Abstract
Summary
We measured serum vitamin D in 8024 Romanian subjects and found a marked seasonal variation with highest levels in September and lowest levels in March. The seasonal variation (early autumn vs. early spring) persisted in all age and sex groups. The prevalence of vitamin D deficiency was very high.
Purpose
Romania is located in Eastern Europe, roughly between 44°N and 48°N latitude. Seasonal variation of serum vitamin D in Romanian subjects is unknown. We assessed the seasonal variation of 25-hydroxy vitamin D [25(OH)D] in Romanian population.
Methods
We retrieved from our endocrinology center database all 25(OH)D measurements between 2012 and 2016. We also evaluated age, sex, diagnosis, and date of blood sampling. The 25(OH)D was measured by two different chemiluminescence or electrochemiluminescence assays.
Results
There were 8024 subjects (median age 50 (37, 62); 1429 men (17.8%)) without a diagnosis of low bone mass (osteopenia or osteoporosis). The median serum 25(OH)D was 18.6 (12.7, 25.4) ng/mL. Of the subjects, 0.73, 14.4, 55.6, and 86.1% had a serum 25(OH)D level below 4, 10, 20, and 30 ng/mL, respectively. Serum 25(OH)D showed a marked seasonal variation with highest levels in September (24.1 [18.3, 30.3] ng/mL) and lowest levels in March (13.5 [9.4, 19.6] ng/mL) (p < 0.001). The seasonal variation (early autumn vs. early spring) persisted in all age and sex groups and was maximal for 21–40 years of age (26.5 (20.8, 33.1) vs. 12.9 (9.7, 17.9) ng/mL) and minimal for >65 years of age (18.6 (13.0, 27.2) vs. 12.7 (7.8, 19.7) ng/mL). Men and women showed similar amplitude of serum 25(OH)D variation.
Conclusion
The prevalence of vitamin D deficiency is high, particularly in the elderly. The data show a strong seasonal variation of serum 25(OH)D in all subgroups of our Romanian population with highest levels in September and lowest levels in March.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vitamin D (VD) is increasingly being recognized as an important factor in bone and general health. However, despite food supplementation in some countries and widely available cholecalciferol-containing medication, vitamin D deficiency and insufficiency are present at a global scale.
Serum 25-hydroxy vitamin D [25(OH)D] measurement is widely used for vitamin D status assessment, and low serum 25(OH)D levels are associated with a number of skeletal and non-skeletal disorders [1].
The serum 25(OH)D levels are influenced by many factors such as VD intake, sun exposure (latitude, climate, personal or cultural habits of sun bathing, clothing), and body mass index [2,3,4]. Despite numerous differences in all these factors among different European countries, there is a very high likelihood of inadequate VD status in all ages but especially in the elderly [5, 6]. Similar results have been reported in Romanian subjects (southeastern European country with four seasons, without mandatory food fortification with vitamin D) [7,8,9].
There is a highly significant correlation between latitude and serum 25(OH)D concentrations across Europe at least [10]. However, the policies for food fortification with VD greatly influence the results. As such, the levels are lower in central Europe than in North America or Scandinavian region (probably reflecting the more consistent VD fortification in these regions) [11]. The serum 25(OH)D concentrations vary with season in subjects of all ages, being lowest during winter and early spring [5, 6]. No study assessing the seasonality of serum VD status has been done on Romanian subjects. Therefore, we aimed to assess this aspect in a large sample of Romanian population.
Methods
Subjects
We retrieved from the electronic database of our endocrinology center all 25(OH)D measurements between May 2012 and November 2016. For each measurement, we also retrieved the corresponding sex, age and diagnosis of the subject, and the date of blood sampling. The study was approved by the Ethical Board of our Institution.
Data retrieved from National Meteorological Administration of Romania showed a stable seasonality for the studied period with not significant differences between similar months or between the studied period and the Romanian climate with regard to total monthly sunshine, rainfall, or mean temperature. Therefore, we were able to group together serum 25(OH)D levels from similar months of all studied years.
