Abstract
In the Neotropics, the effect of agriculture on freshwater habitats is still poorly understood, particularly with respect to sugarcane expansion to meet increased ethanol demands. The variation in taxonomic composition, community attributes and trophic structure of fish assemblages from streams under different riparian zone preservation conditions were studied. Nine stream reaches under the following riparian vegetation conditions were selected: Native Forest (NF), with mostly primary forest; Secondary Forest (SF), which included sites with vegetation in an advanced stage of regeneration surrounded by sugarcane plantations; and Sugarcane sites (SC), located in areas without riparian vegetation, adjacent to sugarcane crops. Forty-one species were collected. The variance partitioning and partial Redundancy Analysis (pRDA) indicated that community composition and trophic structure were significantly explained by riparian condition and spatial variations, while diversity, richness, abundance and biomass were only explained by season and space, with higher diversity and richness at the SF sites. Abundance-Biomass curves (ABC curves) suggested that sugarcane plantations destabilized fish assemblages, especially at the more intensively managed sites, which seem to influence stream integrity and associated fish fauna. We concluded that both physical conditions of streams and some attributes of fish communities reflected the different levels of preservation of the riparian buffers, but given the influence of regional processes which have a pervasive role in shaping local assemblages, land use at the watershed scale seemed to be important especially to explain the higher richness and diversity found at the SF sites.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The influence of land cover on the biological and physical conditions of streams has been intensively studied in temperate regions, indicating that when landscapes are converted from forests and wetlands to agricultural and urban areas, biological communities are affected in predictable ways (Stanfield and Kilgour 2013). Such changes often imply in the reduction of the riparian forests, which affect the functioning of stream ecosystems (Allan et al. 1997). Riparian vegetation acts on regulating aquatic food webs, providing material subsides through inputs of terrestrially derived food, and providing temperature, light and structural protection of habitats (Naiman and Décamps 1997; Pusey and Arthington 2003; Iwata et al. 2012). In addition, riparian zones help maintaining water quality by retaining sediments and contaminants from surrounding areas (Gregory et al. 1991; Wang et al. 1997; Harding et al. 1998; Dudgeon 2008) and maintain in-stream habitat heterogeneity, by regulating flow and discharge, being an important buffer against the impacts of deforestation (Naiman and Décamps 1997). Therefore, riparian vegetation has a multi-scale environmental influence on aquatic communities (e.g., fish and invertebrates) and their trophic dynamics (Gregory et al. 1991; Rabeni and Smale 1995; Allan and Castillo 2007; Lorion and Kennedy 2009) and has become an integral component of management strategies for stream biodiversity conservation (Naiman and Décamps 1997).
Several studies have found negative effects of deforestation on fish in tropical stream ecosystems (Bojsen and Barriga 2002; Wright and Flecker 2004; Casatti et al. 2009; Lorion and Kennedy 2009; Iwata et al. 2012), observing severe reduction in habitat quality, fish diversity, and changes in food resource availability. In agricultural regions, impacts are even more intense, affecting water quality, biodiversity, sedimentation and nutrient levels (Corbi et al. 2006; Riseng et al. 2011). Fewer insect species and intolerant fish species have also been recorded in agricultural areas (Wang et al. 1997; Genito et al. 2002), with observed changes in the diet of characids as reported by Ferreira et al. 2012a in agricultural regions of the Piracicaba River Basin.
Although agricultural expansion alone cannot explain the deforestation rates observed in the past, both processes have long been connected in Brazil (Gibbs et al. 2010). The biggest threat posed by expanding the amount of land under cultivation for energy, or any other use, is the irreversible conversion of virgin ecosystems, which may affect the hydrologic cycle and climate, reducing regional precipitation and increasing temperatures (Goldemberg et al. 2008). Sugarcane is among the economically most important crops in Brazil, with the largest area under cultivation in the world, accounting for approximately one third of global production (Zuubier and Vooren 2008). In the Southeastern region of the country, specifically in the state of São Paulo, the crop has expanded from ~18,000 km2 in 1990 to 52,000 km2 in 2011 (MAPPC 2012), occupying approximately 25 % of the state’s agricultural area. In several cases, riparian zones, considered priority areas for biodiversity conservation (Barletta et al. 2010; SMA 2011), are surrounded by sugarcane fields and recent surveys indicate a deficit of riparian vegetation cover in 1,400,000 hectares in the state, of which 300,000 ha are in sugarcane industry land (SMA 2011). The Piracicaba, Capivari and Jundiaí River Basins (PCJ Basins) have a long history of human occupation and environmental change, with 72.7 % of the basin conformed by pastures and sugarcane (CBH-PCJ 2011). Between 2004 and 2011 there was an increase of 258,682 ha of sugarcane crop area in these basins (IRRIGART 2007; CBH-PCJ 2011).
Land use change toward sugarcane production has raised a number of questions regarding its negative consequences and sustainability. According to Goldemberg et al. 2008, although there are several positive impacts, its negative aspects are related to the destruction or damage of high biodiversity areas, deforestation, soil damage and degradation, water resource contamination or depletion and diminished food security. Several studies have addressed the environmental sustainability issue of sugarcane cultivation with respect to aquatic systems, focusing on sedimentation (Fiorio et al. 2000), transport of contaminants like pesticides, heavy metals, and herbicides (atrazine) (Corbi et al. 2006), the origin of organic matter (Martinelli et al. 1999) and effects of land use on water chemistry and macroinvertebrates (Ometto et al. 2000). Bunn et al. 1997 and Ferreira et al. 2012b used multiple-stable isotope analysis to identify sources of organic carbon supporting food webs in aquatic systems in catchments with different riparian zones. The latter authors showed that in regions where the buffer zone was not covered by riparian forest, there was a significant increase in C4 carbon (sugarcane and pasture). Given the number and importance of the links between riparian and lotic ecosystems, spatial and temporal variation in fish assemblage composition and ecological attributes (i.e., species richness, dominance, abundance and biomass) have been linked to the variation in riparian cover (Godinho 2008).
Considering that the way in which species respond to anthropogenic disturbance is usually complex and strongly influenced by several ecological processes (Hayes et al. 1996), it is often difficult to predict how a particular aquatic-riparian ecosystem will change following a management activity (Bisson et al. 1996). Thus, the aims of this study were to investigate which environmental factors are important in determining the distribution of fish species in streams with riparian zones under different conditions of preservation in the Piracicaba River basin, and the amount of variation that can be assigned solely to the riparian condition or to other factors as season and spatial variability. We hypothesized that streams in sugarcane plantation areas would exhibit altered habitat conditions and fish communities compared with sites with preserved riparian buffers, and that these would support a more balanced community.
Materials and methods
Study area
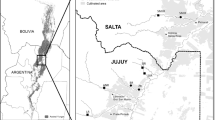
The Piracicaba River Basin spans 12,568 km2, 90.7 % of which are in the State of São Paulo (CBH-PCJ 2010), forming the Piracicaba-Jundiaí-Capivari Basins (PCJ basins). This area contains scattered fragments of Atlantic Forest, in an interface with Seasonal Semideciduous Forests of the Plateau (PCJ 2012). The climate is subtropical, with average annual temperatures between 18 and 22 °C and rainfall of 1,405 mm (Caram 2010; CBH-PCJ 2011). The wet season is between October and March, and the dry season from April to September (Ometto et al. 2000). This basin is highly industrialized (CETESB 2010) and the most populous in São Paulo state, with about five million inhabitants. The basin is mainly occupied by pasture (45 %), agriculture (31 %), natural forest and reforestation (16 %) and urban areas (6 %) (Filoso et al. 2003).
