Abstract
Purpose
Several studies compared the outcomes of laparoscopically completed colorectal resections (LCR) to those requiring conversion to open surgery (COS). However, a comparative analysis between COS patients and patients undergoing planned open surgery (POS) would be useful to clarify if the conversion can be considered a simple drawback or a complication, being cause of additional postoperative morbidity. The aim of this study is to perform a meta-analysis of current evidences comparing postoperative outcomes of COS patients to POS patients.
Methods
A systematic search of Medline, ISI Web of Knowledge, and Scopus was performed to identify studies reporting short-term outcomes of COS and POS patients. Primary outcomes were 30-day overall morbidity and length of postoperative hospital stay. Data were analyzed with fixed-effect modeling, and sensitivity analyses were performed to test the robustness of the results.
Results
Twenty studies involving 30,656 patients undergoing POS and 1935 COS patients were selected. The mean conversion rate was 0.17. Similar 30-day overall morbidity and length of postoperative hospital stay were found in COS and POS patients. Wound infection (OR 1.43, 95 % CI 1.12 to 1.83, p < 0.01) was higher in the COS group. Other results were robust. Outcomes were comparable for patients undergoing resection for different natures of the disease (benign vs. malignant) and at different sites (colon vs. rectum).
Conclusion
Conversions from laparoscopic to open procedure during colorectal resection are not associated with a poorer postoperative outcome compared to patients undergoing planned open surgery, except for a higher risk of wound infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Conversion from laparoscopic to open procedure in colorectal surgery is reported with a widely variable rate (5.2 to 77 %) [1, 2]. Intention-to-treat analyses of randomized controlled trials (RCTs) considering procedures converted to open surgery (COS) for the laparoscopic group have shown that the minimally invasive approach is not inferior to the open approach [3–6]. Nevertheless, it is interesting to analyze the postoperative results in COS patients.
Several studies have compared the outcomes of COS procedures to laparoscopically completed colorectal resections (LCR), in some cases showing increased morbidity [7, 8], mortality [8], and length of hospital stay [9, 10]. However, the right yardstick for patients who require conversion during LCR should be patients undergoing planned open surgery (POS). A comparative analysis would clarify if the conversion has to be considered a simple drawback or a complication, causing additional postoperative morbidity. Evidence comparing these two groups of patients is controversial; some studies showed that COS patients may have a worse outcome in terms of postoperative course [3, 11, 12] and a poorer long-term oncologic outcome [13–15] than POS patients, while other studies showed no differences [16, 17] and one study found better outcome [18].
The aim of this study is to perform a meta-analysis of current evidence, evaluating the short-term outcomes of COS procedures compared to POS ones.
Material and methods
Search strategy and selection criteria
We searched Medline, SCOPUS, and Web of Science with no language, publication date, or publication status restrictions. The last search was run on September 9, 2014, using the following search terms: “laparoscop*,” “pneumoperitoneum,” “conver*,” “colon*,” “colectomy,” “colorectal,” “rectum,” “rectal,” “sigmoid,” “hemicolectomy,” “crohn,” and “ulcerative colitis” (Appendix 1, see Supporting information). The reference list of the identified articles was also checked to identify other potentially relevant studies.
To be considered eligible, a study had to report data on perioperative outcomes in patients undergoing planned open surgery (POS group) and in patients converted to open surgery (COS group) after a failed laparoscopic attempt. Studies including patients undergoing emergency colorectal resections were excluded. Two reviewers (MDs) independently assessed the reports for eligibility at the title and abstract levels. Divergences were resolved by a third reviewer. The full text of selected reports was then retrieved for further analysis.
Data extraction and methodological quality appraisal
Two authors independently extracted data from included studies and filled an electronic database with the following information: first author and publication year, study design, surgeon experience, definition of the term conversion, rate and reason of conversion, and characteristics of study population (age, gender, BMI, ASA index, nature of disease, type of resection, stage of disease).
