Abstract
Background and aims
Understanding how long-term intercropping and phosphorus (P)-fertilizer application affect soil P fractions through P-acquisition strategies is critical to maintaining soil P balance in agroecosystems.
Methods
We established a long-term field experiment with three P-fertilizer application rates (0, 40, and 80 kg P ha−1) and continuously used four intercropping systems of chickpea/maize, faba bean/maize, oilseed rape/maize, soybean/maize and corresponding five monocultures in 2009. We measured aboveground biomass, shoot P content, soil P fractions, P-related root physiological traits, and soil microbe-related parameters of crop species in 2020. We also calculated the apparent soil P balance (P input into soil minus P harvested from crops) using data from 2009 to 2020.
Results
Intercropping enhanced aboveground biomass and shoot P content by 31.2% and 49.4% compared with the weighted means of corresponding monocultures, respectively; intercropping decreased the apparent soil P balance by 37.8% compared with monocultures across three P-fertilizer application rates. Over the 12-year period, chickpea/maize and soybean/maize intercropping systems significantly decreased the soil organic P concentration compared with sole maize; faba bean/maize and oilseed rape/maize intercropping systems significantly decreased soil non-labile P but increased organic P and labile P pool relative to sole maize. Rhizosheath phosphatases and carboxylates (proxied by leaf manganese concentration) might contribute to the depletion of sparingly-available soil P (organic P or non-labile P) in different crop combinations.
Conclusion
The higher rhizosheath acid phosphatase activities and carboxylate concentrations may correlate with efficient utilization of sparingly-available soil P resources in intercropping; effective P-fertilizer input enhanced soil P availability and decreased the P surplus in soil which is crucial to enhance crop P uptake.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Phosphorus (P) is a major limiting nutrient for plant productivity in terrestrial environments (Elser et al. 2007; Vitousek et al. 2010). The concentration of available P in soil is quite low, because soil minerals strongly sorb a large proportion of inorganic P or P is associated with organic matter, and hence P availability cannot meet the requirement of plant growth (Hinsinger 2001). Phosphorus-fertilizer input is, therefore, essential for sustainable food production in agriculture. However, excessive P fertilizers result in large amounts of P accumulating in soil and threaten water quality and biodiversity (Vorosmarty et al. 2010). Soil nutrient balance is an important indicator reflecting the losses to the environment. In recent years, a P surplus has accumulated in most farmland in China (Zhang et al. 2019). Moreover, phosphate rock is turned into P fertilizer for crop yield, but this is a non-renewable resource, and the reserves may become exhausted in 93 to 291 years (Cordell and White 2011; Fixen and Johnston 2012; Johnston et al. 2014). China is the major consumer and producer of P fertilizer, and its P reserves will be depleted within a hundred years with increasing P input (Fixen and Johnston 2012; Yu et al. 2021c). Therefore, it is urgent to develop P-management strategies to improve P-use efficiency and decrease P-fertilizer dependency. Combining species with efficient P-acquisition strategies to mine soil P pools in intercropping systems is a promising and economically viable approach to establishing sustainable food-production systems.
Plant species exhibit divergent P-mobilizing capacities (Li et al. 2014). Faba bean is more efficient at mobilizing P by releasing a large amount of carboxylates and protons in the rhizosphere. In contrast, the proton-releasing capacity of maize is too weak to acidify the rhizosphere (Li et al. 2007; Zhang et al. 2016). Plant species have evolved diverse P-acquisition strategies to obtain sufficient P to meet their growth requirement. For example, oilseed rape and maize mainly depend on adjustments of root morphological traits, e.g., an increase in specific root length and lateral root density, to acquire soil P (Calderon-Vazquez et al. 2009). Chickpea and white lupin release root phosphatases and carboxylates that hydrolyze and mobilize organic P; faba bean relies on both root morphological and physiological responses that mobilize P from insoluble inorganic P forms (Isaac and Borden 2019; Lambers 2022; Li et al. 2007; Lyu et al. 2016). When species with diverse P-acquisition strategies grow together, their P-use efficiency may be improved (Li et al. 2014; Yu et al. 2020).
Intercropping attracts increasing attention as a sustainable cultivation method, which can deliver yield advantage and increase soil fertility (Li et al. 2021; Xing et al. 2023). Enhanced P-use efficiency and P uptake have been observed in intercropping, especially in legume/cereal intercropping systems (Tang et al. 2021; Tian et al. 2020a). The advantage of intercropping in P uptake is mainly mediated by complementarity and facilitation. Facilitation is defined as one or more species increasing a function metric (e.g., plant-availability of nutrients) of their neighbors (Brooker et al. 2015; Yu et al. 2021b). For example, white lupin increases the P-acquisition of intercropped maize via mobilization of insoluble P into soluble P in the rhizosphere (Dissanayaka et al. 2015). Complementarity in P uptake suggests P partitioning between species and reduced interspecific competition, leading to greater P acquisition (Hinsinger 2001). For example, in pigeon pea/cereal intercropping systems, pigeon pea can use iron-bound P, while intercropped cereals rely on calcium-bound P (Ae et al. 1990). Thus, intercropping may allow access to different P fractions via various P-acquisition strategies to enhance P uptake in intercropping systems.
Soil microorganisms play an important role in the soil P cycle (Richardson and Simpson 2011). The P held in the biomass of microorganisms is protected from reactions with soil and is potentially available to plant species (Liebisch et al. 2014; Olander and Vitousek 2004). In addition, soil microorganisms may utilize organic P by producing extracellular enzymes, i.e. alkaline phosphatases, which are only produced by microorganisms (Nannipieri et al. 2011). Also, the phoD gene has been identified in coding for alkaline phosphatases in bacteria (Gomez and Ingram 1995). Different agronomic managements (e.g., nutrient input and cropping systems) may shift microbial functional profiles of P cycling (Dai et al. 2020; Yu et al. 2021a). A greenhouse experiment also showed that soil microorganisms are associated with intercropping overyielding in faba bean/wheat and faba bean/maize intercropping systems (Wang et al. 2021). Hence, associating soil microbial parameters and P-acquisition strategies with P fractions may provide a novel insight into P utilization in agriculture.
Most previous studies focused on soil P-fraction dynamics and P uptake in a single intercropping system, whereas few linked P-mobilizing capacities with soil P pools in diverse intercropping systems (Liao et al. 2020, 2021; Qu et al. 2022; Tian et al. 2020a). Furthermore, the correlations between P-acquisition strategies and P pools in different crop combinations under field conditions are largely unknown. In this study, we aimed to assess the P pools in soil with three P-application rates (0, 40, and 80 kg P ha−1) and with four intercropping systems with different species combinations and corresponding species monocultures, and also to explore how plant P-acquisition strategies impact soil P pools, using a long-term field experiment. We also measured physiological and microbial parameters in the rhizosheath, namely: pH; acid phosphatase activity; alkaline phosphatase activity; soil microbial biomass P; phoD gene abundance. Carboxylates were difficult to measure under field conditions, but leaf manganese concentration ([Mn]) is positively correlated with rhizosheath carboxylate concentrations (Lambers et al. 2021; Yu et al. 2023). Therefore, we measured leaf [Mn] as a proxy for P acquisition via carboxylate release in the rhizosphere. We addressed two key questions: (1) How do P-application rates and intercropping affect productivity, shoot P content, and apparent soil P balance of intercropping systems compared with monocultures? (2) How does intercropping affect soil P pools in different species combinations with different P-acquisition strategies under field conditions?
Material and methods
Study site
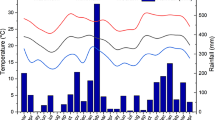
The field experiment is located at Baiyun Experimental Station of the Gansu Academy of Agricultural Sciences, Gansu Province (102˚40′E, 38˚37′N, 1504 m above sea level). This region has a typical arid climate with mean annual temperature and precipitation of 7.7˚C and 150 mm, respectively. Total solar radiation is 5988 MJ m−2 yr−1, and the average annual frost-free period is 170–180 days. The soil (0–20 cm) of the experimental field is classified as calcareous Aridisol and had the following average composition before establishment of the experiment: pH 8.0, organic matter 19.1 g kg−1, total nitrogen (N) 1.08 g kg−1, Olsen-P 20.3 mg kg−1, and available potassium (K) 233 mg kg−1. In 2020, the Olsen-P concentration in the soil under P0, P40, P80 was 4.8 mg kg−1,14.8 mg kg−1, and 29 mg kg−1, respectively.
Experiment design and crop management
The experiment was established in 2009, as a split-plot experimental design with three replicates. The main-plot factor comprised three P-application rates (0, 40, and 80 kg P ha−1 y−1). The 40 kg P ha−1 fertilizer-application rate is recommended by agronomists to meet the nutrient requirement of maize, and 80 kg P ha−1 is the common P-fertilizer application rate for local farmers.
