Abstract
Freshwater ecosystems play a vital role in sustaining human populations; however, these environments are increasingly subject to human interference, driven by land use modifications, species introductions, pollution, and habitat loss. We sampled the ichthyofauna and collected environmental variables from 24 coastal streams in northeastern Brazil. Fish composition, abundance, and biomass served as the response variables, while physicochemical data, hydrological attributes, riparian characteristics, and substrate composition were considered as predictor variables. Our main objective was to evaluate the impact of a riparian land use gradient, ranging from conserved to degraded scenarios (i.e., forested, grassy, and urban streams), on the structure of fish assemblages. To achieve this, the graphical relationship of ABC plots and their W values between the three sets of streams was evaluated, and a Hellinger transformation-based Redundancy Analysis (tb-RDA) was conducted and we compared fish composition among the stream categories using a PERMANOVA test. We identified that the W values of forested and urban streams were significantly different, indicating a shift from k to r-strategists; the tb-RDA revealed three stream categories: (i) forested streams and their association with Anablepsoides bahianus and Parotocinclus cf. jumbo, (ii) grassy streams and their association with Poecilia vivipara and Astyanax cf. bimaculatus, and (iii) urban streams and their relationship with Poecilia reticulata and Oreochromis niloticus. We also found important differences between fish composition along the riparian land use gradient, with urban streams showing a significant divergence from grassy and forested streams. We observed a loss of native species and an introduction of exotic species in the evaluated gradient, consistent with the premises of niche theory.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
For millennia, freshwater resources have been crucial for human settlements, leading to the proximity of houses, farms, and industries to water bodies (Strayer and Dudgeon 2010). Human interference in these environments is widespread across most landscapes of the world, and certain areas have undergone modifications for hundreds of years (Ligeiro et al. 2013). Land use modifications (Utz et al. 2010), the introduction of alien species, pollution, and habitat loss are some of the primary causes of human interference, contributing to the ongoing sixth vertebrate mass extinction (Ceballos et al. 2015, 2020).
Streams represent interconnected ecosystems intimately linked to their surrounding environment (Karr 1998), where physical habitat factors (Karr and Yoder 2004), physicochemical attributes (Allan 2004), and landscape alterations (Walsh et al. 2005) collectively influence the ecological structure of biological communities. As streams are intricately connected to historical and current activities within their watersheds (Harding et al. 1998; Gergel et al. 2002), they can undergo significant degradation at various temporal and spatial scales due to land use changes (Allan 2004). Symptoms of the urban stream syndrome include a flashier hydrograph, elevated concentrations of nutrients and contaminants, altered channel morphology, and reduced biotic richness, with increased dominance of tolerant species (Walsh et al. 2005). The synergistic interplay of these characteristics positions fish stream communities as highly reliable indicators for comprehensive assessments of aquatic ecosystem health (Karr et al. 1986; Otero et al. 2006; Jia and Chen 2013).
Freshwater ecosystems in South America, especially along the Brazilian coast, are characterized by limited legal regulations and face significant threats (Reis et al. 2016). The Atlantic Forest is the primary ecosystem in this region of the country and is also recognized as a biodiversity hotspot (Myers et al. 2000). Northeastern Brazil is a particularly significant biogeographic province, characterized by numerous isolated drainages and a rich endemic ichthyofauna (Ribeiro 2006), which presents a growing challenge for the study of fishes and their habitats amidst rapid anthropogenic changes in the twenty-first century. These anthropogenic changes can also affect the selection of fish species in accordance with their life strategies (Casatti et al. 2006; Moniruzzaman et al. 2021).
The region of Mundaú-Manguaba Estuarine Lagoon Complex (MMELC) in Alagoas state has an estuary region of remarkable environmental and scenic importance. It has a history of human occupation spanning 400 years, driven by its proximity to the state capital (Souza et al. 2004). However, it is currently undergoing urbanization processes and experiencing associated impacts (Walsh et al. 2005). This location encompasses a fragmented landscape comprising preserved forests, agricultural farms, urban housing, industrial zones, and mining areas (Marques 1991; Araújo and Calado 2008; Melo-Magalhães et al. 2009; Cotovicz Junior et al. 2012; Menezes et al. 2012; Guimarães Junior et al. 2017; Santos et al. 2021; Teles 2023). The water resources from MMELC serve as an irrigation source for sugar cane plantations, sugar-alcohol, and petrochemical industries, and it holds enormous potential for the region's tourism sector. It is a vital ecosystem that directly sustains approximately 260 thousand residents in its surrounding areas (Araújo et al. 2011) and indirectly benefits a population of around one million people living in cities connected to it (IBGE 2020).
This study represents the first quantitative ichthyological baseline on streams of the MMELC and serves as a model of studies for other similar environments. The main objective proposed was to evaluate the impact of a riparian land use gradient ranging from conserved to degraded scenarios (i.e., forested, grassy, and urban streams) in fish assemblages of Atlantic forest watershed. To achieve this, we conducted analyses focusing on (i) differences in species composition between the three stream categories, (ii) shifts on a classical evolutionary theory of r and k selection strategies adopted by that fish assemblages, and (iii) the relationship between fish numerical abundances and stream environmental variables (physicochemical, hydrological, riparian, and substrate composition).
We are based on the principles of niche theory proposed by Hutchinson (1957), redefined by Leibold (1995), and revised by Pocheville (2015) and on the elements of the urban stream syndrome (Walsh et al. 2005) to propose our hypothesis. We postulate that a gradient in riparian land use will reflect differences in the fish composition, with a dominance of r-strategist species that exhibit a graphical uplift pattern on the abundance curve in degraded ecosystems (urban and grassy streams), while k-strategist species exhibit a graphical uplift pattern on the biomass curve in conserved environments (forested streams). We also expect that different relationships will be observed between environmental variables and fish numerical abundances in each land use scenario. We hope that this approach enables us to identify potential biological indicators and tools for monitoring this watershed.
Material and methods
Study area
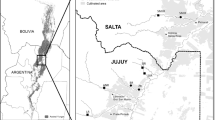
The Mundaú-Manguaba Estuarine Complex (MMELC) is a neotropical estuarine complex located in Alagoas, Brazil. It is situated between coordinates 9° 35′ S and 9° 45′ S and 35° 44′ W to 35° 58′ W. The complex can be characterized as a bar estuary (Wilson 1998), where a semi-continuous structure of sandstone and sand restricts the exchange of water between the ocean and the lagoon, resulting in the formation of a diverse biological community (Fig. 1).
The lower portion of MMELC includes, in addition to the state capital Maceió, the municipalities Santa Luzia do Norte, Coqueiro Seco, Pilar, Satuba, and Marechal Deodoro (Guimarães Junior et al. 2017). The region is composed of the Mundaú́ lagoon (27 km2) and Manguaba lagoons (42 km2) and a region of connecting channels (12 km2) (Menezes et al. 2012). It is one of the main aquatic ecosystems of the state, characterized by its vast spatial extension, ecosystem diversity, and high regional economic relevance (Souza et al. 2004). The downstream portion of MMELC features a coastal plain supported by a tertiary plateau with heights ranging from 50 to 100 m (Ribeiro et al. 2011). The climate is hot and humid, with an average annual temperature of 24 °C (Silva and Souza 2008). It experiences two well-defined seasons: a rainy season from March to August and a dry season from September to February (Araújo and Calado 2008; Menezes et al. 2012).
