Abstract
Many mite species disperse via the air. However, most methods described for the study of aerial dispersal have some limitations in the collection and/or recovery of mites that could be improved. The aim of this study was to describe a rotatory funnel-shaped collector that directs the wind to adhesive surfaces covered with a glycerin-based solution. Tests were conducted on a soccer field at UESC, Ilhéus, Brazil, in four 8-day periods. In total, 330 mites of 52 species of Eriophyidae, Tarsonemidae, Diptilomiopidae, Iolinidae, Triophtydeidae, Astigmata, Tydeidae, Phytoseiidae, Scutacaridae, Oribatida, Ascidae, Dolichocybidae, Eupodidae, Pygmephoridae and Tenuipalpidae were collected. Of the mites captured in the first three periods, 67% were Aceria sp. (Eriophyidae), and in the fourth period 46% were Coccotydaeolus aff. bakeri and Paurotyndareus sp. (Iolinidae). Comparisons between the funnel-shaped collector with the tube-shaped rotatable model of Duffner et al. (J Pest Sci 74:1–6, 2001, adapted from Schliesske 1977) showed that the former captured >3× the number of mites and 2× the number of species. In conclusion, it is expected that the method described here could help in future pest management, and help solve ecological and behavioral problems involving airborne mite dispersal, offering a tool for monitoring, counting and identifying mites, or even other small arthropods, pollen and fungal spores, in experimental and applied studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Although dispersal in small wingless arthropods such as mites can occur by walking, phoresy, anthropogenic action or wind, aerial dispersal may have evolved as an advantageous means for large-scale movement, allowing the colonization of distant hosts by different families of plant-inhabiting mites (Jeppson et al. 1975; Jung and Croft 2001; Osakabe et al. 2008; Hoy 2011; Monteiro et al. 2018). Several morphological and behavioral adaptations seem to facilitate transport of these organisms by wind (Mitchel 1970; Bell et al. 2005; Clotuche et al. 2011; Melo et al. 2014; Kiedrowicz et al. 2017; Laska et al. 2019). Some eriophyoid mites attach their legs or caudal lobes to the plant surface, lifting the abdomen to be transported by air currents (Smith 1960; Nault and Styer 1969; Sabelis and Bruin 1996; Ozman and Goolsby 2005; Melo et al. 2014; Valenzano et al. 2019; Kuczyński et al. 2020). In addition, many spider mites (Tetranychidae) commonly move to the apex of the host plants in large populations, increasing exposition to the wind. Some species use webs for dispersal by ‘ballooning’, attaching web to host’s surface and allowing their ‘body’ to become airborne, or ‘silk balls’ composed of mites and their silk threads, facilitating group dispersal by wind (Bell et al. 2005; Reynolds et al. 2007; Clotuche et al. 2011; Santos et al. 2020).
Besides various types of laboratory methods, as wind tunnels and microenvironment simulations (Gamliel-Atinsky et al. 2009; Michalska et al. 2010; Galvão et al. 2012; Valenzano et al. 2019; Kuczyński et al. 2020; Majer et al. 2021), several trapping methods have been described to study airborne mites’ dispersal in the field, usually involving adhesive surfaces exposed to wind (Davis 1964; Barké et al. 1972; Zhao and Amrine 1997; Michalska et al. 2010; Monfreda et al. 2010; Galvão et al. 2012). Some more sophisticated field methods optimize the capture using structures to collect the wind and direct it to those surfaces (Duffner et al. 2001). The rotatable model of Duffner et al. (2001, adapted from Schliesske 1977), for example, based on a movable PVC tube to direct dominant wind currents to glass slides covered with petroleum jelly (e.g., vaseline), is among the more effective collectors described. Other models use plates, trays, panels and various types of adhesive surfaces covered with motor oil (Zhao and Amrine 1997), mineral oil (Galvão et al. 2012), silicone grease (Staples and Allington 1956; Nault and Styer 1969; Bergh and McCoy 1997; Bergh 2001) and tanglefoot with petroleum ether solutions (Davis 1964; Barké et al. 1972), consuming a long time to screen grease-coated surfaces and to process the often deformed or obscured mites embedded on them, making it difficult to clean specimens before mounting (Nault and Styer 1969; Monfreda et al. 2010). Although water traps allow the mounting without cleaning, the large solution volumes needed for capture generally must be vacuum-filtered using filter papers, what is time consuming and facilitates loss of specimens (Zhao and Amrine 1997; Tixier et al. 1998; Galvão et al. 2012).