The sub-plot factors were nine cropping systems comprising five monocultures: faba bean (Vicia faba L.cv. Lincan No.5), soybean (Glycine max L.cv. Zhonghuang No. 33), chickpea (Cicer arietimum L. cv. Longying No. 1), oilseed rape (Brassica campestris L. cv. Longyou No. 5) and maize (Zea mays L.cv. Xianyu No. 335), and four intercropping systems: faba bean/maize, soybean/maize, chickpea/maize, and oilseed rape /maize. The field was split into 81 plots, with each monocropped plot measuring 4 × 5.5 m2 and each intercropped plot measuring 5.6 × 5.5 m2. For maize, the inter-row distance was 0.4 m, and the inter-plant distance was 0.25 m. For legumes (i.e. faba bean, chickpea, and soybean), the inter-row distance and inter-plant distance were 0.2 m. For oilseed rape, the inter-row distance was 0.2 m, which was planted by broadcast sowing in each row. Every intercropped plot included four 1.4 m-wide intercropping strips, each of which consisted of two rows of maize and three rows of companion crops (i.e. faba bean, chickpea, soybean, and oilseed rape), with a row distance of 0.3 m between maize and the companion crop. In the intercropped area, maize occupied 57% and the companion crop occupied 43%.
All plots were given a basal application of 60 kg K ha−1 as potassium sulfate and corresponding P as superphosphate. Sole maize and intercropping systems were given an identical application of 225 kg N ha−1 as urea, one-third of N fertilizer (75 kg N ha−1) was applied as basal fertilizer, and the remaining N fertilizer was divided equally into two portions and applied at the stem elongation stage and the pre-tasselling stage, respectively (Li et al. 2021; Xing et al. 2023). The monoculture companion crops were given a basal application of 75 kg N ha−1 as urea, which was half of the total N fertilizer during the entire growth period (Li et al. 2021; Xing et al. 2023). Six irrigations were carried out on 12 May, 2 June, 23 June, 3 July, 29 July, and 15 August with 100 mm for monoculture and intercropping plots in 2020. The amounts of fertilizer and irrigation were recommended by local agronomists.
Faba bean and chickpea were sown on March 22 in 2020. Soybean and oilseed rape were sown on April 17 in 2020; maize was sown on April 26. Faba bean, chickpea, and oilseed rape were harvested in early July; maize and soybean were harvested in late September to early October 2020. At maturity, three continuous rows of companion crops and two rows of maize were harvested per plot and separated into grain and straw. All samples were air-dried for two weeks and weighed.
Soil and plant analyses
Soil and mature leaf samples were collected during flowering stage, with samples of faba bean, chickpea, and oilseed rape collected in June, and the samples of soybean and maize were collected in July. We dug up the roots of the five crops from the 0–30 cm soil layer, then shook the roots to remove loosely adhering soil. The rhizosheath soil was brushed from 5–10 individuals according to the different sizes of the crops. Bulk soil samples of 0–20 cm were collected using an auger (3.5 cm diameter) in all plots. In the monoculture plots, the bulk soil was collected from five cores. In the intercropping plots, the bulk soil was separately collected from each crop strip for five cores. The bulk soil samples were air-dried for measurement of soil P fractions and Olsen-P. All rhizosheath soil samples were mixed through a 2-mm sieve and divided into three parts; one was stored at 4˚ for measurement of alkaline and acid phosphatase activity, and microbial biomass P; one was air-dried for measurement of soil pH and P fractions; another subsample was stored at -20˚C for DNA extraction.
Soil pH was measured in a 10 mM calcium chloride extract (1:2.5) by using an S210 pH meter (Mettler Toledo, Zurich, Switzerland). Soil microbial biomass P was determined after fumigation with CHCl3 for 24 h and then extracted with 0.5 M NaHCO3 (Brookes et al. 1982). Olsen-P was extracted with 0.5 M NaHCO3 and measured by molybdenum blue colorimetry (Bao 2005). Alkaline phosphatase and acid phosphatase activity were measured with p-nitrophenol (PNP) as substrate at pH 8.5 and 5.2 using spectrophotometrically at 405 nm, respectively (Tabatabai and Bremner 1969). Shoot P concentration was analyzed using the vanadomolybdate method (Bao 2005). Leaf [Mn] was measured by inductively-coupled plasma optical-emission spectroscopy (ICP-OES; OPTIMA 3300 DV, Perkin-Elmer, Waltham, MA, USA) using mature leaves.
Soil P fractionation
The air-dried bulk and rhizosheath soil samples were sieved through a 0.15-mm mesh and then 0.5 g was used to analyze sequential P fractions. The soil P fractionation was carried out according to a sequential fractionation scheme (Tiessen and Moir 1993) modified from methods described by Hedley et al. (1982). When assessing the soil P fractions, we accept that the biological significance, if any, of some of these fractions is poorly understood (Barrow et al. 2021). In the fractionation scheme, soil P was divided into nine fractions. i.e. resin-extractable P (Resin-P), bicarbonate-extractable P (NaHCO3-Pi and NaHCO3-Po), OH-extractable P (NaOH-Pi and NaOH-Po), dilute HCl-extractable P (1 M HCl-Pi), concentrated HCl-extractable P (conc. HCl-Pi and conc. HCl-Po) and concentrated H2SO4-H2O2-extractable P (residual-P). After adding each extractant, the suspension was first placed on a shaker (200 rpm) for 16 h, then centrifuged at 25,000 g for 10 min at 0˚C; then the corresponding extract was decanted through a 0.45-μm membrane filter into a clean vial for the colorimetric analysis. The inorganic P fraction and residual-P were measured by the method of Murphy and Riley (1962). The total P concentrations (NaHCO3-Pt, NaOH-Pt, and conc. HCl-Pt) were analyzed using the ammonium persulfate digestion method (Li et al. 2008), and the organic P concentrations were obtained by subtracting the inorganic P concentration from the total P concentration (Pt). Total inorganic P was calculated as the sum of Resin-P, NaHCO3-Pi, NaOH-Pi, 1 M HCl-Pi, and conc. HCl-Pi. Total organic P was calculated by adding NaHCO3-Po, NaOH-Po, and conc. HCl-Po. To make comparisons of trends in labile and less-soluble P fractions, we combined the nine P fractions into four P pools according to the activity of the P fraction, including labile P (sum of Resin-P, NaHCO3-Pi, and NaHCO3-Po), moderately-labile P (sum of NaOH-Pi, NaOH–Po, and 1 M HCl-Pi), sparingly-labile P (conc. HCl-Pi and conc. HCl-Po) and non-labile P (residual-P) (Crews and Brookes 2014; Liao et al. 2020).
DNA extraction and sequencing
The DNA was extracted from 0.5 g frozen soil using the FastDNA SPIN kit (MP Bio-medicals, Solon, OH, USA) following the manufacturer’s instructions. The eluted DNA was stored at -20 °C for short-term storage. DNA quality was monitored by a NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies Inc., Wilmington, DE).
Quantitative PCR
Bacterial phoD genes were determined by quantitative polymerase chain reaction (qPCR) with the primers ALPS-F730 (5′-CAGTGGGACGACCACGAGGT-3′) and ALPS-R1101 (5′-GAGGCCGA TCGGCATGTCG-3′) (Sakurai et al. 2008) on the C1000 Touch Thermal Cycler real-time PCR system (BIORAD, Hercules, California, USA). The PCR mixture (25 μL) contained 13 μL of 2 × TB Green Premix Ex Taq (Takara, Japan), 0.5 μL of each primer, 2 μL of DNA, and 9 μL of sterile ddH2O. Cycling conditions were as follows: 95 °C for 3 min, 40 cycles of 95 °C for 5 s, and 58 °C for 30 s (Wei et al. 2019). The standard curve was prepared by serial tenfold dilutions of plasmid. The qPCR results were considered acceptable when the R2 of the standard curve > 0.98.
Calculations
The aboveground biomass produced by intercropping can be compared with the weighted means of two monoculture crops based on their proportions in the intercropping systems. The weighted means of biomass in monocultures were calculated using the following equation:
where a represents maize, while b represents companion crop (i.e. oilseed rape, chickpea, faba bean, or soybean); \({\mathrm{B}}_{\mathrm{monoculture\_a}}\) and \({\mathrm{B}}_{\mathrm{monoculture\_b}}\) represent the biomass of crops a and b in monoculture; \({\mathrm{P}}_{\mathrm{a}}\) and \({\mathrm{P}}_{\mathrm{b}}\) refer to the proportions of the area in the intercropping systems, where \({\mathrm{P}}_{\mathrm{a}}\)= 57% and \({\mathrm{P}}_{\mathrm{b}}\)= 43%. The above formula was also used to calculate the weighted means of shoot P content, soil total inorganic P, soil total organic P and soil residual-P in monocultures.
