Abstract
Purpose
Bioenergetic models are used in cycling to estimate the acute physiological response in terms of oxygen consumption (\({\dot{\text{V}}}\)O2) and lactate concentration ([La]). First, our aim is to review the bioenergetic modelling literature, presenting historical evolution of concepts, techniques and related limitations. Second, our aim is to discuss how and where new approaches can stem and evolve.
Methods
This is a narrative review, where different modelling solutions are compared and qualitatively discussed. First, the principal features of the \({\dot{\text{V}}}\)O2 and [La] kinetics are presented, and then the models available in the literature are compared in light of what aspects of the physiological responses they can describe.
Results
Currently, models can detect most features of \({\dot{\text{V}}}\)O2 and [La] kinetics, but no single existing model appears appropriate for every exercising conditions. Limitations hindering the creation of an ultimate model are: the large variability of an exercise, the required mathematical complexity, and lack of reliable physiological data. To overcome these issues, new modelling solutions are being explored in the emerging AI technologies. However, in AI-models, parameters do not have direct physiological meaning and require massive amounts of experimental data for parameter calibration.
Conclusions
Despite the great efforts made by model developers and exercise physiologists, universal modelling solutions for the variety of potential exercising conditions remain unavailable. At present, further research is needed to assess the accuracy and predictive power of AI models to move the method forward in our field, as it is being done so in many others.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Cycling exercise typically involves frequent changes in external power output and an associated metabolic power demand (MPD) that stresses various energy pathways for energy production [1]. The relative contribution of each key energy system primarily is dictated by the exercise intensity, by the responses of the systems involved in metabolic control [2], exercise duration and endogenous carbohydrate stores availability [3, 4].
Following a step change in power output in the moderate intensity domain, at steady state [5] a fully and evenly aerobic condition can be reached [6] after a brief transitory phase that relies on anaerobic pathways. Energy requirements are met through carbohydrate and lipid oxidation, without net accumulation of blood lactate [7] (e.g., easy riding in the peloton [8]). As the exercise intensity increases, the utilization of lipids is thought to be progressively reduced and the contribution of glycogen breakdown, that ends in lactate production [9], becomes more and more relevant. Above certain individualized intensities [10], the exercise becomes unevenly aerobic [6] and more carbohydrate-dependent [9] and the exercise cannot be maintained indefinitely (e.g., 40 km time trials and high mountain ascents [11]). This threshold is well-explained by studies [12] that used the maximal lactate steady state (MLSS) methodology (considered the gold standard for evaluating this threshold), but also other methods have been used to demarcate the boundary between sustainable-unsustainable exercises (e.g., the critical power CP [13]). At extreme intensities, which are characterised by a limited sustainability (e.g., sprints [14]), intramuscular high-energy phosphates (ATP and PCr) and the anaerobic glycogen breakdown provide the predominant energy required [15] while the contribution of the aerobic metabolism slowly rises over time and becomes essential in reconstituting the depleted energy stores, allowing the repetition of intermittent extreme exercise bouts [16].
A single exercise session triggers a specific acute physiological response that, if repeated, promotes a specific chronic adaptation [17]. Understanding how internal human energy is provisioned to create external power can assist us to understand topics as broad as performance analysis, exercise prescription and exercise physiology. Exercise intensity (i.e., the internal metabolic power demand), can be linked to an associated external power output (P) during cycling that is derived across a cadence (ω) distribution by means of the cycling efficiency [18]. The processes enabling this power output evokes an acute physiological response, typically measured by the oxygen consumption (\({\dot{\text{V}}}\)O2) and the lactate concentration [La], proxies of the oxidative and glycolytic components of the metabolic response, respectively. \({\dot{\text{V}}}\)O2 mirrors oxygen consumption at the level of the working muscles, considered as the aerobic contribution to the exercise [19], while [La] can be used to express the balance between the rate of lactate production and removal [6], and indirectly, the involvement of anaerobic metabolism [20]. The variation of muscular PCr concentrations, can only be assessed with expensive and invasive techniques like the standard biopsy or by 31P magnetic resonance spectroscopy [21].
At steady-state [5], the energy turn-over can be expressed by means of the formula [22]:
where d/dt is the time derivative operator and the three constants (a, b and c) corresponds to the moles of ATP resynthesized by a mole PCr, lactate or oxygen respectively [5]. To use this equation in dynamic conditions (i.e., with a time-varying MPD) we need to include and solve the differential equations for the \({\dot{\text{V}}}\)O2, [La] and PCr responses in a single system of equations, i.e.; a dynamic model. This is made particularly challenging by the different time constants characterising these responses [23], especially in the recovery phase [24].
The goal of a dynamic model is, therefore, to mathematically describe the dynamic behaviour of \({\dot{\text{V}}}\)O2, [La] and PCr. Accurate and reliable models can be highly useful in a number of applications, especially when physiological data collection is problematic. Particularly, \({\dot{\text{V}}}\)O2 and [La] values are used in training prescription [25], to assess the aerobic performance [26] and the training status of endurance athletes [27]. The PCr response to exercise is often neglected mainly because: (1) direct information on PCr is expensive and invasive and (2) PCr contribution during prolonged activities is limited.
Existing models of cycling \({\dot{\text{V}}}\)O2 and [La] are all abstracted using mathematical “hand-written” formulations. The great advantage of this approach is that every equation of the model has a physical/physiological meaning: model developers try to incorporate all the relevant data, and include relationships between relevant variables [e.g., height, age, weight, gender, heart rate (HR) or arterial blood saturation (SaO2)] as a function of the exertion. However, with this approach, we only use few variables in our models because the more variables we add, the more difficult it becomes to make sense of the relationships between them and to express these relationships mathematically. Despite the great work done by sport scientists, exercise physiologists and model developers in the last decades, today there are no modelling solutions that can work for every exercising condition.
The purpose of this narrative review is to provide to model users (typically exercise physiologists or sport scientists) and model developers (typically engineers or mathematicians) a common ground to meet on. We everyday experience a lack of communication between these groups, which may be hindering the development of new and more sophisticated models. However, it would be impossible to extensively review both the entire exercise physiology literature and the different kind of mathematical tools that model developers have in their arsenal. We limit our review to linear dynamic models of \({\dot{\text{V}}}\)O2 and [La], i.e., models that account for time-dependent changes in the metabolic power requirements and that can be written with linear differential operators.
Having said this, first we briefly review the bioenergetic modelling literature; we highlight the limitations that exist, and ultimately the roadblock that has been reached in the area. Second, we introduce potential new solutions to existing limitations on a critical base. The review has been written and organised so to be understandable by both model users and developers: this first introductory section sets the scene on the physiological response to cycling exercise; the second section describes the main features of the \({\dot{\text{V}}}\)O2 and the [La] kinetics; the third section presents the currently available mathematical formulations, the techniques adopted to describe the \({\dot{\text{V}}}\)O2 and the [La] kinetics and the related limitations; the fourth section presents where and how new possible modelling solutions can evolve and how we can bridge the historical results with the modern techniques; the fifth and conclusive section sums up the implications of our arguments.
Bioenergetic modelling
Introduction
Bioenergetic models are used to estimate \({\dot{\text{V}}}\)O2 and [La] responses to cycling exercise. With such models, measurable mechanical variables (typically cadence and power output) represent the system’s input, allowing an estimation of the required metabolic power demand provided by the athlete. Measurable physiological variables (typically \({\dot{\text{V}}}\)O2 and [La]) are considered the system’s outputs that describe the metabolic control of the workload.
Within the realm of cycling science, estimations of the bioenergetic models are used for mainly two distinct purposes: performance analysis and data generation. Performance analysis is usually conducted in a post-process phase and is used, for example, to assess the effect of a treatment on a model parameter or to uncover the most influential parameter [28, 29]. Models adopted in performance analysis should always reflect the system’s physiological features and be consistent with the purpose intended for the model. The goal of data generation initiatives is to create simulated data in a pure predictive fashion. For optimizing pacing strategy, for example, quantities like anaerobic source depletion [30] are commonly adopted, with the goal of finding the best power distribution that minimizes the total race time. Alternatively, training design with bioenergetic modelling aims to find the ideal training intensity distribution that can trigger the optimal training-induced adaptation [31]. Data generation initiatives also include innovative studies attempting to predict physiological variables on a real-time basis using easy-to-obtain information [32]. Two major limitations to the development of accurate and reliable bioenergetic models include data quality and mathematical complexity. Both limitations are direct consequences of the variability of the measured physiological variables alongside an individual physiological response. While external mechanical variables (e.g., speed, power, and cadence) are easily collected in both indoor and outdoor conditions [33], internal physiological variables (e.g., \({\dot{\text{V}}}\)O2, [La]) are more difficult to measure. Experimental data collection of \({\dot{\text{V}}}\)O2 and [La] require highly sophisticated and expensive hardware [34] or invasive techniques [35]. Additionally, such data are always affected by biological noise that makes it harder to recognize distinct characteristics of the response, thereby impacting model parameter choice [36]. Acute physiological responses are highly complex and variable within and between individuals, due to a number of factors that include pedal frequency [37] and prior exercise intensity [38, 39].
