Abstract
Drought stress is a major limiting factor for rice production globally. The current climatic changes have further increased the incidence and duration of droughts worldwide. On the other hand, seaweed extracts have been extensively studied as biostimulants that improve plant growth, nutrition, quality, yield, and stress tolerance of rice (Oryza sativa). Different types of seaweeds have been employed for biostimulant preparation, of which algae is most commonly used. Of particular interest were Galaxaura oblongata and Turbenaria ornate seaweeds, as biostimulants that inoculate with soil in rice plants growing under drought conditions to boost the resilience of rice to drought. The incorporation of drought-stressed-rice soil with two investigated seaweeds led to improved growth, almost regulated levels of photosynthetic pigments, compatible solutes, MDA, hydrogen peroxide, and phenolic compounds as well as flavonoids, along with notably upregulated expression of PIP1;4, PIP2;7, NCDE5, and OsMyb-R1 genes of the rice cultivars Giza 177 and Giza 179, and this enhanced the water status of rice cultivars grown under drought conditions. Furthermore, this study suggested that amending soil with investigated seaweeds enhanced nutrition and osmolyte production, ameliorated MDA, and upregulated investigated drought-responsive genes as well as promoted the growth along with antioxidant reply thoroughly contribute to improving rice plant tolerance to drought stress.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rice (Oryza sativa L.) is a main staple of diets and a major source of food security for more than half of the world’s population; the sustenance of many people depends upon the rice plant (Samal et al. 2021). The production and improvement of rice are affected by several environmental stresses, particularly water deficits, and these stresses have a negative impact on the rice plant’s growth, development, and physiological as well as phytochemical aspects (Datta et al. 2017). Water deficits cause plants to continuously lose water via transpiration or evaporation. During periods of drought, plants close their stomata to prevent evaporation and this reduces the plants’ ability to effect gas exchange. Irrigation is one of the main strategies used to offset water shortages; it enhances the efficiency of water use across the world (Bakry et al. 2012). Water resources in Egypt are very limited, so Egypt has established several water-preserving irrigation practices that can benefit rice plants.
Indeed, drought stress alters the biochemical, physiological, and molecular parameters of almost all crop plants, and these changes help plants to tolerate harsh environmental stresses. The extent of these alterations differs significantly depending upon the plant species and the stress duration and intensity, as well as the growth stage of plants (Abd Elhamid et al. 2016; Ahmed and Sadak 2016; Sadak 2016; Arora et al. 2002). Abiotic stresses such as salinity and drought are global issues threatening the survival of agronomic crops and maintainable production of food (Elewa et al. 2017; Dawood et al. 2017). A water deficit is a restrictive factor during the primary stage of plant growth and development, and it can have harmful effects on a plant’s cell structure and metabolism (Anjum et al. 2003; Bhatt and Rao 2005; Kusaka et al. 2005; Ezzo et al. 2018). Rice may be more susceptible to drought stress than other crops because it is actually grown in water.
Drought damages a plant’s photosynthetic apparatus by inhibiting the content of chlorophyll and the enzyme activities involved in photosynthesis (Monakhova and Chernyadev 2002; Bakry et al. 2019). Moreover, it alters the metabolism of antioxidants, which is considered one of the essential metabolic processes that may enhance plants’ tolerance to drought stress (Da Costa and Huang 2007; Sadak et al. 2020). Notably, the imbalance between the production and elimination (or detoxification) of reactive oxygen species (ROS) induced by water stress causes cellular injuries (Foyer and Noctor 2002; Sadak and Bakry 2020). Meanwhile, some plants can tolerate stresses via developing cellular adaptive mechanisms, including the accumulation of protective compatible solutes such as soluble sugars and proline, as well as the upregulation of oxidative stress markers; these mechanisms help plants to tolerate environmental conditions (Sakamoto and Murata 2002; Abd El-Hameid and Sadak 2020). Consequently, enzymatic and nonenzymatic antioxidant defense mechanisms such as catalase (CAT), ascorbate peroxidase (APX), and peroxidase (POX), as well as ascorbate (AsA), the total phenolic compounds, and the flavonoids, are the most potent defense systems for scavenging ROS (Abd Elhamid et al. 2021).
Furthermore, plants undergo molecular changes to counteract the adverse effects of environmental stresses (El-Esawi et al. 2018, 2019). The water channel proteins called aquaporins form pores in the membranes of biological cells, mainly facilitating the transport of water between cells. Plant aquaporin proteins (AQPs) are intrinsic protein families that are widely distributed in plasma membranes (Quigley et al. 2002; Agre 2006). Plasma membrane intrinsic proteins (PIPs) are the main groups of AQPs, and they are subdivided into two subgroups PIP1 and PIP2 (Kaldenhoff and Fischer 2006). The overexpression of PIP1 and PIP2 genes has improved plant tolerance in response to abiotic stresses and mitigated the negative effects of drought stress (Aharon et al. 2003; Jang et al. 2007; Liu et al. 2013; Ayadi et al. 2019). Concurrently, the abscisic acid (ABA) biosynthetic pathway NCED5 gene encoded 9-cis-epoxycarotenoid dioxygenase 5 (Lefebvre et al. 2006; Yang and Guo 2007). Moreover, the myeloblastosis transcription factor (TF) Myb-R1 is one of the most important classes of TFs, as it has an essential role in regulating a plant’s defense mechanism by mitigating the adverse effects triggered by biotic or abiotic stresses (Erpen et al. 2018).
The global population growth poses a serious threat to food security due to a certain amount of land available for agriculture which decreased with time. Consequently, the development of environmentally friendly products like biofertilizers as well as its influences as a result of modern agriculture has been explored with respect to several crops, including rice. Therefore, the improvement of the yield and crop quality can be achieved by the use of fertilizers. Traditional fertilizers, coming from either organic or inorganic origins play a vital role in agriculture which qualifies the production of food at a broad scale that sustains the global population (Mahanty et al. 2017). The application of seaweeds as biofertilizers is seen as a potential substitute for chemical fertilizers in agricultural due to a vast potential for improving crop productivity and food safety. Seaweeds are macro-aquatic thallophytic algae of the kingdom Planta (Arioli et al. 2015). Seaweeds are a renewable natural resource that adapts to environmental conditions, and they are rich in various types of natural biologically bioactive compounds (Nabti et al. 2017; Bakhoum et al. 2023). The derivation of biofertilizers from seaweeds such as Ascophyllum and Sargassum is considered an ecofriendly activity because these seaweeds are nontoxic, biodegradable, and nonpolluting, as well as harmless to humans and animals (Dhargalkar and Pereira 2005). Therefore, nowadays, seaweeds have the potential to be used, and are used, as bioresources and biofertilizers in sustainable applications instead of chemical fertilizers (Khan et al. 2009; Layek et al. 2018). In addition, the recent application of seaweeds as bio-fertilizers in agriculture and for the improvement of crop plants is considered a tool for preserving the environment. The application of seaweed products in the fields of horticulture and agriculture has led to enhanced seed germination, improved plant development, and increased abilities of plants to tolerate environmental conditions (Zhang and Ervin 2008). Hashem et al. (2019) reported that the potential of different algal taxa such as Ulvalactuca, Cystoseira spp., and Gelidiumcrinale for use as biofertilizers could improve the growth and productivity of canola plants under salt stress conditions. Also, the results obtained by Hussein et al. (2021) confirmed that the application of the seaweeds Ulvafasciata, Cystoseiracompressa, and Laurenciaobtusa as biostimulants had the potential to enhance the salinity tolerance of Vigna sinensis and Zea mays plants.
Irrigation management practices have been described as water-saving as compared with submerging in rice productivity. Moreover, the deficit irrigation adjusts the management use of nitrogen fertilizers is not clear. The influence of deficit irrigation on the efficient nutrient use of rice is important for improving the fertility of the soil and growing rice cultivation. Consequently, this study aimed to elucidate the mechanism by which the rice plant survives drought stress. This was estimated by assaying the plant’s physiological, biochemical, and molecular responses to drought. Attempts were investigated to check the performance of both rice cultivars, Giza 177 and Giza 179 exposed to drought conditions, in terms of their productivity by using seaweeds as biofertilizers in combination with soil also made to enhance the defense mechanisms in both rice cultivars. Finally, this study considered a sustainable approach for reducing water consumption through the management of irrigation practices in improvement the rice production (de Avila et al. 2015; Chen et al. 2021).
Materials and Methods
Plant Material
Two different cultivars of rice (Oryza sativa L) grains, Giza 177 and Giza 179 were attained from the Agriculture Research Center, Rice Research Institute in Giza, Egypt.
Algal Collection
Two seaweed species were collected by handpicking from the Red Sea coast in Egypt, with the littoral zone of rocky shorelines in Hurghada city (27° 15´ 58.45˝ N, 33° 48´ 57.09˝). Algal identification was performed according to (Aleem 1993) which was established by morphological structures determined by microscopic examination. One of them belongs to the division Rhodophyta (red algae) and identified as Galaxaura oblongata (Tricleocarpafragilis) (J. Ellis & Solander) J.V. Lamouroux, while the other one from the Pheaophyta division (brown algae) and identified asTurbenaria ornate (Turner) J. Agardh. After collection, the algal samples were washed with tap water to eliminate salt and sand particles, then air-dried, and finally grounded into small powder using a mill and stored at room temperature for analysis (Table 1).