The soil total inorganic P concentrations in intercropping systems were calculated using the following equation:
where \({\mathrm{P}}_{{\mathrm{i}}\mathrm{\_i}\mathrm{ntercropping\_a}}\) and \({\mathrm{P}}_{{\mathrm{i}}\mathrm{\_i}\mathrm{ntercropping\_b}}\) represent the soil total inorganic P concentration of crops a and b in intercropping. The above formula was also used to calculate the concentrations of soil total organic P, nine soil P fractions, four P pools, aboveground biomass and the shoot P content in intercropping systems.
The apparent P balance is the difference between P inputs and P removal from aboveground biomass, which was calculated using the following equation (Hua et al. 2016; Liao et al. 2021):
where F represents P-application rates (0, 40, and 80 kg ha−1), \({\mathrm{PC}}_{\mathrm{i}}\) represent the shoot P content of cropping systems in i (2009–2020) year.
Statistical analyses
A linear-mixed effect model used the ‘nlme’ package (Pinheiro et al. 2022). First, aboveground biomass, shoot P content, grain yield, grain P content, inorganic P, organic P, and residual-P were tested using cropping system (i.e. monoculture and intercropping), P-application rate, and crop combination as fixed effects, and block as a random effect. Second, the apparent soil P balance at each P-application rate in different crop combinations was separately analyzed using cropping system (i.e. sole maize, sole companion crop, and intercropping combination) as a fixed effect, and block as a random effect. Third, cropping system (i.e. sole maize, sole companion crop, and intercropping combination) and P-application rate, and their interaction effect on the proportion of four soil P pools, rhizosheath soil properties, and bulk Olsen-P of different crop combinations were tested using cropping system and P-application rate as fixed factors; block was treated as a random factor. Tukey’s post-hoc HSD test was conducted at the 5% probability level in linear-mixed effect models.
Principal component analyses (PCA) and PERMANOVA test were conducted using P-related functional traits (rhizosheath acid phosphatase activity, rhizosheath alkaline phosphatase activity, rhizosheath pH, rhizosheath MBP, leaf [Mn], and rhizosheath phoD gene abundance), soil P Pools, total organic P, total inorganic P, Olsen-P of different cropping systems (i.e. sole maize, sole companion crop, and intercropping combination), using the ‘ggbiplot’ package (Vincent 2011). A PERMANOVA test was conducted to calculate the P value between cropping systems using the ‘pairwiseAdonis’ package (Pedro 2017). Finally, we also examined the relationships between P-related functional traits and shoot P content by correlation analyses in each crop combination. All statistical analyses were performed with R version 4.1.3 (R Development Core Team 2022).
Results
Productivity, plant P content, and apparent soil P balance
Aboveground biomass and grain yield differed among cropping systems (Cs: P < 0.001), and P-application rates (P: P < 0.001; Fig. 1; Table S1). Intercropping significantly increased aboveground biomass and grain yield compared with the weighted means of the monoculture crops (Fig. 1). Faba bean/maize, soybean/maize, chickpea/maize, and oilseed rape/maize intercropping increased grain yield by 66.7%, 20.8%, 77.4%, and 68.8% without P application, by 33.2%, 26.4%, 38.8%, and 22.5% under P40, and by 36.2%, 16.5%, 43.6%, and 20.8% under P80, compared with the weighted means of corresponding monocultures, respectively (Fig. 1B). The chickpea/maize intercropping combination showed the greatest yield advantage under three P-application rates. There were no significant differences in aboveground biomass and grain yield among crop combinations (Fig. 1A,B). Aboveground biomass and grain yield tended to increase with increasing P-application rates, and the minimum aboveground biomass was observed at the P0 rate (Fig. 1E,F).
Effects of crop combination (Cc) and cropping system (i.e. monoculture and intercropping, Cs) on aboveground biomass A, grain yield B, shoot phosphorus (P) content C and grain P content D. Effects of P-application rate (factor P) and cropping system (Cs) on aboveground biomass E, grain yield F, shoot P content G and grain P content H in 2020. Aboveground biomass, grain yield, shoot P content and grain P content of the monocultures were calculated as the weighted means of corresponding monoculture crops based on their land proportions in intercropping. P0: 0 kg P ha−1; P40: 40 kg P ha−1; P80: 80 kg P ha−1. FM: faba bean/maize combination; SM: soybean/maize combination; CM: chickpea/maize combination; RM: oilseed rape/maize combination. Lowercase letters indicate differences among treatments if the interaction effect is significant. The same letter means there is no significant difference (Tukey HSD). *, P < 0.05; **, P < 0.01; ***, P < 0.001; ns, not significant
Shoot and grain P content differed among cropping systems, crop combinations, and P-application rates (Fig. 1; Table S1). Intercropping significantly increased grain P content and shoot P content compared with the weighted means of the monoculture crops in all crop combinations (Fig. 1). The oilseed rape/maize combination exhibited significantly greater shoot P content than chickpea/maize and faba bean/maize combinations; the soybean/maize combination exhibited significantly greater grain P content than faba bean/maize combinations (Fig. 1C,D). Phosphorus application significantly increased shoot P content and grain P content; the increase of shoot P content and grain P content in intercropping with P40 and P80 was greater than that in P0 (Fig. 1G,H).
Without P input for 12 years, all treatments showed soil P depletion. In contrast, P surplus was shown in all treatments with 80 kg P ha−1 application annually. Applying 40 kg P ha−1 every year led to a large P surplus for sole companion crops, but a small P surplus for sole maize (33 kg P ha−1). The apparent P balance of intercropping systems under 40 kg P ha−1 was in the range of -50 to + 6 kg P ha−1. Intercropping systems and sole maize accumulated less P soil in than sole companion crops (Table 1).
Effect of cropping system on soil total inorganic P, total organic P, and residual P among crop combinations
We calculated total inorganic P, total organic P, and residual-P to assess the utilization of soil P by crop combinations and cropping systems with different P-application rates. Although there was no significant difference between cropping systems in residual P, total inorganic P, and total organic P of the rhizosheath soil (Fig. S1), intercropping significantly decreased the bulk soil residual-P (P < 0.001). However, there was no significant difference in the concentrations of total inorganic P and total organic P in the bulk soil compared with the weighted means of the monoculture crops (Fig. 2; Table S2). The chickpea/maize combination had the greatest bulk soil total inorganic P concentration, and that of the faba bean/maize combination was the lowest (Fig. 2A). The oilseed rape/maize combination had the greatest bulk soil total organic P concentration, and that of the soybean/maize combination was the lowest. The soybean/maize combination exhibited the greatest bulk soil residual-P concentration among the four crop combinations, and oilseed rape/maize had the lowest soil residual-P concentration (Fig. 2B,C). The interaction effect between crop combination and cropping system was significant in bulk soil inorganic-P and residual-P concentration, in which the faba bean/maize intercropping system showed significantly lower inorganic-P and oilseed rape/maize intercropping system showed significantly lower residual-P concentrations than those of the weighted means of the monoculture crops (Fig. 2A,C). In the rhizosheath soil, we also found a significant interaction effect between crop combination and cropping system in faba bean/maize combination, in which faba bean/maize intercropping system also exhibited a lower inorganic-P concentration than sole faba bean/maize did (Fig. S1). The concentration of soil total inorganic P, total organic P, and residual-P in the bulk soil and rhizosheath soil increased with increasing P-application rates (Fig. 2D,E,F,S1).
Effects of crop combination (Cc) and cropping system (i.e. monoculture and intercropping, Cs) on total soil inorganic phosphorus (P) A, total soil organic P B, and soil residual-P C in the bulk soil. Effects of P-application rate (factor P) and cropping system (Cs) on total soil inorganic phosphorus D, total soil organic phosphorus E, and soil residual-P F in the bulk soil. Total soil inorganic P, total organic P, and residual-P of the monocultures were calculated as the weighted means of corresponding monoculture crops based on their land proportions in intercropping. P0: 0 kg P ha−1; P40: 40 kg P ha−1; P80: 80 kg P ha−1. FM: faba bean/maize combination; SM: soybean/maize combination; CM: chickpea/maize combination; RM: oilseed rape/maize combination. Uppercase letters refer to differences among P-application rates. Lowercase letters indicate differences among treatments if the interaction effect is significant. The same letter means there is no significant difference (Tukey HSD). *, P < 0.05; **, P < 0.01; ***, P < 0.001; ns, not significant
Impacts of P application and cropping system on soil P
In the bulk soil, the moderately-labile soil P pool (the sum of NaOH-Pi, NaOH-Po, and 1 M HCl-Pi) represented the largest proportion of total P in all treatments, accounting for 67% to 74% of total P in all treatments, followed by sparing-labile P, ranging from 11 to 17% (Fig. 3). The proportions of bulk soil P pools were affected by cropping system and P-application rates in different intercropping systems (Fig. 3; Table S3). Phosphorus-application rate significantly increased the labile soil P proportion under the four intercropping systems, but significantly decreased the sparingly-labile P proportion and non-labile P proportion in different intercropping systems (except the sparingly-labile P proportion in the oilseed rape/maize system). The results about the proportions of the four P pools in the rhizosheath soil were similar as those in the bulk soil (Fig. S2).