The MMELC watershed exhibits diversified riparian land use, including urban areas, industrial zones (chlorochemical, mining, and oil companies), agricultural areas (sugar cane cultivation), and some remnants of Atlantic forest fragments confined to protected areas, such as Catolé-Fernão Velho Environmental Protection Area, Santa Rita Environmental Protection Area, Saco da Pedra Ecological Reserve, Maceió Municipal Park, and several private reserves (Bernard et al. 2011; Guimarães Junior et al. 2017). According to ANA (2006), these watersheds are composed of several small drainages of clear water streams that collectively support multiple cities by providing essential water resources.
Sampling sites
In this study, 24 coastal freshwater streams within the MMELC drainage were selected based on the following criteria: first and second-order streams, permanent flow, similarity between sampled stretches, accessibility, and feasibility of collection (maximum depth up to 1.3 m). The sampled streams were categorized into three riparian land use types: ten streams with a predominance of tree vegetation in the riparian buffer (forested), seven streams with shrub and grassy vegetation (grassy), and seven streams with a predominance of urban occupation and exposed soil (urban) (Fig. 2). The inclusion criteria in this category were based on the highest land use percentage within a radius of 1.6 km (Roth et al. 1996). Data on riparian land use were obtained using Google Earth and QGIS tools in order to generate three stream categories. All samplings took place during the dry period. Streams S1 to S16 were sampled in December 2017, and streams S17 to S24 were sampled in November and December 2018.
Sampling of environmental variables
A stream stretch of 80 m was selected in each location, which was divided into transversal transects every 20 m to characterize the hydrological variables: channel width (cm), depth (cm), and velocity (m/s), using FL K2 hydrometric reel. Variables describe riparian composition (grasses, shrubs, trees, exposed soil, sugar cane, bamboo, fine roots, and coarse roots) and substrate composition (silt, sand, gravel, pebble, rock, slab, coarse litter, fine litter, branches and trunks, and decaying substrate). Riparian and substrate composition were obtained through direct observation and quantified as percentage of occupation in each transect along the stream stretch. The average was taken for all environmental variables.
At each sampling stretch, the following physical and chemical parameters were analyzed on-site: pH, temperature (°C), conductivity (µm/cm), and dissolved oxygen (mg.L-1), using METROHM pH meter (826 pH) and portable HANNA oximeter (HI 9146). For the determination of thermotolerant coliforms, water was collected at a fixed point of the stretch using 250 mL sterile and autoclavable plastic bottles. The collected samples were kept under refrigeration at 4 ± 2 °C (APHA 2017) and later analyzed using the most probable number method for thermotolerant coliforms in 100 mL “SMEWW 23rd 2017 9221 B / E” Standard Methods for the Examination of Water and Wastewater (APHA 2017).
For the other chemical variables, a composite sample was collected by mixing several subsamples from the four transects defined in the stretch. The composite sample was collected in a 20-L plastic bucket and then transferred to 1.0-L PET bottles, along with their respective preservatives, for laboratory analyses. Before samples were added, the bottles were homogenized by mixing them with water from the corresponding stretch of each sample.
Samples were sent to the environmental analysis laboratory of “Qualitex Engenharia e Serviços” company for processing. The following parameters were analyzed: total solids, oils and greases, chlorophyll a, ammonia nitrogen, chemical oxygen demand, biochemical oxygen demand, total hardness, nitrate, salinity, chlorides, true color, total dissolved solids, total sulfate, total alkalinity, total phosphorus, mercury, aluminum, and iron. The analysis was conducted using methodologies described in the Standard Methods for the Examination of Water and Wastewater (APHA 2017).
Sampling of biological variables
To collect fish specimens, an electric fishing equipment (220 V AC current, 50–60 Hz, 3.4–4.1 A, 1000 W) was used for 45 min in the same delimited stream stretch of 80 m, and each stretch was sampled only once. Collected specimens were anesthetized with eugenol (CONCEA 2018), fixed in 10% formalin (after a minimum of 48 h), and subsequently preserved in 70% ethanol. Fish identifications were verified by specialists (Fernando Rogério de Carvalho—UFMS and Francisco Langeani Neto – UNESP/SJRP). The biotic data has been grouped, biomass was measured, and specimens were counted before being deposited in the fish collection of the São Paulo State University, campus of São José do Rio Preto (DZSJRP 21208–21315; 22,719–22,721).
Statistical analysis
To assess sample representativeness, we compared the Coleman Rarefaction curve with non-parametric species richness estimators: incidence coverage estimate (ICE) and abundance coverage estimate (ACE) (Colwell and Coddington 1994), using the fossil package (Vavrek 2011) (Supplemental Script 1). The comparative graphical relationship of the abundance biomass curve (ABC) used in the present study was proposed by Warwick (1986) for the diagnosis of pollution in marine environments. In addition to the graphical pattern, the method provides the W value which enables the classification of the streams.
The ABC plots and W value have a theoretical background in the classical evolutionary theory of r- and k-selection (Yemane et al. 2005) and demonstrate the influence of the physical, chemical, and riparian land use predictors on the ichthyofauna (Clarke and Warwick 1994; Casatti et al. 2006) including in this study region. The concepts of r-strategist species (small generalists with rapid sexual maturation, reproduction, and growth) and k-strategist species (large specialists with slow sexual maturation, reproduction, and growth) (Pianka 1970). The W value ranges from − 1 to 1, where negative values indicate environmental disturbance, positive values indicate the absence of environmental disturbance, and values close to zero imply moderate disturbance (Warwick 1986; Warwick et al. 1987; Warwick and Clarke 1994; Magurran 2005). To obtain the ABC graphs and W values for each stream category, the dataset (abundances and biomass) was transformed with the fourth root method. For this analysis, the forams package (Aluizio 2015) was used (Supplemental Script 2). To test differences in W values between stream categories, a Kruskal–Wallis H test and post hoc analysis with Dunn test (with p values adjusted with the Bonferroni method) were conducted. These analyses utilized the FSA (Ogle et al. 2023), lattice (Sarkar 2008), and rcompanion (Mangiafico 2023) packages (Supplemental Script 3). Non-parametric statistical tests were used due to violations of the assumptions of normality and homoscedasticity required for parametric analysis. Levene’s test was used to check for homogeneity of variances, while the Shapiro–Wilk test was used to assess normality.
To determine the potential selection strategy for each species, we examined the pattern of ABC curves for each fish species, as proposed by Casatti et al. (2006). Additionally, we conducted a literature review of their life history attributes, including body size, feeding habits, and reproductive strategies (Pianka 1970). Based on these criteria, we classified species according to their tendency to r-strategists or k-strategists. R-strategists were characterized by their contribution in abundance overlapping biomass, small body size, broad trophic plasticity, and low reproductive requirements. Conversely, k-strategists were characterized by their contribution in biomass overlapping abundances, larger body size, specific trophic preferences, and high reproductive requirements.
