Abstract
Plants have a large diversity of metabolites in order to carry out the complicated plant metabolic pathway in a coordinated manner under normal as well as stressful conditions. These metabolites are further subdivided into primary metabolites which are responsible the for main metabolic pathways that are critical for the survival of plants and secondary metabolites which are not necessary for the main metabolic pathway for growth and development but are involved in developing the ability of the plants to interact with the surrounding adverse environment. Plants produce a diversity of secondary metabolites (PSMs) that serve as defense compounds against herbivores and microorganisms. In addition, some PSMs attract animals for pollination and seed dispersal. Pathogens gain entry into host cell, reproduce there and use biological machinery of host plants which is threat to global crop production. Integrated management strategies based upon minimizing population and use of resistant cultivars can address this potential problem. In the developing world, farmers are less likely to adopt these approaches instead they prefer the use of chemical pesticides. Reckless use of chemical pesticides is destroying our ecosystem, which is why ecofriendly alternatives, like plant-based metabolites to control pathogens, must be explored. Studies conducted on different plant metabolites reported that these metabolites can potentially combat plant pathogens. In this study, we also discuss some of the plant secondary metabolites including alkaloids, flavonoids and phenolics, and antioxidant enzymes like peroxidase, polyphenol oxidase, and chitinase.
Zusammenfassung
Pflanzen verfügen über eine große Vielfalt an Metaboliten, die die komplizierten pflanzlichen Stoffwechselwege sowohl unter normalen als auch unter stressigen Bedingungen koordiniert ablaufen lassen. Diese Metaboliten werden weiter unterteilt in primäre Metaboliten, die für die wichtigsten Stoffwechselwege verantwortlich sind, die für das Überleben der Pflanzen äußerst wichtig sind, und sekundäre Metaboliten, die für die wichtigsten Stoffwechselwege für Wachstum und Entwicklung nicht notwendig sind, aber an der Entwicklung der Fähigkeit der Pflanzen beteiligt sind, mit der umgebenden ungünstigen Umwelt zu interagieren. Pflanzen produzieren eine Vielzahl von Sekundärmetaboliten (PSM), die als Abwehrstoffe gegen Pflanzenfresser und Mikroorganismen dienen. Darüber hinaus locken einige PSM Tiere zur Bestäubung und Samenverbreitung an. Krankheitserreger dringen in die Wirtszelle ein, vermehren sich dort und nutzen die biologische Maschinerie der Wirtspflanzen, was eine Bedrohung für die weltweite Pflanzenproduktion darstellt. Integrierte Bewirtschaftungsstrategien, die auf der Minimierung der Population und der Verwendung resistenter Sorten basieren, können dieses potenzielle Problem lösen. In den Entwicklungsländern sind die Landwirte weniger geneigt, diese Ansätze zu übernehmen, sondern bevorzugen den Einsatz von chemischen Pestiziden. Der rücksichtslose Einsatz von chemischen Pestiziden zerstört unser Ökosystem. Deshalb müssen umweltfreundliche Alternativen erforscht werden, z. B. pflanzliche Stoffwechselprodukte zur Bekämpfung von Krankheitserregern. Studien zu verschiedenen Pflanzenmetaboliten haben ergeben, dass diese Metaboliten potenziell Pflanzenpathogene bekämpfen können. In dieser Studie haben wir auch einige pflanzliche Sekundärmetaboliten wie Alkaloide, Flavonoide und Phenole sowie antioxidative Enzyme wie Peroxidase, Polyphenoloxidase und Chitinase untersucht.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There are over 400,000 vascular plant species on earth, which are the sources of thousands of metabolites; however, the function, utility and structure of these metabolites have only been moderately explored. Plants that are sessile and continuously exposed to changing environments have far more metabolic diversity than any other organism (Mohamed 2011; Mohamed and Akladious 2014; Fang et al. 2019; Younes et al. 2019; Bashandy et al. 2020). These metabolites are responsible for providing protection to plants from the external environment as well as internal issues. Plant metabolites are intermediates and products of metabolism and are categorized into two groups, i.e., primary and secondary (Fig. 1).
Role of primary and secondary metabolites. Isah 2019 (CC BY 4.0, http://creativecommons.org/licenses/by/4.0/)
A primary metabolite is a metabolite that is directly involved in normal growth, development, and reproduction. It usually performs a physiological function in the organism. A primary metabolite is typically present in many organisms or cells. It is also referred to as a central metabolite, which has an even more restricted meaning (present in any autonomously growing cell or organism). Some common examples of primary metabolites include ethanol, lactic acid, and certain amino acids. A primary metabolite plays a very important role in plant metabolism and is essential for the plant existence. Some primary metabolites are precursors of secondary metabolites (Pott et al. 2019; Abdel Latef et al. 2020; Younes et al. 2020). These metabolites serve a variety of purposes in plants, including acting as signaling molecules, having inhibitory and stimulatory effects, having catalytic activity, assisting in plant defense, acting as fuel, and protecting the plant from harmful environmental effects (Abd El-Rahman et al. 2012; Mogazy et al. 2022).
Secondary metabolites (SM) are directly unrelated to the survival of the plant, but they interact with the target molecule responsible for the change in the surrounding environment to ensure the survival of the plant under such conditions (Hussain et al. 2022). Secondary metabolites are low molecular compounds and are tissue specific. The production of secondary metabolites in plants is derived primarily from primary metabolites and has diverse biological functions for the better survival of plants under stress (Fig. 2). These plant metabolites are indispensable organic compounds produced by the plant and participate in several cellular metabolisms to determine the actual physiological state of the cell for the plant’s production and make plants competitive in their surrounding environment (Carrington et al. 2018). These small molecules exert a wide range of effects on plant physiology, like stimulating flowering, setting fruits, maintaining perennial growth, inducing signals for deciduous behavior, regulating stomatal conductance, and photosynthesis. Plants exposed to different stresses cause reductions in plant height, root volume, number of branches, leaf number, leaf area, and secondary metabolites (Mohamed and Abdel-Hamid 2013). These metabolites protect the plant both under biotic stress as they act as antioxidants, osmoprotectants, antimicrobials, and repellents, while under normal conditions, secondary metabolites are either synthesized in very low concentrations or absent. The types of metabolites and their nature directly depend on the physiological condition and developmental stage of the plants, the intensity of the stress, and the type of stress (Jha 2019a). In order to be effective as defense chemicals, plant secondary metabolites evolved as bioactive substances that can interfere with a large number of molecular targets in cells, tissues, and organs of animals or of microbes. The known functions of secondary metabolites are summarized in this review.
Plant growth and development are influenced by a variety of biotic and abiotic challenges, and plants respond to these pressures with a variety of methods and defense systems. Jan et al. 2021 (CC BY 4.0, , http://creativecommons.org/licenses/by/4.0/)
In this review, the bioactive compounds produced in the plant in response to changes responsible for generating stress such as phytopathogenic interaction (biotic stress) are discussed.
Biotic Stress
Biotic stress, which is induced due to infection caused by bacteria, viruses, fungi, and nematodes, is the cause of a remarkable loss in yield and productivity (Mohamed and Abd-El Hameed 2014). Plants’ responses to such biotic stress are incredibly complex, as they involve interaction between two living organisms. Such pathogenic organisms tend to obtain food from the plant, so to achieve that, they kill plant cells and weaken the plant (Jha 2021). At the same time, plants develop multilayer protective mechanisms and induce different defense mechanisms for their protection from such pathogenic interactions. Thus, plant–pathogen interaction is a versatile process where actual interaction takes place between plant and pathogen-derived stimulator biomolecules like lipopolysaccharides, sugars, and proteins. These pathogen-derived stimulator biomolecules are responsible for pathogenicity, and the severity of infection is determined by the pathogen’s ability to colonize inside the plant cell (Jha 2018). In addition, plants produce effector molecules that recognize pathogenic activator chemicals and activate the plant’s defense response by recognizing microbial elicitors via plant receptor proteins on the cell membrane.
Plant Response to Biotic Stress
Plants are susceptible to a variety of biotic and abiotic stresses throughout their lives, and being sessile organisms, they are unable to escape these danger zones. Plants have three ways of expressing their stress responses. Some plants escape stress entirely (for example, ephemeral, short-lived desert plants), while others show susceptibility to stress, which can lead to plant death, and still, others show resistance (Akula and Ravishankar 2011). Plants have evolved complex and tightly regulated mechanisms to defend themselves against a variety of stressors. Plants evolve efficient mechanisms to cope with these challenging environments, such as adjustments in photosynthetic rates, stomatal conductance, transpiration, cell wall architecture, remodeling of membrane structure, and alterations in cell cycle and division rates with overall effects on general growth to fine-tune and metabolism of bioactive compounds (Atkinson et al. 2015). Signaling systems and routes start primary metabolism, which provides biosynthetic intermediates for secondary metabolism in the plant, and so initiate stress and defense responses. Fig. 3 illustrates the stress–response system and the inducible defense system, for example, rely on inducible activation of a large number of defense-related genes, a suite of molecular and cellular processes, and inducible production of a variety of defense-related secondary metabolites (Aly et al. 2012, 2013, 2017; Narayani and Srivastava 2017). Plants synthesize a large number of secondary metabolites in their cells from primary metabolites, and the production of those metabolites is regarded to represent an adaptive capacity for dealing with tough environmental constraints (Caretto et al. 2015).
Plants are generally exposed to various combinations of biotic and abiotic stresses, such as drought, salinity, heavy metals, UV irradiation, pathogenesis, and herbicides (Akladious and Mohamed 2017; Naeem et al. 2020; Ghonaim et al. 2021 Sofy et al. 2021a). In addition, Fig. 4 illustrates that various stresses alter plant morphology and increase levels of phenolic pigments, antioxidant activity, electrolyte leakage, flavonoids, proline, and tocopherol accumulation, all of which impede plant growth (Bano et al. 2016, 2017). Increased reactive oxygen species (ROS) levels in the biological system in response to diverse biotic and abiotic stresses causes oxidative stress, which leads to lipid peroxidation, enzyme inactivation, and DNA damage (Akula and Ravishankar 2011; Atkinson et al. 2015). Plants use a variety of alternative strategies to overcome stress restrictions, including the synthesis of a wide range of secondary metabolites that act as resistance tools. The antioxidative defense mechanism of the plant, as well as several metabolites, help it to survive in severe environments (Bartwal et al. 2013).
Physiological changes in the plant under the influence of various abiotic and biotic stresses. Reprinted by permission from Springer Nature Customer Service Centre GmbH: Springer Nature. Journal of Plant Biology. Plant secondary metabolites synthesis and their regulations under biotic and abiotic constraints. Khare et al. 2020
Biosynthesis of Secondary Metabolites in Plants
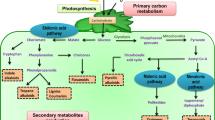
Fig. 5 depicts the production of secondary metabolites and their interconnections/interrelationships with primary metabolism within the plant cell. Alternative mechanisms in plants produce a wide spectrum of secondary metabolites, including terpenes, phenols, and alkaloids, among others (Fig. 5). The mevalonic acid (MVA) and 2‑C-methylerythritol 4‑phosphate (MEP) routes, which occur in the cytosol and plastid, respectively, are two key mechanisms for the production of terpenes. Isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP) are produced from glycolysis products such as pyruvate and glyceraldehyde-3-phosphate and act as universal precursors for all terpenoids found in various cellular compartments (Fig. 5). Terpene’s synthase enzymes are involved in the production of terpenes in many cellular compartments (Khare et al. 2020). Phenolic compounds are synthesized in plants using shikimic acid pathway and the malonic acid pathway (Ghasemzadeh and Ghasemzadeh 2011). The malonic acid pathway has been also reported in fungi and bacteria for the synthesis of phenolics (Cheynier et al. 2013). Phenylalanine ammonia-lyase (PAL) and CHS (chalcone synthase) are the soul enzymes regulate phenolic levels under various stress constraints. Function of PAL, C4H (cinnamate 4‑hydroxylase), 4CL (4-coumarate: CoA ligase), CHS, CHI (chalcone isomerase), F3H (favanone-3-hydroxylase), F30H (favonoid-30-hydroxylase), F3050H (favonoid-3050-hydroxylase), DFR (dihydrofavonol 4‑reductase), FLS (favonol synthase), IFS (isofavone synthase), IFR (isofavone reductase), and UFGT (UDP favonoid glycosyltransferase) were upregulated with the elevated enzymes concentrations (Sharma et al. 2019). Nitrogen-containing secondary metabolites are characterized by the presence of a nitrogen-containing moiety in their structure and amino acids such as lysine, tyrosine and tryptophan act as precursors in their biosynthesis (Ahuja et al. 2012).
