Abstract
The genus Agrilus is one of the most diverse insect genera worldwide. The larval feeding activity causes extensive damage in both forests and orchards. In addition, more than 30 species have been introduced outside their native range so far, including the emerald ash borer Agrilus planipennis Fairmaire. Thus, the availability of efficient trapping protocols for early detection of Agrilus species at entry points is of utmost importance. In this study we tested whether trapping protocols developed for surveillance of A. planipennis in North America were also effective for other Agrilus species. In particular, through a multi-country assessment we compared the efficacy of detecting Agrilus species on: (i) green glue-coated prism traps vs. green Fluon-coated multi-funnel traps when baited with the green leaf volatile (Z)-3-hexenol or left unbaited; and (ii) green multi-panel traps vs. green multi-panel traps baited with dead adult Agrilus beetles (decoys). A total of 23,481 individuals from 45 Agrilus species were caught. Trap design significantly affected both species richness and abundance of Agrilus species in several of the countries where the trapping experiments were carried out, and green prism traps outperformed green multi-funnel traps in most cases. On the contrary, the addition of a (Z)-3-hexenol lure or dead adult beetle decoys on to traps did not improve trap catches. Our study highlights that reliable trap models to survey Agrilus species are already available, but also that there is the clear need to further investigate chemical ecology of Agrilus species to develop semiochemical lures that can improve detection efficacy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Key message
-
The genus Agrilus has > 3,300 species and includes a number of potentially invasive species
-
Efficient trapping protocols are needed for surveillance and early detection of Agrilus species
-
Green glue-coated prism traps generally performed better than green multi-funnel traps
-
The addition of a (Z)-3-hexenol lure or adult beetle decoys to traps did not increase trap catches
Introduction
The genus Agrilus (Coleoptera; Buprestidae) is one of the most diverse insect genera with over 3,300 species described worldwide (Jendek 2016; Kelnarova et al. 2019; Jendek and Grebennikov 2023). Adults mainly feed on leaves of host trees while larvae develop in living subcortical tissues (Kelnarova et al. 2019). The larval feeding activity can be sufficient to kill a host, especially when it has already been weakened by other abiotic (e.g., drought) or biotic (e.g., defoliation) factors (Muzika et al. 2000; Brück-Dyckhoff et al. 2019), causing extensive damage in both forests and orchards (Brown et al. 2015; Zhang et al. 2021; Perish et al. 2023). In addition, the absence of visible evidence of feeding within wood products has likely facilitated invasion by Agrilus species (Ruzzier et al. 2023). More than 30 species have been introduced outside their native range (Ruzzier et al. 2023), including the emerald ash borer A. planipennis Fairmaire, which has caused massive ecological and economic damage in North America (Kovacs et al. 2010; Klooster et al. 2018). Given that further introductions can be expected due to increasing trade and climate change (Brockerhoff and Liebhold 2017; Meurisse et al. 2019; Pureswaran et al. 2022), the availability of efficient trapping protocols for the early detection of introduced Agrilus species at entry points is of utmost importance.
Among the numerous trap types developed for Agrilus monitoring programs around the world (e.g., Poland et al. 2019; Imrei et al. 2020a), glue-coated prism traps and Fluon-coated multi-funnel traps are the most commonly adopted and recommended types (European Food Safety Authority et al. 2020; Evans et al. 2020; Silk et al. 2020a). Indeed, both were found to efficiently catch high numbers of Agrilus species and detect (i.e., the ability to catch a single beetle) them at low density (Francese et al. 2008, 2010a, 2011, 2013; Marshall et al. 2010; Petrice et al. 2013; Crook et al 2014; Brown et al. 2017; Rhainds et al. 2017). Other trap types and trapping strategies for Agrilus species have been developed over the years, including double-decker traps (McCullough and Poland 2017), branch-traps (Domingue et al. 2013), modified multi-funnel traps (Imrei et al. 2020b), fan-traps (Grégoire et al. 2022), and girdled or stressed trap trees (McCullough et al. 2009a, 2009b; Mercader et al. 2013). Some of these other trap types have proven effective at detecting A. planipennis at sites with low population densities (e.g., double-decker traps, girdled trap trees), and for capturing a range of other Agrilus species (e.g., branch traps, modified multi-funnel traps), but with the exception of the modified multi-funnel traps (Imrei et al. 2020b), none are commercially available.
Irrespective of the trap type, visual stimuli present on the trap can strongly affect its efficacy. Agrilus species strongly rely on color vision when searching for mates, suitable brood hosts, and food (Lelito et al. 2007; Domingue et al. 2011). Electroretinogram assays demonstrated that both male and female A. planipennis adults are sensitive to green, blue, red and ultraviolet specific wavelengths (Crook et al. 2009). Subsequent field studies showed that green and purple traps are efficient to catch A. planipennis and other Agrilus species (Crook et al. 2009; Francese et al. 2010b, 2011, 2013; Petrice et al. 2013; Poland and McCullough 2014; Kim et al. 2016; Rhainds et al. 2017; Cavaletto et al. 2020). In particular, specific shades of green (i.e., wavelength range 525–540 nm, and reflectance in the 49–67% range) are very attractive for male A. planipennis (Francese et al. 2010b; Domingue et al. 2012; Poland et al. 2019; Parker et al. 2020) presumably because it resembles foliage where they can find both mates and food (Rodriguez-Soana et al. 2007); thus, green is currently the most commonly adopted trap color for trapping Agrilus species (Rassati et al. 2019; Evans et al. 2020; Sallé et al. 2020; Wittman et al. 2021). Another key visual cue used by males to locate females seems to result from the light scattering effect produced by the microstructures present on elytra of adult beetles (Lelito et al. 2008; Domingue et al. 2014). In fact, dead adult beetles or bioreplicated decoys attached on leaves or plastic sticky cards were found to readily evoke the typical descending flights of males occurring when spotting a mate (Lelito et al. 2008; Domingue et al. 2014, 2015). Nonetheless, whether the presence of dead adult beetles or bioreplicated decoys on traps can increase trap efficacy is still unclear.
Besides visual stimuli, Agrilus species rely on olfactory stimuli when searching for mates, suitable host trees, and food (Pureswaran and Poland 2009; Silk et al. 2019; Imrei et al. 2020a). Many species breed in stressed trees and are particularly attracted to volatiles emitted from their hosts, suggesting that decisions regarding host suitability are made prior to alighting on the tree. For example, significantly more A. planipennis (McCullough et al. 2009a, 2009b), A. bilineatus (Weber) (Dunn et al. 1986), and A. anxius Gory (Silk et al. 2020b) or oak-associated Agrilus species (Sallé et al. 2020) were captured on traps placed on, or adjacent to, stressed (i.e., girdled) host trees compared to healthy host trees. Positive responses to extracts of host foliage and/or bark, as well as synthetic blends of compounds identified in host volatiles, have been observed in A. planipennis (Crook et al. 2008; Grant et al. 2010, 2011; Silk et al. 2011), other Agrilus species (Domingue et al. 2013; Vuts et al. 2016), and other species in the subfamily Agrilinae, namely Coraebus fasciatus (Herbst) (Fürstenau et al. 2012) and Coraebus undatus Fabricius (Fürstenau et al. 2015). Among these volatiles, one of the most adopted is the commercially available green leaf volatile, (Z)-3-hexenol. This compound was found to efficiently attract A. planipennis adult beetles both alone and in combination with other lures (De Groot et al. 2008; Grant et al. 2010, 2011; Silk et al. 2011; Ryall et al. 2012; Crook et al. 2014) and is commonly used as the main attractant in surveillance programs targeting the latter species.