This study describes a method for capturing airborne mites, based on a rotatory funnel-shaped collector to direct the wind to adhesive surfaces covered with glycerin. The rotatory tube-shaped collector adopted by Duffner et al. (2001) was used for comparison.
Materials and methods
The method consists of a rotatory funnel-shaped collector to concentrate air currents with mites over capture units covered with an adhesive solution of glycerin (Figs. 1, 2). The gyratory capacity was provided by a tripod formed by two wheels and a vertical rotatory axis supported by a sturdy base, to allow both wind-driven movement and horizontal funnel stability. The equipment was installed on a soccer field (Fig. 3) at the campus of the Universidade Estadual de Santa Cruz (UESC), located in the Salobrinho district, Ilhéus, BA, Brazil (14°45′35″ S, 39°13′49″ W).
The funnel-shaped collector. a Lateral view. b Anterior view, with the funnel cover removed. c Superior view of the collector tray with capture units attached. d Anterior view of the equipment installed. b bucket, c concrete, cb cross bar, ct collector tray, cw counterweight, fc funnel cover, ff funnel floor, ir inferior rudder, mb middle bar, n nuts, ra rotatory axis, rt receptor tube, sb shelving bracket, sr superior rudder, st support tube, w wheel and wa steel washer
Capture units and equipment installation. a Components of a capture unit. b Collector tray, with capture units attached, inserted above the funnel floor. c Isolated funnel-shaped collector installed. d Panoramic view of the equipment on the soccer field at UESC. e Funnel- and tube-shaped collectors installed simultaneously
Equipment description
The funnel-shaped collector (Figs. 1A, D, 2B, C) consisted of an 8-mm-thick funnel floor made with a PVC plate of 25 × 50 × 78 cm (Fig. 1B) covered by four 0.5-mm-thick aluminum sheets. Sheets were coupled to each other and to the funnel floor with contact adhesive, silver tape and epoxy putty to form a 1.5-m-long funnel, with anterior and posterior aperture widths of 60 and 15 cm, respectively. A PVC square pipe middle bar and three perpendicular wood cross bars were attached under the funnel floor with polyester putty and connected anteriorly in a 90° angle to a perpendicular threaded steel rod (1 cm diameter, 60 cm long) by a shelving bracket, forming the vertical rotatory axis (Fig. 1A, B). The rotatory axis was intended to be fitted with grease, at the time of installation, inside of a galvanized steel receptor tube (15 mm inner diameter, 37 cm long) inserted into a PVC support tube (10 cm inner diameter, 35 cm long) stuck with concrete in the center of a 58-cm-diameter and 60-cm-high plastic bucket (Fig. 1A, B).
To guide the aperture of the collector funnel towards the wind, two 8-mm-thick PVC rudders, a superior and an inferior, were attached to it with polyester putty (Figs. 1A, 2C). Two 3-cm-diameter wheels were attached under the funnel floor sides, with the function of rolling over the border of the bucket in response to wind orientation changes, supporting the floor horizontally together with the rotatory axis. Finally, a counterweight made with a 1-cm-diameter and 1-m-long cylindrical steel bar supporting a 1-kg weight attached to the end was prepared to be fitted inside the middle bar to provide the funnel floor leveling at the installation time.
Capture units (113×) were made of glass microscopy slides with a 3-mm-thick barrier made of a cylindrical rigid wire to the borders of each slide (Fig. 2A). A contact adhesive was used to attach the wire to the glass, and a piece of epoxy putty to connect the wire tips. The internal interface between the wire and the glass was sealed with a lead tetroxide solution. The capture units were attached in 9 transversal rows with double-sided tape over an 8-mm-thick PVC collector tray to be fitted on the funnel floor (Figs. 1C, 2B).