Great contributions to our understanding of \({\dot{\text{V}}}\)O2 kinetics have been made by Wasserman and Whipp [40], Barstow [41], Poole and Jones (with a book entirely devoted to \({\dot{\text{V}}}\)O2 kinetics [42]). In the field of exercising [La], Stegmann [43], Brooks [44], Gladden [45] and Beneke [35] progressively refined our understanding of the area. In the field of the energetics of the muscular exercise and regulatory mechanisms at whole body level, fundamental contribution has been given by scientists of: the “school of Milan” (e.g., di Prampero and Margaria [22, 46] with a first systematic discussion on the oxygen deficit, Cerretelli [47] with the introduction of the early lactate concept, Lador [48] with the application of the double-exponential model to the study of the cardiac output kinetics and Ferretti [49] with a recent book summarizing the latest advances in the field); the “Canadian school” (e.g., Hughson [50] with the first studies investigating \({\dot{\text{V}}}\)O2 kinetics under lower body negative pressure, Faisal et al. [51] for their analyses on the role of the primary exercise on \({\dot{\text{V}}}\)O2 kinetics and lately Beltrame [52] for applying new AI technologies to the problem of modelling exercising \({\dot{\text{V}}}\)O2) and the “British School” (e.g., Rossiter et al. [53] for their research on PCr kinetics and Binzoni [54] for bridging with the “School of Milan”).
Exercising \({\dot{\text{V}}}\)O2 and [La] have been well characterized for on-set [55] and off-set [56] of step exercise, during recovery after exercise [57, 58], for impulsive/pseudo-random/sinusoidal force functions [59,60,61,62], for intermittent exercises [63, 64], for decremental [65] and incremental [66, 67] ramp and graded exercises [68, 69]. A mathematical abstraction of these complex behaviours requires a large number of “what if” scenarios, conditions and exceptions to be expressed in mathematical form. Typically, hand-written formulas are included and coded with the logic of Box 1.
Mathematicians and model developers have the ability to use hand-written formula by choosing from a variety of functions (e.g., exponential functions [70], delayed functions [71] and sigmoidal functions [72]) that can lead to concise and informative mathematical forms. While bioenergetic models have grown in size and complexity over the last decades, solutions for the variety of potential exercising conditions encountered remains unavailable.
Important features of \({\dot{\text{V}}}\)O2 and [La] kinetics to be considered in the development of bioenergetic models are presented separately in the next sessions for an ideal fatigue-free state. This is because a multitude of models available in the literature only focus on one of these variables at time. However, in a model combining both the \({\dot{\text{V}}}\)O2 and [La] responses, the interplay between the aerobic and the anaerobic metabolism and the role of the early blood lactate or the lactate energy equivalent should be considered. The mechanical efficiency and the internal power contributions are also presented.
\({\dot{\text{V}}}\)O2 kinetics
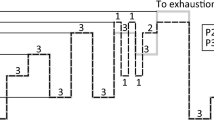
Schematic representations of \({\dot{\text{V}}}\)O2 kinetics have been reported for exercise intensity domains of relevance (Fig. 1) based on ventilatory thresholds VTI (or GET) and VTII (or RCP) [1] (i.e., the intensity thresholds that we can identify with breakpoints in the ventilatory variables during a cardiopulmonary exercising test). These include: moderate (power output lies below the first ventilatory threshold or GET, i.e., P < PVTI), heavy (power output lies between the first and the second ventilatory thresholds or RCP, i.e., PVTI < P < PVTII), severe (power output lies between the second ventilatory threshold or RCP and the minimal power eliciting the maximal oxygen consumption during a cardiopulmonary exercising test, i.e., PVTII < P < PVO2max) [73] and extreme (power output is greater than the minimal power eliciting the maximal oxygen consumption during a cardiopulmonary exercising test). In the case of moderate exercise, a steady state of \({\dot{\text{V}}}\)O2 is attained and is equal to the nominal one (Fig. 1a) as predicted from the MPD/P relationship (see “[La] kinetics” section). Even in the moderate domain, on- and off-kinetics depend on the power output baseline [74], and they show good symmetry (Fig. 1b) [75]. For heavy exercise, still a steady-state is attained and this “excess” \({\dot{\text{V}}}\)O2 (Fig. 1c) can be seen as a change in the MPD/P relationship (see “[La] kinetics” section). In this intensity domain, the on-kinetics is slower than the off-kinetics (Fig. 1d) [75]. For severe exercises, no steady state is shown (Fig. 1e), and the development of a slow component can raise \({\dot{\text{V}}}\)O2 towards \({\dot{\text{V}}}\)O2max [76] without exceeding it [77]. However, exhaustion might occur before \({\dot{\text{V}}}\)O2 could reach the maximal values [78]. \({\dot{\text{V}}}\)O2 can rise, although less significantly, during prolonged heavy exercise, and some authors prefer to refer to this as drift (Fig. 1i) [79]. In the severe intensity domain, the augmented \({\dot{\text{V}}}\)O2 at constant power output can be viewed as a lowered efficiency due to progressive fatigue development and decreased muscle efficiency [80]. When severe exercise ends, \({\dot{\text{V}}}\)O2 drops to a new steady state value dictated by the new reduced intensity with slower kinetics, determining the excess post-exercise oxygen consumption (EPOC) (Fig. 1f) [81]. If the on-kinetics of \({\dot{\text{V}}}\)O2 is dissected, two different phases that correspond to distinct physiological events are evident: the cardiopulmonary phase [or phase-I (Fig. 1g), evident from tON to TD1], and the dominant phase [or phase-II (Fig. 1h), evident after tON + TD1]. In the case of extreme exercise intensities, the \({\dot{\text{V}}}\)O2 response saturates at a maximal value defined by \({\dot{\text{V}}}\)O2max (Fig. 1k) in a time that decreases as the exercise intensity increases [82] (Fig. 1j). However, \({\dot{\text{V}}}\)O2max might not be reached for extreme exercise intensities, especially when power exceeds the ~ 130% of PVO2max [83].
Schematic representations of \({\dot{\mathbf{V}}}\)O2 behaviour in response to a single bout of constant work rate in different intensity domains. For moderate exercises, a steady state is attained and it equals the nominal one (a). The on- and off-kinetics are similar (b). For heavy exercises, a steady state is attained that is greater than the nominal one (c) and the off-kinetics appear to be faster than the on-kinetics, albeit \({\dot{\text{V}}}\)O2 does not return to basal values immediately (d). For severe exercise, no steady state is shown (e) and the off-kinetic is very slow (f) as determined by an additional oxygen consumption (i.e., EPOC). More detailed on-kinetics involves a first phase in which there is an abrupt rise in \({\dot{\mathbf{V}}}\)O2, called phase-I or the cardiopulmonary phase (g) and a phase-II, i.e., the dominant kinetics (h). A drift in the oxygen consumption is reported for prolonged submaximal exercises as a result of rising body temperature (i). In extreme exercise, the time to reach \({\dot{\mathbf{V}}}\)O2max decreases as the exercise intensity increases (j) and the value of \({\dot{\mathbf{V}}}\)O2max is attained and maintained (k) until exhaustion occurs or exercise workload drops
[La] kinetics
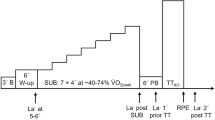
A schematic example of the behaviour of the [La] is reported in Fig. 2 for different exercise intensity domains of interest based on lactate thresholds LTI and LTII (or MLSS) [1] (i.e., the intensity thresholds that we can identify with variations in [La] during a cardiopulmonary exercising test). These include: moderate (below the first lactate threshold, i.e., P < PLTI), heavy (between the first and the second lactate thresholds or CP/MLSS, i.e., PLTI < P < PLTII) and severe (above the second lactate thresholds or CP/MLSS, i.e., P > PLTII). In the case of moderate exercise, lactate clearance exceeds lactate accumulation rate, so any change in lactate concentration is restored and [La] is kept constant at the basal value (Fig. 2a). In the case of heavy exercise intensities, lactate production equals lactate removal and [La] typically stabilises between the 2 and 4 mM [43]. Here, the upper boundary corresponds to the maximal lactate steady state (Fig. 2d). It should, however, be noted that maximal lactate steady state tests have great variability [43] in terms of [La] but they are very repeatable in terms of power output [84], meaning that any correspondence with fixed [La] values of 2 and 4 mM is a simplification of the likely response. In the case of severe exercise, the [La] continues increasing (Fig. 2e) and whole body energy turnover is considerably larger than that indicated by the \({\dot{\text{V}}}\)O2 by an amount that is proportional to the net rate of lactate turnover [6]. Generally, [La] shows a delayed response to the power output and this is particularly evident after severe exercise (Fig. 2f) [85], where a prominent fraction of the total power is in fact provided by the anaerobic metabolism [6]. At the onset of the exercise (tON), regardless of the workload, an increase in the lactate concentration is observed [47]: the early lactate response (Fig. 2a) that gives an overshoot at the start of heavy (Fig. 2c) or severe exercises. When the exercise ends (tOFF), the [La] slowly decreases to the basal value with exponential behaviour (Fig. 2b) [58] with a slow time constant ([La] returns to basal values after ~ 20 min).