Proportions of different soil bulk phosphorus (P) pools in cropping systems (i.e. sole maize, sole companion crop, intercropping combination, Cs) with three P-application rates. Shown are chickpea/maize A, soybean/maize B, faba bean/maize C, and oilseed rape/maize D combinations. Labile P pool includes Resin-P, NaHCO3-Pi, and NaHCO3-Po; moderately-labile P pool includes NaOH-Pi, NaOH-Po, and 1 M HCl-Pi; sparingly-labile P pool includes conc. HCl-Pi and conc. HCl-Po; non-labile P pool is residual-P; P0: 0 kg P ha−1; P40: 40 kg P ha−1; P80: 80 kg P ha−1
In the faba bean/maize combination, the total bulk soil P concentration comprised 82.3% inorganic P, 8.8% Po and 8.9% residual P (Table S4). Among the three systems, sole faba bean exhibited a greater labile-P pool than the faba bean/maize intercropped system and sole maize system (P < 0.01). The faba bean/maize intercropped system and sole faba bean system had less Pi, moderately-labile P and non-labile P than the sole maize (Fig. 4). In the oilseed rape/maize combination, the total bulk soil P concentration was composed of 82.7% Pi, 9.2% Po and 8.1% residual-P (Table S5). Labile-P and sparingly-labile P concentrations among cropping systems were dependent on P-application rate (Fig. 5). Sole maize exhibited a greater non-labile P pool but less soil Po than sole oilseed rape and oilseed rape/maize intercropping systems did. The faba bean/maize intercropping system, oilseed rape/maize intercropping system, and corresponding sole companion crop systems exhibited more soil organic P but less non-labile P than the sole maize system did.
Effects of phosphorus (P)-application rate (factor P) and cropping system (i.e. sole maize, sole faba bean, and intercropping combination, Cs) on bulk soil P in faba bean/maize combination. P0: 0 kg P ha−1; P40: 40 kg P ha−1; P80: 80 kg P ha−1. Uppercase letters refer to differences among P-application rates. The same letter means there is no significant difference (Tukey HSD). *, P < 0.05; **, P < 0.01; ***, P < 0.001; ns, not significant
Effects of phosphorus (P)-application rate (factor P) and cropping system (i.e. sole maize, sole oilseed rape, and intercropping combination, Cs) on bulk soil P in oilseed rape/maize combination. P0: 0 kg P ha−1; P40: 40 kg P ha−1; P80: 80 kg P ha−1. Uppercase letters refer to differences among P-application rates. Lowercase letters indicate differences among treatments if the interaction effect is significant, the middle letters are omitted if there are more than two letters (e.g., the letters ‘bd’ are concisely expressed as ‘bcd’). The same letter means there is no significant difference (Tukey HSD). *, P < 0.05; **, P < 0.01; ***, P < 0.001; ns, not significant
In the chickpea/maize combination, the total soil bulk P concentration comprised 84.6% Pi, 7.2% Po, and 8.2% residual P (Table S6). The sole chickpea and/or chickpea/maize intercropping system showed a greater labile-P pool and Pi but less non-labile P and Po than sole maize did. There were no significant differences among cropping systems in the soil moderately-labile P and sparingly-labile P pools (Fig. 6). In the soybean/maize combination, the total bulk soil P fractions averaged: 84.1% in Pi forms, 6.5% in Po forms, and 9.4% in residual P (Table S7). There were no significant differences among cropping systems in labile-P, moderately-labile P pool and Pi. Sole soybean and soybean/maize intercropping systems exhibited less sparingly-labile soil P and Po than sole maize, in which the decrease of sparingly-labile P pool and Po under P40 and P80 was larger than those without P application (Fig. 7). The concentration of soil Po in the chickpea/maize intercropping system, soybean/maize intercropping system, and corresponding sole companion crop systems was lower than that of sole maize system. Our results also show that the chickpea/maize and soybean/maize systems had a lower percentage of total organic P, but a greater percentage of total inorganic P than the faba bean/maize and oilseed rape/maize systems (Tables S4-S7).
Effects of phosphorus (P)-application rate (factor P) and cropping system (i.e. sole maize, sole chickpea, and intercropping combination, Cs) on bulk soil P in chickpea/maize combination. P0: 0 kg P ha−1; P40: 40 kg P ha−1; P80: 80 kg P ha−1. Uppercase letters refer to differences among P-application rates. The same letter means there is no significant difference (Tukey HSD). *, P < 0.05; **, P < 0.01; ***, P < 0.001; ns, not significant
Effects of phosphorus (P)-application rate (factor P) and cropping system (i.e. sole maize, sole soybean, and intercropping combination, Cs) on bulk soil P in soybean/maize combination. P0: 0 kg P ha−1; P40: 40 kg P ha−1; P80: 80 kg P ha−1. Uppercase letters refer to differences among P-application rates. Lowercase letters indicate differences among treatments if the interaction effect is significant, the middle letters are omitted if there are more than two letters (e.g., the letters ‘ac’ are concisely expressed as ‘abc’). The same letter means there is no significant difference (Tukey HSD). *, P < 0.05; **, P < 0.01; ***, P < 0.001; ns, not significant
Correlations between soil P pools and root physiological traits
The crops showed significant differences in physiological traits in each crop combination. In the oilseed rape/maize combination, sole oilseed rape and oilseed rape/maize intercropping system exhibited greater acid phosphatase activity, greater leaf [Mn], lower pH, and lower phoD gene abundance in rhizosheath than sole maize (Table S4). In the faba bean/maize combination, sole faba bean showed greater rhizosheath microbial biomass P, leaf [Mn] and bulk Olsen P, but a lower phoD gene abundance than sole maize did (Table S4). In the soybean/maize combination, the soybean maize intercropping system showed greater microbial biomass P and leaf [Mn], but lower phoD gene abundance than sole maize did (Table S4). In the chickpea/maize combination, the chickpea/maize intercropping system exhibited lower phoD gene abundance than sole maize did (Table S4).
Principal component analysis (PCA) was used to explore how P-related functional traits and soil P pools were associated with intercropping under different crop combinations. In the chickpea/maize system, PCA axis 1 explained 32.8% of the variance, and PCA axis 2 explained 15.7%. The sole chickpea and chickpea/maize intercropping systems showed less total soil organic P but more inorganic P than sole maize did, while the total soil organic P concentration was negatively correlated with rhizosheath phosphatase activity; pH was negatively correlated with total inorganic P and Olsen P (Figs. 6 and 8A; Table S6). In the soybean/maize system, the two principal components explained 53.1% of the variance. The sole maize system was separated from the sole soybean and soybean/maize intercropping system. The sole soybean and soybean/maize intercropping system showed a lower total soil organic P concentration, which was negatively correlated with rhizosheath acid phosphatase activity, rhizosheath alkaline phosphatase activity, rhizosheath microbial biomass P and leaf [Mn] (Fig. 8B). The two PCA axes in the faba bean/maize and oilseed rape/maize systems accounted for 54.2% and 72.9%, respectively. Sole faba bean, sole oilseed rape, and corresponding intercropping systems showed greater soil total organic P concentrations, but lower residual-P concentrations, and the soil residual-P concentrations were negatively correlated with rhizosheath acid phosphatase activity and alkaline phosphatase activity (Figs.4, 5 and 8C,D). In addition, the intercropping systems and companion crops showed a greater labile P pool, but a lower non-labile P pool concentration than sole maize, except for soybean/maize combinations (Fig. 8E-H). All the companion crops and corresponding intercropping systems exhibited higher rhizosheath acid phosphatase activity, rhizosheath alkaline phosphatase activity, rhizosheath microbial biomass P and leaf [Mn] than sole maize which partly reflected the stronger rhizosheath P-mobilizing capacity (Fig. 8).
