Abstract
Purpose
Choice of renal replacement therapy (RRT) modality may affect renal recovery after acute kidney injury (AKI). We sought to compare the rate of dialysis dependence among severe AKI survivors according to the choice of initial renal replacement therapy (RRT) modality applied [continuous (CRRT) or intermittent (IRRT)].
Methods
Systematic searches of peer-reviewed publications in MEDLINE and EMBASE were performed (last update July 2012). All studies published after 2000 reporting dialysis dependence among survivors from severe AKI requiring RRT were included. Data on follow-up duration, sex, age, chronic kidney disease, illness severity score, vasopressors, and mechanical ventilation were extracted when available. Results were pooled using a random-effects model.
Results
We identified 23 studies: seven randomized controlled trials (RCTs) and 16 observational studies involving 472 and 3,499 survivors, respectively. Pooled analyses of RCTs showed no difference in the rate of dialysis dependence among survivors (relative risk, RR 1.15 [95 % confidence interval (CI) 0.78–1.68], I 2 = 0 %). However, pooled analyses of observational studies suggested a higher rate of dialysis dependence among survivors who initially received IRRT as compared with CRRT (RR 1.99 [95 % CI 1.53–2.59], I 2 = 42 %). These findings were consistent with adjusted analyses (performed in 7/16 studies), which found a higher rate of dialysis dependence in IRRT-treated patients [odds ratio (OR) 2.2–25 (5 studies)] or no difference (2 studies).
Conclusions
Among AKI survivors, initial treatment with IRRT might be associated with higher rates of dialysis dependence than CRRT. However, this finding largely relies on data from observational trials, potentially subject to allocation bias, hence further high-quality studies are necessary.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Acute kidney injury (AKI) is common in critically ill patients and associated with high mortality and morbidity [1]. When AKI is severe, renal replacement therapy (RRT) is often required while disease-specific treatments are applied. RRT is typically provided in two modalities: continuous (CRRT) or intermittent (IRRT). Both modalities achieve a satisfactory degree of metabolic control, and to date, despite numerous observational studies, randomized controlled trials (RCTs) [2–9], and meta-analyses [2, 10–12], neither modality has been found superior in terms of mortality. In contrast, only few studies have specifically focused on the effects of CRRT and IRRT on renal recovery and dialysis dependence among survivors. This question, however, is important because chronic hemodialysis is a major burden for patients, their families, and healthcare systems, and is associated with higher long-term mortality [13–16].
A Cochrane systematic review [10] sought to compare IRRT with CRRT in many aspects including the rate of dialysis dependence. However, only three small, randomized controlled studies [8, 9, 17] were included in this part of the review, and the multiple observational studies reporting renal recovery after RRT were not included.
Accordingly, we sought to systematically review the current literature and to analyze all data on dialysis dependence among critically ill patients who survived an episode of AKI requiring acute RRT. We used intention-to-treat analysis to test the hypothesis that patients assigned to initially receive IRRT might have higher rates of dialysis dependence compared with those assigned to initially receive CRRT.
Methods
We performed this systematic review using the guidelines proposed by the Cochrane Collaboration in the Cochrane Handbook for Systematic Reviews of Interventions (http://www.cochrane-handbook.org).
Study selection criteria
Participants
This review focuses on survivors of critical illness who received RRT for AKI.
Interventions
For the purpose of the review, we use the term “IRRT” to describe intermittent hemodialysis, intermittent hemofiltration, and slow low-efficiency dialysis (SLED). As SLED is substantially different from other intermittent techniques, sensitivity analyses were performed excluding studies reporting data on such modality.
We use the term “CRRT” to describe continuous hemofiltration and/or continuous hemodialysis and/or continuous hemodiafiltration, all intended to run on a continuous basis (24 h/day).
For patients who received both modalities (crossover), we classified patients according to the initial modality administered whenever such data were available (intention-to-treat principle).
Comparators
We compared outcomes according to the initial RRT modality applied on an intention-to-treat basis.
Types of outcome measures
The primary outcome was dialysis dependence among survivors. We assessed dialysis dependence as the need for any form of RRT at the end of the follow-up period.
Types of studies
We included all RCTs and observational studies in English language reporting data on dialysis dependence after RRT for AKI between 2000 and 2012. We excluded reviews, commentaries, and editorials.
Search methods for identification of studies
Study selection
We searched MEDLINE and EMBASE via the OvidSP portal. The keywords/MESH headings used are presented in the ESM Appendix. Two independent investigators (A.G.S. and N.J.G.) carried out the initial search and subsequent study selection. After title screening, we evaluated abstracts for relevance and identified as included, excluded or requiring further assessment. At this stage, if a paper required further assessment, we contacted the study lead investigator by e-mail and/or telephone with a request for further information. We then reviewed the bibliography of selected publications. We corresponded with the authors when missing data were identified. We updated the search in July 2012. All studies that reported data on dialysis dependence after RRT for AKI were included.
For the purpose of meta-analysis we included all studies where simultaneous data on IRRT and CRRT treatment were obtained. Studies in which all patients received a single modality (IRRT or CRRT) or RCTs not comparing IRRT with CRRT were analyzed and presented separately as sensitivity analyses.