Chemicals
The chemicals used in the present work with high purity, obtained from Sigma-Aldrich Chemical Co., Germany, and the organic solvents used with AR grade.
Experimental Design and Growth Condition
Rice grains were immersed for 5 min in sodium hypochlorite solution (1%) to ensure surface sterilization was performed, then washed carefully with tap water. The pot experiment was directed in the Botany Department, Faculty of Science, Ain Shams University. This experiment was performed during the autumn season of 2022 from September to October. Two native rice grains (Oryza sativa L.) cultivars were applied in the current experiment defined; Giza 177 and Giza 179. The pots were separated into 2 groups; the first group was used for cv. Giza 177 and the later one for cv. Giza 179. The rice grains were planted in pots (30 × 18 cm) occupied with standardized clay/sandy (2:1) soil (8 kg). The physical and chemical constituents of the experimental soil are presented in Table 2. The clay/sandy soil was divided into three subgroups for each cultivar. The two series were amended by 10 g w/w from the two investigated seaweeds T. ornate and G. oblongata as well as the third one remains without algal treatment which serves as a control. A randomized complete block design was performed with 3 replicates per each treatment for all analyses, except; 10 different replicates from each treatment were chosen for measuring the growth parameters. The range of relative humidity recorded was 24 and 60%. The maximum and minimum day temperatures were 38 °C and 18 °C, respectively. All pots were provided with sufficient irrigation. Moreover, after 2 weeks, the drought stress condition was exercised in half of each treatment through withholding water capacity as irrigation was displayed every 12 days (Alharbi et al. 2021). The plants after thinning were left-hand to develop with the different treatments for one month, and then seedlings were collected for measuring growth criteria, and physiological, biochemical, and molecular analyses.
Methods
Growth Parameters
The growth parameters were measured after 30 days from sowing. Ten plants were randomly sampled in the morning from each treatment sensibly washed with tap water to remove soil debris, and left on the filter sheets to get rid of water. Plant growth parameters in terms of root and shoot lengths as well as fresh weights of roots and shoots.
Extraction and Estimation of Photosynthetic Pigments
The chlorophylls a and b and carotenoids were extracted and estimated according to the method of Metzner et al. (1965). One gram of leaves was extracted with 85% (v/v) acetone, then the extract was filtered and up to the total volume (100 mL) by acetone. The absorbance was recorded at three wavelengths of 663, 644, and 452.5 nm. The photosynthetic pigment content was assayed according to the following equations:
Extraction and Estimation of Carbohydrates
The method described for carbohydrate extraction was adopted by (Homme et al. 1992). A known weight of fresh plant tissue was extracted with boiled 80% (v/v) ethanol. The homogenate was filtered through filter paper (Whatman No. 1), then was oven-dried at 60°C, followed by dissolving the residue in a known volume of distilled water to get ready for soluble sugar determination. Furthermore, the method used for soluble sugar determination was that of (Blakeney and Mutton 1980). Two mL of dissolved residue was mixed with 10 mL of anthrone reagent, then placed in a boiling water bath for 20 min, then cooled and finally, the absorbance was measured at 620 nm.
Extraction and Estimation of Total Soluble Proteins
Total soluble proteins were extracted by 0.15 N NaCl. The homogenate was centrifuged at 4,000 rpm. A known volume of supernatant was taken to determine the soluble protein content by using the Folin–Ciocalteu reagent according to the method adopted by (Daughaday et al. 1952).
Extraction and Estimation of Proline
Proline was determined according to the method described by (Bates et al. 1973). Fresh plant tissue (0.5 g) was extracted with 3% sulfosalicylic acid, the homogenate was filtered by Whatman No.1 filter paper. Two milliliters of filtrate were added to 2 mL of acid ninhydrin reagent and 2 mL of glacial acetic acid. The absorbance at 520 nm was recorded against toluene as blank.
Extraction and Estimation of Hydrogen Peroxide
The hydrogen peroxide was assayed by Titanium reagent according to the Yu et al. (2003) method. The absorbance was measured by spectrophotometric at 415 nm.
Extraction and Estimation of Lipid Peroxidation Product (MDA)
The level of lipid peroxidation product was determined by determining the amount of malondialdehyde (MDA) conferring to the method of Minotti and Aust (1987). Plant tissue (100 mg) was ground with 2 mL of distilled water, then centrifuged at 4000 rpm for 15 min. The supernatant was added to the same volume of 0.5% thiobarbituric (v/v) dissolved in 20% (w/v) trichloroacetic acid. The mixture was heated at 95 °C for 30 min, after cooling in an ice bath, the absorbance was measured at two wavelengths; 532 and 600 nm.
Extraction and Estimation of Ascorbic Acid (AsA)
Nonenzymatic antioxidants like ascorbic acid were performed by the method of Mukheriee and Choudhuri (1983). Half a gram of fresh plant sample was homogenized with 10 mL of 6% (w/v) trichloroacetic acid. The As A content was determined by a 2% dinitrophenylhydrazine reagent. The absorbance was recorded at 530 nm.
Extraction and Estimation of Total Phenolic Compounds
Total phenols were measured according to the method applied by (Makkar et al. 1993). The extraction method was performed with 80% methanol, then evaporation was carried out and the evaporated residue was up to the known total volume by distilled water. The estimation method was applied by adding 0.5 mL of extract to 0.5 mL Folin–Ciocalteu reagent. The optical density was recorded at 725 nm.
Extraction and Estimation of Flavonoids
According to Harborne (1998), flavonoids were measured based on the reaction of the aluminum chloride colorimetric method. After extraction of flavonoids from the plant sample as carried out in the total phenols method, a known extract volume (1 mL) was added to 0.1 mL (1 M potassium acetate), 1.5 mL (methanol), 0.1 mL (10% aluminum chloride) and 2.8 mL (distilled water). The mixture was left at room temperature for half an hour. Finally, the absorbance was measured at 415 nm.
Extraction and Estimation of Total Antioxidant Capacity
The ferric reducing antioxidant power (FRAP) method was followed for the estimation of total antioxidant capacity as reported by Oyaizu (1986).
DPPH Radical Scavenging Assay
The antioxidant capacity of plant extract was determined through the 1, 1-diphenyl-2-picrylhydrazyl (DPPH) method followed by (Yamaguchi et al. 1998) and calculated conferring to the below equation:
So, A0 is the absorbance of the DPPH blank solution, and A1 is the sample absorbance.
Extraction and Estimation of Antioxidant Enzymes
The method described for enzyme extraction was that according to (Mukheriee and Choudhuri 1983). Catalase (CAT) activity was assayed following the method of Chen et al. (2000). Peroxidase (POX) activity was determined according to the method of Kar and Mishra (1976). Ascorbate peroxidase (APX) activity was assayed as described by (Koricheva et al. 1997).
RT‑PCR Analysis
Total RNA was extracted from leaf tissues using the GeneJet RNA purification kit (Cat. No. K0731, Thermo Scientific, US) conferring to the manufacturer's instructions. The concentration of total RNA and their purity were determined by using NanoDrop (Thermo Scientific, USA); the purity of extracted RNAs (A260/A280) was 1.8–2. The High-Capacity cDNA Reverse-Transcription Kit (Applied Biosystems, US) cat.no. 4368814 was used to synthesize cDNA from isolated RNA. The real-time SYBR Green PCR method was utilized for the PCR analysis, with the Maxima SYBR Green qPCR master mix (Thermo Scientific, US) cat.no. K0251 and primers are listed in Table 3. qPCR was performed on an optical 96-well plate using a real-time polymerase chain reaction (Agilent Stratagene MX3000P, USA) under cycling conditions (10 min at 95 °C, followed by 45 cycles of 15 s at 95 °C, 60 s at 60 °C, and 30 s at 72 °C). The melting curves were examined to confirm the amplicon specificities for SYBR Green-based PCR amplification. A thresholding approach was used to assess relative gene expression. Every sample was tested twice to express relative mRNA levels, the relative quantitation was calculated as 2−ΔΔCt according to the technique applied by Livak and Schmittgen (2001). The resulting cDNA was subjected to PCR for 35 cycles with respective primers designated from the sequence of the OsMyb-R1, OsPIP1;3, OsPIP2;7, OsNCED5 genes (Table 3) using primer premier 5.0 software and were procured from Invitrogen Corporation (Van Allen Way, Carlsbad, Canada). Dream Taq™Green PCR Master Mix (Invitrogen Corporation) was used in the PCR. Amplification yields were run in 1.5% agarose gel 1 × TAE (Tris–Acetate-EDTA) buffer at approximately 120 V, which was separated by electrophoresis, and then visualized via staining the gel in ethidium bromide. For comparative determinations, the control samples and treated PCR yields were run on the same gel. Gel images were analyzed by using Gel Analyzer Pro software (version 3.1).