Principal component analysis (PCA) among plant phosphorus (P)-related traits and soil total bulk soil inorganic P, total bulk soil organic P, bulk soil residual-P and bulk Olsen P of different cropping systems in the four crop combinations in 2020 (A-D).Principal component analysis (PCA) among plant P-related traits and bulk soil P pools and bulk Olsen P of different cropping systems (i.e. sole maize, sole companion crop, and intercropping combination) in the four crop combinations in 2020 (E–H). PC1 represents the first axis, PC2 represents the second axis, and the percentage number represents the proportion of variation the axis could explain. rAC, rhizosheath acid phosphatase activity; rAK, rhizosheath alkaline phosphatase activity; rpH, rhizosheath pH; rMBP, rhizosheath microbial biomass P; leaf [Mn]: leaf manganese concentration; rphoD, rhizosheath phoD gene abundance; Pi, total soil inorganic P; Po, total soil organic P; Moderately-P, moderately-labile P;Sparingly-P, sparingly-labile P. * means the differences between cropping systems by PERMANOVA test. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ns, not significant
Discussion
Intercropping with effective P-application rates reduced the soil P surplus
Overall, our results show that intercropping systems were associated with significantly greater biomass, grain yield and P content than the weighted means of corresponding monocultures, irrespective of P-application rate (Fig. 1). Several experiments and meta-analyses have also shown positive effects of biodiversity on productivity and shoot P content (Mudare et al. 2022; Tang et al. 2021; Yu et al. 2020). After 12 years, oilseed rape/maize intercropping reduced the soil residual-P concentration compared with corresponding monocultures (Fig. 2), while achieving greater shoot P content, indicating intercropping was more efficient at accessing soil P compared with monocultures. Long-term P-fertilizer application has caused a P surplus and increased soil P accumulation (Table 1). Excessive P in soil is highly susceptible to eroding into waterways, resulting in eutrophication of rivers and lakes (Qiu 2010; Zhang et al. 2019). We found that intercropping can reduce the P surplus in soil compared with companion crop monocultures, especially when applying 40 kg P ha−1 fertilizer, as the P surplus was close to zero (Table 1). Our findings corroborate previous studies, which showed a negative effect of intercropping on P balance (Liao et al. 2021, 2020). Our evidence suggests that intercropping with appropriate P-fertilizer application can increase productivity, meet plant P requirements, and better maintain soil P balance than monocultures do.
Phosphorus-fertilizer application rates directly affected soil P. Specifically, P application increased soil inorganic P, labile P pool and moderately-labile P pool in all crop combinations, resulting in a greater labile P proportion, a lower sparingly-labile P proportion, and a lower non-labile P proportion than plots without P fertilizer (Figs. 3, 4, 5, 6 and 7). Similar responses were also found in a faba bean/maize intercropping system with two P-fertilizer rates on calcareous soil (Liao et al. 2021) and in an oilseed rape-rice cropping system with five P-fertilizer rates on acidic soil (Yan et al. 2022). The concentration of the labile soil P pool increased with P-application rates which indicates that P fertilizer might accumulate in the labile P fractions and be available to crops (Tian et al. 2020b). This suggests that supplying P fertilizer is crucial to maintain P bioavailability for long-term cultivation.
The correlations between P-acquisition strategies and soil P pools in different crop combinations
The results of rhizosheath soil P pools may reflect P-mobilization abilities of crop species, while the results exhibited great variation in the field (Fig. S1). We focused on P-mobilization abilities of selected crops and its impact on companion crop in our previous article (An et al. 2023). Here, we pay more attention to the changes in soil P pools which can show the impact of cropping combinations/systems on P pool dynamics over the experimental period. In the present experiment, chickpea/maize and soybean/maize intercropping systems decreased the total soil Po concentration compared with sole maize; faba bean/maize and oilseed rape/maize intercropping systems showed a greater concentration of total organic P, but a lower non-labile soil P concentration than sole maize (Figs. 4, 5, 6, 7, and 8). The difference in soil P pools was related to diverse P-acquisition strategies of crops (Dissanayaka et al. 2015; Li et al. 2004; Liao et al. 2020; Wen et al. 2020). In chickpea/maize and soybean/maize combinations, we found a negative correlation between total soil organic P concentration and rhizosheath acid phosphatase activity (Fig. 8A,B), consistent with previous reports, indicating that chickpea and soybean can hydrolyze organic P by releasing acid phosphatase, leading to belowground facilitation in organic P utilization in intercropping (Belinque et al. 2015; Kong et al. 2014, 2018; Li et al. 2004). In addition, leaf [Mn] (a proxy for carboxylate release) showed a negative correlation with total soil organic P concentration in the soybean/maize combination (Fig. 8). Rhizosheath pH was negative correlated with the pools of Olsen P, labile P and moderately-labile P in chickpea/maize and faba bean/maize combinations (Fig. 8E,G). The exudate release in the rhizosphere caused acidification in the soil, which is critical to mobilize sorbed soil P into available P (Hinsinger et al. 2015; Lambers et al. 2015).
In faba bean/maize and oilseed rape/maize combinations, we did not find a negative correlation between rhizosheath phosphatase activity and soil total organic P concentration, in which rhizosheath phosphatase activity was negatively correlated with the residual soil P concentration (Fig. 8). In the present experiment, faba bean and oilseed rape exhibited greater phosphatase activity than other species did (An et al. 2023); these probably mobilized a non-labile P pool into available P pools, resulting in a greater labile P pool, but a lower non-labile P concentration in intercropping systems than in sole maize (Figs. 4, 5 and 8). Leaf [Mn] was negatively correlated with residual-P concentration in oilseed rape/maize combination (Fig. 8). The combination of carboxylates and enzymes may partly explain the depletion of P in soil (Clarholm et al. 2015; Giles et al. 2017). Therefore, combining species with desirable P-acquisition strategies to fully mine soil P pools in the diverse intercropping systems is crucial for sustainable P management.
The correlations between soil microorganisms and soil P in different crop combinations
Soil microorganisms are integral to soil P cycling in agroecosystems (Dai et al. 2020; Wei et al. 2019). The P held within soil microorganisms accounts for approximately 2% to 10% of the total P concentration, which generally exceeds that in plant biomass (Richardson and Simpson 2011). Microbial biomass P is potentially available to plant species and modulates soil P availability (Liebisch et al. 2014). The immobilized P within the biomass is maintained in labile forms and is protected from reactions with soil (Olander and Vitousek 2004). In this study, sole companion crops and intercropping systems had greater rhizosheath microbial biomass P than sole maize which indicates a potentially stronger competition of soil microorganisms to hold soil P (Fig. 8; Table S8). We also found a positive correlation between rhizosheath microbial biomass P and soil Olsen-P, except in the soybean/maize combination (Fig. 8). This partly supports that microbial biomass P can reflect soil P fertility (Peng et al. 2021). The changes in microbial biomass P are related to P concentration in the soil which depends on P-fertilizer application and agronomic management (Hallama et al. 2021; Liao et al. 2022; Peng et al. 2021).
The phoD gene, encoding an alkaline phosphatase that mediates organic P hydrolysis, is negatively correlated with soil organic P concentration (Fraser et al. 2015). Consistent with these findings, we found negative correlations between soil organic P and rhizosheath phoD gene abundance in oilseed rape/maize combinations (Fig. 8), indicating the contribution of microorganisms in P utilization. However, phoD gene abundance did not show a positive correlation with alkaline phosphatase activity in our study (Fig. 8). Similar results were also found in a soybean pot trial with three P-input rates (Tian et al. 2021).
Sole maize showed significantly higher rhizosheath phoD gene abundance than companion species and intercropping systems (Fig. 8; Table S8) which might be related to the microorganisms associated with arbuscular mycorrhizal fungal hyphae (Lang et al. 2022; Liu et al. 2018). Microorganisms associated with arbuscular mycorrhizal fungal hyphae can hydrolyze organic P and promote organic P turnover (Wipf et al. 2019). Other microbial functional genes of P cycling and microbial compositions need to be tested in future research. Thus, the results of microorganisms may be a consequence of intercropping, species characteristics, and P-fertilizer rate, which is related to P uptake and soil nutrient dynamics (Dai et al. 2020; Roohi et al. 2020; Yang et al. 2022).
The species differed in their P-acquisition strategies as well as associations with soil microorganisms, which are integral to the soil P cycle and mediate P availability, which is a crucial mechanism underpinning belowground facilitation in intercropping systems (Yu et al. 2021a). Intercropping significantly increased productivity and plant P content, compared with monocultures. Among the intercropping systems, chickpea/maize intercropping showed the greatest grain yield advantage, followed by faba bean/maize (Fig. 1). The chickpea/maize intercropping system had less total Po and non-labile P but more labile P than sole maize which correlated with leaf [Mn] and rhizosheath phosphatase activity (Figs. 6 and 8A,E); faba bean/maize intercropping system had less non-labile P but more labile P than sole maize, and the depletion of non-labile P was correlated with pH and rhizosheath phosphatase activity (Figs. 4 and 8C,G). The greater release of carboxylates and phosphatase in the rhizosheath would mobilize sorbed P and organic P into available P pools which promotes P uptake (Clarholm et al. 2015; Li et al. 2014; Giles et al. 2017). Thus, soil P pools and bioavailability, pH, and rhizosheath phosphatase activity played an important role in enhancing plant P uptake, which further contributes to crop productivity of intercropping (Yang et al. 2022).