Data extraction
Data extraction was performed by A.G.S. and confirmed independently by N.J.G. For each study, we recorded the year of publication, the type of study (RCT or observational), and the number of centers involved. We obtained the total number of RRT patients included in each study, and determined how many survived the acute illness and how many were dialysis dependent at the end of the study follow-up. In addition, we collected the following variables when available: duration of follow-up, sex, age, chronic kidney disease (CKD), illness severity score [Acute Physiology and Chronic Health Evaluation (APACHE) II, APACHE III or Simplified Acute Physiology Score (SAPS) II], and use of vasopressors and mechanical ventilation when available. We obtained all results for the whole cohorts and recorded them separately according to RRT modality.
Synthesis of results/statistical analysis
Assessment of risk of bias
We examined RCTs for adequate allocation concealment, randomization process, and balance of baseline characteristics. We assessed study methodology using the Jadad scale [18]. As blinding is virtually impossible when comparing RRT modalities, a score of 3 was considered satisfactory.
For observational trials, we recorded the rule for allocation to either RRT modality to assess allocation bias. Similarly, we extracted data on sex, age, CKD, illness severity score, vasopressors, and mechanical ventilation where available, as all these variables are susceptible to confound the association between choice of RRT modality and dialysis dependence. We recorded the presence of adjusted analyses for dialysis dependence as well as their results. Finally, we assessed selective reporting according to the rate of loss to follow-up.
Data synthesis
We analyzed data using Review Manager version 5.1.4 (The Cochrane Collaboration, Oxford, UK) and Stata release 12.0 (StataCorp, College Station, TX). Due to expected heterogeneity between study protocols, populations, and interventions, we decided a priori to combine results using a random-effects model for all analyses [19].
For dichotomous outcomes, we used relative risk (RR) with 95 % confidence interval (CI) to pool the results.
To enable study comparison, we transformed illness severity scores (SAPS II and APACHE III) into the equivalent APACHE II score, using previously described methodology [20].
We quantified statistical heterogeneity for pooled results using the chi-square and I 2 statistics. We estimated publication bias with a funnel plot.
Stratification
We stratified pooled analyses according to study design (RCT versus observational). We further stratified observational studies according to duration of follow-up, inclusion or exclusion of patients with CKD, and number of centers for the purpose of sensitivity analyses.
We considered RCTs not designed to compare IRRT with CRRT as equivalent to observational studies.
In addition, we separately analyzed studies where RRT was limited to a single modality (only IRRT or CRRT) as direct comparison was not possible. For such comparison, we calculated a pooled OR with 95 % CI (details of calculation presented in ESM Appendix).
Results
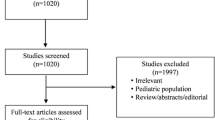
The study selection process is presented in Fig. 1. We identified 383 eligible studies for abstract review. Of these, 146 were selected for full-text search. Finally, 50 studies presented data on dialysis dependence after RRT and were included in this systematic review. Of those, 23 presented outcome data for both modalities (IRRT and CRRT) and were included in the meta-analysis; 7 were randomized controlled trials [8, 9, 17, 21–24] and 16 were observational studies [25–41], including a total of 3,971 patients who survived an episode of AKI requiring RRT [2,255 (CRRT) and 1,716 (IRRT)].
In the other 27 studies (2,536 survivors), a single initial RRT modality was applied to all patients. This modality was IRRT in 11 of these studies (644 survivors) [42–52] and CRRT in 16 (1,892 survivors) [53–68].
Study description, patient demographics, and risk of bias evaluation
Randomized controlled trials
The seven RCTs included in this review are presented in the ESM Appendix. Altogether, these studies report dialysis dependence data for a total of 472 AKI survivors (1,160 patients enrolled). Of those, 240 received IRRT as an initial modality and 232 CRRT. Four of these were single-center studies, and three were multicenter studies.
Although all studies compared IRRT with CRRT, significant heterogeneity between designs was present. In particular, the IRRT arm consisted of slow low-efficiency dialysis (SLED) for two studies [21, 22] as opposed to intermittent hemodialysis for the other five. Hemodynamically unstable patients were excluded in one study [17], while only those with multiple organ dysfunction syndrome were included in another [24]. In addition, imbalances in baseline characteristics between the two groups were present in 3/7 studies, and crossover from allocated modality occurred in 5/7 studies (involving more than 15 % of the patients in 3 of these studies). Studies were all powered to demonstrate a difference in mortality but not in renal recovery to dialysis independence. Finally, four of the studies were graded as “poor quality” (Jadad score 1–2) and three as “satisfactory” (Jadad score 3).
Observational studies
The 16 observational studies included in this review reported data on dialysis dependence in 3,505 AKI survivors (7,158 patients enrolled). Of these, 1,481 received IRRT as an initial modality and 2,024 CRRT. Their baseline characteristics are presented in Table 1.
As presented in the ESM Appendix, modality allocation was likely to be biased in most (14/16) studies as the reasons for choice of RRT modality were not described (13 studies) or CRRT was preferentially applied to patients on inotropic or vasopressor drug support. This risk was considered low in two studies where a before–after study design was applied [40, 41].