Statistical Analysis
The data were expressed as mean values ± standard error (SE). One-way analysis of variance (ANOVA) test was used to perform the statistical analysis using SPSS version 25. Furthermore, apply the least significant difference (LSD) test applied by Snedecor and Cochran (1980) at P-value < 0.05. Finally, Duncan’s multiple range test was carried out to comparisons between intergroups. Further comparisons between intergroup heatmap and PCA (principal comparison analysis) were performed.
Results and Discussion
Drought stress has become a detrimental factor to the growth of rice, as shown in the morphological, physiological, and biochemical, as well as molecular, alterations in rice plants suffering drought (Quampah et al. 2011).
Changes in Growth Parameters
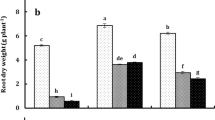
In this study, drought stress altered the length of the roots of the two investigated rice cultivars, Giza 177 and Giza 179, compared with the corresponding control values (Table 4). This might be attributed to the deficits in irrigation intervals experienced by the rice plants. The roots of both investigated rice cultivars were significantly lengthened due to the fact that desiccation caused the root systems to become thick and long so they could reach water (Bouman and Tuong 2001; Lipiec et al. 2013). Moreover, these conditions may also have facilitated improved drought tolerance in the plants via the deep absorption of water from the soil and the reduction of toxic elements and organic acid intermediates, thereby increasing the bioavailability and utilization of some nutrients such as nitrogen and phosphorus, as well as potassium, compared with plants flooded control treatment (Gowda et al. 2011; Uga et al. 2013). On the other hand, the continuous flooding of rice seedlings followed by water deficit irrigation every 12 days significantly reduced the lengths of the shoots and the fresh weights of the shoots and roots of both cultivars (Table 4). Our results are in agreement with those of Ghouri et al. (2021), who postulated that the root length would increase with an increased irrigation period and the shoot length would be reduced in rice plants. Moreover, the amending of the soil with the seaweedGalaxaura oblongata or Turbenaria ornate resulted in significant increases in the previously mentioned growth parameters of both stressed and unstressed rice plants (Table 4). This increase might be a result of the application of seaweed as bio nutrient fertilizers increasing the tolerance to abiotic stress in plants, which the plants can benefit from the water and nutrients in the soil more effectively to the endogenous nutrients of seaweed; as well as the high quantities of organic matter present in seaweeds (Kumareswari and Rani 2015; Layek et al. 2018). The application of seaweed stimulated root proliferation in tomato plants, and this increased the ability of the plants to absorb sufficient nutrients from the soil, leading to plant growth and improvement (Hernández-Herrera et al. 2013). The efficient biostimulation of the investigated seaweeds might be attributed to their total soluble sugars, total antioxidant capacity, flavonoid content, and content of phenolic compounds (Table 1). Moreover, the positive effect of seaweeds as biofertilizers was noticeable in the growth of apple trees, and this could be due to the seaweeds’ adequate supply of essential nutrients and their enhancement of the soil textures, as well as to an improved water-holding capacity (Mazzola and Brown 2010). Nedzarek and Rakusa-Suszczewski (2004) mentioned that the biodegradability of seaweed in soil released great amounts of organic material and various minerals, especially phosphate, NO2, NO3, and NH4, which enhanced the fertility of the soil.
Changes in Photosynthetic Pigments
A water deficit is a great loss of water that induces a disturbance in the structure and metabolism of plant cells. It also ultimately diminishes the water content and leaf turgor and causes the stomata to close; all these activities arrest photosynthesis (Jaleel et al. 2007, 2008). A significant reduction in chlorophylls a and b and the carotenoid content was recorded in the two investigated rice cultivars, Giza 177 and Giza 179, that were cultivated under drought conditions (Table 5). This reduction might be attributed to the inhibition of chlorophyll synthesis or the destruction of chloroplasts in the rice plants and the consequent reduction in the photosynthetic apparatus and inhibition of growth (Monakhova and Chernyadev 2002; Faseela et al. 2019). The results of the current study agreed with those of several other studies (Keyvan 2010; Arjenaki et al. 2012; Embiale et al. 2016; Alharbi et al. 2022) on rice plant species. Consistent with our results, it was recorded that soil amended with G. oblongata or T. ornate ameliorated the effects of water deficit stress. This may have been due to the protection of photosystems against photo-oxidation and the minimization of ROS production triggered by oxidative stress as compared with the controls (see Table 5). Moreover, seaweed application is considered a source of cytokinins, which have protective properties on chloroplasts (Wally et al. 2013), thus, they reflect on the chlorophyll contents. Our findings on the stimulatory effect of the investigated seaweeds on the content of photosynthetic pigments were similar to the findings of Kumar (2014) and Ali et al. (2022) on tomato and okra, respectively, plants.
Changes in Soluble Sugars, Soluble Proteins, Proline, and Lipid Peroxidation Products
Sugars are considered the first organic compounds to be synthesized in plants as metabolic photosynthetic products. A water deficit impacts the metabolism of carbohydrates, and this causes the formation of a group of compatible solutes that act as osmolytes to maintain leaf cell turgor, which can be affected by stress conditions (Rolland et al. 2006; Mahmood et al. 2020). Our study clearly showed that drought stress significantly increased the total soluble sugars in seedlings of the rice cultivars Giza 177 and Giza 179. The percentage was noticeably higher in Giza 177, at 152.3%, than in Giza 179, at 88.9%, compared with their corresponding controls grown under well-watered conditions. The mechanism of accumulation of soluble sugars in plants is recognized as an osmotic regulation that is closely related to the severity of the plant drought stress (Shehab et al. 2010; Usman et al. 2013; Maisura et al. 2014). The percentages of our findings might be attributed to the role of soluble sugars in scavenging ROS, which enhanced water uptake from drying soil and was associated with improved plant tolerance under drought conditions. Our results were in agreement with those of Li et al. (2020) who found that increased drought intensity was responsible for an increased sugar content in maize plants. In addition, the application of either G. oblongata or T. ornate to soil significantly increased the total soluble sugars (Table 6) in both the stressed rice cultivars. Such a percentage of total soluble sugars of seaweed-treated rice plants might be attributed to the effect of the applied algae on the mitigation of the injurious impacts of drought stress on chloroplasts. The results in this study were in agreement with those of El Boukhari et al. (2023), who reported that the application of Fucusspiralis, Ulvalactuca, and Ascophyllum nodosum improved the levels of soluble sugars in faba bean plants as compared with the control as a response to drought stress.
The synthesis of protein, as well as its degradation, is one process affected by drought stress in plants. The soluble protein content is an important sign of the plant’s physiological status (Doganlar and Atmaca 2011). In the current study, the increase in the total soluble protein content was noticed in both Giza 177 and Giza 179 planted under drought stress (see Table 6). This increase was more pronounced in Giza 177. The decrease in the total soluble protein during drought stress could be due to the appearance of new stress proteins (Jiang and Huang 2002). Furthermore, the increment in the total soluble protein content might be the cells compensating for both that have been deactivated due to ROS binding (Seregin and Ivanov 2001; Lamhamdi et al. 2010). Moreover, it could be that drought causes disturbances in the nitrogen metabolism of many plants. The results in Table 6 indicated that the cultivation of the investigated rice cultivars in soil amended with G. oblongata or T. ornate significantly induced the further accumulation of total soluble proteins in both cultivars. The increases in total soluble proteins in stressed plants grown in algae-treated soil were consistent with the findings of Xu and Leskovar (2015) and Kapur et al. (2018) for spinach and strawberry plants, respectively; they reported that seaweeds enhanced the edible quality of plants by stimulating the accumulation of total soluble proteins. The algal extract may have a potential role in diminishing the injurious effects of drought stress on plant productivity. This is due to the rich seaweed, which has vital components that participate in mitigating the adversarial effects of drought stress (El-Sayed et al. 2015; Fouda et al. 2022).