Biosynthesis of secondary metabolites. Reprinted by permission from Springer Nature Customer Service Centre GmbH: Springer Nature. Journal of Plant Biology. Plant secondary metabolites synthesis and their regulations under biotic and abiotic constraints. Khare et al. 2020
Role of Plant Secondary Metabolites Under Biotic Stress
Pathogens have different mechanisms for nutrient acquisition and are classified as necrotrophic, biotrophic, and hemibiotrophic. Biotrophic pathogens require living host tissues that ensure prolonged supply of the carbon and other nutrients to them. Necrotrophic pathogens cause host cell death and acquire nutrients released from dead cells. Hemibiotrophic pathogens obtain nutrients from the host cell by exhibiting the biotrophic phase in early stages of the infection process and then switching to the necrotrophic mode during later stages of their life cycle (Glazebrook 2005). Plant health depends on the natural immunity of the plant, which depends on the induction of preformed defense responses. But such a response is nonspecifically caused by structural barriers and the production of antimicrobial compounds (Fahed et al. 2021). However, successful infection depends on several factors, like the presence of surface receptors for the pathogen on the plant cell surface, the ability of the pathogen to produce toxins or toxic secondary metabolites, whether the pathogen has the ability to overcome plant defense mechanisms, as well as the biochemical, physiological, and genetic makeup of the plants. At the same time, to check infection, plants must have the ability to discriminate between pathogenic interactions and be able to stimulate an efficient defense response to confine pathogenic interactions without compromising their own development. The defense response is induced by the plants upon the recognition of elicitor proteins produced by the pathogen and translocated into the host cell (Abd El-Rahman and Mohamed 2014).
Upon infection, plants induce early defense responses like fortification of the cell wall, reorganization of cytoskeletal, production of reactive oxygen species, phytoalexin synthesis, and later transcription of pathogenesis-related proteins that induce programmed cell death to limit the spread of pathogens (Jha et al. 2021). The stress tolerance ability and environmental adaptation of a plant depend directly on the diversity of secondary metabolites, which can be generally grouped into phenolics, flavonoids, alkaloids, saponins, and terpenoids, and a series of exchanges and transductions of chemical signals takes place in plants upon plant–microbe interactions for the induction of appropriate metabolic responses (Tables 1 and 2) (Mhlongo et al. 2018).
Phenolic Metabolites
It is a group of metabolites that have different numbers of oxy groups and contain one or more phenolic residues. They are known as phenolic residues and are present both in free and conjugated forms. It is located in chloroplasts in young plant cells and in the vacuole in mature cells. Depending upon the function and ultrastructure of chloroplasts, synthesis and accumulation of phenolic metabolites directly takes place from the precursor-like shikimic acid and phenylalanine in the plant cell (Maeda and Dudareva 2012; Hamed et al. 2019). It includes simple phenols and phenols carboxylic acids, quinones and their derivatives, fleukoanthocyans, flavones, catechins, and flavonols, which play a remarkable role in plant metabolism and have antibacterial, allelopathic, and antifungal activity when they interact with elicitor compounds produced by plant pathogens (El-Beltagi et al. 46,47,a, b). Phenolics are one of the prominent secondary metabolites of plants and act as precursors or intermediaries of many metabolic processes. They also have a role in incorporating striking substances for camouflage, for enhanced pollination, as well as protection against herbivores. Phenolics are classified according to their origin and structure (Fig. 6). According to their structure, phenolics are classified into phenolic acids, flavonoids, tannins, coumarins, lignans, quinones, stilbenes, and curcuminoids (Kisiriko et al. 2021).
Phenolic Acids
Phenolic acids and their derivatives represent a varied class of phenolic compounds in plants. The main function of phenolic acids is in the symbiotic interaction of plant–microbe in the plant rhizosphere. It is a secondary metabolite, which is widely distributed in the plant kingdom and is essential for plants’ reproduction, growth, and is produced in response to adverse environmental stimuli (Mohamed et al. 2018; Abu-Shahba et al. 2021, 2022). It has been produced in several different ways as breakdown products of cell wall polymers of lignin, through the phenylpropanoid pathway by shikimic acid precursor, as byproducts of the monolignol pathway (Zabalza et al. 2017). Inplant it may exist either in bounded form or in a soluble form. Bounded phenolic acids form a complex with the protein/cellulose of the cell wall by glycosidic bonds and help in the formation of the cell wall. This incorporation of phenolic acids into the cell wall takes place due to enhanced flux of phenylpropanoid compounds, resulting in the production of benzoic acid and cinnamic acid derivatives, which after esterification can be incorporated into the cell wall in response to biotic stress (Jha 2019a). Different plant phenols have been synthesized from hydroxy-cinnamic acids. These hydroxy-cinnamic acids are the main molecules for the production of diverse groups of phenylpropanoid metabolites in plants for regular plant growth as well as under stress. Phenolic acids present in conjugated form with sugar or ester by forming bonds with hydroxyl groups as well as with simple glycosides or lignin. It is produced through host–pathogen interactions but has a significant role in plant defense (Singh et al. 2021).
As soon as a pathogen infects a plant host, it produces phenolics as an early response, which may lead to a general increase in host metabolism (Mayer et al. 2001). It has been observed that when maize is infected with Glomerella graminicola or C. heterostrophus, the two phenolic caffeic acid esters increase significantly (Pusztahely et al. 2017). Although these polyphenols or phenolic compounds are not poisonous to fungi, their quick accumulation suggested that they could act as a source of additional protective substances (Hardoim et al. 2015). In addition, Pseudomonas aeruginosa, Serratia marcescens, Salmonella choleraesuis, Escherichia coli, Bordetella bronchiseptica, Klebsiella pneumoniae, Bacillus subtilis, and Staphylococcus aureus can be affected by polyphenols such as catechins by altering the permeability and properties of plasma membranes and the production of reactive oxygen species such as hydrogen peroxide (Wang et al. 2018).
Phenolic metabolites play a role in nematode resistance through a variety of mechanisms: (i) browning and the development of non-expandable necrosis in plants, (ii) browning and the delayed production of broad necrosis in plants, (iii) browning and the appearance of non-expandable necrosis in plants, (iv) indoleacetic acid (IAA)-oxidase inhibition promotes auxin accumulation and the production of galls or large cells in plants, (v) IAA-oxidase stimulation promotes auxin breakdown and necrosis in plants (Giebel 1982). Nematodes can activate phenols by decomposing bound phenol into free fl-glycosidases and secreting them into the host tissue (Ohri and Pannu 2010).
Flavonoids
Flavonoids are structurally varied secondary metabolites of plants with polyphenolic structures and are produced by the polypropanoid pathway using phenylalanine as the precursor. All flavonoids contain two six-carbon aromatic rings, a heterocyclic ring having one oxygen atom and a basic C6–C3–C6 skeleton with a huge number of functions in plants, like pigmentation, UV protection, endogenous regulation of plant cell physiology, adaptable plant development, plant defense, and signaling (Li et al. 2021). The polypropanoids act as chemical signal modulators for plant–microbe interaction to attract pollinators, phytoalexins against pathogens, and attractants for symbiotic microbes or repellents for pathogenic interaction (Hodaei et al. 2018). The photosynthetic electron transport is responsible for generation of reactive oxygen species (ROS) that are detoxified by the flavonoid system to avoid oxygen-mediated toxicity. Flavonoids can act as free radical scavengers to protect plants under stress or chelate metal ions responsible for ROS generation to provide resistance to aluminum toxicity, or as potential cytotoxicity by interacting with enzymes responsible for cellular activity (Jha 2020; Jha and Yadav 2021; Jha et al. 2021). It also modulates the polar movement of auxins for controlled stomatal movement and resource allocation for regulated plant growth under stress.
Flavonoids are responsible for the color and odor of flowers and fruits, as well as providing protection against biotic (fungal, bacterial, and viral pathogenic) pathogens (Panche et al. 2016). Flavonoids, such as isoflavones, flavones, and flavanones, have been shown to have antifungal efficacy against a variety of phytopathogenic fungi in recent years (Chepkirui et al. 2014). The fungi toxic mechanisms of flavonoids include destruction of cell walls, cytoplasmic membrane damage, enzyme inhibition, cell death induction, metal ion chelation, efflux pump inhibition, and/or interaction with extracellular and soluble proteins (Mierziak et al. 2014). In addition, Serpa et al. (2012) discovered that the flavone baicalein has antifungal action by blocking the efflux pump and triggering death in Candida albicans. In another study, the isoflavone sedonan A, isolated from Dalea rutico, was discovered to have fungi toxic potential against C. albicans and C. glabrata, blocking efflux mediated pumps and altering intracellular transcription targets (Belofsky et al. 2013). In numerous Candida spp., the chalcone carvacrol has been discovered to damage the cellular cytoplasmic membrane and trigger cell death (Zuzarte et al. 2012). Flavonoids have also been shown to have antiviral effects against viral plant diseases (Wan et al. 2015). Flavonoids have antiphytoviral properties that include inhibiting viral protein synthesis, disrupting viral RNA translation, suppressing viral DNA synthesis, interfering with viral structural proteins, and inhibiting viral genome and enzyme transcription (Zakaryan et al. 2017). The creation of hydrogen bonds between the ring of flavonoids and the nucleic acid bases of viruses can hinder viral DNA and RNA production and polymerases.
Tannins
Tannins are secondary metabolites synthesized through vegetal secondary metabolism that typically act as protecting agents against infection and are associated with plant–environment interactions. Due to its acidic taste, it acts as a repellent for herbivorous and the property of precipitating proteins means it regulates many metabolic pathways. Tannins exist as hydrolysable and condensed tannins (Smeriglio et al. 2017). Hydrolysable tannins are made of gallic acid units connected by an ester bond to glucose, and condensed tannins are polymers of flavan-3-ols such as gallocatechin and catechin. Tannin production in plants is genetically controlled as well as influenced by environmental factors like soil pH, nutrient availability, photoperiod, moisture, and the atmospheric CO2 and O2 ratio. The activity of tannins is directly influenced by polymer chemistry, type of monomer, degree of polymerization, and substitution pattern of the phenolic ring. The main function of tannin is to provide protection against herbivores, microbial pathogens, and insects (Miele et al. 2019).
Tannins and proanthocyanidins have antifungal efficacy against phytopathogenic fungi, according to numerous studies. Anttila et al. (2013) investigated the fungitoxicity of conifer tannins in liquid cultures against eight brown rot fungi, three white rot fungi, and four soft rot fungi. Even at low doses, high growth inhibition was observed for the brown rot fungi tested, but not for the white and soft rot fungi. Tannins and similar phenolic substances inhibit extracellular hydrolases from invading pathogens in temperate trees, preventing fungal degradation of the heartwood. It was also shown that tannins extracted from Acacia mearnsii had antifungal action against Aspergillus niger and Candida sp (Dos Santos et al. 2017). Inhibition of extracellular enzymes (cellulase, pectinase, laccase, xylanase), nutrition deprivation of substrates (metal complexation, protein insolubilization), and inhibition of oxidative phosphorylation are among its antifungal modes of action (Ogawa and Yazaki 2018).