Schematic representations of blood lactate concentration ([La]) behaviours in response to different exercise intensities, beginning in tON and ending in tOFF. At every exercise intensity, [La] shows a delayed response to the exercise. For moderate exercises, after a small peak due to early production (a), lactate returns to the basal value LB exponentially (b). For heavy exercise, after a brief overshoot due to early production (c), a steady state is attained above the basal value (d). For severe exercise, the lactate continues increasing (e) and a delayed peak is evident when exercise ends (f). Irrespective of the exercise intensity, lactate returns to its basal value in an exponential fashion (b)
Including the ‘internal power’ and the mechanical efficiency
Internal power (IP) in cycling has three main contributors [86]—(1) the variation of the kinetic energy of the body, (2) the co-activation of agonist and antagonistic muscles and (3) the frictional/viscous resistance of joints. Observational [87] and theoretical [86] studies report that IP increases linearly with the mass (not the weight [88]) and the moment of inertia of the moving segments and as a power function of ω. However, if a higher ω requires an additional metabolic power, it can be assumed that the faster cadence is detrimental to efficiency, which is not necessarily the case [89]. This of course is a simplification of the issue, but there are different alternatives [90] and model developers should also consider that cadence selection has a large number of biomechanical [91] and metabolic determinants [37]. With the inclusion of the contribution of the internal power, the linear formula for the metabolic power demand becomes:
where k is a compound term for the inertial parameters and γ is a constant parameter (see, e.g., [87]) and MPD0 is the metabolic power demand at rest (i.e., generally assumed equal to the resting \({\dot{\text{V}}}\)O2 value, ≈ 3.5 ml kg−1 min−1 [92]). Typical values of 1/G (Δ efficiency) range from between 18 and 25% [18] (values are close to the efficiency of concentric muscle contraction). G is also known as the “gain” and is usually assumed to be ≈ 10 ml O2 min−1 W−1 for moderate exercise intensities and it gets progressively higher at heavy and severe intensities [75, 78] (≈ 13 ml O2 min−1 W−1). Here, the augmented gain can lead to a \({\dot{\text{V}}}\)O2 excess as high as 230 ml O2 min−1 dependent on the methodology adopted [93] for the calculation. However, it has been suggested that the value of the oxygen gain depends on the exercise intensity domain and the baseline exercise intensity and that it progressively changes with the power output [94].
Existing modelling solutions
\({\dot{\text{V}}}\)O2 models
Models for \({\dot{\text{V}}}\)O2 kinetics have been developed throughout a wide range of different formulations [42]. The simplest and most common form is the first-order form:
where τ is the characteristic time of \({\dot{\text{V}}}\)O2 kinetics (is recognized as one key parameter of aerobic fitness) and \({\dot{\text{V}}}\)O2p is the value of the target \({\dot{\text{V}}}\)O2 (typically inferred from MPD). The solution of the first-order equation in response to a step-input is:
This is why this model is often referred to as a single-phase or mono-exponential model. Where A is the steady state amplitude of the response, calculated as the difference between the target \({\dot{\text{V}}}\)O2p and the baseline \({\dot{\text{V}}}\)O20. First-order \({\dot{\text{V}}}\)O2 models can be used to assess the exercise-induced improvements [95], intermittent training [96], performance [82] and the oxygen debt [97].
A double-exponential (double-phase) model puts together the phase-I and phase-II of the \({\dot{\text{V}}}\)O2 response (Fig. 1g, h). The sum of the time constant of phase-II and the time delay of phase-I has been termed the “effective” time-characteristic or the “mean response time” [98]. However, the choice of a double-exponential model over a single-exponential model is not only mathematical, but it has to be underpinned by important conceptual assumptions made a priori [48]. Phase-I is often assumed to be exponential [99] but there is still a lack of evidence that can support this assumption likely because the asymptote of phase-I is not approached before phase-I/II transition. However, single and double-phase models cannot precisely describe overall \({\dot{\text{V}}}\)O2 kinetics in heavy and severe intensity domains, because the additional \({\dot{\text{V}}}\)O2 requirements (see how in Fig. 1c, e the \({\dot{\text{V}}}\)O2 response deviates from the mono-exponential response). To improve the descriptions, Scheuermann et al. [70] included an additional exponential function in a three-phase model. The slow component of \({\dot{\text{V}}}\)O2 is included as an additional exponential term that switches on after an independent time delay with the onset of the exercise. During heavy exercises, an exponential term with an asymptote of constant amplitude seems adequate [75], but it has to point towards \({\dot{\text{V}}}\)O2max (or above) to correctly represent the \({\dot{\text{V}}}\)O2 slow component mechanism (Fig. 2e). It must also be specified that multi-phase models must include a limit to the maximal values of \({\dot{\text{V}}}\)O2 (Fig. 1k) before they can be adopted to predict exercising \({\dot{\text{V}}}\)O2 values.
Variations in the oxygen gain, kinetics (τ) or time-delays relationships with the power output have been reported by researchers implementing linear \({\dot{\text{V}}}\)O2 models [74, 94]. However, it is important to realise that including these non-linear relationships can lead to non-linear dynamic models of \({\dot{\text{V}}}\)O2 that usually require more complex protocols for their calibration (see “Limitations of hand-written models” section) and additional computational efforts. There are a number of modelling solutions equally justifiable for these function [i.e., G(P) and τ(P)], e.g., a linear profile or an asymptote of increasing amplitude [100] (that could be also used to model the \({\dot{\text{V}}}\)O2 drift mechanism depicted in Fig. 1i), an intensity-dependent [74] or a time-varying τ [101] (that can also be used to describe the fast \({\dot{\text{V}}}\)O2 response to extreme exercises depicted in Fig. 1j), the inclusion of τ as a function of lactate concentration [6] (e.g., τ = λ[La] + τ0 where λ is a constant parameter and τ0 is the characteristic time of \({\dot{\text{V}}}\)O2 kinetics at rest [47]). As comprehensively reviewed in [6], this increase in \({\dot{\text{V}}}\)O2 kinetics (τ) is associated with a greater reliance on anaerobic sources for energy provision. In this last case both \({\dot{\text{V}}}\)O2 and [La] equations must be solved together.
However, model developers must be warned: even if we choose simple linear functions for G(P) and τ(P) (e.g., τ = m · P + q, with m the slope of the linear relationship and q the intercept), we introduce additional parameters (i.e., m and q) that need to be computed. Of course a model that includes these non-linear behaviours is likely to bee more generalizable (it can adapt to a wider range of exercising conditions), but it is usually more difficult to understand and analyse and poses computational issues, like numerical instability. This trade-off between simplicity and accuracy of the model is well-known in the field of mathematical modelling, where between two models with equal predictive power, the simplest one is the most desirable. These concepts are discussed in more depth in “Limitations of hand-written models” section.
Stirling et al. [102] mathematically formulated the multi-phase models of \({\dot{\text{V}}}\)O2 kinetics in the differential form suitable for numerical integrators. They used the model to assess the effects of exercise training on \({\dot{\text{V}}}\)O2 kinetics [103]. However, the original version of the model has been designed for running, and requires amendments to meet cycling activity requirements [104]. Gonzalez and collaborators [71] included a curvilinear characteristics of the P/MPD relationship and the slow component is included as a secondary state variable triggered after an independent time delay and constantly superimposed to the primary component.
While modelling has made physiological inferences drawn from studies that have measured \({\dot{\text{V}}}\)O2 during exercise, it is still not completely clear whether each of the parameters in the models are system descriptors. Further work is required to allow more parameters to offer a testable and justifiable physiological meaning [105].
[La] models
The classic oxygen-debt and anaerobic threshold theories have been formed on the basis that lactate produced during exercise is a dead-end metabolite that relates to the absence of oxygen or to the development of fatigue [106]. Historically, this has resulted in the development of the so-called threshold models, where [La] is linked to \({\dot{\text{V}}}\)O2 by means of static relationships [107]. The more recent lactate shuttle theories [44] suggest that increasing lactate production with increasing power output does not appear to be strictly the result of inadequate oxygen supply [45] and should not be considered a threshold phenomenon [108]. Skeletal muscles and organs like kidney, liver and brain, can be considered as compartments, with the circulating blood providing the route to link the compartments together. Lactate exchange rate correlates with circulating lactate and follows normal concentration gradients. With this in mind, Zouloumian and Freund [58, 109, 110] proposed a model consisting of two communicating compartments—the working muscle and a lactate space. The Freund’s model can be expressed by means of a second order ordinary differential equation that can resemble the delayed behaviour and the overshoot typical of the [La] response to P (Fig. 2) [68]. It is commonly represented in the bi-exponential solution for constant inputs:
Here, A1, A2, A3, λ1, and λ2 are constants that depend on the system characteristics and on their initial conditions, where [La]bp is the lactate production term. Freund’s model has been applied to assess the exercise-induced improvements elicited by intermittent and continuous exercise training [111], to assess [La] kinetics after maximal short term exercise in different populations [112], after high intensity exercises [39] with active/passive recovery [113], to assess training-induced improvements in blood lactate removal ability [114]. For a short-term working period, Freund’s model can accurately describe the time-evolution of [La], suggesting that the model is qualitatively and quantitatively accurate [115]. It may be worth noting, however, that the original model was designed for recovery phases. In fact, in the original formulation, the lactate production term is neglected and a physiological meaning is given to the constant parameters: λ1 represents the lactate production ability and λ2 the lactate clearance ability [116] that depend on intensity and exercise duration. As suggested in the “\({\dot{\text{V}}}\)O2 models” section, it would be appropriate to include power output dependencies in the parameters of the model. This for example would mean to specify equations for λ1(P) and λ2(P), but this would require additional parameters and then more complex protocols for parameter calibration, with all the complications that follow (see “Limitations of hand-written models” section). As previously mentioned, non-linear and explicit time-dependent functions should be included cautiously.