Species in intercropping systems showed greater P-mobilizing capacities while changing soil P pools and enhancing shoot P content and aboveground biomass in the four crop combinations. Thus, it is understandable that plant physiological traits were correlated with different soil P fractions. Previous studies showed that intercropping enhances nutrient-use efficiency, increases nutrient uptake, and enhances crop productivity, while not all crop combinations have an intercropping advantage (Li et al. 2021; Tang et al. 2021). Efficient P-mobilizing species intercropped with inefficient species enhanced the availability of soil P by mobilizing sorbed soil P which increases the P acquisition of inefficient neighbors (Li et al. 2014). Crop species showing greater morphological plasticity in intercropping may acclimate to the heterogeneous environment, leading to P-uptake advantages via interspecific facilitation (Schneider and Lynch 2020; Yu et al. 2020). Furthermore, when P-efficient genotypes are combined with diverse agronomic strategies (i.e. intercropping) with complementary P-acquisition strategies under effective P-fertilizer management, the P-use efficiency may increase (Cong et al. 2020). Thus, establishing desirable species combinations is vital to enhance productivity and shoot P uptake, and maintain the soil P balance under high chemical fertilizer inputs (Cheriere et al. 2020).
Long-term P-fertilizer application results in a large amount of legacy P accumulating in soil or lost to the environment. Soil legacy P represents a secondary P resource, which can substitute for P fertilizer and become a source for crop use (Rowe et al. 2016). Species exhibit different P-acquisition strategies to obtain enough available P for their growth (Li et al. 2007; Zhang et al. 2016) which can help mine legacy P in different soils. Overall, optimizing a rational intercropping system involving complementary P-acquisition strategies is crucial for sustainable P management. The P-fractionation procedures modified by Hedley have limitations, and further analyses should be carried out in conjunction with the methods such as Energy Dispersive X-ray Spectra (EDS) analysis (Barrow et al. 2021).
Conclusion
Our study shows that intercropping significantly increased aboveground biomass, grain yield and plant P content, significantly mitigating soil P balance compared with companion crop monocultures. Under 40 kg ha−1, intercropping can exhibit a relatively high P content and prevent a P surplus in soil. Furthermore, we explored the correlations between P-acquisition strategies and soil P pools in different crop combinations under field conditions. Chickpea/maize and soybean/maize intercropping systems significantly decreased the soil organic P pool compared with sole maize under P-application conditions. Faba bean/maize and oilseed rape/maize intercropping systems significantly decreased the non-labile soil P pool but increased that organic P pool more than the sole maize system did. Furthermore, rhizosheath acid phosphatases and carboxylates may contribute to depleting sparingly-available soil P (organic P or non-labile P pools) in different crop combinations; rhizosheath microbial biomass P and phoD genes may also be related to the increase of soil P availability through mobilizing organic P. These results show that P-acquisition traits of species in intercropping systems change soil P pools to enhance the system’s P uptake while reducing P-fertilizer requirements. Our findings highlight the importance of P-acquisition strategies in designing intercropping systems and in decreasing P accumulation in soil for sustainable P management.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ae N, Arihara J, Okada K, Yoshihara T, Johansen C (1990) Phosphorus uptake by pigeon pea and its role in cropping systems of the Indian subcontinent. Science 248:477–480. https://doi.org/10.1126/science.248.4954.477
An R, Yu R-P, Xing Y, Zhang J-D, Bao X-G, Lambers H, Li L (2023) Enhanced phosphorus-fertilizer-use efficiency and sustainable phosphorus management with intercropping. Agron Sustain Dev 43:57. https://doi.org/10.1007/s13593-023-00916-6
Bao SD (2005) Analysis on Soil and Agricultural Chemistry. China Agri Press
Barrow NJ, Sen A, Roy N, Debnath A (2021) The soil phosphate fractionation fallacy. Plant Soil 459:1–11. https://doi.org/10.1007/s11104-020-04476-6
Belinque H, Pucheu N, Kerber N, Rubio G (2015) Utilization of organic phosphorus sources by oilseed rape, sunflower, and soybean. J Plant Nutr Soil Sci 178:339–344. https://doi.org/10.1002/jpln.201400301
Brooker RW, Bennett AE, Cong WF, Daniell TJ, George TS, Hallett PD, Hawes C, Iannetta PPM, Jones HG, Karley AJ, Li L, McKenzie BM, Pakeman RJ, Paterson E, Schoeb C, Shen J, Squire G, Watson CA, Zhang C, Zhang F, Zhang J, White PJ (2015) Improving intercropping: a synthesis of research in agronomy, plant physiology and ecology. New Phytol 206:107–117. https://doi.org/10.1111/nph.13132
Brookes PC, Powlson DS, Jenkinson DS (1982) Measurement of microbial biomass phosphorus in soil. Soil Biol Biochem 14:319–329. https://doi.org/10.1016/0038-0717(82)90001-3
Calderon-Vazquez C, Alatorre-Cobos F, Simpson-Williamson J, Herrera-Estrella L (2009) Maize under phosphate limitation. In: Bennetzen JL, Hake SC (eds) Handbook of Maize: Its Biology. Springer, New York, NY https://doi.org/10.1007/978-0-387-79418-1
Cheriere T, Lorin M, Corre-Hellou G (2020) Species choice and spatial arrangement in soybean-based intercropping: Levers that drive yield and weed control. Field Crops Res 256:107923. https://doi.org/10.1016/j.fcr.2020.107923
Clarholm M, Skyllberg U, Rosling A (2015) Organic acid induced release of nutrients from metal-stabilized soil organic matter-the unbutton model. Soil Biol Biochem 84:168–176. https://doi.org/10.1016/j.soilbio.2015.02.019
Cong WF, Suriyagoda LDB, Lambers H (2020) Tightening the phosphorus cycle through phosphorus-efficient crop genotypes. Trends Plant Sci 25:967–975. https://doi.org/10.1016/j.tplants.2020.04.013
Cordell D, White S (2011) Peak phosphorus: clarifying the key issues of a vigorous debate about long-term phosphorus security. Sustainability 3:2027–2049. https://doi.org/10.3390/su3102027
Crews TE, Brookes PC (2014) Changes in soil phosphorus forms through time in perennial versus annual agroecosystems. Agric Ecosyst Environ 184:168–181. https://doi.org/10.1016/j.agee.2013.11.022
Dai Z, Liu G, Chen H, Chen C, Wang J, Ai S, Wei D, Li D, Ma B, Tang C, Brookes PC, Xu J (2020) Long-term nutrient inputs shift soil microbial functional profiles of phosphorus cycling in diverse agroecosystems. ISME J 14:757–770. https://doi.org/10.1038/s41396-019-0567-9
Dissanayaka DMSB, Maruyama H, Masuda G, Wasaki J (2015) Interspecific facilitation of P acquisition in intercropping of maize with white lupin in two contrasting soils as influenced by different rates and forms of P supply. Plant Soil 390:223–236. https://doi.org/10.1007/s11104-015-2392-x
Elser JJ, Bracken MES, Cleland EE, Gruner DS, Harpole WS, Hillebrand H, Ngai JT, Seabloom EW, Shurin JB, Smith JE (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1135–1142. https://doi.org/10.1111/j.1461-0248.2007.01113.x
Fixen PE, Johnston AM (2012) World fertilizer nutrient reserves: a view to the future. J Sci Food Agric 92:1001–1005. https://doi.org/10.1002/jsfa.4532
Fraser TD, Lynch DH, Bent E, Entz MH, Dunfield KE (2015) Soil bacterial phoD gene abundance and expression in response to applied phosphorus and long-term management. Soil Biol Biochem 88:137–147. https://doi.org/10.1016/j.soilbio.2015.04.014
Giles CD, George TS, Brown LK, Mezeli MM, Richardson AE, Shand CA, Wendler R, Darch T, Menezes-Blackburn D, Cooper P, Stutter MI, Lumsdon DG, Blackwell MSA, Wearing C, Zhang H, Haygarth PM (2017) Does the combination of citrate and phytase exudation in Nicotiana tabacum promote the acquisition of endogenous soil organic phosphorus? Plant Soil 412:43–59. https://doi.org/10.1007/s11104-016-2884-3