In this study we tested whether trapping protocols most commonly adopted for surveillance of A. planipennis were also effective for other Agrilus species. In particular, we carried out a multi-country assessment comparing the efficacy of detecting Agrilus species on: (i) green glue-coated prism traps vs. green Fluon-coated multi-funnel traps when baited with the green leaf volatile (Z)-3-hexenol or left unbaited; and (ii) green multi-panel traps vs. green multi-panel traps baited with dead adult Agrilus beetles (decoys).
Materials and methods
Study sites and general experimental methods
The three trapping trials were carried out in 2021 and 2022 in nine different countries in Europe and North America: Austria, Canada, France, Germany, Italy, Poland, Slovenia, United Kingdom (UK), and Unites States of America (USA) (Table S1). In USA, three states were involved (Massachusetts, Ohio and Kentucky). Not all countries carried out all three trapping trials (Table S1).
Irrespective of the trapping trial, some general criteria were followed. Traps were always set up in a complete randomized block design within an oak-dominated forest, with 20–40 m spacing between treatments and about 60 m among blocks. Using a big-shot catapult (Hughes et al. 2014) or a carbon-fibre telescopic pole, traps were always suspended in the mid to upper canopy, 10–30 m above the ground, preferably in open and sunny spots and avoiding complete shade under branches. Mid-upper canopy was preferred over ground level because several studies showed that species richness and abundance of Agrilus species are generally higher at the canopy level than close to the ground (e.g., Francese et al. 2008; Crook et al. 2008, 2009; Ryall et al. 2012; Rassati et al. 2019; Sallé et al. 2020). Similarly, open sunny spots were preferred over shaded ones because traps more exposed to sunlight tend to catch more adults than those in shaded locations (e.g., Francese et al. 2008; Lyons et al. 2009). Traps were established in mid to late May, and beetles were collected every 2–4 weeks until mid to late August to cover the main flight period of Agrilus species (i.e., 4–8 collections).
Trial 1: Effect of trap design (green prism traps vs. green multi-funnel traps) on Agrilus detection on unbaited traps
Field trials were conducted in 2021 in Canada, France, Germany, Italy, Slovenia, UK and USA (Massachusetts and Ohio) (Table S1). Green (540 nm, 49% reflectance; see Wittman et al. 2021), glue-coated prism traps were purchased from Andermatt Group (Grossdietwil, Switzerland and Fredericton, New Brunswick, Canada) (Fig. S1A), whereas green (530 nm, 49% reflectance; described by Francese et al. 2011) multi-funnel traps were purchased from ChemTica Internacional (Santo Domingo, Costa Rica) (Fig. S1B). Multi-funnel traps were pre-treated with a 50% solution of Fluon as a trap coating because of its ability to increase trap catch of woodboring beetles (Graham and Poland 2012; Allison et al. 2016) including A. planipennis (Lyons et al. 2012; Francese et al. 2013). Each trap type was replicated 10 times in all but two of the countries (i.e., Slovenia and France) (Table S1). The collecting cup of the multi-funnel traps was filled with approximately 200 ml of 50% propylene glycol solution either in water or in a solution as a pre-mixed marine / RV antifreeze in most of the countries. However, in a few cases either a saturated salt solution (table salt in water) or ventilated cups for dry trapping supplied with an insecticide net were used. The contents of the collecting cups from the multi-funnel traps were transferred to labelled containers and transported back to the laboratory for further analysis (the trap collection cup was reset with fresh solution following each collection). The green prism traps were examined at each collection date and any Agrilus beetles captured on the glue surface of the trap were picked off with forceps and transferred to a labelled collection vial or bag.
Trial 2: Effect of trap design (green prism traps vs. green multi-funnel traps) and (Z)-3-hexenol lure (present vs. absent) on Agrilus detection
Field trials were conducted in 2022 in the same sites and countries as trial 1 in 2021 (Table S1), except for the addition of a site in each of Austria and Sweden and the replacement of the Ohio site with a site in Kentucky in USA. Green prism traps and green multi-funnel traps were obtained from the same companies as in 2021 (i.e., Andermatt and ChemTica Internacional). (Z)-3-hexenol lure pouches were purchased from Synergy Semiochemicals Corporation (Delta, BC, Canada) or purchased together with the traps at Andermatt Group (i.e., Germany), and had a release rate of 50 mg/day at 25ºC. Each treatment was replicated 5–10 times depending on the country (Table S1). Traps were checked as explained above for trial 1.
Trial 3: Effect of decoys on Agrilus detection in green multi-panel traps
Field trials were conducted in 2022 in Canada, France, Italy, Poland, UK, and USA (Massachusetts and Ohio) (Table S1). Green multi-panel traps (i.e., Synergy Semiochemical Multitrap panel trap) were supplied by Synergy Semiochemicals Corporation (Delta, BC, Canada). Three individual dead Agrilus adult specimens were glued onto each panel surface of the multi-panel trap (i.e., 18 individuals/trap) to serve as decoys (Fig. S1C, D). Each country used as decoys the preserved cadavers of the most common Agrilus beetle species captured in the 2021 field trial, namely A. olivicolor Kiesenwetter in Italy, A. laticornis (Illiger) in France and UK, A. biguttatus (Fabricius) in Poland, A. crinicornis Horn in Canada and A. bilineatus in USA. We decided to bait traps with dead adults of a single species and test the effect on multiple species because previous studies showed that the light-scattering effect produced by physical micro-structures present on Agrilus species body is not species-specific (Lelito et al. 2008; Domingue et al. 2011, 2014). Each treatment was replicated 10 times in all countries except France (Table S1). The collecting cup of the multi-panel traps was filled with approximately 200 ml of 50% propylene glycol solution either in water or in a solution as a pre-mixed marine / RV antifreeze in most of the countries, except Canada where a saturated salt solution was used.
Statistical analyses
The effects of trap design (trial 1), trap design, lure and their interaction (trial 2), and decoy presence (trial 3), were tested using generalized linear mixed models (GLMMs). Models were fitted with a Poisson distribution, or negative binomial distribution when data were over dispersed, using a log link-function. We opted for GLMMs over classic ANOVA given the non-normality and the correlated and longitudinal structure of the data. For each country and trial, species richness (i.e., total number of Agrilus species), total abundance (i.e., total number of Agrilus individuals), and the abundance of each Agrilus species (when represented by at least 10 individuals) were considered as response variables. In all cases, we treated the data collected from each trap and sampling period as a distinct statistical unit. This approach ensures more accurate parameter estimates and better model fit compared to pooling data from different sampling rounds. The block identity and the trap identity within each block were included in the models as random factors to account for the temporal pseudoreplication (repeated measures on each trap) and for the nested design of the trials. For trial 2, pairwise multiple comparisons were run using post-hoc tests with Holm correction of p-values. All the analyses were carried out in R (R Core Team 2021). Models were fitted using the ‘glmmTMB’ package (Brooks et al. 2022). Pairwise comparisons were run using the ‘emmeans’ package (Lenth et al. 2022). Models were checked for overdispersion, residual distribution, and residual temporal and spatial autocorrelation using the ‘DHARMa’ package (Hartig 2022).