To reveal gradients in the composition of physical and chemical variables between riparian land use types and species-variable relationships, transformation-based Redundancy Analysis (tb-RDA) was performed (ter Braak and Smilauer 2012) (Supplemental Script 4). Thus, the relative importance of environmental variables in the composition of the fish assemblage by riparian land use was estimated. This method extracts and summarizes the relationship between response and explanatory variables (Legendre and Legendre 1998). All abundance data were transformed using Hellinger series, and all 44 environmental variables collected (Supplemental Table 1) were normalized and arranged in a similarity matrix. Some variables, such as fine and thick roots, sugar cane, bamboo, silt, gravel, pebble, fine litter, thick litter, branches and trunks, decomposing material, slab, chlorophyll a, oils and greases, chemical oxygen demand, total hardness, salinity, solids total dissolved substances, turbidity, alkalinity, and aluminum, were removed due to the positive correlations with the other variables (r ≥ 0.65, p < 0.005). The resulting 21 variables underwent a forward selection procedure (Blanchet et al. 2008), resulting in the removal of variables such as width, speed, conductivity, biochemical demand for oxygen, chlorides, color, sulfate, phosphorus, and total solids. At the end of the selection procedure, 13 variables were retained: depth, dissolved oxygen, mercury, iron, thermotolerant coliforms, pH, nitrogen, rock, exposed soil, sand, nitrate, riparian tree, and shrub cover. An analysis of variance (ANOVA) was used to test the significance of tb-RDA. For these analyses (tb-RDA, correlation matrix, forward selection, and ANOVA), the vegan package (Oksanen et al. 2019) was utilized.
To evaluate the variation in fish species abundance among the stream categories, a non-parametric multivariate analysis method (PERMANOVA with Bray Curtis distances) was employed and was followed by a post hoc pairwise comparison of the Bray Curtis dissimilarity matrices generated by the PERMANOVA analysis (Supplemental Script 5). The PERMANOVA analysis function from the vegan package (Oksanen 2007) and the post hoc pairwise tests were performed using the Adonis function (Martinez-Arbizu 2020). PERMANOVA, as a statistical technique, accounts for the multivariate structure of ecological data and enables testing for significant differences in species abundance. In this study, all analyses were conducted using R version 4.0.4 (Development Core Team R 2011).
Results
Overall, 33,191 fish specimens were collected, belonging to nine orders, 17 families, 22 genera, and 24 species (Supplemental Table 2). Overall, 17,324 g of fish humid biomass (Supplemental Table 3). The estimated richness with ICE and ACE was 24 species for both, indicating a representative sampling. In forested streams, 1,997 individuals were collected and distributed among 14 families and 19 species, two of which were exclusive (Parotocinclus cf. jumbo and Anablepsoides bahianus). In grassy streams, 1694 fishes were captured and distributed among 15 families, with 21 species, five of which were exclusive (Serrapinnus piaba, Awaous tajasica, Hyphessobrycon eques, Microphis lineatus, and Eleotris pisonis). In urban streams, 29,500 specimens were collected and distributed among seven families and eight species, without exclusive species. Cyprinodontiformes dominated in abundance in urban streams, especially Poecilia reticulata, which had a high abundance in six of them. Among all species collected, six were present in the three riparian land uses (Astyanax cf. bimaculatus, Hoplias cf. brasiliensis, Callichthys callichthys, Rhamdia aff. quelen, Poecilia reticulata, Poecilia vivipara, and Synbranchus aff. madeirae).
Forested and grassy streams were considered as environments with low disturbances according to ABC plots (Fig. 3), with a W statistic value close to zero but positive and different from urban sites. Urban streams were classified as environmentally disturbed, according to the negative values of the W and the ABC graphical pattern. According to the Kruskal–Wallis H test, there was a significant difference between the three categories of streams (H(2) = 6.98, p = 0.0305) (Fig. 4), and the only significative difference was found between forested and urban streams (p = 0.0259).
The eigenvalues of two first constrained axes (r2 = 0.40) in the tb-RDA were significant (p = 0.001 and p = 0.049) (Table 1) and the variance of significance test of tb-RDA (ANOVA) was also significant (r2 = 0.51, p = 0.001) (Table 2 and Fig. 5).
Ordination biplot for the transformed-based Redundancy Analysis (tb-RDA). Arrows represent the predictor scores. Green, yellow, and red squares are site scores of forested, grassy, and urban streams, respectively. Gray squares are species scores; the species codes are explained in Supplemental Table 2
Forested streams were associated with high scores of instream descriptors as rocky substrate and dissolved oxygen. In this set of streams, the presence of A. bahianus was exclusive. The grassy streams were associated with the physical attributes such as sand substrate and channel depth, and the abundance of A. cf. bimaculatus was considerable. Poecilia vivipara was associated with the transition from grassy to urban environments, showing a strong relationship with iron concentrations, especially in S23 stream. Urban streams were associated with thermotolerant coliforms and chemical predictors such as nitrate, iron, mercury, and nitrogen, as well as pH values and the presence of P. reticulata (Fig. 5).
The PERMANOVA demonstrated that the fish species abundance was significantly different between the three stream categories (r2 = 0.34, p = 0.001) (Table 3). In the pairwise comparisons (Table 4), significant differences were observed between urban and the other categories of streams.
Discussion
Our study revealed a specious-rich, diverse, and poorly studied ichthyofauna that responded to the environmental gradient in riparian land use, showing consistent differences in fish composition and abundance among forested, grassy, and urban streams. The numerical abundance of fish in urban streams was nearly 15 times higher than in other stream categories, primarily influenced by exotic Cyprinodontiformes. Additionally, forested and grassy streams shared the majority of recorded fish species, highlighting the similarity between these two environmental types. Studies that have evaluated the diversity of ichthyofauna in streams with presence of trees have reported that greater habitat complexity is associated with higher species richness compared to other types of ecosystems (Curtean-Banaduc and Banaduc 2017; Brejão et al. 2018; Virgilio et al. 2018), and this situation could be seen in this study, particularly when we group forest and grassy streams in comparison to urban ones.
In degraded ecosystems, r-strategist species, dominant in abundance, are more present, influencing the overlap of the abundance curve on the graph; while in conserved environments, there is a predominance of k-strategist species that exhibit a graphical uplift pattern on the biomass curve. Despite having different characteristics, forested and grassy environments showed a similar pattern on ABC plots and W values closely clustered and close to zero, with a tendency towards moderately impaired scenarios. Aside from that, grassy and urban streams were also closely related. It is concerning because it shows that forested streams are losing their original characteristics and turning into more disturbed environments, similar to grassy environments, that receive a great physical disturbance. The W values for urban streams showed a pattern where k-strategist species are practically absent, and the r-strategists like the exotic P. reticulata become dominant in terms of abundance. Exotic species are those species present in a habitat but not native to it (Blanco and Basanta 2015) and tend to be more tolerant of habitat modification and chemical pollution, and this is especially true for the globally distributed species P. reticulata (Kennard et al. 2005).
