Abstract
Surgery is developing in the direction of minimal invasiveness, and robotic surgery is becoming increasingly adopted in colonic resection procedures. The ergonomic improvements of robot promote surgical performance, reduce workload for surgeons and benefit patients. Compared with laparoscopy-assisted colon surgery, the robotic approach has the advantages of shorter length of hospital stay, lower rate of conversion to open surgery, and lower rate of intraoperative complications for short-term outcomes. Synchronous robotic liver resection with colon cancer is feasible. The introduction of the da Vinci Xi System (Intuitive Surgical, Inc., Sunnyvale, CA, USA) has introduced more flexibility to colonic operations. Optimization of the suprapubic surgical approach may shorten the length of hospital stay for patients who undergo robotic colonic resection. Single-port robotic colectomy reduces the number of robotic ports for better looking and faster recovery. Intestinal anastomosis methods using totally robotic surgery result in shorter time to bowel function recovery and tolerance to a solid diet, although the operative time is longer. Indocyanine green is used as a tracer to assess blood supplementation in the anastomosis and marks lymph nodes during operation. The introduction of new surgical robots from multiple manufacturers is bound to change the landscape of robotic surgery and yield high-quality surgical outcomes. The present article reviews recent advances in robotic colonic resection over the past five years.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Colon cancer is one of the most common cancers worldwide and is associated with a high mortality rate [1]. The primary treatment method is colonic resection, and surgical approaches vary from open surgery to minimally invasive procedures. Laparoscopic surgery has gradually replaced laparotomy and has become an important approach in recent years. The advent of surgical robots has opened new avenues to operative techniques. The advantages of enlarged three-dimensional views, flexible wrists, and filtration of hand tremors enable surgeons to perform meticulous operations in small spaces, which has ushered in an entirely new experience to operators. The robots may afford superior ergonomic benefits and reduced workload for surgeons compared with laparoscopy [2]. Data from the National Cancer Database in the United States have demonstrated that the use of robot-assisted surgery for colon cancers is rapidly increasing, and being used more frequently in younger and healthier patients [3]. Robotic colonic resection applies not only to malignant tumors but also to some benign diseases, such as inflammatory bowel disease and colonic diverticulum. Meanwhile, innovations in surgical techniques have demonstrated clinical value, such as the suprapubic surgical approach, single-port robotic colectomy, intracorporeal anastomosis, and the use of tracers. The present article reviews recent advances in robotic colonic resection over the past 5 years.
Materials and methods
Studies were obtained from the databases Pubmed (http://www.ncbi.nlm.nih.gov/pubmed/) and Embase (http://www.embase.com) to May 15, 2020. The following text and key words were used in the search: colorectal cancer ("colorectal cancer" OR "colorectal carcinoma" OR "rectal cancer" OR "rectal carcinoma" OR "colon cancer" OR "colorectal cancer" OR "colorectal carcinoma" OR "carcinoma of colon" OR "colorectal neoplasm"), robotic surgery ("robot" OR "robotic" OR "da vinci" OR "davinci") and laparoscopic surgery ("laparoscopies" OR "laparoscopic" OR "laparoscopy"). Logical combinations of related terms were employed to maximize sensitivity. The process of papers screening was show in Fig. 1.
Conventional controversy: robot versus laparoscopy
The efficacy of robotic colonic resection has attracted the attention of surgeons. The comparison of advantages and disadvantages of robotic versus laparoscopic surgery has always been a "hot topic". Comparative studies investigating the effects of robot-assisted (RACS) and laparoscopy-assisted colonic surgery (LACS) remain ongoing and have provided definitive data.
Colectomy
Some studies have included patients who underwent RACS or LACS without precise distinction of tumor sites (Table 1). Estimated blood loss, rate of intraoperative blood transfusion, rate of conversion to open surgery, and rate of complications, such as ileus and anastomotic leakage, were lower using RACS than LACS in some studies, which indicated better short-term outcomes. Several studies have reported shorter time to bowel function recovery and hospital stay in the robotic group, which reflect faster post-operative recovery. In addition to these advantages, longer operative time has been described in some investigations, and higher cost of robot was reported in virtually all studies. Long-term factors, such as overall survival (OS) and disease-free survival (DFS), were comparable using both surgical approaches.
Right hemicolectomy
Some studies have devoted close attention to patients undergoing robot-assisted (RARH) and laparoscopy-assisted right hemicolectomy (LARH) (Table 2). Similar to RACS, the advantages of RARH were primarily reflected by lower rate of conversion to open surgery and complications. Increased harvest of lymph nodes in robotic resection has been reported in several studies. Higher cost and comparable long-term outcomes of RARH versus LARH were similar to the above studies investigating colectomy.
Left hemicolectomy and sigmoidectomy
Studies investigating left hemicolectomy are rare due to the low morbidity of tumors of the left semicolon. Some studies addressing benign lesions, such as diverticulum, have also reported the surgical effects (Table 3). The robot demonstrated good performance in the mobilization of colonic splenic flexure and was feasible for simple and complicated diverticular diseases of the sigmoid. Several studies reported shorter length of hospital stay, lower rate of conversion to open surgery, higher total hospital charges, and comparable postoperative complications in robotic-assisted left hemicolectomy and sigmoidectomy compared with laparoscopic surgery.
