Abstract
Introduction and hypothesis
The aim of the study was to develop a Polish version of the Pelvic Organ Prolapse/Urinary Incontinence Sexual Questionnaire (PISQ) to evaluate sexual function in patients with stress urinary incontinence (SUI) and pelvic organ prolapse (POP) versus a control group.
Methods
Before the validation study, a pilot study of the Polish version of the PISQ was performed in 22 women with pelvic floor dysfunctions. The respondents completed the questionnaire at recruitment and 2 weeks later. Test–retest reliability and internal consistency were determined. The validation study was performed in 249 sexually active women (123 with urodynamic SUI and POP; 126 healthy controls). The study group reported urinary incontinence (UI) at the urogynecological ambulatory clinic, where they underwent urogynecological and urodynamic examinations. All participants completed the questionnaire. PISQ results from both groups were compared and correlated with those of the King’s Health Questionnaire (KHQ) and patient age.
Results
Test–retest reliability was good according to Pearson’s correlation coefficient (r = 0.89, p < 0.001). PISQ had high internal consistency (Cronbach’s α = 0.878). The total PISQ score was significantly lower in the study group compared with the controls (83.6 ± 14.9 vs 95.7 ± 10.3, p < 0.001), as were scores for individual domains: Behavioral/Emotive, Physical, Partner-Related (33.9 ± 10.2 vs 39.8 ± 7.8, 31.7 ± 6.9 vs 37.1 ± 2.8, and 18.0 ± 3.1 vs 19.4 ± 2.6 respectively; p < 0.001), confirming the lower quality of sexual function in women with SUI and POP. A correlation between PISQ and KHQ scores and patient age was confirmed.
Conclusions
The Polish version of the PISQ is a reliable tool for evaluating sexual function in women with POP and UI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sexual function in women is influenced by biological, medical, and psychological factors [1]. In Poland, approximately 87 % of women aged 18–49 years are sexually active, whereas over the age of 50 the rate decreases to 35.7 % [2]. Worldwide, 68 % of women over the age of 21 are sexually active, while the rate decreases to 27 % between the ages of 55 and 95 years [3, 4].
Urinary incontinence (UI) or other urogynecological disorders affect 10–44 % of women [5, 6]. Approximately 46 % of patients with UI or symptoms from the lower urinary tract report sexual disorders [7]. Patients with UI experience anxiety and fear of incontinence during intercourse, which significantly lowers their libido and self-esteem, and causes orgasm problems [7]. UI during intercourse affects 10.6–36 % of women with UI [8, 9], but its effect on the quality of sexual life is not straightforward. Over half of these women (59.8 %) regard the impact of UI during intercourse as being low, 32.3 % as moderate, and only 7.9 % as high [10]. Patients with UI during intercourse restrict the frequency of sexual contact [3], and 6 % of women with UI completely abstain from sexual intercourse [10].
The impact of UI and pelvic organ prolapse (POP) on the quality of sexual function remains unequal. POP is perceived to be the main factor influencing sexual function of affected women. However, overall sexual satisfaction after surgical repair of POP and UI therapy remains at a level similar to that before treatment [11], supporting the theory that psychological factors, e.g., relationships with a partner, play a definitive role as far as quality of sexual life is concerned [12, 13].
Quality of life (QoL) can be evaluated with the use of questionnaires that provide measurable data that can be compared. Among the questionnaires available, those that are disease-specific can detect subtle differences in QoL determined by the nature of the disease, and are the most valuable tools [3]. The International Continence Society has divided all questionnaires on female sexual function into three categories: A, highly recommended; B, recommended; C, optional [14].
The Pelvic Organ Prolapse/Urinary Incontinence Sexual Questionnaire (PISQ) belongs to category B and is the only POP-specific investigative tool. PISQ was designed by Rogers et al. in 2001 [15], and has been further developed, with a short form PISQ-12, and a new version—PISQ, IUGA-Revised (PISQ-IR)—that evaluates the effect of pelvic floor disorders (PFDs) together with anal incontinence on sexual inactivity [16, 17].
The goal of our study was to develop and validate a Polish version of the PISQ to evaluate the quality of sexual function in Polish women with UI and POP.