Tube-shaped collector
The rotatable tube-shaped collector was based on that described by Duffner et al. (2001, adapted from Schliesske 1977), consisting on a chamber of a PVC tube (2 m long, 20 cm inner diameter) to concentrate air currents with mites over capture units covered with an adhesive solution of petroleum jelly (replaced by a glycerin based solution in the present study). To guide the anterior aperture of the collector towards the wind, a rudder was attached to the top of the posterior aperture of the tube. The gyratory capacity was provided by a tripod formed by two wheels and a vertical rotatory axis supported by a sturdy base, similar to those described for the funnel-shaped collector, to allow both wind-driven movement and horizontal tube stability. At the installation time, both ends of the tube were enclosed by a polystyrene ring with an aperture of 10 cm, in order to reduce the diameter of the tube openings to concentrate the wind over the capture units horizontally positioned on a tray in the floor of the tube.
Field installation
Equipment was installed on a soccer field in the UESC at 50 m from the closest fruit and ornamental trees and shrubs planted on the campus (Figs. 2, 3). Each installation period lasted 8 days, with the installation of one funnel-shaped collector in each of the three installation periods in 2020 (from 28 August to 4 September, 21–28 September and from 30 September to 7 October) and of four collectors (two funnels and two tube-shaped collectors simultaneously) in a fourth period in 2021 (4–11 March).
The same system, based on a vertical rotatory axis supported by a bucket, was used to sustain both the funnel- and tube-shaped collectors, leveled to the ground with the help of a manual level and by adjusting the counterweight. Precise positioning of the wheels on the border of each bucket was obtained by adjusting the height of the collectors, through the locking of two steel nuts docked around the threaded rod of the rotatory axis. Finally, the collector trays with the capture units were positioned in the funnel- and tube-shaped collectors, after adding to each unit 1 mL of an adhesive solution composed by a 10:2:1 mixture of glycerin, detergent and water, respectively. Equipment was always installed and removed between 09:00 and 10:00 AM.
Data analysis
The complete area of each capture unit was analyzed under a Leica EZ4 stereomicroscope, with the use of a square (0.5 × 0.5 mm) black opaque plastic basis for orientation and contrast under horizontal illumination. Mites present were removed with a needle, slide mounted in Hoyer’s, kept at 56 °C for 10 days in an oven, and sealed with Glyptal (Krantz and Walter 2009). Mites were determined as species/morphospecies under a phase contrast microscope and counted.
The totals of mite individuals collected in each of the first three installation periods were used for comparison of the funnel-shaped collectors capture efficiency between the periods, considering the possible influence of variable wind speed (km/h), temperature (°C) and rainfall (mm). The totals of mite individuals collected in the various equipment types in the fourth installation period were used for comparison of effectiveness of capture between the funnel- and tube-shaped collectors. Climatic data were obtained from the Comissão Executiva do Plano da Lavoura Cacaueira (CEPEC/CEPLAC), Ilhéus, BA, Brazil, and from https://www.climatempo.com.br.
Results
In total, 330 mites of 52 species were captured: Eriophyidae (12 spp.), Tarsonemidae (12), Diptilomiopidae (5), Iolinidae (5), Triophtydeidae (1), Astigmata (3), Tydeidae (3), Phytoseiidae (2), Scutacaridae (2), Oribatida (2), Ascidae (1), Dolichocybidae (1), Eupodidae (1), Pygmephoridae (1) and Tenuipalpidae (1) (Tables 1, 2). The number of species collected increased from the first (3 spp.) to the second (7), third (15) and fourth (40) installation periods, with Aceria sp. (Eriophyidae) being the most abundant species in the first three periods and Coccotydaeolus aff. bakeri and Paurotyndareus sp. (Iolinidae) in the fourth. Those species, together with Benoinyssus sp. (Eupodidae), Tarsonemus bilobatus (Tarsonemidae), and Abacarus sp. (Eriophyidae), represented 77% of all mites captured.
The mite totals obtained in the funnel-shaped collectors in the first, second and third installation periods were of 31, 36 and 82 specimens, respectively. The average temperature presented similar values, oscillating from 21.5 to 24.8 °C, and wind speed ranged from 0.4 to 1.3 m/s (Fig. 4). Only the accumulated rainfall (1.6 mm3), the number of days with rain (3 days) and the maximum rainfall per day (0.6 mm3) were much lower in the third period than in the first and second periods, both presenting at least 1 day of rainfall higher than 5.6 mm3. On the other hand, the funnel-shaped collectors 1 and 2 captured, respectively, 58 and 80 mites in the fourth period, in which both the average temperature and wind speed were higher than in the first three periods, oscillating from 25.9 to 27.8 °C and from 2.4 to 4.1 m/s.