All subjects have been assessed in C. I. Parhon National Institute of Endocrinology, Bucharest, Romania. As it is the most important endocrinology center in Romania, it covers its whole territory as a tertiary referral center but also works as a secondary referral center of endocrine care for the southern part of the country. It is the particularity of primary care in our country that any clinical suspicion of endocrine disease raised by a primary care physician is referred for confirmation (or invalidation) of the diagnosis to a specialized endocrinology center. In this capacity, apart from difficult patients initially evaluated in other endocrinology centers, our center simultaneously assesses a huge number of patients of all ages referred from primary care. The reasons for referral cover the whole spectrum of endocrine disorders. Each year our center assess over 15,000 different patients.
There were 22,661 25(OH)D measurements. Of these, 14,052 were made on different subjects and 8609 were repeated measurements. In those subjects with multiple assessments available, we used only the first measurement. Of the 14,052 subjects, 6028 (42.9%) had a diagnosis of low bone mass (osteoporosis or osteopenia) and 8024 (57.1%) did not have such a diagnosis. The diagnosis of low bone mass was made by the treating physician of each individual patient, according to protocols being used in our institution (DXA determination with assessment of T-score in subjects over 50 years old and of Z-score in younger subjects as well as careful fragility fracture assessment from patient’s history and symptom-based active search). However, we could retrieve only the final diagnosis from the institution’s electronic database and did not have access to the actual radiological, densitometric, or medical history data. This distinction between patients with or without a low bone mass diagnosis was made because of the higher probability of vitamin D treatment in patients with osteoporosis or osteopenia. All further analysis and subgroups are derived from the group of 8024 subjects without a low bone mass diagnosis. Vitamin D status can be found in Table 1.
25-Hydroxy vitamin D assays
25(OH)D was measured by chemiluminescence on a Liaison XL analyzer (DiaSorin, Saluggia, Italy; with a measuring range 4–150 ng/mL, functional sensitivity 4 ng/mL, and variation coefficient of 20% or by electrochemiluminescence) on a Cobas E601 C analyzer (Roche Diagnostics, Indianapolis, IN; with a measuring range 3–70 ng/mL, functional sensitivity 4.01 ng/mL, and variation coefficient of 18.5%). Vitamin D deficiency was defined as a serum 25(OH)D level less than 10 ng/mL.
Statistical analysis
We tested for normal distribution of 25(OH)D concentrations in the whole group (8024 subjects) and in subgroups (defined by sex, age, or month of blood sampling) using D’Agostino-Pearson test. All groups were positively skewed and normal distribution was rejected. All continuous variables are expressed as median (25th, 75th percentile). Prevalence data are expressed as number (percentage).
The null hypothesis for two continuous variables was tested using Mann-Whitney U test. To quantify the difference between months with highest and lowest 25(OH)D serum concentrations in various subgroups (see Table 2), we log-transformed the variables and applied a t test for independent variables. The differences on log-transformed scale were back-transformed to obtain the ratio of geometric means and its 95% confidence interval (95% CI). Trend testing in age groups was made using the Kruskal-Wallis test.
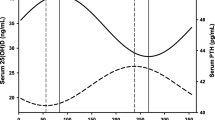
The serum 25(OH)D median monthly values (see Electronic Supplementary Material 1) show a clear periodic disposition with one peak and one nadir per year. For a better description of the annual variation of serum 25(OH)D levels, we plotted all individual 25(OH)D values against the corresponding day of serum sample (counted from the beginning of the study) (Fig. 1). A sine function was fitted using SigmaPlot 12.5 software (San Jose, CA). Minimum and maximum of the curve were found by solving the first derivative of the fitted curve equation.