When specific baseline characteristics were reported according to RRT modality, IRRT patients had lower illness severity scores in 6/8 studies. They required vasopressors (pooled percentage from six studies: 40.1 % for IRRT versus 81.9 % for CRRT, p < 0.0001) or mechanical ventilation less frequently (pooled percentage from five studies 55.8 % for IRRT versus 85.2 % for CRRT, p < 0.0001). Finally, the pooled percentage of patients with CKD was lower among IRRT patients (7.9 % for IRRT versus 10.5 %, p = 0.04). Adjusted analyses taking these confounders into account were performed in seven studies.
Additional studies providing no direct comparison
An additional 27 studies that did not provide direct comparison between IRRT and CRRT were analyzed. Of those, 11 studies reported dialysis dependence data for 644 AKI survivors initially treated with IRRT and 16 in 1,892 survivors initially treated with CRRT.
Patient characteristics per RRT modality are presented in the ESM Appendix. On pooled average, IRRT survivors were younger (57.8 versus 63.5 years old) and had lower APACHE II score (26.8 versus 28.7), and a smaller percentage had pre-existing CKD (5.8 versus 19.4 %) or required mechanical ventilation (77.6 versus 78.9 %). However, a larger percentage of IRRT patients required vasopressors (74 versus 67.6 %).
Finally, the duration of follow-up was shorter in “IRRT studies” [28 days (in 5/11) or until hospital discharge (in 5/11)] as compared with “CRRT studies” [90 days (in 5/16) or until hospital discharge (in 9/19)].
Renal recovery according to dialysis modality
Overall
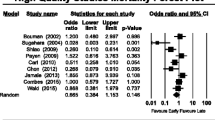
When all studies comparing CRRT with IRRT were pooled (Fig. 2), IRRT was associated with a higher risk for dialysis dependence compared with CRRT (RR 1.73 [1.35–2.20]). There was evidence for moderate heterogeneity (chi square p = 0.02 and I 2 = 44 %).
Randomized controlled trials
Within RCTs (Fig. 2), there was no statistically significant difference in the risk of hemodialysis (HD) dependence between IRRT and CRRT (RR 1.15 [95 % CI 0.78–1.68]). There was no evidence for heterogeneity (chi square p = 0.78, I 2 = 0 %). Similar results were obtained when the two “SLED” studies were excluded from analysis (RR 1.18 [0.79–1.75], I 2 = 0 %) (ESM Appendix). When only studies of “satisfactory” quality according to the Jadad scale were included the RR was 1.48 [0.82–2.66] (I 2 = 0 %).
Observational studies
Within observational studies (Fig. 2), IRRT was associated with a 1.99 relative risk of dialysis dependence compared with CRRT (95 % CI 1.53–2.59). There was evidence for moderate heterogeneity (chi square p = 0.04 and I 2 = 42 %).
This association remained when studies were pooled according to exclusion or inclusion of patients with pre-existing CKD (Fig. 3), follow-up duration (hospital discharge or 90 days), and number of centers involved in the study (additional figures in ESM Appendix).
When adjusted analyses were performed (7/16), the odds ratios for a higher rate of dialysis dependence in IRRT patients ranged from 2.2 to 25 (5 studies) or no difference was found (2 studies).
Additional studies providing no direct comparison
When all dialysis dependence data from studies providing no direct comparison were pooled, IRRT was associated with a higher OR for dialysis dependence (OR 2.30 [95 CI % 1.79–2.96]).
Discussion
Key findings
We performed a systematic review of the literature and identified 50 original studies reporting data on the rate of dialysis dependence among more than 6500 survivors who received RRT for AKI. We found that patients who received IRRT as an initial RRT modality for AKI had a 1.7 times increased risk of remaining dialysis dependent as compared with those who initially received CRRT.
This finding was consistent across subgroups but did not reach statistical significance amongst RCTs. These RCTs, however, were relatively small and of only moderate quality, and did not all include hemodynamically unstable patients. Allocation bias was present in observational trials, with IRRT appearing to be preferentially allocated to patients with lesser illness severity and some degree of chronic kidney disease. Similar findings were present when studies reporting outcomes of a single modality were analyzed.
Comparison with previous studies
To date, observational studies, RCTs [2–9], and meta-analyses [2, 10–12] have failed to demonstrate any survival advantage for IRRT or CRRT in AKI.
Two meta-analyses [11, 69] included renal recovery as an outcome and did not find a difference between IRRT and CRRT. Both of these studies restricted their analyses to RCTs, with similar results to those in the RCT section of this study. However, the limited number of patients and the poor quality of these studies limit the precision of the estimate and the robustness of the findings. Moreover, such a comparison of only 240 versus 232 RCT patients with a rate of dialysis dependence of 15.8 % in the IRRT group would only have a 51 % power to detect even a one-third decrease in relative risk. The present review includes data from observational studies. Such studies, although subject to bias, involve a large number of patients and might be more likely to accurately represent the natural history of an episode of severe AKI.
The association between IRRT and increased dialysis dependence is physiologically plausible. Several animal models [70–72] have shown that renal blood flow autoregulation is lost in AKI. Therefore, any hypotension is likely to decrease renal blood flow and compromise glomerular filtration rate (GFR). Indeed, hemodynamic changes induced by IRRT [73–75] are clinically important [12, 76–81]. Moreover, renal biopsies taken in patients receiving IRRT reveal areas of tubular necrosis consistent with fresh tubular damage [82]. No such concerns have been reported in relation to CRRT [83–86].