Proline is an amino acid with many functions; it performs as a signaling compound, an osmoprotectant, and a stress-tolerance marker (Ahmad et al. 2009; Liu et al. 2011). It adjusts the cells’ osmotic pressure; limits the protein denaturation, membrane constancy, and stabilization of enzymes; protects cells against stress and ROS-triggered damage; and maintains the balance of nutrients via water transport (Kartashov et al. 2008; Ivanov et al. 2013). In the current study, the imposition of drought stress stimulated the buildup of proline in the two investigated rice cultivars. The proline level in the Giza 177 stressed seedlings was higher than that in the Giza 179 stressed seedlings. Soil amendment with G. oblongata or T. ornate stimulated an enormous augmentation of proline. The overexpression of proline was determined by its metabolism and transport through cells, as well as diverse cellular compartments (Szabados and Savoure 2010). Moreover, the massive upregulation of proline biosynthesis under drought conditions was due to the genes involved in this process (Armengaud et al. 2004). The tolerance mechanism was also associated with osmotic adjustment and turgor maintenance. This mechanism led to the upregulation of some osmolytes such as proline, which caused a decline in the cellular osmotic potential and affected the increase of leaf turgor (Armengaud et al. 2004; Trovato et al. 2008), scavenging of ROS, stabilization of protein, and maintenance of membrane integrity (Ashraf and Foolad 2007; Liu et al. 2011). The overproduction of proline in stressed plants was correlated with the plant’s stress tolerance (Anjum et al. 2011). The high levels of proline in stressed rice plants grown in soil amended with G. oblongata or T. ornate caused the plants to be more tolerant than the plants grown in untreated soil (see Table 6). Previous studies reported an enhancement in the accumulation of proline content in seaweed-treated plants grown under drought conditions, thus might be due to the lipophilic components (LPC) existing in seaweeds, as these could enhance the proline content (Sangha et al. 2014). Previous studies reported that the application of A. nodosum algae caused an increase in proline levels in drought-stressed bean plants (Renuka and Rathinavel 2006; Carvalho et al. 2018).
Notably, drought stimulated the liberation of ROS as hydrogen peroxide, as well as the superoxide radical, so it was able to first attack the phospholipids of the plasma membrane and deactivate the enzymes of the SH group. In response to drought conditions, the generation of ROS exceeded the capacity of the antioxidant defense system and caused a loss of membrane stability; this was allied with an intensification in permeability and an injury of integrity (Blokhina et al. 2003). Therefore, the capability of plasma membranes to regulate the rates of entry and egress of ions in and out of a cell was considered a sign of membrane injury. The results of this investigation, as shown in Table 6, showed that drought stress induced a significant intensification in the lipid peroxidation product malondialdehyde (MDA) in Giza 177 and Giza 179. The increase in MDA levels was parallel with the increase in hydrogen peroxide (H2O2) levels in the two rice cultivars, and this resulted in increased membrane fluidity and, consequently, enhanced membrane permeability (Reddy et al. 2004; Zhou et al. 2007). Cultivar Giza 177 exhibited greater increases in MDA and H2O2 content than cultivar Giza 179; this could be evidence of a greater degree of membrane damage and, thereby, a high susceptibility of Giza 177 to drought compared with Giza 179 (see Table 6 and Fig. 1). The reduction in membrane damage in both rice cultivars when grown in soil amended with G. oblongata or T. ornate, as evident by the lower values of MDA and H2O2, could be due to the high activity of antioxidant enzymes in stressed rice seedlings. An increase in the activities of antioxidant enzymes imposed by drought stress might buffer the increased generation of ROS and enhance the protective strategy for eliminating the oxidative damage generated by drought stress (Gunes et al. 2007). The fact that the H2O2 level was reduced in the drought-stressed seedlings of both rice cultivars suggested that the higher activities of scavengers such as CAT, POX, and APX were sufficient to eliminate H2O2 (Bray et al. 2000). The data in this study were in agreement with those of Jacomassi et al. (2022), who reported that the MDA concentration was reduced in sugarcane plants grown in soil amended with seaweed extract.
Changes in the Activities of Some Antioxidant Enzymes
Plants have enzymatic and nonenzymatic antioxidants which can serve as an effective defense system. It can attenuate the augmented production of ROS and build up a protective mechanism to eliminate the damage inflicted by oxidative stress (Diaz et al. 2001). The enzymatic antioxidants assayed in this work were peroxidase (POX), catalase (CAT), and ascorbate peroxidase (APX), and the nonenzymatic compounds assayed included ascorbic acid, total phenols, and flavonoids. In plants, there is a balance between antioxidant enzymes and ROS that extremely alterations, hindering plant growth in stressful conditions and reducing plant homeostasis; decline in ROS accumulation in plants treated with seaweeds (Dutot et al. 2012). The activities of CAT, POX, and APX in the present study were markedly increased in the stressed rice cultivars grown in soil amended with G. oblongata or T. ornate (Table 7). The application of G. oblongata or T. ornate caused increases in the activities of CAT and POX in stressed rice seedlings, and this was associated with a significant reduction in the H2O2 level. Furthermore, antioxidant enzyme activities improved with the application of seaweeds, which mitigate ROS damage and return cellular homeostasis in plants (Elansary et al. 2017). The increase in the enzyme activities of CAT, POX, and APX under conditions of abiotic stress reduces the risk of membrane damage and returns plants to homeostasis (Gill et al. 2002). The present results showed increased activities of APX, CAT, and POX in rice plants grown in soil amended with our investigated seaweeds (Table 7). The results were in accordance with the results of Sujata et al. (2023), who showed that seaweeds were responsible for increased antioxidant enzymes in Brassica juncea (L.). This increase might be attributed to the potential for seaweed extracts to upregulate the activities of the antioxidant enzymes APX, CAT, and POX; this would be related to their endogenous bioactive components (O’Sullivan et al. 2011). The results in this study agreed with the findings of Mansori et al. (2015), who reported that when the seaweeds Ulva rigida and Fucus spiralis were applied to bean plants exposed to drought conditions, the result was the activation of the antioxidant enzymatic system, including CAT and APX, and an increase in total phenolic compounds, which led to improved protection of the plants against peroxidation triggered by drought stress.
Changes in Levels of Nonenzymatic Antioxidant Compounds such as Ascorbic Acid (AsA), Total Phenolic Compounds, and Flavonoids
Abiotic stresses such as drought, salinity, and temperature extremes can enhance the assembly of secondary metabolites such as phenols (Çirak et al. 2007). In this study, the content of total phenols and flavonoids showed a significant increase in both rice cultivars under investigation exposed to drought stress compared with fully irrigated plants (Figs. 2 and 3). Polyphenols are considered the most important secondary metabolite class of antioxidants, and they efficiently inhibit lipid peroxidation products by scavenging ROS; therefore, the polyphenols increase the adaptation to abiotic oxidative stress (Pandey and Rizvi 2009; Krol et al. 2014; Stagnari et al. 2016). Other studies showed an accumulation of phenolic compounds in response to drought in Spanish (Xu and Leskovar 2015), tomatoes (Patanè et al. 2021), and wheat (Ali et al. 2022). In addition, algal treatments of soil with either G. oblongata or T. ornate induced further accumulations of total phenols and flavonoids in both stressed rice cultivars (Fig. 2). This accumulation might be the cause of the strong effect of algal extracts in increasing the polyphenol content, especially the content of flavonoids, in stressed plants. Also, due to the accumulation of some enzymes such as phenyl ammonia lyase (PAL) as well as chalcone synthase (Naikoo et al. 2019). Mansori et al. (2015) mentioned that the application of the seaweeds U. rigida and F. spiralis induced the accumulation of total phenols in bean plants.
The nonenzymatic antioxidant ascorbic acid (AsA) was measured in both the investigated rice cultivars exposed to drought stress (Fig. 4). A more pronounced increase was observed in Giza 179, which was 73.6% over the control value. Our results supported the findings of Pourghasemian et al. (2020) for sesame plants exposed to water deficit conditions. These findings may be attributed to the highly significant antioxidant capacity of AsA to detoxify ROS, minimize MDA levels, and mitigate other types of physiological deterioration in plants; it is considered one of the defense mechanisms plants employ for drought tolerance (Miller et al. 2010; Xu et al. 2010). Moreover, soil amended with either G. oblongata or T. ornate led to increased AsA levels in the two investigated rice cultivars (see Fig. 4).
In this study, the total antioxidant capacity of rice seedlings and the evaluation of drought tolerance in rice plants were determined by fluorescence recovery by photobleaching (FRAP) and the DPPH assay for screening plants for stress tolerance (Rekika et al. 2005; Ozgen et al. 2006a, b). FRAP found that the antioxidant capacity markedly increased in both rice cultivars under drought stress compared with the fully irrigated plants (Fig. 5). Moreover, the application of G. oblongata or T. ornate extract to soil increased the total antioxidant capacity levels in both the investigated stressed rice cultivars. This finding may be because the seaweeds preserved the highest antioxidant activity in rice plants by forming a protective barrier on the surface of the plants and inhibiting the reduction in antioxidant activity. On the other hand, the evaluation of the total antioxidant capacity by the DPPH scavenging method showed an opposite trend to that of the FRAP method in both rice cultivars grown under drought stress compared with their corresponding controls (see Figs. 5 and 6).