The present study focused on two sub-basins of the Piracicaba River Basin, the Atibaia (2,868 km2) and Jaguari River Basins (3,290 km2) (CBH-PCJ 2010). The Atibaia River Basin has widespread eutrophication, is predominantly agricultural land (50.52 %) (Piacente 2005), with 16.4 % native vegetation (IRRIGART 2006). The Jaguari River Basin is intensively deforested due to urbanization and expansion of industrial and agricultural activities (72.4 %) retaining only 17.7 % of its natural vegetation (Whately and Cunha 2007).
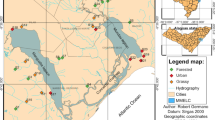
Nine stream reaches were selected based on field observations of the condition of the riparian buffer zones. The width of this zone and the degree of complexity of the forest canopy (i.e., presence of multi-strata forest vegetation) were used to classify them into three categories (Native Forest, Secondary Forest and Sugarcane). The absence of point sources of pollution, similar dimensions and location within the same basin were also considered. The stream reaches were gentle-gradient riffle/pool types (Rosgen 1994) and varied from 1st to 3rd order. Each riparian condition had similar numbers of sites in each order. Percent land cover classes for each watershed and a relief model with a 15 m spatial resolution were obtained using the Geographic Information System (ArcInfo 9.3) based on the digital cartographic databases (Brazilian Institute of Geography and Statistics - IBGE) scale 1:50,000. The direct contribution area was delimited for each watershed based on topography, contour, and hydrographic layers. The land use types included native forests, secondary forests, sugarcane, and the category “others” (pasture, bare soils and perennial crops). Native forest comprised dense ombrophilous forests and lowland vegetation, while secondary forests comprised semi-deciduous and secondary dense ombrophilous vegetation.
Three stream reaches were selected under each of the following riparian conditions (Fig. 1): primary forest buffers within forested areas (Native Forest - NF); riparian vegetation consisting of secondary forest, with sugarcane fields in adjacent areas (Secondary Forest - SF); sparse or absent riparian vegetation, with surrounding sugarcane fields (Sugarcane - SC). Native Forest sites presented dense woody vegetation on both banks, with buffer widths of 12–18 m. The catchment was composed by both secondary and primary forest, and absence of sugarcane fields (Table 1). Secondary forest reaches were located within sugarcane properties in Cosmópolis (SP) showing clear signs of deforestation with a relatively open understory and 6–12 m of riparian buffer zones. This forest type results from the natural regeneration of vegetation in areas where primary forest was cut down. In such cases, land was almost always used temporarily for agriculture or pasture and forest emerged spontaneously after these activities were abandoned. In these zones, vegetation was dominated by sparse scrub, several pioneer species, grasses, bamboos, the castor-oil plant (Ricinus communis) and Cecropia sp. Sugarcane was abundant in the catchment (35–63 %), followed by a mix of primary native and secondary forests (Table 1).
In Sugarcane sites, riparian vegetation consisted primarily of Brachiaria type grasses. Sugarcane fields were sparse and riparian vegetation was composed of shrubs in the Açude Stream (AÇ). Severe erosion, with the presence of gullies, was also observed along this reach. The other two reaches (SF 1 and SF 2), were subject to different sugarcane management activities. During the study period, the plantation ranged between ratoon cane and reform classes, i.e., the stage at which sugarcane has gone through more than one cut and the stage when natural productivity decreases indicating the need for renovation, respectively (CANASAT 2011; SMA 2011). In the dry season, sugarcane fields next to sample reaches were harvested, while in the rainy season they were in more advanced stages of development. Sugarcane was dominant at the catchment scale, attaining ≈ 90 % of land cover (Table 1).
All sample reaches were located in the Jaguari River Basin, except for SF 1 and SF 2 sites, which were located in the Atibaia River Basin.
Environmental data and fish sampling
The following physical and chemical parameters were recorded at each site with a HORIBA U-22 multiparameter analyzer during dry (August-September/2011) and rainy (March and December/2011) seasons: temperature (°C), turbidity (NTU), dissolved oxygen (mg.L-1), pH, conductivity (μS.cm-1), and total dissolved solids (mg.L-1). Water samples were also collected and kept on ice for laboratory analysis of nitrate (μg.L-1), nitrite (μg.L-1), ammonium (μg.L-1), total nitrogen (μg.L-1), orthophosphate (μg.L-1), total phosphorus (μg.L-1), and total suspended solids (mg.L-1) (APHA 2005).
In-stream variables were recorded during site surveys within the 50-m long reaches where fish were sampled. These variables included measures of width (m), depth (m) and water velocity (m.s-1) (Marques and Argento 1988). Flow rate (m3.s-1) was measured with a General Oceanics flow meter (2031H/2135) and calculations made according to Santos et al. 2001. The proportion of canopy cover, riffles, pools, trunks and roots (%) and substrate categories were estimated visually at each stream reach. Substrate was coarsely categorized as sand (<3 mm); fine gravel (≅3–49 mm), coarse gravel (≅50–149 mm); small boulders (≅150–300 mm) and large boulders (>300 mm).
A rapid assessment protocol (Habitat Quality Index - HQI) was used to quantify the habitat disturbance level of the sampling sites, based on the methodology proposed by Callisto et al. 2002. Site characteristics were evaluated through different sets of attributes including the type of occupation of the water body, margins and/or erosion on river banks and siltation, anthropogenic changes, vegetation cover, odor, oil, water and substrate quality. The sum of scores gives the final index ranging from 0 to 100, where 0–40 means “impacted”, 41–60 “moderately impacted” and > 61 is “natural” habitat.
Fish samples were collected during the dry and rainy seasons using electrofishing equipment (Honda Generator - EU10i 1,000 W, 120 V, 60 Hz). Three successive removals in 50 m stretches were performed at each site, following the 3-catch removal method (Zippin 1958), resulting in a constant fishing effort (~40 min for each removal) at each locality. The reach was set at this length in order to maintain the uniformity of the riparian conditions along the sampling site, avoiding other anthropogenic influences as channelization or dredging. This length is similar to the one used by Teixeira-de Mello et al. 2014 to evaluate the efficiency of electrofishing for assessing richness, density and size distribution in small streams located in different climatic and biogeographical regions, including neotropical streams. Three passes have also proved to be adequate for obtaining quantitative data in Atlantic Forest streams (Mazzoni et al. 2000), while Humpl and Lusk 2006 have shown that data from two-pass sampling is more precise than one pass only for detecting fish community structure in streams.
Specimens were fixed in 10 % formalin, transferred to 70 % alcohol, identified according to Britski 1972, measured and weighed in the laboratory. Identification was confirmed at the Museum of Zoology, University of São Paulo, Brazil, where voucher specimens were deposited (MZUSP 110954 -110972).