Gomez PF, Ingram LO (1995) Cloning, sequencing and characterization of the alkaline phosphatase gene (phoD) from Zymomonas mobilis. FEMS Microbiol Lett 125:237–245
Hallama M, Pekrun C, Pilz S, Jarosch KA, Frac M, Uksa M, Marhan S, Kandeler E (2021) Interactions between cover crops and soil microorganisms increase phosphorus availability in conservation agriculture. Plant Soil 463:307–328. https://doi.org/10.1007/s11104-021-04897-x
Hedley MJ, Stewart JWB, Chauhan BS (1982) Changes in inorganic and organic soil-phosphorus fractions induced by cultivation practices and by laboratory incubations. Soil Sci Soc Am J 46:970–976. https://doi.org/10.2136/sssaj1982.03615995004600050017x
Hinsinger P (2001) Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review. Plant Soil 237:173–195. https://doi.org/10.1023/a:1013351617532
Hinsinger P, Herrmann L, Lesueur D, Robin A, Trap J, Waithaisong K, Plassard C (2015). Impact of roots, microorganisms and microfauna on the fate of soil phosphorus in the rhizosphere. In: Plaxton WC, Lambers H (Eds.), Phosphorus Metabolism in Plants 377–407 https://doi.org/10.1002/9781118958841.ch13
Hua KK, Zhang WJ, Guo ZB, Wang DZ, Oenema O (2016) Evaluating crop response and environmental impact of the accumulation of phosphorus due to long-term manuring of vertisol soil in northern China. Agric Ecosyst Environ 219:101–110. https://doi.org/10.1016/j.agee.2015.12.008
Isaac ME, Borden KA (2019) Nutrient acquisition strategies in agroforestry systems. Plant Soil 444:1–19. https://doi.org/10.1007/s11104-019-04232-5
Johnston AE, Poulton PR, Fixen PE, Curtin D (2014) Phosphorus: its efficient use in agriculture. Adv Agron 123:177–228. https://doi.org/10.1016/B978-0-12-420225-2.00005-4
Kong YB, Li XH, Ma J, Li WL, Yan GJ, Zhang CY (2014) GmPAP4, a novel purple acid phosphatase gene isolated from soybean (Glycine max), enhanced extracellular phytate utilization in Arabidopsis thaliana. Plant Cell Rep 33:655–667. https://doi.org/10.1007/s00299-014-1588-5
Kong YB, Li XH, Wang B, Li WL, Du H, Zhang CY (2018) The soybean purple acid phosphatase GmPAP14 predominantly enhances external phytate utilization in plants. Front Plant Sci 9:292. https://doi.org/10.3389/fpls.2018.00292
Lambers H (2022) Phosphorus acquisition and utilization in plants. Annu Rev Plant Biol 73:17–42. https://doi.org/10.1146/annurev-arplant-102720-125738
Lambers H, Hayes PE, Laliberté E, Oliveira RS, Turner BL (2015) Leaf manganese accumulation and phosphorus-acquisition efficiency. Trends Plant Sci 20:83–90. https://doi.org/10.1016/j.tplants.2014.10.007
Lambers H, Wright IJ, Pereira CG, Bellingham PJ, Bentley LP, Boonman A, Cernusak LA, Foulds W, Gleason SM, Gray EF, Hayesf PE, Kooyman RM, Malhi Y, Richardson SJ, Shane MW, Staudinger C, Stock WD, Swarts ND, Turner BL, Turner J, Veneklaas EJ, Wasaki J, Westoby M, Xu YG (2021) Leaf manganese concentrations as a tool to assess belowground plant functioning in phosphorus-impoverished environments. Plant Soil 461:43–61. https://doi.org/10.1007/s11104-020-04690-2
Lang M, Zhang CY, Su WH, Chen XX, Zou CQ, Chen XP (2022) Long-term P fertilization significantly altered the diversity, composition and mycorrhizal traits of arbuscular mycorrhizal fungal communities in a wheat-maize rotation. Appl Soil Ecol 170:104261. https://doi.org/10.1016/j.apsoil.2021.104261
Li H, Shen J, Zhang F, Clairotte M, Drevon JJ, Le Cadre E, Hinsinger P (2008) Dynamics of phosphorus fractions in the rhizosphere of common bean (Phaseolus vulgaris L.) and durum wheat (Triticum turgidum durum L.) grown in monocropping and intercropping systems. Plant Soil 312:139–150. https://doi.org/10.1007/s11104-007-9512-1
Li L, Li SM, Sun JH, Zhou LL, Bao XG, Zhang HG, Zhang FS (2007) Diversity enhances agricultural productivity via rhizosphere phosphorus facilitation on phosphorus-deficient soils. Proc Natl Acad Sci USA 104:11192–11196. https://doi.org/10.1073/pnas.0704591104
Li L, Tilman D, Lambers H, Zhang F-S (2014) Plant diversity and overyielding: insights from belowground facilitation of intercropping in agriculture. New Phytol 203:63–69. https://doi.org/10.1111/nph.12778
Li SM, Li L, Zhang FS, Tang C (2004) Acid phosphatase role in chickpea/maize intercropping. Ann Bot 94:297–303. https://doi.org/10.1093/aob/mch140
Li XF, Wang ZG, Bao XG, Sun JH, Yang SC, Wang P, Wang CB, Wu JP, Liu XR, Tian XL, Wang Y, Li JP, Wang Y, Xia HY, Mei PP, Wang XF, Zhao JH, Yu RP, Zhang WP, Che ZX, Gui LG, Callaway RM, Tilman D, Li L (2021) Long-term increased grain yield and soil fertility from intercropping. Nat Sustain 4:943–950. https://doi.org/10.1038/s41893-021-00767-7
Liao D, Zhang CC, Lambers H, Zhang FS (2021) Changes in soil phosphorus fractions in response to long-term phosphate fertilization under sole cropping and intercropping of maize and faba bean on a calcareous soil. Plant Soil 463:589–600. https://doi.org/10.1007/s11104-021-04915-y
Liao D, Zhang CC, Lambers H, Zhang FS (2022) Adding intercropped maize and faba bean root residues increases phosphorus bioavailability in a calcareous soil due to organic phosphorus mineralization. Plant Soil 476:201–218. https://doi.org/10.1007/s11104-022-05309-4
Liao D, Zhang CC, Li HG, Lambers H, Zhang FS (2020) Changes in soil phosphorus fractions following sole cropped and intercropped maize and faba bean grown on calcareous soil. Plant Soil 448:587–601. https://doi.org/10.1007/s11104-020-04460-0
Liebisch F, Keller F, Huguenin-Elie O, Frossard E, Oberson A, Bünemann EK (2014) Seasonal dynamics and turnover of microbial phosphorusin a permanent grassland. Biol Fertil Soils 50:465–475. https://doi.org/10.1007/s00374-013-0868-5
Liu F, Xu YJ, Han GM, Wang W, Li XY, Cheng BJ (2018) Identification and functional characterization of a maize phosphate transporter induced by mycorrhiza formation. Plant Cell Physiol 59:1683–1694. https://doi.org/10.1093/pcp/pcy094
Lyu Y, Tang H, Li H, Zhang F, Rengel Z, Whalley WR, Shen J (2016) Major crop species show differential balance between root morphological and physiological responses to variable phosphorus supply. Front Plant Sci 7:1939. https://doi.org/10.3389/fpls.2016.01939
Mudare S, Kanomanyanga J, Jiao XQ, Mabasa S, Lamichhane JR, Jing JY, Cong WF (2022) Yield and fertilizer benefits of maize/grain legume intercropping in China and Africa: A meta-analysis. Agron Sustain Dev 42:81. https://doi.org/10.1007/s13593-022-00816-1
Murphy J, Riley JP (1962) A modified single solution method for the determination of phosphate in natural waters. Anal Chim Acta 27:31–36. https://doi.org/10.1016/S0003-2670(00)88444-5
Nannipieri P, Giagnoni L, Landi L, Renella G (2011) Role of phosphatase enzymes in soil. In: EK Bünemann, A Oberson, E Frossard (Eds) Phosphorus in action. Springer-Verlag, Berlin, Heidelberg 215–243 https://doi.org/10.1007/978-3-642-15271-9_9
Olander LP, Vitousek PM (2004) Biological and geochemical sinks for phosphorus in soil from a wet tropical forest. Ecosystems 7:404–419. https://doi.org/10.1007/s10021-004-0264-y
Pedro MA (2017) pairwiseAdonis: Pairwise Multilevel Comparison using Adonis. R package version 0.4
Peng Y, Duan Y, Huo W, Xu M, Yang X, Wang X, Wang B, Blackwell MSA, Feng G (2021) Soil microbial biomass phosphorus can serve as an index to reflect soil phosphorus fertility. Biol Fertil Soils 57:657–669. https://doi.org/10.1007/s00374-021-01559-z
Pinheiro JC, Bates DM and R Core Team (2022) nlme: Linear and nonlinear mixed effects models. R package version 3.1–157. https://CRAN.R-project.org/package=nlme
Qiu J (2010) Phosphate fertilizer warning for China. Nat. News, 29 September https://doi.org/10.1038/news.2010.498
Qu JH, Li LJ, Zhao PY, Han DY, Zhao XY, Zhang YL, Han L, Wang Y (2022) Impact of phosphorous fertilization on rape and common vetch intercropped fodder and soil phosphorus dynamics in North China. Agriculture-Basel 12 https://doi.org/10.3390/agriculture12111949
R Development Core Team (2022) R: A language and environment for statistical computing, v.4.1.3. Vienna, Austria: R Foundation for Statistical Computing. [WWW document] URL https:// www.R-project.org/
