Abstract
The performance of quantitative PCR-methods for the determination of beef and pork fraction in sausage was tested in an interlaboratory trial. Twelve different laboratories analysed four sausages of different composition of beef and pork by using four sausages of known fraction of meat as calibrators. Although different PCR-methods were applied, the precision of all results was better than 16% and the trueness better than 25%. The main reason for the good performance is the use of common calibrators emphasizing the importance of the quality of calibrators for molecular analysis. Thus, we conclude, that PCR-based detection methods are suitable for quantitative control of meat fractions in sausages.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fried sausages of veal (“Kalbsbratwurst”) are consumed in bulk quantities in Switzerland and its neighbouring countries. By law “Kalbsbratwurst” must be composed of at least 50/100 g veal and less pork [1]. As veal is much more expensive than pork, fraud may be attractive for producers in a highly competitive market. The determination of meat fractions in processed food such as sausages is therefore an important issue for official control laboratories. Usually, fraud encloses sausages with diminished content of veal. Only seldom the sausages contain no veal at all. To prosecute producers for their fraud, analytical methods must be able to quantify all expected components in this complex matrix.
Precision and accuracy are crucial performance criteria to ensure comparable results between control laboratories. Although PCR-based methods cannot distinguish veal from beef, they proved their applicability for the analysis of mixed processed food already in the past [8]. The precision and accuracy of PCR-methods for food species was usually estimated by comparing results generated by the same method [2–7, 9–11] and relative standard deviations (RSD) of about 30% were reported.
A basic problem of DNA-based methods influencing their accuracy is the composition of the sample. Sausages contain different tissue material, ranging from fatty bacon to fatless meat and connective tissue. These different tissues exhibit different concentrations of DNA and therefore the initial weight of meat in sausages may not be reflected by the species-specific DNA proportion received after DNA-isolation. This may lead to biased results generated by DNA-based detection methods. One approach often applied to compensate this possible lack of accuracy is to use appropriate reference material as calibrators simulating the real production process. In this study, four different reference sausages were produced as calibrators containing 30–60/100 g veal fraction in pork according to a traditional recipe of “Kalbsbratwurst”. Twelve laboratories from Switzerland and Germany used their own DNA extraction and PCR methods to determine the fractions of veal in four unknown samples using the above mentioned reference sausages for quantification. Here we present the results of this approach.
Materials and methods
Reference sausages
Reference sausages were produced according to the composition described in Table 1. According to a traditional recipe of “Kalbsbratwurst”, 10 kg of each sausage mixture was produced by the Master Butcher School Spiez, Switzerland. Reference sausages were analysed on the content of water, fat, total protein, connective tissue protein and muscle protein. The principal components were consistent with an average, commercially obtainable “Kalbsbratwurst” as estimated in a market analysis from 1965 to 1997 [12] (data not shown).
Samples A–D
Sample sausages A and B were produced similarly to the reference sausages by the Master Butcher School Spiez, Switzerland. Sample C was a sausage from the market. Sample D is a local speciality with a slightly different composition, mainly by addition of milk soaked white bread. The overall fat content is similar to the other sample sausages but with a higher relative fat proportion originating from veal lumbar fat. Thus, the composition of the unknown sausages differed significantly. An overview on the detailed composition of the samples is given in Table 2.
DNA based methods
A compilation of DNA-isolation and real-time PCR methods used is shown in Table 3.
Results
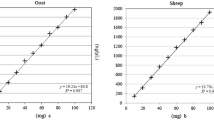
Eleven datasets of a total of 14 datasets produced by all 12 laboratories were used for the statistical evaluation. Three datasets did not correspond to the minimal requirements as previously agreed upon, and were excluded. The included data sets are presented in Table 4. All participants used the standard reference sausages to calibrate their assays. The statistical analysis of the datasets revealed that all mean values were very close to the true values of the recipe. The largest bias was observed for sample D with −5.6/100 g. Real-time PCR based detection methods often display a relative standard deviation (RSDR) of 30% [13]. In this study, different PCR-methods were performed in participating laboratories. Therefore, we expected an elevated value for the RSD, taking into account that different PCR-systems may differ in their performance caracteristics. In contrary to this expectation, a maximal RSDR value of 18% was found (sample C; mean value 48.8/100 g, reproducibility sR 8.9/100 g) leading to an extended uncertainty of measurement of 18/100 g (Table 5). Not unexpectedly, results of samples A and B, which were produced similarly to the reference sausages used as calibrators, exhibited the smallest values for repeatability (sr 1.4/100 and 3.5/100 g) and for reproducibility (sR 2/100 and 3.6/100 g) resulting in a minimal extended uncertainty of measurement of 4.3/100 g. It is worth mentioning that we found no significant difference between results generated with or without sample homogenization prior to DNA extraction, and no differences dependent on sample size, DNA extraction method and thermocycler used.