Data analysis
As an initial approach, we examined the species-environment relationships using a Canonical Correspondence Analysis (CCA) on the software CANOCO 4.5 (ter Braak and Šmilauer P 2002), to identify patterns between assemblages. Step-wise forward selection was used to reduce the environmental variables to those most correlated with the axes (ter Braak 1987). A cutoff point of p = 0.05 was used to incorporate variables into the final models. The significance of each variable for each axis was determined using the null matrices obtained by the Monte Carlo permutation test (Hope 1968) considering 9999 permutations and a significance level of p = 0.05. Data in percentages were square root transformed, while other values were transformed to log (x + 1). Shannon-Wiener Diversity (H′), Simpson Dominance (λ′) (Krebs 1989) and Margalef Richness (d) (Ludwig and Reynolds 1988) were calculated for each site and season using PRIMER 6 software (Clarke and Gorley 2006).
By means of a pRDA we run a variance partitioning analysis (Borcard et al. 1992; Peres-Neto et al. 2006) to understand the influences of riparian condition (Sugarcane, Secondary Forest, Native Forest), season (Dry and Rainy) and space (distance among sampled sites) on abiotic variables and community descriptors. Partial RDA considers the idea that some species and environmental variables may share a common spatial structuring, and is based on pre-existing methods of canonical ordination [RDA (van den Wollenberg 1977) and CCA (ter Braak 1986)]. Recently, variance partitioning has become popular among community ecologists to disentangle the influences of multiple sets of predictors on community structure (Dray et al. 2012) measuring the fractions assigned to pure and shared effects.
The abiotic variables were represented by a matrix of physical, chemical and in-stream structures, while fish assemblages were described by three matrices of: fish composition (species-site matrix), diversity/abundance (Margalef richness, Shannon-Wiener diversity, Simpson dominance, biomass – g/m2, and density – individuals/m2), and trophic composition (densities - g/m2 of omnivores, detritivores, carnivores and insectivores). The classification of trophic groups was based on several studies (Casatti and Castro 1998; Gibran et al. 2001; Esteves and Alexandre 2011; Castro et al. 2004; Ferreira 2007; Braga and Gomiero 2009; Costa and Schulz 2010). The Hellinger transformation of species abundance and trophic composition matrices was applied to the data previously to the pRDA analysis. From IBGE maps (scale 1:50,000) rivers channels were vectorized using AutoCAD 2007, in order to measure geographical distances among sampling sites. Then, a principal coordinate analysis (PCOA) was applied to pairwise-distance matrix among sites and eigenvectors used to describe the spatial configuration of sampled sites. Variance partitioning analyses, pRDA and PCOA were done in R software (R Development Core Team 2014). Permutation procedures were used (1,000 permutations) to test the significance of riparian condition, space and season considering the significance level at 0.05.
Stress levels of fish communities based on species abundance and biomass at each sampling site were compared using Abundance and Biomass Curves (ABC Curves) (Warwick 1986) in PRIMER 6 (Clarke and Gorley 2006). The difference between the two curves is given by the W statistic that ranges from -1 to 1, where a negative sign indicates that the biomass curve lies below the abundance curve and suggests a disturbed community (Clarke and Warwick 2001).
Results
Abiotic stream characteristics
Native Forest sites presented the highest values of conductivity, ammonium, nitrite, nitrate, orthophosphate, total nitrogen, canopy cover, percentage of pools and HQI, while SC sites showed the highest values of total phosphorus and lowest canopy cover (Table 2). The best habitat conditions (HQI) were recorded in the rainy season when NF and SF sites were considered ‘natural’ (>61). Sugarcane sites ranged from ‘impacted’ (0–40) to ‘altered’ (41–60), except in the Açude stream (AÇ) where the habitat was classified as ‘natural’ in the dry season and altered in the rainy season. The mean monthly temperature and rainfall in the study area ranged from 16.5 to 25.8 °C and from 2.9 mm to 500.3 mm in the dry and rainy seasons, respectively (CIIAGRO 2012). The variance partitioning analysis related to the abiotic variables showed that shared effects among riparian condition, space and season were not significant, with only pure effects being observed. These variables were significantly influenced by riparian condition (r2 adj = 0.19, F = 3.64, p = 0.002), seasonality (r2 adj = 0.13, F = 4.37, p = 0.001) and space (r2 adj = 0.16, F = 3.32, p = 0.001) (Fig. 2a). Shared effects were in general not important except for a small fraction of 8 % between riparian condition and space. An overlap between sugarcane and native forested sites along the first pRDA axis was observed. This axis was predominantly influenced by flow, total suspended solids and water velocity on the negative extremity. The SF sites differed from the other riparian conditions along the second pRDA axis mainly due to its wider and deeper channels and higher flow (Fig. 3a).
Biplot of Partial Redundancy Analysis (pRDA) showing the influence of riparian conditions on abiotic variables (a), species abundance (b), diversity/abundance measures (c) and trophic composition (d). The biplots show only pure effects associated with riparian condition, excluding the effects of space and season. See species acronyms in Table 3
Fish assemblages
A total of 1,325 individuals of 41 fish species were collected. Bryconamericus iheringii and Astyanax sp. were the most abundant species in NF sites. Gymnotus carapo, Astyanax paranae, Astyanax altiparanae and Imparfinis mirini were numerically dominant in the SF sites, while Phalloceros spp. was the most abundant species in sugarcane reaches. Twenty-two species occurred exclusively in one of the three different riparian conditions, with seven occurring only in NF; 11 in SF; and four in SC reaches (Table 3). The percentage of rare species for each riparian condition (i.e., with ≤ five individuals) was higher at the SF (68.9 %) and SC (55.6 %), followed by NF sites (47.4 %).
The first two axes in the CCA explained 48.9 % of variation in the data, most of which (26.8 %) was explained by axis 1 (Fig. 4). Orthophosphate (p = 0.0002), riffles (p = 0.001) and depth (p = 0.003) explained most of the variance in this analysis. Axis 2 explained 22.1 % of data variation, with higher contribution of nitrate (p = 0.0005) and stream width (p = 0.005). Species such as Characidium zebra and Apareiodon affinis, were associated with deep sites with a high proportion of riffles, while Gymnotus carapo, and Hyphessobrycon eques, among others, showed preference for slow flowing habitats, such as the Tributary of Macaco Branco (NF), Jacutinga (SF) and São Francisco streams (SC). The benthic catfish Imparfinis mirini, occurred both in SF sites and the Açude stream (SC), which differed from other SC sites because of the low ortophosphate levels and higher proportion of pebbles, to which Corydoras aeneus, Hypostomus ancistroides, and Gymnotus pantherinus were associated.
Representation of the first two axes of the Canonical Correspondence Analysis (CCA) for fish communities from nine stream sites from the Jaguari and Atibaia River Basins. Dry season (open symbols) and rainy season (filled symbols): ■ Native Forest; ● Secondary Forest; ▲ Sugarcane. Vector lines indicate the relationship of the significant environmental variables to the ordination axis; line length is proportional to its relative significance. Ort Orthophosphate, Nitra Nitrate, Wid Width, Ri Riffles, Peb Pebbles and Dep Depth. See species acronyms in Table 3
Community composition was significantly influenced by riparian condition (r2 adj = 0.29, F = 4.78, p = 0.001) and space (r2 adj = 0.34, F = 5.30, F = 0.001), but not by seasonality (r2 adj ≈ 0, F = 0.43, p = 0.924) (Fig. 2b). The different riparian conditions showed little overlap, suggesting that assemblage composition was a good descriptor of riparian conditions under different preservation conditions. The first axis of pRDA separated the NF sites with predominance of Geophagus brasiliensis from SC sites with predominance of Hypostomus ancistroides and Gymnotus pantherinus. SF sites grouped on the positive extreme of the second pRDA axis and were described mainly by the predominance of Gymnotus carapo and Astyanax altiparanae (Fig. 3b).
