Abstract
Context
Natural regenerating forests are rapidly expanding in the tropics. Forest transitions have the potential to restore biodiversity. Spatial targeting of land use policies could improve the biodiversity benefits of reforesting landscapes.
Objective
We explored the relative importance of landscape attributes in influencing the potential of tree cover increase to restore native woody plant biodiversity at the landscape scale.
Methods
We developed land use scenarios that differed in spatial patterns of reforestation, using the Pangor watershed in the Ecuadorian Andes as a case study. We distinguished between reforestation through natural regeneration of woody vegetation in abandoned fallows and planted forests through managed plantations of exotic species on previously cultivated land. We simulated the restoration of woody plant biodiversity for each scenario using LANDIS-II, a process-based model of forest dynamics. A pair-case comparison of simulated woody plant biodiversity for each scenario was conducted against a random scenario.
Results
Species richness in natural regenerating fallows was considerably higher when occurring in: (i) close proximity to remnant forests; (ii) areas with a high percentage of surrounding forest cover; and (iii) compositional heterogeneous landscapes. Reforestation at intermediate altitudes also positively affected restoration of woody plant species. Planted exotic pine forests negatively affected species restoration.
Conclusions
Our research contributes to a better understanding of the recolonization processes of regenerating forests. We provide guidelines for reforestation policies that aim to conserve and restore woody plant biodiversity by accounting for landscape attributes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Although tropical deforestation continues at a rapid rate, there is also evidence of a regrowth of tropical and subtropical forests in some regions. Regrowth occurs in various ways. For example, through regenerating forests that naturally establish after human or natural disturbance of the original forest vegetation (Chokkalingam and de Jong 2001; Chazdon 2014) or planted forests composed of trees established through planting of native or exotic species (FAO 2015). To date, we still lack an accurate estimate of the extent of regenerating or planted tropical forests (but see Asner et al. 2009; Meyfroidt and Lambin 2011). Numerous studies have emphasized the critical importance of regenerating forests in tropical landscapes (Dunn 2004; Chazdon 2008; DeClerck et al. 2010), but have also emphasized the differences between forest transition pathways to restore biodiversity and ecosystem functions (Meyfroidt and Lambin 2011; Chazdon 2014).
In Ecuador, land reforms and governmental programs (e.g., the payment for carbon sequestration program PROFAFOR) have stimulated forest plantations from the 1960s onwards, leading to the conversion of large areas of natural grasslands to exotic pine plantations in the highlands (Farley 2007, 2010). Economic and demographic forces, such as land abandonment with economic development, have also favored natural regeneration of tropical forests (Rudel et al. 2005; Lambin and Meyfroidt 2010) and have been observed in the Ecuadorian Andes (Balthazar et al. 2015). Ecuadorian conservation policies (e.g., the payment for ecosystem services program SocioBosque) have started to recognize the role of forest regrowth at the landscape level and will incorporate reforestation programs in the future (de Koning et al. 2011).
Natural regenerating and planted forests affect the spatial configuration and composition of landscapes and therefore habitat distribution. Landscape structure has been suggested as an important factor that determines the effectiveness of local biodiversity conservation management in human-modified landscapes (Tscharntke et al. 2012). Even though landscape fragmentation has been recognized as a key factor in biodiversity loss (Fischer and Lindenmayer 2007), the effect of landscape patterns on restoration of biodiversity and ecosystem functions in reforesting landscapes is still not well understood (Hall et al. 2012; Leite et al. 2013; Melo et al. 2013). Well-designed approaches of reforestation policies could improve natural regeneration of native species at a landscape scale.
The objective of this study was to better understand the relative importance of landscape attributes in influencing the potential of tree cover increase to restore native woody plant richness and composition at the landscape scale. We focused on human-modified landscapes in the Ecuadorian Andes, where extensive farming and grazing takes place. We used a process-based vegetation model to simulate forest cover and species dynamics. We distinguished between reforestation through woody vegetation gain by natural, unassisted regeneration of abandoned fallows, and planted forests through managed plantations of exotic species on previously cultivated lands. We focused on the richness and composition of woody plants, as the diversity and structural characteristics of vegetation in regrowth forests were important determinants of faunal assemblages (Evelyn and Stiles 2003; Kanowski et al. 2006; Bowen et al. 2007). This research provides guidelines for reforestation policies that aim to enhance biodiversity restoration in human-modified landscapes.
Background
Many studies that have explored the effects of landscape patterns on species richness have been based on costly and time consuming fieldwork, carried out over short time periods (<5 years). In the tropics, available data reflect a sampling bias towards young regenerating forests (Chazdon et al. 2009b) and long-term datasets are scarce (Chazdon et al. 2009a; Gardner et al. 2009). Model simulations allow considerably more experimentation with various landscape patterns and over much longer time periods than possible in the field.
In this study, we used the spatially explicit landscape simulation model LANDIS-II (Scheller et al. 2007) to test various hypotheses on landscape factors influencing the recovery of woody species richness and composition resulting from tree cover increase in a tropical landscape. LANDIS-II is a well-established model that simulates the dynamics of forested landscapes over large spatial scales by incorporating ecological processes, such as succession, disturbance, and seed dispersal. In Latin America, LANDIS-II has been used to support conservation planning in a dry-land environment in Chile (Newton et al. 2011), to assess the potential for forest restoration in two Mexican landscapes under different disturbance regimes (Cantarello et al. 2011), and to predict the spatial extent of forest restoration (Birch et al. 2010). Thompson et al. (2011) showed that LANDIS-II could simulate forest to non-forest transitions.
The ecological value of forest regeneration is influenced by many factors. Beyond the local site characteristics (i.e., climate, soil fertility), landscape dynamics affect the initial establishment, species composition, and persistence of regenerating forests (Chazdon et al. 2009b). Landscape attributes (e.g., distance to forest remnants, local forest cover, heterogeneity of surrounding land covers) and temporal dynamics (e.g., time since abandonment) have a strong influence on the reforestation trajectory (Chazdon 2003). We tested seven hypotheses based on the literature (Table 1), which are justified here.
Local forest regrowth is linked with features of the surrounding landscapes through source populations and processes of seed dispersal. The extent and distribution of forest cover in the surrounding landscape serves as the external ecological memory and influences the recruitment of forest species during re-colonization (Chazdon 2014). In previous studies, the presence of old-growth forest remnants promoted the reassembly of tropical communities (Norden et al. 2009). Proximity to mature forests was particularly important during forest regrowth, as remnant vegetation provided a seed source (Guariguata and Ostertag 2001; Mesquita et al. 2001; Günter et al. 2007). The lack of dispersal of forest seeds was found to be one of the most important barriers to the restoration of tropical montane forest on abandoned pastures (Holl et al. 2000). We thus expect that the recovery of species richness and composition in regenerating fallows positively relates to a greater proximity to natural forest remnants (hypothesis 1) and a higher percentage of surrounding natural forest cover (hypothesis 2).
The diversity of different natural land covers (i.e., compositional heterogeneity) is also important during species regeneration (Gardner et al. 2009). Fahrig et al. (2011) suggested that biodiversity is expected to increase through the accumulation of species as more natural cover types are added to the landscape. Compositional heterogeneity was a key component for explaining landscape species richness (Wagner et al. 2000). We expect that a higher compositional heterogeneity of surrounding natural land covers positively affects the recovery of species richness and composition in regenerating fallows (hypothesis 3).