Discussion
Our study clearly demonstrates that the reliable, accurate and precise measurement of the beef fraction of sausages is feasible independent of the applied PCR-based detection method. As expected, repeatability and reproducibility values are better for samples closely resembling the reference sausages used as calibrators. Of course, it is not realistic to produce reference sausages for every single type of sausage on the market. However, it can be done for those types of products where legal compositional requirements have to be enforced.
Further studies will be needed to validate the application range of a given set of reference sausages. Such studies will help to define a minimal set of reference sausages for the DNA-based quantitative detection of meat fractions. Analytical limitations might be expected outside the range covered by the actual set of reference sausages. But it is also of particular interest to quantify minor amounts of non-declared meat additions in terms of a good manufacturing praxis (GMP) or the meat fraction of improper species in certain ethnic specialities.
We resume that the presented approach is feasible and gives accurate and precise results independent of the applied PCR-methods.
References
Schweizerische Verordnung über Lebensmittel tierischer Herkunft vom 23.November 2005 (SR 817.022.108) Art. 8 Abs. 5
Binke R, Spiegel K, Schwägele F (2005) Vergleichende Untersuchung von mitochondrialen und nukleären Gensequenzen zur Identifizierung von tierischen Bestandteilen in Fleischerzeugnissen mittels PCR. Mitteilungsblatt der Fleischforschung Kulmbach 44(169):201–210
Lopez-Andreo M, Lugo L, Garrido-Pertierra A, Prieto MI, Puyet A (2005) Identification and quantitation of species in complex DNA mixtures by real-time polymerase chain reaction. Anal Biochem 339:73–82
Hird H, Chisholm J, Brown J (2005) The detection of commercial duck species in food using a single probe-multiple species-specific primer real time PCR assay. Eur Food Res Technol 221:559–563
Rodriguez MA, Garcia T, Gonzales I, Asensio L, Hernandez PE, Martin R (2004) Quantiation of mule duck in goose foie gras using TaqMan real-time polymerase chain reaction. J Agric Food Chem 52:1478–1483
Laube I, Spiegelberg A, Butschke A, Zagon J, Schauzu M, Kroh L, Broll H (2003) Methods for the detection of beef and pork in foods using real-time polymerase chain reaction. Int J Food Sci Technol 38:111–118
Calvo JH, Osta R, Zaragoza P (2002) Quantitative PCR detection of pork in raw and heated ground beef and pâté. J Agric Food Chem 50:5265–5267
Meyer R, Candrian U, Lüthy J (1994) Detection of pork in heated meat products by the polymerase chain reaction. J AOAC Int 77(3):617–622
Laube I, Butschke A, Zagon J, Spiegelberg A, Schauzu M, Bögl K-W, Kroh LW, Broll H (2001) Nachweisverfahren für Rindfleisch in Lebensmitteln unter Anwendung der TaqManTM-Technologie. Bundesgesundheitsbl 44:326–330
Brodmann P, Moor D (2003) Sensitive and semi-quantitative TaqManTM real-time polymerase chain reaction systems for the detection of beef (Bos taurus) and the detection of the family Mammalia in food and feed. Meat Sci 65:599–607
Sawyer J, Wood C, Shanahan D, Gout S, McDowell D (2003) Real-time PCR for quantitative meat species testing. Food Control 14:579–583
Swiss Food Manual (SLMB) Chapter 11 Meat and Meat Products (1999); 4.3.2. Beurteilungskriterien
Hubner P, Waiblinger Hu, Pietsch K, Brodmann P (2001) Validation of PCR methods for quantitation of genetically modified plants in food. J AOAC Int 84(6):1855–1864
Acknowledgments
We thank all participating laboratories: Coop-Labor, Service de la consommation du canton Neuchâtel, Chemisches und Veterinäruntersuchungsamt Freiburg, Bundesinstitut für Risikobewertung Berlin, Chemisches und Veterinäruntersuchungsamt Dresden, Thüringer Landesamt für Lebensmittelsicherheit undVerbraucherschutz Jena. Special thank goes to the butchery Hoesli Geb. AG for the production of Sample D and to SQTS for the quality control of reference sausages.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eugster, A., Ruf, J., Rentsch, J. et al. Quantification of beef and pork fraction in sausages by real-time PCR analysis: results of an interlaboratory trial. Eur Food Res Technol 227, 17–20 (2008). https://doi.org/10.1007/s00217-007-0686-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-007-0686-9