Coumarins
Coumarins are a large class of phenolic substances in plants and contain fused benzene and α‑pyrone rings. It is also considered a member of the family of plant-derived secondary metabolites and is also produced via the phenylpropanoid pathway (Reen et al. 2018). There are more than 1300 types of coumarins as secondary metabolites that exist in plants. Coumarins have polar structures and exist in a free state or as glycosides. They have the ability to absorb UV light and are photosensitive, so they are structurally altered under natural light. Coumarins have a defensive role against phytopathogens, a protective response to abiotic stresses, regulation of phytohormones, and protection against oxidative stress. Coumarins in plant roots have an iron-mobilizing ability that helps in iron uptake from iron-deprived soils by plant roots. Coumarin has antibacterial activity against a wide spectrum of Gram-positive bacteria (Reen et al. 2018).
Phenolics build up at the infected plant’s infected spot, inhibiting the microorganism’s general growth and development as a result of cell death caused by the hypersensitive reaction (Lincoln et al. 2018). Biosynthesis of lignin proceeds from L‑phenylalanine via 4‑coumaric acid and the CoA-esters of 4‑coumaric, ferulic, and sinapic acids to the corresponding alcohols, which supposedly polymerize under the action of a peroxidase and aid in fighting biotic stress agents (Peperidou et al. 2017; Bi et al. 2017). Ferulic acid and coumaric acid are lignin precursors (Goleniowski et al. 2013). They have key roles in physical and chemical defense against pathogens, as well as structural functions, enzyme activities, nutrition uptake, protein synthesis, photosynthesis, and allelopathy (Leváková and Lacko-Bartošová 2017).
Lignans
Lignans are naturally occurring dimeric phenylpropanoids and nonflavonoid metabolites in plants. The dimeric phenylpropanoids that are in lignans have a bond between carbons C8 and C80. On the basis of carbon skeletons, the position of incorporated oxygen, structural patterns, and structural patterns, it is classified into eight classes: furan, dibenzylbutane, furofuran, aryltetralin, aryltetralin, dibenzylbutyrolactone, dibenzylbutyrolactol, and arylnaphthalene, which can exist in free form or its derivatives and glycosides. Lignans, due to their unique structure, can act as efficient antioxidants and protect plants from the detrimental ROS generated in excess under stress. In scavenging ROS, lignans can act as a primary antioxidant (Kumar and Singh 2015). In plants, mostly lignans exist in free form and protect plants from oxidative stress by scavenging free radicals during growth and development. Lignans play a significant role in plant–pathogen defense by blocking microbe-derived degradative enzymes like cellulases, polygalacturonases, glucosidases, and laccases (Davin et al. 2008). Furthermore, lignans have been suggested to act as insect antifeedants by altering the insect endocrine system (Harmatha and Dinan 2003).
Lignin
Lignin is the second most abundant natural polymer on the planet, after cellulose. Three basic phenolic molecules (monolignols) in the cell wall combine to produce this polymer (C6–C3)n (Alejandro et al. 2012). The formation of a physical barrier against invading pathogens by lignin deposition in the cell wall serves an important defense function in the plant. It also improves plant growth and development, cell wall rigidity, and hydrophobic characteristics (Schuetz et al. 2014).
Lignin is well-known for its ability to defend against phytopathogenic fungi. It exhibits antifungal properties, serves as a physical barrier to prevent fungal invasion, and inhibits the spread of fungal toxins (Xie et al. 2018). Many pathogenic fungi have been shown to deposit lignin during their attack. Marques et al. (2018) investigated the cell wall-associated defense responses in smut-resistant and susceptible sugarcane cultivars against infection with Sporisorium scitamineum, the smut causative agent. Furthermore, when lignin was tested in vitro against the fungal pathogen Diplodia pinea, the cause of tip blight and canker in Austrian pine, it showed the most significant antifungal activity compared to the other phenolic compounds (Sherwood and Bonello 2013). Downregulation of several lignin biosynthesis-related genes, on the other hand, was found to boost plant immunity. Suppression of HCT in Arabidopsis and Medicago resulted in the buildup of salicylic acid and activation of certain pathogenesis-related genes, resulting in improved plant immunity (Gallego-Giraldo et al. 53,54,a, b). Lignification is a plant defense strategy against pathogens and herbivores because it increases the physical toughness of plant tissues, making them indigestible to insects and other herbivores (Ahmad et al. 2021a).
Quinones
Quinones are found in a wide variety of plant families with remarkable structural diversity and have a function in photosynthesis. Quinones have cyclic, α, β-diketone structures, which on reduction from hydroquinones have phenols with two hydroxyl groups. Quinones are classified into anthraquinones, benzoquinones, anthracyclinones, naphthoquinones, and condensed quinines. Among all types, anthraquinones comprise the largest group of natural quinines (Laohavisit et al. 2020). Many quinones are derived from different pathways in plants; many are derived from common shikimate pathways, many from acetate–malonate pathways, and a few from the oxidative modification of secondary metabolites. Most quinonoid compounds may occur as glycosides in plants. Quinones, for example, are extremely beneficial to plants because they generate complex derivatives by interacting with proteins and blocking herbivores from macerating proteomic substances (Easwar et al. 2017). The presence of these molecules in amino acids causes insects to have difficulty digesting proteins, which hinders pest growth. Reactive oxygen species such as peroxides, superoxide, and singlet oxygen, which are implicated in a variety of biotic and abiotic stressors, are likewise reduced by phenols. Plants are protected from these challenges by these phenolic chemicals, which initiate the start of defense enzymes (Saddique et al. 2018).
Stilbenes
Stilbenes are a small family of plant secondary metabolites derived from the phenylpropanoid pathway, with a common structure of a 14-carbon skeleton having two benzene rings centrally linked with an ethylene bridge. So, it exists in two possible stereoisomers, cis and trans, but naturally, in plants, it exists in the trans form. Stilbenes are responsible for providing protection against abiotic and biotic stress (Valletta et al. 2021). On one hand, it counterbalances the aggression, exerting a direct toxic effect on the pathogen, while on the other hand, it acts as an antioxidant, protecting the cells from oxidative damage. Stilbenes may potentially affect the development of fungi. Conidia treated with sublethal or lethal concentrations of resveratrol or pterostilbene show cytological abnormalities, such as curved germ tubes, protoplasmic retraction in the dead hyphal tip cell, cytoplasmic granulation of conidia, disruption of the plasma membrane, and regrowth of a secondary or tertiary germ tube from the surviving conidium. Some of these characteristics have been reported previously for another phytoalexin/pathogen relationships (Adrian et al. 1997). Membrane peroxidation could be one of the stilbenes probable mechanisms of action (Lee et al. 2017). In Botrytis cinerea dormant conidia, pterostilbene destroyed the endoplasmic reticulum, as well as the nuclear and mitochondrial membranes. Natural and synthesized stilbenes have a positive connection between antifungal activity and hydrophobicity, implying that pterostilbene is more active than the less hydrophobic resveratrol due to greater cytoplasmic membrane diffusion (Caruso et al. 2011).
Curcuminoids
Curcuminoids are natural polyphenol compounds and plant secondary metabolites that accumulate in plants at low concentrations during very long growth periods. Curcumin’s molecule is similar to ubiquinols, and other phenols known to have strong antioxidant properties. Curcuminoids are produced by type III polyketide synthases (PKS) in plants and consist of two phenylpropanoid units chemically derived from the amino acid phenylalanine and connected by a central carbon unit derived from malonyl–coenzyme A (Chungloo et al. 2021). Curcumin exhibited the antiviral activity against coxsackievirus by reduction of viral RNA expression, protein synthesis, and virus titer (Chungloo et al. 2021).
Alkaloid Metabolites
Alkaloids are one of the largest groups of secondary metabolites, with exceptionally different biosynthetic pathways and structures. They are low molecular weight nitrogen containing heterocyclic ring compounds having a heterocyclic ring consisting of a nitrogen atom and is widely distributed in vascular plants. There are more than 20,000 different types of alkaloids that have been reported to date and are responsible for improving plant resistance against stress (Thawabteh et al. 2019). Alkaloids (such as quinolones, metronidazole, and others) have been proven to have antimicrobial effect through blocking enzyme activity or other methods. Squalamine, a polyamine alkaloid, works on Gram-negative bacteria by disrupting their outer membranes and depolarizing Gram-positive bacterial membranes through a detergent-like mechanism of action (Alhanout et al. 2010). Alkaloids have been classified depending on their natural/biochemical origin and chemical structure, and they can be derived from different biosynthetic pathways: the polyketide and terpenoid pathways, the lysine, nicotinic acid, and ornithine pathways, the purine, and histidine pathways, and the shikimate pathway. The induction of alkaloids in plants takes place in response to wounds in plants, pathogen attacks, changes in irradiance intensities, and the presence of herbivores (Helmi and Mohamed 2016). Plant alkaloid secondary metabolites are also responsible for enhancing plant reproductive rates by attracting pollinators as well as improving seed dispersal. Accumulation of alkaloids is also frequently responsive to developmental signals such as modification associated with fruit setting and flowering (War et al. 2018). It has a defensive role against biotic stress. Due to its bitter taste or toxic nature, it acts as a predator repellent (Mohamed and Abd-El Hameed 2014).
Plant alkaloids are essential for plant defense. Plant alkaloids have the opportunity to generate both beneficial and poisonous potent bioactive compounds, which is both a blessing and a curse (Cushnie et al. 2014). Alkaloids are one of the mechanisms by which plants defend themselves against a wide range of potential environmental hazards. Biological agents are one of the most dangerous causes of plant diseases (including fungi and bacteria). Pathogens form a strong bond with their hosts in order to reduce and block plant defenses, while also promoting the nutrient release. Plants defend themselves against infections via a range of techniques, including the synthesis of poisonous chemicals (Freeman and Beattie 2008). The antibacterial effect of alkaloids containing quaternary ammonium groups causes the bacterial cell to die immediately by breaking the negatively charged bacterial cell membrane, followed by the release of K+ ions and other cytoplasmic components (Suzuki et al. 2018). Suzuki et al. (2018) reported antimicrobial effects of β-carboline derivatives against bacteria and fungi, including S. aureus, E. coli, Candida albicans, Candida intermedia, and Candida krusei, and concluded that naturally occurring β‑carboline derivatives and their nitrogen-containing alkylated analogs could be synthesized. Many plants have been identified as a source of antibacterial alkaloids, according to Tiku (2018).
The alkaloid cocaine found in coca and is effective against both Gram-negative and Gram-positive cocci. Piperine has an antibacterial and antifungal alkaloid produced from Piper nigrum, against Lactobacillus sp., Micrococcus sp., Escherichia coli, and Escherichia faecalis. Long et al. (2018) investigated the antibacterial and antifungal properties of six alkaloids isolated from the areal section of Kopsia ruticose against two Gram-positive bacteria and five Gram-negative bacteria, as well as five pathogens. The majority of alkaloids are toxin-like and play an important role in plant defense against microbial infection and herbivore attack. Because alkaloids have particular features such as bitter flavor, interruption of protein activity after consumption and metabolization, and central nervous system change, they play a vital role that has been documented in numerous times in the scientific literature as protective agents against herbivores (Matsuura and Fett-Neto 2015).