Gene Expression by RT-PCR
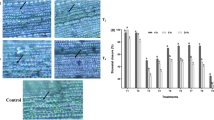
Plants have numerous types of aquaporin proteins, and they participate in the tolerance of and maintain the water balance of stressed plants (Hu et al. 2012; Ayadi et al. 2019). The plasma membrane intrinsic proteins (PIPs) belong to the AQP family and have a potential role in transporting water through the plasma membranes in many types of plants (Martre et al. 2002; Tyerman et al. 2002). In the present study, the results obtained from real-time polymerase chain reaction (RT-PCR) analysis revealed that drought stress induced a downregulation of PIP1;4 and PIP2;7 expression. Our results were in accordance with those of Porcel et al. (2006), who studied PIPs in Glycine max and Lactuca sativa plants. This downregulation was accompanied by overexpression of the PIP1;4 and PIP2;7 genes in stressed rice plants grown in soil amended with G. oblongata or T. ornate (Fig. 7a and b) compared with their corresponding controls. This overexpression might have had a main role in retarding the growth of seedlings in response to drought conditions (Jang et al. 2007). The most pronounced downregulated gene expression value was observed in the PIP2;7 genes of Giza 179. In addition, the NCDE5 gene related to abscisic acid biosynthesis showed downregulation under drought stress in the Giza 179 cultivar; however, a reverse pattern was significantly observed in the rice-sensitive cultivar Giza 177 (see Fig. 7c). The soil amended with either G. oblongata or T. ornate enhanced the overexpression of the NCDE5 gene in the two stressed rice cultivars compared with the controls (see Fig. 7c). This might be due to stimulation by the PIP1;4 and PIP2;7genes of the synthesis of ABA in stressed plants, which played an efficient role in the tolerance of plants exposed to drought (Li et al. 2015). In the present study, the application of investigated seaweeds induced a partial stomatal closure, associated with changes in the expression levels of genes involved in ABA-responsive and antioxidant systems in response to drought stress to eliminate ROS and improve the drought tolerance of rice plants. Our results are in alliance with the findings of Ali et al. (2022) on ork plants. The maximum overexpression of the NCDE5 gene was noticeable in Giza 177; it reached an approximately 16-fold change. Transcription factors (TFs) are considered to be common regulators of many vital processes of plants, beginning during development and continuing with defense mechanisms. MYB is one TF that can play a crucial role in the maintenance of a plant’s tolerance to environmental stresses (Erpen et al. 2018). As the data in Fig. 7d show, the downregulation of Myb-R1gene expression in both Giza 177 and Giza 179 was observed under drought conditions. The amendment of soil by either G. oblongata or T. ornate improved the overexpression of the Myb-R1gene in the two stressed rice cultivars compared with the controls (see Fig. 7d). Chauhan et al. (2020) like us, found a significant difference in the overexpression of the OsMyb-R1 gene in rice plants grown in treated soil. Our results suggested that the overexpression of the OsMyb-R1gene, when triggered by drought stress, could be a main factor in the control of the physiological, biochemical, and molecular responses to stresses (Zhang et al. 2014).
Influence of amended soil with either Turbenaria ornate or Galaxaura oblongata on the relative mRNA gene expression fold change, which A represented PIP1;4, B PIP2;7, C NCDE 5; and D OsMyb-R1 of the two rice cultivars Giza 177 and Giza 179 grown under drought condition. Results are shown as a mean of three replicates; the bars on the column show ± SE
Finally, to summarize the comparisons between groups, the interrelationships among the investigated variables in terms of Pearson’s correlation test were plotted as a heatmap (Fig. 8). The blue boxes in Fig. 8 indicate a positive correlation between variables, and the red boxes indicate a negative correlation. The principle component analysis (PCA) summarized the similarities and arranged the different samples in four groups: group 1 consisted of samples 11, 12, 5, and 6; group 2 of samples 8, 9, 3, and 2; group 3 of samples 10 and 4; and group 4 of samples 1 and 7 (Fig. 9).
Heatmap showing the correlation between different treatments. The numbers represented in this figure showed the different treatments as follows: 1 and 7, control; 2 and 8, Turbenaria ornate; 3 and 9, Galaxaura oblongata; 4 and 10, drought; 5 and 11, drought + Turbenaria ornate; and 6 and 12, drought + Galaxaura oblongata of cv. Giza 177 and cv. Giza 179, respectively
Conclusions
In this finding, we examined and studied the mechanism of drought as well as the relative effect of seaweeds G. oblongata and T. ornate incorporated soil as biostimulant on two rice cultivars’ growth and crop productivity by determining some physiological and biochemical analyses as well as molecular aspects. Rice plants grown in algal-enriched soil under drought conditions presented adaption by accumulating some compatible solutes such as proline and soluble sugar to regulate cellular turgor and produce antioxidants like phenolic compounds and AsA accompanied with diminished the levels of MDA and H2O2 to counter oxidative damage. They also upregulation PIP1;4, PIP2;7, NCDE5, and Myb-R1 drought-responsive genes, improving rice tolerance and stress resilience. We found that this mechanism of tolerance worked better in the drought-resistant cultivar Giza 179 than in the drought-sensitive cultivar Giza 177. Further research is needed for comprehensive insights.
Data Availability
The manuscript has associated data in a data repository. The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
References
Abd El-Hameid AR, Sadak MS (2020) Impact of glutathione on enhancing sunflower growth and biochemical aspects and yield to alleviate salinity stress. Biocatal Agric Biotechnol 29:101744
Abd Elhamid EM, Sadak MS, Tawfik MM (2016) Physiological response of Fenugreek plant to the application of proline under different water regimes. Res J Pharmac Biol Chem Sci 7(3):580–594
Abd Elhamid EMA, Sadak MS, Ezzo MI, Abdalla AM (2021) Impact of glycine betaine on drought tolerance of Moringa oleifera plant grown under sandy soil. Asian J Plant Sci 20:578–589
Agre P (2006) The aquaporin water channels. Proc Am Thorac Soc 3:5–13
Aharon R, Shahak Y, Wininger S, Bendov R, Kapulnik Y, Galili G (2003) Overexpression of a plasma membrane aquaporin in transgenic tobacco improves plant vigor under favorable growth conditions but not under drought or salt stress. Plant Cell 15:439–447
Ahmad S, Ahmad R, Ashraf MY, Ashraf M, Waraich EA (2009) Sunflower (Halianthusannuus L.) to drought stress at germination and seedling stages. Pak J Bot 41:647–654
Ahmed MMR, Sadak MS (2016) Effect of putrescine foliar application on wheat genotypes (Triticum aestivum L.) under water stress conditions. Int J Pharm Technol Res 9(8):94–102
Aleem AA (1993) The marine algae of Alexandria; Alexandria privately published: Alexandria, Egypt p: 139
Alharbi BM, Abdulmajeed AM, Hassan H (2021) Biochemical and molecular effects induced by triacontanol in acquired tolerance of rice to drought stress. Genes 12:1119
Alharbi K, Amin MA, Ismail MA, Ibrahim MTS, Hassan SE, Fouda A, Eid AM, Said HA (2022) Alleviate the drought stress on Triticumaestivum L. using the algal extracts of Sargassum latifolium and Corallina elongate versus the commercial algal products. Life 12:1757
Ali O, Ramsubhag A, Farrell AD, Jayaraman J (2022) Foliar application of seaweed extracts influences the phytomicrobiome dynamics in tomato and sweet pepper plants. J Appl Phycol 34:3219–3235
Anjum F, Yaseen M, Rasul E, Wahid A, Anjum S (2003) Water stress in barley (HordeumvulgareL.). I. Effect on morphological characters. Pak J Agric Sci 40:43–44
Anjum NA, Umar S, Iqbal M, Khan NA (2011) Cadmium causes oxidative stress in mung bean by affecting the antioxidant enzyme system and ascorbate-glutathione cycle metabolism. Russ J Plant Physiol 58:92–99
Arioli T, Mattner SW, Winberg PC (2015) Applications of seaweed extracts in Australian agriculture: Past, present and future. J Appl Phycol 275:2007–2015
Arjenaki GFG, Jabbari AR, Morshedi A (2012) Evaluation of drought stress on relative water content, chlorophyll content and mineral elements of wheat (Triticumaestivum L.) varieties. Intl J Agricult Crop Sci 4(11):726–729
Armengaud P, Thiery L, BuhotN G-D, Savouré A (2004) Transcriptional regulation of proline biosynthesis in Medicagotruncatula reveals developmental and environmental specific features. PhysiolgiaPlantarum 120(3):442–450