The diversity/abundance matrix was significantly influenced by seasonality (r2 adj = 0.15, F = 3.89, p = 0.022) and space (r2 adj = 0.20, F = 3.17, p = 0.034), but not by riparian condition (r2 adj = 0.13, F = 2.36, p = 0.077) (Fig. 2c). This weak influence of riparian condition reflected on a great overlap among sites on the pRDA. Nevertheless, SF sites were associated to higher richness and Shannon diversity as shown on the negative extreme of the first pRDA axis (Fig. 3c).
As for assemblage composition, trophic structure of fish communities was influenced only by riparian condition (r2 adj = 0.44, F = 5.75, p = 0.011) and space (r2 adj = 0.35, F = 4.77, p = 0.013), but not by seasonal variations (r2 adj ≈ 0, F = 0.76, p = 0.407) (Fig. 2d). The first axis of pRDA described a gradient of sites with predominance of omnivores to sites with predominance of insectivores, which were related to SF sites. Despite the statistical significance, there was great overlap among sites with different type riparian conditions (Fig. 3c) suggesting that trophic structure may be a weak predictor of this kind of modification.
The results of the ABC curves and their corresponding W values (Fig. 5), indicated that the NF and SF sites had preserved and moderately impacted communities (W ranging from -0.141 to 0.23), while most impacted conditions were observed in the SC sites (W ranging from -0.44 to 0.18).
ABC Curves based on abundance (□) and biomass (●) of fish sampled at the nine stream reaches from the Jaguari and Atibaia River Basins in the dry season. Streams: A Macaco Branco, B Tributary of Macaco Branco (Native Forest), C Ponte Funda, D Canguçu, E Jacutinga (Secondary Forest), F São Francisco 1, G Açude (Sugarcane). The Josias stream (Native Forest) and São Francisco 2 (Sugarcane) were not included because of single species dominance
Discussion
Abiotic variables
Although water quality met the guidelines proposed by the Brazilian regulatory arm of the Ministry of Environment (CONAMA 2005) for aquatic life preservation (class 02), physicochemical variables were different for the three riparian vegetation types. Native Forest sites had higher conductivity, total dissolved solids and nitrogen compounds, which can be related to litter accumulation and decomposition on the stream bed (Esteves 1998). According to Nislow 2005, vegetation type may influence nutrient retention processes, with older forests retaining naturally fewer nutrients than younger forests, and releasing them into the river. Similar results were found by Burcham 1988; Lorion and Kennedy 2009, and Riseng et al. 2011, who reported higher conductivity in dense forested sites. Nevertheless, these values are low when compared to other studies in the Piracicaba River Basin, which reported conductivity between 130 to 272.8 μS.cm -1 (Ometto et al. 2000; Esteves and Alexandre 2011).
Native Forest stream reaches were structurally more complex, as reflected by a higher HQI, and proportion of pools and trunks. Forests formed by older vegetation are more vulnerable to being blown down, increasing the supply of large woody debris (LWD) into the channel, as well as the frequency of pools (Nislow 2005). This can significantly increase nutrient and material retention in the stream by creating transient storage zones, thus increasing habitat and flow path complexity (Bilby and Likens 1980). The LWD can also greatly impact fish habitat by increasing pool habitat area and pool frequency, which is important for survival of aquatic biota, supporting a more diverse and structured fish community (Casatti et al. 2006b).
The variance partitioning analysis of abiotic characteristics showed that the effect of riparian condition that is spatially structured accounted for only 8 % of total variation. This effect is probably due to nearby sites that also have similar riparian conditions. Nevertheless, this shared effect was smaller than those assigned solely to riparian conditions (19 %), space (16 %) or season (18 %). Residual variation accounted for 49 % and this may be attributed to other non-measured underlying factors. In fact, we did not quantify other substances as agrochemicals heavily used during the production of sugarcane that can potentially be harmful to the environment (Smeets et al. 2006) nor the influence of larger scale factors at the watershed scale. The search for other environmental factors (metals and organochlorinated compounds) as those reported by Corbi et al. 2006 in streams located in sugarcane areas could thus be considered in other studies, increasing the explanation of the remaining variation.
Species-environment relationships
The number of species observed in this study (41) was high when compared to those found by Gerhard 2005, who conducted a survey in 60 watersheds in the Corumbataí River, also in the Piracicaba River Basin. This author used electrofishing in low order streams and found 51 species, whereas Esteves and Alexandre 2011, with a comparable sampling effort to our study recorded 35 species in the Guamium River (Piracicaba River Basin). Results indicated the predominance of Bryconamericus iheringii in NF sites; this appears to be a species characteristic of more pristine environments (Hued and Bistoni 2005). Individuals of B. iheringii are active swimmers, which dwell in flowing environments, and mostly prey on items carried by the current (Ferreira et al. 2012a), which may explain their presence in more heterogeneous environments. Phalloceros spp. is an opportunistic species that occupies disturbed habitats (Casatti et al. 2008; Araújo et al. 2009) such as the SC sites where it was most abundant. Several species were associated to specific microhabitats as shown by the CCA. Species of the Parodontidae (e.g., Apareiodon affinis) and Crenuchidae (e.g., Characidium zebra) families occurred in microhabitats with higher proportion of riffles, as observed in the SF and NF sites. Most species in these families inhabit fast flowing waters and stay close to the bottom where they scrape the substrate in search of food (Marceniuk and Hilsdorf 2010). Gymnotus carapo occurred in shallow waters characteristic of SF reaches, particularly in the Jacutinga stream, where grasses were abundant on the river banks. This species appears to prefer river banks and backwaters where submerged marginal vegetation is abundant (Ferreira and Casatti 2006; Oyakawa et al. 2006), which may be related to their ability to tolerate and explore structurally simplified streams (Casatti et al. 2006a). The low HQI, high TSS and high turbidity recorded at the SF and SC sites, indicated that these sites presented altered environmental conditions, with visible effects of erosion. Different species associations were observed at these sites, as well as in the São Francisco stream, where Hyphessobrycon eques occurred in shallow flowing waters. This species is known to prefer standing waters with abundant macrophytes and is frequently found in disturbed environments (Oyakawa et al. 2006; Marceniuk and Hilsdorf 2010). In fact, H. eques occurred exclusively in stretches where floating pennywort Hydrocotyle ranunculoides abundance was high. This is an invasive macrophyte from the Araliaceae that is currently spreading in waterways; it is highly adaptable to a wide range of environmental conditions, reducing the level of dissolved oxygen (Bonants and Edema 2012) and disrupting natural erosion-deposition processes (DEFRA 2010). Hypostomus ancistroides, Corydoras aeneus, Gymnotus pantherinus and I. mirini occurred in the Açude stream, a SC site with a higher HQI, despite heavy siltation. Both H. ancistroides and I. mirini have been associated with higher siltation related to the removal of riparian vegetation (Casatti et al. 2005; Furlan et al. 2012) and urbanized streams (Tófoli et al. 2013), indicating their adaptability to altered conditions.