Aphids are among the most economically damaging pests of plants, transmitting plant viruses and, as a result, causing fungal infection and an adverse influence on photosynthetic activity by intaking plant juices with their stylets and secreting honeydew, resulting in serious economic–financial losses around the world (Yan et al. 2018). Aphids are small sap-sucking insects that infest a variety of economically significant vegetable and fruit crops throughout the world, including apple (Malus domestica), crab apple (Malus sylvestris), and papaya (Carica papaya). Yan et al. (2018) tested the insecticidal effects of ten alkaloids extracted from Lycoris radiate against Aphis citricola, including amabiline, deoxytazettine, deoxydihydrotazettine, 3‑epimacronine, galantamine, 11-hydroxygalanthamine, N‑allylnorgalanthamine, 11β-hydroxygalanthamine, lycorine, and colchicine. The first nine alkaloids examined were shown to have potential aphicidal activity against A. citricola. In vivo and in vitro, N‑allylnorgalanthamine had the best inhibitory activity as an aphicidal against A. citricola. Ricinine alkaloid possesses insecticidal effect against Atta sexdens and can be found in all parts of the Ricinus communis plant (Santos et al. 2018).
Saponins Metabolites
An assorted group of secondary metabolites identified by their unique structure, having triterpene or steroidal aglycone linked with sugar chains and involved in many biological/physiological activities for the induction plant defense system (Moses et al. 2014). They are chemically complex, naturally occurring, functionally and structurally diverse phytochemicals broadly distributed in plants. The foaming and emulsifying properties of the saponins are due to their highly amphipathic nature because of the presence of hydrophilic sugar molecules with a hydrophobic aglycone backbone (Moses et al. 2014). Saponin content is highly dynamic in plants and is directly influenced by external stimuli such as biotic stress due to pathogenic interaction. Accumulation and synthesis of saponins are considered as a part of an integrated defense mechanism and considerable saponin content alteration has been observed under pathogenic interaction in plants. During pathogenic interaction, the saponin content in the infected cell changed dramatically due to hydrolysis for infection or degradation by the pathogen (He et al. 2019).
Saponins are generally located in the vacuoles of plant cells in an inactive form and readily become metabolically active upon pathogenic interaction stimuli, as well as being involved in plant mutualistic symbioses and helping plants respond positively to external factors. Phytochemical studies revealed that plants are especially abundant in saponins that could be induced by anticandidal action (Maatalah et al. 2012). Saponins are mostly recognized to play a role in plants’ antimicrobial defenses. Their ability to form complexes with sterols in the microorganism’s membrane and cause disruption of the membrane is the basis of the processes that underpin their behavior (Sreij et al. 2019). The creation of holes and the loss of membrane integrity are saponins’ principal modes of action against fungus. Saponins have been known to have antifungal properties for plants for decades (Gruiz 1996). The mode of action is similar to that of saponins, which have hemolytic activity. The oat avenacin triterpene saponin was found to have the following pattern of action: in the first stage, aglycones are inserted into the membrane and then bound to sterols (Korchowiec et al. 2015). The next stage aids in the association of sugar residues and the creation of sterol–saponin complexes. It also helps with membrane lipid reorganization, the creation of holes, and cell lysis (Coleman et al. 2010).
Plants have also developed a variety of strategies for using secondary metabolites to combat insects (War et al. 2012). Healthy and sustainable agriculture may be looking for plant-derived chemicals as a source for biopesticide development (Silva et al. 2012). Several plant secondary metabolites have been identified as insect pest product feeding deterrents (Nawrot and Harmatha 2012). Saponins have a direct effect on insect pest reproduction and growth due to their repellent or deterrent properties (Singh and Kaur 2018). These reduce food consumption and disturb the insect’s digestive activities due to toxicity and poor digestibility, raising mortality rates (Adel et al. 2000). Saponins appear to alter the intestinal digestion process by forming complexes with digestive enzymes such as proteases. Saponins break the mucosal cells of the intestine’s inner lining due to their membrane permeability (Qasim et al. 2020).
Terpene Metabolites
Terpenes are a diverse group of volatile compounds produced by plants as secondary metabolites that are responsible for the color, odor, and flavor of plant parts. It is one of the most diverse groups of chemicals and natural products with a unique group of hydrocarbons. The term “terpene” usually refers to a hydrocarbon molecule, while terpenoids are modified terpenes. Due to the complexity of these volatile blends, the terpenes are responsible for protecting plants from herbivores, i.e., by attracting natural enemies of the attacking herbivores (Singh and Sharma 2015). Similarly, terpenoids enable plants to interact with other organisms like neighboring plants, insects, and pathogens as well as act as building blocks for other metabolites for the synthesis of carotenoids, sterols, turpentine, phytol tails of chlorophyll, and some plant hormones. Terpenoids are produced in response to biotics either constitutively or induced as members of herbivore-induced plant volatiles and are synthesized from the five-carbon precursor isopentenyl pyrophosphate unit and its isomer dimethylallyl pyrophosphate. The diversity of terpenes in the plant is due to its universal precursor’s terpene synthases. Many plants and flowers have specific aromas due to the presence of terpenes and terpenoids as primary constituents of their essential oils (Dhifi et al. 2016). It not only has a role against herbivores but also has a role in attracting pollinators, UV-light shielding, protection against toxicity, acting as allelopathic agents, and signal transduction.
Phytoalexins
Plants have complex defense systems against phytopathogens, in which the production of low molecular weight secondary metabolites with antimicrobial activity is known as phytoalexins. It lowers the molecular weight of antimicrobial compounds produced by the plants in response to biotics and is a part of a complex defense system to enable plants to check invading microorganisms (Dhawi et al. 2021). Phytoalexins inhibit the growth of pathogenic fungi, bacteria, and nematodes and are toxic in nature. More than 350 phytoalexins have been characterized to date in plant families. Phytoalexins are antimicrobial compounds that accumulate at the site of pathogenic microorganism interaction and are responsible for inhibition/destruction of the growth of the pathogen as well as regulating plant-parasite interactions (Tiku 2020). Due to their lipophilic nature, phytoalexins are able to cross the plasma membrane and act inside the plant cell. Due to the high number of hydroxyl groups and substituents, it is acidic in nature and is responsible for toxicity in the plant. The mode of action of phytoalexins against fungi includes the destruction of the plasma membrane, disorganization of fungal cellular contents, granulation of cytoplasmic content, inhibition of spore germination, inhibition of mycelial growth, and germ tube elongation (Mansour et al. 2020). Different phytoalexins are produced by different pathways. But the most common are the phenylpropanoid-polymalonic acid pathway, the indole phytoalexin pathway, and the geranyl-geranyl diphosphate and methylerythritol phosphate pathways. The biosynthesis of phytoalexin is regulated by several endogenous molecules like phytohormones like salicylic acid, cytokinins, auxins, ethylene, jasmonic acid, and abscisic acid, as well as transcriptional regulation of defense-related genes, phosphorylation-based cascades (Amer et al. 2021). It also depends on the nature of the infecting pathogen, and the induction of regulatory mechanisms decides the nature of the induced phytoalexin (Sofy et al. 2021b). Phytoalexins limit sporulation, development, and growth of hyphae of plant pathogenic fungi. Plant defense by phytoalexins against microbes or pathogens depends on the rate of synthesis and their accumulation in plant tissues (Duke 2018).
Pathogenesis-Related Proteins
Due to their biochemical properties, pathogenesis-related proteins are low molecular weight proteins, which are able to persist in adverse environments. They are very stable at low pH, at which all other plant proteins become denatured with extreme isoelectric points and are relatively resistant to proteolytic cleavage. Pathogenesis-related proteins are mainly extracellular in nature, but some are located in the vacuole, in intercellular space, and on the cell wall. Pathogenesis-related proteins are typically host-specific proteins stimulated in plants during pathogenic attacks by bacteria, viruses, fungi, insects, nematodes, herbivores, and after wounding or related situations (Ali et al. 2018). Accumulation of pathogenesis-related proteins is an essential constituent of plant innate immune responses, which are induced during pathogen attack and accumulate locally in the infected leaf and are responsible for the activation of systemic acquired resistance or hypersensitive response. The enzymes like β‑1,3‑glucanases, peroxidase, chitinases, phenylalanine ammonia-lyase, and polyphenol oxidase are considered pathogenesis-related proteins and directly act on the pathogen or indirectly via induction of host resistance (Mohamed et al. 2021). Peroxidase (EC 1.11.1.7), a well-known class of PR protein is stimulated by pathogen infection in the host plant tissues. They relate to the PR(pathogenesis-related)-protein 9 subfamily (Van Loon 1997) and are defined as limiting the cellular spread of infection by creating structural barriers or generating highly toxic environments through massive ROS and reactive nitrogen species (RNS) development (Passardi et al. 2005). Phenylalanine ammonia-lyase (PAL) (EC 4.3.1.5), a primary metabolism enzyme of phenylpropanoids seems to play an important role in the synthesis of many secondary defense-related compounds, such as phenols and lignin (Tahsili et al. 2014). Polyphenol oxidase (PPO) (EC 1.10.3.2) another defense-related enzyme is considered to catalyze phenol oxidation to quinones that are harmful to pathogens (Mohammadi and Kazemi 2002). These enzymes are increasingly critical in the direct resistance of plants to pathogenic agents. During plant growth, phenylpropanoid compounds not only attain different outstanding roles but also serve as essential protectants towards different biotic and abiotic stress Pathogenesis-related proteins exhibit a high-degree of pathogen specificity and are synchronized at the level of transcription. Phytoalexins limit sporulation, development, and growth of hyphae of plant pathogenic fungi. Plant defense by phytoalexins against microbes or pathogens depends on the rate of synthesis and their accumulation in plant tissues (Duke 2018).
β-1,3-Glucanase
β‑Glucanase is a common pathogenesis-related protein of the plant defense system, which is induced in plants in response to pathogenic interaction and is broadly distributed in higher plants. β‑Glucanase enzyme catalyzes the hydrolytic cleavage of the β‑1,3‑glucans linkage which is the main structural component of the fungal cell wall, and this enzyme has also been produced by many microbes, to check the growth of other organisms to reduce the competition for food and niche in its surrounding as well as to avail food from the competitor for its own growth and development (Jha et al. 2021). This enzyme in plants serves as a defense enzyme as it inhibits and blocks the growth of pathogens and the enzyme produces oligosaccharides via hydrolysis, which then elicit a cascade of systemic resistance in the plants and promote induction of other related PR proteins and defense-related products (Singh et al. 2014). In addition to plant protection response, β‑glucanase is involved in various plants’ developmental and metabolic activities like pollen germination, bud dormancy, flower growth, fruit ripening, and seed germination (Jha 2019b).
It has been proposed that β‑1,3‑glucanases hydrolyze fungal cell walls, causing fungal cells to lyse when they are defending against fungus. β‑1,3‑Glucanases produce the synthesis of oligosaccharide elicitors in response to pathogen encounters, which elicit the production of additional PR proteins or low molecular weight antifungal chemicals such phytoalexins (Klarzynski et al. 2000). β‑1,3‑Glucanases are usually expressed at low concentration in plants, but when plants are challenged by fungal, bacterial, or viral pathogens, the β‑1,3‑glucanase enzyme increases dramatically (Lusso and Kuc 1995). Class I β‑1,3‑glucanases and class I chitinases showed synergistic effect in pathogen defense. Class I β‑1,3‑glucanase accumulated only at the site of tobacco mosaic virus (TMV) infection in tobacco plants, while class II and III β‑1,3‑glucanases accumulated both at the site of infection and systemically (Livne et al. 1997).