The analysis of tb-RDA supports the predetermined three sets of streams and reinforces the relationship between species with specific environmental variables:
-
(a)
Forested streams were characterized by a heterogeneous habitat with two exclusive species, A. bahianus and P. cf. jumbo, where their presence could be considered an indicator of a more conserved environment. Both species have special adaptations to live in forested streams, marked by the presence of rocky substrate and high dissolved oxygen concentrations, as found by Costa (2008) in other Atlantic Forest streams. The Parotocinclus genus was also recorded in streams with gravel bottom, clear, shallow, moderate flowing, and high oxygenated waters (Garavello 1977; Sarmento-Soares et al. 2009). These data corroborate other studies that evaluate the biodiversity in freshwater environments with tree cover, confirming that riparian tree vegetation provides greater habitat complexity (i.e., roots, submerged, woody debris, and deciduous leaves) providing micro and meso-habitats that favor rare species in terms of abundance and consequently increase the total richness of fish species (Curtean-Banaduc and Banaduc 2017; Brejão et al. 2018; Virgilio et al. 2018)
-
(b)
Grassy streams were characterized by the dominance of A. cf. bimaculatus and P. vivipara and influenced by the descriptors channel depth, sand substrate, and grass cover. Both species are considered generalists, presenting wide niche breadth (Silva-Camacho et al. 2014) and have omnivorous diets, feeding on aquatic and terrestrial invertebrates, detritus, plants, and insect larvae (Neves and Monteiro 2003). Luke et al. (2019) report that, although the structure of fish communities in grassy/shrubby riparian streams is more similar to those in reference forests, effects on species richness, abundance, and biomass can still be observed, which is also evident in this study. Water depth is an important abiotic factor influencing fish dominance in terms of biomass (Souza et al. 2008), and in our study, this pattern is likely related to channel erosion and siltation process. We also observed other generalist species associated with grassy streams, such as S. piaba, G. cf. brasiliensis, and P. kennedyi, which further reinforces the advantage of this feeding habit in environmentally disturbed scenarios. A similar pattern of dominance by generalist species was reported by Casatti et al. (2009)
-
(c)
Urban streams showed low fish richness compared to grassy and forested streams, with dominance in terms of abundance of generalists, tolerant, and exotic species, such as P. reticulata and O. niloticus. This environmental pattern is strongly associated with environmental disturbance and ongoing impact (Walrath et al. 2016; Ruaro et al. 2018). Impaired streams have poor food availability, favoring species with flexible diets and specialized reproductive strategies that are less vulnerable to environmental degradation (Cruz et al. 2013). Poecilia reticulata and O. niloticus are considered exotic species capable of surviving in highly contaminated environments (Yesilbudak and Erdem 2014; Gomes-Silva et al. 2020). These species were associated with the descriptors nitrate, nitrogen, iron, mercury, pH, and thermotolerant coliforms. In this context, urban activities contribute to the deposition of nitrogen compounds, heavy metals, fecal contamination, and altered pH values, mainly due to the suppression of riparian vegetation and sewage discharge (Buda and Dewalle 2009; Kaushal et al. 2011; Connor et al. 2014; Wang et al. 2014; Crisigiovanni et al. 2020). The urban streams studied align with the pattern suggested by Hale et al. (2014) where human activities increase disturbances that can alter chemical and physical characteristics of the water body, which in turn influence biological characteristics. Miller et al. (2021) observed significant changes in the functional composition of fish assemblages in response to urbanization, favoring detritivory species with hypoxia tolerance, while ruralization favored only the herbivory feeding habit
The results of the PERMANOVA pairwise post hoc analysis confirm significant differences between forested and grassy streams when compared to the urban streams group. This distinction is reinforced by the graphical positioning of the majority of forested and grassy streams, situated on an axis opposite to that of urban streams. Studies that associate species with land use gradients mainly consider the trophic habit of species (generalists or specialists), demonstrating the importance of understanding how the fish fauna structure responds to gradients (Allan 2004; Casatti et al. 2010, 2012; Teresa and Romero 2010; Pease et al. 2015; Ortega et al. 2021). The combination of methodologies performed in this study reveals structural differences in the fish community related to riparian land uses in the MMELC, and the high abundance of P. reticulata and O. niloticus is considered an indicator of poor water quality in this condition.
In describing and comparing the relationship between fish fauna community structure and environmental variables (physicochemical, hydrological, riparian, and substrate composition), we found different responses of the ichthyofauna (composition, abundance, and biomass) to environmental disturbances. This can be explained in the light of niche theory (Hutchinson 1957; Leibold 1995), which considers the niche as a visualization of the ecological mechanisms, the conjunction of the responses to, and of the impacts on, the environmental factors. In this study, we found that the land use types act as an environmental filter that determines habitat and meso-habitat characteristics, selecting the fish species pool. Our findings underscore the significance of riparian vegetation cover, affirming the correlation between the ecotone and ichthyofauna, as discussed by Casatti et al. (2009). The existence of riparian vegetation in lotic environments is linked to heightened diversity and habitat complexity, offering optimal conditions for sustaining aquatic biodiversity, even in moderately impaired scenarios.
In a present scenario of urban expansion and the lack of conservation initiatives, we proposed that the entire MMELC region could be negatively affected by the urban stream syndrome (Walsh et al. 2005; Booth et al. 2016). This worldwide phenomenon considers that human changes in land and riverscapes directly affect fish communities, leading to the loss of sensitive taxa and the substitution and increase of tolerant and exotic species (Walters et al. 2003; Komínková 2012), as was evident in this study.
With the data obtained in this study, it is possible to affirm that especially the Cyprinodontiformes have a better response to the gradients of land use, with the poeciliids being rarely found in the stretches with good physical evaluation, where there is the presence of the Rivulidae member A. bahianus. In streams with predominance of grassy vegetation, there were only poeciliids, with few individuals of the species P. reticulata and an abundance of P. vivipara. In the urban streams with a high level of environmental disturbance, the population structure of these two species was inverted, P. vivipara was rarely present, and P. reticulata showed a high abundance, being sampled in all stretches of the category.
We consider that these fish species respond to a gradient of degradation (forested, grassy, and urban streams), constituting the observation of their presence and abundance as bioindicators for the environmental quality of streams. Recent studies have confirmed this pattern with the high presence of the alien species P. reticulata and a decrease in native fish species in highly degraded urban streams (Miller et al. 2021; Ortega et al. 2021). It was confirmed that fish are good indicators and are considered as “sentinels of the environment” (Karr 1998), and we proposed a set of species that could be used as indicators of water quality in coastal tropical streams from northeastern Brazil.
