Abstract
The soil environment is getting contaminated with heavy metals and other pollutants due to unattended anthropogenic and natural activities. Plants grown in such contaminated soils use different mechanisms to regulate and combat the deleterious effects of metal ions by producing several metabolites and antioxidants to combat oxidative damage. Among them, secondary metabolites actively participate in the alleviation of stress by acting as metal chelators or via direct scavenging of the reactive oxygen species. Plant-sourced secondary metabolites (SMs) are organic compounds that are highly inducible in stress experiencing plants. Though secondary metabolites are non-essential, but are directly involved in the healthy interaction of plants with the environment. As they strengthen the plants by making them adaptable to the environment and counteract the deleterious effects of pollutants. These SMs also act as basic immune-protector in medicinal plants for traditional medicine in different cultures of the world. The chemical structure of these metabolites mainly corresponds to the antioxidant behavior. Depending upon chemical structure, these metabolites can be classified into various types like phenolics, alkaloids, saponins, terpenes, lipids, carbohydrates, nitrogen, sulfur containing compounds, etc. Plant experiencing abiotic stress produces SMs from primary metabolites using the various pathways to help plants combat the stressed environment. Though mainly stress initiated, the production of these SMs in a plant is usually very less and varies among different plant species and affected by the strength of exogenous stimuli. Inorganic pollutants like mineral salts, gaseous toxins, heavy metals, pesticides, and aerosols are major factors involved in the initiation of SMs production in plants. Abiotic stress signals like methyl jasmonate, jasmonic acid, salicylic acid, calcium, polyamines, serotonin, abscisic acid, plant growth regulators, nutrient stress, and micro-drought serve as stimuli for SMs production. Cell culture technologies are a basic source of in vitro production of secondary metabolites for studying their effects on plants and for plant growth improvement purposes. This chapter addresses the nature and role of secondary metabolites in plant systems under abiotic stress conditions.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Agricultural and atmospheric contamination with heavy metal species and other inorganic pollutants like alkali salts is a major issue regarding food security and human health. Environmental risks linked with inorganic pollutants vary largely owing to complex interactions at extracellular and intracellular levels (Saha et al. 2017). Salts of the alkali group affect the physicochemical properties of the soil resulting in the interruption of the plant–soil–water relationships, nutrient use efficiency, bioavailability, and cycling. Toxic metal ions interact with soil colloids more strongly than salts depending on their speciation and elemental nature. Even at their low concentration and less mobility in the soil, they disrupt the metabolic processes by affecting the physiology of the plants. Heavy metals or trace elements are the group of metalloids having density and atomic number greater than 5 g cm−3 and 20, respectively (Alloway 2011). Some of these trace elements (zinc, copper, nickel, molybdenum, manganese, iron) are essential for the structural and biochemical processes in plants like proper growth, tissue development, electron transport, redox reactions, and many other metabolic processes (Andresen et al. 2018). While non-essential heavy metals including lead (Pb), mercury (Hg), cadmium (Cd), arsenic (As), etc. have been found to be toxic for plants growth with no known biological function along with food chain contamination even at minute concentrations (Shahid et al. 2017). Variability in spatial distribution and contamination of soils with heavy metals is owing to anthropogenic and natural sources, while the rapid increase in pollutants from last some decades in some ecosystems is typically linked with unmanaged anthropogenic activities (Sarma et al. 2011; Street 2012; Xiong et al. 2016a, b). Industrial effluents, mining, metallurgy, chemical fertilizers, urbanization, and transportation tend to increase the mass of pollutants in the environmental matrices (Megateli et al. 2009; De Lurdes Dinis and Fiúza 2010). The ultimate sink of these pollutants is the soil where they degrade the soil properties and badly affect the growth of plants. These toxic pollutants get absorbed into plants via root system, translocate to the aerial portion causing metabolic dysfunction and food chain contamination (Zheljazkov et al. 2006, 2008a, b; Baye and Hymete 2009; Chaiyarat et al. 2010; Li et al. 2010; Carrubba and Scalenghe 2012; Ebrahim et al. 2012; Shahid et al. 2017). Plants use different strategies to cope with these inorganic pollutants including physiological, morphological, genetic, and biochemical mechanisms (Sharma and Dietz 2009; Schreck et al. 2012).
Exposure to heavy metals results in overproduction of the reactive oxygen species, which are then detoxified by different transcriptional responses and epigenetic modifications (Dutta et al. 2018). Induction of heavy metals persuades the production of secondary metabolites (SMs) in the plants which help in mitigating their toxic effects. Primary metabolites like amino acids, carbohydrates, and lipids serve as base materials and are involved in the production of secondary metabolites (Hatami and Ghorbanpour 2016). Plant secondary metabolites have the least essential role in the functioning of the plants; however, they serve for the adaptive and defense interaction of the plant with its environment (Ramakrishna and Ravishankar 2011). About 100,000 types of secondary metabolites have been discovered so far with weight <150 kDa and contributing <1% of the plant total dry matter (Oksman-Caldentey and Inzé 2004). Depending on their biosynthetic pathways, they have been classified into nitrogen compounds (alkaloids) and nitrogen-deficient (terpenes and phenolics) (Kasote et al. 2015; Pagare et al. 2015). Accumulation of secondary metabolites in the plant tissues is often subjected to different types of stresses and signal molecules and gets regulated by many plant factors like evolution and genetic behavior, growing conditions, climate, mineral elements, and pollutants concentrations (Street 2012; Hatami and Ghorbanpour 2016; Yang et al. 2018). The induced stress of heavy metals and other inorganic molecules leads to the production of secondary metabolites through stimulation of the defensive system of the plants (Asgari Lajayer et al. 2017). SMs also add in the tastes, odors, toxins, and colors in plants (Mazid et al. 2011; Pagare et al. 2015) and are the distinctive sources of food additives, flavors, pharmaceutically active substances, and industrial biochemicals (Heitefuss 2011; Ramakrishna and Ravishankar 2011). Chemicals involved in the synthesis of SMs are calcium, salicylic acid, abscisic acid, jasmonates, polyamines, and nitric oxides (Tuteja and Sopory 2008). Plant secondary metabolites can also be synthesized using plant tissue cultures and it is a subject of much interest in research owing to their large applications (Gonçalves and Romano 2018). But their considerable yield and extraction present challenges that need to be solved yet as part of biological and phyto-chemical investigations. Successful production and extraction begin with cautious selection and preparation of the plants used. During this process, it is necessary to minimize the contamination that may get extracted with concerned compounds.