Chitinases
Chitinases are also pathogenesis-related proteins, which are induced in the plant cells in response to pathogenic interactions. Different proteins with differences in structure, substrate specificity, localization, and action are integrated into the chitinase group. They may act on the nonreducing end of the chitin chain (exochitinases) or act randomly inside the polymer (endochitinases). Plant chitinases catalyzes the hydrolysis of the β‑1,4 linkages between N‑acetylglucosamine and N‑acetylmuramic acid residues in the peptidoglycan of bacteria and the bond of chitin of fungal cell walls and also catalyzes the deacetylation of chitin. Chitin is abundant in the cell walls of fungi, algae, and bacteria, as well as in the exoskeletons of invertebrates. As a result, the enzyme chitinase inhibits fungal growth by hydrolyzing fungal hyphal tips with exposed nascent chitin chains, whereas the mature cell wall chitin layer is cross-linked with polysaccharides and overlaid by proteins, making it inaccessible to chitinase hydrolysis and secretory chitinases presumably have different roles in defense reactions (Rana and Farhana 2019). Cellular chitinases are released after fungal penetration and affect cell integrity, while apoplastic chitinases directly act on fungal hyphae invading the intercellular space and indirectly by releasing fungal elicitors. Vacuolar chitinases are effectively induced by ethylene/jasmonate pathways and also induce local hypersensitive responses. Chitinases are considered as molecular targets of selection in plant–pathogen co-evolution (Balint-Kurti 2019). Plant chitinases alone are unable to effectively degrade harder chitin structures of fungi as they usually affect only the hyphal tip, but when co-expressed with β‑1,3‑glucanase, these two enzymes act synergistically against fungal pathogen (Abd El-Rahman et al. 2012).
Phenylalanine Ammonia-Lyase
Phenylalanine ammonia-lyase (PAL) is a plant enzyme that catalyzes the nonoxidative elimination of ammonia from L‑phenylalanine into trans-cinnamic acid, which acts as the precursor of a variety of phenylpropanoids like flavonoids, coumarins, and lignins. In plants, a remarkable amount of total fixed carbon is allocated by this enzyme as it is the first enzyme in the general phenylpropanoid pathway. The phenylpropanoid pathway is among the main secondary metabolic pathways responsible for the production of many secondary metabolites like lignin, stilbenes, suberin, flavonoids, phytoalexins, and coumarins (Jha 76,77,c, d). Phenylalanine ammonia-lyase activity is extremely regulated during the development of plants for the cell- and growth-specific induction of pigments, flavonoids, and lignin and is also modulated according to the changes in the surrounding environment like infection, wounding, and various biotic stresses involved in adaptation or protection. The level of PAL activity is directly related to developmental stage, types of tissue, organ, and plant genotype and is affected by a number of factors, including light, temperature, growth regulators, inhibitors of RNA and protein synthesis, wounding, and mineral nutrition (Abd El-Rahman and Mohamed 2014).
Polyphenol Oxidase
Polyphenol oxidase (PPO) has three active domains, including a C-terminal region, an N‑terminal plastid transit peptide, and a highly conserved type-three copper center, which catalyze the oxidation of numerous phenols to o-quinones and are widely distributed in plants. In the presence of free molecular oxygen, two different reactions take place by polyphenol oxidases: monophenolase catalyzes the hydroxylation of monophenols to o-diphenols, and then diphenols catalyze the oxidation of the o-diphenols to respective o-quinones (Ahmad et al. 2021b). The products of o-quinones are very reactive molecules that undergo nonenzymatic secondary reactions for the formation of a brown color complex called melanin. It induced several defensive reactions like quinine to form cross-linking with another phenolic compound/proteins, resulting in the formation of physical barriers for the entry of pathogens or alkylation of pathogen cellular proteins to affect their metabolic activity or reduce the bioavailability of essential elements for pathogenic infection, direct toxicity of quinines, or the generation of reactive oxygen species, which act in defense signaling. The enzyme polyphenol oxidase is present everywhere in the plant but is most commonly located in the chloroplasts and plays a critical role in plant defense against biotic stress. In chloroplasts, this enzyme also has a protective role in photosynthetic electron transport, which is the site for the production of the high rate of oxygen for proper photosynthetic activity (Moustafa-Farag et al. 2020; Mohamed et al. 2016).
Peroxidase
Peroxidase (POD), an oxidoreductive enzyme that partakes in the processes of cell wall polysaccharide such as phenol oxidation, suberization, auxin metabolism, phytoalexin synthesis, cross-linking of cell wall components and host plant cell lignification during the defensiveness reaction against pathogenic viruses and bacterium (Sofy et al. 2021b). In several plant processes, it provides a broad-spectrum activity, including plant immune responses to biotic stresses (Helmi and Mohamed 2016). Peroxidase is an important enzyme in the biochemical defense mechanisms of plants and is also involved in plant metabolism after infection (Helmi and Mohamed 2016). The expression of POD activity in plant species mediated by various pathogens like viruses (Sofy et al. 2021b), bacteria (Hussain et al. 2022) and fungi (El-Mahdy et al. 2021).
Effect of Phenolics in Postharvest Management of Fruits and Vegetable
In plant pathology diseases produced in fruits are called “rot” and the fungi which cause them are called “pathogens.” In the postharvest period, these alterations cause deterioration of the fruit from harvesting until being consumed or processed (Viñas et al. 2013). Losses due to postharvest diseases have been estimated at around 20% in developed countries, and up to 50% in developing countries (Janisiewicz and Korsten 2002). Hypersensitive response takes place during the first few minutes of interaction, at the pathogen’s point of entry and in adjacent cells. This occurs as a “suicide” program, active and organized, and not because of a passive cell collapse produced by the pathogen attack or toxins resulting from it (Torres et al. 2006). In this cellular process, protein kinase and phosphatase enzymes are activated, and there are also changes in membrane permeability and intracellular ion concentration (Zhao et al. 2005). At the same time, rapid activation of reactive oxygen species (ROS) occurs; this process includes the production of substances such as superoxide (O2–), hydroxyl radical (OH–) and hydrogen peroxide (H2O2), which are responsible for the oxidative burst that indicates that the host has successfully recognized the pathogen. This takes place in two phases: the first is transient and nonspecific and occurs in the first few minutes of interaction, while the second appears hours after the pathogen attack and is associated with the establishment of defenses (Grant and Loake 2000).
ROS can also form physical barriers at the infection site by cross-linking of cell wall glycoproteins or via oxidative cross-linking of precursors during biosynthesis of polymers such as lignin and suberin (Torres 2010). These polymers can also be directly toxic, degrading the wall of the fungi and bacteria (Treutter 2006). In this regard, Vilanova et al. (2012) observed lignin production in unripe apples infected with Penicillium digitatum and Penicillium expansum one day after inoculation; on the other hand, the lignin increases in unripe oranges occurred after 48 h with both pathogens, decreasing in ripe fruits (Vilanova et al. 2012). Apples treated with ROS-inducing compounds, such as guaiacol, also showed increased lignin production, which reduced the incidence of P. expansum (Valentines et al. 2005). Valentines et al. (2005) observed in immature apples infected with P. expansum higher oxidation potential, and in turn they were less susceptible than mature fruits, suggesting that peroxidase is involved in pathogen resistance. It was also observed that oranges infected with P. digitatum reduced the levels of hydrogen peroxide, superoxide dismutase and catalase, after 72 h, confirming the role of the pathogen in the deactivation of the fruit’s ROS production mechanisms (Torres et al. 2011).
Most of these metabolites are from the phenylpropanoid pathways, such as flavonoids, isoflavones, coumarins, stilbenes, dihydrophenanthrenes and other phenols. In unripe avocados, a significant increase in epicatechin levels occur six hours after being infected with Colletotrichum gloeosporioides (Beno-Moualem and Prusky 2000). Increases in polymethoxylated flavones (PMFs), chlorogenic acid (CGA), and scoparone in oranges infected with P. digitatum were also observed (Ballester et al. 2013). Caffeic acid (CA) and CGA have also been proposed as resistance factors in peach against brown rot caused by Monilinia fructicola (Lee and Bostock 2007). CA is able to affect the virulence of this pathogen through cellular redox regulation, increasing the activity of glutathione reductase, which in turn can maintain a high proportion of reduced glutathione (GSH) to oxidized glutathione (GSSG) in the cell (Chiu et al. 2013). These compounds are also capable of inhibiting the expression of virulence genes such as mfcut1 and mfpg1, which encode for a cutinase and a polygalacturonase, respectively, and they also inhibit the formation of appressoria (Lee and Bostock 2007).
Conclusion
Plants being sessile entities are often subjected to varied biotic stresses. They have developed an alternative defense mechanism that involves a vast variety of secondary metabolites to serve as tools to cope up with various stress conditions. The exposure of plant cells to biotic stresses initiates multilevel reaction cascades that consequently leads to production and accumulation of various secondary metabolites (phenolic, flavonoids, phytoalexins and pathogenic related proteins). Various enzymatic and non-enzymatic molecules like peroxidase, polyphenol oxidase, phenylalanine ammonia-lyase and chitinase comprising the antioxidative defense system comes into play to counteract the undesirable effect of ecological stresses. In the present review, the influence of different biotic factors including herbivory and pathogenesis on the production of secondary metabolites and their roles in stress tolerance mechanism in plants are summarized.