Factors influencing fish assemblages
Comparisons among our three riparian conditions suggested that this component had significant effects on some aspects of the fish assemblages, which included species composition and trophic structure, while richness, diversity, biomass and total density were not influenced by riparian condition, but only by seasonality and space. As for the abiotic stream conditions, part of the variability could not be explained by our data set, a situation commonly found in other studies (Borcard et al. 1992; Alexandre et al. 2010; Ferreira et al. 2014). High unexplained variances are not unusual in metacommunity studies (Melo et al. 2011), and although the explained data sets may seem proportionally low, the underlying causes found to be significant can nevertheless be considered as important in the structuring of these communities as stressed by Borcard et al. 1992. This analysis is further complicated by the fact that intrinsic and extrinsic local processes (e.g., competition, predation and disturbance) have been considered important to explain local fish assemblage structure, as well as the role of regional processes which have a pervasive role in shaping local assemblages (Schlosser 1995). More recently, several temperate region studies have shown that the variability in results from studies of landscape influences on streams can be attributed to confounding effects within the predictor data sets, regional variation in responses, catchment size-dependent responses and use of different types of metrics and measurement approaches (Stanfield and Kilgour 2013). However, studies on the effects of riparian vegetation on aquatic communities have stressed the importance of local influence, because riparian buffers help the preservation of mesohabitats and other in-stream features (Birnbaum et al. 2007; Lorion and Kennedy 2009; Casatti et al. 2012).
Several studies have shown positive correlations between nutrient levels and fish biomass and abundance (Murphy and Meehan 1991; Nislow 2005). Fish abundance can change in agricultural regions (Lammert and Allan 1999), deforested sites (Bojsen and Barriga 2002), and in streams with different intensities of physical habitat degradation and water chemistry (Casatti et al. 2006b). Although some studies have observed a decrease in fish biomass in streams within agricultural areas (Karr et al. 1985) and habitats with impaired environmental conditions (Casatti et al. 2006a), Bojsen and Barriga 2002 and Lorion and Kennedy 2009 found that biomass did not differ between sites with different riparian conditions. Our results agree with the latter findings and may be attributed to the influence of other factors such as space, which might be a limiting factor for total fish biomass (Bojsen and Barriga 2002). A balance between the disappearance of intolerant species at the SC sites, and an increase of tolerant species such as the r strategist Phalloceros spp. that may benefit from the larger amounts of food (periphyton and macroinvertebrates) in these altered streams may also explain the obtained results.
Richness and diversity measures, found to be higher at the SF sites could also not be explained by the influence of the riparian conditions, but only by spatial and seasonal factors. Although SF sites were apparently at an advanced stage of riparian vegetation regeneration, at the catchment scale, land use patterns presented relatively high levels of sugarcane cultivation at these sites, which may be considered a disturbance. Considering that when environmental disturbances are frequent, communities are dominated by opportunistic species, with low species diversity and when disturbances are rare, diversity is lower due to resource competition, one may suppose that in environments with intermediate levels of disturbance, diversity is high due to the presence of both opportunistic and intolerant species (Begon and Mortimer 1986), as predicted by the Intermediate Disturbance Hypothesis (Connell 1978). Wang et al. 1997 have shown that agriculture has a small influence on habitat quality or biotic integrity in watersheds with less than 50 % agriculture, suggesting that there may be a threshold level at which agricultural effects begin to manifest themselves.
Results also indicated an important influence of both space and riparian conditions on the trophic structure of the fish communities, with SF sites presenting higher values of insectivore species. Other guilds as omnivores and carnivores were similar between riparian conditions, while detritivores, especially Phalloceros spp. were more abundant at SC sites. Zeni and Casatti 2014 found greater availability of aquatic insects in homogenous streams of the northwest region of the state of São Paulo, and this was associated with the presence of submerged plants such as grasses and the opportunistic behavior of fish. Wichert and Rapport 1998 indicate that a higher proportion of insectivores and piscivores is expected in less disturbed systems than in heavily degraded sites, because as degradation intensifies, those species at the top of the trophic structure are expected to disappear first. In fact, SF sites showed lower canopy cover, which may have favored the development of grasses, to which the insectivorous G. carapo was highly associated.
In our analysis of the community attributes, it also has to be considered that conditions within the different riparian areas, especially sugarcane reaches, were not homogeneous with respect to impairment level and intensity of management activity. Unlike the Açude stream, where sugarcane plantations were not intensively managed, two sites (SF-1 and SF-2) were subject to intense management activities where heavy machinery exposed bare soils during harvest. Thus communities and physical conditions may have responded with different intensities to the local land use, suggesting that future sampling schemes could be conducted in order to understand the influence of different sugarcane management activities or site abandonment on stream communities.
The ABC curves reflected the effects of the different riparian conditions on fish assemblages, indicating that SC sites were subject to stress according to Warwick 1986 classification. From an ecological perspective, the interpretation of these plots is based on the r-K selection gradient, where species with long development times and large size are represented by few individuals, but are dominant in terms of biomass. In this case, the biomass curve lies above the abundance curve (Clarke and Warwick 2001; Clarke and Gorley 2006). According to these authors, in moderately affected sites, K strategist species are partially eliminated, reducing inequality between biomass and abundance, so that the curves are close and may even cross. Impacted sites favor the presence of small, r-strategist species, which become numerically dominant, in which case the abundance curve lies above the biomass curve. Forested (NF, SF) sites showed a dominance of small Characidae like B. iheringii (up to 11.4 cm total length), several Astyanax species (5.0–11.5 cm total length) and the Gymnotiform G. carapo (up to 76 cm total length). Their occurrence in these habitats may be related to the increased heterogeneity of these reaches, where pools, riffles, trunks and grasses may have provided spatial refuge not just from predation but also for the metabolic demands of maintaining position in flowing environments. The shape of ABC curves is influenced by the presence of large bodied species, which in these sites consisted of a large sedentary piscivore (H. malabaricus), the armored catfish H. ancistroides and the electric knifefish G. carapo. Despite the abundance of the siluriform I. mirini (up to 9.3 cm total length) in one SC site (AÇ), the dominance of the small viviparous Phalloceros spp. (up to 3 cm total length) seemed to account for the shifts in the ABC curves, indicating that agricultural disturbances were major stressors for these streams. Similar results were found for disturbed streams in northeastern São Paulo, where the exotic viviparous Poecilia reticulata was characteristic of reaches without canopy cover, with severe siltation, and mesohabitat simplification (Casatti et al. 2006b). Some authors consider the results of ABC curves may be biased by an influx of recruits of dominant species (Yemane et al. 2005), as well as the migration of large bodied species. However, this seems unlikely in this case, as most of the dominant species in the NF and SF sites were small sized Characidae, known to perform only short-distance reproductive migrations (Vazzoler 1996), while no typical large seasonal species occurred at the studied reaches.
Although there is some controversy regarding the relative influence of land use and land cover at the different scales, with some studies favoring the basin level (Wang et al. 1997; Harding et al. 1998) and others favoring the local scale (Stauffer et al. 2000; Birnbaum et al. 2007), this study showed that both physical conditions of streams and some attributes of fish communities reflected the different levels of preservation of the riparian buffers. These findings have important implications for the conservation of fish biodiversity and watershed management practices, especially when considering that only 7.9 % of native forest remains in the PCJ basin (CBH-PCJ 2011). Considering that only 5 % of the sugarcane areas in the State of São Paulo are of riparian vegetation (SMA 2011), a target to be met is natural vegetation restoration along streams on sugarcane fields, with strips of 30 m for rivers <10 m wide being proposed by the Brazilian Forest Code (n° 12.651, May 2012). Therefore, proper planning, improvement of land use practices, and protection of streams and riparian ecosystems are important issues to consider regarding environmental sustainability of sugarcane production as it was shown that fish assemblages were disrupted in sugarcane sites, probably as a consequence of low environmental integrity.