2 Classification of Secondary Metabolites
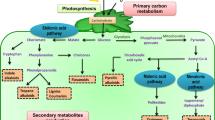
Among various organic molecules that are being synthesized by plants in response to stress signals is a special class called secondary metabolites. They are not necessary for cell life but have a role in making the plant adaptable to its surroundings. Having a unique structure with a carbon skeleton is the basic property of the SMs (Pagare et al. 2015). These SMs can be classified into various classes depending upon their chemical structure, composition, solubility, or pathway through which they are synthesized (McMurry 2014). Based upon structural composition SMs can be classified into enzymatic and non-enzymatic (antioxidants) (Hasanuzzaman et al. 2019). These antioxidants are very important in combating stresses in plants making them an integral part of plant biochemistry (Kapoor et al. 2019). Among enzymatic antioxidants, ascorbate peroxidase, dehydroascorbate reductase, monodehydroascorbate reductase, glutathione reductase, catalase, superoxide dismutase, glutathione peroxidase, peroxiredoxin, and glutathione-transferase are well known (Noctor et al. 2014; Hasanuzzaman et al. 2017a). While ascorbate, carotenoids, flavonoids, glutathione, and tocopherols are non-enzymatic antioxidants (Gill and Tuteja 2010). Based upon the biosynthetic pathway of SMs, another classification has been proposed in which SMs are divided into three major groups: terpenoids, phenolics, and nitrogenous/sulfur compounds. Terpenoids represent the biggest group of SMs in plants (Nagegowda 2010). They are biosynthesized via common origin from glycolytic or acetyl Co-A intermediates. Most of the diverse terpenes’ structures produced by plants are in their defense against feeding deterrents and are toxic in nature. Terpenes have been classified in accordance with the number of 5-carbon units present in their structure. Hence, it includes hemi (C-5), mono (C-10), sesqui (C-15), di (C-20), sester (C-25), tri (C-30), and tetra (C-40) terpenes (Nagegowda and Gupta 2020). Pyrethroid (terpenoid) occurring in the leaves of chrysanthemum species shows strong insecticidal effects and is an ingredient of many commercial insecticides owing to its low environmental persistency. In gymnosperms plants, commonly found terpenes are limonene, α-pinene, and myrcene. Similarly, sesquiterpenes including costunolides, abscisic acid; diterpenes including abietic acid, phorbol; tetraterpenes including carotenoids family and many high molecular weight polyterpenes have been reported as beneficial SMs in abiotic stress mitigation (Pagare et al. 2015).
A large variety of phenolic compounds are also produced in the plants as SMs for defense purposes (Colak et al. 2019; Jiang et al. 2019). These compounds contain phenolic acids, lignin, anthocyanidin (Taiz and Zeiger 2006), coumarin derived by shikimic acid pathways (Brooker et al. 2008), furano, psoralene (Ali et al. 2008), flavonoids, and isoflavonoids (Sreevidya 2006; Lake et al. 2009; Saviranta et al. 2010). Phenolic metabolites specially phenylpropanoids and flavonoids get oxidize by peroxidases and help in scavenging H2O2, phenolic/POX system (Michalak 2006). Several phenolic SMs have been reported in plants for stress tolerance like ascorbate peroxidase (Esmaeilzadeh et al. 2017), ascorbic acid (Maleki et al. 2017), catalase, dehydroascorbic acid, cytosolic dehydroascorbate reductase, glutathione reductase, glutathione, glutathione disulfide, monodehydroascorbate, monodehydroascorbate reductase, peroxidase, and superoxide dismutase (Michalak 2006; Blokhina et al. 2003; Kovacik et al. 2009a, b). Nitrogen-containing SMs include alkaloids, defensins, GSL, and N-glycosides, while sulfur-containing SMs include phytoalexins, allicin, S-glycosides, and thionins (Saito 2004; Grubb and Abel 2006; Halkier and Gershenzon 2006; Figueiredo et al. 2008; Street 2012). Some nitrogen-containing SMs also include phenolic acids (Taiz and Zeiger 2006).
3 Production of Secondary Metabolites in Plants
Most of the secondary metabolites are produced in plants by three pathways, via the shikimate pathway, polyketide pathway, and isoprenoid pathway. After the formation of the basic skeletons, SMs get modified according to the species-specific compounds. Aromatic compounds are mainly sourced from the shikimate pathway. While phenylpropanoid pathway is involved in the synthesis of lignin, anthocyanins, and flavonoids which are major SMs in stress conditions including heavy metal and alkali salts pollution (Pagare et al. 2015). Phenylpropanoid is one of the central metabolic pathways in terms of carbon flux as about 20% of the total metabolism in a plant cell goes through this pathway. Fundamental to SM products in this pathway is the enzyme phenylalanine ammonia-lyase which changes phenylalanine into trans-cinnamic acid through non-oxidative deamination. Isoprenoid pathway is another important pathway in this regard including terpenoids production which is 1/3 of the plants SMs (Pagare et al. 2015).