References
Abu-Shahba MS, Mansour MM, Mohamed HI, Sofy MR (2021) Comparative cultivation and biochemical analysis of iceberg lettuce grown in sand soil and hydroponics with or without microbubble and microbubble. J Soil Sci Plant Nutr 21:389–403
Abu-Shahba MS, Mansour MM, Mohamed HI, Sofy MR (2022) Biosorptive Removal of Cadmium Ions from hydroponic Solution with indigenous garlic peel and mercerized garlic peel on lettuce productivity. Sci Horti 293:110727
Adel MM, Sehnal F, Jurzysta M (2000) Effects of alfalfa saponins on the moth Spodoptera littoralis. J Chem Ecol 26:1065–1078
Adrian M, Jeandet P, Veneau J, Westo LA, Bessis R (1997) Biological activity of resveratrol, a stilbenic compound from grapevines, against Botrytis cinerea, the causal agent for gray mold. J Chem Ecol 23:1689–1702
Ahmad G, Khan AA, Mohamed HI (2021a) Impact of the low and high concentrations of fly ash amended soil on growth, physiological response and yield of pumpkin (Cucurbita moschata Duch. Ex Poiret L.). Environ Sci Poll Res 28:17068–17083
Ahmad G, Khan A, Khan AA, Ali A, Mohhamad HI (2021b) Biological control: a novel strategy for the control of the plant parasitic nematodes. Antonie Van Leeuwenhoek 114:885–912
Ahuja I, Kissen R, Bones AM (2012) Phytoalexins in defense against pathogens. Trends Plant Sci 17:73–90
Akladious SA, Mohamed HI (2017) Physiological role of exogenous nitric oxide in improving performance, yield and some biochemical aspects of sunflower plant under zinc stress. Acta Biol Hung 68(1):101–114
Akula R, Ravishankar GA (2011) Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal Behav 6:1720–1731
Alejandro S, Lee Y, Tohge T, Sudre D, Osorio S, Park J, Bovet L, Geldner N, Fernie AR, Martinoia E (2012) AtABCG29 is a monolignol transporter involved in lignin biosynthesis. Curr Biol 22:1207–1212
Alhanout K, Malesinki S, Vidal N, Peyrot V, Rolain JM, Brunel JM (2010) New insights into the antibacterial mechanism of action of squalamine. J Antimicrob Chemother 65:1688–1693
Ali S, Ganai BA, Kamili AN, Bhat AA, Mir ZA, Bhat JA, Tyagi A, Islam ST, Mushtaq M, Yadav P, Rawat S (2018) Pathogenesis-related proteins and peptides as promising tools for engineering plants with multiple stress tolerance. Microbiol Res 212:29–37
Aly AA, Mansour MTM, Mohamed HI, Abd-Elsalam KA (2012) Ascorbic acid of seeds and proteins of leaves as biochemical markers for resistance of flax to powdery mildew disease. Romanian Agri Res 29:63–69
Aly AA, Mohamed HI, Mansour MTM, Omar MR (2013) Suppression of powdery mildew on flax by foliar application of essential oils. J Phytopathol 161:376–381
Aly AA, Mansour MTM, Mohamed HI (2017) Association of increase in some biochemical components with flax resistance to powdery mildew. Gesunde Pflanz 69(1):47–52
Amer A, Ghoneim M, Shoala T, Mohamed HI (2021) Comparative studies on French basil (Ocimum basilicum L. cv. Grand verde) as affected by alternatives spraying with humic, salicylic, and glycyrrhizic acids and their nanocomposites. Environ Sci Pollu Res 28:47196–47212
Anttila AK, Pirttilä AM, Häggman H, Harju A, Venäläinen M, Haapala A, Holmbom B, Julkunen TR (2013) Condensed conifer tannins as antifungal agents in liquid culture. Holzforschung 67(7):825–832
Atkinson NJ, Jain R, Urwin PE (2015) The response of plants to simultaneous biotic and abiotic stress. Combined stresses in plants. Springer, Cham, pp 181–201
Balint-Kurti P (2019) The plant hypersensitive response: concepts, control and consequences. Mol Plant Pathol 20(8):1163–1178
Ballester AR, Lafuente MT, González-Candelas L (2013) Citrus phenylpropanoids and defence against pathogens. Part II: Gene expression and metabolite accumulation in the response of fruits to Penicillium digitatum infection. Food Chem 136:285–291
Bano C, Singh NB, Sunaina S (2016) Differential responses of pea seedlings to salicylic acid under UV‑B stress. Trop Plant Res 3(3):586–591
Bano C, Amist N, Singh NB, Sunaina S (2017) UV‑B radiation escalates allelopathic effect of benzoic acid on Solanum lycopersicum L. Sci Hort 220:199–205
Bartwal A, Mall R, Lohani P, Guru SK, Arora S (2013) Role of secondary metabolites and brassinosteroids in plant defense against environmental stresses. J Plant Growth Regul 32(1):216–232
Bashandy SR, Abd-Alla MH, Dawood MF (2020) Alleviation of the toxicity of oily wastewater to canola plants by the N2-fixing, aromatic hydrocarbon biodegrading bacterium Stenotrophomonas maltophilia-SR1. Applied Soil Ecol 154:103654
Belofsky G, Kolaczkowski M, Adams E, Schreiber J, Eisenberg V, Coleman CM, Zou Y, Ferreira D (2013) Fungal ABC transporter-associated activity of isoflavonoids from the root extract of Dalea formosa. J Nat Prod 76(5):915–925
Beno-Moualem D, Prusky D (2000) Early events during quiescent infection development by Colletotrichum gloeosporioides in unripe avocado fruits. Phytopathology 90:553–559
Bi B, Tang J, Han S, Guo J, Miao Y (2017) Sinapic acid or its derivatives interfere with abscisic acid homeostasis during Arabidopsis thaliana seed germination. BMC Plant Biol 17(1):99
Buonaurio R, Moretti C, da Silva DP, Cortese C, Ramos C, Venturi V (2015) The olive knot disease as a model to study the role of interspecies bacterial communities in plant disease. Front Plant Sci 6:434
Caretto S, Linsalata V, Colella G, Mita G, Lattanzio V (2015) Carbon fluxes between primary metabolism and phenolic pathway in plant tissues under stress. Inter J Mol Sci 16:26378–26394
Carrington Y, Guo J, Le CH, Fillo A, Kwon J, Tran LT, Ehlting J (2018) Evolution of a secondary metabolic pathway from primary metabolism: shikimate and quinate biosynthesis in plants. Plant J 95:823–833
Caruso F, Mendoza L, Castro P, Cotoras M, Aguirre M, Matsuhiro B, Isaacs M, Rossi M, Viglianti A, Antonioletti R (2011) Antifungal activity of resveratrol against Botrytis cinerea is improved using 2‑Furyl derivatives. Plos One 6:10
Chepkirui C, Matasyoh JC, Wagara IN, Nakavuma J (2014) Antifungal activity of flavonoids isolated from Monanthotaxis littoralis against mycotoxigenic fungi from maize. Am J Chem Appl 1(4):54–60
Cheynier V, Comte G, Davies KM, Lattanzio V, Martens S (2013) Plant phenolics: recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol Biochem 72:1–20
Chiu CM, You BJ, Chou CM, Yu PL, Yu FY, Pan SM, Bostock RM, Chung KR, Lee MH (2013) Redox status-mediated regulation of gene expression and virulence in the brown rot pathogen Monilinia fructicola. Plant Pathol 62(4):809–819
Chungloo D, Tisarum R, Samphumphuang T, Sotesaritkul T, Cha-Um S (2021) Regulation of curcuminoids, photosynthetic abilities, total soluble sugar, and rhizome yield traits in two cultivars of turmeric (Curcuma longa) using exogenous foliar paclobutrazol. Not Bot Horti Agrobot Cluj Napoca 49(3):12445
Coleman JJ, Okoli I, Tegos GP, Holson EB, Wagner FF, Hamblin MR, Mylonakis E (2010) Characterization of plant-derived saponin natural products against Candida albicans. ACS Chem Biol 5:321–332
Cushnie T, Cushnie B, Lamb A (2014) Alkaloids: an overview of their antibacterial, antibiotic-enhancing, and antivirulence activities. Inter J Antimicrob Agents 44:377–386
Davin LB, Jourdes M, Patten AM, Kim KW, Vassão DG, Lewis NG (2008) Dissection of lignin macromolecular configuration and assembly: comparison to related biochemical processes in allyl/propenyl phenol and lignan biosynthesis. Nat Prod Rep 25:1015–1090
Dhawi F, El-Beltagi HS, Abdel-Mobdy YE, Salah SM, Ghaly IS, Abdel-Rahim EA, Soliman AM, Mohamed HI (2021) Synergistic impact of the pomegranate peels and its nanoparticles against the infection of Tobacco Mosaic Virus (TMV). Fresenius Environ Bull 30(1):731–746
Dhifi W, Bellili S, Jazi S, Bahloul N, Mnif W (2016) Essential oils’ chemical characterization and investigation of some biological activities: a critical rev med. Medicines 3(4):25
Dos Santos C, Vargas Á, Fronza N, Dos SJHZ (2017) Structural, textural and morphological characteristics of tannins from Acacia mearnsii encapsulated using sol-gel methods: applications as antimicrobial agents. Colloids Surf B Biointerfaces 151:26–33
Duke SO (2018) Interaction of chemical pesticides and their formulation ingredients with microbes associated with plants and plant pests. J Agric Food Chem 66(29):7553–7561
Easwar RD, Divya K, Prathyusha IVSN, Krishna RC, Chaitanya KV (2017) Insect-resistant plants. In: Current developments in biotechnology and bioengineering. Elsevier, Amsterdam, pp 47–74
El-Beltagi HS, Mohamed HI, Abdelazeem AS, Youssef R, Safwat G (2019a) GC-MS analysis, antioxidant, antimicrobial and anticancer activities of extracts from Ficus sycomorus fruits and leaves. Not Bot Horti Agrobot Cluj Napoca 47(2):493–505
El-Beltagi HS, Mohamed HI, Elmelegy AA, Eldesoky SE, Safwat G (2019b) Phytochemical screening, antimicrobial, antioxidant, anticancer activities and nutritional values of cactus (Opuntia ficus Indicia) pulp and peel. Fresenius Environ Bull 28(2A):1534–1551
El-Mahdy OM, Mohamed HI, Mogazy AM (2021) Biosorption effect of Aspergillus niger and Penicillium chrysosporium for Cd and Pb contaminated soil and their physiological effects on Vicia faba L. Environ Sci Pollut Res 28(47):67608–67631
El-Rahman ASS, Mohamed HI (2014) Application of benzothiadiazole and Trichoderma harzianum to control faba bean chocolate spot disease and their effect on some physiological and biochemical traits. Acta Physiol Plant 36(2):343–354
El-Rahman ASS, Mazen MM, Mohamed HI, Mahmoud NM (2012) Induction of defence related enzymes and phenolic compounds in lupin (Lupinus albus L.) and their effects on host resistance against Fusarium wilt. European J Plant Pathol 134(1):105–116
Fahed L, Beyrouthy ME, Ouaini N, Eparvier V, Stien D, Vitalini S, Iriti M (2021) Antimicrobial activity and synergy investigation of Hypericum scabrum essential oil with antifungal drugs. Molecules 26:6545
Fang X, Wang L, Ishikawa R (2019) Arabidopsis FLL2 promotes liquid–liquid phase separation of polyadenylation complexes. Nature 569:265–269
Freeman BC, Beattie GA (2008) An overview of plant defenses against pathogens and herbivores. Plant Pathol Microbiol. https://doi.org/10.1094/phi-i-0226-01
Gallego-Giraldo L, Escamilla-Trevino L, Jackson LA, Dixon RA (2011a) Salicylic acid mediates the reduced growth of lignin down-regulated plants. Proc Natl Acad Sci U S A 108:20814–20819
Gallego-Giraldo L, Jikumaru Y, Kamiya Y, Tang Y, Dixon RA (2011b) Selective lignin downregulation leads to constitutive defense response expression in alfalfa (Medicago sativa L.). New Phytol 190:627–639
Ghasemzadeh A, Ghasemzadeh N (2011) Flavonoids and phenolic acids: role and biochemical activity in plants and human. J Med Plants Res 5(31):6697–6703
Ghonaim MM, Mohamed HI, Omran AAA (2021) Evaluation of wheat salt stress tolerance using physiological parameters and retrotransposon-based markers. Genet Resour Crop Evol 68:227–242
Giebel J (1982) Mechanism of resistance to plant nematodes. Annu Rev Phytopathol 20(1):257–279
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43:205–227
Goleniowski M, Bonfill M, Cusido R, Palazón J (2013) Phenolic acids. In: Ramawat K, Mérillon JM (eds) Natural products. Springer, Berlin/Heidelberg
Grant JJ, Loake GJ (2000) Role of reactive oxygen intermediates and cognate redox signaling in disease resistance. Plant Physiol 124:21–29