Results
General results
A total of 23,481 individuals from 45 Agrilus species were caught (Table S2). Six species were represented by more than 1,000 individuals; among them, A. laticornis was the most abundant species (6,665 individuals), followed by A. olivicolor (4,136), A. angustulus (Illiger) (3,158), A. obscuricollis Kiesenwetter (2,069), A. hastulifer Ratzeburg (1,296), and A. crinicornis Horn (1,240). By contrast, 21 species were represented by less than 10 individuals (Table S2). Three countries were represented by more than 2,000 individuals, from 10 or more different species, i.e., France (8,048), Austria (5,521) and Italy (2,449). Only Poland and Sweden, in which only one trapping experiment was conducted, were represented by less than 200 individuals (Table S2). In general, species richness at the sampling sites tended to decrease with increasing latitude.
Trial 1: Effect of trap design (green prism traps vs. green multi-funnel traps) on Agrilus detection on unbaited traps
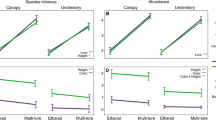
Trap type significantly affected both species richness and abundance of Agrilus species in France (Fig. 1A, I), Italy (Fig. 1C, K), UK (Fig. 1E, M) and Ohio (Fig. 1H, P), where green prism traps always outperformed green multi-funnel traps. On the contrary, no significant effect of trap type was observed in Germany (Fig. 1B, J), Slovenia (Fig. 1D, L), Canada (Fig. 1F, N), or Massachusetts (Fig. 1G, O) neither for species richness nor abundance.
Means from the generalized linear mixed models (Poisson or negative binomial distribution with a log link-function) testing the effect of trap type on the mean number of species (i.e., species richness) and mean number of individuals (i.e., abundance) of Agrilus species collected per trap per sampling period in each of the eight different countries where the trial was carried out in 2021 (trial 1). Trap type: PT = green prism traps; MF = green multi-funnel traps. P-values: * = 0.05–0.01; ** = 0.01–0.001; *** = < 0.001; ns = not significant (> 0.05). Bars are green for countries where a significant effect of trap type was found and grey for countries where the effect was not significant. Error bars indicate the 95% positive confidence interval. Note the nonlinearity of the y-axis (due to log-link function in the models)
At the species level, trap type significantly affected abundance of 14 of the 33 Agrilus species collected in 2021, even though the effect was not evident in all countries where the species were collected (Table 1). In countries where the effect of trap type was significant, A. angustulus, A. arcuatus (Say), A. celti Knull, A. convexicollis Redtenbacher, A. curtulus Mulsant & Rey, A. hastulifer, A. laticornis, A. masculinus Horn, A. obscuricollis, A. olivicolor, A. putillus Say, and A. viridis L. were caught significantly more in green prism traps than green multi-funnel traps, while the opposite trend was observed only for A. bilineatus (Table 1). The only species for which the effect of trap type was not consistent between countries was A. sulcicollis Lacordaire (i.e., better performance of green prism traps in France and green multi-funnel traps in Slovenia) (Table 1).
For species composition, most Agrilus species were collected by both trap types (i.e., 23), seven exclusively by green prism traps and three exclusively by green multi-funnel traps (Fig. S2A). When singletons and doubletons were excluded, no species was found exclusively in green multi-funnel traps, two exclusively in green prism traps, and 21 in both trap types (Fig. S2A).
Trial 2: Effect of trap design (green prism traps vs. green multi-funnel traps) and (Z)-3-hexenol lure (present vs. absent) on Agrilus detection
Both species richness and abundance of Agrilus species were affected by trap type in Austria (Figs. 2A, 3A), France (Figs. 2B, 3B), Italy (Figs. 2D, 3D), Slovenia (Figs. 2E, 3E), Sweden (Figs. 2F, 3F), UK (Figs. 2G, 3G), Kentucky (Figs. 2I, 3I) and Massachusetts (Figs. 2J, 3J), where green prism traps outperformed green multi-funnel traps in all sites but Massachusetts, where the opposite trend was observed. On the contrary, there were no significant effects of trap type on species richness or abundance in Canada (Figs. 2H, 3H) and Germany (Figs. 2C, 3C). The (Z)-3-hexenol lure and the interaction between trap type and lure never affected species richness or abundance.
Means from the generalized linear mixed models (Poisson or negative binomial distribution with a log link-function) testing the effect of trap type, (Z)-3-hexenol lure and their interaction on the mean number of species (i.e., species richness) of Agrilus species collected per trap per sampling period in each of the ten different countries where the trial was carried out in 2022 (trial 2). (Z)-3-hexenol lure: UN = unbaited; BA = baited. Trap type: PT = green prism traps; MF = green multi-funnel traps. P-values: * = 0.05–0.01; ** = 0.01–0.001; *** = < 0.001. Bars are green for countries where a significant effect of either trap type, lure or their interaction was found and grey for countries where the effect of the latter variables were not significant. Error bars indicate the 95% positive confidence interval. Note the nonlinearity of the y-axis (due to log-link function in the models)
Means from the generalized linear mixed models (Poisson or negative binomial distribution with a log link-function) testing the effect of trap type, (Z)-3-hexenol lure and their interaction on the mean number of individuals (i.e., abundance) of Agrilus species collected per trap per sampling period in each of the ten different countries where the trial was carried out in 2022 (trial 2). (Z)-3-hexenol lure: UN = unbaited; BA = baited. Trap type: PT = green prism traps; MF = green multi-funnel traps. P-values: * = 0.05–0.01; ** = 0.01–0.001; *** = < 0.001. Bars are green for countries where a significant effect of either trap type, lure or their interaction was found and grey for countries where the effect of the latter variables were not significant. Error bars indicate the 95% positive confidence interval. Note the nonlinearity of the y-axis (due to log-link function in the models)
Trap type also significantly affected abundance of 15 out of the 36 Agrilus species collected in trial 2, but the effect was not evident in all countries where the species were collected (Table 2). In countries where the effect of trap type was significant, A. angustulus, A. celti, A. convexicollis, A. geminatus (Say), A. graminis Kiesenwetter, A. hastulifer, A. laticornis, A. lecontei Saunders, A. obscuricollis, A. olivicolor, A. putillus, A. sulcicollis, and A. viridis were caught significantly more in green prism traps than green multi-funnel traps, while the opposite trend was observed only for A. arcuatus and A. biguttatus (Table 2).
For species composition, most Agrilus species were collected by all treatments irrespective whether singletons and doubletons were included or excluded (i.e., 21) (Fig. S2B). Only a few species were instead found to be exclusive to certain treatments, and this value was even lower when singletons and doubletons were excluded (Fig. S2B).