The production and concentration of the SMs in plants are dependent on the soil environment and plant resistance. Exposure to stress like inorganic pollutants and HMs can produce reactive oxygen species in the plants which are damaging to the cell functions. They can limit plant growth and productivity severely (Pandey et al. 2017). Expressions of certain genes get an increase in response to the ROS (Tuteja 2007; Nakashima et al. 2009; Roy 2016). These genes are involved in the production of SMs to combat with the ROS and help in their scavenging. A stress signal is responded when recognized at the cellular level in the form of SMs formation as a protective function. Edreva et al. (2007) reported phenyl amides and polyamines accumulation in the tobacco and bean plants under abiotic stress conditions suggesting the role of these SMs as antioxidants. Similarly, accumulation of anthocyanin is induced by several environmental stress signal including wounds, drought, nutrient deficiency, high-intensity light, blue light, UV-rays, and pathogen attack (Winkel-Shirley 2001; Truong et al. 2018; Hu et al. 2020; Zhang et al. 2020; Zheng et al. 2020).
Salts usually incur both osmotic and ionic stress resulting in the increase or decrease of specific SMs in plants (Mahajan and Tuteja 2005). Truong et al. (2018) reported enhanced production of anthocyanins under low nitrate and high salt stress. Contrary to this, Daneshmand et al. (2009) described that salt stress reduced anthocyanins concentration in salt-sensitive plant species. Similarly, Petrusa and Winicov (1997) explained that salt tolerant plant alfalfa quickly doubled its root proline contents, while salt-sensitive plant’s rate of increase in proline contents was slow. Torre-González et al. (2018) reported proline accumulation linked with salt tolerance in Solanum lycopersicum L. Pedranzani et al. (2003) also reported endogenous jasmonic acid accumulation in tomato plants under salt stress. Methyl jasmonate and salicylic acid are well-known elicitors for a number of SMs including terpenoid, alkaloids, and phenolic phytoalexins in medicinal plants (Singh and Dwivedi 2018). Polyphenol synthesis often occurs owing to biotic or abiotic stresses (Muthukumarasamy et al. 2000; Popović et al. 2016). Increasing salinity has also been reported for high polyphenol contents in many plants (Parida and Das 2005). Navarro et al. (2006) described high levels of phenolic compounds in moderately saline soil in red pepper and have been reported many times as salinity response. Plant polyamines also get involved in the salinity response. Mutlu and Bozcuk (2007) reported changes inbound and free polyamine concentration in the roots of sunflower due to salinity.
Heavy metals also influence the production of SMs as they are toxic in nature and cause the production of ROS leading to increase the SMs production via specific signals. A number of studies are present that describes the role of HMs in inducing the plant defense system. Copper exhibited the stimulation for the production of betalains in Beta vulgaris (Trejo-Tapia et al. 2001). Silver and cadmium enhanced the production of two tropane alkaloids (hyoscyamine and scopolamine) in Brugmansia candida (Angelova et al. 2006). Lanthanum has been found involved in the production of taxol in the Taxus species. A decrease in putrescine has been observed in the leaf disks of sunflower (Jacobsen et al. 1992). But there was no effect of chromium exposure on the spermidine and spermine level in leaves except putrescine which increased with increasing Cr level and exposure time via leaves. Lin and Kao (1999) also reported an increase in the level of putrescine in the leaves of rice on exposure to Cu, while the centration of spermine decreased. Production of SMs in plants varies largely depending on the stress conditions, HMs, salt types, and species of plants.
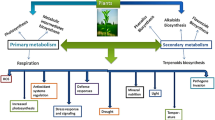
4 Roles of Secondary Metabolites in Plants
Secondary metabolites have a prominent role in the protection of plants against abiotic as well as biotic stress factors although they are considered insignificant for development and growth processes (Schafer and Wink 2009; Ncube and Staden 2015). It is thought that most of the known SMs are involved in the chemical defensive system of the plants from millions of years as plants are suffering from the invaders (Wink 1999). High concentrations of SMs result in more resistance in plants, while their production is costly for the plants leading to a decrease in their growth and reproduction (Siemens et al. 2002). Plants get damaged when the amount of ROS exceed than the antioxidant or detoxification mechanisms capacity. So, plants have developed extensive protective systems to get rid of ROS in the form of SMs. They can stop the oxidation process by blocking the detrimental oxidation chain reaction to save plants (Sgherri et al. 2003; Karuppanapandian et al. 2011). Low molecular weight antioxidants like vitamin E, vitamin C, and phenolic acids, etc. are considered most important under inorganic pollutant stress (Kasote et al. 2015). They are also necessary for the defense of cellular components and are substantial scavenger of many ROS but unable to cope with metastable hydroperoxides like reducing radicals (Chaudiere and Ferrari-iliou 1999). In this regard superoxide dismutase, ascorbate peroxidase, dehydroascorbate reductase, monodehydroascorbate reductase, glutathione reductase, and catalase participate in ascorbate glutathione cycle, a highly established detoxification system (Noctor 2002; Hasanuzzaman et al. 2019). Ascorbate peroxidase is an important peroxidase to detoxify H2O2 acting both in chloroplasts and cytosol using ascorbic acid as a reductant in the ascorbate glutathione cycle (Smirnoff 2000).