Gruiz K (1996) Fungitoxic activity of Saponins: practical use and fundamental principles, saponins used in traditional and modern medicine. Springer, Berlin, pp 527–534
Hamed MM, El-Mobdy AMA, Kamel MT, Mohamed HI, Bayoumi AE (2019) Phytochemical and biological activities of two asteraceae plants Senecio vulgaris and Pluchea dioscoridis L. Pharmacology 2:101–121
Hardoim PR, Van Overbeek LS, Berg G, Pirttilä AM, Compant S, Campisano A, Sessitsch A (2015) The hidden world within plants: ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol Mol Biol Rev 79(3):293–320
Harmatha J, Dinan L (2003) Biological activities of lignans and stilbenoids associated with plant–insect chemical interactions. Phytochem Rev 2:321–330
He Y, Hu Z, Li A, Zhu Z, Yang N, Ying Z, He J, Wang C, Yin S, Cheng S (2019) Recent advances in biotransformation of saponins. Molecules 24:2365
Helmi A, Mohamed HI (2016) Biochemical and ulturasturctural changes of some tomato cultivars to infestation with Aphis gossypii Glover (Hemiptera: Aphididae) at Qalyubiya, Egypt. Gesunde Pflanz 68:41–50
Hernandez-Cumplido J, Giusti MM, Zhou Y, Kyryczenko-Roth V, Chen YH, Rodriguez-Saona C (2018) Testing the ‘plant domestication-reduced defense’ hypothesis in blueberries: the role of herbivore identity. Arthropod Plant Interact 12(4):483–493
Hodaei M, Rahimmalek M, Arzani A, Talebi M (2018) The effect of water stress on phytochemical accumulation, bioactive compounds and expression of key genes involved in flavonoid biosynthesis in Chrysanthemum morifolium L. Ind Crops Prod 120:295–304
Hussain T, Khan AA, Mohamed HI (2022) Potential efficacy of biofilm-forming biosurfactant Bacillus firmus HussainT-Lab.66 against Rhizoctonia solani and mass spectrometry analysis of their metabolites. Int J Peptide Res Therap 28:3. https://doi.org/10.1007/s10989-021-10318-5
Isah T (2019) Stress and defense responses in plant secondary metabolites production. Biol Res 52:39
Jan R, Asaf S, Numan M, Kim KM (2021) Plant secondary metabolite biosynthesis and transcriptional regulation in response to biotic and abiotic stress conditions. Agronomy 11:968
Janisiewicz WJ, Korsten L (2002) Biological control of postharvest diseases of fruits. Ann Rev Phytopathol 40:411–441
Jha Y (2018) Induction of anatomical, enzymatic, and molecular events in maize by PGPR under biotic stress. P 125–141. In: Meena V (ed) Role of rhizospheric microbes in soil. Springer, Singapore, p 398
Jha Y (2019a) Higher induction of defense enzymes and cell wall reinforcement in maize by root associated bacteria for better protection against Aspergillus niger. J Plant Prot Res 59(3):341–349
Jha Y (2019b) Endophytic bacteria mediated anti-autophagy and induced catalase, β‑1, 3‑glucanases gene in paddy after infection with pathogen Pyricularia grisea. Ind Phytopathol 72(1):99–106
Jha Y (2019c) Endophytic bacteria-mediated regulation of secondary metabolites for the growth induction in Hyptis suaveolens under stress. In: Egamberdieva D, Tiezzi A (eds) Medically important plant biomes: source of secondary metabolites. Microorganisms for Sustainability, vol 15. Springer, Singapore
Jha Y (2019d) Regulation of water status, chlorophyll content, sugar, and photosynthesis in maize under salinity by mineral mobilizing bacteria. In: Book photosynthesis productivity and environmental stress, pp 75–93
Jha Y (2020) Plant microbiomes with phytohormones attribute for plant growth and adaptation under the stress conditions. In: Agriculture, microorganisms for sustainability, pp 85–103
Jha Y (2021) Macrophytes as a potential tool for crop production by providing nutrient as well as protection against common phyto pathogen. Highlights Biosci. https://doi.org/10.36462/H.BioSci.202103
Jha Y, Yadav AN (2021) Piriformospora indica: biodiversity, ecological significances, and biotechnological applications for agriculture and allied sectors. In: Abdel-Azeem AM, Yadav AN, Yadav N, Usmani Z (eds) Industrially important fungi for sustainable development. Fungal biology. Springer, Cham
Jha Y, Dehury B, Kumar SPJ, Chaurasia A, Singh UB, Yadav MK, Angadi UB, Ranjan R, Tripathy M, Subramanian RB, Kumar S, Simal-Gandara J (2021) Delineation of molecular interactions of plant growth promoting bacteria induced β‑1,3‑glucanases and guanosine triphosphate ligand for antifungal response in rice: a molecular dynamics approach. Mol Biol Rep. https://doi.org/10.1007/s11033-021-07059-5
Kanno H, Hasegawa M, Kodama O (2012) Accumulation of salicylic acid, jasmonic acid and phytoalexins in rice, Oryza sativa, infested by the white-backed planthopper, Sogatella furcifera (Hemiptera: Delphacidae). Appl Entomol Zool 47(1):27–34
Khare S, Singh NB, Singh A, Hussain I, Niharika K, Yadav V, Bano C, Yadav RK, Amist N (2020) Plant secondary metabolites synthesis and their regulations under biotic and abiotic constraints. J Plant Biol 63(3):203–216
Kisiriko M, Anastasiadi M, Terry LA, Yasri A, Beale MH, Ward JL (2021) Phenolics from medicinal and aromatic plants: characterisation and potential as biostimulants and bioprotectants. Molecules 26:6343
Klarzynski O, Plesse B, Joubert JM, Yvin JC, Kopp M, Kloareg B, Fritig B (2000) Linear β‑1, 3 glucans are elicitors of defense responses in tobacco. Plant Physiol 124(3):1027–1038
Korchowiec B, Gorczyca M, Wojszko K, Janikowska M, Henry M, Rogalska E (2015) Impact of two different saponins on the organization of model lipid membranes. Biochim Biophys Acta Biomembr 1848:1963–1973
Kumar CM, Singh SA (2015) Bioactive lignans from sesame (Sesamum indicum L.): evaluation of their antioxidant and antibacterial effects for food applications. J Food Sci Technol 52(5):2934–2941
Laohavisit A, Wakatake T, Ishihama N (2020) Quinone perception in plants via leucine-rich-repeat receptor-like kinases. Nature 587:92–97
Latef AAAH, Dawood MF, Hassanpour H, Rezayian M, Younes NA (2020) Impact of the static magnetic field on growth, pigments, osmolytes, nitric oxide, hydrogen sulfide, phenylalanine ammonia-lyase activity, antioxidant defense system, and yield in lettuce. Biology 9(7):172
Lee MH, Bostock RM (2007) Fruit exocarp phenols in relation to quiescence and development of Monilinia fructicola infections in Prunus spp.: a role for cellular redox? Phytopathology 97(3):269–277
Lee G, Joo Y, Kim SG, Baldwin IT (2017) What happens in the pith stays in the pith: tissue -localized defense responses facilitate chemical niche differentiation between two spatially separated herbivores. Plant J 92:414–425
Leváková L, Lacko-Bartošová M (2017) Phenolic acids and antioxidant activity of wheat species: a review. Agriculture 63(3):92–101
Li B, Fan R, Sun G (2021) Flavonoids improve drought tolerance of maize seedlings by regulating the homeostasis of reactive oxygen species. Plant Soil 461:389–405
Lincoln JE, Sanchez JP, Zumstein K, Gilchrist DG (2018) Plant and animal PR1 family members inhibit programmed cell death and suppress bacterial pathogens in plant tissues. Mol Plant Path 19(9):2111–2123
Livne B et al (1997) TMV-induced expression of tobacco β‑glucanase promoter activity is mediated by a single inverted, GCC motif. Plant Sci 130(2):159–169
Long SY, Li CL, Hu J, Zhao QJ, Chen D (2018) Indole alkaloids from the aerial parts of Kopsia fruticosa and their cytotoxic, antimicrobial and antifungal activities. Fitoterapia 129:145–149
Lusso M, Kuc J (1995) Evidence for transcriptional regulation of β‑1, 3‑glucanase as it relates to induced systemic resistance of tobacco to blue mold. Mol Plant Microbe Interact 8(3):473–475
Maatalah MB, Bouzidi NK, Bellahouel S, Merah B, Fortas Z, Soulimani R, Saidi S, Derdour A (2012) Antimicrobial activity of the alkaloids and saponin extracts of Anabasis articulate. J Biotechnol Pharm Res 3:54–57
Maddox CE, Laur LM, Tian L (2010) Antibacterial activity of phenolic compounds against the Phytopathogen Xylella fastidiosa. Curr Microbiol 60(1):53–58
Maeda H, Dudareva N (2012) The shikimate pathway and aromatic amino Acid biosynthesis in plants. Annu Rev Plant Biol 63:73–105
Mandhania S, Sangwan RS, Siwach SS, Pundir SR, Sangwan O, Janu A (2018) Role of biochemical constituents and minerals against cotton leaf curl disease in cotton. J Environ Biol 39:221–227
Mansour MTM, Aly AA, Habeb MM, Mohamed HI (2020) Control of cotton seedling damping-off by treating seed with inorganic salts. Gesunde Pflanz 72:273–283
Marques JP, Hoy JW, Appezzato-da-Glória B, Viveros AF, Vieira ML, Baisakh N (2018) Sugarcane cell wall-associated defense responses to infection by Sporisorium scitamineum. Frontiers Plant Sci 23(9):698
Matsuura HN, Fett-Neto AG (2015) Plant alkaloids: main features, toxicity, and mechanisms of action. Plant Toxins. https://doi.org/10.1007/978-94-007-6728-7_2-1
Mayer AM, Staples RC, Gil-ad NL (2001) Mechanisms of survival of necrotrophic fungal plant pathogens in hosts expressing the hypersensitive response. Phytochemistry 58(1):33–41
Mendes IDS, Rezende MOO (2014) Assessment of the allelopathic effect of leaf and seed extracts of Canavalia ensiformis as post emergent bioherbicides: a green alternative for sustainable agriculture. J Environ Sci Health Part B 49(5):374–380
Mhlongo MI, Piater LA, Madala NE, Labuschagne N, Dubery IA (2018) The chemistry of plant–microbe interactions in the rhizosphere and the potential for metabolomics to reveal signaling related to defense priming and induced systemic resistance. Front Plant Sci 9:112
Miele S, Tegli S, Izquierdo CG, Cerboneschi M, Bargiacchi E (2019) Hydrolysable tannins in agriculture. Tannins-structural properties, biological properties and current knowledge
Mierziak J, Kostyn K, Kulma A (2014) Flavonoids as important molecules of plant interactions with the environment. Molecules 19(10):16240–16265
Mogazy AM, Mohamed HI, El-Mahdy OM (2022) Calcium and iron nanoparticles: a positive modulator of innate immune responses in strawberry plants against Botrytis cinerea. Process Biochem 115:128–145
Mohamed HI (2011). Molecular and Biochemical Studies on the Effect of Gamma Rays on Lead Toxicity in Cowpea (Vigna sinensis) Plants. Biol Trace Elem Res 144:1205–1218
Mohamed H, Abd-El Hameed AG (2014) Molecular and biochemical markers of some Vicia faba L. genotype in response to storage insect pests infestation. J Plant Int 9(1):618–626
Mohamed HI, Abdel-Hamid AME (2013) Molecular and biochemical studies for heat tolerance on four cotton genotypes (Gossypium hirsutum L.). Romanian Biotechnol Lett 18(6):7223–7231
Mohamed HI, Akladious SA (2014). Influence of garlic extract on enzymatic and non enzymatic antioxidants in soybean plants (Glycine max) grown under drought stress. Life Sci J 11(3s):46–58
Mohamed HI, Elsherbiny EA, Abdelhamid MT (2016) Physiological and biochemical responses of Vicia faba plants to foliar application with zinc and iron. Gesunde Pflanz 68:201–212
Mohamed HI, Akladious SA, Ashry NA (2018) Evaluation of water stress tolerance of soybean using physiological parameters and retrotransposon-based markers. Gesunde Pflanz 70:205–215
Mohamed HI, Mohammed AHM, Mohamed NM, Ashry NA, Zaky LM, Mogazy AM (2021) Comparative effectiveness of potential elicitors of soybean plant resistance against Spodoptera Littoralis and their effects on secondary metabolites and antioxidant defense system. Gesunde Pflanz 73:273–285