Neglecting the lactate production term has important mathematical and conceptual consequences. First of all, the model becomes a static non-linear function and it loses its mechanical meaning. Second, we cannot apply this equation to the prediction of [La] in response to varying power output. Possible modelling solutions for the lactate production term can be proposed by means of the lactate energy equivalent [117]. Provided that any increase 1 mM in [La] is equivalent to the energy released by the consumption of ~ 3 ml O2 per kg of body mass [6], and neglecting the PCr contribution, we could relate the lactate production term with the difference between the metabolic power demand and \({\dot{\text{V}}}\)O2. However, this would not be sufficient to explain constant [La] values during heavy exercises (Fig. 2d) and it would require an additional power output dependent term.
Introducing a parameter for the lactate energy equivalent, Moxnes and Sandbakk proposed a mathematical model [72] for the lactate production and removal during whole-body exercise that can be adapted for cycling [118] and that can be used to estimate both \({\dot{\text{V}}}\)O2 and [La] kinetics with the same system of equations. The peculiarity of this model resides within the use of a sigmoidal function (tanh) that can be used to model smooth transitions between exercise intensities [119] and to describe the lactic acid buffering capacity. This model can be adopted to predict [La] in response to varying power output, but the large number of parameters with questionable physiological meaning impairs its predictive ability.
Limitations of hand-written models
The development of new and accurate hand-written models is mainly limited by the mathematical complexity required to describe the physiological phenomena of interest. It is difficult even to fathom how one would “hand-write” models for \({\dot{\text{V}}}\)O2 or [La] that can include the complicated internal power contribution, along with time-varying and intensity-dependent gains, τs and λs. The complexity of a model then depends upon the extension of the underlying physiological mechanism of interest: complex models are made to cover a large number of different kinetics (therefore, require a large number of parameters), whereas simpler models can only cover limited aspects of the physiological response (therefore, require a limited number of parameters).
The main problem is that every parameter of the model must be estimated within an acceptable level of confidence and that the number of parameters widens the confidence interval (CI) of the estimates (usually computed with statistical techniques like the bootstrap method [120]) and reduces the number of degrees of freedom (i.e., the difference between the number of data points and the number of parameters). Usually parameters are estimated by a model fitting procedure, i.e., an optimization problem consisting of minimizing an objective function by selecting the parameters from within an allowed set and computing the value of the objective function. The objective function typically consists in a sum of the squared differences between the simulated (the data generated with the model) and the experimental data points. The optimization problem is then solved by means of a nonlinear programming solver (e.g., implemented in the fminsearch [121] or particleswarm [122] functions in Matlab software). The higher the number of the parameters of the model, the less their effect on the fitting is discernible because the presence of local minima. In addition, if parameters are interdependent, the performances of the model (in terms of objective function) is poorly sensitive to variations in the parameters. In this case we say that the optimization problem is poorly conditioned and that the choice of the parameters is not making relevant difference on the value of the objective function [123]. As a consequence, the number of the parameters and the presence of interdependent parameters have a detrimental effect on the CI of the parameters. It can be assumed that, if the change in the error with experimental data is sufficient to offset the loss in degrees of freedom associated with the number of parameters, then the inclusion of a greater number of parameters is statistically justified [124]. However, if the CI of the estimates is larger than the smallest worthwhile changes [125] considered for those parameters, then the number of data points should be increased [126, 127]. Given that the testing protocols should elicit all the different behaviours that the model wants to describe, this requires long and articulated protocols for parameter estimation, so the number of individuals that can be tested is reduced if a more complex model is involved. Conversely, for simple models, a larger pool of individuals can be tested for different trials.
Besides mathematical complexity, data quality is also a limiting factor in the development of new bioenergetic models. High-quality data is characterized by a high signal to noise S/N ratio and by an adequate number of data points. Increasing the workload is the most straightforward method to increase the magnitude of the \({\dot{\text{V}}}\)O2 and [La] responses, but, as a consequence, the exercise becomes less sustainable. Noise reduction can be obtained by averaging over a different number of trials or by applying different smoothing techniques [128]. Most of the difficulties in modelling the different phases of \({\dot{\text{V}}}\)O2 kinetics lie behind the noise. In fact, the different phases of exercising \({\dot{\text{V}}}\)O2 only become discernible when they emerge from the noise of the background component of the fundamental profile. Noise affecting \({\dot{\text{V}}}\)O2 data can be approximated with a stochastic uncorrelated Gaussian noise of ≈ 120 ml O2 min−1 [13] (the assumption of normality has been questioned [129]) independent from the metabolic rate [36] while with respect to [La], uncertainty in measurements can range from ≈ 10 to ≈ 25% of the absolute value [130]. The number of data points is highly determined by the sampling rate (e.g., breath-to-breath for \({\dot{\text{V}}}\)O2 or blood sample time for [La]). The number of data points increases the number of degrees of freedom and narrows the CI of the parameter estimates. A possible strategy to increase \({\dot{\text{V}}}\)O2 data points and improve parameter estimation [131] is by interpolating at 1-s. This is reasonable for \({\dot{\text{V}}}\)O2 breath-to-breath signals, as the average respiratory frequency is ≈ 3.5 s for moderate exercises and the main dynamic of \({\dot{\text{V}}}\)O2 is ≈ 25–50 s [36]. In the case of [La], data points are typically spares (e.g., every minute [112], every 30 s [39], 10 s [58], up to real-time during bed rest [132]). During a classic Wingate test, [La] can rise as fast as ≈ 5 mM min−1 [133] indicating that exercising [La] could have surprisingly fast kinetics compatible to that of the \({\dot{\text{V}}}\)O2. This suggests that interpolation error may become relevant if sampling or interpolation time is not compatible with variations in the power output. Another method to improve the quality of the dataset is to exclude outliers or data points that are not likely to be part of the physiological response of interest and to avoid estimates influenced by unassociated portions of the response (e.g., see comparisons between [134, 38]).
To summarize: (1) simple models are best suited for performance analysis, as they have sufficient validity and sensitivity to identify worthwhile changes in the parameters characterizing the response, (2) complex models are best suited for data generation and simulations, as they cover a wide range of different scenarios, but the low confidence in parameter estimation impairs their predictive ability. The stages outlined in the conceptual scheme of Fig. 4 (left half) offer a structured format for the design of “hand-written” bioenergetic models.
Alternative approaches to bioenergetic modelling to predict cycling \({\dot{\text{V}}}\)O2 and [La]
If the understanding of the underlying physiological mechanism is not a priority, then we can rely on a different class of models, where a number of free-adjustable parameters without physiological meaning is included. With this approach, parameters are tuned until the model behaves with satisfactory accuracy like a specific instance of the real system. This is the approach of the Artificial Intelligence (AI) technologies, where the general goal is to recreate with computers the cognitive functions of the human mind, e.g., learning and problem solving [135]. AI algorithms, and in particular, machine learning and deep learning [136] have been adopted in vast and complicated conditions like health care [137], motor vehicle operations [138] and physical activity type classification [139]. In sports, machine learning have been used to treat cycle training data [140] or swimming performance data [141]. However, while AI is rapidly advancing forward in other fields, it is still in its infancy in cycling science [142].
In the realm of the AI-related technologies, neural networks are a biologically inspired programming paradigm which allows a computer to learn from experimental data through machine learning techniques [143]. At first, as with hand-written models, examples that specify the correct output (y) for a given set of inputs (x) are used to train the model, i.e., to adjust the parameters of the neural network. Second, the model is applied to process a data set where only the x variables are available to estimate the y. Training a neural network also involves an optimization problem that typically requires a high amount of numerical computation due to the great number of parameters. However, while in “hand-written” models it is often possible to retrieve an analytical formulation for the exact solution and speed up the optimization process with faster and robust solvers [123], machine learning algorithms usually refine the estimates of the solution via iterative processes. In fact, most learning algorithms are based on a class of optimization algorithms called stochastic gradient descent [144] that require massive amount of data and computational efforts [143].
Two common machine learning task are classification and regression. In a classifier, y variables take class labels, and the algorithm returns a membership identification probability. In a regressor, the y variables take continuous values and the program can learn to find the best relationship to represent the data. A classifier can be trained to provide the probability of \({\dot{\text{V}}}\)O2 and [La] across different training zones (Fig. 3a), whereas a regressor can be used to estimate exact values of \({\dot{\text{V}}}\)O2 and [La] (Fig. 3b) for a given power output.