References
Alexandre CV, Esteves KE, De Moura e Mello MAM (2010) Analysis of fish communities along a rural–urban gradient in a neotropical stream (Piracicaba River Basin, São Paulo, Brazil). Hydrobiologia 64:97–114
Allan JD, Castillo MM (2007) Stream ecology: Structure and function of running waters. Kluwer Academic Publishers, Dordrecht
Allan JD, Erickson DL, Fay J (1997) The influence of catchment land use on stream integrity across multiple spatial scales. Freshw Biol 37:149–161
APHA (2005) Standard methods for the examination of water and freshwater. American Public Health Association, Washington
Araújo FG, Peixoto MG, Pinto BCT, Teixeira TP (2009) Distribution of guppies Poecilia reticulata (Peters, 1860) and Phalloceros caudimaculatus (Hensel, 1868) along a polluted stretch of the Paraíba do Sul River, Brazil. Braz J Biol 69:4148
Barletta M, Jaureguizar AJ, Baigun C et al (2010) Fish and aquatic habitat conservation in South America: a continental overview with emphasis on neotropical systems. J Fish Biol 76:2118–2176
Begon M, Mortimer M (1986) Population ecology, a unified study of animals and plants. Blackwell Scientific Publications, London
Bilby RE, Likens GE (1980) Importance of organic debris dams in the structure and function of stream ecosystems. Ecology 61:1107–1113
Birnbaum JSK, Winemiller O, Shen L et al (2007) Associations of watershed vegetation and environmental variables with fish and crayfish assemblages in headwater streams of the Pedernales River, Texas. River Res Appl 996:979–996
Bisson PA, Raphael MG, Foster AD, Jones LLC (1996) Influence of site and landscape features on vertebrate assemblages in small streams. Proc Wood Compat Initiative Workshop 6:61–72
Bojsen BH, Barriga R (2002) Effects of deforestation on fish community structure in Ecuadorian Amazon streams. Freshw Biol 47:2246–2260
Bonants P, Edema M (2012) Q-bank – Comprehensive databases on quarantine plant pests and diseases. http://www.q-bank.eu/. Accessed 22 November 2013
Borcard D, Legendre P, Drapeau PD (1992) Partialling out the spatial component of ecological variation. Ecology 73:1045–1055
Braga FMS, Gomiero LM (2009) Alimentação de peixes na microbacia do Ribeirão Grande, Serra da Mantiqueira oriental, SP. Biota Neotrop 9:207–212
Britski HA (1972) Peixes de água doce do Estado de São Paulo – Sistemática. In: Comissão interestadual da bacia Paraná- Uruguay. Poluição e Piscicultura, notas sobre ictiologia, poluição e piscicultura. FSPUSP e Instituto de Pesca, São Paulo, pp 79–108
Bunn SE, Davies PM, Kellaway DM (1997) Contributions of sugar cane and invasive pasture grass to the aquatic food web of a tropical lowland stream. Mar Freshw Res 48:173–179
Burcham J (1988) Fish communities and environmental characteristics of two lowland streams in Costa Rica. Rev Biol Trop 36:273–285
Callisto M, Ferreira WR, Moreno P, Goulart M, Petrucio M (2002) Aplicação de um protocolo de avaliação rápida da diversidade de habitats em atividades de ensino e pesquisa (MG-RJ). Acta Limnol Bras 14:91–98
CANASAT (2011) Monitoramento da Cana-de-açúcar via imagens de satélite. http://www.dsr.inpe.br/laf/canasat/. Accessed 05 January 2012
Caram RO (2010) Mudanças no uso e cobertura do solo e resposta hidrológica da bacia do rio Piracicaba. Dissertation, University of São Paulo
Casatti L, Castro RMC (1998) A fish community of the São Francisco River headwaters riffles, Southeastern Brazil. Ichthyol Explor Fres 9:229–242
Casatti L, Ferreira CP, Carvalho FR (2009) Grass-dominated stream sites exhibit low fish species diversity and dominance by guppies: an assessment of two tropical pasture river basins. Hydrobiologia 632:273–283
Casatti L, Ferreira CP, Langeani F (2008) A fish-based biotic integrity index for the assessment of lowland streams in Southeastern Brazil. Hydrobiologia 623:173–189
Casatti L, Langeani F, Ferreira CP (2006a) Effects of physical habitat degradation on the stream fish assemblage structure in a pasture region. Environ Manag 38:974–982
Casatti L, Langeani F, Silva AM, Castro RMC (2006b) Stream fish, water and habitat quality in a pasture dominated basin, Southeastern Brazil. Braz J Biol 66:681–696
Casatti L, Rocha FC, Pereira DC (2005) Habitat use by two species of Hypostomus (Pisces, Loricariidae) in Southeastern Brazilian streams. Biota Neotrop 5:1–9
Casatti L, Teresa FB, Gonçalves-Souza T et al (2012) From forests to cattail: how does the riparian zone influence stream fish? Neotrop Ichthyol 10:205–214
Castro RMC, Casatti L, Santos HF et al (2004) Estrutura e composição da ictiofauna de riachos da bacia do Rio Grande no Estado de São Paulo, Sudeste do Brasil. Biota Neotrop 4:57–95
CBH-PCJ Comitê das bacias hidrográficas dos rios Piracicaba, Capivari e Jundiaí (2010) Relatório da situação dos recursos hídricos da UGRHI 5. http://www.comitepcj.sp.gov.br/download/RS/PCJ_RS-2010_RelatorioFinal.pdf. Accessed 05 February 2012
CBH-PCJ Comitê das bacias hidrográficas dos rios Piracicaba, Capivari e Jundiaí (2011) Relatório da situação dos recursos hídricos da UGRHI 5. http://www.comitepcj.sp.gov.br/download/RS/PCJ_RS-2011_RelatorioFinal.pdf . Accessed 18 March 2012
CETESB Companhia de Tecnologia de Saneamento Ambiental (2010) Qualidade das águas superficiais no Estado de São Paulo. Série Relatórios, São Paulo
CIIAGRO Centro Integrado de Informações Agrometeorológicas (2012). http://www.ciiagro.sp.gov.br. Accessed 10 February 2012
Clarke KR, Gorley RN (2006) Software Primer v6 user Manual/Tutorial. Primer-E, Plymouth
Clarke KR, Warwick RM (2001) Change in marine communities: An approach to statistical analysis and interpretation. Primer-E, Plymouth
CONAMA Conselho Nacional de Meio Ambiente (2005) Resolução no 357 de 17 de março de 2005. Diário Oficial da República Federativa do Brasil, Poder Executivo, Brasília (DF)
Connell JH (1978) Diversity in tropical rain forests and coral reefs. Science 199:1302–1310
Corbi JJ, Strixino ST, dos Santos A, Del Grande M (2006) Environmental diagnostic of metals and organochlorinated compounds in streams near sugar cane plantations activity (São Paulo State, Brazil). Quim Nova 29:61–65
Costa PF, Schulz UH (2010) The fish community as an indicator of biotic integrity of the streams in the Sinos River basin, Brazil. Braz J Biol 70:195–205
DEFRA – Department for Environment, Food & Rural Affairs (2010) Floating Pennywort, Hydrocotyle ranunculoides. https://secure.fera.defra.gov.uk/nonnativespecies/factsheet/downloadFactsheet.cfm?speciesId=1766. Accessed 20 March 2013