Richardson AE, Simpson RJ (2011) Soil microorganisms mediating phosphorus availability. Plant Physiol 156:989–996. https://doi.org/10.1104/pp.111.175448
Roohi M, Arif MS, Yasmeen T, Riaz M, Rizwan M, Shahzad SM, Ali S, Bragazza L (2020) Effects of cropping system and fertilization regime on soil phosphorous are mediated by rhizosphere-microbial processes in a semi-arid agroecosystem. J Environ Manage 271:111033. https://doi.org/10.1016/j.jenvman.2020.111033
Rowe H, Withers PJA, Baas P, Chan NI, Doody D, Holiman J, Jacobs B, Li HG, MacDonald GK, McDowell R, Sharpley AN, Shen JB, Taheri W, Wallenstein M, Weintraub MN (2016) Integrating legacy soil phosphorus into sustainable nutrient management strategies for future food, bioenergy and water security. Nutr Cycl Agroecosyst 104:393–412. https://doi.org/10.1007/s10705-015-9726-1
Sakurai M, Wasaki J, Tomizawa Y, Shinano T, Osaki M (2008) Analysis of bacterial communities on alkaline phosphatase genes in soil supplied with organic matter. Soil Sci Plant Nutr 54:62–71. https://doi.org/10.1111/j.1747-0765.2007.00210.x
Schneider HM, Lynch JP (2020) Should root plasticity be a crop breeding target? Front Plant Sci 11:546. https://doi.org/10.3389/fpls.2020.00546
Tabatabai M, Bremner J (1969) Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol Biochem 1:301–307. https://doi.org/10.1016/0038-0717(69)90012-1
Tang XY, Zhang CC, Yu Y, Shen JB, van der Werf W, Zhang FB (2021) Intercropping legumes and cereals increases phosphorus use efficiency; a meta-analysis. Plant Soil 460:89–104. https://doi.org/10.1007/s11104-020-04768-x
Tian J, Kuang X, Tang M, Chen X, Huang F, Cai Y, Cai K (2021) Biochar application under low phosphorus input promotes soil organic phosphorus mineralization by shifting bacterial phoD gene community composition. Sci Total Environ 779:146556. https://doi.org/10.1016/j.scitotenv.2021.146556
Tian J, Tang M, Xu X, Luo S, Condron LM, Lambers H, Cai K, Wang J (2020a) Soybean (Glycine max(L.) Merrill) intercropping with reduced nitrogen input influences rhizosphere phosphorus dynamics and phosphorus acquisition of sugarcane (Saccharum officinarum). Biol Fertil Soils 56:1063–1075. https://doi.org/10.1007/s00374-020-01484-7
Tian JH, Lu X, Chen QQ, Kuang XZ, Liang CY, Deng LS, Lin DJ, Cai KZ, Tian J (2020b) Phosphorus fertilization affects soybean rhizosphere phosphorus dynamics and the bacterial community in karst soils. Plant Soil. https://doi.org/10.1007/s11104-020-04662-6
Tiessen H, Moir JO (1993) Characterization of available P bysequential extraction. Soil sampling methods of analysis 824. Lewis Publisher, Boca Ration 75–86 https://doi.org/10.1201/9781420005271.ch25
Vincent QV (2011) ggbiplot: A ggplot2 based biplot. R package version 0.55. http://github.com/vqv/ ggbiplot
Vitousek PM, Porder S, Houlton BZ, Chadwick OA (2010) Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen-phosphorus interactions. Ecol Appl 20:5–15. https://doi.org/10.1890/08-0127.1
Vorosmarty CJ, McIntyre PB, Gessner MO, Dudgeon D, Prusevich A, Green P, Glidden S, Bunn SE, Sullivan CA, Liermann CR, Davies PM (2010) Global threats to human water security and river biodiversity. Nature 467:555–561. https://doi.org/10.1038/nature09440
Wang G, Bei S, Li J, Bao X, Zhang J, Schultz PA, Li H, Li L, Zhang F, Bever JD, Zhang J (2021) Soil microbial legacy drives crop diversity advantage: linking ecological plant-soil feedback with agricultural intercropping. J Appl Ecol 58:496–506. https://doi.org/10.1111/1365-2664.13802
Wei X, Hu Y, Razavi BS, Zhou J, Shen J, Nannipieri P, Wu J, Ge T (2019) Rare taxa of alkaline phosphomonoesterase-harboring microorganisms mediate soil phosphorus mineralization. Soil Biol Biochem 131:62–70. https://doi.org/10.1016/j.soilbio.2018.12.025
Wen Z, Pang J, Tueux G, Liu Y, Shen J, Ryan MH, Lambers H, Siddique KHM (2020) Contrasting patterns in biomass allocation, root morphology and mycorrhizal symbiosis for phosphorus acquisition among 20 chickpea genotypes with different amounts of rhizosheath carboxylates. Funct Ecol 34:1311–1324. https://doi.org/10.1111/1365-2435.13562
Wipf D, Krajinski F, van Tuinen D, Recorbet G, Courty PE (2019) Trading on the arbuscular mycorrhiza market: from arbuscules to common mycorrhizal networks. New Phytol 223:1127–1142. https://doi.org/10.1111/nph.15775
Xing Y, Yu RP, An R, Yang N, Wu JP, Ma HY, Zhang JD, Bao XG, Lambers H, Li L (2023) Two pathways drive enhanced nitrogen acquisition via a complementarity effect in long-term intercropping. Field Crops Res 293:108854. https://doi.org/10.1016/j.fcr.2023.108854
Yan JY, Ren T, Wang KK, Li HZ, Li XK, Cong RH, Lu JW (2022) Improved crop yield and phosphorus uptake through the optimization of phosphorus fertilizer rates in an oilseed rape-rice cropping system. Field Crops Res 286:108614. https://doi.org/10.1016/j.fcr.2022.108614
Yang ZY, Zhang YP, Wang YZ, Zhang HF, Zhu QR, Yan BJ, Fei JC, Rong XM, Peng JW, Luo GW (2022) Intercropping regulation of soil phosphorus composition and microbially-driven dynamics facilitates maize phosphorus uptake and productivity improvement. Field Crops Res 287:108666. https://doi.org/10.1016/j.fcr.2022.108666
Yu H, Wang F, Shao M, Huang L, Xie Y, Xu Y, Kong L (2021a) Effects of rotations with legume on soil functional microbial communities involved in phosphorus transformation. Front Microbiol 12:661100. https://doi.org/10.3389/fmicb.2021.661100
Yu R-P, Lambers H, Callaway RM, Wright AJ, Li L (2021b) Belowground facilitation and trait matching: two or three to tango? Trends Plant Sci 26:1227–1235. https://doi.org/10.1016/j.tplants.2021.07.014
Yu R-P, Li XX, Xiao ZH, Lambers H, Li L (2020) Phosphorus facilitation and covariation of root traits in steppe species. New Phytol 226:1285–1298. https://doi.org/10.1111/nph.16499
Yu R-P, Su Y, Lambers H, van Ruijven J, An R, Yang H, Yin XT, Xing Y, Zhang WP, Li L (2023) A novel proxy to examine interspecific phosphorus facilitation between plant species. New Phytol 239:1637–1650. https://doi.org/10.1111/nph.19082
Yu WJ, Li HG, Nkebiwe PM, Li GH, Mueller T, Zhang JL, Shen JB (2021c) Estimation of the P fertilizer demand of China using the LePA model. Front Environ Sci 9:759987. https://doi.org/10.3389/fenvs.2021.759984
Zhang DS, Zhang CC, Tang XY, Li HG, Zhang FS, Rengel Z, Whalley WR, Davies WJ, Shen JB (2016) Increased soil phosphorus availability induced by faba bean root exudation stimulates root growth and phosphorus uptake in neighbouring maize. New Phytol 209:823–831. https://doi.org/10.1111/nph.13613
Zhang WN, Tang XM, Feng XH, Wang EL, Li HG, Shen JB, Zhang FS (2019) Management strategies to optimize soil phosphorus utilization and alleviate environmental risk in China. J Environ Qual 48:1167–1175. https://doi.org/10.2134/jeq2019.02.0054
Funding
This study was financially supported by the National Natural Science Foundation of China (Project Nos: 32130067, 32101297). Rui-Peng Yu was also financially supported by the fellowship of China Postdoctoral Science Foundation (Project No: 2022T150703). We thank all members for helping maintain the long-term field experiment.
Author information
Authors and Affiliations
Contributions
Long Li, Ran An, Rui-Peng Yu, Yi Xing, Xing-Guo Bao, and Jiu-Dong Zhang designed and managed the experiment. Ran An, Rui-Peng Yu, and Yi Xing collected data and performed analyses. Ran An drafted the paper, and Rui-Peng Yu, Hans Lambers and Long Li contributed substantially to the revisions.
Corresponding author
Ethics declarations
Competing interest
The author declare to have no competing interests.
Additional information
Responsible Editor: Andrew Fletcher.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
An, R., Yu, RP., Xing, Y. et al. Intercropping efficiently utilizes phosphorus resource in soil via different strategies mediated by crop traits and species combination. Plant Soil 497, 705–725 (2024). https://doi.org/10.1007/s11104-023-06426-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-06426-4