Isoflavonoids are well-known SMs for their role in defense against ROS (Posmyk et al. 2009). ROS are the major stress-causing agents under heavy metal and other salt pollution. Antioxidant enzymes including superoxide dismutase, catalase, proline oxidase, ascorbate peroxidase, glutathione peroxidase, and glutathione reductase are the most effective agents for scavenging ROS under metal stress. Phenolic SMs are the substrate for these peroxidase enzymes and found to be the first line of defense regarding various stress factors including HMs and other pollutants (Novak et al. 2004; Posmyk et al. 2009). Glutathione (GSH) is a major form of organic sulfur and also serves as an effective antioxidant in stress conditions (Kang and Kim 2007). Trichomes a specialized type of cells show high enzymatic activities for the synthesis of many phytochelatins and GSH required for the detoxification of the HMs (Tian et al. 2017). GSH directly takes part as an antioxidant in mitigating oxidative stress as well as a reducing agent for other antioxidants like ascorbic acid and scavenge O3 produced by ROS (Nocito et al. 2002). It also helps plants in detoxification of cyto-toxins and xenobiotics by sequestering them to the vacuole (Hasanuzzaman et al. 2017b).
Phenolic compounds are potential antioxidants produced by plants against heavy metal stress (Colak et al. 2019). A lot of studies have been reported about metals induced production of peroxidases and phenolic compounds in plants. The ability of phenolic compounds to alleviate HMs stress is owing to their high propensity to chelate metals. They own carboxyl and hydroxyl groups which make them capable of binding copper and iron (Jung et al. 2003). Yang and Pan (2013) have reported high exudation of phenolic compounds by roots of Helianthus annuus L. exposed to HMs. While Moran et al. (1997) described that chelating ability is due to the high nucleophilic property of the aromatic rings rather than specifically due to chelating groups in the molecules. There is another mechanism involved in the antioxidant nature of the phenolic compounds. Heavy metal ions produce lipid alkoxyl-radicals by the hemolytic breakdown of O–O bonds and decomposition of lipid hydroperoxides, which induce free radical-chain oxidation. Phenolic SMs trap these lipid alkoxyl-radicals and help in the inhibition of lipid peroxidation. It depends on the structure, position, and number of hydroxyl groups of the molecule (Milic et al. 1998). Arora et al. (2000) described that phenolic SMs especially flavonoids also have the ability to modify the peroxidation kinetics through lipid packing order. They help in the stabilization of the membranes by reducing their fluidity, restrict peroxidative reactions, and decrease the diffusion of free radicals (Arora et al. 2000; Blokhina et al. 2003). In addition to their protein binding ability, procyanidins and flavonols interact with membrane phospholipids via hydrogen bonding with polar heads of phospholipids (Verstraeten et al. 2003). Hence, these compounds can get accumulated at the surfaces of the membranes both inside and outside of the cell. This suggests that flavonoids help in maintaining the integrity of the membranes by inhibiting the access of damaging species to the hydrophobic expanse of the bilayer along with others that can affect the rheology of the membrane and can initiate oxidative damage to the cell.
5 Advances in Synthesis of Secondary Metabolites
There are a lot of ways of production of secondary metabolites like plant-based, tissue culture, and strategies like traditional and metabolic engineering (Gonçalves and Romano 2018). With increasing consumer demand for safe product provision, plant-derived product demand is on all-time rise (Lucera et al. 2012). Though chemosynthesis can be used to produce various simple plant products with simple chemical structure but producing SMs like alkaloids via this process is not economical (Stevenson and Szczeklik 2006; Greger 2017). Environmental constraints and restrictions can also limit the extraction of some plant products from naturally grown plants (Yue et al. 2014). Plant farming for the extraction of SMs is a very time-consuming process and plant tissue culture seems to be more appropriate in this regard as it can facilitate mass production of SMs (Isah et al. 2017). Plant culture-based production of SMs is comparably neat, pesticide/herbicide and microbe contamination free system with more efficiency (Verpoorte et al. 2002; Murthy et al. 2014; Ochoa-Villarreal et al. 2016) making them suitable for commercial scale production (Kolewe et al. 2008). In tissue culture-based production of SMs, undifferentiated plant tissue callus is preferred as plant cell is considered as a totipotent in its mechanical and biochemical machinery (Yue et al. 2014). Tissue culture allows us the production of various novel plant-based SMs whose production from native plants was otherwise difficult (Ochoa-Villarreal et al. 2016; de Pádua et al. 2012). Tissue culture has been successfully used to produce SMs like artemisinin (Baldi and Dixit 2008), ajmalicine (Ten Hoopen et al. 2002), taxol (Patil et al. 2014; Sharma and Zafar 2016), resveratrol (Farag and Hassan 2004), and ginsenosides. For improvement in the production of secondary metabolites, nutrient precursor feeding and stimuli provision can boost yield, while other metabolic engineering approaches are also viable to use.