Results
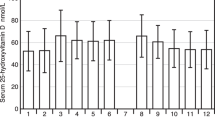
Serum 25(OH)D showed a strong seasonal variation with a nadir in early spring and peak levels at the end of summer. The lowest serum 25(OH)D concentration was found in March (13.5 [9.36, 19.63] ng/mL) and the highest in September (24.1 [18.29, 30.32] ng/mL) (p < 0.001). However, even at the end of summer, the prevalence of VD deficiency was still present in the subjects evaluated (3.6%) and was 9.2% in subjects over 65 years of age. The daily distribution of 25(OH)D serum concentrations in the whole group (8024 subjects) during the study period can be found in Fig. 1. The sinusoidal function equation between serum 25(OH)D level f(x) and the day of the blood sampling x (R = 0.3136; p < 0.001) is f(x) = 20.0027 + 4.2977 ∙ sin(2πx/361.8367–2.8119). The period of the fitted sine function is 362 days, remarkably close to 365, showing a good approximation of seasonality. The lowest serum 25(OH) concentration (min f(x) = 15.705) corresponds to beginning of March, while the highest concentration (max f(x) = 24.30) corresponds to beginning of September. Serum 25(OH)D concentration by month in 8024 subjects without a diagnosis of low bone mass and 6028 subjects with a diagnosis of low bone mass can be found in Electronic Supplementary Material 1.
Serum 25(OH)D by age can be found in Electronic Supplementary Material 2. In all months with the exception of December, there was a significant trend (p < 0.001) towards lower 25(OH)D serum concentration with advancing age. All age groups showed similar variations of 25(OH)D with a nadir in early spring (March/April) and the peak in late summer (August) or early autumn (September) (Table 2). This distribution was more pronounced in 11–20 and 21–40 years of age groups (ratio of geometric means between peak and nadir 1.81 and 1.94, respectively) than in 41–65 and over 65 years of age groups (ratio of geometric means 1.74 and 1.46, respectively).
Distribution of serum 25(OH)D by sex and month can be found in Fig. 2. Males had higher levels of 25(OH)D than females all over the year. Both sexes showed a similar peak of serum 25(OH)D in early spring (March for females, April for males) and nadir at the end of summer (August for females, September for males) (Table 2).
Discussion
Romania is located in Europe, in the Northern Hemisphere, roughly between 44° and 48°. It has a typical temperate climate with four seasons. This study presents the seasonal variation of serum 25(OH)D levels in a large sample of Romanian population. To our knowledge, it is the first study to assess serum 25(OH)D across the year in Romania and adds to others in order to fill up the European map [12].
Our study showed a nadir of serum 25(OH)D in early spring (March) for the whole sample or March/April for various subgroups and a peak level in early autumn (September) for the whole sample or August/September for subgroups. The time of nadir and peak levels was largely similar in all subgroups. However, the magnitude of the variation diverged significantly for age groups: maximal for 21–40 years of age group and minimal for >65 years of age group. Unfortunately, in our sample, the 0–10 years of age subgroup was underrepresented and no firm conclusion can be drawn for it. Men and women showed similar amplitude of serum 25(OH)D.
Studies from countries at the same latitude as Romania showed results close to ours. In a study on 206 Hungarian men [13], the peak serum 25(OH)D was found during summer (33 ng/mL as compared to 26.3 ng/mL in September in our study) and the nadir levels during winter (23 ng/mL as compared to 15.1 ng/mL in April in our study). As both levels are higher than in the Romanian population, we can presume a more intense vitamin D supplementation. Hungarian women [14] showed a similar seasonal variation with the lowest level of 17 ng/mL in the spring (compared to 13 ng/mL in March in our study) and the highest level of 21 ng/mL in the autumn (compared to 23.6 ng/mL in August in our study) in a study evaluating 319 women. Interestingly, although the spring-autumn variation is preserved in these Hungarian postmenopausal women, the amplitude of the variation is much smaller than in our population. In a large Swiss study [15], both the methodology and results closer resembled our results on Romanian population. That study enrolled 1682 patients from general practice. At the end of summer (September) and at the end of winter (March), the median serum 25(OH)D were 21.3 and 16.6 ng/mL, very similar to our results (24.1 and 13.5 ng/mL). Unfortunately, the Swiss study did not provided data on age or sex subgroups.