Example of two different kinds of artificial neural networks that can be adopted in oxygen consumption \({\dot{\text{V}}}\)O2 and blood lactate concentration [La] prediction by means of easy-to-obtain inputs, such as: power (P), cadence (ω), heart rate (HR), respiratory frequency (RF), and oxygen saturation (SaO2). a A classifier is used to assign a label to the different data inputs. In the case of a classifier, the output variable takes a class label, such as the intensity domains (e.g., basal, moderate, hard, severe). b A regressor is used to estimate the exact value of \({\dot{\text{V}}}\)O2 and [La] from input data. Here, the output variables take continuous values
With a neural network approach, adding more measurable variables means supplying useful information to learn input/output relationships. Particularly, additional inputs can be useful if they: (1) bear strong relationships with the output variables, (2) are independent of each other, and (3) are always available. A number of easy-to-obtain inputs can be used to predict exercising \({\dot{\text{V}}}\)O2 and [La] data on a real time basis. For example, x datasets can be formed by the concomitant collection of power output, cadence, heart rate (HR), respiratory frequency (RF) and blood arterial saturation (SaO2). Additional information is provided by the simultaneous collection of different physiological markers, and used to predict a more accurate response of the target outputs, i.e., the \({\dot{\text{V}}}\)O2 and [La], so that:
Neural networks are made to create relationships between x and y, without a priori defined structures that automatically discern relevant and un-relevant correlations. The absence of these conditions makes neural networks more flexible than “hand-written” models and their application requires less knowledge about the system being modelled (i.e., in our case, \({\dot{\text{V}}}\)O2 and [La] kinetics). Furthermore, when a sufficient number of parameters are included, neural networks can approximate any function to the desired level of accuracy [145]. This allows the representation of very complex systems across a large variety of exercising conditions. If individual characteristics change, knowledge from historical data can be transferred and adapted to different individual characteristics (transfer learning) [146].
Particularly, among the many different classes of neural networks, great contribution can be provided by the so-called deep, convolutional and recurrent neural networks. Deep neural networks (as opposed to shallow neural networks) have multiple hidden layers of neurons between the input and output layers and have been used in the prediction of \({\dot{\text{V}}}\)O2 kinetics during treadmill walking [147], activities of daily living [139, 148] and cycling [149]. Convolutional neural networks are a special class of deep neural networks specifically designed for image recognition and classification [150]. They have been used to detect arrhythmias from ECG signals [151], but unfortunately they have not been already embedded in portable HR monitors that could be used in outdoor cycling [152]. Recurrent neural networks have the ability to include past values of the inputs and to keep memory of recent exercise history, thereby being particularly suited for time-series analysis [153]. Recurrent neural networks are currently applied in health care and diagnostics [154], in lactate threshold detection in running [155], in ventilatory thresholds estimation from cardiopulmonary exercising test data [156] and hold potential to be exploited for estimating cycling \({\dot{\text{V}}}\)O2.
AI techniques suffer from two major limitations, however. First, such models involve a “black box” approach [157] and the knowledge of the complex internal mathematical formulation is limited (the model is not self-explanatory, and hence the term “black box”). Second, the approach requires substantial training data to cover the variety of learnt processes, with sufficient data density needed for all exercising conditions (otherwise over-fitting may occur). To this extent, large datasets may be provided by the advent of the current and future wearable sensor industry, capable of non-invasively acquiring a large array of metabolic markers [158]. These large datasets, the high number of parameters, alongside the aforementioned condition-specific training process, will together require massive collaborative and computational efforts.
With AI, instead of writing a program by hand (Box 1) for each specific exercising condition (Figs. 1, 2), a computer program can be written so that the computer can determine by itself the relationships between parameters and inputs for different exercising conditions. When we compare against the complexity and work involved in writing out by hand all the possible combinations involved, AI solutions become extremely appealing and exciting. It should be clear, however, that with AI, we are replacing known knowledge and procedures with one single big one. Thus, it can become difficult if not impossible to elucidate underlying physiological mechanisms from a neural network, as relationships between variables may be hidden. We believe that a trade-off solution can be found in a hybrid approach (see, e.g., [159]), where physiology equations are combined together with neural networks. In these models it is possible to incorporate prior knowledge about the bioenergetics (e.g., the first-order model of \({\dot{\text{V}}}\)O2 kinetics) into a neural network that is used to estimate non-linear/time-dependent parameters [e.g., G(P), τ(P), λ1(P) or λ2(P) introduced in “\({\dot{\text{V}}}\)O2 models” and “[La] models” sections]. As a consequence, a hybrid model typically requires a significantly smaller number of experimental data and parameters than a pure neural network [160]. To the best of our knowledge, this promising approach has yet to be tested against \({\dot{\text{V}}}\)O2 and [La] data.
As it has been done for “hand-written” models, we offer the different critical stages in the creation of an AI-bioenergetic model in Fig. 4 (right side). It must, therefore, clear that there is not a superior approach, and that the best modelling solution depends on the purpose intended for the model. In an age where new communication devices are offering new ways to collect big amount of data [161], the collaborative work between sport scientists and mathematicians will be instrumental for enabling future solutions towards a more effective utilization of this data. Neural networks and machine learning [136], by their very nature of how they function, may in effect thrive on using a large volume of information in a productive and informative way: model developers have new and complementary tools in their toolboxes. Currently, however, extra work is needed to embed these innovative technologies and their hybrid combinations in portable devices and reach the athletes on the field.
Decision process and stages of the creation of a bioenergetic model following the “hand-written” model (left side) and the AI-model (e.g., neural network NN) (right-side) approaches. The best modelling solution depends on the purpose intended for the model and on data availability. In both cases, a trade-off exists between the mathematical complexity of the model and generalizability to a variety of exercising conditions
Conclusions
In the field of modelling \({\dot{\text{V}}}\)O2 and [La] responses to cycling exercise, despite the great amount of synergistic work done by model developers and exercise physiologists together, a single model or approach that can fit all the possible exercising conditions is currently unavailable. Limits to current model development lie behind the mathematical complexity required to describe the physiological responses to the exercise and to the high variability of the data characterizing this response. Due to a reduced number of parameters, “hand-written” models typically show low flexibility to adapt to changes in exercise conditions. Among the AI-related technologies, neural networks fit well in a framework where many physiological variables that are easy-to-obtain can be used to generate predictions of cycling \({\dot{\text{V}}}\)O2 and [La] in a great variety of exercise conditions. However, in neural networks, models parameters do not have direct physiological meaning and require big datasets for calibration. We hope that this review might be the start of a conversation that brings together these exciting technologies into exercise physiology and sport science understanding and application.
References
Faria EW, Parker DL, Faria IE (2005) The science of cycling: physiology and training: part 1. Sports Med Auckl NZ 35:285–312
Grassi B, Poole DC, Richardson RS et al (1996) Muscle O2 uptake kinetics in humans: implications for metabolic control. J Appl Physiol Bethesda Md 1985 80:988–998