Synchronous colonic resection with liver metastases
Liver metastases are particular focus of surgeons in improving the survival of patients with colon cancer. Unfortunately, only 25% of colon cancer patients with liver metastases are suitable candidates for liver resection [27]. Dwyer et al. [28] described synchronous robotic surgery for patients with stage IV colorectal cancer with liver metastases. This retrospective review of prospectively collected data, however, included only six patients. Liver treatment was performed first in consideration of intraoperative bleeding risk. The authors reported that the robotic approach contributed to low blood loss (150–1000 mL), appropriate length of hospital stay (3–10 days), and no 30-day mortality. This study supported the potential benefits of synchronous robotic liver resection with colon cancer.
It is clear from the above studies investigating different surgical methods and tumor sites that most reported shorter length of hospital stay, lower rate of conversion to open surgery, and lower rate of intraoperative complications in robotic colonic resection, while long-term indexes in all studies, such as OS and DFS, were not significantly different from the laparoscopic approach (Table 4). The high cost of robotic surgery, mainly coming from high selling price, expensive consumables and daily maintenance expense, however, has become an important factor that restricts its application in surgery. The surgical robot, appeared as an advanced tool, do bring some benefits to patients though the long-term efficacy is comparable with laparoscopy, and studies with multicenter and long-term follow-up are needed.
New tools and methods
With advances in engineering and technology, surgical robots, such as the da Vinci robotic system, are continually improved. Together, new ideas and innovative methods, such as the suprapubic surgical approach, single-port robotic colectomy, intracorporeal anastomosis and the use of tracers, are applied in the clinic. These new changes have demonstrated promising potential in robotic colonic resection.
Outcomes using the da Vinci Xi system
The da Vinci Xi (dVXi) surgical system is the fourth generation of surgical robot from Intuitive Surgical, Inc. (Sunnyvale, CA, USA), which attracted the attention of surgeons as soon as it was introduced to the market. The versatility of this new system, including integrated table motion, more sophisticated arms, and complex imaging units, enables it to perform a wide range of colonic procedures, from complex multiquadrant colectomies to intracorporeal anastomosis in a narrow space. The dVXi is also flexible in the complete mobilization of the colonic splenic flexure. Research investigating surgical outcomes of the dVXi has been reported continuously in recent years (Table 5). Use of the dVXi may result in shorter operative time, less estimated blood loss, and faster postoperative recovery than its predecessor, the da Vinci Si (dVSi), shorter length of hospital stay, and shorter duration of postoperative ileus than the laparoscopic approach. Long-term outcomes using this system, however, are currently lacking.
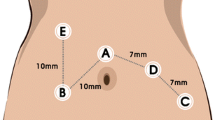
Optimization of the suprapubic surgical approach
Exploration of new surgical approaches, such as the optimization of the suprapubic method, is of clinical significance. A convenient surgical approach can increase the fluency of the operation and reduce collision of the robotic arms. The suprapubic surgical approach means colonic resection performed with horizontal linear placement of ports in the suprapubic area, especially applied in the robotic right hemicolectomy (Fig. 2). Hamilton et al. [37] reviewed technical and perioperative outcomes using the dVXi and dVSi systems with suprapubic port placement (SPPP) or traditional port placement (TPP) in 138 patients who underwent totally robotic right hemicolectomy (RRHC). The authors reported that SPPP had more advantages than TPP, with less console time and shorter hospital stay. Yeo et al. [38] developed a potentially universal SPPP strategy for robotic colectomy with complete mesocolic excision (CME) and central vascular ligation using the dVXi robotic system from cadaveric models. Lee et al. [39], from Korea, and Schulte Am Esch et al. [40], from Germany, separately described robotic right hemicolectomy using the suprapubic access strategy, and the perioperative effects were relatively satisfactory. The above studies reported optimistic short-term effects, and optimization of the suprapubic surgical approach may be widely used in the future when application conditions and long-term efficacy are further clarified.
Application of single-port robotic surgery
Surgeons are attempting to reduce the number of robotic ports for better looking and faster recovery in robotic colonic resection. Single-port robotic (SPR) surgery has begun to be applied in the clinic, which just as its name implies, performs colonic resection through a single-incision with the assistance of robot (Fig. 3). From the United States, Juo et al. [41] completed 1 case of SPR total colectomy and reported that it was a feasible procedure associated with a small increase in operative time. In another study from the United States, Marks et al. [42] reported 2 cases, and Bae et al. [43] from Korea reported 23 cases of SPR left colectomy, indicating that this method was feasible and safe. Spinoglio et al. [44] successfully performed 3 cases of SPR right colectomies and completed intracorporeal anastomosis using the da Vinci single-site platform through a suprapubic incision. A systemic review [45] of current studies revealed that SPR surgery for colonic diseases was feasible and safe with acceptable perioperative outcomes (early postoperative complications 0–36.4% and hospital stay 2–9 days, comparable with those of multi-port robotic surgery). Presently, research investigating SPR colectomy is limited by small sample sizes. The results of larger-sample and longer-term studies, therefore, are eagerly anticipated.