The Newcastle–Ottawa scale (NOS) was used to assess the quality of a study on a scale of 9, with a greater score indicating better quality. Further assessment of the selection bias was made by considering if the COS and POS groups were comparable for six variables: age, gender, American Society Anesthesiology (ASA) index, body mass index (BMI), site of resection, and stage of disease. Thus, every study was considered to be at low (5–6), medium (3–4), or high risk (0–2) of selection bias, depending on the number of variables that were balanced. If a variable was missing, it was considered not to be balanced.
Outcome analysis
Primary outcomes were 30-day overall morbidity and length of postoperative hospital stay. Secondary outcomes were 30-day mortality, operating time, estimated blood loss, anastomotic leak, re-operation, postoperative bleeding, postoperative bowel obstruction, wound infection, pneumonia, sepsis, cardiovascular complications, and deep venous thrombosis. The odds ratio (OR) and 95 % confidence interval (95 % CI) were used as summary measures for discrete outcomes, while the weighted mean difference (WMD) and 95 % CI were used as summary measures for continuous outcomes. In the absence of statistical heterogeneity, the fixed-effect Mantel-Hænzel model was used. Otherwise, a random-effect DerSimonian and Laird model was used. The heterogeneity among the studies was tested by the Q statistic and quantified by the I 2 statistic. As a guide, I 2 values of <25 % indicated low heterogeneity, 25–50 % indicated moderate heterogeneity, and >50 % indicated high [19] heterogeneity. For dichotomous analyses with zero count cells, 0.5 was added to each cell. Subgroup analysis was planned to establish whether the type of disease (cancer) or type of resection (rectal, colonic) affected the results. The presence of a correlation between the year of the study and the conversion rate was assessed using Spearman’s rho statistic.
Sensitivity analyses
Additional analyses were performed to test if the results were robust to our methodological assumptions. When a fixed-effect model was used, the meta-analysis was repeated using a random-effect model. The influence of each individual study on the analysis was investigated by omitting each study in turn and re-estimating the summary effect and the heterogeneity.
To further explore whether the results were affected by potential confounding factors, a meta-regression analysis was performed. Study characteristics (year of publication, study design, surgeon experience, conversion rate), study quality (NOS), and risk of selection bias were tested as potential effect modifiers. Publication bias was assessed by graphical inspection of the funnel plot to detect asymmetry. Symmetry of the funnel plot was also tested using Egger’s linear regression method and Harbord’s modified test. Statistical analyses were performed using STATA 12 statistical software (STATA Corp, College Station, Texas, USA). The study was realized according to the preferred reporting items for systematic reviews and meta-analyses (PRISMA) statement [20].
Results
Study selection
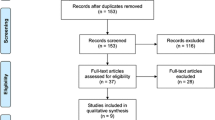
The search of the Medline, Web of Science, and Scopus databases provided a total of 4617 citations. Three additional studies [3, 21, 22] were identified by checking the references. One thousand eight hundred forty-seven duplicated studies were found and removed. Of the 2770 remaining, 2691 studies were discarded because they clearly did not meet the inclusion criteria after reviewing the title or abstract. The full text of the remaining 79 articles was examined in more detail. After excluding 59 studies, 20 studies [11, 12, 15, 16, 18, 23–36] were included in the meta-analysis (Fig. 1). No relevant unpublished studies were found.
Study characteristics and quality assessment
The selected studies included 41,741 patients: 30,656 patients underwent POS, while 11,085 patients had an LCR. Of these, 1935 were converted to an open procedure. The mean conversion rate was 0.17, ranging from 0.07 to 0.46. The more recent the study, the lower the conversion rate was (Spearman’s rho −0.68, p = 0.002). However, reasons for conversion to open surgery (intraoperative findings vs. complication) remained constant over the years (Spearman’s rho −0.1, p = 0.64). Characteristics of included studies are shown in Table 1. The indication for surgery was colorectal cancer in 13 studies [3, 15, 21, 22, 25–31, 33, 34], while seven studies [11, 12, 16, 18, 23, 24, 32] analyzed resections both for malignant and benign disease. Five studies included only rectal resections [15, 21, 30, 31, 34] while five studies analyzed colonic resections only [11, 16, 22, 26, 27]. Table 2 shows the assessment of the risk of bias. The definition of the term “conversion” in the selected studies is reported in Table 3.