Dray S, Pélissier R, Couteron P et al (2012) Community ecology in the age of multivariate multiscale spatial analysis. Ecol Monogr 82:257–275
Dudgeon D (2008) Tropical stream ecology. Academic Press, London
Esteves FA (1998) Fundamentos de Limnologia. Interciências, Rio de Janeiro
Esteves KE, Alexandre CV (2011) Development of an index of biotic integrity based on fish communities to assess the effects of rural and urban land use on a stream in Southeastern Brazil. Int Rev Hydrobiol 96:296–317
Ferreira A, Paula FR, Ferraz SFB, Gerhard P, Kashiwaqui EAL, Cyrino JEP, Martinelli LA (2012a) Riparian coverage affects diets of characids in neotropical streams. Ecol Freshw Fish 21:12–22
Ferreira A, Cyrino JEP, Duarte-Neto PJ, Martinelli LA (2012b) Permeability of riparian forest strips in agricultural, small subtropical watersheds in south-eastern Brazil. Mar Freshw Res 63:1272–1282
Ferreira CP, Casatti L (2006) Integridade biótica de um córrego na Bacia do Alto Rio Paraná avaliada por meio da comunidade de peixes. Biota Neotrop 6:1–25
Ferreira FC, Silva AT, Gonçalves CS, Jr P (2014) Disentangling the influences of habitat structure and limnological predictors on stream fish communities of a coastal basin, southeastern Brazil. Neotrop Ichthyol 12:177–186
Ferreira KM (2007) Biology and ecomorphology of stream fishes from the Mogi-Guaçu. Neotrop Ichthyol 5:311–326
Filoso S, Martinelli LA, Williams MR, Lara LB, Krusche A, Ballester MV, Victoria R, Camargo PB (2003) Land use and nitrogen export in the Piracicaba River basin, Southeast Brazil. Biogeochemistry 65:275–294
Fiorio PR, Demattê JAM, Sparovek G (2000) Cronologia e impacto ambiental do uso da terra na Microbacia Hidrográfica do Ceveiro, em Piracicaba, SP. Pesq Agrop Brasileira 35:671–679
Furlan N, Esteves KE, Quináglia GA (2012) Environmental factors associated with fish distribution in an urban neotropical river (Upper Tietê River Basin, São Paulo, Brazil). Environ Biol Fish 96:77–92
Genito D, Gburek WJ, Sharpley AN (2002) Response of stream macro invertebrates to agricultural land cover in a small watershed. J Freshw Ecol 17:109–119
Gerhard P (2005) Comunidades de peixes de riachos em função da paisagem da bacia do rio Corumbataí, Estado de São Paulo. Dissertation, University of São Paulo
Gibbs HK, Ruesch AS, Achard F, Clayton MK, Holmgren P, Ramankutty N, Foley JA (2010) Tropical forests were the primary sources of new agricultural land in the 1980s and 1990s. Proc Natl Acad Sci U S A 107:16732–16737
Gibran FZ, Ferreira KM, Castro RMC (2001) Diet of Crenicichla britskii (Perciformes: Cichlidae) in a stream of Rio Aguapeí basin, upper Rio Paraná system, Southeastern Brazil. Biota Neotrop 1:1–5
Godinho FN (2008) The influence of riparian vegetation on freshwater fish. In: Arizpe D, João AM, Rabaça E (eds) Sustainable riparian zones - a management guide. Generalitat Valenciana, Spain, pp 96–100
Goldemberg J, Coelho ST, Guardabassi P (2008) The sustainability of ethanol production from sugarcane. Energ Policy 36:2086–2097
Gregory SV, Swanson FJ, Mckee WA, Cummins KW (1991) An ecosystem perspective of riparian zones. Bioscience 41:540–551
Harding JS, Benfield EF, Bolstad PV, Helfman GS, Jones EBD III (1998) Stream biodiversity: the ghost of land use past. Proc Natl Acad Sci U S A 95:14843–14847
Hayes JP, Adam MD, Bateman D et al (1996) Integrating research and forest management in riparian áreas of the Oregon Coast Range. West J Appl For 11:85–89
Hope ACA (1968) A simplified Monte Carlo significance test procedure. J R Stat Soc 30:582–598
Hued AC, Bistoni MA (2005) Development and validation of a biotic index for evaluation of environmental quality in the central region of Argentina. Hydrobiologia 543:279–298
Humpl M, Lusk S (2006) Effect of multiple electro-fishing on determining the structure of fish communities in small streams. Folia Zool 55:315–322
IRRIGART - Engenharia e Consultoria em Recursos Hídricos (2006) Plano de gerenciamento integrado para remediação e proteção dos recursos hídricos da Sub-Bacia do Atibaia com ênfase no Reservatório de Salto Grande - Americana – SP. http://www.comitepcj.sp.gov.br/download/PGISaltoGrande_Relatorio.pdf. Accessed 20 November 2011
IRRIGART Engenharia e Consultoria em Recursos Hídricos (2007) Bacias hidrográficas dos rios Piracicaba, Capivari e Jundiaí: situação dos recursos hídricos 2004/2006 - relatório síntese. http://www.comitepcj.sp.gov.br/download/RS/RS-04-06_Relatorio-Sintese.pdf. Accessed 12 November 2011
Iwata T, Nakano S, Inoue M (2012) Impacts of past riparian deforestation on stream communities in a tropical rain forest. Ecol Appl 13:461–473
Karr JR, Toth LA, Dudley DR (1985) Fish communities of Midwestern rivers: a history of degradation. Bioscience 35:90–95
Krebs CJ (1989) Ecological methodology. Harper Collins Publishers, New York
Lammert M, Allan JD (1999) Assessing biotic integrity of streams: effects of scale in measuring the influence of land use/cover and habitat structure on fish and macroinvertebrates. Environ Manag 23:257–270
Lorion CM, Kennedy BP (2009) Riparian forest buffers mitigate the effects of deforestation on fish assemblages in tropical headwater streams. Ecol Appl 19:468–479
Ludwig A, Reynolds F (1988) Statistical ecology: A primer on methods and computing. John Wiley & Sons, New York
MAPPC (2012) Municipal Agricultural Production and Population Census (Instituto Brasileiro de Geografia e Estatistica. http://go.nature.com/vdm5lC. Accessed 12 February 2014
Marceniuk AP, Hilsdorf AWS (2010) Peixes das cabeceiras do Rio Tietê e Parque das Neblinas. Canal 6, Bauru
Marques VS, Argento MSF (1988) O uso de flutuadores para avaliação da vazão de canais fluviais. Geociências 7:173–186
Martinelli LA, Ballester MV, Krusche A, Victoria RL, Camargo PB, Bernardes M, Ometto JPHB (1999) Land-cover changes and δ13C composition of riverine particulate organic matter in the Piracicaba River Basin (southeast region of Brazil). Limnol Oceanogr 44:1826–1833
Mazzoni R, Fenerich-Verani N, Caramaschi EP (2000) Electrofishing as a sampling technique for coastal stream fish populations and communities in the Southeast of Brazil. Braz J Biol: 205–216
Melo AS, Schneck F, Hepp LU, Simões NR, Siqueira T, Bini LM (2011) Focusing on variation: methods and applications of the concept of beta diversity in aquatic ecosystems. Acta Limnol Bras 23:318–331
Murphy ML, Meehan RW (1991) Stream ecosystem. Am Fish Soc Spec Publ 19:17–46
Naiman RJ, Décamps H (1997) The ecology of interfaces : riparian zones. Annu Rev Ecol Syst 28:621–658