Moreover, it can be anticipated that a significant decline in fish species richness and the degradation of stream habitat will result in the loss of various ecosystem goods and services that are beneficial to human populations (Colvin et al. 2019). These include, among others, essential resources like clean water and food, as well as the regulation of microclimates and the esthetic value derived from the natural environment (Ferreira et al. 2023). We expect that this approach could support future initiatives of conservation and management of degraded coastal streams to restore the ecosystem goods and services vital to us.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Agência Nacional de Águas ANA (2006) Plano de ações e gestão integrada do complexo estuarino lagunar Mundaú-Manguaba (CELMM): Resumo Executivo. Available in: http://arquivos.ana.gov.br/institucional/sge/CEDOC/Catalogo/2006/CELMM.pdf. Accessed 8 Jun 2023
Allan JD (2004) Landscapes and riverscapes: the influence of land use on stream ecosystems. Annu Rev Ecol Evol Syst 35:257–284. https://doi.org/10.1146/annurev.ecolsys.35.120202.110122
Aluizio R (2015) Foraminifera and community ecology analyses. R Package Documentation. https://search.r-project.org/CRAN/refmans/forams/html/00Index.html. Accessed 4 Dec 2023
Alvim MCC, Peret AC (2004) Food resources sustaining the fish fauna in a section of the Upper São Francisco River in Três Marias, MG. Brazil Braz J Biol 64(2):195–202. https://doi.org/10.1590/S1519-69842004000200003
American Public Health Association APHA (2017) Standard methods for the examination of water and wastewater. Washington, D.C
Amorim, PF, Costa WJM (2022) Evolution and biogeography of Anablepsoides killifishes shaped by neotropical geological events (Cyprinodontiformes, Aplocheilidae). Zool Scr 51(4). https://doi.org/10.1111/zsc.12539
Andreata JV (2012) Reprodução e alimentação de algumas espécies de peixes da lagoa Rodrigo de Freitas. Rio De Janeiro Oecolog Austr 16(3):501–524. https://doi.org/10.4257/oeco.2012.1603.11
Araújo MSLC, Calado TCS (2008) Bioecologia do caranguejo-uçá Ucides cordatus (Linnaeus) no complexo estuarino lagunar Mundáu/Manguaba (CELMM), Alagoas, Brasil. RGCI 8(2):169–181. https://doi.org/10.5894/rgci141
Araújo MP, Costa TLF, Carreira RS (2011) Esteróis como indicadores do acúmulo de esgotos domésticos em sedimentos de um sistema estuarino-lagunar tropical (Mundaú-Manguaba, AL). Quím Nova 34(1):64–70. https://doi.org/10.1590/S0100-40422011000100013
Ayala-Pérez LA, Miranda JR, Hernández DF, López AS, Romero GEM (2015) Ictiofauna marina y costera de Campeche. Universidad Autónoma de Campeche, Universidad Autónoma Metropolitana-Xochimilco, Campeche
Barros NHC, Lima LTB, Araújo AS, Gurgel LL, Chellappa NT, Chellappa S (2016) Estudos sobre as táticas e as estratégias reprodutivas de sete espécies de peixes de água doce do Rio Grande de Norte, Brasil. Holos 3:84–103. https://doi.org/10.15628/holos.2016.3648
Bernard E, Melo FPL, Pinto SRR (2011) Challenges and opportunities for biodiversity conservation in the Atlantic forest in face of bioethanol expansion. Trop Conserv Sci 4(3):267–275. https://doi.org/10.1177/194008291100400305
Blanchet FG, Legendre P, Borcard D (2008) Forward selection of explanatory variables. Ecology 89:2623–2632. https://doi.org/10.1890/07-0986.1
Blanco E, Basanta M (2015) Especies invasoras: cambios na investigación ecolóxica ao longo de medio século. Nova Acta Cient Compostelana (Biol) 22:1–13. https://revistas.usc.gal/index.php/nacc/article/view/2426
Booth DB, Roy AH, Smith B, Capps KA (2016) Global perspectives on the urban stream syndrome. Freshw Sci 35(1):412–420. https://www.journals.uchicago.edu/doi/full/10.1086/684940
Brejão GL, Gerhard P, Zuanon J (2013) Functional trophic composition of the ichthyofauna of forest streams in eastern Brazilian Amazon. Neotrop Ichthyol 11:361–373. https://doi.org/10.1590/S1679-62252013005000006
Brejão GL, Hoeinghaus DJ, Pérez-Mayorga MA, Ferraz SF, Casatti L (2018) Threshold responses of Amazonian stream fishes to timing and extent of deforestation. Conserv Biol 32(4):860–871. https://doi.org/10.1111/cobi.13061
Buda AR, Dewalle DR (2009) Dynamics of stream nitrate sources and flow pathways during storm flows on urban, forest and agricultural watersheds in central Pennsylvania, USA. Hydrol Process 23:3292–3305. https://doi.org/10.1002/hyp.7423
Casatti L, Langeani F, Ferreira CP (2006) Effects of physical habitat degradation on the stream fish assemblage structure in a pasture region. Environ Manage 38(6):974–982. https://doi.org/10.1007/s00267-005-0212-4
Casatti L, Ferreira CP, Carvalho FR (2009) Grass dominated stream sites exhibit low fish species diversity and dominance by guppies: an assessment of two tropical pasture rivers basins. Hydrobiologia 632:273–283. https://doi.org/10.1007/s10750-009-9849-y
Casatti L, Teresa FB, Souza TG, Bessa E, Manzotti AR, Gonçalves CS, Zeni JO (2012) From forest to cattail: how does the riparian zone influence stream fish? Neotrop Ichthyol 10(1):205–214. https://doi.org/10.1590/S1679-62252012000100020
Casatti L, Romero RM, Teresa FB (2010) Fish community structure along conservation gradient in Bodoquena plateau stream, central west of Brazil. Acta Limnol Bras 22:50–59. http://hdl.handle.net/11449/22491
Ceballos G, Ehrlich PR, Barnosky AD, Garcia A, Pringle RM, Palmer TM (2015) Accelerated modern human-induced species losses: entering the sixth mass extinction. Sci Adv 1:1–5. https://doi.org/10.1126/sciadv.1400253
Ceballos G, Ehrlich PR, Raven PH (2020) Vertebrates on the brink as indicators of biological annihilation and the sixth mass extinction. Proc Natl Acad Sci 117:13596–13602. https://doi.org/10.1073/pnas.1922686117
Clarke KR, Warwick RN (1994) Change in marine communities: an approach to statistical analysis and interpretation. Primer-E, Plymouth
Colvin SAR, Sullivan SP, Shirey PD, Colvin RW, Winemiller KO, Hughes RM, Fausch KD, Infante DM, Olden JD, Bestgen KR, Danehy RJ, Eby L (2019) Headwater streams and wetlands are critical for sustaining fish, fisheries, and ecosystem services. Fisheries 44(2):73–91. https://doi.org/10.1002/fsh.10229
Colwell RK, Coddington JA (1994) Estimating terrestrial biodiversity through extrapolation. Phil Trans R Soc Lond Series B: Biol Sci 345(1311):101–118. https://doi.org/10.1002/fsh.10229
Connor NP, Sarraino S, Frantz DE, Bushaw-Newton K, MacAvoy SE (2014) Geochemical characteristics of an urban river: influences of an anthropogenic landscape. Appl Geochem 47:209–216. https://doi.org/10.1016/j.apgeochem.2014.06.012
Conselho Nacional de Controle de Experimentação Animal CONCEA (2018) Diretrizes da prática de eutanásia do CONCEA. Brasília, DF https://www.mctic.gov.br/mctic/export/sites/institucional/institucional/concea/arquivos/legislacao/resolucoes_normativas/Resolucao-Normativa-n-37-Diretriz-da-Pratica-de-Eutanasia_site-concea.pdf. Accessed 22 Apr 2019