Trial 3: Effect of decoys on Agrilus detection in green multi-panel traps
The presence of adult Agrilus decoys on green multi-panel traps did not affect species richness and abundance of Agrilus species in any country (Table 3). At the species level, a significant effect of the Agrilus decoys was observed only on 3 of the 25 Agrilus species collected, i.e., A. hastulifer in France, A. sulcicollis in Poland, and A. bilineatus in Ohio (Table 3). Traps baited with decoys of A. laticornis collected fewer A. hastulifer than did unbaited traps. Similarly, traps baited with decoys of A. sulcicollis collected fewer A. sulcicollis than did unbaited traps. Conversely, traps baited with A. bilineatus decoys collected more A. geminatus than did unbaited traps (Table 3).
For species composition, most Agrilus species were collected by both unbaited and Agrilus decoy-baited multi-panel traps (i.e., 21), two exclusively by unbaited multi-panel traps and two exclusively by decoy-baited multi-panel traps (Fig. S2C). When singletons and doubletons were excluded, no species was found exclusively in unbaited multi-panel traps, one exclusively in decoy-baited multi-panel traps, and 21 in both trap types (Fig. S2C).
Discussion
More than 30 Agrilus species have been introduced outside their native range, including European species introduced into North American forests (e.g., A. sulcicollis) and North American species introduced into European forests (e.g., A. bilineatus) (Hızal and Arslangündoğdu 2018). For this reason, the availability of efficient trapping protocols that can be integrated in surveillance programs is of utmost importance (Poland and Rassati 2019; Nahrung et al. 2023). Through extensive trapping trials in Europe and North America, we showed that trap types commonly used for early detection and monitoring of A. planipennis (i.e., green prism traps and multi-funnel traps) are reliable also for detection of other Agrilus species, even though depending on the scope of the surveillance programs one trap design might be more suitable than the other. In addition, our trials indicated that baiting traps with (Z)-3-hexenol or adult beetle decoys does not seem to increase general trap efficacy.
Trap design significantly affected both species richness and abundance of Agrilus species in several of the countries where the trapping experiments were carried out. In particular, when a significant effect was found, green prism traps outperformed green multi-funnel traps in all cases but one. A very similar pattern was observed also at the species level. The efficacy of colored prism traps in attracting Agrilus species is well known (Francese et al. 2008; Petrice et al. 2013; Poland et al. 2019), and this trap design has been used for monitoring the spread of the emerald ash borer in USA and Canada. Nonetheless, the higher efficacy of prism traps compared to multi-funnel traps that we found was rarely observed in previous studies (e.g., Francese et al. 2005), which often highlighted a similar efficacy of the two trap designs or even a higher efficacy of multi-funnel traps (Francese et al. 2011, 2016; Crook et al. 2014; Poland et al. 2019). We suggest three possible reasons why green prism traps generally performed better than green multi-funnel traps in our study. First, previous Agrilus trapping studies have primarily focused on A. planipennis, while our study assessed the responses of other Agrilus species which may display different behaviors and preferences. Secondly, compared to multi-funnel traps, the glue-coated prism traps may have retained a greater proportion of beetles that alighted on the trap surface. Although Fluon-coating increases the capture rate of beetles in multi-funnel and intercept panel traps (Allison et al. 2011; Graham and Poland 2012), studies suggest that a proportion of beetles that contact the surface of multi-funnel traps do not fall into the collecting cup (Allison et al. 2014). Thirdly, adult Agrilus individuals landing on prism traps remain attached to the panel and could act as positive visual cues for other Agrilus species (Domingue et al. 2014, 2015) whereas this does not occur in multi-funnel traps.
Besides overall efficacy, other factors to consider when deciding to use prism traps or multi-funnel traps for Agrilus surveys include the need to reuse traps for multiple years, ease of deployment, and the overall goal of the trapping program. Prism traps are discarded after one field season while multi-funnel traps can be reusable for many years if properly maintained (Francese et al. 2011). Additionally, at each trap check prism traps must be thoroughly examined in the field, which is time consuming particularly in bad weather, while multi-funnel traps are simply emptied by pouring the content of the collector cup in a plastic container (Francese et al. 2011). Prism traps may also be at a disadvantage in areas with high dust and pollen counts. This debris can coat the surface of the trap and render the glue inefficient. Glue-coated surfaces in hot, sunny locations can also slough off of the trap surface taking samples with it (Francese et al 2011). Furthermore, removal of beetles from prism traps and subsequent treatments to remove the glue from their body can damage them, negatively affecting morphological identification. Several studies showed that multi-funnel traps also efficiently catch longhorn beetles (Rassati et al. 2019), bark and ambrosia beetles (Marchioro et al. 2020; Thurston et al. 2022), and leaf-dwelling beetles (Sallé et al. 2020), while whether this is true also for sticky prism traps is still unclear (e.g., Skvarla and Holland 2011). Thus, despite our results suggest that for generic surveillance programs targeting Agrilus species green prism traps might be preferred over green multi-funnel traps, the latter trap design might be preferred when targeting Agrilus species that do not show preferences for a specific trap design as found for more than half of the species caught in this study.
We also found that the addition of the green leaf volatile (Z)-3-hexenol did not affect species richness, total abundance, or abundance of any Agrilus species in any of the countries where the trapping trials were carried out. This result is in contrast to the results of previous studies showing that (Z)-3-hexenol significantly increased trap catches of A. planipennis (De Groot et al. 2008; Grant et al. 2010, 2011; Silk et al. 2011), A. auroguttatus (Coleman et al. 2014), and A. sulcicollis (Domingue et al. 2014). However, lack of positive response to (Z)-3-hexenol has previously been observed for A. anxius (Silk et al. 2019), A. angustulus, A. graminis, A. laticornis, and A. obscuricollis (Domingue et al. 2013). Because so many of the same foliar and cortical volatile compounds are shared among different genera of trees, it follows that Agrilus species and other beetles that use olfactory cues during host selection, likely respond most positively to particular blends and relative ratios of volatiles emitted from suitable hosts, more so than any individual compound. Indeed, van Wijk et al. (2011) suggested that the blend of compounds emitted from a suitable host is perceived as a distinct odor, different from its individual components.
Our field trials also indicated that the inclusion of adult beetle decoys on trap panels did not affect species richness and total abundance of most Agrilus species; however there did appear to be an effect for three individual species. Two species, A. laticornis and A. sulcicollis, were seemingly repelled by the presence of adult beetles of a different or the same species, respectively, while A. geminatus seemed to be attracted by the presence of adult beetles of A. bilineatus. The general lack of an effect might be due to the small number of Agrilus individuals (i.e., three) that we glued on each trap panel. It is possible that any attractive effects of the decoys were obviated by stronger combined effects of the green color and the large size of the multi-panel traps, contrary to what was observed when smaller traps (i.e., branch-traps) were used (Lelito et al. 2008; Domingue et al. 2014). The few significant repellent or attractive effects observed at the species level might be due to greater sensitivity of certain species to light scattering effects produced by adult beetles. In a previous study, only individuals of A. sulcicollis were significantly more attracted to traps baited with decoys than unbaited traps, while no effect was observed for A. angustulus and A. laticornis (Domingue et al. 2013). In addition, different species might display different mating behavior (Lelito et al. 2011), leading to heterogeneous responses to decoy-baited traps.