Romijn JA, Coyle EF, Sidossis LS et al (1993) Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am J Physiol Endocrinol Metab 265:E380–E391. https://doi.org/10.1152/ajpendo.1993.265.3.E380
Nummela A, Rusko H (1995) Time course of anaerobic and aerobic energy expenditure during short-term exhaustive running in athletes. Int J Sports Med 16:522–527. https://doi.org/10.1055/s-2007-973048
Ferretti G, Fagoni N, Taboni A et al (2017) The physiology of submaximal exercise: the steady state concept. Respir Physiol Neurobiol 246:76–85. https://doi.org/10.1016/j.resp.2017.08.005
di Prampero PE, Ferretti G (1999) The energetics of anaerobic muscle metabolism: a reappraisal of older and recent concepts. Respir Physiol 118:103–115
Jones AM, Vanhatalo A (2017) The ‘critical power’ concept: applications to sports performance with a focus on intermittent high-intensity exercise. Sports Med 47:65–78. https://doi.org/10.1007/s40279-017-0688-0
Padilla S, Mujika I, Orbananos J et al (2001) Exercise intensity and load during mass-start stage races in professional road cycling. Med Sci Sports Exerc 33:796–802
Hawley JA, Leckey JJ (2015) Carbohydrate dependence during prolonged, intense endurance exercise. Sports Med 45:5–12. https://doi.org/10.1007/s40279-015-0400-1
Antonutto G, Di Prampero PE (1995) The concept of lactate threshold: a short review. J Sports Med Phys Fitness 35:6–12
Lucia A, Hoyos J, Chicharro JL (2001) Physiology of professional road cycling. Sports Med 31:325–337. https://doi.org/10.2165/00007256-200131050-00004
Faude O, Kindermann W, Meyer T (2009) Lactate threshold concepts: How valid are they? Sports Med 39:469–490. https://doi.org/10.2165/00007256-200939060-00003
Keir DA, Fontana FY, Robertson TC et al (2015) Exercise intensity thresholds: identifying the boundaries of sustainable performance. Med Sci Sports Exerc 47:1932–1940
Menaspà P, Quod M, Martin D et al (2015) Physical demands of sprinting in professional road cycling. Int J Sports Med 36:1058–1062
Sahlin K (2014) Muscle energetics during explosive activities and potential effects of nutrition and training. Sports Med 44:167–173. https://doi.org/10.1007/s40279-014-0256-9
Burnley M, Jones AM (2007) Oxygen uptake kinetics as a determinant of sports performance. Eur J Sport Sci 7:63–79. https://doi.org/10.1080/17461390701456148
Lundby C, Jacobs RA (2016) Adaptations of skeletal muscle mitochondria to exercise training: mitochondria and exercise. Exp Physiol 101:17–22. https://doi.org/10.1113/EP085319
Ettema G, Loras HW (2009) Efficiency in cycling: a review. Eur J Appl Physiol 106:1–14
Whipp BJ, Ward SA, Rossiter HB (2005) Pulmonary O2 uptake during exercise: conflating muscular and cardiovascular responses. Med Sci Sports Exerc 37:1574–1585
Brooks GA (2000) Intra-and extra-cellular lactate shuttles. Med Sci Sports Exerc 32:790–799
Rossiter HB, Ward SA, Kowalchuk JM et al (2002) Dynamic asymmetry of phosphocreatine concentration and O 2 uptake between the on- and off-transients of moderate- and high-intensity exercise in humans. J Physiol 541:991–1002. https://doi.org/10.1113/jphysiol.2001.012910
di Prampero PE (1981) Energetics of muscular exercise. Rev Physiol Biochem Pharmacol 89:143–222
Gastin PB (2001) Energy system interaction and relative contribution during maximal exercise. Sports Med 31:725–741. https://doi.org/10.2165/00007256-200131100-00003
Tomlin DL, Wenger HA (2001) The relationship between aerobic fitness and recovery from high intensity intermittent exercise. Sports Med 31:1–11. https://doi.org/10.2165/00007256-200131010-00001
Buchheit M, Laursen PB (2013) High-intensity interval training, solutions to the programming puzzle. Sports Med 43:313–338
Bosquet L, Léger L, Legros P (2002) Methods to determine aerobic endurance. Sports Med 32:675–700. https://doi.org/10.2165/00007256-200232110-00002
Billat LV (1996) Use of blood lactate measurements for prediction of exercise performance and for control of training: recommendations for long-distance running. Sports Med 22:157–175. https://doi.org/10.2165/00007256-199622030-00003
Olds T (2001) Modelling human locomotion. Sports Med 31:497–509
Jeukendrup AE, Martin J (2001) Improving cycling performance. Sports Med 31:559–569
Sundström D, Carlsson P, Tinnsten M (2014) Comparing bioenergetic models for the optimisation of pacing strategy in road cycling. Sports Eng 17:207–215. https://doi.org/10.1007/s12283-014-0156-0
Barbosa LF, Denadai BS, Greco CC (2016) Endurance performance during severe-intensity intermittent cycling: effect of exercise duration and recovery type. Front Physiol 7:602. https://doi.org/10.3389/fphys.2016.00602
Beltrame T, Amelard R, Villar R et al (2016) Estimating oxygen uptake and energy expenditure during treadmill walking by neural network analysis of easy-to-obtain inputs. J Appl Physiol 121:1226–1233. https://doi.org/10.1152/japplphysiol.00600.2016
Passfield L, Jg H, Jobson S et al (2017) Knowledge is power: issues of measuring training and performance in cycling. J Sports Sci 35:1426–1434. https://doi.org/10.1080/02640414.2016.1215504
Louhevaara V, Ilmarinen J, Oja P (1985) Comparison of three field methods for measuring oxygen consumption. Ergonomics 28:463–470. https://doi.org/10.1080/00140138508963155
Beneke R, Leithäuser RM, Ochentel O (2011) Blood lactate diagnostics in exercise testing and training. Int J Sports Physiol Perform 6:8–24
Lamarra N, Whipp BJ, Ward SA, Wasserman K (1987) Effect of interbreath fluctuations on characterizing exercise gas exchange kinetics. J Appl Physiol Bethesda Md 1985 62:2003–2012
Beneke R, Alkhatib A (2014) High cycling cadence reduces carbohydrate oxidation at given low intensity metabolic rate. Biol Sport 32:27–33. https://doi.org/10.5604/20831862.1126325
Burnley M, Jones AM, Carter H, Doust JH (2000) Effects of prior heavy exercise on phase II pulmonary oxygen uptake kinetics during heavy exercise. J Appl Physiol Bethesda Md 1985 89:1387–1396
Chatel B, Bret C, Edouard P et al (2016) Lactate recovery kinetics in response to high-intensity exercises. Eur J Appl Physiol 116:1455–1465. https://doi.org/10.1007/s00421-016-3420-0
Wasserman K, Hansen JE, Sue DY et al (2005) Principles of exercise testing and interpretation: including pathophysiology and clinical applications. Lippincott Williams & Wilkins, Philadelphia
Barstow TJ, Molé PA (1991) Linear and nonlinear characteristics of oxygen uptake kinetics during heavy exercise. J Appl Physiol 71:2099–2106
Jones AM, Poole DC (2013) Oxygen uptake kinetics in sport, exercise and medicine. Routledge, Abingdon
Stegmann H, Kindermann W, Schnabel A (1981) Lactate kinetics and individual anaerobic threshold. Int J Sports Med 02:160–165. https://doi.org/10.1055/s-2008-1034604
Brooks GA (1991) Current concepts in lactate exchange. Med Sci Sports Exerc 23:895–906
Gladden LB (2004) Lactate metabolism: a new paradigm for the third millennium. J Physiol 558:5–30
di Prampero PE, Margaria R (1968) Relationship between O2 consumption, high energy phosphates and the kinetics of the O2 debt in exercise. Pflüg Arch 304:11–19. https://doi.org/10.1007/BF00586714
Cerretelli P, Pendergast D, Paganelli WC, Rennie DW (1979) Effects of specific muscle training on VO2 on-response and early blood lactate. J Appl Physiol 47:761–769
Lador F, Azabji Kenfack M, Moia C et al (2006) Simultaneous determination of the kinetics of cardiac output, systemic O2 delivery, and lung O2 uptake at exercise onset in men. Am J Physiol Regul Integr Comp Physiol 290:R1071–R1079. https://doi.org/10.1152/ajpregu.00366.2005
Ferretti G (2015) Energetics of muscular exercise. Springer, Cham
Hughson RL, Cochrane JE, Butler GC (1993) Faster O2 uptake kinetics at onset of supine exercise with than without lower body negative pressure. J Appl Physiol 75:1962–1967. https://doi.org/10.1152/jappl.1993.75.5.1962
Faisal A, Beavers KR, Robertson AD, Hughson RL (2009) Prior moderate and heavy exercise accelerate oxygen uptake and cardiac output kinetics in endurance athletes. J Appl Physiol 106:1553–1563. https://doi.org/10.1152/japplphysiol.91550.2008
Beltrame T, Hughson RL (2017) Linear and non-linear contributions to oxygen transport and utilization during moderate random exercise in humans: aerobic system linearity in frequency domain. Exp Physiol 102:563–577. https://doi.org/10.1113/EP086145
Rossiter HB (2010) Exercise: kinetic considerations for gas exchange. In: Terjung R (ed) Comprehensive Physiology. Wiley, Hoboken
Binzoni T, Ferretti G, Schenker K, Cerretelli P (1992) Phosphocreatine hydrolysis by 31P-NMR at the onset of constant-load exercise in humans. J Appl Physiol 73:1644–1649. https://doi.org/10.1152/jappl.1992.73.4.1644
Wasserman K (1987) Determinants and detection of anaerobic threshold and consequences of exercise above it. Circulation 76:VI29–VI39
Cerretelli P, Shindell D, Pendergast D et al (1977) Oxygen uptake transients at the onset and offset of arm and leg work. Respir Physiol 30:81–97
Gaesser GA, Brooks GA (1984) Metabolic bases of excess post-exercise oxygen consumption: a review. Med Sci Sports Exerc 16:29–43