6 Inorganic Pollutant Stress and SMs Production in Plants
The SMs are widely produced by plant species to combat stress and strengthening defense mechanisms (Isah 2019). Among various abiotic stresses, heavy metals are very much potent and persistent in modern-day agriculture. Plants tend to cope with this stress via the production of various SMs in real-time out of which plant hormones are very important. Under abiotic stress, out of all SMs, phytohormones are very important which tend to regulate mineral homeostasis for plants. Phytohormones production (triggered by abiotic stress) results in the regulation of enzymatic activities and SMs production which is helpful in stress mitigation (Wani et al. 2016). Under heavy metal stress, plant tends to regulate the production of abscisic acid (ABA) which acts as a signaling compound regulating gene expression for stress mitigation in plants (O’Brien and Benková 2013). Heavy metals like Cd, Hg, Cu, As turns ABA gene expression on causing a surge in endogenous ABA concentration (Bücker-Neto et al. 2017). Another phytohormone, auxin, or indole acetic acid (IAA) is also helpful in increasing plant growth under normal and stress conditions (Kazan 2013). Similarly, gibberellic acid (GA) is also involved in plant adaption to provide resistance towards various abiotic stresses like HMs (Maggio et al. 2009). Cytokinin (CK) hormone is involved in inter-hormonal signaling under heavy metal stress and sometimes works antagonistically to ABA (Ha et al. 2012). Another phytohormone, salicylic acid (SA) production in plants is also observed to be a defensive response towards heavy metal stress (Rivas-San Vicente and Plasencia 2011) as not only it can detoxify HMs stress but also enhance the activities of antioxidants (Metwally et al. 2003). Ethylene production is enhanced under minute HMs stress, but severe HMs toxicity tends to decrease ethylene production (Gora and Clijsters 1989).
Besides phytohormones, trace metals also alter the composition of other SMs like Ni inhibiting anthocyanin in plants (Hawrylak et al. 2007). A decrease in anthocyanin levels is due to inhibition in the activity of l-phenylalanine ammonia-lyase (Krupa et al. 1996). Copper and Cadmium (Cu and Cd) have been reported to increase shikonin in Lithospermum callus cultures upon toxic accumulation (Mizukami et al. 1977). Similarly, Cu and Co stress have been reported to enhance betalains production in Beta vulgaris (Trejo-Tapia et al. 2001). Various other heavy metals have been reported to affect the production of SMs which is primarly plant’s defensive response against HMs stress. Similarly, heavy metal toxicity also alters SMs production in the medicinal plants where these compounds are responsible for the medicinal properties of plants. A heavy metal derived inactivation of plant metabolites is due to the loss of specific enzymes or damage to biochemical cycles involved in SMs production (Pandey et al. 2007). Nickel toxicity in Hypericum perforatum has shown a 15–20 folds’ decrease in hyperforin production. While induction of phenolic compounds has been reported in Ni, Al, and Cu toxicity in wheat, maize, and Mascarene Island leaf flower (Winkel-Shirley 2002; Michalak 2006). A chapter summarized by Nasim and Dhir (2009) has reviewed several types of research on the effect of heavy metals on SMs production by medicinal plants and various plant species have been shown to do so.
Heavy metals aim to activate ROS production in plant species upon toxic concentration leading to cellular degradation of biochemical machinery. To cope with ROS production, plants tend to accumulate several antioxidant SMs like flavonoids, lignin, phenolic acids, tocopherol, stilbenes, tannins, and organic acids (Hou et al. 2003). Other antioxidants are enzymatic (catalase, (CAT), superoxide dismutase (SOD), guaiacol peroxidase (GPX), ascorbate peroxidase (APX), and glutathione reductase (GR)) and non-enzymatic (glutathione (GSH), ascorbate (AsA), carotenoids) that act as the scavengers of ROS (Michalak 2006; Rastgoo et al. 2011; Sharma et al. 2012). Various studies have reported the regulation of antioxidants by heavy metal stimuli. Bankajj et al. (2016) reported an increased activity of GPX while a decreased activity of CAT and APX in Atriplex halimus under Cu stress. Cadmium toxicity in Kosteletzkya virginica has caused a net increase in endogenous GSH, tocopherol, SA and GR activities (Han et al. 2012).
7 Exogenous Applications of SMs for Combating Stress
Under abiotic stress like salinity and HM, plants tend to produce various SMs (metabolites, phytohormones, and antioxidants) like IAA, SA, GA, ethylene, CK, brassinosteroids, ascorbates, glutathione, and crotonids. Exogenous application of some of these SMs is helpful for plant to counter abiotic stresses as well.
Phytohormones are essential to plant biochemical machinery products to counter abiotic stress, improve plant growth, and enhance plant defense mechanisms (Sytar et al. 2018). Bali et al. (2019) reported that exogenous application of jasmonic acid activates the production of essential SMs in tomato tissues, triggers stress suppression genes, and detoxifies Pb toxicity. Similarly, exogenous application of auxins to Arabidopsis thaliana has shown a remarkable decrease in Cu (Peto et al. 2011; Yuan et al. 2013), Cd (Yuan and Huang 2015), and As (Srivastava et al. 2012) uptake and toxicities. Similarly, the exogenous application of ABA decreased Cd root–shoot translocation in Arabidopsis thaliana (Perfus-Barbeoch et al. 2002). Another important class of phytohormones BRs is also helpful in HMs derived stress mitigation in the plant via increasing antioxidants defense as reported for Zn (Arora et al. 2010), Pb (Rady and Osman 2012), and Cr (Choudhary et al. 2011) toxicity. Salicylic acid (SA) has also been reported to have a mitigative effect on barley plants experiencing Cd toxicity (Tamás et al. 2015).
