in endothelium-lined lymphovascular channels without underlying muscular walls (Hasui et al. 1992). Routine light microscopic examination with hematoxylin and eosin was performed to identify LVI. Immunohistochemical techniques were not routinely used as to match standard practice of pathologists.
Statistical analyses
The clinical and pathological covariates between the two groups with or without LVI were evaluated by a Chi squared test for categorical variables and an independent t test for continuous variables. Further evaluation was performed for the node-negative subgroup to assess predictability of disease recurrence and survival. Patients with negative pathological lymph nodes and those who did not undergo lymphadenectomy were grouped together for the purpose of analysis. Progression-free (PFS), cancer-specific (CSS) and overall survival (OS) curves were generated by a Kaplan–Meier analysis, with survival rate differences assessed with log rank statistics. Univariate and multivariate Cox proportional hazard models were applied to address the effect of LVI for tumor recurrence after adjusting for presence of preoperative hydronephrosis and hypertension, BMI, tumor stage, and lymph node invasion. Statistical analysis was performed with IBM SPSS software package version 21.0 (Statistical Package for Social Sciences™, Chicago, IL, USA). A two-tailed p value < 0.05 was considered significant for all analyses.
Results
Baseline characteristics of patients with or without LVI
The median age of 453 patients enrolled in the study was 69 years, with a median and mean follow-up duration of 23.2 months and 37.1 months, respectively (range 0–172 months). LVI was present in 132 (29.1%) patients, and baseline characteristics were similar to the LVI absent group (n = 321, 70.9%) (Table 1). The LVI (+) group had significantly higher age (68.79 vs 65.44, p = 0.025), lower preoperative GFR (62.65 vs 70.40, p = 0.002), and higher rate of concurrent bladder cancer at the time of surgery (27.3% vs 18.1%, p = 0.028). There was no difference in tumor laterality or location, nor with patient characteristics such as gender, ECOG performance status, or pre-existing comorbidities (DM, HTN).
Perioperative and pathological outcomes
Operation methods, selected based on the surgeon’s discretion, was significantly different between the two groups, with a larger percentage of patients with LVI undergoing open surgery rather than laparoscopic or robotic operations (Table 1). There was no significant difference in average operation time (230.25 vs 227.03 min, p = 0.685). The LVI (−) group had significantly less estimated blood loss (239.35 vs 325.98 ml, p = 0.020) and less transfusion rates (15.6% vs 28.8%, p = 0.010), which can be attributed to the larger percentage of patients having underwent robotic surgery (36.8% vs 21.2%).
Final pathologic outcomes include significantly larger tumor volume (5.45 vs 3.99 cm), higher tumor grade, and higher rates of lymph node invasion (29.5% vs 2.2%) in the LVI (+) group. Positive surgical margins (15.9% vs 2.8%), venous/perineural invasion (42.0% vs 3.8%), squamous/glandular metaplasia (33.3% vs 17.4%), and CIS (22.7 vs 12.1%) were frequently observed in patients with LVI (p < 0.001). There was no difference in the percentage of ureteroscopic biopsy, interval from preoperative ureteroscopy to nephroureterectomy, and postoperative complications.
Progression, cancer-specific mortality, and overall mortality outcomes
During the follow-up period, 59.2% (268) of total patients experienced tumor recurrence (Table 2). 77.3% (102) of patients with positive LVI had recurrent tumor, most frequently occurring in lymph nodes (51.5%), followed by lung (28.8%), bladder (26.5%), bone (19.7%), and liver (15.9%). Other sites of metastasis include contralateral ureter and renal pelvis (9.9%), pelvic cavity (3.0%), colorectal (4.5%), adrenal gland (3.0%), and brain (3.0%) in LVI (+) groups. There were single incidences of neck and spleen metastasis in the LVI (+) group and penile solid tumor metastasis in the LVI (−) group. 46 (10.2%) of all patients died during the follow-up period, 32 (7.1%) deaths of whom were cancer-related. LVI was associated with worse PFS as well as CSS and OS (p < 0.001). 5-year PFS in all patients was 35.4% in LVI (−) vs. 17.2% in LVI (+); 5-year CSS in all patients 94.6% in LVI (−) vs. 75.1% in LVI (+); and 5-year OS in all patients 91.1% in LVI (−) vs. 70.8% in LVI (+) (Fig. 1).