Nislow KH (2005) Forest change and stream fish habitat: lessons from “Olde” and New England. J Fish Biol 67:186–204
Ometto JPHB, Martinelli LA, Ballester MV, Gessner A, Krusche AV, Victoria RL, Williams M (2000) Effects of land use on water chemistry and macroinvertebrates in two streams of the Piracicaba river basin, south-east Brazil. Freshw Biol 44:327–337
Oyakawa OT, Akama A, Mautari KC, Nolasco JC (2006) Peixes de riachos da Mata Atlântica nas Unidades de Conservação do Vale do Ribeira de Iguape no Estado de São Paulo. Editora Neotropica, São Paulo
Peres-Neto PR, Legendre P, Dray S, Borcard D (2006) Variation partitioning of species data matrices: estimation and comparison of fractions. Ecology 87:2614–2625
PCJ (Comitês das Bacias Hidrográficas dos Rios Piracicaba, Capivari e Jundiaí) (2012) Plano das bacias hidrográficas dos Rios Piracicaba, Capivari e Jundiaí 2010 a 2020 - relatório síntese. ArtPrinter Gráficos, São Paulo
Piacente FJ (2005) Agroindústria canavieira e o sistema de gestão ambiental: o caso das usinas localizadas nas bacias hidrográficas dos rios Piracicaba, Capivari e Jundiaí. Dissertation, University of Campinas
Pusey BJ, Arthington AA (2003) Importance of the riparian zone to the conservation and management of freshwater fish : a review. Mar Freshw Res 54:1–16
R Development Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rabeni CF, Smale MA (1995) Effects of siltation on stream fishes and the potential mitigating role of the buffering riparian zone. Hydrobiologia 303:211–219
Riseng CM, Wiley MJ, Black RW, Munn MD (2011) Impacts of agricultural land use on biological integrity: a causal analysis. Ecol Appl 21:3128–3146
Rosgen DL (1994) A classification of natural rivers. Catena 22:169–199
Santos I, Fill HD, Sugai MRVB, Buba H, Kishi RT, Marone D, Lautert LF (2001) Hidrometria aplicada. Instituto de Tecnologia para o Desenvolvimento, Curitiba
Schlosser IJ (1995) Critical landscape attributes that influence fish population dynamics in headwater streams. Hydrobiologia 303:71–81
SMA - Secretaria do Meio Ambiente (2011) Biodiversidade no Estado de São Paulo. Secretaria de Estado do Meio Ambiente, São Paulo
Smeets E, Junginger M, Faaij A, Walter A, Dolzan P (2006) Sustainability of Brazilian bio-ethanol . http://www.chem.uu.nl/nws/www/general/ personal/smeets_a_files/nieuw/E2006-110. Report NWS- E-2006-110, ISBN 90-8672-012-9
Stanfield W, Kilgour BW (2013) How proximity of land use affects stream fish and habitat. River Res Appl 29:891–905
Stauffer JC, Goldstein RM, Newman RM (2000) Relationship of wooded riparian zones and runoff potential to fish community composition in agricultural streams. Can J Fish Aquat Sci 57:307–316
Teixeira-De Mello F, Kristensen EA, Meerhoff M, González-Bergonzoni I, Baattrup-Pedersen A, Iglesias C, Jeppesen E (2014) Monitoring fish communities in wadeable lowland streams: comparing the efficiency of electrofishing methods at contrasting fish assemblages. Environ Monit Assess 186:1665–1677
ter Braak CFJ (1986) Canonical correspondence analysis: a new eigenvector technique for multivariate direct gradient analysis. Ecology 67:1167–1179
ter Braak CJF (1987) CANOCO - a FORTRAN Program for canonical community ordination by partial, Detrended, Canonical correspondence analysis, Principal components analysis, and Redundance analysis. Agricultural Mathematics Group, Wageningen, The Netherlands
ter Braak CJF, Šmilauer P (2002) CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (version 4.5), Ithaca, NY, USA
Tófoli RM, Alves GH, Higut R, Cunico AM, Hahn NS (2013) Diet and feeding selectivity of a benthivorous fish in streams: responses to the effects of urbanization. J Fish Biol 83:39–51
van den Wollenberg AL (1977) Redundancy analysis. An alternative for canonical correlation analysis. Psychometrika 42:207–219
Vazzoler AEA (1996) Biologia da reprodução de peixes teleósteos: teoria e prática. EDUEM, Maringá
Wang L, Lyons J, Kanehl P, Gatti R (1997) Influences of watershed land use on habitat quality and biotic integrity in Wisconsin streams. Fisheries 22:6–12
Warwick RM (1986) A new method for detecting pollution effects on marine macrobenthic communities. Mar Biol 92:557–562
Whately M, Cunha P (2007) Cantareira 2006: um olhar sobre o maior manancial de água da Região Metropolitana de São Paulo. Instituto Socioambiental, São Paulo
Wichert G, Rapport D (1998) Fish community structure as a measure of degradation and rehabilitation of Riparian systems in an agricultural drainage basin. Environ Manag 22:425–443
Wright JP, Flecker AS (2004) Deforesting the riverscape: the effects of wood on fish diversity in a Venezuelan piedmont stream. Biol Conserv 120:439–447
Yemane D, Field JG, Leslie RW (2005) Exploring the effects of fishing on fish assemblages using abundance biomass comparison (abc) curves. ICES J Mar Sci 62:374–379
Zeni JO, Casatti L (2014) The influence of habitat homogenization on the trophic structure of fish fauna in tropical streams. Hydrobiologia 726:259–270
Zippin C (1958) An evaluation of the removal method of estimating animal populations. Biometrics 12:163–169
Zuubier P, Vooren JV (2008) Sugarcane ethanol: Contributions to climate change mitigation and the environment. Wageningen Academic Publishers, Netherlands
Acknowledgments
The authors thank the Instituto de Pesca (APTA/SAA) for providing infrastructure; its staff and students for their help in field work (Sérgio Luiz da Silva, Paulo Henrique Gusmão, Ludmila Cristina Baldi and Katerine Pereira Carvalho); Fundação de Amparo à Pesquisa do Estado de São Paulo for financial support (Proc. 2010/52069-6); Conselho Nacional de Desenvolvimento Científico e Tecnológico for the MsC scholarship (Proc. 154078/2010-7); Osvaldo T. Oyakawa for fish identification; Cíntia Kameyama for macrophyte identification; Mirna Ferracini for map drawing; Antônio Olinto, André Vaz, Leonardo Tachibana and Eduardo Paes for help with statistical analyses and José Carlos Perdigão and Walter Lima for help in locating sampling sites. This study was approved by the Ethics Committee of the Fisheries Institute of São Paulo (CEEAIP) (Protocol 07/2010).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
dos Santos, F.B., Ferreira, F.C. & Esteves, K.E. Assessing the importance of the riparian zone for stream fish communities in a sugarcane dominated landscape (Piracicaba River Basin, Southeast Brazil). Environ Biol Fish 98, 1895–1912 (2015). https://doi.org/10.1007/s10641-015-0406-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-015-0406-4