Outcome analysis
All studies provided information on 30-day mortality, and the Forest plot showed no difference between COS and POS (OR, 1.1; 95 % CI, 0.83 to 1.46). The incidence of overall postoperative 30-day morbidity was reported in 13 studies [3, 12, 18, 21, 23–25, 28–31, 33, 34]. The results were similar in the two groups (OR, 1.16; 95 % CI, 0.86 to 1.56) although a significant heterogeneity was found (Q, 35.5; p = 0; I 2 = 66.2 %) (Fig. 2). Conversion to open surgery was associated with a higher incidence of postoperative pneumonia (OR, 1.54; 95 % CI, 1.16 to 2.04) as reported in nine studies [3, 16, 22–24, 26, 27, 32, 34] (Fig. 3a). Furthermore, the rate of wound infection was compared in 11 studies [3, 11, 12, 15, 16, 22–24, 27, 32, 34], and it was found to be higher in COS group (OR, 1.43; 95 % CI, 1.12 to 1.83) (Fig. 3b). The length of hospital stay was reported in 14 studies [3, 12, 15, 16, 18, 24–28, 31, 32, 34] (Fig. 4) with no difference between COS and POS (WMD, −0.12; 95 % CI, −1.14 to 0.89; significant heterogeneity Q, 65.10; p = 0, I 2 = 96.9 %) as for anastomotic leak rate (OR, 1.08; 95 % CI, 0.88 to 1.33) that was recorded in seven studies [3, 11, 15, 24, 29, 32, 34]. Operating time [11, 12, 16, 18, 22, 24–26, 28, 34, 37] was longer in the COS group (WMD, 57.59; 95 % CI, 44.55 to 70.63), while no difference was found in blood loss (WMD, 36.34: 95 % CI, −122.79 to 195.48), reported in five studies [18, 22, 23, 26, 28]. Among other secondary outcomes investigated, no differences were found in rates of re-operation, postoperative bleeding, postoperative obstruction, sepsis, cardiac complication, and deep venous thrombosis (Table 4). Subgroup analysis confirmed the results when studies were considered according to the nature of disease and site of resection (Table 5).
Sensitivity analysis and publication bias
The results obtained using a fixed-effect model were confirmed by repeating the analysis using a random-effect model (Table 4). The influence analysis showed that after exclusion of the studies by Hewett et al. [27] (OR, 1.64; 95 % CI, 0.92 to 2.91) or Guillou at al. [3] (OR, 1.35; 95 % CI, 0.98 to 1.86), there was no more difference in the risk of pneumonia. Meta-regression analysis showed that the results were not influenced by the quality of the study, the selection bias, or other study characteristics (Table 6). The funnel plot showed symmetry (Fig. 5), which was confirmed by Egger’s and Harbord’s modified tests (p > 0.6 for all tests).
Discussion
Several studies have compared the outcomes of COS procedures to laparoscopic completed colorectal resections. However, the right term for comparison of patients who required COS should be patients undergoing POS. Surgeons should answer the question, “Would the patient’s outcome have changed if the operation had been planned primarily as an open case?” In addition, when informing the patient about the procedure, a failed laparoscopic attempt should be presented not simply as a drawback, but as a complication, if the conversion is associated with a poorer postoperative outcome than POS.