Among various SMs, glutathione is one of the most cited and reported non-enzymatic antioxidants and has a nullification effect on HM toxicity if applied exogenously. Glutathione exogenous application has shown stress mitigation effects against Ni (Khan and Khan 2014; Khan et al. 2016), Cd (Khan et al. 2015, 2016), As (Sakai et al. 2010; Dixit et al. 2016), Zn (Barrameda-Medina et al. 2014; Khan and khan 2014), Cu (Mostofa et al. 2015), Al (Ruiz et al. 2006), and Pb (Yuan and Huang 2015).
8 Summary and Prospects
Plants are considered to be more capable of tolerating the heavy metals and salts pollution in the environment than other organisms. Oxidative metabolism is one of the major mechanisms involved in the tolerance behavior of plants. They have salinity tolerance strategies which also help in conferring the heavy metals toxicity. The organization of enzymatic and non-enzymatic pathways of the antioxidant system helps the plants in providing defense against inorganic pollutants. Phenolic, terpenoid, nitrogen, sulfur, etc. compounds increase tolerance of the plants against various stress elicitors. The accumulation of osmoprotectants, metallothioneins, phytochelatins, etc. regulates the plant’s normal metabolism. So, a better understanding of these secondary metabolites pathways, processes, and formations along with their precursor can help to mitigate the inorganic pollutant stress as well as other stress factors on plants. Exogenous application of these SMs and their precursors need a thorough understanding and comprehensive study to evaluate their effects on plant growth. Many studies are present indicating the effects of SMs on plants but controversy is present about their exogenous application and plant-specific effects. Some cases are present where plants showed a decrease in growth on SMs overexpression which needs further research to explore the benefits of SMs regarding their optimum concentration. It is a vast area of research that can help scientists in dealing with heavy metals and salinity stress for plants. Exogenous application or elicitation genes understanding of the SMs can be a novel option in conferring stress in plants.
Abbreviations
- ABA:
-
Abscisic acid
- Al:
-
Aluminum
- APX:
-
Ascorbate peroxidase
- As:
-
Arsenic
- AsA:
-
Ascorbate
- BRs:
-
Brassinosteroids
- C:
-
Carbon
- CAT:
-
Catalase
- Cd:
-
Cadmium
- CK:
-
Cytokinin
- Co:
-
Cobalt
- Cr:
-
Chromium
- Cu:
-
Copper
- GA:
-
Gibberellic acid
- GPX:
-
Guaiacol peroxidase
- GR:
-
Glutathione reductase
- GSH:
-
Glutathione
- GSL:
-
Glucosinolates
- H2O2:
-
Hydrogen peroxide
- Hg:
-
Mercury
- HMs:
-
Heavy metals
- IAA:
-
Indole acetic acid
- kDa:
-
Kilodalton
- Ni:
-
Nickel
- O:
-
Oxygen
- O3:
-
Ozone
- Pb:
-
Lead
- POX:
-
Peroxidase
- ROS:
-
Reactive oxygen species
- SA:
-
Salicylic acid
- SMs:
-
Secondary metabolites
- SOD:
-
Superoxide dismutase
- UV:
-
Ultraviolet
- Zn:
-
Zinc
References
Ali ST, Mahmooduzzafar-Abdin MZ, Iqbal M (2008) Ontogenetic changes in Foliar features and psoralen content of Psoralea corylifolia Linn. Exposed to SO2 stress. J Environ Biol 29:661–668
Alloway BJ (2011) Heavy metals in soil, 3rd edn. Wiley, New York
Andresen E, Peiter E, Küpper H (2018) Trace metal metabolism in plants. J Exp Bot 69:909–954
Angelova Z, Georgiev S, Roos W (2006) Elicitation of plants. Biotechnol Biotechnol Equip 20:72–83. https://doi.org/10.1080/13102818.2006.10817345
Arora A, Byrem TM, Nair MG, Strasburg GM (2000) Modulation of liposomal membrane fluidity by flavonoids and isoflavonoids. Arch Biochem Biophys 373:102–109. https://doi.org/10.1006/abbi.1999.1525
Arora P, Bhardwaj R, Kumar Kanwar M (2010) 24-epibrassinolide induced antioxidative defense system of Brassica juncea L. under Zn metal stress. Physiol Mol Biol Plants 16:285–293. https://doi.org/10.1007/s12298-010-0031-9
Asgari Lajayer B, Ghorbanpour M, Nikabadi S (2017) Heavy metals in contaminated environment: destiny of secondary metabolite biosynthesis, oxidative status and phytoextraction in medicinal plants. Ecotoxicol Environ Saf 145:377–390. https://doi.org/10.1016/j.ecoenv.2017.07.035
Baldi A, Dixit VK (2008) Yield enhancement strategies for artemisinin production by suspension cultures of Artemisia annua. Bioresour Technol 99:4609–4614. https://doi.org/10.1016/j.biortech.2007.06.061
Bali S, Jamwal VL, Kohli SK, Kaur P, Tejpal R, Bhalla V, Ohri P, Gandhi SG, Bhardwaj R, Al-Huqail AA, Siddiqui MH, Ali HM, Ahmad P (2019) Jasmonic acid application triggers detoxification of lead (Pb) toxicity in tomato through the modifications of secondary metabolites and gene expression. Chemosphere 235:734–748. https://doi.org/10.1016/j.chemosphere.2019.06.188