Costa WJEM (2008) Redescription of Rivulus bahianus (Cyprinodontiformes: Rivulidae) a killifish from the Atlantic forest of northeastern Brazil. Ichthyol Explor Fres 19(4):297–300. https://pfeil-verlag.de/wp-content/uploads/2015/05/ief19_4_02.pdf
Cotovicz Junior LC, Brandini N, Knoppers BA, Souza WFL, Medeiros PRP (2012) Comparação de modelos de índice de estado trófico do complexo estuarino lagunar Mundaú-Manguaba, (AL). Geochim Bras 26(1):7–18. https://doi.org/10.21715/gb.v26i1.353
Crisigiovanni EL, Nascimento EAD, Godoy RFB, Oliveira-Filho PCD, Vidal CMDS, Martins KG (2020) Inadequate riparian zone use directly decreases water quality of a low-order urban stream in southern Brazil. Rev Ambient Água 15(2):1–11. https://doi.org/10.4136/ambi-agua.2451
Cruz BB, Teshima FA, Cetra M (2013) Trophic organization and fish assemblage structure as disturbance indicators in headwater stream of lower Sorocaba River basin, São Paulo, Brazil. Neotrop Ichthyol 11(1):171–178. https://doi.org/10.1590/S1679-62252013000100020
Curtean-Banaduc A, Banaduc D (2017) The status of Romanogobio uranoscopus (Agassiz, 1828) species, in Maramures Mountain Nature Park (Romania). Transylv Rev Syst Ecol Rev 19(1):71–84. https://doi.org/10.1515/trser-2017-0007
Development Core Team R (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Austria
Feitosa FS, Rezende CF (2020) Trophic ecology of the fish Leporinus piau (Characiformes: Anostomidae) in an area influenced by a dam in the Parnaíba river. Rev Biol Trop 68(2):426–439. https://doi.org/10.15517/rbt.v68i2.38641
Ferreira V, Albariño R, Larrañaga A, LeRoy CJ, Masese FO, Moretti MS (2023) Ecosystem services provided by small streams: an overview. Hydrobiologia 850:2501–2535. https://doi.org/10.1007/s10750-022-05095-1
Fricke R, Eschmeyer WN, van der Laan R (eds) (2023) Eschmeyer’s catalog of fishes: genera, species, references. http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp. Accessed 9 Jul 2023
Garavello JC (1977) Systematics and geographical distribution of the genus Parotocinclus Eigenmann & Eigenmann, 1889 (Ostariophysi, Loricariidae). Arq Zool 28:1–37. https://doi.org/10.11606/issn.2176-7793.v28i4p1-37
Gergel SE, Turner MG, Miller JR, Melack JM, Stanley EH (2002) Landscape indicators of human impacts to riverine systems. Aquat Sci 64:118–128. https://doi.org/10.1007/s00027-002-8060-2
Gomes-Silva G, Cyubahiro E, Wronski T, Riesch R, Apio A, Plath M (2020) Water pollution affects fish community structure and alters evolutionary trajectories of invasive guppies (Poecilia reticulata). Sci Total Environ 730:e138912. https://doi.org/10.1016/j.scitotenv.2020.138912
Guimarães Junior SAM, Nascimento MC, Silva DJRP (2017) Impactos do uso da terra no complexo estuarino lagunar Mundaú - Manguaba – Alagoas – Brasil. Rev Contexto Geogr 2(3):86–99. https://doi.org/10.28998/contegeo.v2i3.6137
Hale R, Barbee NC, Swearer SE (2014) Assessing the likely responses by fishes to stream bank rehabilitation in a large, urban estuary. Aust Ecol 39:479–489. https://doi.org/10.1111/aec.12106
Harding JS, Benfield EF, Bolstad PV, Helfman GS, Jones EBD (1998) Stream biodiversity: the ghost of land use past. Proc Natl Acad Sci USA 95(25):14843–14847. https://doi.org/10.1073/pnas.95.25.1484
Hutchinson GE (1957) Concluding remarks. Cold Spring Harb Symp Quant Biol 22:415–417
Instituto Brasileiro de Geografia e Estatística IBGE (2020) Censo Demográfico: população residente no Brasil. https://ftp.ibge.gov.br/Estimativas_de_Populacao/Estimativas_2020/estimativa_dou_2020.pdf Acess: 20/06/2023.
Jarduli LR, Pereira AD, Garcia DAZ, Ferraz JD, Geller IV, Orsi ML (2021) Distribution modeling of Psellogrammus kennedyi (Eigenmann, 1903) and new records in the lower Paranapanema river, Brazil. Check List 17(5):1277–1284. https://doi.org/10.15560/17.5.1277
Jia YT, Chen YF (2013) River health assessment in a large river: bioindicators of fish population. Ecol Indic 26:24–32. https://doi.org/10.1016/j.ecolind.2012.10.011
Karr JR (1998) Rivers as sentinels: using the biology of rivers to guide landscape management. In: Naimam RJ, Bilby RE (eds) River ecology and management lessons from the Pacific coastal ecorregion. Springer Verlag, New York, pp 502–528
Karr JR, Yoder CO (2004) Biological assessment and criteria improve total maximum daily load decision making. J Environ Eng 130(6):594–604. https://doi.org/10.1061/(ASCE)0733-9372(2004)130:6(594)
Karr JR, Fausch KD, Angermeier PL, Yant PR, Schlosser IJ (1986) Assessing biological integrity in running waters: a method and its rationale. Publications Committee, Illinois
Kaushal SS, Groffman PM, Band LE, Elliot EM, Shields CA, Kendall C (2011) Tracking nonpoint source nitrogen pollution in human impacted watersheds. Environ Sci Technol 45(19):8225–8232. https://doi.org/10.1021/es200779e
Kennard MJ, Arthington AH, Pusey BJ, Harch BD (2005) Are alien fish a reliable indicator of river health? Freshw Biol 50:174–193. https://doi.org/10.1111/j.1365-2427.2004.01293.x
Knight JM (2010) Invasive ornamental fish: a potential threat to aquatic biodiversity in peninsular India. J Threat Taxa 2(2):700–704. https://doi.org/10.11609/JoTT.o2179.700-4
Kominkova D (2012) The urban stream syndrome - a mini-review. Open Environ Biol Monit J 5(1):24–29. https://doi.org/10.2174/1875040001205010024
Legendre P, Legendre L (1998) Numerical ecology. Elsevier, Amsterdan
Leibold MA (1995) The niche concept revisited: mechanistic models and community context. Ecology 76(5):1371–1382. https://doi.org/10.2307/1938141
Ligeiro R, Hughes RM, Kaufmann PM, Macedo DR, Firmiano KR, Ferreira WR, Oliveira D, Melo AS, Callisto M (2013) Defining quantitative stream disturbance gradients and the additive role of habitat variation to explain macroinvertebrates taxa richness. Ecol Indic 25(1):45–57. https://doi.org/10.1016/j.ecolind.2012.09.004
Linhares JCS, Resende Manna LR, Mazzoni R, Rezende CF, Silva, JRF (2014) Reproductive tactics optimizing the survival of the offspring of Cichlasoma orientale (Perciformes: Cichlidae). Rev Biol Trop 62(3):1007–1018. https://www.scielo.sa.cr/scielo.ph=sci_arttext&tlng=en
Lucena C, Soares H (2016) Review of species of the Astyanax bimaculatus “caudal peduncle spot” subgroup sensu Garutti & Langeani (Characiformes, Characidae) from the rio La Plata and rio São Francisco drainages and coastal systems of southern Brazil and Uruguay. Zootaxa 4072:101. https://doi.org/10.11646/zootaxa.4072.1.5
Luke SH, Slade EM, Gray CL, Annammala KV, Drewer J, Williamson J, Agama AL, Ationg M, Mitchell SL, Vairappan CS, Struebig MJ (2019) Riparian buffers in tropical agriculture: scientific support, effectiveness and directions for policy. J Appl Ecol 56:85–92. https://doi.org/10.1111/1365-2664.13280
Magurran AE (2005) Measuring biological diversity. Blackwell Publishing, New Jersey