The main finding of the study is that the postoperative course of COS patients does not differ from that of POS patients, except for a higher risk of wound infection. No difference was found in 30-day overall morbidity between COS and POS patients. Previously, two studies found a higher morbidity in the COS group [3, 12], but Slim et al. [12] reported data from an early laparoscopic experience. Consistent with this result, there was no difference in the length of postoperative hospital stay. Moreover, our analysis did not show any difference in the risk of anastomotic complication after conversion, in contrast to findings by Belizon et al. [11] and Slim et al. [12]. As expected, the operating time was longer in the COS group, which could explain the higher risk of pneumonia in these patients [3] revealed in our analysis. However, this result was not robust, since it depended on the singular inclusion of two [3, 27]. In particular, the study by Hewett et al. [27] has high risk of selection bias. Thus, we think that no conclusion should be drawn about the risk of pneumonia in COS patients. Despite the conversion rate decreasing in more recent studies, the outcomes of COS patients were substantially comparable to those of POS patients over the years. Consistent with this result, surgeon experience and the reason for conversion (findings vs. intraoperative complications) did not worsen postoperative outcomes in COS patients. This could be because most of the intraoperative complications that occurred could be repaired without having a significant impact on the postoperative course.
Some methodological aspects and limitations of this study should be considered. An inherent risk of selection bias is present in this analysis. The COS group is a negatively selected group, as patients requiring conversion are usually older, have more comorbidities, or have an advanced stage of disease [38, 39]. Additionally, in a nonrandomized setting, patients could have been selected for POS in view of the same characteristics. Each of these aspects alone could cause a poorer postoperative outcome, independently from the procedure (POS or COS).
In addition to the study quality (NOS), we assessed the presence of a selection bias by considering if the groups (POS and COS) were comparable for six variables (age, gender, ASA index, BMI, site of resection, stage of disease), which are risk factors for conversion and could act as confounding factors [38, 39]. The meta-regression analysis showed that the results did not change according to the NOS score, the number of variables balanced, and the status of each of these variables (balanced vs. not balanced). Another potential confounding factor is the number of previous surgeries. Unfortunately, this factor could not be considered in this assessment, since it was not reported in most of the studies. However, this and other unknown confounding factors are more likely to have been a cause of higher morbidity in the COS group than in our analysis, which does not show a substantial difference of morbidity.
The design of the included studies was heterogeneous (Table 1). We should point out that in this comparison, RCTs lose their advantages. Although POS and LCR groups are fully comparable because of randomization, the COS group is not comparable to the POS group as a result of the negative selection (Table 2). In view of the unpredictability of the conversion event [40], an observational study remains as the only conceivable evidence to study this topic. Compared to retrospective studies, prospective ones have a lower risk of measurement bias. However, sensitivity analysis showed that the results were comparable for prospective and retrospective studies. Study populations were heterogeneous according to the nature of disease (benign vs. malignant), the site of the disease (colon vs. rectum), and consequently, the type of operation. Subgroup analyses revealed comparable outcomes for these categories of patients.
Inter-study heterogeneity was present in the analysis of overall morbidity. This is a composite outcome, and a varying definition of the composition could account for this heterogeneity. This hypothesis is supported by the fact that no statistical heterogeneity was present in the analysis of each single outcome of morbidity. The definition of conversion varied across the studies, as there is still no consensus on this term. Nevertheless, the varying definition of conversion did not correspond with significant heterogeneity of the outcomes. This suggests that in most of the studies, the definition differed mainly on a formal level.
Intention-to-treat analyses of RCTs have shown that a minimally invasive approach is not inferior to the open approach when COS procedures are considered in the laparoscopic group [3–6]. This evidence might support a surgeon’s choice to attempt a minimally invasive colorectal resection when a laparoscopic operation is feasible and there are no obvious contraindications. We believe that our results further support this strategy, adding valuable information that the postoperative course of converted patients does not differ from that of patients undergoing POS, except for a higher risk of wound infection.