Trapping programs are commonly carried out at entry points or other high-risk sites (e.g., Rassati et al. 2015a,b; Rabaglia et al. 2019; Mas et al. 2023), but their efficacy strongly depends on the availability of efficient trapping protocols for the target group of species. Our study highlighted that both green prism traps and green multi-funnel traps can be considered reliable trap models to monitor Agrilus species, but also that the commonly used (Z)-3-hexenol is not universally attractive to all Agrilus species. Given that host volatiles and pheromones, especially when combined, can be used to increase trap attractiveness (Parker et al. 2020), more efforts should be done to discover new species-specific and generic lures to be used in surveillance programs for Agrilus species. A direct comparison of the three trap designs tested in this study would also be useful to understand whether green multi-panel traps can be eventually more efficient than green prism-traps and/or green multi-funnel traps. Despite the lack of an effect of adult beetle decoys glued on traps, future studies should further investigate whether it is possible to reproduce the light scattering effect on trap panels, for example using more adult beetles as decoys or exploiting technological advances to reproduce the microstructures present on beetle elytra on surfaces that can be then attached to trap panels. Traps of other colour wavelengths (and designs) should be also further evaluated (Imrei et al. 2020a). Finally, additional multi-country experiments involving more continents should be conducted. As shown in this and other studies (e.g., Roques et al. 2023), sharing information from trapping experiments conducted on different continents provides us with direct information on the efficacy of traps, lures, and other factors for detecting target species potentially at risk of introduction to our respective continents.
Author’s contributions
DW, JAF, JS, and DR conceived the study; DR and GS wrote the first draft of the manuscript; BH, GS, GCa, EB, SD, EKLF, EGB, SMD, DLP, EF, CH, JMG, RP, KS, EG, TH, MdG, AS, DW, AE, GH, WH, JC conducted field experiments; GCu, EF, MG, CK, JMG, KVR, EG, AS, DW, AE, JC, WH identified beetle specimens; GS, DR and MdG analyzed the data; BH, DW, DR, MdG, JS, JAF, AS, GH, AMR acquired funds. All authors contributed to and approved the manuscript.
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding authors on reasonable request.
Code availability
Not applicable.
References
Allison JD, Wood Johnson C, Meeker JR, Strom BL, Butler SM (2011) Effect of aerosol surface lubricants on the abundance and richness of selected forest insects captured in multiple-funnel and panel traps. J Econ Entomol 104:1258–1264
Allison JD, Bhandari BD, McKenney JL, Millar JG (2014) Design factors that influence the performance of flight intercept traps for the capture of longhorned beetles (Coleoptera: Cerambycidae) from the subfamilies Lamiinae and Cerambycinae. PLoS ONE 9:e93203
Allison JD, Graham EE, Poland TM, Strom BL (2016) Dilution of fluon before trap surface treatment has no effect on longhorned beetle (Coleoptera: Cerambycidae) captures. J Econ Entomol 109:1215–1219
Brockerhoff EG, Liebhold AM (2017) Ecology of forest insect invasions. Biol Invasions 19:3141–3159
Brooks M, Bolker B, Kristensen K, Maechler M, Magnusson A, McGillycuddy M, Skaug H, Nielsen A, Berg C, Van Bentham K, Sadat N, Lüdecke D, Lenth R, O’Brien J, Geyer CJ, Jagan M, Wiernik B, Stouffer DB (2022) Generalized linear mixed models using template model builder. R package ‘glmmTMB’, version 1.1.5
Brown N, Inward DJG, Jeger M, Denman S (2015) A review of Agrilus biguttatus in UK forests and its relationship with acute oak decline. Forestry 88:53–63
Brown N, Jeger M, Kirk S, Williams D, Xu X, Pautasso M, Denman S (2017) Acute oak decline and Agrilus biguttatus: the co-occurrence of stem bleeding and D-shaped emergence holes in Great Britain. Forests 8:87
Brück-Dyckhoff C, Petercord R, Schopf R (2019) Vitality loss of European beech (Fagus sylvatica L.) and infestation by the European beech splendour beetle (Agrilus viridis L., Buprestidae, Coleoptera). For Ecol Manag 432:150–156
Cavaletto G, Faccoli M, Marini L, Spaethe J, Magnani G, Rassati D (2020) Effect of trap color on captures of bark-and wood-boring beetles (Coleoptera; Buprestidae and Scolytinae) and associated predators. Insects 11:749
Coleman TW, Chen Y, Graves AD, Hishinuma SM, Grulke NE, Flint ML, Seybold SJ (2014) Developing monitoring techniques for the invasive goldspotted oak borer (Coleoptera: Buprestidae) in California. Environ Entomol 43:729–743
Crook DJ, Khrimian A, Francese JA, Fraser I, Poland TM, Mastro VC (2008) Development of a host-based semiochemical lure for trapping emerald ash borer Agrilus planipennis (Coleoptera: Buprestidae). Environ Entomol 37:356–365
Crook DJ, Francese JA, Zylstra KE, Fraser I, Sawyer AJ, Bartels DW, Lance DR, Mastro VC (2009) Laboratory and field response of the emerald ash borer (Coleoptera: Buprestidae), to selected regions of the electromagnetic spectrum. J Econ Entomol 102:2160–2169
Crook DJ, Francese JA, Rietz ML, Lance DR, Hull-Sanders HM, Mastro VC, Silk PJ, Ryall KL (2014) Improving detection tools for emerald ash borer (Coleoptera: Buprestidae): comparison of multifunnel traps, prism traps, and lure types at varying population densities. J Econ Entomol 107:1496–1501
De Groot P, Grant GG, Poland TM, Scharbach R, Buchan L, Nott RW, Macdonald L, Pitt D (2008) Electrophysiological response and attraction of emerald ash borer to green leaf volatiles (GLVs) emitted by host foliage. J Chem Ecol 34:1170–1179
Domingue MJ, Csóka G, Tóth M, Vétek G, Pénzes B, Mastro V, Baker TC (2011) Field observations of visual attraction of three European oak buprestid beetles toward conspecific and heterospecific models. Entomol Exp Appl 140:112–121
Domingue MJ, Lelito JP, Fraser I, Mastro VC, Tumlinson JH, Baker TC (2012) Visual and chemical cues affecting the detection rate of the emerald ash borer in sticky traps. J Appl Entomol 137:77–87
Domingue MJ, Imrei Z, Lelito JP, Muskovits J, Janik G, Csóka G, Mastro VC, Baker TC (2013) Trapping of European buprestid beetles in oak forests using visual and olfactory cues. Entomol Exp Appl 148:116–129
Domingue MJ, Lakhtakia A, Pulsifer DP, Hall LP, Badding JV, Bischof JL, Martín-Palma RJ, Imrei Z, Janik G, Mastro VC, Hazen M, Baker T (2014) Bioreplicated visual features of nanofabricated buprestid beetle decoys evoke stereotypical male mating flights. Proc Natl Acad Sci USA 111:14106–14111