Use of intracorporeal anastomosis
Intracorporeal (IC) anastomosis of the intestinal canal during colectomy is a relatively new surgical method that changes the way in which the colon is pulled from the body through a surgical incision before anastomosis. This anastomosis method reduces the pulling of the intestinal tract to a certain extent, which may reduce postoperative complications. A systematic review and meta-analysis [46] from Italy found a higher rate of IC anastomosis in robotic right colectomy than in the laparoscopic group. Ngu et al. [47] from Singapore reported that robotic group recorded statistically shorter time for IC anastomosis, greater lymph node harvest and similar postoperative recovery and complication rate compared with laparoscopic group in right hemicolectomy with IC anastomosis. Some studies have verified the safety and feasibility of robotic IC anastomosis, and some reported shorter extraction site incisions, earlier bowel recovery, fewer complications, and lower rate of conversion, anastomotic leakage, surgical site infection, and incisional hernia, but longer operative time in IC than extracorporeal (EC) anastomosis, which indicated that IC had broad prospects for application in colonic surgery (Table 6). There remains a lack of long-term results of IC versus EC in robotic colonic resection.
Development of totally robotic surgery
The proportion of total robotic colectomies without laparoscopic assistance has recently increased. Studies investigating totally robotic colonic resection have reported safety and feasibility. A retrospective study from China by Liu et al. [54] included 64 cases of totally robotic right hemicolectomy (TRRH) and 128 cases of RARH and compared short- and long-term outcomes. The authors reported longer mean operative time, incision length, lower postoperative pain score and shorter time to pass flatus in the TRRH compared with RARH group. The 3-year OS and DFS rate were comparable between the two groups. Ozben et al. [55] completed a retrospective review of 37 patients undergoing totally robotic right-sided CME with a mean operative time of 289.8 ± 85.3 min, estimated blood loss of 77.4 ± 70.5 mL, 41.8 ± 11.9 harvested lymph nodes, and mean length of hospital stay of 6.6 ± 3.7 days. Scotton et al. [56] reported a longer operative time and faster bowel function recovery and tolerance to solid diet in CME with totally robotic right colectomy versus hybrid robot-assisted right colectomy. However, the difference was not statistically significant.
The superiority of this method, however, needs further confirmation. Meanwhile, research investigating long-term efficacy is urgently needed. Totally robotic surgery with intracorporeal anastomosis may replace the existing hybrid robot-assisted extracorporeal anastomosis as the mainstream surgical approach in the future.
Use of tracers
In recent years, the use of tracers in surgery has changed the face of colectomy, and indocyanine green (ICG) is the most commonly used tracer in colonic resection. ICG, a fluorescent tricarbocyanine compound, which rapidly binds to plasma proteins when injected intravenously, can produce fluorescence under infrared excitation. Currently, research investigating ICG assessing blood supply of anastomotic stoma to prevent anastomotic leakage is increasing, especially in laparoscopic colorectal surgery. In a systematic review and meta-analysis of intraoperative anastomotic testing in colorectal surgery from Treviglio Hospital (Lombardy, Italy), Rausa et al. [57] suggested that using ICG for blood supply assessment may reduce the anastomotic leakage rate. Yang et al. [58] reported that the use of ICG facilitated the delineation of the vascular anatomy, and Munechika et al. [59] demonstrated that high ligation of the inferior mesenteric artery for descending colon cancer under ICG fluorescence imaging was safe and effective in a pilot study. Van den Bos et al. [60] reported that both subjective and measured fluorescence intensity of ICG appeared to be related to anastomotic leakage in a clinical pilot study including 30 patients undergoing either laparoscopic or robotic anastomotic colorectal surgery.
Except for the assessment of vascularization of the colic stump, ICG can also be used as a tracer for lymph nodes in colectomy, which helps complete lymph node dissection more efficiently. Park et al. [61] performed a retrospective study and found that real-time ICG fluorescence imaging of lymph nodes may improve the performance of radical D3 lymph node dissection during laparoscopic hemicolectomy for advanced right-sided colon cancer.
Regrettably, this type of research investigating ICG in robotic colectomy is relatively rare. “Firefly” technology, integrated on the dVXi, enables tracers, such as ICG, to complete the assessment of colon perfusion and identification of lymph nodes more efficiently. Therefore, it is foreseeable that tracers will play an increasingly important role in colonic resection, and we believe that many reports describing the value of ICG in robotic colectomy are forthcoming.
Learning curve of robotic surgery
The learning curve represents the number of cases for surgeons to achieve plateau performance through a new procedure. Pernar et al. [62] found that 19–128 cases of robotic colorectal operation were needed for surgeons. Shaw et al. [63] made a conclusion after retrospectively reviewed 62 patients that overall complications were reduced after first 15 cases. Symer et al. [64] reported that iatrogenic complications were reduced after surgeons completed 27 cases of robotic colorectal resection in a study including 2763 procedures. Gerbaud et al. [65] further reported that a surgeon, who was experienced in laparoscopic surgery, may not cause any increase on the morbidity rate of complications when started to perform robotic right colectomy with intracorporeal anastomosis. It can be seen from the above studies that, for surgeons, the learning curve of robotic surgery was relatively short, which was convenient for rapid clinical application.