LVI status, as well as BMI, tumor stage (≥ T3), lymph node invasion, preoperative hydronephrosis, and hypertension, were established as potential predictors of PFS on univariate analysis (Table 3). Adjusting for the significant factors on multivariate analysis demonstrated LVI was an independent predictor of PFS (HR = 1.480; 95% CI = 1.071–2.045; p = 0.018). Subgroup analysis in patients with pN0/x suggests LVI to have a similar negative impact on PFS, CSS, and OS, with 5-year PFS 36.2% in LVI (−) vs. 24.3% in LVI (+), 5-year CSS 95.3% in LVI (−) vs. 82.6% in LVI (+), and 5-year OS 92.0% in LVI (−) vs. 76.5% in LVI (+) (all p < 0.001). However, LVI was not found to be a statistically significant predictor of CSS or OS on multivariate analysis (HR = 1.809, p = 0.202; HR = 1.737, 0.158, respectively).
Additional survival analysis was done comparing patients with T2 and T3 disease, separated by presence of LVI. While pathologic (p) T2 with LVI (+) group and pT3 with LVI (−) showed a notable separation in the PFS Kaplan–Meier curve (Fig. 2), such difference was diminished in CSS and OS, with no statistical significance between the two groups (p = 0.188, p = 0.461, p = 591, respectively). However, patients with pT3 and LVI (+) had significantly poor PFS, CSS, and OS when compared to pT3 and LVI (−) patients (p = 0.016, p = 0.003, p = 0.001, respectively).
Kaplan–Meier curves showing a progression-free survival, b cancer-specific survival, c overall survival in stage pT2 and T3 patients who underwent RNU for UTUC according to LVI [blue line: pT2 and LVI (−), 122 patients, green line: pT2 and LVI (+), 25 patients, yellow line: pT3 and LVI (−), 63 patients, purple line: pT3 and LVI (+), 20 patients]
Discussion
The impact of LVI on the prognostic outcome of patients undergoing RNU for UTUC remains to be debated, as certain previous literature associate the pathological profile with poor outcome (Ku et al. 2013; Mellouli et al. 2017; Novara et al. 2010), while others have failed to establish a significant correlation (Colin et al. 2012; Mathieu 2015; Yoshida et al. 2017). In this study, we retrospectively analyzed our single institutional experience with patients who underwent RNU for UTUC. In line with previous reports (Kikuchi et al. 2005, 2009; Saito et al. 2007), approximately 30% of our patients had LVI, which was associated with higher rate of concurrent bladder cancer at time of surgery, as well as increased risk of positive margin status, venous/perineural invasion, squamous/glandular differentiation, and CIS. Univariate and multivariate analyses indicated LVI to be an independent predictor of progression-free survival (HR = 1.480; 95% CI = 1.071–2.045; p = 0.018), which was consistent in the pN0/x subgroup. Our findings support findings by previous research postulating tumor staging to be an important prognostic factor (Zigeuner and Pummer 2008), with tumor stage over ≥ T3 to have a HR of 1.611 on multivariate analysis (p value = 0.002). Progression related to LVI occurred most frequently in lymph nodes as well as lung, bladder, bone, and liver. The metastatic pattern closely resembles previous literature by Shinagare et al. (2011), where metastasis occurred most frequently in lymph node (75%), lung (65%), liver (54%), and bone (39%). Literature on sites of metastasis in UTUC are limited, and the high prevalence of recurrence (59% overall; 77.3% in LVI (+) vs. 51% in LVI (−), p < 0.001) highlight the possibility of micrometastases prior to surgery.
Previous articles have established the clinicopathological association of LVI in lymph-node positivity and poor prognostic outcome in other urological malignancies including penile, prostate, and bladder cancer (Ku et al. 2013). Intuitively, LVI provides a key pathway of tumor cell dissemination and metastatic spread, with initial preferential entry into local lymphatic and vascular systems and sequential invasion through routes of natural drainage (Pepper 2001). Previous meta-analysis of 17 trials including a total of 4896 patients demonstrated LVI to be a significant predictor of mortality in UTUC (Ku et al. 2013). LVI in bladder cancer also predicted shorter PFS and OS in patients undergoing radical cystectomy, with a pooled hazard ratio of 1.57 and 1.59, respectively (Mari et al. 2018). Based on these studies, some have argued for its inclusion in urothelial carcinoma TNM staging (Kikuchi et al. 2009), but it is inherently difficult to assess LVI at a morphological level, which inevitably leads to internal inconsistencies between pathologists (Verhoest et al. 2011). Additional analysis via immunohistochemical staining, which is only performed in select equivocal cases and not routinely used in clinical practice, may be obtained to achieve increased accuracy (Godfrey et al. 2012).