Arora A, Sairam RK, Sriuastava GC (2002) Oxidative stress and antioxidative system in plants. Curr Sci 82:1227–1238
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59:206–216
Ayadi M, Brini F, Masmoudi K (2019) Overexpression of a wheat aquaporin gene, TdPIP2;1, enhances salt and drought tolerance in transgenic durum wheat cv. Maali Int J Mole Sci 20:2389
Bakhoum GSH, Tawfik MM, Kabesh MO, Sadak MS (2023) Potential role of algae extract as a natural stimulating for wheat production under reduced nitrogen fertilizer rates and water deficit. Biocatal Agric Biotechnol 51:102794
Bakry BA, El-Hariri DM, Sadak MS, El-Bassiouny HMS (2012) Drought stress mitigation by foliar application of salicylic acid in two linseed varieties grown under newly reclaimed sandy soil. J Appl Sci Res 8(7):3503–3514
Bakry BA, Sadak MS, El-Karamany MF, Tawfik MM (2019) Sustainable production of two wheat cultivars under water stress conditions. Plant Arch 19(2):2307–2315
Bates LS, Waldren RP, Tear ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Bhatt RM, Rao NKS (2005) Influence of pod load response of okra to water stress. Indian J Plant Physiol 10:54–59
Blakeney AB, Mutton LL (1980) A simple colorimetric method for the determination of sugar in fruit and vegetables. J Sci Food Agric 31(9):889–897
Blokhina O, Virolainen E, Fagerstedt KV (2003) Antioxidants, oxidative damage and oxygen deprivation stress. Ann Bot 91:179–194
Bouman BAM, Tuong TP (2001) Field water management to save water and increase its productivity in irrigated rice. Agric Water Manag 49(1):11–30
Bray EA, Bailey-Serres J, Weretilnyk E (2000) Responses to abiotic stresses. Biochemistry and molecular biology of plant 1158–1203
Carvalho MEA, de Camargo e Castro PR, Gaziola SA, Azevedo RA (2018) Is seaweed extract an elicitor compound? Changing proline content in drought-stressed bean plants. Comunicata Scientiae 9(2):292–297
Chauhan R, Awasthi S, Indoliya Y, Chauhan AS, Mishra S, Agrawal L, Srivastava S, Dwivedi S, Singh PC, Mallick S, Chauhan PS (2020) Transcriptome and proteome analyses reveal selenium mediated amelioration of arsenic toxicity in rice (Oryza sativa L.). J Hazard Mater 122–122
Chen Y, Cao XD, Lu Y, Wang XR (2000) Effects of rare earth metal ions and their EDTA complexes on antioxidant enzymes of fish liver. Bull Environ Contam Toxicol 65:357–365
Chen X, Yang S, Ding J, Jiang Z, Sun X (2021) Effects of biochar addition on rice growth and yield under water-saving irrigation. Water 13:209–220
Çirak C, Radušienė J, Ivanauskas L, Janulis V (2007) Variation of bioactive secondary metabolites in Hypericumoriganifolium during its phenological cycle. Acta Physiolgiae Plantarum 29:197–203
Da Costa M, Huang B (2007) Drought survival and recuperative ability of bentgrass associated with abscisic acid and cytokinin production. J Am Horticult Sci 132:60–66
Datta A, Ullah H, Ferdous Z (2017) Water management in rice. In: Chauhan BS, Jabran K, Mahajan G (eds) Rice production worldwide. Springer, Dordrecht, the Netherlands, pp 255–277
Daughaday WH, Lowry OH, Rosebrough NJ, Fields WS (1952) Determination of cerebrospinal fluid protein with the folin phenol reagent. J Lab Clin Med 39:663–665
Dawood MG, El-Awadi ME, Abdel-Baky YR, Sadak MS (2017) Physiological role of ascobin on quality and productivity of sunflower plants irrigated with sodium chloride solution. Agricult Eng Intl 16–26
de Avila LA, Martini LFD, Mezzomo RF, Refatti JP, Campos R, Cezimbra DM, Machado ELO, Massey JH, Carlesso R, Marchesan E (2015) Rice water use efficiency and yield under continuous and intermittent irrigation. Climatol Water Manag 107(2):442–448
Dhargalkar VK, Pereira N (2005) Seaweed: Promising plant of themillennium. Sci Cult 71(3):60–66
Diaz J, Bernal A, Pomar F, Merino F (2001) Induction of shikimate dehydrogenase and peroxidase in pepper (Capsicum annuum L.) seedlings in response to copper stress and its relation to lignification. Plant Sci 161(1):179–188
Doganlar ZB, Atmaca M (2011) Influence of airborne pollution on Cd, Zn, Pb, Cu, and Al accumulation and physiological parameters of plant leaves in Antakya (Turkey). Water, Air, Soil Pollution 214(1–4):509–523
Dutot M, Fagon R, Hemon M, Rat P (2012) Antioxidant, anti-inflammatory, and anti-senescence activities of a phlorotannin-rich natural extract from brown seaweed Ascophyllum nodosum. Appl Biochem Biotechnol 167:2234–2240
El Boukhari ME, Barakate M, Drissi B, Bouhia Y, Lyamlouli K (2023) Seaweed extract biostimulants differentially act in mitigating drought stress on faba bean (Viciafaba L.). J Plant Growth Regul 42:5642–5652
Elansary HO, Yessoufou K, Abdel-Hamid AME, El-Esawi MA, Ali HM, Elshikh MS (2017) Seaweed extracts enhance salam turfgrass performance during prolonged irrigation intervals and saline shock. Front Plant Sci 8:830
El-Esawi MA, Al-Ghamdi AA, Ali HM, Alayafi AA, Witczak J, Ahmad M (2018) Analysis of genetic variation and enhancement of salt tolerance in french pea. Int J Mole Sci 19:2433
El-Esawi M, Al-Ghamdi AA, Ali HM, Ahmad M (2019) Overexpression of AtWRKY30 transcription factor enhances heat and drought stress tolerance in wheat (Triticum aestivum L.). Genes 10:163
Elewa TA, Sadak MS, Saad AM (2017) Proline treatment improves physiological responses in quinoa plants under drought stress. Biosci Res 14(1):21–33
El-Sayed S, Hellal F, Nofal O, El-Karamany M, Bakry B (2015) Influence of algal extracts on yield and chemical composition of moringa and alfalfa grown under drought condition. Int J Environ 4:151–157
Embiale A, Hussein M, Husen A, Sahile S, Mohammed K (2016) Differential sensitivity of Pisumsativum L. cultivars to water-deficit stress: Changes in growth, water status, chlorophyll fluorescence and gas exchange attributes. J Agron 15(2):45–57
Erpen L, Devi HS, Grosser JW, Dutt M (2018) Potential use of the DREB/ERF, MYB, NAC and WRKY transcription factors to improve abiotic and biotic stress in transgenic plants. Plant Cell Tissue Organ Culture 132:1–25
Ezzo M, Ebtihal M, Elhamid A, Sadak MS, Abdalla AM (2018) Improving drought tolerance of moringa plants by using trehalose foliar treatments. Biosci Res 5(4):4203–4214
Faseela P, Sinisha AK, Brestic M, Puthur JT (2019) Chlorophyll a fluorescence parameters as indicators of a particular abiotic stress in rice. Photosynthesis 57(SI):108–115
Fouda A, Eid AM, Abdelkareem A, Said HA, El-Belely EF, Alkhalifah DHM, Alshallash KS, Hassan SED (2022) Phycosynthesized zinc oxide nanoparticles using marine macroalgae, UlvafasciataDelile, characterization, antibacterial activity, photocatalysis, and tanning wastewater treatment. Catalysts 12:756
Foyer CH, Noctor G (2002) Oxygen processing in photosynthesis: Regulation and signalling. New Phytol 146:359–388
Ghouri F, Ali Z, Naeem M, Ul-Allah S, Babar M, Baloch FS, Chattah WS, Shahid MQ (2021) Effects of silicon and selenium in alleviation of drought stress in rice. SILICON 14:1–9
Gill PK, Sharma AD, Singh P, Bhullar SS (2002) Osmotic stress-induced changes in germination, growth and soluble sugar content of Sorghum bicolor (L.) Moench seeds. Bulg J Plant Physiol 28:12–25
Gowda VRP, Henry A, Yamauchi A (2011) Root biology and genetic improvement for drought avoidance in rice. Food Crop Res 122:1–13
Gunes A, Inal A, Bagci EG, Pilbeam DJ (2007) Silicon mediated changes of some physiological and enzymatic parameters symptomatic for oxidative stress in spinach and tomato grown in sodic-B toxic soil. Plant Soil 290:103–114
Harborne JB (1998) Photochemical methods: a guide to modern technique of plant analysis, 3rd Ed. Champman and Hall, London, UK p. 302
Hashem HA, Mansour HA, El-Khawas SA, Hassanein RA (2019) The potentiality of marine macro-algae as bio-fertilizers to improve the productivity and salt stress tolerance of canola (Brassica napus L.) plants. Agronomy 9:146
Hernández-Herrera RM, Santacruz-Ruvalcaba F, Ruiz-López MA, Norrie J, Hernández-Carmona G (2013) Effect of liquid seaweed extracts on growth of tomato seedlings (Solanum lycopersicum L.). J Appl Phycol 26:619–628
Homme PM, Gonzalez B, Billard J (1992) Carbohydrate content, frutane and sucrose enzyme activities in roots, stubble and leaves of rye grass (Lolium perenne L.) as affected by sources/link modification after cutting. J Plant Physiol 140:282–291
Hu W, Yuan Q, Wang Y, Cai R, Deng X, Wang J, Zhou S, Chen M, Chen L, Huang C, Ma Z, Yang G, He G (2012) Overexpression of a wheat aquaporin gene, TaAQP8, enhances salt Stress tolerance in transgenic tobacco. Plant Cell Physiol 53(12):2127–2141
Hussein MH, Eltanahy E, Al Bakry AF, Elsafty N, Elshamy MM (2021) Seaweed extracts as prospective plant growth bio-stimulant and salinity stress alleviator for Vigna sinensis and Zea mays. J Appl Phycol 33:1273–1291