Freund H, Gendry P (1978) Lactate kinetics after short strenuous exercise in man. Eur J Appl Physiol 39:123–135
Lamarra N (1990) Variables, constants, and parameters: clarifying the system structure. Med Sci Sports Exerc 22:88–95
Swanson GD (1990) Assembling control models from pulmonary gas exchange dynamics. Med Sci Sports Exerc 22:80–87
Casaburi R, Whipp BJ, Wasserman K, Koyal SN (1978) Ventilatory and gas exchange responses to cycling with sinusoidally varying pedal rate. J Appl Physiol 44:97–103. https://doi.org/10.1152/jappl.1978.44.1.97
Haouzi P, Fukuba Y, Casaburi R et al (1993) O2 uptake kinetics above and below the lactic acidosis threshold during sinusoidal exercise. J Appl Physiol 75:1683–1690. https://doi.org/10.1152/jappl.1993.75.4.1683
Bangsbo J (2000) Physiology of intermittent exercise. Exerc Sport Sci 5:53–65
Gharbi A, Chamari K, Kallel A et al (2008) Lactate kinetics after intermittent and continuous exercise training. J Sports Sci Med 7:279
Yano T, Yunoi T, Horiuchi M (2000) Kinetics of oxygen uptake during decremental ramp exercise. J Sports Med Phys Fitness 40:11
Boone J, Bourgois J (2012) The oxygen uptake response to incremental ramp exercise. Sports Med 42:511
Fukuba Y, UsuI S, Munaka M (1989) New mathematical modelling of blood lactate kinetics during ramp mode exercise in man. Jpn J Physiol 39:325–334
Oyono-Enguelle S, Marbach J, Heitz A et al (1990) Lactate removal ability and graded exercise in humans. J Appl Physiol Bethesda Md 1985 68:905–911
Grassi B, Quaresima V, Marconi C et al (1999) Blood lactate accumulation and muscle deoxygenation during incremental exercise. J Appl Physiol Bethesda Md 1985 87:348–355
Scheuermann BW, Hoelting BD, Noble ML, Barstow TJ (2001) The slow component of O2 uptake is not accompanied by changes in muscle EMG during repeated bouts of heavy exercise in humans. J Physiol 531:245–256
Artiga Gonzalez A, Bertschinger R, Brosda F et al (2015) Modeling oxygen dynamics under variable work rate. SCITEPRESS-Science and Technology Publications, Setúbal, pp 198–207
Moxnes JF, Sandbakk Ø (2012) The kinetics of lactate production and removal during whole-body exercise. Theor Biol Med Model 9:7
Poole DC, Burnley M, Vanhatalo A et al (2016) Critical power: an important fatigue threshold in exercise physiology. Med Sci Sports Exerc 48:2320
Wilcox SL, Broxterman RM, Barstow TJ (2016) Constructing quasi-linear V̇O2 responses from nonlinear parameters. J Appl Physiol 120:121–129. https://doi.org/10.1152/japplphysiol.00507.2015
Özyener F, Rossiter HB, Ward SA, Whipp BJ (2001) Influence of exercise intensity on the on- and off-transient kinetics of pulmonary oxygen uptake in humans. J Physiol 533:891–902. https://doi.org/10.1111/j.1469-7793.2001.t01-1-00891.x
Gaesser GA, Poole DC (1996) The slow component of oxygen uptake kinetics in humans. Exerc Sport Sci Rev 24:35–70
Esposito F, Schena F, Ferretti G (2006) Phase III \({\dot{\text{V}}}\)O2 increase does not lead to \({\dot{\text{V}}}\)O2 values higher than \({\dot{\text{V}}}\)O2 max during prolonged intense exercises in humans. Sport Sci Health 1:146–152
Iannetta D, Inglis EC, Fullerton C et al (2018) Metabolic and performance-related consequences of exercising at and slightly above MLSS. Scand J Med Sci Sports 28:2481–2493. https://doi.org/10.1111/sms.13280
Kalis J, Freund BJ, Joyner MJ et al (1988) Effect of beta-blockade on the drift in O2 consumption during prolonged exercise. J Appl Physiol 64:753–758
Grassi B, Rossiter HB, Zoladz JA (2015) Skeletal muscle fatigue and decreased efficiency: two sides of the same coin? Exerc Sport Sci Rev 43:75–83
Bahr R (1992) Excess postexercise oxygen consumption–magnitude, mechanisms and practical implications. Acta Physiol Scand Suppl 605:1
Billat V, Morton R, Blondel N et al (2000) Oxygen kinetics and modelling of time to exhaustion whilst running at various velocities at maximal oxygen uptake. Eur J Appl Physiol 82:178–187
Hill DW, Poole DC, Smith JC (2002) The relationship between power and the time to achieve VO(2max). Med Sci Sports Exerc 34:709–714
Hauser T, Adam J, Schulz H (2013) Comparison of selected lactate threshold parameters with maximal lactate steady state in cycling. Int J Sports Med 35:517–521. https://doi.org/10.1055/s-0033-1353176
Goodwin ML, Harris JE, Hernández A, Gladden LB (2007) Blood lactate measurements and analysis during exercise: a guide for clinicians. J Diabetes Sci Technol 1:558–569
Minetti AE (2011) Bioenergetics and biomechanics of cycling: the role of “internal work”. Eur J Appl Physiol 111:323–329
Francescato M, Girardis M, Di Prampero P (1995) Oxygen cost of internal work during cycling. Eur J Appl Physiol 72:51–57
Bonjour J, Capelli C, Antonutto G et al (2010) Determinants of oxygen consumption during exercise on cycle ergometer: the effects of gravity acceleration. Respir Physiol Neurobiol 171:128–134. https://doi.org/10.1016/j.resp.2010.02.013
Di Prampero P, Cortili G, Mognoni P, Saibene F (1979) Equation of motion of a cyclist. J Appl Physiol 47:201–206
Brooks HP, Andrews MH, Gray AJ, Osborne MA (2013) Comparison of models for the physiological estimation of internal mechanical power in cycling. J Sci Cycl 2:58
Abbiss CR, Peiffer JJ, Laursen PB (2009) Optimal cadence selection during cycling. Int SportMed J 10:1–15
Mcmurray RG, Soares J, Caspersen CJ, Mccurdy T (2014) Examining variations of resting metabolic rate of adults: a public health perspective. Med Sci Sports Exerc 46:1352–1358. https://doi.org/10.1249/MSS.0000000000000232
Bell C, Paterson DH, Kowalchuk JM et al (2001) A comparison of modelling techniques used to characterise oxygen uptake kinetics during the on-transient of exercise. Exp Physiol 86:667–676
Spencer MD, Murias JM, Kowalchuk JM, Paterson DH (2011) Pulmonary O2 uptake and muscle deoxygenation kinetics are slowed in the upper compared with lower region of the moderate-intensity exercise domain in older men. Eur J Appl Physiol 111:2139–2148. https://doi.org/10.1007/s00421-011-1851-1
Jones AM, Burnley M (2009) Oxygen uptake kinetics: an underappreciated determinant of exercise performance. Int J Sports Physiol Perform 4:524
Millet G, Libicz S, Borrani F et al (2003) Effects of increased intensity of intermittent training in runners with differing VO2 kinetics. Eur J Appl Physiol 90:50–57
Bangsbo J (1998) Quantification of anaerobic energy production during intense exercise. Med Sci Sports Exerc 30:47–52
Whipp BJ, Ward SA (1990) Physiological determinants of pulmonary gas exchange kinetics during exercise. Med Sci Sports Exerc 22:62–71
Lador F, Tam E, Azabji Kenfack M et al (2008) Phase I dynamics of cardiac output, systemic O2 delivery, and lung O2 uptake at exercise onset in men in acute normobaric hypoxia. AJP Regul Integr Comp Physiol 295:R624–R632. https://doi.org/10.1152/ajpregu.00797.2007
Bearden SE, Henning PC, Bearden TA, Moffatt RJ (2004) The slow component of VO2 kinetics in very heavy and fatiguing square-wave exercise. Eur J Appl Physiol 91:586–594. https://doi.org/10.1007/s00421-003-1009-x
Whipp B, Rossiter H, Ward S (2002) Exertional oxygen uptake kinetics: a stamen of stamina? Biochem Soc Trans 30:237–247
Stirling J, Zakynthinaki M, Saltin B (2005) A model of oxygen uptake kinetics in response to exercise: including a means of calculating oxygen demand/deficit/debt. Bull Math Biol 67:989–1015
Stirling J, Zakynthinaki M, Billat V (2008) Modeling and analysis of the effect of training on kinetics and anaerobic capacity. Bull Math Biol 70:1348–1370
Artiga Gonzalez A (2016) A comparison of models for oxygen consumption. In: Proceedings of the workshop modelling in endurance sports. University of Konstanz
Whipp BJ, Stirling JR, Zakynthinaki MS (2009) Point: counterpoint. The kinetics of oxygen uptake during muscular exercise do/do not manifest time-delayed phases. J Appl Physiol 107:1669
Ferguson BS, Rogatzki MJ, Goodwin ML et al (2018) Lactate metabolism: historical context, prior misinterpretations, and current understanding. Eur J Appl Physiol 118:691–728. https://doi.org/10.1007/s00421-017-3795-6
Green HJ, Hughson RL, Orr GW, Ranney DA (1983) Anaerobic threshold, blood lactate, and muscle metabolites in progressive exercise. J Appl Physiol 54:1032–1038
Hughson RL, Weisiger KH, Swanson GD (1987) Blood lactate concentration increases as a continuous function in progressive exercise. J Appl Physiol Bethesda Md 1985 62:1975–1981
Zouloumian P, Freund H (1981) Lactate after exercise in man: II. Mathematical model. Eur J Appl Physiol 46:135–147. https://doi.org/10.1007/BF00428866
Zouloumian P, Freund H (1981) Lactate after exercise in man: III. Properties of the compartment model. Eur J Appl Physiol 46:149–160. https://doi.org/10.1007/BF00428867
Gharbi A, Chamari K, Kallel A et al (2008) Lactate kinetics after intermittent and continuous exercise training. J Sports Sci Med 7:279
Beneke R, Hütler M, Jung M, Leithäuser RM (2005) Modeling the blood lactate kinetics at maximal short-term exercise conditions in children, adolescents, and adults. J Appl Physiol 99:499–504