In our present study, subgroup analysis of patients with pN0/x showed negative impact of LVI on PFS, which is in line with a previous report by Liu et al. (Liu et al. 2019) that indicated LVI to be a significant predictor of PFS and CSS in node-negative patients (HR = 3.732 and 3.825, respectively). Also, survival analysis on subgroups of pT2 and T3 has shown a trend towards poor prognosis, especially in pT3 patients with LVI compared to patients without LVI. This is comparable to reports by Danzig et al. (2018), which looked at the prognostic impact of LVI in patients with UTUC limited to the renal pelvis, where patients with pT3 LVI (+) disease had an adverse survival pattern that was comparable to pT4 LVI (−) disease, even when controlling for lymph node status on multivariate analysis. Based on our results, certain groups with pT2 LVI (+) disease show a survival pattern that is not statistically different from pT3 LVI (−), which merits further investigation and possible consideration in the selection for adjuvant treatment.
While adjuvant chemotherapy is not currently recommended due to ambiguous results from observational studies (Necchi et al. 2018; Rouprêt et al. 2018), some reports have suggested an overall survival benefit in patients with locally advanced pT3/T4 tumor and/or positive lymph nodes (Seisen et al. 2017). In contrast, poor postoperative renal function makes cisplatin-based chemotherapy inadequate in a large percentage of patients with UTUC, making the role of adjuvant chemotherapy controversial (Lee et al. 2018). In light of recent evidence supporting poor prognosis in LVI (+), node-negative patients, further risk stratification based on LVI status could provide additional information for selection of candidates for adjuvant chemotherapy.
Our study had several limitations. First, this was a single institution, retrospective study, which by design has the risk of intrinsic bias. The retrospective nature of the study further obscures the possibility of higher rates of micro-LN metastasis in patients with positive LVI despite perioperative clinical N0 or Nx disease. Also, most patients did not undergo routine lymphadenectomy, which may have led to inaccurate assessment of node invasion at the time of surgery. However, the percentage of patients who underwent lymphadenectomy (n = 141, 31.1%) do not differ significantly from previous studies, ranging from 30 to 40% (Kikuchi et al. 2009; Lee et al. 2015). Our study also did not assess the mean or median number of lymph nodes dissected; however, previous reports have shown that number of removed lymph nodes did not predict PFS and CSS (Kim et al. 2010). Also, LVI was shown to be a statistically significant predictor of PFS but not CSS or OSS, which may be in part due to the relatively short median and mean follow-up durations (23.2 months and 37.1 months, respectively). Finally, the long study interval spanning from 2003 to 2018 over a period of 15 years may not accurately reflect the current clinical practice. Future large-scale randomized prospective trials may be necessary to further elucidate the effect of LVI on patient prognosis, but this may be limited by the relatively low incidence of the disease.
In conclusion, LVI is an independent predictor of adverse PFS in patients undergoing RNU for UTUC, which is also apparent in the node-negative subgroup. Further risk stratification for staging and prognosis estimation may provide additional criteria to guide selection of patients for adjuvant chemotherapy. Future prospective studies should be performed to further validate our results.
References
Colin P et al (2012) Influence of positive surgical margin status after radical nephroureterectomy on upper urinary tract urothelial carcinoma survival. Ann Surg Oncol 19:3613–3620
Danzig MR et al (2018) Prognostic importance of lymphovascular invasion in urothelial carcinoma of the renal pelvis. Cancer 124:2507–2514. https://doi.org/10.1002/cncr.31372
Epstein JI, Amin MB, Reuter VR, Mostofi FK, Committee BCC (1998) The World Health Organization/International Society of Urological Pathology consensus classification of urothelial (transitional cell) neoplasms of the urinary bladder. Am J Surg Pathol 22:1435–1448
Godfrey MS, Badalato GM, Hruby GW, Razmjoo M, McKiernan JM (2012) Prognostic indicators for upper tract urothelial carcinoma after radical nephroureterectomy: the impact of lymphovascular invasion. BJU Int 110:798–803
Greene FL, Page D, Fleming I, Fritz A, Balch C, Haller D, Morrow M (2002) AJCC (American Joint Committee on Cancer) cancer staging manual. Springer, New York
Hasui Y, Nishi S, Kitada S, Osada Y, Asada Y (1992) The prognostic significance of vascular invasion in upper urinary tract transitional cell carcinoma. J Urol 148:1783–1785
Kikuchi E, Horiguchi Y, Nakashima J, Hatakeyama N, Matsumoto M, Nishiyama T, Murai M (2005) Lymphovascular invasion independently predicts increased disease specific survival in patients with transitional cell carcinoma of the upper urinary tract. J Urol 174:2120–2124