Ivanov A, Pawlikowski J, Manoharan I, van Tuyn J, Nelson DM, Rai TS, Shah PP, Hewitt G, Korolchuk VI, Passos JF, Wu H, Berger SL, Adams PD (2013) Lysosome-mediated processing of chromatin in senescence. J Cell Biol 202(1):129–143
Jacomassi LM, de Viveiros JO, Oliveira MP (2022) A seaweed extract-based biostimulant mitigates drought stress in sugarcane. Front Plant Sci 13:865291
Jaleel CA, Gopi R, Panneerselvam R (2008) Growth and photosynthetic pigments responses of two varieties of Catharanthusroseus to triadimefon treatment. Comp Rend Biol 331:272–277
Jaleel CA, Manivannan P, Sankar B, Kishorekumar A, Gopi R, Somasundaram R, Panneerselvam R (2007) Induction of drought stress tolerance by ketoconazole in Catharanthus roseus is mediated by enhanced antioxidant potentials and secondary metabolite accumulation. Colloids Surf B: Biointerfaces 60:201–206
Jang JY, Lee SH, Rhee JY, Chung GC, Ahn SJ, Kang H (2007) Transgenic Arabidopsis and tobacco plants overexpressing an aquaporin respond differently to various abiotic stresses. Plant Mol Biol 64:621–632
Jiang Y, Huang B (2002) Protein alterations in tall fescue in response to drought stress and abscisic acid. Crop Sci 42:202–207
Kaldenhoff R, Fischer M (2006) Functional aquaporin diversity in plants. Biochimica et Biophysica Acta 1134–1141
Kapur B, Sarıda MA, Çeliktopuz E (2018) Health and taste related compounds in strawberries under various irrigation regimes and bio-stimulant application. Food Chem 263:67–73
Kar M, Mishra D (1976) Catalase, peroxidase and polyphenol oxidase activities during rice leaf senescence. Plant Physiol 57:315–319
Kartashov AV, Radyukina NL, Ivanov YuV, PashkovskiiPP SNI, Kuznetsov VV (2008) Role of antioxidant systems in wild plant adaptation to salt stress. Russ J Plant Physiol 55:463–468
Keyvan S (2010) The effects of drought stress on yield, relative water content, proline, soluble carbohydrates and chlorophyll of bread wheat cultivars. J Animal Plant Sci 8(3):1051–1060
Khan W, Rayirath UP, Subramanian S, Jithesh MN, Rayorath P, Hodges DM, Critchley A, Craigie JS, Norrie J, Prithiviraj B (2009) Seaweed extracts as biostimulants of plant growth and development. J Plant Growth Regul 28:386–399
Koricheva J, Roy S, Vranjic JA, Haukioja E, Hughe PR, Hanninen O (1997) Antioxidant responses to simulated acid rain and heavy metal deposition in brich seedlings. Environ Pollut 95:249–258
Krol A, Amarowicz R, Weidner S (2014) Changes in the composition of phenolic compounds and antioxidant properties of grapevine roots and leaves (Vitisvinifera L.) under continuous of long-term drought stress. Acta Physiologica Plantarum 36:1491–1499
Kumar A (2014) Activity and mechanism of Ascophyllum nodosum extract induced salinity tolerance in tomato. Master’s Thesis, Dalhousie University Halifax, Halifax, NS, Canada
Kumareswari T, Rani MS (2015) Utilization of seaweeds to enhance growth and nutritive status of Amaranthuscaudatus L. Int J Res Stud Biosci 3:9–15
Kusaka M, Ohta M, Fujimura T (2005) Contribution of inorganic components to osmotic adjustment and leaf folding for drought tolerance in pearl millet. Physiologia Plantarum 125:474–489
Lamhamdi M, Bakrim A, Aarab A, Lafont R, Sayah FA (2010) Comparison of lead toxicity using physiological and enzymatic parameters on spinach (Spinacia oleracea) and wheat (Triticum aestivum) growth. Mor J Biol 6(7):64–73
Layek J, Das A, Idapuganti RG, Sarka D, Ghosh A, Zodape ST, Lal R, Yadav GS, Panwar AS, Ngachan S (2018) Seaweed extract as organic bio-stimulant improves productivity and quality of rice in eastern Himalayas. J Appl Phycol 30:547–558
Lefebvre V, North H, Frey A, Sotta B, Seo M, Okamoto M, Nambara E, Marion-Poll A (2006) Functional analysis of Arabidopsis NCED6 and NCED9 genes indicates that ABA synthesized in the endosperm is involved in the induction of seed dormancy. Plant J 45:309–319
Li S, Li X, Wei Z, Liu F (2020) ABA-mediated modulation of elevated CO2 on stomatal response to drought. Curr Opin Plant Biol 19:30117–30117
Li J, Ban L, Wen H, Wang Z, Dzyubenko N, Chapurin V, Gao H, Wang X (2015) An aquaporin protein is associated with drought stress tolerance. Biochem Biophys Res Commun 208–213
Lipiec J, Doussan C, Nosalewicz A, Kondracka K (2013) Effect of drought and heat stresses on plant growth and yield: A review. Int Agrophys 27:463–477
Liu C, Liu Y, Guo K, Fan D, Li G, Zheng Y (2011) Effect of drought on pigments, osmotic adjustment and antioxidant enzymes in six woody plant species in karst habitats of Southwestern China. Environ Exp Bot 71:174–183
Liu W, Yu K, He T, Li F, Zhang D, Liu J (2013) The low temperature induced physiological responses of Avenanuda L., a cold-tolerant plant species. Transfus. Apher. Science, Article ID 658793, 7 pages
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real time quantitative PCR and the 2a Act method. Methods 25:402–408
Mahanty T, Bhattacharjee S, Goswami M, Bhattacharyya P, Das B, Ghosh A, Tribedi P (2017) Biofertilizers: A potential approach for sustainable agriculture development. Environ Sci Pollut Res 24:3315–3335
Mahmood T, Abdullah M, Ahmar S, Yasir M, Iqbal MS, Yasir M (2020) Incredible role of osmotic adjustment in grain yield sustainability under water scarcity conditions in wheat (Triticum aestivum L.). Plants 9:1208
Maisura M, Chozin MA, Lubis I, Junaedinand A, Ehara H (2014) Some physiological character responses of rice under drought conditions in a paddy system. J Int Soc Southeast Asian Agric Sci 20(1):104–114
Makkar HPS, Blümmel M, Borowy NK, Becker K (1993) Gravimetric determination of tannins and their correlations with chemical and protein precipitation methods. J Sci Food Agr 61(2):161–165
Mansori M, Chernane H, Latique S, Benaliat A, Hsissou D, El Kaoua M (2015) Seaweed extract effect on water deficit and antioxidative mechanisms in bean plants (Phaseolus vulgaris L.). J Appl Phycol 27:1689–1698
Martre P, Raphae M, Barrieu F, Gretchen B, North PS, Nobel MJ, Chrispeels (2002) Plasma membrane aquaporins play a significant role during recovery from water deficit1. Plant Physiol 130:2101–2110
Mazzola M, Brown J (2010) Efficacy of brassicaceous seed meal formulations for the control of apple replant disease in conventional and organic production systems. Plant Dis 94:835e842
Metzner H, Rau H, Senger H (1965) Untersuchungenzursynchronisierbarkeiteinzelnerpigmentmangel-mutanten von Chlorella. Planta 65:186–194
Miller G, Suzuki N, Cift-Yilmaz S, Mittler R (2010) Reactive oxygen species homeostasis and signaling during drought and salinity stresses. Plant Cell Environ 33:453–467
Minotti G, Aust SD (1987) The requirement for iron (III) in the initiation of lipid peroxidation by iron (II) and hydrogen peroxide. J Biol Chem 262(3):1098–1104
Monakhova OF, Chernyadev II (2002) Protective role of kartolin-4 in wheat plants exposed to soil drought. Appl Environ Microbiol 38:373–380
Mukheriee SP, Choudhuri MA (1983) Implication of water stress-induced changes in the level of endogenous ascorbic acid and hydrogen peroxide in vigna seedling. Physiol Plant 58:166–170
Nabti E, Jha B, Hartmann A (2017) Impact of seaweeds on agricultural crop production as biofertilizer. Int J Environ Sci Technol 14:1119–1134
Naikoo MI, Dar MI, Raghib F, Jaleel H, Ahmad B, Raina A (2019) Role and regulation of plants phenolics in abiotic stress tolerance: An overview, In Plant signaling molecules: role and regulation under stressful environments. Woodhead Publishing, Elsevier, Amsterdam, pp 157–168
Nedzarek A, Rakusa-Suszczewski S (2004) Decomposition of macroalgae and the release of nutrients into Admiralty Bay, King George Island, Antarctica. Polar Biosci 17:26–35
O’Sullivan AM, O’Callaghan YC, O’Grady MN, Queguineur B, Hannify D, Troy DJ, Kerry JP, O’Brien NH (2011) Vitro and cellular antioxidant of seaweed extracts prepared from five brown seaweeds harvested in spring from the west coast of Ireland. Food Chem 126:1064–1070
Oyaizu M (1986) Studies on products of browning reaction prepared from glucoseamine. Jap J Nutri 44:307–314
Ozgen M, Tulio AZ Jr, Reese RN, Scheerens JC, Miller AR (2006b) Modified 2,2-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid method to measure antioxidant capacity of selected small fruits and comparison to ferric reducing antioxidant power and 2,2,-diphenyl-1-picrylhydrazyl methods. J Agron Food Chem 54:1151–1157
Ozgen M, Tulio AZ, Jr, Chanon AM, Janakiraman N, Reese RN, Miller AR, Scheerens JC(2006a) Phytonutrient accumulation and antioxidant capacity at eight developmental stages of black raspberry fruit. Horticulture science 41: 1082. (abstr.)