References
Gervaz P, Pikarsky A, Utech M, Secic M, Efron J, Belin B, Jain A, Wexner S (2001) Converted laparoscopic colorectal surgery. Surg Endosc 15:827–832. doi:10.1007/s004640080062
Schwandner O, Schiedeck TH, Bruch H (1999) The role of conversion in laparoscopic colorectal surgery: do predictive factors exist? Surg Endosc 13:151–156
Guillou PJ, Quirke P, Thorpe H, Walker J, Jayne DG, Smith AM, Heath RM, Brown JM (2005) Short term endpoints of conventional versus laparoscopic-assisted surgery in patients with colorectal cancer (MRC CLASICC trial): multicentre, randomised controlled trial. Lancet 365:1718–1726, doi:1010.1016/S0140-6736(05)66545-2
Neudecker J, Klein F, Bittner R, Carus T, Stroux A, Schwenk W (2009) Short-term outcomes from a prospective randomized trial comparing laparoscopic and open surgery for colorectal cancer. Br J Surg 96:1458–1467. doi:10.1002/bjs.6782
Veldkamp R, Kuhry E, Hop WCJ, Jeekel J, Kazemier G, Bonjer HJ, Haglind E, Påhlman L, Cuesta MA, Msika S, Morino M, Lacy AM (2005) Laparoscopic surgery versus open surgery for colon cancer: short-term outcomes of a randomised trial. Lancet Oncol 6:477–484, doi:10.1016/S1470-2045(05)70221-7
Van der Pas MH, Haglind E, Cuesta MA, Fürst A, Lacy AM, Hop WC, Bonjer HJ (2013) Laparoscopic versus open surgery for rectal cancer (COLOR II): short-term outcomes of a randomised, phase 3 trial. Lancet Oncol 14:210–218. doi:10.1016/S1470-2045(13)70016-0
Yamamoto S, Fukunaga M, Miyajima N, Okuda J, Konishi F, Watanabe M (2009) Impact of conversion on surgical outcomes after laparoscopic operation for rectal carcinoma: a retrospective study of 1,073 patients. J Am Coll Surg 208:383–389. doi:10.1016/j.jamcollsurg.2008.12.002
Marusch F, Gastinger I, Schneider C, Scheidbach H, Konradt J, Bruch HP, Köhler L, Bärlehner E, Köckerling F (2001) Importance of conversion for results obtained with laparoscopic colorectal surgery. Dis Colon Rectum 44:207–214, discussion 214–6
Lord SA, Larach SW, Ferrara A, Williamson PR, Lago CP, Lube MW (1996) Laparoscopic resections for colorectal carcinoma. A three-year experience. Dis Colon Rectum 39:148–154
Le Moine M-C, Fabre J-M, Vacher C, Navarro F, Picot M-C, Domergue J (2003) Factors and consequences of conversion in laparoscopic sigmoidectomy for diverticular disease. Br J Surg 90:232–236. doi:10.1002/bjs.4035
Belizon A, Sardinha CT, Sher ME (2006) Converted laparoscopic colectomy: what are the consequences? Surg Endosc 20:947–951. doi:10.1007/s00464-005-0553-3
Slim K, Pezet D, Riff Y, Clark E, Chipponi J (1995) High morbidity rate after converted laparoscopic colorectal surgery. Br J Surg 82:1406–1408
Ptok H, Kube R, Schmidt U, Köckerling F, Gastinger I, Lippert H (2009) Conversion from laparoscopic to open colonic cancer resection—associated factors and their influence on long-term oncological outcome. Eur J Surg Oncol 35:1273–1279. doi:10.1016/j.ejso.2009.06.006
Moloo H, Mamazza J, Poulin EC, Burpee SE, Bendavid Y, Klein L, Gregoire R, Schlachta CM (2004) Laparoscopic resections for colorectal cancer: does conversion survival? Surg Endosc 18:732–735. doi:10.1007/s00464-003-8923-1