Mangiafico SS (2023) rcompanion: functions to support extension education program evaluation. Rutgers Cooperative Extension, New Brunswick, New Jersey. version 2.4.30. https://CRAN.R-project.org/package=rcompanion/
Manna LR, Rezende CF, Mazzoni R (2017) Effect of body size on microhabitat preferences in stream-dwelling fishes. J App Ichthyol 33(2):193–202. https://doi.org/10.1111/jai.13320
Manna LR, Villéger S, Rezende CF, Mazzoni R (2019) High intraspecific variability in morphology and diet in tropical stream fish communities. Ecol Freshw Fish 28(1):41–52. https://doi.org/10.1111/eff.12425
Marques JGW (1991) Aspectos ecológicos da etnoictiologia dos pescadores no complexo estuarino lagunar Mundaú/Manguaba, Alagoas. PhD Thesis, Universidade Estadual de Campinas. http://www.repositorio.unicamp.br/handle/REPOSIP/315947
Martinez-Arbizu P (2020) PairwiseAdonis: pairwise multilevel comparison using Adonis. R Package Version 0.0.1. https://github.com/pmartinezarbizu/pairwiseAdonis. Accessed 20 Nov 2023
Mazzoni R, Iglesias-Rios R (2002) Environmentally related life history variations in Geophagus brasiliensis. J Fish Biol 61(6):1606–1618. https://doi.org/10.1111/j.1095-8649.2002.tb02501.x
Melo CED, Machado FDA, Pinto-Silva V (2004) Feeding habits of fish from a stream in the savanna of central Brazil, Araguaia Basin. Neotrop Ichthyol 2(1):37–44. https://doi.org/10.1590/S1679-62252004000100006
Melo-Magalhães EM, Medeiros PRP, Lira MCA, Koening ML, Moura NA (2009) Determination of eutrophic areas in Mundaú/Manguaba lagoons, Alagoas-Brasil, through studies of the phytoplanktonic community. Braz J Biol 62(2):271–280. https://doi.org/10.1590/S1519-69842009000200006
Menezes APD, Araújo MLSC, Calado TCS (2012) Bioecologia de Goniopsis cruentata (Latreille, 1803) (Decapoda, Grapsidae) do complexo estuarino lagunar Mundaú/Manguaba, Alagoas, Brasil. Nat Resour 2(2):37–49. https://doi.org/10.6008/ESS2237-9290.2012.002.0004
Miller NOR, Cunico AM, Gubiani EA, Piana PA (2021) Functional responses of stream fish communities to rural and urban land uses. Neotrop Ichthyol 19(3):e200134. https://doi.org/10.1590/1982-0224-2020-0134
Moniruzzaman M, Bhowmick AR, Karan S, Mukherjee J (2021) Spatial heterogeneity within habitat indicates the community assemblage pattern and life strategy. Ecol Indic 123:e107365. https://doi.org/10.1016/j.ecolind.2021.107365
Myers N, Mittermeier RA, Mittermeier CG, Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403(6772):853–858. https://doi.org/10.1038/35002501
Neves FM, Monteiro LR (2003) Body shape and size divergence among populations of Poecilia vivipara in coastal lagoons of south-eastern Brazil. J Fish Biol 63:928–941. https://doi.org/10.1046/j.1095-8649.2003.00199.x
Njiru M, Ojuok JE, Okeyo-Owuor JB, Muchiri M, Ntiba MJ, Cowx IG (2006) Some biological aspects and life history strategies of Nile tilapia Oreochromis niloticus (L.) in Lake Victoria, Kenya. Afri J Ecol 44(1):30–37. https://doi.org/10.1111/j.1365-2028.2006.00610.x
Ogle DH, Doll JC, Wheeler AP, Dinno A (2023) FSA: simple fisheries stock assessment methods. R package version 0.9.4 https://CRAN.R-project.org/package=FSA
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D et al (2019) Vegan: community ecology package. R package Available from: https://CRAN.R-project.org/package=vegan
Ortega JCG, Baccani L, Dorado-Rodrigues TF, Strussmann C, Fernandez IM, Morales J, Mateus L, Silva HP, Penha J (2021) Effects of urbanization and environmental heterogeneity on fish assemblages in small streams. Neotrop Ichthyol 19(3):e210050. https://doi.org/10.1590/1982-0224-2021-0050
Otero MEB, Spach HL, Queiroz GMLN, Santos C, Silva ALC (2006) O uso de atributos das assembléias de peixes para avaliar a integridade biótica em habitats rasos das baías de Antonina e Paranaguá, Paraná. Acta Biol Paraná 35:69–82. https://doi.org/10.5380/abpr.v35i0.6875
Pease AA, Taylor JM, Winemiller KO, King RS (2015) Ecoregional, catchment, and reach-scale environmental factors shape and functional traits structure of stream fish assemblages. Hydrobiologia 753(1):265–283. https://doi.org/10.1007/s10750-015-2235-z
Pianka ER (1970) On r- and K-selection. Am Nat 104(940):592–597. https://doi.org/10.1086/282697
Pocheville A (2015) The ecological niche: history and recent controversies. In: Heams T, Huneman P, Lecointre G, Silberstein M (eds) Handbook of evolutionary thinking in the sciences. Springer, Berlim, pp 547–586. https://doi.org/10.1007/978-94-017-9014-7_26
Quirino BA, Carniatto N, Gaiotto JV, Fugi R (2015) Seasonal variation in the use of food resources by small fishes inhabiting the littoral zone in a neotropical floodplain lake. Aquatic Ecol 49(4):431–440. https://doi.org/10.1007/s10452-015-9535-2
Reis RE, Albert JS, Dario FD, Mincarone MM, Petry P, Rocha LA (2016) Fish biodiversity and conservation in south America. J Fish Biol 89:12–47. https://doi.org/10.1111/jfb.13016
Ribeiro AC (2006) Tectonic history and the biogeography of the freshwater fishes from the coastal drainages of eastern Brazil: an example of faunal evolution associated with a divergent continental margin. Neotrop Ichthyol 4(2):225–246. https://doi.org/10.1590/S1679-62252006000200009
Ribeiro MA, Knoppers BA, Carreira RS (2011) Fontes e distribuição de matéria orgânica sedimentar no complexo estuarino lagunar de Mundaú-Manguaba/AL, utilizando esteróis e alcoóis como indicadores. Quim Nova 34(7):11111118. https://doi.org/10.1590/S0100-40422011000700002
Rocha FD, Casatti L, Pereira DC (2009) Structure and feeding of a stream fish assemblage in southeastern Brazil: evidence of low seasonal influences. Acta Limnol Bras 21(1):123–134. hdl.handle.net/11449/22484
Roth NE, Allan D, Erickson DL (1996) Landscape influences on stream biotic integrity assessed at multiple spatial scales. Landsc Ecol 11(3):141–156. https://doi.org/10.1007/BF02447513
Ruaro R, Mormul RP, Gubiani EA, Piana PA, Cunico AM, Graça WJ (2018) Non-native fish species are related to the loss of ecological integrity in neotropical streams: a multimetric approach. Hydrobiologia 817:413–430. https://doi.org/10.1007/s10750-018-3542-y
Santos MCD, da Silva Filho RC, Leite ACR, Nascentes CC, Botero WG, Santos JC (2021) Evaluation of potentially toxic elements in Mundaú Lagoon (Maceió, AL-Brazil): systematic environmental monitoring of water and food quality. J Braz Chem Soc 32:1762–1772. https://doi.org/10.21577/0103-5053.20210067
Sarkar D (2008) Lattice: multivariate data visualization with R. Springer, New York. https://doi.org/10.1007/978-0-387-75969-2
Sarmento-Soares LM, Lehmann PA, Martins-Pinheiro RF (2009) Parotocinclus arandai, a new species of Hypoptopomatine catfish (Siluriformes: Loricariidae) from the upper rios Jucuruçu and Buranhém, states of Bahia and Minas Gerais, Brazil. Neotrop Ichthyol 7(2):191–198. https://doi.org/10.1590/S1679-62252009000200009