Island biography and metapopulation theories predict that the largest patches hold the most species as a result of lower extinction rates (MacArthur and Wilson 1967; Hanski 1999). In highly fragmented tropical landscapes, the size of remnant forest patches was positively related to plant species density (Arroyo-Rodríguez et al. 2009; Hernández-Stefanoni et al. 2011). We expect that a larger size of regenerating forest patches positively affects the recovery of species richness and composition (hypothesis 4).
It has been shown that plant species richness increased over time during forest regrowth (Guariguata and Ostertag 2001; Barlow et al. 2007; Dent and Wright 2009; Norden et al. 2009), making the age structure of regenerating forests an important determinant of landscape-level biodiversity. At initial stages of reforestation, typical old-growth species and endemic forest specialists may be missing, but they might be able to establish later (Chazdon 2003). We expect that time since abandonment of regenerating fallows positively affects the recovery of species richness and composition (hypothesis 5).
Altitude is a well-known driver of compositional variation in Andean vegetation (Jørgensen and Ulloa Ulloa 1994). Individuals of the same species have been found to differ in growth rate across their altitudinal ranges due to changes in temperature (Rapp et al. 2012). Recovery rates in cloud forests might lag behind due to lower temperatures at higher altitudes that cause trees to grow slowly. We expect that a higher elevation of regenerating abandoned fallows negatively affects the recovery of species richness and composition (hypothesis 6).
Finally, studies showed that the ecological benefits of plantations depend largely on the land cover they replace and whether planted trees are native or introduced species (Bremer and Farley 2010). Most exotic forest plantations established in high altitude grasslands in Ecuador negatively affected the water retention and carbon storage capacity of páramo soils (Molina et al. 2012; Farley et al. 2013) and offered little value for biodiversity conservation (Hofstede et al. 2002). We expect that planted forests using exotic pine species negatively affects the recovery of species richness and composition in previously cultivated areas (hypothesis 7).
We simulated restoration of woody plant richness and composition using LANDIS-II over a series of land-use scenarios that explored a range of variation in the seven key landscape attributes introduced above (Table 1). This approach facilitated the comparison of different landscape architectures of reforestation to evaluate the relative importance of landscape attributes on biodiversity restoration.
Data and methods
Study area
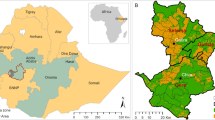
The Pangor watershed (78°50′–79°01′W, 1°43′–1°58′S) located in the Chimborazo province is a headwater basin of ~282 km2 and ranges from 1431 to 4333 masl (Fig. 1a). The mean annual precipitation is ~1400 mm and the topography is characterized by steep slopes, with over half of the catchment above 25°. Soils developed on volcanic ash deposits (mainly Andosols) and are characterized by a high water retention capacity and soil organic matter content when undisturbed (Buytaert et al. 2005). However, these soils become susceptible to erosion when cleared or cultivated.
Temperature, precipitation, and solar radiation are strongly correlated with elevation, leading to an altitudinal zonation of vegetation observed throughout the Ecuadorian Andes. The treeless area above 3400 masl is covered by grass páramo (i.e., pajonal), mainly consisting of bunch grasses with scattered herbs and shrubs. Forests of Polylepis trees occur sporadically in this zone, supporting additional woody species. Grass páramos can support a high number of species and are considered natural vegetation cover, although scientists have claimed it is man-made through burning during the Holocene (White 2013), consequently lowering the historical treeline (Jørgensen and Ulloa Ulloa 1994). Below the treeline, upper montane forest (>3000 masl) and montane forest (<3000 masl) cover the catchment. Upper montane forest is characterized by contorted tree growth with many multi-stemmed trees or giant shrubs. Montane forest (i.e., Andean cloud forest) is composed of a rich diversity of taller trees cover by diverse epiphyte flora and lianas.
Balthazar et al. (2015) highlighted a shift from net deforestation to net reforestation in the Pangor watershed in the early 1990s based on a 50-year time series consisting of aerial photographs and satellite images. Most of the lowland in the catchment has been cleared of its natural vegetation for small-scale agriculture of mainly beans, potatoes, and maize, while extensive grazing occurs on the high altitude grasslands. Since the late 1980s, pine plantations were established in the higher parts of the catchment (>3200 masl), after being promoted by governmental programs that linked the Ministry of Agriculture with rural landowners (Farley 2007, 2010). Approximately five percent of the watershed was covered with pine plantations by 2009 (Vanacker et al. 2013). Additionally, some abandoned agricultural lands were left for natural regeneration (Balthazar et al. 2015).
Forest landscape model
LANDIS-II is a stochastic, spatially-explicit, raster- and process-based model that simulates the dynamics of forested landscapes on large spatial and temporal scales by incorporating ecological processes, such as succession, disturbance, and seed dispersal (Scheller et al. 2007). Species composition and succession is driven by the user-defined life history characteristics of each modeled species (i.e., longevity, minimum age at reproduction, shade tolerance, minimum/maximum seed dispersal distances, and resprout probability) and their establishment coefficient. This coefficient governs the probability of each species to establish in different subdivisions of the landscapes that are homogeneous in environmental conditions (i.e., ecoregions). For each cell, the presence and absence of 10-year age cohorts is monitored. A more detailed description of LANDIS-II can be found elsewhere (He et al. 1999; Mladenoff 2004; Scheller et al. 2007; http://www.landis-ii.org).
Field survey data
To determine the floristic composition across the Pangor watershed, we collected species data in September and October 2013 from twelve 0.02 ha transects (40 × 5 m) (Table S1). These transects were parallel to the contour lines of the slope in montane forest (n = 3), upper montane forest (n = 2), natural regenerating forest (n = 2), pine plantations (n = 4), and páramo (n = 1). Jorge Caranqui collected additional species data in 2007 from five 0.02 ha transects (50 × 4 m) in montane forest (unpublished data), resulting in a total of eight montane forest plots. Plots were selected using a subjective sampling approach, to identify sample units that are assumed to be representative of the entire population.
In each plot, plant height and diameter at breast height (DBH) for all woody plants >1 cm DBH were recorded. Based on collected specimens, all individuals were subsequently identified at the Herbario QCA at the Pontificia Universidad Católica del Ecuador. Plant species names and authorities were according to Jørgensen and León-Yánez (1999). We excluded six individuals that could not be identified to genus level.
Model parameterization
LANDIS-II is designed to accept raster imagery as spatially explicit input. The required input maps are an ecoregion map that describes the ecological conditions affecting species establishment and an initial communities map that describes the distribution and age of cohorts for each species at the onset of simulations. All maps were produced and manipulated using ArcGIS 10.2.2 (© 2014 ESRI Inc., Redlands, California, USA).
Ecoregions
Vegetation zonation in the Ecuadorian Andes is strongly related to altitude. We selected the elevation zonation as proposed by Jørgensen and Ulloa Ulloa (1994). We generated a total of 11 active ecoregions by overlaying a Digital Elevation Model and a beam solar irradiation map (Table S2). All regions above 3,700 masl and all agricultural raster cells were inactive during simulations as they mainly consisted of herbaceous and small shrub vegetation in most of our study area. The species establishment probabilities were derived from MaxEnt models (Phillips et al. 2006) as the average probability of occurrence for each species in each ecoregion. Input data for these models included species occurrence data from the MOBOT Tropicos® database (©2014 Missouri Botanical garden, USA) and bioclimatic variables extracted from the Worldclim database (Hijmans et al. 2005).