Materials and methods
The PISQ includes 31 items, divided into three domains, labeled Behavioral/Emotive, Physical, and Partner-Related. The answers to the questions pertaining to sexual function are marked on a five-point Likert scale, ranging from 0, never, to 4, always. Only item 5 is evaluated from 0, does not masturbate, to 5, always masturbates. Higher PISQ scores correspond to better sexual function. The maximum total score is 125 [15]. The individual domains include the following questions: Behavioral/Emotive—1, 2, 5, 6, 7, 8, 9, 10, 12, 22, 23, 24, 26, 27; Physical—11, 13, 16, 17, 18, 19, 20, 21, 25, 30; and Partner-Related—3, 4, 14, 15, 28, 31.
The King’s Health Questionnaire (KHQ), a disease-specific questionnaire for the assessment of QoL among patients with UI, was used to investigate criterion-related validity. The questionnaire consists of 21 items, divided into eight domains [18]. These domains are labeled General Health Perception, Incontinence Impact, Role Limitation, Physical Limitations, Social Limitations, Personal Relationships, Emotions, and Sleep and Energy. The scores within the domains range from 0 to 100, with 0 representing the best and 100 the worst health status. KHQ has been shown to have excellent reliability and validity, and sensitivity to change [18, 19].
Validation of the questionnaire
The Polish version of the PISQ questionnaire was developed according to the required criteria [20]. Two independent certified translators translated the questionnaire into Polish. Back translation was performed by a certified translator, who confirmed the integrity of the document. Next, a multidisciplinary team, consisting of urogynecologists, psychologists, and a biologist corrected the text and approved the final version of the questionnaire.
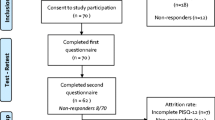
A pilot study of the Polish version of the questionnaire was carried out in a group of 22 women with PFDs attending an outpatient clinic of the Center for Family Medicine at the Medical University of Gdańsk. The patients completed a questionnaire at the first appointment and again after 2 weeks. The results were compared to evaluate test–retest reliability.
Validation of the PISQ was then performed in a group of 249 women, including 123 patients with stress urinary incontinence (SUI) and POP, and 126 healthy controls. All patients were sexually active, gave their informed consent to participate in the study, and completed the QoL questionnaires. All women from the study group reported UI at the urogynecological ambulatory clinic and all were investigated according to the standards of the International Continence Society. Additional questions about UI and sexual function were included in the gynecological and obstetric history. All patients underwent a complete urogynecological examination including: gynecological examination, POP assessment according to the Pelvic Organ Prolapse Quantification System (POP-Q), cough provocation tests in the supine and standing positions, pelvic floor muscle assessment, and introital and transvaginal ultrasonography. Finally, a urodynamic examination was performed using Solar® (Medical Measurement Systems, Enschede, the Netherlands). It included uroflowmetry, filling cystometry, pressure flow study, and profilometry. Urodynamically confirmed SUI and POP (at least stage I in the POP-Q system) were necessary for inclusion in the study group.
The control group included 126 consecutive patients attending the Gynecological and Obstetric Ambulatory Clinic, the Center for Family Medicine, Medical University of Gdańsk, between 2011 and 2012. All controls were patients without UI, with no history of surgery for UI or POP, and whose gynecological examination did not show any PFDs. Patients from both groups completed the PISQ. Additionally, the women from the study group filled out the KHQ, but 4 patients failed to complete it. High scores in the KHQ and low scores in the PISQ represented a poor QoL. The main outcome measures were mean scores of the questionnaires: PISQ in the study and control groups, and KHQ in the study group. Both the total scores and individual domain scores were evaluated. The impact of SUI and POP on the sexual function of the affected women was analyzed and compared with the control sample.
The study was approved by the Local Ethics Committee and all patients gave their informed consent to participate in the study. The privacy of all subjects was maintained. The Declaration of Helsinki was followed.
For information on and licensing of the validated Polish King’s Health Questionnaire (KHQ), contact the MAPI Research Trust, Lyon, France (E-mail: PROinformation@mapi-trust.org; www.mapi-trust.org).
Statistical analysis
Statistical analysis was performed using SPSS for Windows 17.0 (SPSS, Chicago, IL, USA). Continuous variables were expressed as mean ± standard deviation. Categorical variables were expressed as percentages of the total group. A p value <0.05 was considered statistically significant.
Based on the literature, the sample size for the test–retest calculation of PISQ was 20 [16]. The desired correlation coefficient to confirm the reproducibility of the questionnaire results was at least 0.7. For this level of significance the minimum sample size was 9. In the study, the correlation coefficient was 0.89, which was higher than the assumed minimum level of significance. Based on data in the literature of mean PISQ scores in sexually active women with UI and/or POP [15], and assuming 85 % study power and an α error of 0.05, we calculated that each study group should have at least 89 patients. Post-hoc analysis of the results revealed that the power of the study reached 100 %.