Silva DF, Souza FAS (2008) Proposta de manejo sustentável para o complexo estuarino lagunar Mundaú Manguaba (AL). Rev Bras Geogr Fis 1(2):78–94. https://doi.org/10.26848/rbgf.v1.2.p78-94
Silva MJD, Ramos TPA, Diniz VD, Ramos RTDC, Medeiros ESF (2014) Ichthyofauna of Seridó/Borborema: a semi-arid region of Brazil. Biota Neotrop 14(3):1–6. https://doi.org/10.1590/1676-06032014007713
Silva-Camacho DS, Santos JNS, Gomes RS, Araújo FG (2014) Ecomorphological relationships among four Characiformes fish species in a tropical reservoir in south-eastern Brazil. Zoologia 31(1):28–34. https://doi.org/10.1590/S1984-46702014000100004
Souza RC, Reis RS, Fragoso Junior CR, Souza CF (2004) Uma análise da dragagem do complexo estuarino lagunar Mundaú Manguaba em Alagoas através de um modelo numérico hidrodinâmico bidimensional – resultados preliminares. Rev Bras Rec Hídr 9(4):21–31. https://doi.org/10.21168/rbrh.v9n4.p21-31
Souza UP, Costa RC, Martins IA, Fransozo A (2008) Relationships among Sciaenidae fish (Teleostei: Perciformes) and Penaeoidea shrimp (Decapoda: Dendrobranchiata) biomass from the north coast of São Paulo state, Brazil. Biota Neotrop 8(1):84–92. https://doi.org/10.1590/S1676-06032008000100011
Strayer DL, Dudgeon D (2010) Freshwater biodiversity conservation: recent progress and future challenges. J N Am Benthol Soc 29(1):344–358. https://doi.org/10.1899/08-171.1
Teixeira RL (1994) Abundance, reproductive period, and feeding habits of eleotrid fishes in estuarine habitats of north-east Brazil. J Fish Biol 45(5):749–761. https://doi.org/10.1111/j.1095-8649.1994.tb00941.x
Teles RC (2023) Migração forçada e mineração: a cidade de Maceió sob a ótica dos direitos humanos. Rev Direito e Prax 14(1):517–535. https://doi.org/10.1590/2179-8966/2023/73120
ter Braak CJF, Smilauer P (2012) CANOCO reference manual and user’s guide: software for ordination (version 5). Microcomputer Power, New York. https://research.wur.nl/en/publications/canoco-reference-manual-and-users-guide-software-for-ordination-v
Teresa FB, Romero RM (2010) Influence of the riparian zone phytophysiognomies on the longitudinal distribution of fishes: evidence from a Brazilian savanna stream. Neotrop Ichthyol 8(1):163–170. https://doi.org/10.1590/S1679-62252010000100019
Utz RM, Hildebrand RH, Raesly RL (2010) Regional differences in patterns of fish species loss with changing land use. Biol Conservat 143:688–699. https://doi.org/10.1016/j.biocon.2009.12.006
Vavrek MJ (2011) Fossil: palaeoecological and palaeogeographical analysis tools. Palaeontologia Electronica 14(1):1–16. https://cran.r-project.org/web/packages/fossil/index.html. Accessed 5 Dec 2023
Virgilio LR, Ramalho WP, Silva JCB, Susçuarana MS, Brito CH, Vieira LJS (2018) Does riparian vegetation affect fish assemblage? A longitudinal gradient analysis in three Amazonian streams. Acta Sci Biol Sci 40:e42562–e42562. https://doi.org/10.4025/actascibiolsci.v40i1.42562
Walrath JD, Dauwalter DC, Reinke D (2016) Influence of stream condition on habitat diversity and fish assemblage in an impaired Upper Snake River Basin watershed. Trans Am Fish Soc 145(4):821–834. https://doi.org/10.1080/00028487.2016.1159613
Walsh CJ, Roy AH, Feminella JW, Cottingham PD, Groffman PM, Morgan RP (2005) The urban stream syndrome: current knowledge and the search for a cure. J N Am Benthol Soc 24(3):706–723. https://doi.org/10.1899/04-028.1
Walters DM, Leigh DS, Bearden AB (2003) Urbanization, sedimentation, and the homogenization of fish assemblages in the Etowah River basin, USA. In: Kronvang B (ed) The interactions between sediments and water. Development in hydrology. Springer, Dordrecht, pp 5–10. https://doi.org/10.1007/978-94-017-3366-3_2
Wang S, Bernhardt ES, Wright JP (2014) Urban stream denitrifier communities are linked to lower functional resistance to multiple stressors associated with urbanization. Hydrobiologia 726:13–23. https://doi.org/10.1007/s10750-013-1747-7
Warwick RM (1986) A new method for detecting pollution effects on marine macrobenthic communities. Mar Biol 92:557–562. https://doi.org/10.1007/BF00392515
Warwick RM, Clarke KR (1994) Relearning the ABC: taxonomic changes and abundance/biomass relationships in disturbed benthic communities. Mar Biol 118(4):739–744. https://doi.org/10.1007/BF00347523
Warwick RM, PearsonRuswahyuni TH (1987) Detection of pollution effects on marine macrobenthic: further evaluation of the species abundance/biomass method. Mar Biol 95:193–200. https://doi.org/10.1007/BF00409005
Wilson JG (1998) The biology of estuarine management. St Edmundsburry Press Ltd, Suffolk
Yemane D, Field JG, Leslie RW (2005) Exploring the effects of fishing on fish assemblages using abundance biomass comparison (ABC) curves. J Mar Sci 62:374–379. https://doi.org/10.1016/j.icesjms.2005.01.009
Yesilbudak B, Erdem C (2014) Cadmium accumulation in gill, liver, kidney, and muscle tissues of common carp, Cyprinus carpio, and Nile tilapia. Oreochromis Niloticus Bull Environ Contam Toxicol 92(5):546–550. https://doi.org/10.1007/s00128-014-1228-3
Acknowledgements
This study was made possible by funding provided by the Fundação de Amparo à Pesquisa de Alagoas (FAPEAL – Proc. 60030 995/2016). MAPM receives research support from Ministerio de Ciencia, Tecnología e Innovación, MinCiencias, and UPTC (Research call: # 848–2019; Research project code: SGI–3000). The authors thank Fabricio Barreto Teresa for their comments about statistical analysis, Frederico Passos for their help with R software routines, Fernando Rogério de Carvalho and Francisco Langeani Neto for their assistance with fish identification, Jouel Bruno de Souza Lima for their contribution to the construction of electric fishing equipment, and Lilian Casatti and Maurício Cetra for pushing us forward, reviewers, and chief editors for their careful and precise assessment. We also thank our colleagues Michel Santos, Robert França, and Alexandre Lopes for their assistance in the field work and Marina Pascoalino and Angelo Manzotti for their photographic registers. We thank the IFAL for funding the manuscript translation and for logistical support.
Funding
Fapeal, 60030 995/2016, Renato De Mei Romero, Ministerio de Ciencia, Tecnología e Innovación, # 848–2019, María Angélica Pérez-Mayorga
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. The fish collection and transport were authorized by The Chico Mendes Institute for Biodiversity Conservation-ICMBio (Biodiversity Authorization and Information System-SISBIO license 60910–1, 26/Oct/2017).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
da Silva, R.G.A., Pérez-Mayorga, M.A. & Romero, R.M. Coastal freshwater stream fish fauna from a threatened estuarine lagoon complex in northeastern Brazil. Environ Biol Fish 106, 2141–2156 (2023). https://doi.org/10.1007/s10641-023-01495-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-023-01495-x