Source Comissão Executiva do Plano da Lavoura Cacaueira (CEPEC/CEPLAC) and https://www.climatempo.com.br
Rainfall, average wind speed and temperature on the first (28 Aug–4 Sep), second (21–28 Sep) and third (30 Sep–7 Oct) installation periods in 2020, and on the fourth installation period (4–11 Mar 2021), at Ilhéus, BA, Brazil.
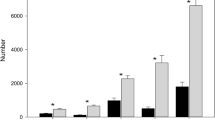
In total 181 mites of 40 species were captured in the two funnel and two tube-shaped collectors in the fourth period (Table 2). The numbers of mites (138) and species (35) captured in the two funnel-shaped collectors were higher than in the two tube-shaped collectors (43 mites, 15 species). Twenty-five species were found exclusively in the funnel-shaped collectors, whereas only five spp. were exclusive for the tube-shaped collectors and 10 spp. were found in both.
Considering each equipment separately, the funnel-shaped collectors 1 and 2 captured, respectively, 58 and 80 individuals, whereas the tube-shaped collectors 1 and 2 captured, respectively, only 26 and 17 specimens. The numbers of species collected in each individual equipment were also higher in the funnel-shaped collectors 1 and 2, with 27 and 17 species, respectively, against only 10 and 8 species captured in the tube-shaped collectors 1 and 2.
Discussion
Mites captured from the wind represented a large number of families and species diversity, proving that the equipment worked not only for capturing plant-inhabiting mites, but also other mite groups. Unlike most studies on the dispersal of plant-inhabiting mites (Davis 1964; Barké et al. 1972; Bergh and McCoy 1997; Bergh 2001; Duffner et al. 2001; Galvão et al. 2012), the equipment here described was not installed inside areas of monocultures infested by pest mites, where their densities in the wind are expected to be higher, indicating its efficiency in the capture of airborne mites in a distance of at least 50 m from the closest fruit and ornamental trees and shrubs. Moreover, since the composition and dominance of species captured in different periods probably reflect temporary abundances in their populations, the efficiency of the method capturing this variation may be useful in basic and applied studies involving temporal fluctuations in mite dispersal.
Regardless of the methods used for capture, plant-inhabiting mites have been widely represented among the groups that are aerially dispersed (Staples and Allington 1956; Davis 1964; Nault and Styer 1969; Barké et al. 1972; Bergh and McCoy 1997; Bergh 2001; Duffner et al. 2001). This was confirmed in the present work only in the first three installation periods, in which 94% of the mites collected were representative of Diptilomiopidae, Eriophyidae, Tarsonemidae and Tydeidae, with Aceria sp. and Abacarus sp. among the most abundant species. On the other hand, the collection of 70% of non-exclusively plant-inhabiting mites in the fourth period, represented by Iolinidae, Triophtydeidae, Eupodidae, Pygmephoridae, Dolichocybidae, Scutacaridae, Astigmata and Oribatida, with Coccotydaeolus aff. bakeri and Paurotyndareus sp. among the most abundant species, may have been the result of the low rainfall and higher temperature in that period. As most of those mite families are reported from soil, litter, moss, dry grass, fungi, tree branches, bird nests, insects and other substrates (Wood 1965; Kazmierski 1980; Walter et al. 2009; Theron et al. 2012), it is likely that a possible increase in the suspended sediments under those conditions may have facilitated their dispersal by the wind. In addition, it is known that high rainfall rates are generally unfavorable to mite population increase, whereas dry periods and low precipitation levels are favorable (Hoy 2011), and perhaps those propitious conditions could explain the higher numbers of mites captured in the third and fourth periods.