Domingue MJ, Pulsifer DP, Lakhtakia A, Berkebile J, Steiner KC, Lelito JP, Hall LP, Baker TC (2015) Detecting emerald ash borers (Agrilus planipennis) using branch traps baited with 3D-printed beetle decoys. J Pest Sci 88:267–279
Dunn JP, Kimmerer TW, Nordin GL (1986) The role of host tree condition in attack of white oaks by the twolined chestnut borer, Agrilus bilineatus (Weber) (Coleoptera: Buprestidae). Oecologia 70:596–600
European Food Safety Authority (EFSA), Schans J, Schrader G, Delbianco A, Graziosi I, Vos S (2020) Pest survey card on Agrilus planipennis. EFSA Support Publ 17:1945E
Evans HF, Williams D, Hoch G, Loomans A, Marzano M (2020) Developing a European Toolbox to manage potential invasion by emerald ash borer (Agrilus planipennis) and bronze birch borer (Agrilus anxius), important pests of ash and birch. Forestry 93:187–196
Francese JA, Mastro VC, Oliver JB, Lance DR, Youssef N, Lavallee SG (2005) Evaluation of colors for trapping Agrilus planipennis (Coleoptera: Buprestidae). J Entomol Sci 40:93–95
Francese JA, Oliver JB, Fraser I, Lance DR, Youssef N, Sawyer AJ, Mastro VC (2008) Influence of trap placement and design on capture of the emerald ash borer (Coleoptera: Buprestidae). J Econ Entomol 101:1831–1837
Francese JA, Fraser I, Rietz ML, Crook DJ, Lance DR, Mastro VC (2010a) Relation of color, size, and canopy placement of prism traps in determining capture of emerald ash borer (Coleoptera: Buprestidae). Can Entomol 142:596–600
Francese JA, Crook DJ, Fraser I, Lance DR, Sawyer AJ, Mastro VC (2010b) Optimization of trap color for emerald ash borer (Coleoptera: Buprestidae). J Econ Entomol 103:1235–1241
Francese JA, Fraser I, Lance DR, Mastro VC (2011) Efficacy of multifunnel traps for capturing emerald ash borer (Coleoptera: Buprestidae): effect of color, glue, and other trap coatings. J Econ Entomol 104:901–908
Francese JA, Rietz ML, Mastro VC (2013) Optimization of multifunnel traps for emerald ash borer (Coleoptera: Buprestidae): influence of size, trap coating, and color. J Econ Entomol 106:2415–2423
Francese JA, Sorensen B, Booth EG, Hilszczański J, Schwartzberg E, Youssef N, Lopez VM, Furtado M, Spichiger SE, Barringer L, Hansen J, Oliver, Lance DR (2016) Using purple and green multi-funnel traps as general survey tools for wood-boring beetles. Otis Laboratory Accomplishments 2016 https://www.aphis.usda.gov/plant_health/science-technology/otis-lab-accomps-2016.pdf, pp 68–69. Accessed 21 July 2023
Fürstenau B, Rosell G, Guerrero A, Quero C (2012) Electrophysiological and behavioral responses of the black-banded oak borer, Coroebus florentinus, to conspecific and host-plant volatiles. J Chem Ecol 36:378–388
Fürstenau B, Quero C, Riba JM, Rosell G, Guerrero A (2015) Field trapping of the flathead oak borer Coroebus undatus (Coleoptera: Buprestidae) with different traps and volatile lures. Insect Sci 22:139–149
Graham EE, Poland TM (2012) Efficacy of fluon conditioning for capturing cerambycid beetles in different trap designs and persistence on panel traps over time. J Econ Entomol 105:395–401
Grant GG, Ryall KL, Lyons DB, Abou-Zaid MM (2010) Differential response of male and female emerald ash borers (Col., Buprestidae) to (Z)-3-hexenol and manuka oil. J Appl Entomol 134:26–33
Grant GG, Poland TM, Ciaramitaro T, Barry Lyons D, Jones GC (2011) Comparison of male and female emerald ash borer (Coleoptera: Buprestidae) responses to phoebe oil and (Z)-3-hexenol lures in light green prism traps. J Econ Entomol 104:173–179
Grégoire JC, Caiti E, Hasbroucq S, Molenberg JM, Willenz S (2022) When the beetles hit the fan: the fan-trap, an inexpensive, light and scalable insect trap under a creative commons license, for monitoring and experimental use. Insects 13:1122
Hartig F (2022) Residual diagnostics for hierarchical (multi-level/mixed) regression models. R package ‘DHARMa’, version 0.4.6.
Hızal E, Arslangündoğdu Z (2018) The first record of two-lined chestnut borer Agrilus bilineatus (Weber, 1801) (Coleoptera: Buprestidae) from Europe. Entomol News 127:333–335
Hughes CC, Johns RC, Sweeney JD (2014) A technical guide to installing beetle traps in the upper crown of trees. J Acad Entomol Soc 10:12–18
Imrei Z, Lohonyai Z, Csóka G, Muskovits J, Szanyi S, Vétek G, Fail J, Tóth M, Domingue MJ (2020a) Improving trapping methods for buprestid beetles to enhance monitoring of native and invasive species. Forestry 93:254–264
Imrei Z, Lohonyai Z, Muskovits J, Matula E, Vuts J, Fail J, Gould PJL, Birkett MA, Tóth M, Domingue MJ (2020b) Developing a non-sticky trap design for monitoring jewel beetles. J Appl Entomol 144:224–231
Jendek E (2016) Taxonomic, nomenclatural, distributional and biological study of the genus Agrilus (Coleoptera: Buprestidae). J Insect Biodivers 4:1–57
Jendek E, Grebennikov VV (2023) Summary of native geographic distribution of all 3341 species of the most speciose animal genus Agrilus (Coleoptera: Buprestidae). J Insect Biodivers 39:32–78
Kelnarova I, Jendek E, Grebennikov VV, Bocak L (2019) First molecular phylogeny of Agrilus (Coleoptera: Buprestidae), the largest genus on Earth, with DNA barcode database for forestry pest diagnostics. Bull Entomol Res 109:200–211
Kim SHS, Trammel CE, Lewis BA, Johnson DT (2016) Comparison of color attractiveness for Agrilus ruficollis (Coleoptera: Buprestidae): potential for a simple trap. J Econ Entomol 109:1799–1806
Klooster WS, Gandhi KJ, Long LC, Perry KI, Rice KB, Herms DA (2018) Ecological impacts of emerald ash borer in forests at the epicenter of the invasion in North America. Forests 9:250
Kovacs KF, Haight RG, McCullough DG, Mercader RJ, Siegert NW, Liebhold AM (2010) Cost of potential emerald ash borer damage in US communities, 2009–2019. Ecol Econ 69:569–578
Lelito JP, Fraser I, Mastro VC, Tumlinson JH, Böröczky K, Baker TC (2007) Visually mediated ‘paratrooper copulations’ in the mating behavior of Agrilus planipennis (Coleoptera: Buprestidae), a highly destructive invasive pest of North American ash trees. J Insect Behav 20:537–552
Lelito JP, Fraser I, Mastro VC, Tumlinson JH, Baker TC (2008) Novel visual-cue-based sticky traps for monitoring of emerald ash borers, Agrilus planipennis (Col., Buprestidae). J Appl Entomol 132:668–674
Lelito JP, Domingue MJ, Fraser I, Mastro VC, Tumlinson JH, Baker TC (2011) Field investigation of mating behaviour of Agrilus cyanescens and Agrilus subcinctus. Can Entomol 143:370–379
Lenth RV, Buerkner P, Herve M, Love J, Miguez F, Riebl H, Singmann H (2022) Estimated marginal means, aka least-squares means. R package ‘emmeans’, version 1.8.2.