Clinical implications and future studies
Trials in critically ill patients with AKI have targeted mortality as the primary outcome [55, 73]. However, for survivors, limiting disabilities and maximizing quality of life are of major importance [87, 88]. Dialysis dependence negatively impacts quality of life [89] and is financially burdensome [13–15]. Thus, future studies or comparative trials of RRT modality should focus on dialysis dependence as a major outcome of interest.
Strengths and limitations
To the best of our knowledge, this study is the first to systematically assess the effect of RRT modality on dialysis dependence among patients who survived an episode of AKI requiring RRT. It included data from more than 6,500 patients, 50 studies, and 31 countries, from both large observational studies and randomized controlled studies, and all types of adult critically ill who survived an episode of AKI requiring RRT.
However, this study has several important limitations. First, as we report an association, no inferences of causality can be made. Second, this association is largely dependent upon observational studies and might have been affected by allocation bias. However, factors susceptible to confound the association that were recorded do not support this assertion. In particular, when direct comparative data were available, patients allocated to IRRT had lower levels of illness severity and required mechanical ventilation and vasopressors less frequently. Of even greater relevance, our findings were consistent between studies that did or did not exclude patients with pre-existing CKD, an important risk factor for nonrecovery. This finding makes the possible impact of CKD on nonrecovery among IRRT patients an implausible explanation for our observations. Finally, when adjusted analyses were performed, IRRT was found to be associated with a greater risk of dialysis dependence in all but two studies.
Third, we focused on AKI survivors because dialysis dependence at time of death is rarely reported. We therefore can only report on conditional, not absolute, dialysis dependence. However, the benefit of recovery to dialysis dependence followed by death within 90 days of treatment initiation is low.
Fourth, CRRT may increase the risk of death. Thus, those patients who might have remained dialysis dependent, had they survived, simply died and were therefore not counted. However, there is no convincing evidence in the literature to suggest an association between the choice of RRT modality and mortality after correction for confounders such as illness severity, and need for vasopressors and mechanical ventilation [2, 10–12].
We used the intention-to-treat principle. However, in most studies, patients crossed between modalities or often such data were not reported. Thus, we cannot study the possibility of a dose effect on nonrecovery. However, the fact that many patients were exposed to IRRT only for a part of their overall RRT time implies that our intention-to-treat analysis would logically underestimate the nonrecovery risk of IRRT.
Finally, studies utilizing SLED as an RRT modality have been considered as IRRT. However, as SLED is a hybrid technology combining properties from both IRRT and CRRT, we have presented results including and excluding such studies. These emerging technologies might have a role in future clinical practice, but further studies are required.
Conclusions
Currently available randomized controlled trials do not allow a definitive conclusion on whether choice of initial RRT modality is associated with greater renal recovery rates. Analysis of observational trials suggests that initial support with IRRT might be associated with a higher rate of RRT dependence amongst survivors who received RRT for AKI. As these studies might be associated with allocation bias and given the human and public health implications of these findings, large studies focusing on renal recovery after AKI according to choice of RRT are needed to fully understand the effects of initial modality choice on subsequent dialysis dependence.
References
Uchino S, Kellum JA, Bellomo R, Doig GS, Morimatsu H, Morgera S, Schetz M, Tan I, Bouman C, Macedo E, Gibney N, Tolwani A, Ronco C (2005) Acute renal failure in critically ill patients: a multinational, multicenter study. JAMA 294:813–818
Kellum JA, Angus DC, Johnson JP, Leblanc M, Griffin M, Ramakrishnan N, Linde-Zwirble WT (2002) Continuous versus intermittent renal replacement therapy: a meta-analysis. Intensive Care Med 28:29–37
Kierdorf H (1991) Continuous versus intermittent treatment: clinical results in acute renal failure. Contrib Nephrol 93:1–12
Bosworth C, Paganini EP, Cosentino F, Heyka RJ (1991) Long-term experience with continuous renal replacement therapy in intensive-care unit acute renal failure. Contrib Nephrol 93:13–16