Initial communities
A land cover map with a spatial resolution of 30 m was made for 2010 based on a supervised classification method using Worldview-2 imagery, with an average accuracy of 80.4 % and a Kappa coefficient of 77.5 % (Marie-Pierre de Wouters and Vincent Balthazar—unpublished data). Six land cover types were identified: (1) natural forest remnants (i.e., cloud and upper montane forest); (2) shrub vegetation mainly characterized by natural regeneration; (3) sub páramo; (4) grass páramo; (5) agriculture; and (6) pine plantations (Fig. 1).
The initial distribution, composition, and age structure of forest communities for each cell in the landscape was determined by combining the land cover classification with the field survey data. We estimated ages of individuals by dividing the measured height with the maximum documented height, multiplied with the maximum documented longevity for each species.
Species attributes
We selected a subset of the 19 most abundant species based on their importance value index, which is the sum of relative density, relative frequency, and relative dominance found during the field survey (Mishra 1968). For each modeled species, we extracted life history characteristics from the literature and from a consultation of local experts (Table S3).
Scenarios
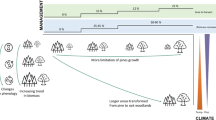
To explore the relative importance of landscape attributes on the re-colonization of biodiversity during reforestation, we developed alternative land-use scenarios that differed in the pattern of reforestation (Table 1). First, we estimated the rate of past natural regeneration based on post-classification change detection between land-cover classifications of two satellite imagery for 1991 and 2009 (Balthazar et al. 2015), using Markov Chain Analysis (MCA) within the Land Change Modeler (LCM) in IDRISI Taiga v.17.01 (©2015 Clark Labs, Clark University, Worcester, USA). We projected the area of natural regenerating forests from 2010 to the year 2055 to ~943 ha (i.e., 10,476 raster cells). All natural regeneration during simulations took place in former agricultural raster cells and at equal time intervals of 15 years (i.e., year 2010, 2025, and 2040) to disintegrate the influence of time since onset of regeneration on species richness.
The spatial allocation of raster cells under reforestation varied for each scenario, altering the: (i) proximity to remnant forest; (ii) percentage of surrounding forest cover; (iii) compositional heterogeneity of surrounding natural land-cover; (iv) patch size; (v) elevation; and (vi) type of reforestation (i.e., planted pine forests). We created one random reforestation scenario with a similar area of reforestation that served as a reference to which all other scenarios were compared. The proximity to remnant forest was calculated as the Euclidean distance to the nearest forested cell. The percentage of forest cover in the surrounding landscape was calculated in a 200 m radius surrounding the focal cell. Compositional heterogeneity was determined as the Simpson’s diversity index, which represents the probability that any two pixels selected at random were different patch types, calculated for a 200 m radius moving window using FRAGSTATS software V.4.2 (McGarigal et al. 2012). We varied patch size by increasing from 150 m to 250 m the buffer size in which reforestation occurred around randomly selected raster cells. For the plantation scenario, an age-cohort of pines was planted at the onset of regeneration at random locations across the landscape. We created a total of one random, one planted forest, and 15 reforestation scenarios (Table 2; Fig. S1).
We implemented the Base Harvest (v2.2) extension of LANDIS-II (Gustafson et al. 2000) to simulate reforestation during simulations. At each harvesting time step, raster cells identified for reforestation were completely harvested until the onset of reforestation (i.e., 2010, 2025, and 2040). Simulations were conducted for 55 years. Each scenario was replicated three times with a different random seed number to account for within-scenario stochasticity. The time steps were 5 years for tree succession and 1 year for the harvesting extension.
Data analysis
The Age Cohort Statistics v1.0 extension of LANDIS-II generated raster maps for each time step containing for each pixel: (i) the minimum and maximum age across all species; (ii) the presence or absence of all species; and (iii) the total number of tree species. All output maps were analyzed using the raster package (Hijmans 2014) in R (R Development Core Team 2010). We recalculated species richness maps to contain only individuals >1 year.
At the landscape scale, we calculated the mean and standard deviations for the area in hectares containing a number of species and for the cover percentage of selected species over time for three repeated simulations of each scenario. We selected the following species for analysis, based on their life characteristics (e.g., shade-tolerance, seed dispersal limitations): Aegiphila bogotensis, Baccharis latifolia, Boehmeria aspera, Clusia multiflora, Gynoxys spp., Hedyosmum luteynii, and Podocarpus glomeratus.
We compared simulated species richness for reforesting fallows between each scenario and the random scenario using a pair matched case-comparison method. This approach accounts for the imbalance in co-variables (i.e., altitude, solar irradiance, time since abandonment) that influences species establishment during simulations. We conducted a genetic matching with replacement in R (Sekhon 2011). This algorithm found the optimal pair of matching that achieves covariance balance. We assessed the causal effect of a given landscape attribute on the simulated species richness based on matched data by comparing means of each scenario against the random scenario, using Wilcoxon-Mann-Withney tests. In the subsequent results, we only included the results for scenarios that showed a significantly different average species richness compared to the random scenario for the matching analyses. These scenarios are indicated in the text as ‘significant scenarios’. All other results are included in the appendices and are indicated in the text as ‘non-significant scenarios’.
Descriptive statistics were used to calculate the mean cover percentage of reforesting fallows containing a number of species. In addition we calculated α-, and β-diversity values to assess the changes in landscape biodiversity, using the vegan package in R (Oksanen et al. 2007). We calculated α-diversity as the mean species richness in reforesting raster cells (Whittaker 1972) and β-diversity as the dissimilarity between two reforesting raster cells based on counts at each cell, using the Bray-Curtis index (Bray and Curtis 1957). We also calculated the cover percentage of selected species over time for reforesting fallows in each scenario.
Results
Field survey
A total of 80 woody species were encountered in the field sites, although taxonomic uncertainty over some genera led to congeneric species being grouped under the same species life history characteristics in the model. The most important tree species were Hedyosmum luteynii and Aegiphila bogotensis in montane forest, Berberis spp. and Brachyotum ledifolium in upper montane forest, and Miconia spp. and Baccharis latifolia in regrowth forest (for all species encountered, see Table S4). A total of 49 species were found in montane forest with an average richness of 12 species per plot. Natural regenerated forests were species rich with ~15 species per plot. However, species composition did not resemble montane forests completely, containing more shade-intolerant pioneer species. In the páramo plot, we found a total of 12 woody species. Pine plantations contained little or no undergrowth. We encountered more than two shrub species only in one plantation that was less densely planted with pines and located in close proximity to remnant forest.
Species restoration, proximity, and cover percentage of remnant forest patches
Consistent with hypotheses 1 and 2, we found significantly higher species richness for regenerating fallows located within 400 m from forest remnants and surrounded by more than 30 % cover of forest remnants compared to random reforestation (P < 0.05, two-sided Wilcoxon-test; Fig. 3). For these scenarios, α-diversity values were high but β-diversity values were low and decreasing, indicating high mean species richness and a small differentiation between species occurring in fallows (Fig. 5). When forest cover dropped below 30 %, species richness recovery in natural regenerating forests was significantly lower (P < 0.05, two-sided Wilcoxon-test; Fig. 3) and more natural regenerating forests remained uncolonized (Fig. 4).