Lyons DB, de Groot P, Jones GC, Scharbach R (2009) Host selection by Agrilus planipennis (Coleoptera: Buprestidae): inferences from sticky-band trapping. Can Entomol 141:40–52
Lyons DB, Lavallee R, Kyei-Poku G, van Frankenhuyzen K, Johny S, Guertin C, Francese JA, Jones GC, Blais M (2012) Towards the development of an autocontamination trap to manage populations of emerald ash borer (Coleoptera: Buprestidae) with native entomopathogenic fungus Beauveria bassiana. J Econ Entomol 105:1929–1939
Marchioro M, Rassati D, Faccoli M, Van Rooyen K, Kostanowicz C, Webster V, Mayo P, Sweeney J (2020) Maximizing bark and ambrosia beetle (Coleoptera: Curculionidae) catches in trapping surveys for longhorn and jewel beetles. J Econ Entomol 113:2745–2757
Marshall JM, Storer AJ, Fraser I, Mastro VC (2010) Efficacy of trap and lure types for detection of Agrilus planipennis (Col., Buprestidae) at low density. J Appl Entomol 134:296–302
Mas H, Santoiemma G, Lencina JL, Gallego D, Pérez-Laorga E, Ruzzier E, Rassati D (2023) Investigating beetle communities in and around entry points can improve surveillance at national and international scale. NeoBiota 85:145–165
McCullough DG, Poland TM (2017) Building double-decker traps for early detection of emerald ash borer. J Vis Exp 128:e55252
McCullough DG, Poland TM, Anulewicz AC, Cappaert D (2009a) Emerald ash borer (Coleoptera: Buprestidae) attraction to stressed or baited ash trees. Environ Entomol 38:1668–1679
McCullough DG, Poland TM, Cappaert D (2009b) Attraction of the emerald ash borer to ash trees stressed by girdling, herbicide treatment, or wounding. Can J For Res 39:1331–1345
Mercader RJ, McCullough DG, Bedford JM (2013) A comparison of girdled ash detection trees and baited artificial traps for Agrilus planipennis (Coleoptera: Buprestidae) detection. Environ Entomol 42:1027–1039
Meurisse N, Rassati D, Hurley BP, Brockerhoff EG, Haack RA (2019) Common pathways by which non-native forest insects move internationally and domestically. J Pest Sci 92:13–27
Muzika RM, Liebhold AM, Twery MJ (2000) Dynamics of twolined chestnut borer Agrilus bilineatus as influenced by defoliation and selection thinning. Agric For Entomol 2:283–289
Nahrung HF, Liebhold AM, Brockerhoff EG, Rassati D (2023) Forest insect biosecurity: processes, patterns, predictions, pitfalls. Annu Rev Entomol 68:211–229
Parker K, Ryall K, Aukema BH, Silk P (2020) Early detection of Agrilus planipennis: investigations into the attractive range of the sex pheromone (3Z)-lactone. Entomol Exp Appl 168:166–173
Perish PK, Shanovich HN, Koch AN, Lindsey AR, Aukema BH (2023) The hazel stem borer, Agrilus pseudocoryli (Coleoptera: Buprestidae), as a pest of hybrid hazelnuts. J Econ Entomol 116:496–504
Petrice TR, Haack RA, Poland TM (2013) Attraction of Agrilus planipennis (Coleoptera: Buprestidae) and other buprestids to sticky traps of various colors and shapes. Great Lakes Entomol 46:13–30
Poland TM, McCullough DG (2014) Comparison of trap types and colors for capturing emerald ash borer adults at different population densities. Environ Entomol 43:157–170
Poland TM, Rassati D (2019) Improved biosecurity surveillance of non-native forest insects: a review of current methods. J Pest Sci 92:37–49
Poland TM, Petrice TR, Ciaramitaro TM (2019) Trap designs, colors, and lures for emerald ash borer detection. Front For Glob Change 2:80
Pureswaran DS, Poland TM (2009) Host selection and feeding preference of Agrilus planipennis (Coleoptera: Buprestidae) on ash (Fraxinus species). Environ Entomol 38:757–765
Pureswaran D, Meurisse N, Rassati D, Liebhold AM, Faccoli M (2022) Climate change and invasion by non-native bark and ambrosia beetles. In: Hostetter RW, Gandhi K (eds) Bark beetle management, ecology and climate change. Academic Press, New York, pp 3–30
R Core Team (2021). R: a language and environment for statistical computing. https://www.r-project.org/
Rabaglia RJ, Cognato AI, Hoebeke ER, Johnson CW, LaBonte JR, Carter ME, Vlach JJ (2019) Early detection and rapid response: a 10-year summary of the USDA Forest Service program of surveillance for non-native bark and ambrosia beetles. Am Entomol 65:29–42
Rassati D, Faccoli M, Petrucco Toffolo E, Battisti A, Marini L (2015a) Improving the early detection of alien wood-boring beetles in ports and surrounding forests. J Appl Ecol 52:50–58
Rassati D, Faccoli M, Marini L, Haack RA, Battisti A, Petrucco Toffolo E (2015b) Exploring the role of wood waste landfills in early detection of non-native wood-boring beetles. J Pest Sci 88:563–572
Rassati D, Marini L, Marchioro M, Rapuzzi P, Magnani G, Poloni R, Di Giovanni F, Mayo P, Sweeney J (2019) Developing trapping protocols for wood-boring beetles associated with broadleaf trees. J Pest Sci 92:267–279
Rhainds M, Kimoto T, Galko J, Nikolov C, Ryall K, Brodersen G, Webster V (2017) Survey tools and demographic parameters of Slovakian Agrilus associated with beech and poplar. Entomol Exp Appl 162:328–335
Rodriguez-Soana CR, Miller JR, Poland TM, Kuhn TM, Otis GW, Turk T, Ward DL (2007) Behaviors of adult Agrilus planipennis (Coleoptera: Buprestidae). Gt Lakes Entomol 40:1–15
Roques A, Ren L, Rassati D, Shi J, Akulov E, Audsley N, Auger Rozenberg MA, Avtzis D, Battisti A, Bellanger R, Bernard A, Bernadinelli I, Branco M, Cavaletto G, Cocquempot C, Contarini M, Courtial B, Courtin C, Denux O et al (2023) Worldwide tests of generic attractants, a promising tool for early detection of non-native cerambycid species. NeoBiota 84:169–209
Ruzzier E, Haack RA, Curletti G, Roques A, Volkovitsh MG, Battisti A (2023) Jewels on the go: exotic buprestid around the world (Coleoptera: Buprestidae). NeoBiota 84:107–135
Ryall K, Silk PJ, Mayo P, Crook D, Khrimian A, Cossé AA, Sweeney J, Scarr T (2012) Attraction of Agrilus planipennis Fairmaire (Coleoptera: Buprestidae) to a volatile pheromone: effects of release rate, host volatile and trap placement. Environ Entomol 41:648–656