Bankajj I, Sleimi N, Gómez-Cadenas A, Pérez-Clemente RM (2016) NaCl protects against Cd and Cu-induced toxicity in the halophyte Atriplex halimus. Spanish J Agric Res 14:e0810. https://doi.org/10.5424/sjar/2016144-10117
Barrameda-Medina Y, Montesinos-Pereira D, Romero L, Blasco B, Ruiz JM (2014) Role of GSH homeostasis under Zn toxicity in plants with different Zn tolerance. Plant Sci 227:110–121
Baye H, Hymete A (2009) Lead and cadmium accumulation in medicinal plants collected from environmentally different sites. Bull Environ Contam Toxicol 84:197–201. https://doi.org/10.1007/s00128-009-9916-0
Blokhina O, Virolainen E, Fagerstedt KV (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Bot 91:179–194
Brooker N, Windorski J, Blumi E (2008) Halogenated coumarins derivatives as novel seed protectants. Commun Agric Appl Biol Sci 73:81–89
Bücker-Neto L, Paiva ALS, Machado RD, Arenhart RA, Margis-Pinheiro M (2017) Interactions between plant hormones and heavy metals responses. Genet Mol Biol 40:373–386. https://doi.org/10.1590/1678-4685-GMB-2016-0087
Carrubba A, Scalenghe R (2012) The scent of Mare Nostrum: medicinal and aromatic plants in Mediterranean soils. J Sci Food Agric 92:1150–1170. https://doi.org/10.1002/jsfa.5630
Chaiyarat R, Suebsima R, Putwattana N, Kruatrachue M, Pokethitiyook P (2010) Effects of soil amendments on growth and metal uptake by Ocimum gratissimum grown in Cd/Zn-contaminated soil. Water Air Soil Pollut 214:383–392. https://doi.org/10.1007/s11270-010-0430-0
Chaudiere J, Ferrari-Iliou R (1999) Intracellular antioxidants: from chemical to biochemical mechanisms. Food Chem Toxicol 37:949–962. https://doi.org/10.1016/s0278-6915(99)00090-3
Choudhary SP, Kanwar M, Bhardwaj R, Gupta BD, Gupta RK (2011) Epibrassinolide ameliorates Cr (VI) stress via influencing the levels of indole-3-acetic acid, abscisic acid, polyamines and antioxidant system of radish seedlings. Chemosphere 84:592–600. https://doi.org/10.1016/j.chemosphere.2011.03.056
Colak N, Torun H, Gruz J, Strnad M, Ayaz FA (2019) Exogenous N-Acetylcysteine alleviates heavy metal stress by promoting phenolic acids to support antioxidant defence systems in wheat roots. Ecotoxicol Environ Saf 181:49–59
Daneshmand F, Arvin MJ, Kalantari KM (2009) Physiological responses to NaCl stress in three wild species of potato in vitro. Acta Physiol Plant 32:91–101. https://doi.org/10.1007/s11738-009-0384-2
De Lurdes Dinis M, Fiúza A (2010) Exposure assessment to heavy metals in the environment: measures to eliminate or reduce the exposure to critical receptors. In: Environmental heavy metal pollution and effects on child mental development, pp 27–50
de Pádua RM, Meitinger N, Filho JDDS, Waibel R, Gmeiner P, Braga FC, Kreis W (2012) Biotransformation of 21-O-acetyl-deoxycorticosterone by cell suspension cultures of Digitalis lanata (strain W.1.4). Steroids 77:1373–1380. https://doi.org/10.1016/j.steroids.2012.07.016
Dixit G, Singh AP, Kumar A, Mishra S, Dwivedi S, Kumar S, Trivedi PK, Pandey V, Tripathi RD (2016) Reduced arsenic accumulation in rice (Oryza sativa L.) shoot involves sulfur mediated improved thiol metabolism, antioxidant system and altered arsenic transporters. Plant Physiol Biochem 99:86–96
Dutta S, Mitra M, Agarwal P, Mahapatra K, De S, Sett U, Roy S (2018) Oxidative and genotoxic damages in plants in response to heavy metal stress and maintenance of genome stability. Plant Signal Behav 13:8
Ebrahim AM, Eltayeb MH, Khalid H, Mohamed H, Abdalla W, Grill P, Michalke B (2012) Study on selected trace elements and heavy metals in some popular medicinal plants from Sudan. J Nat Med 66:671–679. https://doi.org/10.1007/s11418-012-0630-6
Edreva AM, Velikova VB, Tsonev TD (2007) Phenylamides in plants. Russ J Plant Physiol 54:287–301. https://doi.org/10.1134/s1021443707030016
Esmaeilzadeh M, Karbassi A, Bastami KD (2017) Antioxidant response to metal pollution in Phragmites australis from Anzali wetland. Mar Pollut Bull 119:376–380
Farag AM, Hassan MA (2004) Purification, characterization and immobilization of a keratinase from Aspergillus oryzae. Enzym Microb Technol 34:85–93. https://doi.org/10.1016/j.enzmictec.2003.09.002
Figueiredo AC, Barroso JG, Pedro LG, Scheffer JJC (2008) Factors affecting secondary metabolite production in plants: volatile components and essential oils. Flavour Fragr J 23:213–226. https://doi.org/10.1002/ffj.1875
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930. https://doi.org/10.1016/j.plaphy.2010.08.016
Gonçalves S, Romano A (2018) Production of plant secondary metabolites by using biotechnological tools. In: Vijayakumar R, Raja SSS (eds) Secondary metabolites - sources and applications. IntechOpen. https://doi.org/10.5772/intechopen.76414
Gora L, Clijsters H (1989) Effect of copper and zinc on the ethylene metabolism in Phaseolus Vulgaris L. In: Biochemical and physiological aspects of ethylene production in lower and higher plants, pp 219–228