The combined use of adhesive surfaces based on a glycerin solution with a rotatory collector to concentrate the prevailing wind probably implied a great advantage of the proposed method. Most methods for trapping airborne plant-inhabiting mites use oils, glues and petroleum products as adhesives (Davis 1964; Barké et al. 1972; Zhao and Amrine 1997; Bergh and McCoy 1997; Bergh 2001; Duffner et al. 2001; Galvão et al. 2012), consuming time to process and often deforming or obscuring the mites (Nault and Styer 1969; Monfreda et al. 2010). Others use water with detergents (Zhao and Amrine 1997; Tixier et al. 1998; Monfreda et al. 2010; Galvão et al. 2012), requiring filtration and increasing the chances of specimen loss. Among the studies mentioned above, only Duffner et al. (2001) used a rotatable equipment to direct the dominant wind currents to microscope slides provided with an adhesive surface (vaseline in their case, instead of the glycerin-based solution adopted in the present study). The direct comparisons conducted in the present work between the funnel-shaped collector with the tube-shaped collector of Duffner et al. (2001) showed that the former collected more than 3× the number of mites and 2× the number of species captured in the tube-shaped collector. Such improved efficiency confirmed the expectation that the funnel-shaped collector may concentrate more air currents than the tube-shaped collector, increasing the chances of capturing a greater abundance and diversity of mites.
Based on the efficiency of the method described here, it is expected that the combined use of the funnel-shaped collector with a glycerin-based adhesive surface could help solve future pest management, control, and ecological and behavioral problems involving airborne dispersal of mites, offering qualitative and quantitative information on the population dynamics of mites in future experimental and/or applied studies. Its probable application in the study of the dispersal of insects, other small wingless arthropods, or even of pollen and fungal spores, could also be tested.
Data availability
Data provided.
Code availability
Not applicable.
References
Barké HE, Davis R, Hunter PE (1972) Studies on a peach silver mite Aculus cornutus. J Ga Entmol Soc 7:171–178
Bell JR, Bohan DA, Shaw EM, Weyman GS (2005) Ballooning dispersal using silk: world fauna, phylogenies, genetics and models. Bull Entomo Res 95:69–114. https://doi.org/10.1079/BER2004350
Bergh JC (2001) Ecology and aerobiology of dispersing citrus rust mites (Acari: Eriophyidae) in Central Florida. Environ Entomol 30:318–326. https://doi.org/10.1603/0046-225X-30.2.318
Bergh JC, McCoy CW (1997) Aerial dispersal of citrus rust mite (Acari: Eriophyidae) from Florida Citrus Groves. Environ Entomol 26:256–264. https://doi.org/10.1093/ee/26.2.256
Clotuche G, Mailleux AC, Fernadéz AA, Deneubourg JL, Detrain C, Hance T (2011) The formation of collective silk balls in the spider mite Tetranychus urticae Koch. PLoS ONE 6:e18854. https://doi.org/10.1371/journal.pone.0018854
Davis R (1964) Autecological studies of Rhynacus breitlowi Davis (Acarina: Eriophyidae). Florida Entomol Soc 47:113–122
Duffner K, Schruft G, Guggenheim R (2001) Passive dispersal of the grape rust mite Calepitrimerus vitis Nalepa 1905 (Acari, Eriophyoidea) in vineyards. J Pest Sci 74:1–6
Galvão AS, Melo JWS, Monteiro VB, Lima DB, De Moraes GJ, Gondim MGC Jr (2012) Dispersal strategies of Aceria guerreronis (Acari: Eriophyidae), a coconut pest. Exp Appl Acarol 57(1):1–13
Gamliel-Atinsky E, Freeman S, Sztejnberg A, Maymon M, Ochoa R, Belausov E, Palevsky E (2009) Interaction of the mite Aceria mangiferae with Fusarium mangiferae, the causal agent of mango malformation disease. Phytopathology 99:152–159
Hoy MA (2011) Agricultural acarology: introduction to integrated mite management. CRC Press, Boca Raton, p 430