Sallé A, Parmain G, Nusillard B, Pineau X, Brousse R, Fontaine-Guenel T, Ledet R, Vincent-Barbaroux C, Bouget C (2020) Forest decline differentially affects trophic guilds of canopy-dwelling beetles. Ann For Sci 77:1–21
Silk PJ, Ryall K, Mayo P, Lemay MA, Grant G, Crook D, Cossè A, Fraser I, Sweeney JD, Barry Lyons D, Pitt D, Scarr T, Magee D (2011) Evidence for a volatile pheromone in Agrilus planipennis Fairmaire (Coleoptera: Buprestidae) that increases attraction to a host foliar volatile. Environ Entomol 40:904–916
Silk PJ, Mayo P, Ryall K, Roscoe L (2019) Semiochemical and communication ecology of the emerald ash borer, Agrilus planipennis (Coleoptera: Buprestidae). InSects 10:323
Silk PJ, Ryall K, Roscoe L (2020a) Emerald ash borer, Agrilus planipennis (Coleoptera: Buprestidae), detection and monitoring in Canada. Forestry 93:273–279
Silk PJ, Ryall KL, Grant G, Roscoe LE, Mayo P, Williams M, LeClair G, Kimoto T, Williams D, Rutledge C (2020b) Tree girdling and host tree volatiles provides a useful trap for bronze birch borer Agrilus anxius Gory (Coleoptera: Buprestidae). For Int J For Res 93:265–272
Skvarla MJ, Holland JD (2011) Nontarget insects caught on emerald ash borer purple monitoring traps in western Pennsylvania. Northern J Appl For 28:219–221
Thurston GS, Slater A, Nei I, Roberts J, McLachlan Hamilton K, Sweeney JD, Kimoto T (2022) New Canadian and provincial records of Coleoptera resulting from annual Canadian Food Inspection Agency surveillance for detection of non-native, potentially invasive forest insects. Insects 13:708
van Wijk M, de Bruijn PJA, Sabelis MW (2011) Complex odor from plants under attack: herbivore’s enemies react to the whole, not its parts. PLoS ONE 6:e21742
Vuts J, Woodcock CM, Sumner ME, Caulfield JC, Reed K, Inward DJ, Leather SR, Pickett JA, Birkett MA, Denman S (2016) Responses of the two-spotted oak buprestid, Agrilus biguttatus (Coleoptera: Buprestidae), to host tree volatiles. Pest Manag Sci 72:845–851
Wittman JT, Silk P, Parker K, Aukema BH (2021) Optimizing early detection strategies: defining the effective attraction radius of attractants for emerald ash borer Agrilus planipennis Fairmaire. Agric For Entomol 23:527–535
Zhang X, Zhang YL, Zhang P, Cui ZJ, Han P, Gao GZ, Poland TM, Zalucki MP, Lu ZZ (2021) Agrilus mali Matsumura (Coleoptera: Buprestidae) density and damage in wild apple Malus sieversii (Rosales: Rosaceae) forests in Central Eurasia under four different management strategies. Entomol Gener 41:257–266
Acknowledgements
This study was carried out as part of the Euphresco project 2020-A-337 “Developing and assessing surveillance methodologies for Agrilus beetles”. We thank Matthias Becker, Madeline Bidinger, Olivia Bigham, Andreas Daxer, Hannah Doerger, Stephie Feltgen, Martin Križaj, Julia Lankisch, Maximilian Lorbeer, Alice Martinelli, Paolo Paolucci, Olivia Ruhlman, Dahlia Seibert, Duncan Self, Cailie Sitawi, Tom Staton, Silke Steinmöller, Melanie Studera, Tobias Wille and Alisha Yerovi for field and laboratory assistance. We also thank Andreas Baderschneider (Lower Saxony State Forest), Richard Boyce, Dick Durtsche, New Brunswick Department of Natural Resources and Energy Development, Urbarialgemeinde Neckenmarkt, Bob Thurston, Nicole Kelleher (Massachusetts Department of Conservation and Recreation), Northern Kentucky University Research and Education Field Station and Ohio Department of Natural Resources for permission to carry out the experiments at the selected sites. Finally, we thank Wolfgang Barries, Joshua Basham (USDA-APHIS MRP), and Lawrence Barringer (Pennsylvania Department of Agriculture) for help in determination of Agrilus species.
Funding
Open access funding provided by Università degli Studi di Padova. BH received funding from the German Federal Ministry of Food and Agriculture (BMEL); JS received funding from the Canadian Food Inspection Agency (project #02316/N-000338.001.01) and Natural Resources Canada, Canadian Forest Service under the Pest Risk Management Program; GH received funding from the Austrian Federal Ministry for Agriculture, Forestry, Regions and Water Management (Project no. 101607); DR was funded by the DOR program of the University of Padua and by the Agritech National Research Center that received funding from the European Union Next-GenerationEU (Piano Nazionale di Ripresa e Resilienza (PNRR)—Missione 4 Componente 2, Investimento 1.4–D.D. 1032 17 June 2022, CN00000022); GS was funded by The Foundation Blanceflor Boncompagni Ludovisi, née Bildt; AS was funded by the Région Centre-Val de Loire (Project CANOPEE no. 2018-00124136); MdG was funded by the Administration of the Republic of Slovenia for Food Safety, Veterinary Sector and Plant Protection (Contract No. C2337-23-000026) and Slovenian Research Agency (Research Programme P4-0107); DW was funded by the Department of Environment, Food & Rural Affairs (Defra); AMR was partially funded by USDA cooperative agreements AP20PPQS&T00C173, AP21PPQS&T00C163, and AP22PPQS&T00C145 to Xavier University, and AP21PPQS&T00C152, and AP22PPQS&T00C187 to Pennsylvania Department of Agriculture; JAF sample identification was funded through cooperative agreements AP21PPQS&T00C152, and AP22PPQS&T00C187 to Pennsylvania Department of Agriculture; JAF was funded by the USDA APHIS PPQ Pest Detection Program as well as USDA PPA 7721 projects 3.0712.01 (2021) and 3.0047.01 (2022).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Ethical approval
This article does not contain any studies with human participants or vertebrates performed by any of the authors.
Additional information
Communicated by Antonio Biondi.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Santoiemma, G., Williams, D., Booth, E.G. et al. Efficacy of trapping protocols for Agrilus jewel beetles: a multi-country assessment. J Pest Sci 97, 1795–1810 (2024). https://doi.org/10.1007/s10340-023-01728-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-023-01728-z