Simulated species composition of regenerating fallows resembled natural forests more when occurring in the proximity to natural forest remnants and in areas with a high percentage of remnant forest cover in the surrounding. In these scenarios, montane forest species, such as A. bogotensis, C. multiflora, H. luteynii, and P. glomeratus, were present in a larger number of regenerating fallows compared to other scenarios (Fig. 6a, d, f, h).
Species restoration and compositional heterogeneity
Consistent with hypothesis 3, we found significantly higher species richness in regenerating fallows with high compositional heterogeneity of surrounding natural cover types compared to random reforestation (P < 0.05, two-sided Wilcoxon-test; Fig. 3). Regenerating fallows located in compositional heterogeneous surroundings were increasingly dissimilar in species over time (Fig. 5b), indicating enhanced biodiversity at the landscape level (Fig. 2). These regenerating fallows were more often occupied by species typical of montane (Fig. 6a, d, h), shrub (Fig. 6c), and páramo (Fig. 6e) vegetation.
Species restoration and patch size
Contrary to hypothesis 4, our simulations showed no significant difference between the average species richness of regenerating forest of either small or large patch sizes compared to random reforestation (Fig. 3). Patch size scenarios also did not differ in terms of species richness at the landscape scale (Fig. 2) or recolonization dynamics over time (Fig. S3).
Boxplot showing the median, 25–75 % quartiles, and non-outlier ranges (in black), and the average and standard deviations (in grey) of simulated species richness after 55 years in reforesting fallows over three runs for each scenario. The horizontal dashed line indicates average species richness for the random scenario. Compared to the random scenario, average species richness was significantly higher for the distance < 200 m, distance 200–400 m, forest cover > 50 %, forest cover > 30 %, diversity index > 50 %, altitude 2400–3000 m, and altitude 3000–3400 m scenarios and significantly lower for the forest cover < 30 %, altitude < 2400 m, altitude > 3400 m, and plantation scenarios. Significant differences are indicated by asterisks (P < 0.05, two-sided Wilcoxon-test)
Species restoration over time
Species establishment in abandoned fallows was fast during all natural regeneration scenarios (Fig. 4). Consistent with hypothesis 5, most species rapidly increased their cover percentage in fallows after abandonment in all scenarios, except for the plantation scenario (Fig. 6). The presence of short-lived, shade-intolerant shrub species declined several decades after colonization (Fig. 6c, e), due to increasing levels of simulated shade during recolonization. This decline resulted in a decrease in species richness over time, shown by diminishing α-diversity values for all scenarios (Fig. 5a).
The percentage of reforesting fallows containing a number of woody species over time since abandonment for significant scenarios. Re-colonization of abandoned agricultural lands was dynamic during simulations. In particular, the distance < 200 m, forest cover > 50 %, forest cover > 30 %, diversity index > 50 %, and altitude 3000–3400 m scenarios indicated a relatively large area that contained a large number of species
The development of the simulated a mean species diversity per cell (i.e., α-diversity) and b differentiation among these sites (i.e., β-diversity) over time for significant scenarios. α-Diversity decreased over time for all scenarios, whereas β-diversity increased for diversity index > 50 %, altitude 3000–3400 m, and altitude > 3400 m scenarios, whereas it decreased for the remaining scenarios. Note the discontinued y-axis
Species restoration and altitude
Consistent with hypothesis 6, species richness in regenerating fallows was significantly lower at high altitudes (>3400 masl) and significantly higher at intermediate altitudes (between 2400 and 3400 masl) compared to random reforestation (P < 0.05, two-sided Wilcoxon-test; Fig. 3). Contrary to hypothesis 6, however, we found significant lower species richness for regenerating fallows located below 2,400 masl compared to random reforestation (P < 0.05, two-sided Wilcoxon-test; Fig. 3), because the lower basin of the catchment has been almost completely cleared of natural forest remnants. Regenerating fallows located between 3,000 and 3,400 masl showed the highest α-diversity values (Fig. 5a). Species associated with lower elevations, were more abundant in naturally regenerated forests below 2,400 m (Fig. 6c, g), while species that were able to establish and grow at high altitudes increased their cover percentage at intermediate altitudes (between 3000 and 3400 masl) (Fig. 6e, f, h).
The cover percentage of natural regenerated fallows occupied by each selected species over time under significant scenarios. a Aegiphila bogotensis; b Baccharis latifolia; c Boehmeria aspera; d Clusia multiflora; e Gynoxys spp.; f Hedyosmum luteynii; g Miconia spp.; and h Podocarpus glomeratus. Shapes correspond to scenarios. Values presented are means for three repeated simulations for reforestation onset in 2010. Note that the y-axis values are not fixed
Species restoration in planted forests
Consistent with hypothesis 7, we found significantly lower species richness for planted forest on previously cultivated land compared to random reforestation (P < 0.05, two-sided Wilcoxon-test; Fig. 3). Planted forests using exotic pine species prevented the establishment of all other species (Figs. 4, 5a, 6), leading to a diminished overall species richness at the landscape scale (Fig. 2).
Discussion
Species restoration, proximity, and cover percentage of remnant forest patches
Our results demonstrate that remnant vegetation was an important seed source that determined the recovery of both species richness and composition during natural regeneration of abandoned fallows. Consistent with our results, other studies indicate a smaller numbers of individuals and species in abandoned fields under reforestation located further away from forests (e.g., Mesquita et al. 2001; Robiglio and Sinclair 2011). In the tropical mountain rainforest of Southern Ecuador, the total number of species found in natural regenerating forest decreased from 47 to 31 with increasing distance from the forest edge (Günter et al. 2007). In a review of more than 30 years of ecological restoration in Brazil, Rodrigues et al. (2009) have stated that the distance to the forest edge reduces species richness and diversity long after the onset of forest regeneration. Our results support the idea that below a 20–30 % threshold of natural surrounding habitat, isolation becomes an important factor determining species richness (Andrén 1994; Fahrig 1998, 2003).
Species restoration and compositional heterogeneity
Compositional heterogeneity increased the recovery of species richness and composition during our simulations, due to the accumulation of species associated with different natural cover types. Similarly, Kumar et al. (2006) found that compositional heterogeneity (i.e., Simpson’s diversity index) was positively correlated with native and non-native plant species richness for 180 plots in the Rocky Mountains. This effect was strongest at an intermediate spatial extent (240 m) (Kumar et al. 2006), which is comparable to the spatial scale of 200 m at which we found a positive effect of compositional heterogeneity on species richness. An increase in patch density (i.e., increased compositional heterogeneity), which resulted from low levels of land use, enhanced plant diversity in a tropical forest landscape in Mexico (Hernández-Stefanoni et al. 2011). In a modeling approach, it was found that plant diversity was highest in heterogeneous landscapes with more small patches and low aggregation (Steiner and Köhler 2003).
Besides compositional heterogeneity, it has also been suggested that species diversity is positively correlated with an increasing complexity of patch shapes of different cover types (i.e., configurational heterogeneity) (Fahrig et al. 2011). In practice, these two dimensions are difficult to control independently (Fahrig et al. 2011). This study only focused on compositional heterogeneity. The nature of landscape heterogeneity-biodiversity relationships still needs further exploration. The effect of compositional heterogeneity on biodiversity likely depends on contextual factors, such as historical landscape patterns, time frame, and the nature of reforestation.