Kikuchi E et al (2009) Lymphovascular invasion predicts clinical outcomes in patients with node-negative upper tract urothelial carcinoma. J Clin Oncol 27:612
Kim DS, Lee YH, Cho KS, Cho NH, Chung BH, Hong SJ (2010) Lymphovascular invasion and pT stage are prognostic factors in patients treated with radical nephroureterectomy for localized upper urinary tract transitional cell carcinoma. Urology 75:328–332
Ku JH, Byun S-S, Jeong H, Kwak C, Kim HH, Lee SE (2013) Lymphovascular invasion as a prognostic factor in the upper urinary tract urothelial carcinoma: a systematic review and meta-analysis. Eur J Cancer 49:2665–2680
Lee H-Y et al (2015) Prognostic significance of lymphovascular invasion in upper urinary tract urothelial carcinoma is influenced by tumor location. Ann Surg Oncol 22:1392–1400
Lee BH et al (2018) Renal function recovery after radical nephroureterectomy for upper tract urothelial carcinoma. World J Urol 36:257–263
Liu W, Zhou Z, Dong D, Sun L, Zhang G (2019) Prognostic Value of Lymphovascular Invasion in node-negative upper urinary tract urothelial carcinoma Patients undergoing radical nephroureterectomy. Yonsei Med J 60:174–181
Mari A, Kimura S, Foerster B, Abufaraj M, D’Andrea D, Gust KM, Shariat SF (2018) A systematic review and meta-analysis of lymphovascular invasion in patients treated with radical cystectomy for bladder cancer. In: Urologic oncology: seminars and original investigations, vol 6, Elsevier, Amsterdam, pp 293–305
Mathieu R et al (2015) Survivin is not an independent prognostic factor for patients with upper tract urothelial carcinoma: a multi-institutional study. In: Urologic oncology: seminars and original investigations, vol 11, Elsevier, Amsterdam, pp 495, e415–495, e422
Mellouli M et al (2017) Prognostic role of lymphovascular invasion in patients with urothelial carcinoma of the upper urinary tract. Urol J 14:5008–5012
Necchi A et al (2018) Adjuvant chemotherapy after radical nephroureterectomy does not improve survival in patients with upper tract urothelial carcinoma: a joint study by the European Association of Urology-Young Academic Urologists and the Upper Tract Urothelial Carcinoma Collaboration. BJU Int 121:252–259
Novara G et al (2010) Prognostic role of lymphovascular invasion in patients with urothelial carcinoma of the upper urinary tract: an international validation study. Eur Urol 57:1064–1071
Pepper MS (2001) Lymphangiogenesis and tumor metastasis: myth or reality? Clin Cancer Res 7:462–468
Rouprêt M et al (2018) European association of urology guidelines on upper urinary tract urothelial carcinoma: 2017 update. Eur Urol 73:111–122
Saito K, Kawakami S, Fujii Y, Sakura M, Masuda H, Kihara K (2007) Lymphovascular invasion is independently associated with poor prognosis in patients with localized upper urinary tract urothelial carcinoma treated surgically. J Urol 178:2291–2296
Seisen T et al (2017) Effectiveness of adjuvant chemotherapy after radical nephroureterectomy for locally advanced and/or positive regional lymph node upper tract urothelial carcinoma. J Clin Oncol 35:852–860
Shinagare AB, Fennessy FM, Ramaiya NH, Jagannathan JP, Taplin ME, Van den Abbeele AD (2011) Urothelial cancers of the upper urinary tract: metastatic pattern and its correlation with tumor histopathology and location. J Comput Assist Tomogr 35:217–222
Van der Poel H, Antonini N, Van Tinteren H, Horenblas S (2005) Upper urinary tract cancer: location is correlated with prognosis. Eur Urol 48:438–444
Verhoest G et al (2011) Predictive factors of recurrence and survival of upper tract urothelial carcinomas. World J Urol 29:495–501
Yoshida T, Kinoshita H, Shimada S, Sugi M, Matsuda T (2017) Preoperative pyuria is a poor prognostic factor in patients with urothelial carcinoma of the upper urinary tract after surgery. Clin Genitourinar Cancer 15:e543–e550
Zigeuner R, Pummer K (2008) Urothelial carcinoma of the upper urinary tract: surgical approach and prognostic factors. Eur Urol 53:720–731
Funding
The present study did not receive any funding.
Author information
Authors and Affiliations
Contributions
JJO: protocol/project development. CHY, SL, SKH, S-SB, SEL: data collection or management. SHS: data analysis, manuscript writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest related to this manuscript.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Institutional review board approval was obtained (no. SNUBH-B-1907-552-110).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Song, S.H., Ye, C.H., Lee, S. et al. Association between lymphovascular invasion and oncologic outcomes among upper urinary tract urothelial carcinoma patients who underwent radical nephroureterectomy. J Cancer Res Clin Oncol 145, 2863–2870 (2019). https://doi.org/10.1007/s00432-019-03020-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-019-03020-z