Surgical robots from multiple manufacturers
Since the turn of the century, robot applied in colonic surgery has been synonymous with the da Vinci robotic surgical system. The introduction of new robotic platforms will grow considerably in the near future as several manufacturers are active in the developing stages of robotic systems.
"MicroHand S", a surgical robot from China, has entered clinical trials. Some studies have also reported good performance and application prospects. Yi et al. [66] reported 10 surgical procedures with the assistance of MicroHand S without intraoperative complications or technical problems. Luo et al. [67] retrospectively analyzed 45 patients with sigmoid colon cancer who underwent MicroHand S or da Vinci robotic-assisted surgery. In patients with sigmoid colon cancer, the Da Vinci surgical system did not demonstrate obvious clinical advantages compared with the MicroHand S surgical system. However, the MicroHand S surgical platform demonstrated advantages in terms of the hospitalization cost and length of postoperative bedtime. The outcome of this study indicated that the MicroHand S surgical system may have good prospects for application in surgical fields in China.
The novel Senhance robotic system (TransEnterix Surgical Inc., Morrisville, NC, USA) has been used in a variety of applications in Europe and approved for limited clinical use in the United States. In a study from Germany, Darwich et al. [68] collected 12 patients who underwent sigmoid resection using the Senhance surgical robotic system and confirmed that this robotic system can be used safely in sigmoid resection for diverticular disease after adequate training. Samalavicius et al. [69] performed a prospective analysis of the first 100 robotic surgeries using the Senhance robotic system in Lithuania, and reported that this robotic system was feasible and safe in general surgery.
In addition to the MicroHand S and Senhance systems, “Hugo RAS” from Medtronic Inc. (Dublin, Ireland) and “Versius” from CMR Surgical Ltd. (Cambridge, United Kingdom) have demonstrated promising potential in clinical applications. The emergence of surgical robots from multiple manufacturers is bound to change da Vinci’s market share and the cost of surgery is expected to decrease, which ultimately benefits patients.
Dissenting opinions regarding robotics
Although most studies have reported that robotic-assisted colectomy yielded equal or better surgical outcomes than the laparoscopic approach, a study from Johns Hopkins University School of Medicine (Baltimore, MD, USA) by Lo et al. [70] reached a different conclusion. The authors conducted a retrospective study of prospectively collected data to examine the impact of minimally invasive surgical approaches in frailty (defined as a loss of physiological reserve and association with adverse health outcomes, including disability, hospitalization, and mortality) with colon cancer. A total of 37,977 colectomies, performed between 2012 and 2016, were included and the primary outcome measure was 30-day postoperative complications. The study concluded that frailer patients experienced increased complication rate and were more likely to develop major complications. Results of this study serve as a reminder to be cautious when performing robotic colon surgery on certain subsets of the population, such as frail individuals.
Summary
Advances in the development of minimally invasive surgery and the application of surgical robots with ergonomic advantages in colonic resection have afforded several benefits to patients. Comparative studies investigating the efficacy of robots and laparoscopies remain the focus of research in colon surgery. Simultaneously, the application of the optimized suprapubic surgical approach, single-port robotic surgery, robotic intracorporeal anastomosis, totally robotic surgery, and tracers for vessels and lymph nodes are gradually popularized in colonic resection. The introduction of new surgical robots from multiple manufacturers will reduce the burden of healthcare cost and bring a new look to the field of colonic surgery. With innovative approaches and emerging robotic technologies, we believe that robotic colonic resection will have good application prospects.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424. https://doi.org/10.3322/caac.21492
Kuo LJ, Ngu JC, Lin YK, Chen CC (2020) Tang YH (2020) A pilot study comparing ergonomics in laparoscopy and robotics: beyond anecdotes, and subjective claims. J Surg Case Rep 2:rjaa005. https://doi.org/10.1093/jscr/rjaa005