The seasonal variation of serum 25OHD concentrations is not limited to southeastern Europe, and it has been previously confirmed in many studies, all across the world [10, 16, 17]. Even more, the seasonal variation has also been noted in subjects taking cholecalciferol supplements [18].
Men had constantly higher levels of 25(OH)D compared to females, a result consistent with most of those reported in the literature, on population of all ages [19,20,21,22,23,24]. However, there is no accepted explanation for this variation. Even more, in certain populations, the relation is opposite with VD deficiency being more prevalent in men [25]. As vitamin D is fat soluble and stored in the adipose tissue [26], these gender differences could be related to gender-related variations in body fat content.
Young subjects consistently have higher serum 25OHD concentrations compared to elderly subjects. This has been widely recognized previously [27]. The explanation is related to the decreased sun exposure in the elderly and the progressive impairment with age of the skin ability to produce vitamin D3 [28].
The other study on serum 25(OH)D in Romanian population [9] included only postmenopausal women and did not present any date on seasonal variation. However, the distribution of serum concentrations was similar to ours. In the study of Grigorie et al. [9], the median, year-round, serum 25(OH)D, was 16.76 ng/mL, slightly lower than 18.6 ng/mL in our population that also included men and younger females. The prevalence of vitamin D deficiency (22.2%) was intermediate between our data from March (29.9%) and August (5.6%) in the female population.
The biggest limitation is that the study population is not a general population sample of Romania. Our endocrinology center receives patients from all counties but patients from Southern Romania (between 44°N and 46°N) are overrepresented. Also, the oral intake of vitamin D (dietary or medication) was not assessed in our study. However, we tried to minimize this bias by selecting only the first value (in case of serial measurements) and by excluding from the study the patients with a diagnosis of low bone mass (osteopenia or osteoporosis) who are prone to take drugs containing vitamin D. As shown in Table 1, patients with a diagnosis of low bone mass had significantly higher serum 25(OH)D levels than patients without such a diagnosis, suggesting vitamin D supplementation. Unfortunately, we did not have access to any data concerning body mass index, comorbidities, medication, and tobacco or alcohol use which could potentially influence vitamin D status. However, there is no reason to suspect that our sample differs significantly from the Romanian population in this respect. Finally, the number of subjects differs widely by month (414 in August vs. 1011 in October). As such, the vitamin D status of our population may not be extrapolated to the whole Romanian population.
The main advantage of our approach is the very large number of subjects. This allowed us to do multiple subgroup analysis (by month, age, or sex) with sufficient statistical power. Although a hospital-based population does not mirror the general population in terms of vitamin D status, we can reasonably presume that seasonal variation of serum 25(OH)D is preserved. The seasonal variation was robust for all age or sex subgroups, adding strength to the data. All age and sex subgroups had similar distribution over months (e.g., the percent of a subgroup from the total number of patients was constant every month).
In conclusion, our data show a strong seasonal variation of serum 25(OH)D in Romanian population similar to other countries of the same latitude. Also, the prevalence of vitamin D deficiency is high even at the end of summer, particularly in the elderly.