Ströhlein MA, Grützner K-U, Jauch K-W, Heiss MM (2008) Comparison of laparoscopic vs. open access surgery in patients with rectal cancer: a prospective analysis. Dis Colon Rectum 51:385–391. doi:10.1007/s10350-007-9178-z
Casillas S, Delaney CP, Senagore AJ, Brady K, Fazio VW (2004) Does conversion of a laparoscopic colectomy adversely affect patient outcome? Dis Colon Rectum 47:1680–1685. doi:10.1007/s10350-004-0692-4
Rottoli M, Bona S, Rosati R, Elmore U, Bianchi PP, Spinelli A, Bartolucci C, Montorsi M (2009) Laparoscopic rectal resection for cancer: effects of conversion on short-term outcome and survival. Ann Surg Oncol 16:1279–1286. doi:10.1245/s10434-009-0398-4
Gonzalez R, Smith CD, Mason E, Duncan T, Wilson R, Miller J, Ramshaw BJ (2006) Consequences of conversion in laparoscopic colorectal surgery. Dis Colon Rectum 49:197–204. doi:10.1007/s10350-005-0258-7
Higgins JPT, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560. doi:10.1136/bmj.327.7414.557
Moher D, Liberati A, Tetzlaff J, Altman DG (2010) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 8:336–341. doi:10.1016/j.ijsu.2010.02.007
Laurent C, Leblanc F, Wütrich P, Scheffler M, Rullier E (2009) Laparoscopic versus open surgery for rectal cancer: long-term oncologic results. Ann Surg 250:54–61. doi:10.1097/SLA.0b013e3181ad6511
Curet MJ, Putrakul K, Pitcher DE, Josloff RK, Zucker KA (2000) Laparoscopically assisted colon resection for colon carcinoma perioperative results and long-term outcome. 1062–1066. doi:10.1007/s004640000092
Senagore AJ, Luchtefeld MA, Mackeigan JM, Mazier WP, Lloyd LR, Hoffman JP (1993) Open colectomy versus laparoscopic colectomy—are there differences. Am Surg 59:549–554
Begos DG, Arsenault J, Ballantyne GH (1996) Laparoscopic colon and rectal surgery at a VA hospital. Analysis of the first 50 cases. Surg Endosc 10:1050–1056
Bouvet M, Mansfield PF, Skibber JM, Curley SA, Ellis LM, Giacco GG, Madary AR, Ota DM, Feig BW (1998) Clinical, pathologic, and economic parameters of laparoscopic colon resection for cancer. Am J Surg 176:554–558
Kaiser AM, Kang J-C, Chan LS, Vukasin P, Beart RW (2004) Laparoscopic-assisted vs. open colectomy for colon cancer: a prospective randomized trial. J Laparoendosc Adv Surg Tech A 14:329–334. doi:10.1089/lap.2004.14.329
Hewett PJ, Allardyce RA, Bagshaw PF, Frampton CM, Frizelle FA, Rieger NA, Smith JS, Solomon MJ, Stephens JH, Stevenson ARL (2008) Short-term outcomes of the Australasian randomized clinical study comparing laparoscopic and conventional open surgical treatments for colon cancer: the ALCCaS trial. Ann Surg 248:728–738. doi:10.1097/SLA.0b013e31818b7595
Martinek L, Dostalik J, Gunkova P, Gunka I, Vavra P, Zonca P (2012) Impact of conversion on outcome in laparoscopic colorectal cancer surgery. Videosurgery Other Miniinvasive Tech 7:74–81. doi:10.5114/wiitm.2011.25799
Rottoli M, Stocchi L, Geisler DP, Kiran RP (2012) Laparoscopic colorectal resection for cancer: effects of conversion on long-term oncologic outcomes. Surg Endosc 26:1971–1976. doi:10.1007/s00464-011-2137-8
Mroczkowski P, Hac S, Smith B, Schmidt U, Lippert H, Kube R (2012) Laparoscopy in the surgical treatment of rectal cancer in Germany 2000–2009. Color Dis 14:1473–1478. doi:10.1111/j.1463-1318.2012.03058.x