Kruczynski K, Irvine-Bird K, Toffelmire EB, Morton AR (1993) A comparison of continuous arteriovenous hemofiltration and intermittent hemodialysis in acute renal failure patients in the intensive care unit. ASAIO J 39:M778–M781
Rialp G, Roglan A, Betbese AJ, Perez-Marquez M, Ballus J, Lopez-Velarde G, Santos JA, Bak E, Net A (1996) Prognostic indexes and mortality in critically ill patients with acute renal failure treated with different dialytic techniques. Ren Fail 18:667–675
Swartz RD, Messana JM, Orzol S, Port FK (1999) Comparing continuous hemofiltration with hemodialysis in patients with severe acute renal failure. Am J Kidney Dis 34:424–432
Uehlinger DE, Jakob SM, Ferrari P, Eichelberger M, Huynh-Do U, Marti HP, Mohaupt MG, Vogt B, Rothen HU, Regli B, Takala J, Frey FJ (2005) Comparison of continuous and intermittent renal replacement therapy for acute renal failure. Nephrol Dial Transpl 20:1630–1637
Augustine JJ, Sandy D, Seifert TH, Paganini EP (2004) A randomized controlled trial comparing intermittent with continuous dialysis in patients with ARF. Am J Kidney Dis 44:1000–1007
Rabindranath K, Adams J, Macleod AM, Muirhead N (2007) Intermittent versus continuous renal replacement therapy for acute renal failure in adults. Cochrane Database Syst Rev: CD003773
Pannu N, Klarenbach S, Wiebe N, Manns B, Tonelli M (2008) Renal replacement therapy in patients with acute renal failure: a systematic review. JAMA 299:793–805
Bagshaw SM, Berthiaume LR, Delaney A, Bellomo R (2008) Continuous versus intermittent renal replacement therapy for critically ill patients with acute kidney injury: a meta-analysis. Crit Care Med 36:610–617
Gopal I, Bhonagiri S, Ronco C, Bellomo R (1997) Out of hospital outcome and quality of life in survivors of combined acute multiple organ and renal failure treated with continuous venovenous hemofiltration/hemodiafiltration. Intensive Care Med 23:766–772
Go AS, Parikh CR, Ikizler TA, Coca S, Siew ED, Chinchilli VM, Hsu CY, Garg AX, Zappitelli M, Liu KD, Reeves WB, Ghahramani N, Devarajan P, Faulkner GB, Tan TC, Kimmel PL, Eggers P, Stokes JB (2010) The assessment, serial evaluation, and subsequent sequelae of acute kidney injury (ASSESS-AKI) study: design and methods. BMC Nephrol 11:22
Parikh CR, Coca SG, Smith GL, Vaccarino V, Krumholz HM (2006) Impact of chronic kidney disease on health-related quality-of-life improvement after coronary artery bypass surgery. Arch Intern Med 166:2014–2019
Parikh A, Shaw A (2012) The economics of renal failure and kidney disease in critically ill patients. Crit Care Clin 28: 99–111, vii
Mehta RL, McDonald B, Gabbai FB, Pahl M, Pascual MT, Farkas A, Kaplan RM, Collaborative Group for Treatment of ARFitICU (2001) A randomized clinical trial of continuous versus intermittent dialysis for acute renal failure. Kidney Int 60:1154–1163
Jadad AR, Murray WE (2007) Randomized controlled trials: questions, answers and musings. Blackwell, Oxford
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Schneider AG, Lipcsey M, Bailey M, Pilcher DV, Bellomo R (2012) Relationship between illness severity scores in acute kidney injury. Crit Care Resusc 14:53–55
Abe M, Okada K, Suzuki M, Nagura C, Ishihara Y, Fujii Y, Ikeda K, Kaizu K, Matsumoto K (2010) Comparison of sustained hemodiafiltration with continuous venovenous hemodiafiltration for the treatment of critically ill patients with acute kidney injury. Artif Organs 34:331–338
Kumar VA, Yeun JY, Depner TA, Don BR (2004) Extended daily dialysis vs. continuous hemodialysis for ICU patients with acute renal failure: a two-year single center report. Int J Artif Organs 27:371–379
Lins RL, Elseviers MM, Van der Niepen P, Hoste E, Malbrain ML, Damas P, Devriendt J (2009) Intermittent versus continuous renal replacement therapy for acute kidney injury patients admitted to the intensive care unit: results of a randomized clinical trial. Nephrol Dial Transpl 24:512–518
Vinsonneau C, Camus C, Combes A, Costa de Beauregard MA, Klouche K, Boulain T, Pallot J-L, Chiche J-D, Taupin P, Landais P, Dhainaut J-F, Hemodiafe Study Group (2006) Continuous venovenous haemodiafiltration versus intermittent haemodialysis for acute renal failure in patients with multiple-organ dysfunction syndrome: a multicentre randomised trial. Lancet 368:379–385
Andrikos E, Tseke P, Balafa O, Cruz DN, Tsinta A, Androulaki M, Pappas M, Ronco C (2009) Epidemiology of acute renal failure in ICUs: a multi-center prospective study. Blood Purif 28:239–244
Bagshaw SM, Mortis G, Godinez-Luna T, Doig CJ, Laupland KB (2006) Renal recovery after severe acute renal failure. Int J Artif Organs 29:1023–1030
Bell M, Granath F, Schon S, Ekbom A, Martling CR (2007) Continuous renal replacement therapy is associated with less chronic renal failure than intermittent haemodialysis after acute renal failure. Intensive Care Med 33:773–780