Taoutaou Z, Granier P, Mercier B et al (1996) Lactate kinetics during passive and partially active recovery in endurance and sprint athletes. Eur J Appl Physiol 73:465–470
Messonnier L, Freund H, Denis C et al (2006) Effects of training on lactate kinetics parameters and their influence on short high-intensity exercise performance. Int J Sports Med 27:60–66
Freund H, Zouloumian P (1981) Lactate after exercise in man: IV. Physiological observations and model predictions. Eur J Appl Physiol 46:161–176. https://doi.org/10.1007/BF00428868
Freund H, Zouloumian P (1981) Lactate after exercise in man: I. Evolution kinetics in arterial blood. Eur J Appl Physiol 46:121–133. https://doi.org/10.1007/BF00428865
Margaria R, Cerretelli P, Diprampero PE et al (1963) Kinetics and mechanism of oxygen debt contraction in man. J Appl Physiol 18:371–377
Zignoli A, Savoldelli A, Biral F et al (2014) Application to cycling of a bioenergetic model: towards a multi-level biomechanical model for global cyclist performance analysis. J Sci Cycl 3:76
Dahmen T (2012) Optimization of pacing strategies for cycling time trials using a smooth 6-parameter endurance model. In: Yong J (ed) Proceedings of 2012 pre-olympic congress on sports science and computer science in sport. World Academic Union, Edgbaston
Efron B, Tibshirani R (1986) Bootstrap methods for standard errors, confidence intervals, and other measures of statistical accuracy. Stat Sci 1:54–75
Lagarias JC, Reeds JA, Wright MH, Wright PE (1998) Convergence properties of the Nelder–Mead simplex method in low dimensions. SIAM J Optim 9:112–147
Eberhart R, Kennedy J (1995) A new optimizer using particle swarm theory. In: MHS’95. Proceedings of the sixth international symposium on micro machine and human science, IEEE, pp 39–43
Nocedal J, Wright S (2006) Numerical optimization. Springer, Berlin
Motulsky HJ, Ransnas LA (1987) Fitting curves to data using nonlinear regression: a practical and nonmathematical review. FASEB 1:365–374
Batterham AM, Hopkins WG (2006) Making meaningful inferences about magnitudes. Int J Sports Physiol Perform 1:50
Hopkins WG, Hawley JA, Burke LM (1999) Design and analysis of research on sport performance enhancement. Med Sci Sports Exerc 31:472–485
Hopkins WG (2000) Measures of reliability in sports medicine and science. Sports Med 30:1–15
Robergs RA, Dwyer D, Astorino T (2010) Recommendations for improved data processing from expired gas analysis indirect calorimetry. Sports Med 40:95–111
Stirling JR, Zakynthinaki M (2009) Counterpoint: the kinetics of oxygen uptake during muscular exercise do not manifest time-delayed phases. J Appl Physiol 107:1665–1667. https://doi.org/10.1152/japplphysiol.00158.2009a
Medbø JI, Mamen A, Holt Olsen O, Evertsen E (2000) Examination of four different instruments for measuring blood lactate concentration. Scand J Clin Lab Invest 60:367–380. https://doi.org/10.1080/003655100750019279
Francescato MP, Cettolo V, Bellio R (2014) Confidence intervals for the parameters estimated from simulated O2 uptake kinetics: effects of different data treatments: confidence intervals of estimated parameters of O2 uptake kinetics. Exp Physiol 99:187–195. https://doi.org/10.1113/expphysiol.2013.076208
De Tymowski C, Soussi S, Depret F, Legrand M (2017) On-line plasma lactate concentration monitoring in critically ill patients. Crit Care. https://doi.org/10.1186/s13054-017-1738-6
Beneke R, Jumah MD, Leithäuser RM (2007) Modelling the lactate response to short-term all out exercise. Dyn Med 6:10. https://doi.org/10.1186/1476-5918-6-10
Gerbino A, Ward SA, Whipp BJ (1996) Effects of prior exercise on pulmonary gas-exchange kinetics during high-intensity exercise in humans. J Appl Physiol Bethesda Md 1985 80:99–107
Luger GF (2005) Artificial intelligence: structures and strategies for complex problem solving. Pearson Education, London
LeCun Y, Bengio Y, Hinton G (2015) Deep learning. Nature 521:436–444. https://doi.org/10.1038/nature14539
Esteva A, Kuprel B, Novoa RA et al (2017) Dermatologist-level classification of skin cancer with deep neural networks. Nature 542:115–118
Jones N (2014) The learning machines. Nature 505:146
Montoye AHK, Pivarnik JM, Mudd LM et al (2016) Comparison of activity type classification accuracy from accelerometers worn on the hip, wrists, and thigh in young, apparently healthy adults. Meas Phys Educ Exerc Sci 20:173–183. https://doi.org/10.1080/1091367X.2016.1192038
Jobson SA, Passfield L, Atkinson G et al (2009) The analysis and utilization of cycling training data. Sports Med 39:833–844. https://doi.org/10.2165/11317840-000000000-00000
Silva AJ, Costa AM, Oliveira PM et al (2007) The use of neural network technology to model swimming performance. J Sports Sci Med 6:117–125
Ofoghi B, Zeleznikow J, MacMahon C, Raab M (2013) Data mining in elite sports: a review and a framework. Meas Phys Educ Exerc Sci 17:171–186. https://doi.org/10.1080/1091367X.2013.805137
Goodfellow I, Bengio Y, Courville A (2016) Deep learning. MIT Press, New York
Kiefer J, Wolfowitz J (1952) Stochastic estimation of the maximum of a regression function. Ann Math Stat 23:462–466
Hornik K, Stinchcombe M, White H (1989) Multilayer feedforward networks are universal approximators. Neural Netw 2:359–366. https://doi.org/10.1016/0893-6080(89)90020-8
Thrun S, Pratt L (2012) Learning to learn. Springer, Berlin
Beltrame T, Amelard R, Villar R et al (2016) Estimating oxygen uptake and energy expenditure during treadmill walking by neural network analysis of easy-to-obtain inputs. J Appl Physiol 121:1226–1233. https://doi.org/10.1152/japplphysiol.00600.2016
Beltrame T, Amelard R, Wong A, Hughson RL (2017) Prediction of oxygen uptake dynamics by machine learning analysis of wearable sensors during activities of daily living. Sci Rep 7:45738. https://doi.org/10.1038/srep45738
Borror A, Mazzoleni M, Coppock J et al (2019) Predicting oxygen uptake responses during cycling at varied intensities using an artificial neural network. Biomed Hum Kinet 11:60–68. https://doi.org/10.2478/bhk-2019-0008
LeCun Y, Bengio Y (1995) Convolutional networks for images, speech, and time series. Handb Brain Theory Neural Netw 3361:1995
Rajpurkar P, Hannun AY, Haghpanahi M, et al (2017) Cardiologist-level arrhythmia detection with convolutional neural networks. ArXiv Prepr ArXiv170701836
Gajda R, Biernacka EK, Drygas W (2018) Are heart rate monitors valuable tools for diagnosing arrhythmias in endurance athletes? Scand J Med Sci Sports 28:496–516. https://doi.org/10.1111/sms.12917
Längkvist M, Karlsson L, Loutfi A (2014) A review of unsupervised feature learning and deep learning for time-series modeling. Pattern Recognit Lett 42:11–24. https://doi.org/10.1016/j.patrec.2014.01.008
Lipton ZC, Kale DC, Elkan C, Wetzell R (2015) Learning to diagnose with LSTM recurrent neural networks. ArXiv Prepr ArXiv151103677
Etxegarai U, Portillo E, Irazusta J et al (2017) Estimation of lactate threshold with machine learning techniques in recreational runners. Appl Soft Comput. https://doi.org/10.1016/j.asoc.2017.11.036
Zignoli A, Fornasiero A, Stella F et al (2019) Expert-level classification of ventilatory thresholds from cardiopulmonary exercising test data with recurrent neural networks. Eur J Sport Sci. https://doi.org/10.1080/17461391.2019.1587523
Holm EA (2019) In defense of the black box. Science 364:26–27. https://doi.org/10.1126/science.aax0162
Darwish A, Hassanien AE (2011) Wearable and implantable wireless sensor network solutions for healthcare monitoring. Sensors 11:5561–5595
Di Dino A, Biral F, Bosetti P (2011) Hybrid modeling of non-linear mechanical systems: the case of a vehicle shock absorber. In: Volume 4: 8th international conference on multibody systems, nonlinear dynamics, and control, parts A and B. ASME, Washington, DC, USA, pp 1121–1130
Psichogios DC, Ungar LH (1992) A hybrid neural network-first principles approach to process modeling. AIChE J 38:1499–1511. https://doi.org/10.1002/aic.690381003
Møller M, Wedderkopp N, Myklebust G et al (2018) Validity of the SMS, Phone, and medical staff Examination sports injury surveillance system for time-loss and medical attention injuries in sports. Scand J Med Sci Sports 28:252–259. https://doi.org/10.1111/sms.12869
Acknowledgements
We are grateful to the Fondazione Cassa di Risparmio di Trento e Rovereto (CARITRO) for partially supporting this research. We thank the reviewers for spending a substantial amount of time looking over the manuscript.
Funding
Financial support has been received from the Fondazione CARITRO (Grant No. 2017.0379).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zignoli, A., Fornasiero, A., Bertolazzi, E. et al. State-of-the art concepts and future directions in modelling oxygen consumption and lactate concentration in cycling exercise. Sport Sci Health 15, 295–310 (2019). https://doi.org/10.1007/s11332-019-00557-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11332-019-00557-x