References
Holick MF (2007) Vitamin D deficiency. N Engl J Med 357(3):266–281. https://doi.org/10.1056/NEJMra070553
Burgaz A, Akesson A, Oster A, Michaëlsson K, Wolk A (2007) Associations of diet, supplement use, and ultraviolet B radiation exposure with vitamin D status in Swedish women during winter. Am J Clin Nutr 86(5):1399–1404
Hintzpeter B, Mensink GB, Thierfelder W et al (2008) Vitamin D status and health correlates among German adults. Eur J Clin Nutr 62(9):1079–1089. https://doi.org/10.1038/sj.ejcn.1602825
Hypponen E, Power C (2007) Hypovitaminosis D in British adults at age 45 y: nationwide cohort study of dietary and lifestyle predictors. Am J Clin Nutr 85(3):860–868
Andersen R, Molgaard C, Skovgaard LT et al (2005) Teenage girls and elderly women living in northern Europe have low winter vitamin D status. Eur J Clin Nutr 59(4):533–541. https://doi.org/10.1038/sj.ejcn.1602108
van der Wielen RP, Lowik MR, van den Berg H et al (1995) Serum vitamin D concentrations among elderly people in Europe. Lancet 346(8969):207–210. https://doi.org/10.1016/S0140-6736(95)91266-5
Capatina C, Carsote M, Caragheorgheopol A, Poiana C, Berteanu M (2014) Vitamin d deficiency in postmenopausal women—biological correlates. Maedica (Buchar) 9(4):316–322
Chirita-Emandi A, Socolov D, Haivas C, Calapiș A, Gheorghiu C, Puiu M (2015) Vitamin D status: a different story in the very young versus the very old Romanian patients. PLoS One 10(5):e0128010. https://doi.org/10.1371/journal.pone.0128010
Grigorie D, Sucaliuc A, Ivan M, Neacsu E, Popa O, Diaconescu A (2008) High prevalence of vitamin D deficiency in 1048 Romanian women with postmenopausal osteoporosis. Acta Endocrinol (Buc) 4(1):33–45. https://doi.org/10.4183/aeb.2008.33
Lips P, Duong T, Oleksik A, Black D, Cummings S, Cox D, Nickelsen T (2001) A global study of vitamin D status and parathyroid function in postmenopausal women with osteoporosis: baseline data from the multiple outcomes of raloxifene evaluation clinical trial. J Clin Endocrinol Metab 86(3):1212–1221. https://doi.org/10.1210/jcem.86.3.7327
McKenna MJ (1992) Differences in vitamin D status between countries in young adults and the elderly. Am J Med 93(1):69–77. https://doi.org/10.1016/0002-9343(92)90682-2
Spiro A, Buttriss JL (2014) Vitamin D: an overview of vitamin D status and intake in Europe. Nutr Bull 39(4):322–350. https://doi.org/10.1111/nbu.12108
Bhattoa HP, Nagy E, More C, Kappelmayer J, Balogh A, Kalina E, Antal-Szalmas P (2013) Prevalence and seasonal variation of hypovitaminosis D and its relationship to bone metabolism in healthy Hungarian men over 50 years of age: the HunMen Study. Osteoporos Int 24(1):179–186. https://doi.org/10.1007/s00198-012-1920-2
Bhattoa HP, Bettembuk P, Ganacharya S, Balogh A (2004) Prevalence and seasonal variation of hypovitaminosis D and its relationship to bone metabolism in community dwelling postmenopausal Hungarian women. Osteoporos Int 15(6):447–451. https://doi.org/10.1007/s00198-003-1566-1
Merlo C, Trummler M, Essig S, Zeller A (2015) Vitamin D deficiency in unselected patients from Swiss primary care: a cross-sectional study in two seasons. PLoS One 10(9):e0138613. https://doi.org/10.1371/journal.pone.0138613
Papapetrou PD, Triantaphyllopoulou M, Karga H, Zagarelos P, Aloumanis K, Kostakioti E, Vaiopoulos G (2007) Vitamin D deficiency in the elderly in Athens, Greece. J Bone Miner Metab 25(3):198–203. https://doi.org/10.1007/s00774-006-0746-4