Cartin-Ceba R, Haugen EN, Iscimen R, Trillo-Alvarez C, Juncos L, Gajic O (2009) Evaluation of “loss” and “end stage renal disease” after acute kidney injury defined by the risk, injury, failure, loss and ESRD classification in critically ill patients. Intensive Care Med 35:2087–2095
Chang JW, Yang WS, Seo JW, Lee JS, Lee SK, Park S-K (2004) Continuous venovenous hemodiafiltration versus hemodialysis as renal replacement therapy in patients with acute renal failure in the intensive care unit. Scand J Urol Nephrol 38:417–421
Elseviers MM, Lins RL, Van der Niepen P, Hoste E, Malbrain ML, Damas P, Devriendt J, SHARF Investigators (2010) Renal replacement therapy is an independent risk factor for mortality in critically ill patients with acute kidney injury. Crit Care 14:R221
Garcia-Fernandez N, Perez-Valdivieso JR, Bes-Rastrollo M, Vives M, Lavilla J, Herreros J, Monedero P, GEDRCC (2011) Timing of renal replacement therapy after cardiac surgery: a retrospective multicenter Spanish cohort study. Blood Purif 32:104–111
Gonwa TA, Mai ML, Melton LB, Hays SR, Goldstein RM, Levy MF, Klintmalm GB (2001) Renal replacement therapy and orthotopic liver transplantation: the role of continuous veno-venous hemodialysis. Transplantation 71:1424–1428
Jacka MJ, Ivancinova X, Gibney RTN (2005) Continuous renal replacement therapy improves renal recovery from acute renal failure. Can J Anaesth 52:327–332
Lin Y-F, Ko W-J, Chu T-S, Chen Y-S, Wu V-C, Chen Y-M, Wu M-S, Chen Y-W, Tsai C-W, Shiao C-C, Li W-Y, Hu F-C, Tsai P-R, Tsai T-J, Wu K-D, NSARF Study Group (2009) The 90-day mortality and the subsequent renal recovery in critically ill surgical patients requiring acute renal replacement therapy. Am J Surg 198:325–332
Lins RL, Elseviers MM, Daelemans R (2006) Severity scoring and mortality 1 year after acute renal failure. Nephrol Dial Transpl 21:1066–1068
Marshall MR, Creamer JM, Foster M, Ma TM, Mann SL, Fiaccadori E, Maggiore U, Richards B, Wilson VL, Williams AB, Rankin APN (2011) Mortality rate comparison after switching from continuous to prolonged intermittent renal replacement for acute kidney injury in three intensive care units from different countries. Nephrol Dial Transpl 26:2169–2175
Park J, Gage BF, Vijayan A (2005) Use of EPO in critically ill patients with acute renal failure requiring renal replacement therapy. Am J Kidney Dis 46:791–798
Swartz RD, Bustami RT, Daley JM, Gillespie BW, Port FK (2005) Estimating the impact of renal replacement therapy choice on outcome in severe acute renal failure. Clin Nephrol 63:335–345
Uchino S, Bellomo R, Kellum JA, Morimatsu H, Morgera S, Schetz M, Tan I, Bouman C, Macedo E, Gibney N, Tolwani A, Oudemans-Van Straaten H, Ronco C (2007) Patient and kidney survival by dialysis modality in critically ill patients with acute kidney injury. Int J Artif Organs 30:281–292
Waldrop J, Ciraulo DL, Milner TP, Gregori ID, Kendrick AS, Richart CM, Maxwell RA, Barker DE (2005) A comparison of continuous renal replacement therapy to intermittent dialysis in the management of renal insufficiency in the acutely ill surgical patient. Am Surg 71:36–39
Khanal N, Marshall MR, Ma TM, Pridmore PJ, Williams AB, Rankin APN (2012) Comparison of outcomes by modality for critically ill patients requiring renal replacement therapy: a single-centre cohort study adjusting for time-varying illness severity and modality exposure. Anaesth Intensive Care 40:260–268
Albright RC, Smelser JM, McCarthy JT, Homburger HA, Bergstralh EJ, Larson TS (2000) Patient survival and renal recovery in acute renal failure: randomized comparison of cellulose acetate and polysulfone membrane dialyzers. Mayo Clin Proc 75:1141–1147
Bahar I, Akgul A, Ozatik MA, Vural KM, Demirbag AE, Boran M, Tasdemir O (2005) Acute renal failure following open heart surgery: risk factors and prognosis. Perfusion 20:317–322
Faulhaber-Walter R, Hafer C, Jahr N, Vahlbruch J, Hoy L, Haller H, Fliser D, Kielstein JT (2009) The Hannover Dialysis Outcome study: comparison of standard versus intensified extended dialysis for treatment of patients with acute kidney injury in the intensive care unit. Nephrol Dial Transpl 24:2179–2186
Gabriel DP, Caramori JT, Martim LC, Barretti P, Balbi AL (2008) High volume peritoneal dialysis vs daily hemodialysis: a randomized, controlled trial in patients with acute kidney injury. Kidney Int 73(Suppl 108):S87–S93
Kumpers P, Hafer C, David S, Hecker H, Lukasz A, Fliser D, Haller H, Kielstein JT, Faulhaber-Walter R (2010) Angiopoietin-2 in patients requiring renal replacement therapy in the ICU: relation to acute kidney injury, multiple organ dysfunction syndrome and outcome. Intensive Care Med 36:462–470