Penninckx F, Kartheuser A, Van de Stadt J, Pattyn P, Mansvelt B, Bertrand C, Van Eycken E, Jegou D, Fieuws S (2013) Outcome following laparoscopic and open total mesorectal excision for rectal cancer. Br J Surg 100:1368–1375. doi:10.1002/bjs.9211
Kang CY, Halabi WJ, Chaudhry OO, Nguyen V, Ketana N, Carmichael JC, Pigazzi A, Stamos MJ, Mills S (2013) A nationwide analysis of laparoscopy in high-risk colorectal surgery patients. J Gastrointest Surg 17:382–391. doi:10.1007/s11605-012-2096-y
Kolfschoten NE, van Leersum NJ, Gooiker GA, Marang van de Mheen PJ, Eddes EH, Kievit J, Brand R, Tanis PJ, Bemelman WA, Tollenaar RA, Meijerink J, Wouters MW (2013) Successful and safe introduction of laparoscopic colorectal cancer surgery in Dutch hospitals. Ann Surg 257:916–921. doi:10.1097/SLA.0b013e31825d0f37
Rickert A, Herrle F, Doyon F, Post S, Kienle P (2013) Influence of conversion on the perioperative and oncologic outcomes of laparoscopic resection for rectal cancer compared with primarily open resection. Surg Endosc 27:4675–4683. doi:10.1007/s00464-013-3108-z
Taylor EF, Thomas JD, Whitehouse LE, Quirke P, Jayne D, Finan PJ, Forman D, Wilkinson JR, Morris EJ (2013) Population-based study of laparoscopic colorectal cancer surgery 2006–2008. Br J Surg 100:553–560. doi:10.1002/bjs.9023
Koo H, Kim H, Yun S, Lee W, Cho Y, Park Y, Yun J, Kim K (2013) Comparison of short-term and long-term outcomes in open surgery group, laparoscopic surgery group and conversion to open surgery group during laparoscopic colorectal cancer surgery. Dis Colon Rectum 56:E272
Jayne DG, Thorpe HC, Copeland J, Quirke P, Brown JM, Guillou PJ (2010) Five-year follow-up of the Medical Research Council CLASICC trial of laparoscopically assisted versus open surgery for colorectal cancer. Br J Surg 97:1638–1645. doi:10.1002/bjs.7160
Bhama AR, Charlton ME, Schmitt MB, Cromwell JW, Byrn JC (2014) Factors associated with conversion from laparoscopic to open colectomy using the National Surgical Quality Improvement Program (NSQIP) database. Color Dis. doi:10.1111/codi.12800
Masoomi H, Moghadamyeghaneh Z, Mills S, Carmichael JC, Pigazzi A, Stamos MJ (2015) Risk factors for conversion of laparoscopic colorectal surgery to open surgery: does conversion worsen outcome? World J Surg. doi:10.1007/s00268-015-2958-z
Cima RR, Hassan I, Poola VP, Larson DW, Dozois EJ, Larson DR, O’Byrne MM, Huebner M (2010) Failure of institutionally derived predictive models of conversion in laparoscopic colorectal surgery to predict conversion outcomes in an independent data set of 998 laparoscopic colorectal procedures. Ann Surg 251:652–658. doi:10.1097/SLA.0b013e3181d355f7
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supporting information
(DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Giglio, M.C., Celentano, V., Tarquini, R. et al. Conversion during laparoscopic colorectal resections: a complication or a drawback? A systematic review and meta-analysis of short-term outcomes. Int J Colorectal Dis 30, 1445–1455 (2015). https://doi.org/10.1007/s00384-015-2324-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-015-2324-5