The Mann–Whitney U test was used to compare two groups and the Kruskal–Wallis test was used to compare more than two groups. Post-hoc tests were employed for statistically significant results. All statistical tests were two-sided. Spearman’s rho was used to analyze the relationship between variables. Cronbach’s α was used to measure internal consistency. Pearson’s correlation coefficient (r) was used to measure test–retest reliability.
Results
The comparative analysis of demographic data of the study and control populations are presented in Table 1. The women in the study group had higher body mass index and parity, and were more often multiparous compared with the controls. The study group also included fewer employed women and a greater number of old-aged pensioners, disability pensioners, and blue-collar workers. Subjects in the study group were less well-educated than the controls.
In the study group, SUI grade 1 was diagnosed in 46 (37.4 %), grade 2 in 57 (46.3 %), and grade 3 in 20 (16.3 %) patients. POP-Q stage I was diagnosed in 17 (13.8 %), stage II in 98 (79.7 %), and stage III in 8 (6.5 %) patients. No stage IV POP-Q cases were found.
Assessment of internal consistency
The PISQ has good internal consistency (Cronbach’s α = 0.878). However, even the authors of the questionnaire observed lower reliability in the Partner-Related domain, with high overall internal consistency of the test [15] (Table 2).
Assessment of stability
Analysis of the correlation between scores for individual scales was performed to evaluate test–retest reliability using Pearson’s correlation coefficient (r) (Table 3). The analysis revealed a strong positive correlation between consecutive scores on the scale for QoL assessment (Pearson’s correlation coefficient (r) = 0.89, p < 0.001).
Assessment of construct validity
Construct validity examines scale scores based on some known groups or in relation to other measures, and refers to the ability of a measurement tool (e.g., a survey, test) to actually measure the psychological concept being studied [21]. Construct validity was evaluated by comparing the questionnaire results of the SUI and POP study group with controls who had no PFDs. The analysis revealed significantly lower scores in all domains of the study group compared with controls, confirming the lower quality of sexual function of the group with SUI and POP (Table 4).
Assessment of criterion-related validity
Criterion-related validity was evaluated by comparing PISQ results with the KHQ previously validated in Polish. Spearman’s rho was determined to examine the correlation between PISQ questions and KHQ scales (Table 5). The correlation analysis revealed that higher scores in the Behavioral/Emotive domain corresponded with higher general health perceptions. Higher quality of sexual life in the Physical domain signified lower levels of all KHQ scores, which in turn meant higher QoL. Higher scores in the Partner-Related domain corresponded to lower levels of all KHQ scores (better QoL). The results were statistically significant with the exception of the following domains: Incontinence Impact, Physical Limitations, and Severity Measures. A higher level of the overall perception of the quality of sexual function (total PISQ score) represented better QoL on all KHQ scales apart from the Incontinence Impact and Severity Measures domains.
In addition, analysis of the PISQ scores with regard to age was carried out, as age is considered an important factor that negatively affects sexual function [22]. Pearson’s correlation coefficient (r) revealed a statistically significant correlation r = −0.24; p = 0.007 (test strength = 0.86). Thus, the older the respondents, the lower they perceived the quality of their sexual function, which again confirmed the criterion-related validity of the PISQ.
Discussion
Efforts are continuing to design a tool for the adequate analysis of sexual function in women. The original English version of the PISQ is widely used to evaluate the influence of surgical management on sexual function in women with UI and POP [23, 24]. It has also been used in numerous other languages, but a Polish questionnaire has been unavailable until now [25, 26].
This study indicated that the validated Polish version of the PISQ is a good tool for assessing the quality of sexual function in women with UI and POP. The study group had significantly lower scores compared with controls in the total PISQ and in all domains, thus confirming the lower quality of sexual function of women with SUI and POP.
Internal consistency determined whether the items within a domain measured the same aspects of the concept [27]. The PISQ in the study group had high internal consistency, with Cronbach’s α of 0.878. Cronbach’s α calculated by the authors of the questionnaire was 0.85 for the entire questionnaire, and 0.86, 0.77, and 0.43 for the Behavioral/Emotive, Physical, and Partner-Related domains respectively [15]. This was consistent with our study population, where Cronbach’s α for the Partner-Related domain was also lower. Nevertheless, the entire questionnaire has high internal consistency, making it a reliable diagnostic tool.