Jeppson LR, Keifer HH, Baker EW (1975) Mites injurious to economic plants. University of California Press, Berkeley, p 614
Jung C, Croft BA (2001) Aerial dispersal of phytoseiid mites (Acari: Phytoseiidae): estimating falling speed and dispersal distance of adult females. Oikos 94:182–190. https://doi.org/10.1034/j.1600-0706.2001.11044.x
Kazmierski A (1980) Coccotydaeolus rafalskii sp. nov. (Acari: Prostigmata) a new species of Tydeid mite from Poland. Acarologia 21:361–366
Kiedrowicz A, Kuczyński L, Lewandowski M, Proctor H, Skoracka A (2017) Behavioural responses to potential dispersal cues in two economically important species of cereal-feeding eriophyid mites. Sci Rep. https://doi.org/10.1038/s41598-017-04372-7
Krantz GW, Walter DE (2009) A manual of acarology, 3rd edn. Texas Tech University Press, Lubbock, p 807
Kuczyński L, Radwańska A, Karpicka-Ignatowska K, Laska A, Lewandowski M, Rector BG, Majer A, Raubic A, Skoracka A (2020) A comprehensive and cost-effective approach for investigating passive dispersal in minute invertebrates with case studies of phytophagous eriophyid mites. Exp Appl Acarol 82:17–31
Laska A, Rector BG, Skoracka A, Kuczynski L (2019) Can your behaviour blow you away? Contextual and phenotypic precursors to passive aerial dispersal in phytophagous mites. Anim Behav 155:141–151
Majer A, Laska A, Hein G, Kuczyński L, Skoracka A (2021) Hitchhiking or hang gliding? Dispersal strategies of two cereal-feeding eriophyoid mite species. Exp Appl Acarol 85(131–146):6. https://doi.org/10.1007/s10493-021-00661-z
Melo JWS, Lima DB, Sabelis M, Pallini A, Gondim MGC Jr (2014) Behaviour of coconut mites preceding take-off to passive aerial dispersal. Exp Appl Acarol 64:429–443. https://doi.org/10.1007/s10493-014-9835-6
Michalska K, Skoracka A, Navia D, Amrine JW (2010) Behavioural studies on eriophyoid mites: an overview. Exp Appl Acarol 51:31–59. https://doi.org/10.1007/s10493-009-9319-2
Mitchel R (1970) An analysis of dispersal in mites. Am Nat 104:425–431
Monfreda R, Lekveishvili M, Petanovic R, Amrine JW Jr (2010) Collection and detection of eriophyoid mites. Exp Appl Acarol 51:273–282. https://doi.org/10.1007/s10493-009-9315-6
Monteiro VB, Silva VF, Lima DB, Guedes RNC, Gondim MGC Jr (2018) Pesticides and passive dispersal: acaricide- and starvation-induced take-off of the predatory mite Neoseiulus baraki. Pest Manag Sci 74:1272–1278. https://doi.org/10.1002/ps.4851
Nault LR, Styer WE (1969) The dispersal of Aceria tulipae and three other grass-infesting eriophyid mites in Ohio. Ann Entomol Soc Am 62:1443–1455
Osakabe MH, Isobe H, Kasai A, Masuda R, Kubota S, Umeda M (2008) Aerodynamic advantages of upside-down take-off for aerial dispersal in Tetranychus spider mites. Exp Appl Acarol 44:165–183. https://doi.org/10.1007/s10493-008-9141-2
Ozman SK, Goolsby JA (2005) Biology and phenology of the eriophyid mite, Floracarus perrepae, on its native host in Australia, Old World climbing fern Lygodium microphyllum. Exp Appl Acarol 35:197–213. https://doi.org/10.1007/s10493-004-4186-3
Reynolds AM, Bohan DA, Bell JR (2007) Ballooning dispersal in arthropod taxa: conditions at take-off. Biol Lett 3:237–240. https://doi.org/10.1098/rsbl.2007.0109
Sabelis MW, Bruin J (1996) Evolutionary ecology: life history patterns, food plant choice and dispersal. In: Lindquist EE, Sabelis MW, Bruin J (eds) Eriophyoid mites—their biology, natural enemies and control. World crop pests, vol 6. Elsevier Science Publishing, Amsterdam, pp 329–366
Santos FA, Rolim GS, Nachman GS, Andrade DJ (2020) Using mathematical models to describe aerial dispersal and silk ball formation of peanut red spider mite, Tetranychus ogmophallos (Acari: Tetranychidae). Exp Appl Acarol 81:85–102