Konstantinidis IT, Ituarte P, Woo Y, Warner SG, Melstrom K, Kim J, Singh G, Lee B, Fong Y, Melstrom LG (2019) Trends and outcomes of robotic surgery for gastrointestinal (GI) cancers in the USA: maintaining perioperative and oncologic safety. Surg Endosc. https://doi.org/10.1007/s00464-019-07284-x
Huerta S, Meier J, Emuakhagbon VS, Favela J, Argo M, Polanco PM, Augustine MM, Pham T (2020) A comparative analysis of outcomes of open, laparoscopic, and robotic elective (procto-) colectomies for benign and malignant disease. J Robot Surg. https://doi.org/10.1007/s11701-020-01069-4
Chiu CC, Hsu WT, Choi JJ, Galm B, Lee MG, Chang CN, Liu CC, Lee CC (2019) Comparison of outcome and cost between the open, laparoscopic, and robotic surgical treatments for colon cancer: a propensity score-matched analysis using nationwide hospital record database. Surg Endosc 33(11):3757–3765. https://doi.org/10.1007/s00464-019-06672-7
Polat F, Willems LH, Dogan K, Rosman C (2019) The oncological and surgical safety of robot-assisted surgery in colorectal cancer: outcomes of a longitudinal prospective cohort study. Surg Endosc 33(11):3644–3655. https://doi.org/10.1007/s00464-018-06653-2
Zhu XL, Yan PJ, Yao L, Liu R, Wu DW, Du BB, Yang KH, Guo TK, Yang XF (2019) Comparison of short-term outcomes between robotic-assisted and laparoscopic surgery in colorectal cancer. Surg Innov 26(1):57–65. https://doi.org/10.1177/1553350618797822
Ozben V, de Muijnck C, Karabork M, Ozoran E, Zenger S, Bilgin IA, Aytac E, Baca B, Balik E, Hamzaoglu I, Karahasanoglu T, Bugra D (2019) The da Vinci Xi system for robotic total/subtotal colectomy vs. conventional laparoscopy: short-term outcomes. Tech Coloproctol 23(9):861–868. https://doi.org/10.1007/s10151-019-02066-y
Sheng S, Zhao T, Wang X (2018) Comparison of robot-assisted surgery, laparoscopic-assisted surgery, and open surgery for the treatment of colorectal cancer: a network meta-analysis. Medicine 97(34):e11817. https://doi.org/10.1097/md.0000000000011817
Pinar I, Fransgaard T, Thygesen LC, Gogenur I (2018) Long-term outcomes of robot-assisted surgery in patients with colorectal cancer. Ann Surg Oncol 25(13):3906–3912. https://doi.org/10.1245/s10434-018-6862-2
Kulaylat AS, Mirkin KA, Puleo FJ, Hollenbeak CS, Messaris E (2018) Robotic versus standard laparoscopic elective colectomy: where are the benefits? J Surg Res 224:72–78. https://doi.org/10.1016/j.jss.2017.11.059
Nolan HR, Smith BE, Honaker MD (2018) Operative time and length of stay is similar between robotic assisted and laparoscopic colon and rectal resections. J Robot Surg 12(4):659–664. https://doi.org/10.1007/s11701-018-0798-1
Fransgaard T, Pinar I, Thygesen LC, Gogenur I (2018) Association between robot-assisted surgery and resection quality in patients with colorectal cancer. Surg Oncol 27(2):177–184. https://doi.org/10.1016/j.suronc.2018.03.003
Benlice C, Aytac E, Costedio M, Kessler H, Abbas MA, Remzi FH, Gorgun E (2017) Robotic, laparoscopic, and open colectomy: a case-matched comparison from the ACS-NSQIP. Int J Med Robot Comput Assist Surg. https://doi.org/10.1002/rcs.1783
Zhang X, Wei Z, Bie M, Peng X, Chen C (2016) Robot-assisted versus laparoscopic-assisted surgery for colorectal cancer: a meta-analysis. Surg Endosc 30(12):5601–5614. https://doi.org/10.1007/s00464-016-4892-z
Waters PS, Cheung FP, Peacock O, Heriot AG, Warrier SK, O’Riordain DS, Pillinger S, Lynch AC, Stevenson ARL (2019) Successful patient-oriented surgical outcomes in robotic vs laparoscopic right hemicolectomy for cancer—a systematic review. Colorect Dis . https://doi.org/10.1111/codi.14822
Yozgatli TK, Aytac E, Ozben V, Bayram O, Gurbuz B, Baca B, Balik E, Hamzaoglu I, Karahasanoglu T, Bugra D (2019) Robotic complete mesocolic excision versus conventional laparoscopic hemicolectomy for right-sided colon cancer. J Laparoendosc Adv Surg Tech A. https://doi.org/10.1089/lap.2018.0348
Park JS, Kang H, Park SY, Kim HJ, Woo IT, Park IK, Choi GS (2019) Long-term oncologic after robotic versus laparoscopic right colectomy: a prospective randomized study. Surg Endosc 33(9):2975–2981. https://doi.org/10.1007/s00464-018-6563-8
Haskins IN, Ju T, Skancke M, Kuang X, Amdur RL, Brody F, Obias V, Agarwal S (2018) Right colon resection for colon cancer: does surgical approach matter? J Laparoendosc Adv Surg Tech A 28(10):1202–1206. https://doi.org/10.1089/lap.2018.0148