Test–retest reliability, evaluated using Pearson’s correlation coefficient (r) between subsequent measures for individual domains of the quality of sexual function, revealed a strong, statistically significant, positive correlation with subsequent domain scores.
The PISQ and KHQ results were compared to confirm criterion-related validity. A correlation between the PISQ and KHQ scores was found, and KHQ is a popular and reliable tool for the assessment of QoL in women with UI [18]. It is important to note that KHQ is used to evaluate general health-related QoL, and not only sexual functioning, and questions about sexual activity (bladder problems affecting the relationship with a partner, sex, and family life) are only included in the Personal Relationship domain. Thus, it is important that there was a statistically significant correlation between that particular domain and PISQ scores.
In the PISQ, the Behavior/Emotive domain measures the frequency of sexual activity, the desired frequency, the ability to have an orgasm, physical reactions accompanying sexual activity, and the overall satisfaction with one’s sexual life [13]. In our study, this correlated with the general health score of the KHQ. The Physical domain of the PISQ correlated with all KHQ domains, highlighting the problems associated with UI, POP, and anal incontinence during sexual activity, and the related feelings that lead to restrictions of sexual activity.
The Partner-Related domain evaluated the influence of a partner’s dysfunction on sexual activity, e.g., erectile and ejaculation dysfunction, and active avoidance of sexual activity and intercourse due to the presence of symptoms related to UI and POP [13].
The impact of UI and POP on the quality of sexual function has been confirmed [7, 11]. However, it has been demonstrated that the severity of UI does not always correspond to the quality of sexual life among women with UI and POP [3]. KHQ Severity Measures did not correlate with the general PISQ score, with the exception of the Physical domain. The Severity Measures domain includes items concerning problems related to pad usage, fluid intake restriction, changing underclothes, smell, and feeling of embarrassment resulting from UI; therefore, a correlation with the PISQ Physical domain may be anticipated. However, other domains of the PISQ, such as the Partner-Related and Behavioral/Emotive domains, influence the overall score of the quality of sexual function.
Conclusions
The Polish version of the PISQ is a valid, reliable, and consistent tool for evaluating sexual function in a Polish population of women with POP and UI. Its results correlate with KHQ scores and allow the quality of sexual function in women with SUI and POP to be assessed and compared.
References
Dalpiaz O, Kerschbaumer A, Mitterberger M, Pinggera GM, Colleselli D, Bartsch G, Strasser H (2008) Female sexual dysfunction: a new urogynaecological research field. BJU Int 101(6):717–721
Izdebski Z (2012) Seksualność Polaków na początku XXI wieku—stadium badawcze. Wydawnictwo Uniwersytetu Jagiellońskiego, Kraków [in Polish]
Rogers GR, Villarreal A, Kammerer-Doak D, Qualls C (2001) Sexual function in women with and without urinary incontinence and/or pelvic organ prolapse. Int Urogynecol J Pelvic Floor Dysfunct 12(6):361–365
Tannenbaum C, Corcos J, Assalian P (2006) The relationship between sexual activity and urinary incontinence in older women. J Am Geriatr Soc 54(8):1220–1224
Parazzini F, Colli E, Origgi G, Surace M, Bianchi M, Benzi G, Artibani W (2000) Risk factors for urinary incontinence in women. Eur Urol 37(6):637–643
Hunskaar S, Lose G, Sykes D, Voss S (2004) The prevalence of urinary incontinence in women in four European countries. BJU Int 93(3):324–330
Salonia A, Zanni G, Nappi RE, Briganti A, Dehò F, Fabbri F, Colombo R, Guazzoni G, Di Girolamo V, Rigatti P, Montorsi F (2004) Sexual dysfunction is common in women with lower urinary tract symptoms and urinary incontinence: results of a cross-sectional study. Eur Urol 45(5):642–648, discussion 648
Moran PA, Dwyer PL, Ziccone SP (1999) Urinary leakage during coitus in women. J Obstet Gynaecol 19(3):286–288