Pandey KB, Rizvi SI (2009) Plant polyphenols as dietary antioxidants in human health and disease. Oxid Medical and Cell Longev 2(5):270–278
Patanè C, Siah S, Pellegrino A, Salvatore L, Cosentino SL (2021) Fruit yield, polyphenols, and carotenoids in long shelf-life tomatoes in response to drought stress and rewatering. Agronomy 11:1943
Porcel R, Aroca R, Azco´n R, Ruiz-Lozano JM (2006) PIP aquaporin gene expression in arbuscula rmycorrhizal Glycine max and Lactuca sativa plants in relation to drought stress tolerance. Plant Mole Biol 60:389–404
Pourghasemian N, Moradi R, Naghizadeh M, Landberg T (2020) Mitigating drought stress in sesame by foliar application of salicylic acid, beeswax waste and licorice extract. Agric Water Manag 231(31):105997
Quampah A, Wang RM, Shamsi IH, Jilani G, Zhang Q, Hua S, Xu H (2011) Improving water productivity by potassium application in various rice genotypes. Int J Agric Biol 13:9–17
Quigley F, Rosenberg JM, Shachar-Hill Y, Bohnert HJ (2002) From genome to function: The Arabidopsis aquaporins. Genome Biol 3:1–17
Reddy AR, Chaitanya KV, Vivekanandan M (2004) Drought induced responses of photosynthesis and antioxidant metabolism in higher plants. J Plant Physiol 161:1189–1202
Rekika D, Khanizadeh S, Deschenes M, Levasseur A, Charles MT (2005) Antioxidant capacity and phenolic content of selected strawberry genotypes. Horticult Sci 40:1777–1781
Renuka R, Rathinavel S (2006) Alleviating potential of seaweed liquid extract of Enteromorpha prolifera on salt induced changes in cluster bean plants. Plant Arch 6:845–848
Rolland F, Baena-Gonzalez E, Sheen J (2006) Sugar sensing and signaling in plants: Conserved and novel mechanisms. Ann Rev Plant Biol 57:675–709
Sadak MS (2016) Mitigation of drought stress on Fenugreek plant by foliar application of trehalose. Intl J Chem Technol Res 9(2):147–155
Sadak MS, Bakry BA (2020) Alleviation of drought stress by melatonin foliar treatment on two flax varieties under sandy soil. Physiol Mole Biol Plant 26(5):907–919
Sadak MS, El-Enany MAM, Bakry BA, Abdallah MMS, El-Bassiouny HMS (2020) Signal molecules improving growth, yield and biochemical aspects of wheat cultivars under water stress. Asian J Plant Sci 19:35–53
Sakamoto A, Murata N (2002) The role of glycine betaine in the protection of plants from stress: Clues from transgenic plants. Plant Cell Environ 25:163–171
Samal AC, Bhattacharya P, Biswas P, Maity JP, Bundschuh J, Santra SC (2021) Variety-specific arsenic accumulation in 44 different rice cultivars (O. sativa L.) and human health risks due to co-exposure of arsenic-contaminated rice and drinking water. J Hazard Mater 704:124–804
Sangha JS, Kelloway S, Critchley AT, Prithiviraj B (2014) Seaweeds (macroalgae) and their extracts as contributors of plant productivity and quality: The current status of our understanding. Adv Bot Res 71:189–219
Seregin IV, Ivanov VB (2001) Physiological aspects of cadmium and lead toxic effects on higher plants. Russ J Plant Physiol 48:523–544
Shehab GG, Ahmed OK, El-Beltagi HS (2010) Effects of various chemical agents for alleviation of drought stress in rice plants (Oryza sativa L.). Notulae Botanicae horticulture Agrobotanici Cluj Napoca 38(1):139–148
Snedecor GW, Cochran WG (1980) Statistical methods, 5th edn. Iowa State University Press, Ames, Iowa, USA, p, p 276
Stagnari F, Galieni A, Pisante M (2016) Drought stress effects on crop quality. In: Ahmad P (ed) In Water stress and crop plants: A sustainable approach, vol 2. New York, NY, USA: John Wiley & Sons Limited, pp 375–392
Sujata GV, Baliyan V, Avtar R, Mehrotra S (2023) Alleviating drought stress in Brassica juncea (L.) Czern & Coss. by foliar application of biostimulantsorthosilicic acid and seaweed extract. Appl Biochem Biotechnol 195:693–721
Szabados L, Savoure A (2010) Proline: A multifunctional amino acid. Trend Plant Sci 15(2):89–97
Trovato M, Matioli R, Costantino P (2008) Multiple roles of proline in plant stress tolerance and development. Rendiconti Lincei 19:325–346
Tyerman SD, Niemietz CM, Bramley H (2002) Plant aquaporins: Multifunctional water and solute channels with expanding roles. Plant Cell Environ 25:173–194
Uga Y, Sugimoto K, Ogawa S, Rane J, Ishitani M, Hara N (2013) Control of root system architecture by deeper rooting 1 increases rice yield under drought conditions. Natl Gen 45:1097–1102
Usman M, Fishan Z, Ahsan RA, Iqbal A, Sarfaraz ZN, Haq Z (2013) Morphological, physiological and biochemical attributes as indicators for drought tolerance in rice (Oryzasativa L.). Eur J Biol Sci 5(1):23–28
Wally OSD, Critchley AT, Hiltz D, Craigie JS, Han X, Zaharia LI, Abrams SR, Prithiviraj B (2013) Regulation of phytohormone biosynthesis and accumulation in arabidopsis following treatment with commercial extract from the marine macroalga, Ascophyllum nodosum. J Plant Growth Regul 32(2):324–339
Xu C, Leskovar DI (2015) Effects of A. nodosum seaweed extracts on spinach growth, physiologyand nutrition value under drought stress. Sci Hortic 183:39–47
Xu J, Tian YS, Peng RH, Xiong AS, Zhu B, Jin XF, Gao F, Fu XY, Hou XL, Yao QH (2010) AtCPK6, a functionally redundant and positive regulator involved in salt/drought stress tolerance in Arabidopsis. Planta 231:1251–1260
Yamaguchi T, Takamura H, Matoba T, Terao J (1998) HPLC method for evaluation of the free radical scavenging activity of foods by using 1,1-diphenyl-2-picrylhydrazyl. Biosci Biotechnol Biochem 62(6):1201–1204
Yang J, Guo Z (2007) Cloning of a 9-cis-epoxycarotenoid dioxygenase gene (SgNCED1) from Stylosanthesguianensis and its expression in response to abiotic stresses. Plant Cell Reprod 26:1383–1390
Yu CW, Murph TM, Lin CH (2003) Hydrogen peroxide-induces chilling tolerance in mung bean mediated through ABA-independent glutathione accumulation. Funct Plant Biol 30:955–963
Zhang X, Ervin EH (2008) Impact of seaweed extract-based cytokinins and zeatinriboside on creeping bentgrass heat tolerance. Crop Sci 48:364–370
Zhang L, Liu G, Zhao G, Xia C, Jia J, Liu X, Kong X (2014) Characterization of a wheat R2R3-MYB transcription factor gene, TaMYB19, involved in enhanced abiotic stresses in Arabidopsis. Plant Cell Physiol 55(10):1802–1812
Zhou J, Wang X, Jiao Y, Qin Y, Liu X, He K, Chen C, Ma L, Wang J, Xiong L, Zhang Q, Fan L, Deng XW (2007) Global genome expression analysis of rice in response to drought and high-salinity stresses in shoot, flag leaf, and panicle. Plant Mol Biol 63:591–608
Author information
Authors and Affiliations
Contributions
Conceptualization, N.M.H and R.M.A.; methodology, N.M.H and R.M.A.; validation, N.M.H and R.M.A.; investigation, N.M.H.; writing—original draft preparation, N.M.H.; writing—review and editing, N.M.H and R.M.A.All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alshegaihi, R.M., Helal, N.M. The Biostimulant Potential of the Seaweeds Galaxaura oblongata and Turbenaria ornate in Improving Drought Tolerance in Rice Plant (Oryza sativa L.). Plant Mol Biol Rep (2024). https://doi.org/10.1007/s11105-024-01479-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11105-024-01479-w