Spinoglio G, Bianchi PP, Marano A, Priora F, Lenti LM, Ravazzoni F, Petz W, Borin S, Ribero D, Formisano G, Bertani E (2018) Robotic versus laparoscopic right colectomy with complete mesocolic excision for the treatment of colon cancer: perioperative outcomes and 5-year survival in a consecutive series of 202 patients. Ann Surg Oncol 25(12):3580–3586. https://doi.org/10.1245/s10434-018-6752-7
Kang J, Park YA, Baik SH, Sohn SK, Lee KY (2016) A comparison of open, laparoscopic, and robotic surgery in the treatment of right-sided colon cancer. Surg Laparosc Endosc Percutan Tech 26(6):497–502. https://doi.org/10.1097/sle.0000000000000331
Alharthi S, Reilly M, Arishi A, Ahmed AM, Chulkov M, Qu W, Ortiz J, Nazzal M, Pannell S (2020) Robotic versus laparoscopic sigmoid colectomy: analysis of healthcare cost and utilization project database. Am Surg 86(3):256–260. https://doi.org/10.1177/000313482008600337
Bastawrous AL, Landmann RG, Liu Y, Liu E, Cleary RK (2020) Incidence, associated risk factors, and impact of conversion to laparotomy in elective minimally invasive sigmoidectomy for diverticular disease. Surg Endosc 34(2):598–609. https://doi.org/10.1007/s00464-019-06804-z
Grass F, Crippa J, Mathis KL, Kelley SR, Larson DW (2019) Feasibility and safety of robotic resection of complicated diverticular disease. Surg Endosc 33(12):4171–4176. https://doi.org/10.1007/s00464-019-06727-9
Kim JC, Lee JL, Yoon YS, Kim CW, Park IJ, Lim SB (2018) Robotic left colectomy with complete mesocolectomy for splenic flexure and descending colon cancer, compared with a laparoscopic procedure. Int J Med Robot Comput Assis Surg 14(5):e1918. https://doi.org/10.1002/rcs.1918
Crolla R, Tersteeg JJC, van der Schelling GP, Wijsman JH, Schreinemakers JMJ (2018) Robot-assisted laparoscopic resection of clinical T4b tumours of distal sigmoid and rectum: initial results. Surg Endosc 32(11):4571–4578. https://doi.org/10.1007/s00464-018-6210-4
Dorr NM, Bartels M, Morgul MH (2020) Current treatment of colorectal liver metastasis as a chronic disease. Anticancer Res 40(1):1–7. https://doi.org/10.21873/anticanres.13921
Dwyer RH, Scheidt MJ, Marshall JS, Tsoraides SS (2018) Safety and efficacy of synchronous robotic surgery for colorectal cancer with liver metastases. J Robot Surg 12(4):603–606. https://doi.org/10.1007/s11701-018-0813-6
Hill A, McCormick J (2020) In experienced hands, does the robotic platform impact operative efficiency Comparison of the da Vinci Si versus Xi robot in colorectal surgery. J Robot Surg. https://doi.org/10.1007/s11701-020-01055-w
Fleming CA, Westby D, Ullah MF, Mohan HM, Sehgal R, Bolger JC, O’Leary DP, McNamara E, Korpanty G, El Bassiouni M, Condon E, Coffey JC, Peirce C (2020) A review of clinical and oncological outcomes following the introduction of the first robotic colorectal surgery programme to a university teaching hospital in Ireland using a dual console training platform. J Robot Surg. https://doi.org/10.1007/s11701-020-01073-8
Huang YM, Huang YJ, Wei PL (2019) Colorectal Cancer Surgery using the Da Vinci Xi and Si systems: comparison of perioperative outcomes. Surg Innov 26(2):192–200. https://doi.org/10.1177/1553350618816788
Beltzer C, Knoerzer L, Bachmann R, Axt S, Dippel H, Schmidt R (2019) Robotic versus laparoscopic sigmoid resection for diverticular disease: a single-center experience of 106 cases. J Laparoendosc Adv Surg Tech A 29(11):1451–1455. https://doi.org/10.1089/lap.2019.0451
Liang JT, Huang J, Chen TC (2019) Standardize the surgical technique and clarify the relevant anatomic concept for complete mobilization of colonic splenic flexure using da Vinci Xi((R)) robotic system. World J Surg 43(4):1129–1136. https://doi.org/10.1007/s00268-018-04882-z
Protyniak B, Jorden J, Farmer R (2018) Multiquadrant robotic colorectal surgery: the da Vinci Xi vs Si comparison. J Robot Surg 12(1):67–74. https://doi.org/10.1007/s11701-017-0689-x
Jimenez-Rodriguez RM, Quezada-Diaz F, Tchack M, Pappou E, Wei IH, Smith JJ, Nash GM, Guillem JG, Paty PB, Weiser MR, Garcia-Aguilar J (2019) Use of the Xi robotic platform for total abdominal colectomy: a step forward in minimally invasive colorectal surgery. Surg Endosc 33(3):966–971. https://doi.org/10.1007/s00464-018-6529-x
Ngu JC, Sim S, Yusof S, Ng CY, Wong AS (2017) Insight into the da Vinci(R) Xi - technical notes for single-docking left-sided colorectal procedures. Int J Med Robot Comput Assist Surg. https://doi.org/10.1002/rcs.1798
Hamilton AER, Chatfield MD, Johnson CS, Stevenson ARL (2019) Totally robotic right hemicolectomy: a multicentre case-matched technical and peri-operative comparison of port placements and da Vinci models. J Robot Surg. https://doi.org/10.1007/s11701-019-01014-0