Species restoration and patch size
Our finding that patch size of regenerating fallows did not affect species richness or composition contradicts other studies. Several researchers have suggested that smaller patches support smaller populations, which increases the probability of extinction (Ellstrand and Elam 1993; Fischer and Lindenmayer 2007). Harrison (1999) found a higher local endemic plant diversity (α-diversity) and among-site differentiation (β-diversity) on small patches when comparing communities from patchy and continuous habitats. In Yucatan Peninsula in Mexico, larger regenerating forest patches with fewer edges had higher woody species diversity compared to small regenerating forest patches of irregular shapes (Hernández-Stefanoni et al. 2011). Arroyo-Rodríguez et al. (2009) found that patch size explained density-area relationships of plants in highly fragmented landscapes. However, richness of native and non-native plants did not increase with size of planted revegetation established on agricultural land in Gippsland southeastern Australia (Munro et al. 2009), which might be the result of insufficient maturity to display significant species density-area relationships.
Fragmentation in human-modified landscapes leads to the decrease in mean patch size and thus the creation of more forest edges (Fischer and Lindenmayer 2007). Several studies have suggested that light-demanding generalist, exotic, and invasive species may benefit from edge environments in tropical forest remnants (Harper et al. 2005; Tabarelli et al. 2010). Natural regenerating forests that persist as isolated small patches were recognized as edge-affected habitats (Tabarelli et al. 2012) and represented a suitable habitat for a subset of species only (Barlow et al. 2007). The absence of species-area relationships in the LANDIS-II model could explain the lack of patch size effect during model simulations. Nonetheless, empirical work demonstrates the importance of patch size for species conservation (Arroyo-Rodríguez et al. 2009; Hernández-Stefanoni et al. 2011).
Species restoration over time
As found by other studies, age structure of regenerating fallows was an important determinant for biodiversity recovery (Guariguata and Ostertag 2001; Barlow et al. 2007; Dent and Wright 2009; Norden et al. 2009). In a quantitative review of the value of natural regenerating forests across the tropic, it was found that similarity to old-growth forests increased rapidly with age (Dent and Wright 2009). Interestingly, we found a decrease in species richness over time for all scenarios, due to the decline of short-lived, shade-intolerant pioneer species several years after colonization. This process indicates a shift in successional stages of reforestation, with shade-intolerant, short-lived pioneers being replaced by long-lived pioneers and generalist species (Wirth et al. 2009). Long-term species time-series data are rare for tropical forest sites (Gardner et al. 2009). As a consequence, recolonization trajectories over longer time periods are not well understood. In temperate forests, however, the decline in species richness as forest succession shifts to later stages is well recorded (e.g., Horn 1974; Vetaas 1997; Bonet and Pausas 2004; Amici et al. 2013).
Species restoration and altitude
In the Andes, altitude is an important factor that affects plant community composition (Young 1993; Lauer and Rafiqpoor 2000), which supports our findings. Variations in species composition related to altitude were most obvious in disturbed forest fragments and played a role in recolonization of regenerating pastures (MacLaren et al. 2014). It has been suggested that the creation of more open areas at high altitudes in the Andes, due to agriculture and pasture expansion, benefits the establishment of woody species associated with tree line, alpine shrub land, or páramo (Sarmiento 2002; MacLaren et al. 2014). Consistent with our findings, regenerating pastures in highland areas may prove beneficial for woody species associated with high altitudes in lower regions previously covered with mature forest. This phenomenon is thought to be widespread in the Andes (Sarmiento 2002). Nevertheless, the establishment of small pioneer shrub or tree species, such as B. latifolia, promoted the regrowth of other sensitive species (Posada et al. 2000). At very high altitudes (>3400 masl), growth of woody species lagged behind due to lower temperatures causing trees to grow slowly (Rapp et al. 2012). Natural regeneration of abandoned fallows is therefore limited at high altitudes.
Species restoration in planted forests
Our simulations showed that planted pine forests provided no benefits for biodiversity recovery. Nonetheless, almost all forest plantation activities in Ecuador have involved the planting of exotic species, such as Pinus and Eucalyptus spp. In regions with a high endemic biodiversity, such as the highlands of Ecuador, exotic plantations posed a high risk to native vegetation (Bremer and Farley 2010) and altered hydrological properties of soils (Farley et al. 2004; Molina et al. 2012). Plantations may provide habitats for certain species and increase forest connectivity if managed with long rotations and if located around natural forest fragments (Meyfroidt and Lambin 2011).
Lindenmayer et al. (2009) found that compositional heterogeneity at the landscape scale partly determines the level of biodiversity supported by plantation landscapes. Bird species richness was significantly reduced by 4-9 species in patches of native eucalypt forest where surrounding stands of exotic Pinus radiata were clear-felled compared to patches where surrounding pine stands remained unlogged (Lindenmayer et al. 2009). However, spatial heterogeneity was not accounted for in our plantation scenario and may affect species recovery at the landscape scale.
Limitations
Forest landscape models explicitly represent spatial processes such as seed dispersal and disturbance spread. The value of forest models does not lie in the exact prediction of future conditions, but in their potential to explore the consequences of explicitly stated assumptions. They allow for considerable experimentation with different landscape patterns and human processes that would not be possible in the field given time and money constraints. Nevertheless, the dependence on relationships estimated in the past to predict future dynamics is a limitation to all forest models (Gustafson 2013), which makes it difficult to perform rigorous model validation. In the tropics, there is a lack of long-term data on the ecological behavior of forests, which hinders model parameterization. The greatest uncertainties for model parameterization in this study were: (i) the dispersal ability of modeled species; (ii) establishment probabilities of tree species across the landscape; and (iii) initial forest composition and age structure. We addressed these uncertainties by including high resolution remote sensing imagery, MaxEnt models, scientific literature, and consultations of local experts.
Some other factors that potentially affect future species distribution were not taken into account, such as climate change, differences in microhabitat, and seed dispersal. Andean plant species are predicted to shift their distributions to higher altitudes in response to changes in temperature and humidity (Feeley and Silman 2010). Microhabitat differences in soil fertility can also strongly affect competition and growth during re-colonization (Guariguata and Ostertag 2001). Additionally, animal movement is influenced by landscape patterns and affects seed rain (Jesus et al. 2012). We neglected the presence of seed banks from which seeds of mostly shrubs and short-lived pioneer trees may establish at the time of abandonment during natural regeneration (Holl 2007). Finally, in the Ecuadorian Andes, many farmers leave remnant trees and shrubs on agricultural lands and, in some cases, allow vegetation to regenerate (Middendorp, pers. obs.). Our assumption that cultivated lands were bare may have underestimated the natural regeneration potential during model simulations.
Conclusion and policy recommendations
Forest regeneration might prove fundamental to future conservation initiatives, but the value of regenerating forests is context dependent. Conservation policies have started to recognize the role of forest regrowth at the landscape level (Harvey et al. 2008). Reducing deforestation and increasing forest cover became a national priority under the Ecuadorian National Development Plan, known as the Plan for Good Living 2009–2013. For example, reforestation plans will be incorporated in the SocioBosque program, in which the Ministry of Environment (MAE) offers yearly monetary payments to private and communal landholders in return for maintaining forest cover (de Koning et al. 2011). A better integration of restoration ecology and landscape ecology is needed for mainstreaming successful programs (Metzger and Brancalio 2013). Our research provides specific guidelines for the design of reforestation projects that enhance biodiversity restoration in human-modified landscapes. Spatial targeting of reforestation should take into account the presence of natural remnant forest patches, the spatial pattern of the landscape, altitudinal gradients, and the type of regenerating forest, as summarized below.