Mezquita-Raya P, Munoz-Torres M, Luna JD et al (2001) Relation between vitamin D insufficiency, bone density, and bone metabolism in healthy postmenopausal women. J Bone Miner Res 16(8):1408–1415. https://doi.org/10.1359/jbmr.2001.16.8.1408
Holick MF, Chen TC, Lu Z, Sauter E (2007) Vitamin D and skin physiology: a D-lightful story. J Bone Miner Res 22(Suppl 2):V28–V33. https://doi.org/10.1359/jbmr.07s211
Atli T, Gullu S, Uysal AR, Erdogan G (2005) The prevalence of vitamin D deficiency and effects of ultraviolet light on vitamin D levels in elderly Turkish population. Arch Gerontol Geriatr 40(1):53–60. https://doi.org/10.1016/j.archger.2004.05.006
Gannage-Yared MH, Chemali R, Yaacoub N et al (2000) Hypovitaminosis D in a sunny country: relation to lifestyle and bone markers. J Bone Miner Res 15(9):1856–1862. https://doi.org/10.1359/jbmr.2000.15.9.1856
El-Hajj Fuleihan G, Nabulsi M, Choucair M et al (2001) Hypovitaminosis D in healthy schoolchildren. Pediatrics 107(4):E53. https://doi.org/10.1542/peds.107.4.e53
Krieg MA, Cornuz J, Jacquet AF, Thiébaud D, Burckhardt P (1998) Influence of anthropometric parameters and biochemical markers of bone metabolism on quantitative ultrasound of bone in the institutionalized elderly. Osteoporos Int 8(2):115–120. https://doi.org/10.1007/BF02672506
Semba RD, Houston DK, Bandinelli S, Sun K, Cherubini A, Cappola AR, Guralnik JM, Ferrucci L (2010) Relationship of 25-hydroxyvitamin D with all-cause and cardiovascular disease mortality in older community-dwelling adults. Eur J Clin Nutr 64(2):203–209. https://doi.org/10.1038/ejcn.2009.140
Tolppanen AM, Fraser A, Fraser WD, Lawlor DA (2012) Risk factors for variation in 25-hydroxyvitamin D(3) and D(2) concentrations and vitamin D deficiency in children. J Clin Endocrinol Metab 97(4):1202–1210. https://doi.org/10.1210/jc.2011-2516
Lagunova Z, Porojnicu AC, Lindberg F, Hexeberg S, Moan J (2009) The dependency of vitamin D status on body mass index, gender, age and season. Anticancer Res 29(9):3713–3720
Wortsman J, Matsuoka LY, Chen TC, Lu Z, Holick MF (2000) Decreased bioavailability of vitamin D in obesity. Am J Clin Nutr 72(3):690–693
Lester E, Skinner RK, Wills MR (1977) Seasonal variation in serum-25-hydroxyvitamin-D in the elderly in Britain. Lancet 1(8019):979–980
MacLaughlin J, Holick MF (1985) Aging decreases the capacity of human skin to produce vitamin D3. J Clin Invest 76(4):1536–1538. https://doi.org/10.1172/JCI112134
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Electronic supplementary material
ESM 1
Serum 25(OH)D concentration by month in 8024 subjects without a diagnosis of low bone mass (white bars) and 6028 subjects with a diagnosis of low bone mass (gray bars). The boxes represent the median (25, 75 percentile). The whiskers and small squares below and above represent 10th and 90th percentiles and 5th and 95th percentiles respectively. (GIF 39 kb)
ESM 2
Serum 25(OH)D concentration by age group and month in winter and spring (top panel) or summer and autumn (lower panel). White bars = 0–10 years; gray bars = 11–20 years; rightwards dashed gray bars = 21–40 years; leftwards dashed gray bars = 41–65 years; dotted bars= > 65 years of age (GIF 46 kb)
(GIF 47 kb)
Rights and permissions
About this article
Cite this article
Niculescu, D.A., Capatina, C.A.M., Dusceac, R. et al. Seasonal variation of serum vitamin D levels in Romania. Arch Osteoporos 12, 113 (2017). https://doi.org/10.1007/s11657-017-0407-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-017-0407-3