Yeo SA, Noh GT, Han JH, Cheong C, Stein H, Kerdok A, Min BS (2017) Universal suprapubic approach for complete mesocolic excision and central vascular ligation using the da Vinci Xi((R)) system: from cadaveric models to clinical cases. J Robot Surg 11(4):399–407. https://doi.org/10.1007/s11701-016-0664-y
Lee HJ, Choi GS, Park JS, Park SY, Kim HJ, Woo IT, Park IK (2018) A novel robotic right colectomy for colon cancer via the suprapubic approach using the da Vinci Xi system: initial clinical experience. Ann Surg Treat Res 94(2):83–87. https://doi.org/10.4174/astr.2018.94.2.83
Schulte Am Esch J, Iosivan SI, Steinfurth F, Mahdi A, Forster C, Wilkens L, Nasser A, Sarikaya H, Benhidjeb T, Kruger M (2019) A standardized suprapubic bottom-to-up approach in robotic right colectomy: technical and oncological advances for complete mesocolic excision (CME). BMC Surg 19(1):72. https://doi.org/10.1186/s12893-019-0544-2
Juo YY, Obias V (2015) Robot-assisted single-incision total colectomy: a case report. Int J Med Robot Comput Assist Surg 11(1):104–108. https://doi.org/10.1002/rcs.1593
Marks JH, Salem JF, Anderson BK, Josse JM, Schoonyoung HP (2020) Single-port robotic left colectomy: first clinical experience using the SP robot (rSILS). Tech Coloproctol 24(1):57–63. https://doi.org/10.1007/s10151-019-02129-0
Bae SU, Jeong WK, Baek SK (2017) Robot-assisted colectomy for left-sided colon cancer: comparison of reduced-port and conventional multi-port robotic surgery. J Laparoendosc Adv Surg Tech A 27(4):398–403. https://doi.org/10.1089/lap.2016.0427
Spinoglio G, Lenti LM, Ravazzoni F, Formisano G, Pagliardi F, Marano A (2015) Evaluation of technical feasibility and safety of Single-Site robotic right colectomy: three case reports. Int J Med Robot Comput Assist Surg 11(2):135–140. https://doi.org/10.1002/rcs.1609
Bae SU, Jeong WK, Baek SK (2017) Current status of robotic single-port colonic surgery. Int J Med Robot Comput Assist Surg. https://doi.org/10.1002/rcs.1735
Solaini L, Bazzocchi F, Cavaliere D, Avanzolini A, Cucchetti A, Ercolani G (2018) Robotic versus laparoscopic right colectomy: an updated systematic review and meta-analysis. Surg Endosc 32(3):1104–1110. https://doi.org/10.1007/s00464-017-5980-4
Ngu JC, Ng YY (2018) Robotics confers an advantage in right hemicolectomy with intracorporeal anastomosis when matched against conventional laparoscopy. J Robot Surg 12(4):647–653. https://doi.org/10.1007/s11701-018-0793-6
Widmar M, Aggarwal P, Keskin M, Strombom PD, Patil S, Smith JJ, Nash GM, Garcia-Aguilar J (2020) Intracorporeal anastomoses in minimally invasive right colectomies are associated with fewer incisional hernias and shorter length of stay. Dis Colon Rectum 63(5):685–692. https://doi.org/10.1097/dcr.0000000000001612
Emile SH, Elfeki H, Shalaby M, Sakr A, Bassuni M, Christensen P, Wexner SD (2019) Intracorporeal versus extracorporeal anastomosis in minimally invasive right colectomy: an updated systematic review and meta-analysis. Tech Coloproctol 23(11):1023–1035. https://doi.org/10.1007/s10151-019-02079-7
Al Natour RH, Obias V, Albright J, Wu J, Ferraro J, Akram WM, McClure AM, Shanker BA, Cleary RK (2019) A propensity score matched comparison of intracorporeal and extracorporeal techniques for robotic-assisted sigmoidectomy in an enhanced recovery pathway. J Robot Surg 13(5):649–656. https://doi.org/10.1007/s11701-018-00910-1
Kelley SR, Duchalais E, Larson DW (2018) Robotic right colectomy with intracorporeal anastomosis for malignancy. J Robot Surg 12(3):461–466. https://doi.org/10.1007/s11701-017-0759-0
Jung KU, Park Y, Lee KY, Sohn SK (2015) Robotic transverse colectomy for mid-transverse colon cancer: surgical techniques and oncologic outcomes. J Robot Surg 9(2):131–136. https://doi.org/10.1007/s11701-015-0502-7
Lujan HJ, Molano A, Burgos A, Rivera B, Plasencia G (2015) Robotic right colectomy with intracorporeal anastomosis: experience with 52 consecutive cases. J Laparoendosc Adv Surg Tech A 25(2):117–122. https://doi.org/10.1089/lap.2014.0199
Liu D, Li J, He P, Tang C, Lei X, Jiang Q, Li T (2019) Short- and long-term outcomes of totally robotic versus robotic-assisted right hemicolectomy for colon cancer: a retrospective study. Medicine 98(13):e15028. https://doi.org/10.1097/md.0000000000015028
Ozben V, Aytac E, Atasoy D, Erenler Bayraktar I, Bayraktar O, Sapci I, Baca B, Karahasanoglu T, Hamzaoglu I (2019) Totally robotic complete mesocolic excision for right-sided colon cancer. J Robot Surg 13(1):107–114. https://doi.org/10.1007/s11701-018-0817-2