Holt BG, White JJ, Kuthiala A, Fall P, Szerlip HM (2008) Sustained low-efficiency daily dialysis with hemofiltration for acute kidney injury in the presence of sepsis. Clin Nephrol 69:40–46
Franzen D, Rupprecht C, Hauri D, Bleisch JA, Staubli M, Puhan MA (2010) Predicting outcomes in critically ill patients with acute kidney injury undergoing intermittent hemodialysis—a retrospective cohort analysis. Int J Artif Organs 33:15–21
Iyem H, Tavli M, Akcicek F, Buket S (2009) Importance of early dialysis for acute renal failure after an open-heart surgery. Hemodial Int 13:55–61
Ponikvar JB, Rus RR, Kenda RB, Bren AF, Ponikvar RR (2001) Low-flux versus high-flux synthetic dialysis membrane in acute renal failure: prospective randomized study. Artif Organs 25:946–950
Schiffl H, Lang SM, Fischer R (2002) Daily hemodialysis and the outcome of acute renal failure. N Engl J Med 346:305–310
Al-Malki H, Sadek M, Rashed A, Asim M, Fituri O, Abbass M (2009) Acute renal failure in the State of Qatar: presentation and outcome. Transpl Proc 41:1530–1532
Boussekey N, Chiche A, Faure K, Devos P, Guery B, D’Escrivan T, Georges H, Leroy O (2008) A pilot randomized study comparing high and low volume hemofiltration on vasopressor use in septic shock. Intensive Care Med 34:1646–1653
Beitland S, Moen H, Os I (2010) Acute kidney injury with renal replacement therapy in trauma patients. Acta Anaesthesiol Scand 54:833–840
Bellomo R, Cass A, Cole L, Finfer S, Gallagher M, Lo S, McArthur C, McGuinness S, Myburgh J, Norton R, Scheinkestel C, Su S (2009) Intensity of continuous renal-replacement therapy in critically ill patients. N Engl J Med 361:1627–1638
Ng KP, Chanouzas D, Fallouh B, Baharani J (2012) Short and long-term outcome of patients with severe acute kidney injury requiring renal replacement therapy. QJM 105:33–39
Saudan P, Niederberger M, De Seigneux S, Romand J, Pugin J, Perneger T, Martin PY (2006) Adding a dialysis dose to continuous hemofiltration increases survival in patients with acute renal failure. Kidney Int 70:1312–1317
Van Der Voort PHJ, Boerma EC, Koopmans M, Zandberg M, De Ruiter J, Gerritsen RT, Egbers PHM, Kingma WP, Kuiper MA (2009) Furosemide does not improve renal recovery after hemofiltration for acute renal failure in critically ill patients: a double blind randomized controlled trial. Crit Care Med 37:533–538
Bouman CS, Oudemans-Van Straaten HM, Tijssen JG, Zandstra DF, Kesecioglu J (2002) Effects of early high-volume continuous venovenous hemofiltration on survival and recovery of renal function in intensive care patients with acute renal failure: a prospective, randomized trial. Crit Care Med 30:2205–2211
Chung KK, Juncos LA, Wolf SE, Mann EE, Renz EM, White CE, Barillo DJ, Clark RA, Jones JA, Edgecombe HP, Park MS, Albrecht MC, Cancio LC, Wade CE, Holcomb JB (2008) Continuous renal replacement therapy improves survival in severely burned military casualties with acute kidney injury. J Trauma Inj Infect Crit Care 64:S179–S185 (discussion S185–177)
Kowalik MM, Lango R, Klajbor K, Musial-Swiatkiewicz V, Kolaczkowska M, Pawlaczyk R, Rogowski J (2011) Incidence- and mortality-related risk factors of acute kidney injury requiring hemofiltration treatment in patients undergoing cardiac surgery: a single-center 6-year experience. J Cardiothorac Vasc Anesth 25:619–624
Lines SW, Cherukuri A, Murdoch SD, Bellamy MC, Lewington AJP (2011) The outcomes of critically ill patients with acute kidney injury receiving renal replacement therapy. Int J Artif Organs 34:2–9
Luckraz H, Gravenor MB, George R, Taylor S, Williams A, Ashraf S, Argano V, Youhana A (2005) Long and short-term outcomes in patients requiring continuous renal replacement therapy post cardiopulmonary bypass. Eur J Cardiothorac Surg 27:906–909
Oudemans-van Straaten HM, Bosman RJ, Koopmans M, van der Voort PH, Wester JP, van der Spoel JI, Dijksman LM, Zandstra DF (2009) Citrate anticoagulation for continuous venovenous hemofiltration. Crit Care Med 37:545–552
Soubrier S, Leroy O, Devos P, Nseir S, Georges H, d’Escrivan T, Guery B (2006) Epidemiology and prognostic factors of critically ill patients treated with hemodiafiltration. J Crit Care 21:66–72
Tolwani AJ, Campbell RC, Stofan BS, Lai KR, Oster RA, Wille KM (2008) Standard versus high-dose CVVHDF for ICU-related acute renal failure. J Am Soc Nephrol 19:1233–1238
Vats HS, Dart RA, Okon TR, Liang H, Paganini EP (2011) Does early initiation of continuous renal replacement therapy affect outcome: experience in a tertiary care center. Ren Fail 33:698–706
Hussain S, Piering W, Mohyuddin T, Saleh M, Zhu Y-R, Hannan M, Cohen E (2009) Outcome among patients with acute renal failure needing continuous renal replacement therapy: a single center study. [Erratum appears in Hemodial Int. 2010 Jan; 14(1):152 Note: Hanan, Mary [corrected to Hannan, Mary]]. Hemodial Int 13:205–214