We emphasize that the conservation and regeneration of natural remnant forest patches is crucial. A considerable fraction of human-modified landscapes no longer serves as a source of seed dispersal due to habitat loss. This may impose a limitation for natural forest regeneration (Zimmerman et al. 2000). The proximity and cover percentage of surrounding remnant forest patches is an important factor that determines the success of natural regeneration after abandonment. Subsequent management and conservation of regenerating forests is essential, because regenerating forests need to reach a certain age to become a source of propagules (van Breugel et al. 2013).
Additionally, we suggest that diversity and spatial arrangement of habitat patches determines the successful recolonization of the species that inhabit different environmental conditions. Based on our research, we recommend maintaining compositional heterogeneity as an important guideline in conservation management. Even though our results did not show a significant effect of patch size on species restoration, we recommend stimulating the regeneration of large and aggregated patches of natural regenerating forests. Habitat-specialists that need large areas of contiguous habitat for their survival may disappear from landscapes with high fragmentation and compositional heterogeneity (Fahrig et al. 2011). The limitations of conservation efforts to small forest fragments only risks biotic homogenization of forest-requiring species, as shown in the northern parts of the Atlantic Forest region in Brazil (Lôbo et al. 2011).
Natural regeneration following fallow abandonment often occurs on marginal lands (Crk et al. 2009), such as upland areas (Asner et al. 2009). Our results suggest, however, that reforestation in upland areas might not be enough for species recovery. We recommend that natural regeneration be encouraged at various altitudes, allowing a variety of species associated with different elevation ranges to recover at the landscape scale.
Finally, we found that planted forests using exotic pine species provided no benefits for species recovery. Active planting of native species might stimulate restoration of native biodiversity, especially in regions were natural land covers are strongly diminished.
References
Amici V, Santi E, Filibeck G, Diekmann M, Geri F, Landi S, Scoppola A, Chiarucci A (2013) Influence of secondary forest succession on plant diversity patterns in a Mediterranean landscape. J Biogeogr 40(12):2335–2347
Andrén H (1994) Effects of habitat fragmentation on birds and mammals in landscapes with different proportions of suitable habitat: a review. Oikos 71(3):355–366
Arroyo-Rodríguez V, Pineda E, Escobar F, Benítez-Malvido J (2009) Value of small patches in the conservation of plant-species diversity in highly fragmented rainforest. Conserv Biol 23(3):729–739
Asner GP, Rudel TK, Aide TM, Defries R, Emerson R (2009) A contemporary assessment of change in humid tropical forests. Conserv Biol 23(6):1386–1395
Balthazar V, Vanacker V, Molina A, Lambin EF (2015) Impacts of forest cover change on ecosystem services in high Andean mountains. Ecol Indic 48:63–75
Barlow J, Gardner TA, Araujo IS, Ávila-Pires TC, Bonaldo AB, Costa JE, Esposito MC, Ferreira LV, Hawes J, Hernandez MIM, Hoogmoed MS, Leite RN, Lo-Man-Hung NF, Malcolm JR, Martins MB, Mestre LAM, Miranda-Santos R, Nunes-Gutjahr AL, Overal WL, Parry L, Peters SL, Ribeiro-Junior MA, da Silva MNF, da Silva Motta C, Peres CA (2007) Quantifying the biodiversity value of tropical primary, secondary, and plantation forests. PNAS 104(47):18555–18560
Birch JC, Newton AC, Aquino CA, Cantarello E, Echeverría C, Kitzberger T, Schiappacasse I, Garavito NT (2010) Cost-effectiveness of dryland forest restoration evaluated by spatial analysis of ecosystem services. PNAS 107(50):21925–21930
Bonet A, Pausas J (2004) Species richness and cover along a 60-year chronosequence in old-fields of southeastern Spain. Plant Ecol 174(2):257–270
Bowen ME, McAlpine CA, House APN, Smith GC (2007) Regrowth forests on abandoned agricultural land: a review of their habitat values for recovering forest fauna. Biol Conserv 140(3–4):273–296
Bray JR, Curtis JT (1957) An ordination of the upland forest communities of Southern Wisconsin. Ecol Monogr 27(4):325–349
Bremer L, Farley K (2010) Does plantation forestry restore biodiversity or create green deserts? A synthesis of the effects of land-use transitions on plant species richness. Biodivers Conserv 19(14):3893–3915
Buytaert W, Wyseure G, De Bièvre B, Deckers J (2005) The effect of land-use changes on the hydrological behaviour of Histic Andosols in south Ecuador. Hydrol Process 19(20):3985–3997
Cantarello E, Newton AC, Hill RA, Tejedor-Garavito N, Williams-Linera G, López-Barrera F, Manson RH, Golicher DJ (2011) Simulating the potential for ecological restoration of dryland forests in Mexico under different disturbance regimes. Ecol Model 222(5):1112–1128
Chazdon RL (2003) Tropical forest recovery: legacies of human impact and natural disturbances. Perspect Plant Ecol 6(1–2):51–71
Chazdon RL (2008) Beyond deforestation: restoring forests and ecosystem services on degraded lands. Science 320(5882):1458–1460
Chazdon RL (2014) Second growth: the promise of tropical forest regeneration in an age of deforestation. The University of Chicago Press, Chicago
Chazdon RL, Harvey CA, Komar O, Griffith DM, Ferguson BG, Martínez-Ramos M, Morales H, Nigh R, Soto-Pinto L, Van Breugel M, Philpott SM (2009a) Beyond reserves: a research agenda for conserving biodiversity in human-modified tropical landscapes. Biotropica 41(2):142–153
Chazdon RL, Peres CA, Dent D, Sheil D, Lugo AE, Lamb D, Stork NE, Miller SE (2009b) The potential for species conservation in tropical secondary forests. Conserv Biol 23(6):1406–1417
Chokkalingam U, de Jong W (2001) Secondary forest: a working definition and typology. International Forestry Review 3(1):19–26
Crk T, Uriarte M, Corsi F, Flynn D (2009) Forest recovery in a tropical landscape: what is the relative importance of biophysical, socioeconomic, and landscape variables? Landscape Ecol 24(5):629–642
de Koning F, Aguiñaga M, Bravo M, Chiu M, Lascano M, Lozada T, Suarez L (2011) Bridging the gap between forest conservation and poverty alleviation: the Ecuadorian Socio Bosque program. Environ Sci Policy 14(5):531–542
DeClerck FAJ, Chazdon R, Holl KD, Milder JC, Finegan B, Martinez-Salinas A, Imbach P, Canet L, Ramos Z (2010) Biodiversity conservation in human-modified landscapes of Mesoamerica: past, present and future. Biol Conserv 143(10):2301–2313
Dent DH, Wright SJ (2009) The future of tropical species in secondary forests: a quantitative review. Biol Conserv 142(12):2833–2843
Dunn RR (2004) Recovery of faunal communities during tropical forest regeneration. Conserv Biol 18(2):302–309