Scotton G, Contardo T, Zerbinati A, Tosato SM, Orsini C, Morpurgo E (2018) From laparoscopic right colectomy with extracorporeal anastomosis to robot-assisted intracorporeal anastomosis to totally robotic right colectomy for cancer: the evolution of robotic multiquadrant abdominal surgery. J Laparoendosc Adv Surg Tech A 28(10):1216–1222. https://doi.org/10.1089/lap.2017.0693
Rausa E, Zappa MA, Kelly ME, Turati L, Russo A, Aiolfi A, Bonitta G, Sgroi LG (2019) A standardized use of intraoperative anastomotic testing in colorectal surgery in the new millennium: is technology taking over? A systematic review and network meta-analysis. Tech Coloproctol 23(7):625–631. https://doi.org/10.1007/s10151-019-02034-6
Yang Y, Peacock O, Malakorn S, Limvorapitak T, Zafar SN, Chang GJ (2020) Superior mesenteric vein-first approach for robotic salvage surgery with indocyanine green fluorescence angiography. Ann Surg Oncol. https://doi.org/10.1245/s10434-020-08222-1
Munechika T, Kajitani R, Matsumoto Y, Nagano H, Komono A, Aisu N, Morimoto M, Yoshimatsu G, Yoshida Y, Hasegawa S (2020) Safety and effectiveness of high ligation of the inferior mesenteric artery for cancer of the descending colon under indocyanine green fluorescence imaging: a pilot study. Surg Endosc. https://doi.org/10.1007/s00464-020-07556-x
van den Bos J, Jongen A, Melenhorst J, Breukink SO, Lenaerts K, Schols RM, Bouvy ND, Stassen LPS (2019) Near-infrared fluorescence image-guidance in anastomotic colorectal cancer surgery and its relation to serum markers of anastomotic leakage: a clinical pilot study. Surg Endosc 33(11):3766–3774. https://doi.org/10.1007/s00464-019-06673-6
Park SY, Park JS, Kim HJ, Woo IT, Park IK, Choi GS (2020) Indocyanine green fluorescence imaging-guided laparoscopic surgery could achieve radical D3 dissection in patients with advanced right-sided colon cancer. Dis Colon Rectum 63(4):441–449. https://doi.org/10.1097/dcr.0000000000001597
Pernar LIM, Robertson FC, Tavakkoli A, Sheu EG, Brooks DC, Smink DS (2017) An appraisal of the learning curve in robotic general surgery. Surg Endosc 31(11):4583–4596. https://doi.org/10.1007/s00464-017-5520-2
Shaw DD, Wright M, Taylor L, Bertelson NL, Shashidharan M, Menon P, Menon V, Wood S, Ternent CA (2018) Robotic colorectal surgery learning curve and case complexity. J Laparoendosc Adv Surg Tech A 28(10):1163–1168. https://doi.org/10.1089/lap.2016.0411
Symer MM, Sedrakyan A, Yeo HL (2019) Case Sequence Analysis of the Robotic Colorectal Resection Learning Curve. Dis Colon Rectum 62(9):1071–1078. https://doi.org/10.1097/dcr.0000000000001437
Gerbaud F, Valverde A, Danoussou D, Goasguen N, Oberlin O, Lupinacci RM (2019) Experience with transitioning from laparoscopic to robotic right colectomy. JSLS. https://doi.org/10.4293/jsls.2019.00044
Yi B, Wang G, Li J, Jiang J, Son Z, Su H, Zhu S, Wang S (2017) Domestically produced Chinese minimally invasive surgical robot system “Micro Hand S” is applied to clinical surgery preliminarily in China. Surg Endosc 31(1):487–493. https://doi.org/10.1007/s00464-016-4945-3
Luo D, Liu Y, Zhu H, Li X, Gao W, Li X, Zhu S, Yu X (2019) The MicroHand S robotic-assisted versus Da Vinci robotic-assisted radical resection for patients with sigmoid colon cancer: a single-center retrospective study. Surg Endosc. https://doi.org/10.1007/s00464-019-07107-z
Darwich I, Stephan D, Klockner-Lang M, Scheidt M, Friedberg R, Willeke F (2020) A roadmap for robotic-assisted sigmoid resection in diverticular disease using a Senhance Surgical Robotic System: results and technical aspects. J Robot Surg 14(2):297–304. https://doi.org/10.1007/s11701-019-00980-9
Samalavicius NE, Janusonis V, Siaulys R, Jasenas M, Deduchovas O, Venckus R, Ezerskiene V, Paskeviciute R, Klimaviciute G (2020) Robotic surgery using Senhance((R)) robotic platform: single center experience with first 100 cases. J Robot Surg 14(2):371–376. https://doi.org/10.1007/s11701-019-01000-6
Lo BD, Leeds IL, Sundel MH, Gearhart S, Nisly GRC, Safar B, Atallah C, Fang SH (2020) Frailer patients undergoing robotic colectomies for colon cancer experience increased complication rates compared with open or laparoscopic approaches. Dis Colon Rectum 63(5):588–597. https://doi.org/10.1097/dcr.0000000000001598
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, H., Xu, M., Liu, R. et al. The art of robotic colonic resection: a review of progress in the past 5 years. Updates Surg 73, 1037–1048 (2021). https://doi.org/10.1007/s13304-020-00969-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-020-00969-2