Schliesske J (1977) Untersuchungen zur Morphologie, Biologie und Verbreitung von Aculus fockeui Nal. et Trt. (Acari: Eryophyoidea) in Niedersachsen. PhD Thesis, Techn. Univ. Hannover
Smith BD (1960) The behaviour of the black currant gall mite (Phytoptus ribi Nal.) during the free-living phase of its life cycle. Ann Rep Long Ashton Agric Hort Res Sta 1959:130–136
Staples R, Allington WB (1956) Streak mosaic of wheat in Nebraska and its control. Res Bull Univ Nebr (Linc. campus), Agric Exp Stn no. 178, p 41
Theron N, Roets F, Dreyer LL, Esler KJ, Ueckermann EA (2012) A new genus and eight new species of Tydeoidea (Acari: Trombidiformes) from Protea species in South Africa. Int J Acarol 38(3):257–273. https://doi.org/10.1080/01647954.2011.619576
Tixier MS, Kreiter S, Auger P, Weber M (1998) Colonization of Languedoc vineyards by phytoseiid mites (Acari: Phytoseiidae): influence of wind crop environment. Exp Appl Acarol 22:523–542. https://doi.org/10.1023/A:1006085723427
Valenzano D, Bari G, Valeria M, de Lillo E (2019) Off-host survival of Eriophyoidea and remarks on their dispersal modes. Exp Appl Acarol 79:21–33. https://doi.org/10.1007/s10493-019-00417-w
Walter DE, Lindquist EE, Smith IM, Cook DR, Krantz GW (2009) Chapter thirteen: order Trombidiformes. In: Krantz GW, Walter DE (eds) A manual of acarology, 3rd edn. Texas Tech University Press, Lubbock, pp 233–420
Wood TG (1965) New and described species of Tydeidae (Acari) from moorland soils in Britain. Acarologia 7:663–672
Zhao S, Amrine JW Jr (1997) A new method for studying aerial dispersal behavior of eriophyoid mites (Acari: Eriophyoidea). Syst Appl Acarol 2:107–110
Acknowledgements
To Uilson V. Lopes, George A. Sodré and ‘Seção de Climatologia’ of ‘Comissão Executiva do Plano da Lavoura Cacaueira’, Ilhéus, BA, Brazil (CEPEC/CEPLAC), for their support and availability of the climatic dada, to ‘Universidade Estadual de Santa Cruz’, Ilhéus, BA, Brazil (UESC), for the availability of the soccer field and the PVC tubes for the tests, to G. F. Camargo, for the PVC plates, to Carlos H.W. Flechtmann (ESALQ, USP, Piracicaba, SP), André S.G. Sousa (UESC) and Rafaela A. da Silva (UESC), for part of the mite identifications. Special thanks are also due to James W. Amrine Jr. and Joachim Schliesske for the copy of Schliesske (1977), and to José W. S. Melo (UFCE, Fortaleza, CE) and Martin R. Alvarez (UESC) for suggestions. This research was partially supported by the ‘Fundação de Amparo à Pesquisa do Estado da Bahia (FAPESB)’, through the Msc fellowship to ACS (BOL1692/2019, BOL0372/2020 and BOL0476/2020).
Funding
This research was partially supported by the ‘Fundação de Amparo à Pesquisa do Estado da Bahia (FAPESB)’, through the Msc fellowship to ACS (BOL1692/2019, BOL0372/2020 and BOL0476/2020).
Author information
Authors and Affiliations
Contributions
Material preparation, data collection and analyses were performed by ACS, TRML, MSSC and ARO at UESC, Ilhéus. MGCGJ, ACL, NJF and GLB contributed with mite identifications, discussion of the results, and with major revisions. The first draft of the manuscript was written by ACS and ARO, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. This article is based on the dissertation of ACS, advised by ARO and MGCGJ at Programa de Pós Graduação em Produção Vegetal (PPGPV/UESC), Ilhéus, BA, Brazil.
Corresponding author
Ethics declarations
Conflict of interest
There’s no conflict/competing interest.
Ethical approval
Not applicable.
Consent to participate
All authors consented to participate.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Santos, A.C., Leite, T.R.M., Cunha, M.S.S. et al. A rotatory funnel-shaped collector for trapping airborne mites in a glycerin-based adhesive surface. Exp Appl Acarol 86, 189–200 (2022). https://doi.org/10.1007/s10493-022-00687-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-022-00687-x