Mohammadi M, Kazemi H (2002) Changes in peroxidase and polyphenol oxidase activities in susceptible and resistant wheat heads inoculated with Fusarium graminearum and induced resistance. Plant Sci 162:491–498
Moses T, Papadopoulou KK, Osbourn A (2014) Metabolic and functional diversity of saponins, biosynthetic intermediates and semi-synthetic derivatives. Crit Rev Biochem Mol Boil 49(6):439–462
Moustafa-Farag M, Mohamed HI, Mahmoud A, Elkelish A, Misra AN, Guy KM, Kamran M, Ai S, Zhang M (2020) Salicylic acid stimulates antioxidant defense and osmolyte metabolism to alleviate oxidative stress in watermelons under excess boron. Plants 9(6):724
Naeem M, Basit A, Ahmad I, Mohamed HI, Wasila H (2020) Effect of salicylic acid and salinity stress on the performance of tomato. Gesunde Pflanz 72:393–402
Narayani M, Srivastava S (2017) Elicitation: a stimulation of stress in in vitro plant cell/tissue cultures for enhancement of secondary metabolite production. Phytochem Rev 16:1227–1252
Nawrot J, Harmatha J (2012) Phytochemical feeding deterrents for stored product insect pests. Phytochem Rev 11:543–566
Ogawa S, Yazaki Y (2018) Tannins from Acacia mearnsii De wild. Bark: tannin determination and biological activities. Molecules 23(4):E837
Ohri P, Pannu SK (2010) Effect of phenolic compounds on nematodes—a review. J Appl Nat Sci 2(2):344–350
Panche AN, Diwan AD, Chandra SR (2016) Flavonoids: an overview. J Nutr Sci 5:e47
Passardi F, Cosio C, Penel C, Dunand C (2005) Peroxidases have more functions than a Swiss army knife. Plant Cell Rep 24:255–265
Peperidou A, Pontiki E, Hadjipavlou-Litina D, Voulgari E, Avgoustakis K (2017) Multifunctional cinnamic acid derivatives. Molecules 22(8):1247
Pott DM, Osorio S, Vallarino JG (2019) From central to specialized metabolism: an overview of some secondary compounds derived from the primary metabolism for their role in conferring nutritional and organoleptic characteristics to fruit. Front Plant Sci 28(10):835
Pusztahely T, István H, Pócsi IJ (2017) Plant-fungal interactions: special secondary metabolites of the biotrophic, necrotrophic, and other specific interactions. In: Fungal metabolites. Academic Press, London, pp 1–58
Qasim M, Islam W, Ashraf HJ, Ali I, Wang L (2020) Saponins in insect pest control. In: Co-evolution of secondary metabolites, pp 897–924
Rana KI, Farhana NA (2019) Chitinases potential as biocontrol. Biomed J Sci Tech Res 14(5):2629
Reen FJ, Gutiérrez-Barranquero JA, Parages ML, Gara OF (2018) Coumarin: a novel player in microbial quorum sensing and biofilm formation inhibition. Appl Microbiol Biotechnol 102(5):2063–2073
Saddique M, Kamran M, Shahbaz M (2018) Differential responses of plants to biotic stress and the role of metabolites. In: Plant metabolites and regulation under environmental stress. Elsevier, San Diego, pp 69–87
Sade D, Shriki O, Cuadros-Inostroza A, Tohge T, Semel Y, Haviv Y, Brotman Y (2014) Comparative metabolomics and transcriptomics of plant response to tomato yellow leaf curl virus infection in resistant and susceptible tomato cultivars. Metabolomics 11(1):81–97
Santos PM, Batista DL, Ribeiro LA, Boffo EF, de Cerqueira MD, Martins D, de Castro RD, de Souz-Neta LC, Pinto EL, Zambotti-Villela E, Colepicolo P, Fernandez LG, Canuto GA, Ribeiro PR (2018) Identification of antioxidant and antimicrobial compounds from the oilseed crop Ricinus communis using a multiplatform metabolite profiling approach. Ind Crop Prod 124:834–844
Schuetz M, Benske A, Smith RA, Watanabe Y, Tobimatsu Y, Ralph J, Demura T, Ellis B, Samuels AL (2014) Laccases direct lignification in the discrete secondary cell wall domains of protoxylem. Plant Physiol 166:798–807
Serpa R, Franca EJ, Furlaneto-Maia L, Andrade CG, Diniz A, Furlaneto MC (2012) In vitro antifungal activity of the flavonoid baicalein against Candida species. J Med Microbiol 61:1704–1708
Sharma A, Shahzad B, Rehman A, Bhardwaj R, Landi M, Zheng B (2019) Response of phenylpropanoid pathway and the role of polyphenols in plants under abiotic stress. Molecules 24(13):2452
Sherwood P, Bonello P (2013) Austrian pine phenolics are likely contributors to systemic induced resistance against Diplodia pinea. Tree Physiol 33(8):845–854
Silva AX, Jander G, Samaniego H, Ramsey JS, Figueroa CC (2012) Insecticide resistance mechanisms in the green peach aphid Myzus persicae (Hemiptera: aphididae) I: a transcriptomic survey. Plos One 7:e36366
Singh B, Kaur A (2018) Control of insect pests in crop plants and stored food grains using plant saponins: a review. LWT 87:93–101
Singh B, Sharma RA (2015) Plant terpenes: defense responses, phylogenetic analysis, regulation and clinical applications. 3 Biotech 5(2):129–151
Singh RK, Rai N, Singh M, Singh SN, Srivastava K (2014) Selection of tomato genotypes resistant to tomato leaf curl virus disease using biochemical and physiological markers. J Agric Sci 153(04):646–655
Singh S, Kaur I, Kariyat R (2021) The multifunctional roles of polyphenols in plant-herbivore interactions. Int J Mol Sci 22:1442
Smeriglio A, Barreca D, Bellocco E, Trombetta D (2017) Proanthocyanidins and hydrolysable tannins: occurrence, dietary intake and pharmacological effects. Br J Pharmacol 174(11):1244–1262
Sofy MR, Mohamed HI, Dawood MFA, Abu-Elsaoud AM, Soliman MH (2021a) Integrated usage of Trichoderma harzianum and biochar to ameliorate salt stress on spinach plants. Arch Agronomy Soil Sci. https://doi.org/10.1080/03650340.2021.1949709
Sofy AR, Sofy MR, Hmed AA, Dawoud RA, Refaey EE, Mohamed HI, El-Dougdoug NK (2021b) Molecular characterization of the Alfalfa mosaic virus infecting Solanum melongena in Egypt and the control of its deleterious effects with melatonin and salicylic acid. Plants 10(3):459
Soltys D, Krasuska U, Bogatek R, Gniazdow A (2013) Allelochemicals asbio-herbicides—present and perspectives. In: Price AJ, Kelton JA (eds) Herbicides—current research and case studies in use. InTech, pp 517–542
Sreij R, Dargel C, Schweins R, Pr’evost S, Dattani R, Hellweg T (2019) Aescincholesterol complexes in DMPC model membranes: a DSC and temperature dependent scattering study. Sci Rep 9:1–15
Stevenson PC, Nicolson SW, Wright GA (2017) Plant secondary metabolites in nectar: impacts on pollinators and ecological functions. Funct Ecol 31:65–75
Suzuki K, Nomura I, Ninomiya M, Tanaka K, Koketsu M (2018) Synthesis and antimicrobial activity of b‑carboline derivatives with N2-alkyl modifications. Bioorg Med Chem Lett 28:2976–2978
Tahsili J, Sharifi M, Safaie N, Esmaeilzadeh-Bahabadi S, Behmanesh M (2014) Induction of lignans and phenolic compounds in cell culture of Linum album by culture filtrate of Fusarium graminearum. J Plant Interact 9:412–417
Thawabteh A, Juma S, Bader M, Karaman D, Scrano L, Bufo SA, Karaman R (2019) The biological activity of natural alkaloids against herbivores, cancerous cells and pathogens. Toxins 11(11):656
Tiku AR (2018) Antimicrobial compounds and their role in plant defense. In: Singh A, Singh I (eds) Molecular aspects of plant-pathogen interaction. Springer, Singapore, pp 283–307
Tiku AR (2020) Antimicrobial compounds (phytoanticipins and phytoalexins) and their role in plant defense. In: Mérillon JM, Ramawat K (eds) Co-evolution of secondary metabolites. Reference series in phytochemistry. Springer, Cham
Torres MA (2010) ROS in biotic interactions. Physiol Plant 138:414–429
Torres MA, Jones JDG, Dangl JL (2006) Reactive oxygen species signaling in response to pathogens. Plant Physiol 141:373–378
Torres R, Teixidó N, Usall J, Abadias M, Mir M, Larrigaudiere C, Viñas I (2011) Antioxidant activity of oranges after infection with the pathogen Penicillium digitatum or treatment with the biocontrol agent Pantoea agglomerans CPA‑2. Biol Control 57:103–109
Treutter D (2006) Significance of flavonoids in plant resistance: a review. Environ Chem Lett 4:147–157
Valentines MC, Vilaplana R, Torres R, Usall J, Larrigaudiere C (2005) Specific roles of enzymatic browning and lignifications in apple disease resistance. Postharvest Biol Technol 36:227–234
Valletta A, Iozia LM, Leonelli F (2021) Impact of environmental factors on stilbene biosynthesis. Plants 10(1):90
Van Loon LC (1997) Induced resistance in plants and the role of pathogenesis-related proteins. Eur J Plant Pathol 103:753–765
Vilanova L, Teixidó N, Torres R, Usall J, Viñas I (2012) The infection capacity of P. expansum and P. digitatum on apples and histochemical analysis of host response. Int J Food Microbiol 157:360–367
Viñas I, Abadias M, Teixidó N, Usall J, Torres R (2013) Aspectos Básicos de la Patología de la Poscosecha. In: Viñas I, Recasens I, Usall J, Graell J (eds) Poscosecha de manzana, pera y melocotón. Mundi Prensa, Madrid, pp 203–242
Wan Z, Hu D, Li P, Xie D, Gan X (2015) Synthesis, antiviral bioactivity of novel 4‑thioquinazoline derivatives containing chalcone moiety. Molecules 20(7):11861–11874
Wang L, Sun R, Zhang Q, Luo Q, Zeng S, Li X, Liu Z (2018) An update on polyphenol disposition via coupled metabolic pathways. In: Expert opinion on drug metabolism & toxicology. Ashley, London, pp 1–15
War AR, Paulraj MG, Ahmad T, Buhroo AA, Hussain B, Ignacimuthu S, Sharma HC (2012) Mechanisms of plant defense against insect herbivores. Plant Signal Behav 7:1306–1320
War AR, Taggar GK, Hussain B, Taggar MS, Nair RM, Sharma HC (2018) Plant defence against herbivory and insect adaptations. Aob Plants 10(4):ply37
Xie M, Zhang J, Tschaplinski TJ, Tuskan GA, Chen J‑G, Muchero W (2018) Regulation of lignin biosynthesis and its role in growth-Defense Tradeoffs. Front Plant Sci 9:1427
Yan H, Xie N, Zhong C, Su A, Hui X, Zhang X, Jin Z, Li Z, Feng J, He J (2018) Aphicidal activities of Amaryllidaceae alkaloids from bulbs of Lycoris radiata against Aphis citricola. J Ind Crop Prod 123:372–378
Younes NA, Dawood MF, Wardany AA (2019) Biosafety assessment of graphene nanosheets on leaf ultrastructure, physiological and yield traits of Capsicum annuum L. and Solanum melongena L. Chemosphere 228:318–327
Younes NA, Dawood MF, Wardany AA (2020) The phyto-impact of fluazinam fungicide on cellular structure, agro-physiological, and yield traits of pepper and eggplant crops. Environ Sci Pollu Res 27(15):18064–18078
Zabalza A, Orcaray L, Fernández-Escalada M, Zulet-González A, Royuela M (2017) The pattern of shikimate pathway and phenylpropanoids after inhibition by glyphosate or quinate feeding in pea roots. Pestic Biochem Physiol 141:96–102
Zakaryan H, Arabyan E, Oo A, Zandi K (2017) Flavonoids: promising natural compounds against viral infections. Arch Virol 162(9):2539–2551
Zhao J, Davis LC, Verpoorte R (2005) Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol Adv 23(4):283–333
Zuzarte M, Vale-Silva L, Goncalves MJ, Cavaleiro C, Vaz S, Canhoto J, Pinto E, Salgueiro L (2012) Antifungal activity of phenolic-rich Lavandula multifida L. essential oil. Eur J Clin Microbiol Infect Dis 31(7):1359–1366
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Y. Jha and H.I. Mohamed declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Jha, Y., Mohamed, H.I. Plant Secondary Metabolites as a Tool to Investigate Biotic Stress Tolerance in Plants: A Review. Gesunde Pflanzen 74, 771–790 (2022). https://doi.org/10.1007/s10343-022-00669-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10343-022-00669-4