Ellstrand NC, Elam DR (1993) Population genetic consequences of small population size: implications for plant conservation. Annu Rev Ecol Syst 24:217–242
Evelyn MJ, Stiles DA (2003) Roosting requirements of two frugivorous bats (Sturnira lilium and Arbiteus intermedius) in fragmented Neotropical forest. Biotropica 35(3):405–418
Fahrig L (1998) When does fragmentation of breeding habitat affect population survival? Ecol Model 105(2–3):273–292
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol S 34:487–515
Fahrig L, Baudry J, Brotons L, Burel FG, Crist TO, Fuller RJ, Sirami C, Siriwardena GM, Martin JL (2011) Functional landscape heterogeneity and animal biodiversity in agricultural landscapes. Ecol Lett 14(2):101–112
FAO (2015) Global forest resources assessment 2015. FAO, Rome
Farley KA (2007) Grasslands to tree plantations: forest transition in the Andes of Ecuador. Ann Assoc Am Geogr 97(4):755–771
Farley KA (2010) Pathways to forest transition: local case studies from the Ecuadorian Andes. J Lat Am Geogr 9(2):7–26
Farley KA, Bremer LL, Harden CP, Hartsig J (2013) Changes in carbon storage under alternative land uses in biodiverse Andean grasslands: implications for payment for ecosystem services. Conserv Lett 6(1):21–27
Farley KA, Kelly E, Hofstede R (2004) Soil organic carbon and water retention after conversion of grasslands to pine plantations in the Ecuadorian Andes. Ecosystems 7(7):729–739
Feeley KJ, Silman MR (2010) Land-use and climate change effects on population size and extinction risk of Andean plants. Glob Change Biol 16(12):3215–3222
Fischer J, Lindenmayer DB (2007) Landscape modification and habitat fragmentation: a synthesis. Global Ecol Biogeogr 16(3):265–280
Gardner TA, Barlow J, Chazdon R, Ewers RM, Harvey CA, Peres CA, Sodhi NS (2009) Prospects for tropical forest biodiversity in a human-modified world. Ecol Lett 12(6):561–582
Guariguata MR, Ostertag R (2001) Neotropical secondary forest succession: changes in structural and functional characteristics. Forest Ecol Manag 148(1–3):185–206
Günter S, Weber M, Erreis R, Aguirre N (2007) Influence of distance to forest edges on natural regeneration of abandoned pastures: a case study in the tropical mountain rain forest of Southern Ecuador. Eur J Forest Res 126(1):67–75
Gustafson EJ (2013) When relationships estimated in the past cannot be used to predict the future: using mechanistic models to predict landscape ecological dynamics in a changing world. Landscape Ecol 28(8):1429–1437
Gustafson EJ, Shifley SR, Mladenoff DJ, Nimerfro KK, He HS (2000) Spatial simulation of forest succession and timber harvesting using LANDIS. Can J Forest Res 30(1):32–43
Hall J, Van Holt T, Daniels A, Balthazar V, Lambin EF (2012) Trade-offs between tree cover, carbon storage and floristic biodiversity in reforesting landscapes. Landscape Ecol 27(8):1135–1147
Hanski I (1999) Metapopulation ecology. Oxford University Press, Oxford
Harper KA, Macdonald SE, Burton PJ, Chen J, Brosofske KD, Saunders SC, Euskirchen ES, Roberts D, Jaiteh MS, Esseen PA (2005) Edge influence on forest structure and composition in fragmented landscapes. Conserv Biol 19(3):768–782
Harrison S (1999) Local and regional diversity in a patchy landscape: native, alien, and endemic herbs on serpentine. Ecology 80(1):70–80
Harvey CA, Komar O, Chazdon R, Ferguson BG, Finegan B, Griffith DM, MartÍNez-Ramos M, Morales H, Nigh R, Soto-Pinto L, Van Breugel M, Wishnie M (2008) Integrating agricultural landscapes with biodiversity conservation in the Mesoamerican hotspot. Conserv Biol 22(1):8–15
He HS, Mladenoff DJ, Boeder J (1999) An object-oriented forest landscape model and its representation of tree species. Ecol Model 119(1):1–19
Hernández-Stefanoni JL, Dupuy J, Tun-Dzul F, May-Pat F (2011) Influence of landscape structure and stand age on species density and biomass of a tropical dry forest across spatial scales. Landscape Ecol 26(3):355–370
Hijmans RJ (2014) raster: geographic data analysis and modeling. R package version 2.3-12 http://CRAN.R-project.org/package=raster
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25(15):1965–1978
Hofstede RGM, Groenendijk JP, Coppus R, Fehse JC, Sevink J (2002) Impact of pine plantations on soils and vgetation in the Ecuadorian high Andes. Mt Res Dev 22(2):159–167
Holl KD (2007) Old field vegetation succession in the Neotropics. In: Cramer VA, Hobbs RJ (eds) Old fields: dynamics and restoration of abandoned farmland. Island Press, Washington, pp 93–118
Holl KD, Loik ME, Lin EHV, Samuels IA (2000) Tropical montane forest restoration in Costa Rica: overcoming barriers to dispersal and establishment. Restor Ecol 8(4):339–349
Horn HS (1974) The ecology of secondary succession. Ann Rev Ecol Syst 5:25–37
Jesus FM, Pivello VR, Meirelles ST, Franco GADC, Metzger JP (2012) The importance of landscape structure for seed dispersal in rain forest fragments. J Veg Sci 23:1126–1136
Jørgensen PM, León-Yánez S (1999) Catalogue of the vascular plants of Ecuador. Monogr Syst Bot Missouri Bot Gard 75: i–viii, 1–1182
Jørgensen PM, Ulloa Ulloa C (1994) Seed plants of the High Andes of Ecuador: a checklist. University of Aarhus, Aarhus
Kanowski JJ, Reis TM, Catterall CP, Piper SD (2006) Factors affecting the use of reforested sites by reptiles in cleared rainforest landscapes in tropical and subtropical Australia. Restor Ecol 14(1):67–76
Kumar S, Stohlgren TJ, Chong GW (2006) Spatial heterogeneity influences native and nonnative plant species richness. Ecology 87(12):3186–3199
Lambin EF, Meyfroidt P (2010) Land use transitions: socio-ecological feedback versus socio-economic change. Land Use Policy 27(2):108–118
Lauer W, Rafiqpoor MD (2000) Páramo de Papallacta: a physiogeographical map 1: 50,000 of the area around the Antisana (Eastern Cordillera of Ecuador). Erdkunde 54:20–33
Leite MdS, Tambosi LR, Romitelli I, Metzger JP (2013) Landscape ecology perspective in restoration projects for biodiversity conservation: a review. Brazilian Journal Nature Conservation 11(2):108–118
Lindenmayer DB, Wood JT, Cunningham RB, Crane M, Macgregor C, Michael D, Montague-Drake R (2009) Experimental evidence of the effects of a changed matrix on conserving biodiversity within patches of native forest in an industrial plantation landscape. Landscape Ecol 24(8):1091–1103
Lôbo D, Leão T, Melo FPL, Santos AMM, Tabarelli M (2011) Forest fragmentation drives Atlantic forest of northeastern Brazil to biotic homogenization. Divers Distrib 17(2):287–296
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton University Press, Princeton
MacLaren C, Buckley H, Hale R (2014) Conservation of forest biodiversity and ecosystem properties in a pastoral landscape of the Ecuadorian Andes. Agrofor Syst 88(2):369–381
McGarigal K, Cushman S, Ene E (2012) FRAGSTATS v4: Spatial pattern analysis program for categorical and continuous maps. University of Massachusetts, Amherst