Rabindranath KS, Adams J, Shail R, Macleod AM, Muirhead N (2009) Does dialysis therapy modality matter in acute renal failure? A systematic review of randomised controlled trials comparing intermittent and continuous renal replacement therapy. Br J Intensive Care 19:110–121
Kelleher SP, Robinette JB, Miller F, Conger JD (1987) Effect of hemorrhagic reduction in blood pressure on recovery from acute renal failure. Kidney Int 31:725–730
Adams PL, Adams FF, Bell PD, Navar LG (1980) Impaired renal blood flow autoregulation in ischemic acute renal failure. Kidney Int 18:68–76
Matthys E, Patton MK, Osgood RW, Venkatachalam MA, Stein JH (1983) Alterations in vascular function and morphology in acute ischemic renal failure. Kidney Int 23:717–724
Palevsky PM, Zhang JH, O’Connor TZ, Chertow GM, Crowley ST, Choudhury D, Finkel K, Kellum JA, Paganini E, Schein RM, Smith MW, Swanson KM, Thompson BT, Vijayan A, Watnick S, Star RA, Peduzzi P (2008) Intensity of renal support in critically ill patients with acute kidney injury. N Engl J Med 359:7–20
Davenport A (2006) Intradialytic complications during hemodialysis. Hemodial Int 10:162–167
Manns M, Sigler MH, Teehan BP (1997) Intradialytic renal haemodynamics–potential consequences for the management of the patient with acute renal failure. Nephrol Dial Transpl 12:870–872
Lameire N, Van Biesen W, Vanholder R, Colardijn F (1998) The place of intermittent hemodialysis in the treatment of acute renal failure in the ICU patient. Kidney Int Suppl 66:S110–S119
Abdeen O, Mehta RL (2002) Dialysis modalities in the intensive care unit. Crit Care Clin 18:223–247
Selby NM, Lambie SH, Camici PG, Baker CS, McIntyre CW (2006) Occurrence of regional left ventricular dysfunction in patients undergoing standard and biofeedback dialysis. Am J Kidney Dis 47:830–841
Burton JO, Jefferies HJ, Selby NM, McIntyre CW (2009) Hemodialysis-induced repetitive myocardial injury results in global and segmental reduction in systolic cardiac function. Clin J Am Soc Nephrol 4:1925–1931
Davenport A, Will EJ, Davidson AM (1993) Improved cardiovascular stability during continuous modes of renal replacement therapy in critically ill patients with acute hepatic and renal failure. Crit Care Med 21:328–338
Van der Schueren G, Diltoer M, Laureys M, Huyghens L (1996) Intermittent hemodialysis in critically ill patients with multiple organ dysfunction syndrome is associated with intestinal intramucosal acidosis. Intensive Care Med 22:747–751
Conger JD (1990) Does hemodialysis delay recovery from acute renal failure? Semin Dial 3:146–148
Kramer JR, Hertzer NR, Taylor PC (1977) Traumatic arterial embolism producing arteriovenous fistula. Heart Lung 6:1023–1026
Kohen JA, Whitley KY, Kjellstrand CM (1985) Continuous arteriovenous hemofiltration: a comparison with hemodialysis in acute renal failure. Trans Am Soc Artif Intern Organs 31:169–175
Baldwin IC, Elderkin TD (1995) Continuous hemofiltration: nursing perspectives in critical care. New Horiz 3:738–747
Yang L, Besschetnova TY, Brooks CR, Shah JV, Bonventre JV (2010) Epithelial cell cycle arrest in G2/M mediates kidney fibrosis after injury. Nat Med 16:535–543 (531 p following 143)
Hayes JA, Black NA, Jenkinson C, Young JD, Rowan KM, Daly K, Ridley S (2000) Outcome measures for adult critical care: a systematic review. Health Technol Assess 4:1–111
Ramsay P (2011) Health-related quality of life: implications for critical care interventional studies and why we need to collaborate with patients. Curr Opin Crit Care 17:510–514
Johansen KL, Smith MW, Unruh ML, Siroka AM, O’Connor TZ, Palevsky PM (2010) Predictors of health utility among 60-day survivors of acute kidney injury in the Veterans Affairs/National Institutes of Health Acute Renal Failure Trial Network Study. Clin J Am Soc Nephrol 5:1366–1372
Acknowledgments
We thank Drs. Vijayan, Lins, Ahlstrom, Andrikos, Garcia-Fernandes, and Marshall for making data available. This study was funded by the Austin Hospital Anaesthesia and Intensive Care Trust Fund. Preliminary results of this study were presented at the 32nd ISICEM conference and published as an abstract in Critical Care. Dr. Bagshaw is supported by a Canada Research Chair in Critical Care Nephrology and Clinical Investigator Award from Alberta Innovates-Health Solutions.
Conflicts of interest
Drs. Bellomo and Bagshaw have acted as occasional paid consultant for Gambro Pty ltd over the last five years. All other authors stated that they had no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Schneider, A.G., Bellomo, R., Bagshaw, S.M. et al. Choice of renal replacement therapy modality and dialysis dependence after acute kidney injury: a systematic review and meta-analysis. Intensive Care Med 39, 987–997 (2013). https://doi.org/10.1007/s00134-013-2864-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-013-2864-5