Espuña Pons M, Puig Clota M (2008) Coital urinary incontinence: impact on quality of life as measured by the King’s Heath Questionnaire. Int Urogynecol J Pelvic Floor Dysfunct 19(5):621–625
Kizilkaya Beji N, Yalcin O, Ayyildiz EH, Kayir A (2005) Effect of urinary leakage on sexual function during sexual intercourse. Urol Int 74(3):250–255
Ozel B, White T, Urwitz-Lane R, Minaglia S (2006) The impact of pelvic organ prolapse on sexual function in women with urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct 17(1):14–17
Barber MD, Visco AG, Wyman JF, Fantl JA, Bump RC (2002) Continence Program for Women Research Group. Sexual function in women with urinary incontinence and pelvic organ prolapse. Obstet Gynecol 99(2):281–289
Kammerer-Doak D (2009) Assessment of sexual function in women with pelvic floor dysfunction. Int Urogynecol J Pelvic Floor Dysfunct 20 [Suppl 1]:S45–S50
Omotosho TB, Rogers RG (2009) Shortcomings/strengths of specific sexual function questionnaires currently used in urogynecology: a literature review. Int Urogynecol J Pelvic Floor Dysfunct 20 [Suppl 1]:S51–S56
Rogers RG, Kammerer-Doak D, Villarreal A, Coates K, Qualls C (2001) A new instrument to measure sexual function in women with urinary incontinence or pelvic organ prolapse. Am J Obstet Gynecol 184(4):552–558
Rogers RG, Coates KW, Kammerer-Doak D, Khalsa S, Qualls C (2003) A short form of the pelvic organ prolapse/urinary incontinence sexual questionnaire (PISQ-12). Int Urogynecol J Pelvic Floor Dysfunct 14(3):164–168
Rogers RG, Rockwood TH, Constantine ML, Thakar R, Kammerer-Doak DN, Pauls RN, Parekh M, Ridgeway B, Jha S, Pitkin J, Reid F, Sutherland SE, Lukacz ES, Domoney C, Sand P, Davila GW, Espuna Pons ME (2013) A new measure of sexual function in women with pelvic floor disorders (PFD): the Pelvic Organ Prolapse/Incontinence Sexual Questionnaire, IUGA-Revised (PISQ-IR). Int Urogynecol J 24(7):1091–1103
Kelleher CJ, Cardozo LD, Khullar V, Salvatore S (1997) A new questionnaire to assess the quality of life of urinary incontinent women. Br J Obstet Gynaecol 104(12):1374–1379
Yip SK, Chan A, Pang S, Leung P, Tang C, Shek D, Chung T (2003) The impact of urodynamic stress incontinence and detrusor overactivity on marital relationship and sexual function. Am J Obstet Gynecol 188(5):1244–1248
Guillemin F, Bombardier C, Beaton D (1993) Cross-cultural adaptation of health-related quality of life measures: literature review and proposed guidelines. J Clin Epidemiol 46(12):1417–1432, review
Hendriks EJ, Bernards AT, Berghmans BC, de Bie RA (2007) The psychometric properties of the PRAFAB-questionnaire: a brief assessment questionnaire to evaluate severity of urinary incontinence in women. Neurourol Urodyn 26(7):998–1007
Weber AM, Walters MD, Schover LR, Mitchinson A (1995) Sexual function in women with uterovaginal prolapse and urinary incontinence. Obstet Gynecol 85(4):483–487
Rogers RG, Kammerer-Doak D, Darrow A, Murray K, Qualls C, Olsen A, Barber M (2006) Does sexual function change after surgery for stress urinary incontinence and/or pelvic organ prolapse? A multicenter prospective study. Am J Obstet Gynecol 195(5):e1–e4
Komesu YM, Rogers RG, Kammerer-Doak DN, Barber MD, Olsen AL (2007) Posterior repair and sexual function. Am J Obstet Gynecol 197(1):101.e1–101.e6
Su TH, Lau HH (2010) Validation of a Chinese version of the short form of the pelvic organ prolapse/urinary incontinence sexual questionnaire. J Sex Med 7(12):3940–3945
Cam C, Sancak P, Karahan N, Sancak A, Celik C, Karateke A (2009) Validation of the short form of the pelvic organ prolapse/urinary incontinence sexual questionnaire (PISQ-12) in a Turkish population. Eur J Obstet Gynecol Reprod Biol 146(1):104–107
Fitzpatrick R, Davey C, Buxton MJ, Jones DR (1998) Evaluating patient-based outcome measures for use in clinical trials. Health Technol Assess 2(14):i–iv, 1–74
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Funding
None.
Financial disclaimer
None.
Rights and permissions
About this article
Cite this article
Grzybowska, M.E., Wydra, D.G. & Smutek, J. Validation of the Polish version of the Pelvic Organ Prolapse/Urinary Incontinence Sexual Questionnaire. Int Urogynecol J 27, 781–786 (2016). https://doi.org/10.1007/s00192-015-2883-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-015-2883-6