Melo FPL, Arroyo-Rodríguez V, Fahrig L, Martínez-Ramos M, Tabarelli M (2013) On the hope for biodiversity-friendly tropical landscapes. Trends Ecol Evol 28(8):462–468
Mesquita RCG, Ickes K, Ganade G, Williamson GB (2001) Alternative successional pathways in the Amazon Basin. J Ecol 89(4):528–537
Metzger JP, Brancalio PHS (2013) Challenges and opportunities in applying a landscape ecology perspective in ecological restoration: a powerful approach to shape Neolandscapes. Brazilian Journal Nature Conservation 11(2):103–107
Meyfroidt P, Lambin EF (2011) Global forest transition: prospects for an end to deforestation. Annu Rev Environ Resour 36(1):343–371
Mishra R (1968) Ecology workbook. Oxford and IBH Company, New Delhi
Mladenoff DJ (2004) LANDIS and forest landscape models. Ecol Model 180(1):7–19
Molina A, Vanacker V, Balthazar V, Mora D, Govers G (2012) Complex land cover change, water and sediment yield in a degraded Andean environment. J Hydrol 472–473:25–35
Munro NT, Fischer J, Wood J, Lindenmayer DB (2009) Revegetation in agricultural areas: the development of structural complexity and floristic diversity. Ecol Appl 19(5):1197–1210
Newton AC, Echeverría C, Cantarello E, Bolados G (2011) Projecting impacts of human disturbances to inform conservation planning and management in a dryland forest landscape. Biol Conserv 144(7):1949–1960
Norden N, Chazdon RL, Chao A, Jiang Y-H, Vílchez-Alvarado B (2009) Resilience of tropical rain forests: tree community reassembly in secondary forests. Ecol Lett 12(5):385–394
Oksanen J, Kindt R, Legendre P, O’Hara B, Stevens MHH, Oksanen MJ, Suggests M (2007) The vegan package. Community ecology package 2013
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190(3–4):231–259
Posada JM, Aide TM, Cavelier J (2000) Cattle and weedy shrubs as restoration tools of tropical montane rainforest. Restor Ecol 8(4):370–379
R Development Core Team (2010) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Viena
Rapp JM, Silman MR, Clark JS, Girardin CAJ, Galiano D, Tito R (2012) Intra- and interspecific tree growth across a long altitudinal gradient in the Peruvian Andes. Ecology 93(9):2061–2072
Robiglio V, Sinclair F (2011) Maintaining the conservation value of shifting cultivation landscapes requires spatially explicit interventions. Environ Manag 48(2):289–306
Rodrigues RR, Lima RAF, Gandolfi S, Nave AG (2009) On the restoration of high diversity forests: 30 years of experience in the Brazilian Atlantic Forest. Biol Conserv 142(6):1242–1251
Rudel TK, Coomes OT, Moran M, Achard F, Angelsen A, Xu J, Lambin EF (2005) Forest transitions: towards a global understanding of land use change. Global Environ Change 15(1):23–31
Sarmiento FO (2002) Antrhopogenic change in th elandscapes of highland Ecuador. Geogr Rev 92(2):213–234
Scheller RM, Domingo JB, Sturtevant BR, Williams JS, Rudy A, Gustafson EJ, Mladenoff DJ (2007) Design, development, and application of LANDIS-II, a spatial landscape simulation model with flexible temporal and spatial resolution. Ecol Model 201(3–4):409–419
Sekhon JS (2011) Multivariate and propensity score matching software with automated balance optimization: the matching package for R. J Stat Softw 42(7):1–52
Steiner NC, Köhler W (2003) Effects of landscape patterns on species richness: a modelling approach. Agric Ecosyst Environ 98(1–3):353–361
Tabarelli M, Aguiar AV, Ribeiro MC, Metzger JP, Peres CA (2010) Prospects for biodiversity conservation in the Atlantic Forest: lessons from aging human-modified landscapes. Biol Conserv 143(10):2328–2340
Tabarelli M, Santos BA, Arroyo-Rodríguez Victor, Melo FPL (2012) Secondary forests as biodiversity repositories in human-modified landscapes: insights from the Neotropics. Boletim do Museu Paraense Emílio Goeldi: Ciências Naturais 7(3):319–328
Thompson JR, Foster DR, Scheller R, Kittredge D (2011) The influence of land use and climate change on forest biomass and composition in Massachusetts. USA. Ecol Appl 21(7):2425–2444
Tscharntke T, Tylianakis JM, Rand TA, Didham RK, Fahrig L, Batáry P, Bengtsson J, Clough Y, Crist TO, Dormann CF, Ewers RM, Fründ J, Holt RD, Holzschuh A, Klein AM, Kleijn D, Kremen C, Landis DA, Laurance W, Lindenmayer D, Scherber C, Sodhi N, Steffan-Dewenter I, Thies C, van der Putten WH, Westphal C (2012) Landscape moderation of biodiversity patterns and processes - eight hypotheses. Biol Rev 87(3):661–685
van Breugel M, Hall JS, Craven D, Bailon M, Hernandez A, Abbene M, van Breugel P (2013) Succession of ephemeral secondary forests and their limited role for the conservation of floristic diversity in a human-modified tropical landscape. PLoS ONE 8(12):e82433
Vanacker V, Balthazar V, Molina A (2013) Anthropogenic activity triggering landslides in densely populated mountain areas. In: Margottini C, Canuti P, Sassa K (eds) Landslide science and practice. Springer, Berlin, pp 163–167
Vetaas OR (1997) The effect of canopy disturbance on species richness in a central Himalayan oak forest. Plant Ecol 132(1):29–38
Wagner H, Wildi O, Ewald K (2000) Additive partitioning of plant species diversity in an agricultural mosaic landscape. Landscape Ecol 15(3):219–227
White S (2013) Grass páramo as hunter-gatherer landscape. The Holocene 23(6):898–915
Whittaker RH (1972) Evolution and measurement of species diversity. Taxon 21:213–251
Wirth C, Messier C, Bergeron Y, Frank D, Fankhänel A (2009) Old-growth forest definitions: a pragmatic view. In: Wirth C, Gleixner G, Heimann M (eds) Old-growth forests, ecological studies. Springer, Berlin, pp 11–33
Young KR (1993) Tropical timberlines: changes in forest structure and regeneration between two Peruvian timberline margins. Arctic Alpine Res 25(3):167–174
Zimmerman JK, Pascarella JB, Aide TM (2000) Barriers to forest regeneration in an abandoned pasture in Puerto Rico. Restor Ecol 8(4):350–360
Acknowledgments
This research was supported by the Belgian Federal Science Policy Programme BELSPO (Project Stereo II SR/00/133) and the Fonds National de la Recherche Scientifique (FNRS, Brussels, Belgium). We acknowledge the collaboration and hospitality offered by the communities living in the study area, in particular the family Paguay. We are grateful for the data provided by Jorge Caranqui from the Herbario Politecnico CHEP in Riobamba, Ecuador, for the contribution of Dr. Vincent Balthazar, and for the advices of Prof. Veerle Vanacker and Dr. Patrick Meyfroidt. We thank the Pontificia Universidad Católica del Ecuador and the Ecuadorian Ministerio del Ambiente for their collaboration. This manuscript was greatly improved thanks to constructive comments by two anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Middendorp, R.S., Pérez, A.J., Molina, A. et al. The potential to restore native woody plant richness and composition in a reforesting landscape: a modeling approach in the Ecuadorian Andes. Landscape Ecol 31, 1581–1599 (2016). https://doi.org/10.1007/s10980-016-0340-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-016-0340-7