Greger H (2017) Phytocarbazoles: alkaloids with great structural diversity and pronounced biological activities. Phytochem Rev 16:1095–1153. https://doi.org/10.1007/s11101-017-9521-5
Grubb CD, Abel S (2006) Glucosinolate metabolism and its control. Trends Plant Sci 11:89–100. https://doi.org/10.1016/j.tplants.2005.12.006
Ha S, Vankova R, Yamaguchi-Shinozaki K, Shinozaki K, Tran LSP (2012) Cytokinins: metabolism and function in plant adaptation to environmental stresses. Trends Plant Sci 17:172–179. https://doi.org/10.1016/j.tplants.2011.12.005
Halkier BA, Gershenzon J (2006) Biology and biochemistry of glucosinolates. Annu Rev Plant Biol 57:303–333. https://doi.org/10.1146/annurev.arplant.57.032905.105228
Han R-M, Lefèvre I, Albacete A, Pérez-Alfocea F, Barba-Espín G, Díaz-Vivancos P, Quinet M, Ruan CJ, Hernández JA, Cantero-Navarro E, Lutts S (2012) Antioxidant enzyme activities and hormonal status in response to Cd stress in the wetland halophyte Kosteletzkya virginica under saline conditions. Physiol Plant 147:352–368
Hasanuzzaman M, Nahar K, Anee TI, Fujita M (2017a) Exogenous silicon attenuates cadmium-induced oxidative stress in Brassica napus L. by modulating AsA-GSH pathway and glyoxalase system. Front Plant Sci 8:1061. https://doi.org/10.3389/fpls.2017.01061
Hasanuzzaman M, Nahar K, Anee TI, Fujita M (2017b) Glutathione in plants: biosynthesis and physiological role in environmental stress tolerance. Physiol Mol Biol Plants 23:249–268
Hasanuzzaman M, Bhuyan MHMB, Anee TI, Parvin K, Nahar K, Al Mahmud J, Fujita M (2019) Regulation of ascorbate-glutathione pathway in mitigating oxidative damage in plants under abiotic stress. Antioxidants 8:384
Hatami M, Ghorbanpour M (2016) Retracted chapter: changes in phytochemicals in response to rhizospheric microorganism infection. In: Microbial-mediated induced systemic resistance in plants, pp 1–14
Hawrylak B, Matraszek R, Szymańska M (2007) Response of lettuce (Lactuca Sativa L.) to selenium in nutrient solution contaminated with nickel. Veg Crop Res Bull 67:63–70. https://doi.org/10.2478/v10032-007-0031-7
Heitefuss R (2011) Functions and biotechnology of plant secondary metabolites, 2nd edn, annual plant reviews, vol 39. J Phytopathol 159:72–72
Hou WC, Lin RD, Cheng KT, Hung YT, Cho CH, Chen CH, Hwang SY, Lee MH (2003) Free radical-scavenging activity of Taiwanese native plants. Phytomedicine 10:170–175. https://doi.org/10.1078/094471103321659898
Hu J, Fang H, Wang J, Yue X, Su M, Mao Z, Zou Q, Jiang H, Guo Z, Yu L, Feng T, Lu L, Peng Z, Zhang Z, Wang N, Chen X (2020) Ultraviolet B-induced MdWRKY72 expression promotes anthocyanin synthesis in apple. Plant Sci 292:110377
Isah T (2019) Stress and defense responses in plant secondary metabolites production. Biol Res 52:39. https://doi.org/10.1186/s40659-019-0246-3
Isah T, Umar S, Mujib A, Sharma MP, Rajasekharan PE, Zafar N, Frukh A (2017) Secondary metabolism of pharmaceuticals in the plant in vitro cultures: strategies, approaches, and limitations to achieving higher yield. Plant Cell Tissue Organ Cult 132:239–265. https://doi.org/10.1007/s11240-017-1332-2
Jacobsen S, Hauschild MZ, Rasmussen U (1992) Induction by chromium ions of chitinases and polyamines in barley (Hordeum vulgare L.) and rape (Brassica napus L. ssp. oleifera). Plant Sci 84:119–128. https://doi.org/10.1016/0168-9452(92)90125-6
Jiang S, Han S, He D, Cao G, Fang K, Xiao X, Yi J, Wan X (2019) The accumulation of phenolic compounds and increased activities of related enzymes contribute to early defense against walnut blight. Physiol Mol Plant Pathol 108:101433
Jung C, Maeder V, Funk F et al (2003) Release of phenols from Lupinus albus L. roots exposed to Cu and their possible role in Cu detoxification. Plant and Soil 252:301–312. https://doi.org/10.1023/a:1024775803759
Kang SY, Kim YC (2007) Decursinol and decursin protect primary cultured rat cortical cells from glutamate-induced neurotoxicity. J Pharm Pharmacol 59:863–870. https://doi.org/10.1211/jpp.59.6.0013
Kapoor D, Singh S, Kumar V, Romero R, Prasad R, Singh J (2019) Antioxidant enzymes regulation in plants in reference to reactive oxygen species (ROS) and reactive nitrogen species (RNS). Plant Gene 19:100182
Karuppanapandian T, Moon JC, Kim C, Manoharan K, Kim W (2011) Reactive oxygen species in plants: their generation, signal transduction, and scavenging mechanisms. Aust J Crop Sci 5:709–725
Kasote DM, Katyare SS, Hegde MV, Bae H (2015) Significance of antioxidant potential of plants and its relevance to therapeutic applications. Int J Biol Sci 11:982–991
Kazan K (2013) Auxin and the integration of environmental signals into plant root development. Ann Bot 112:1655–1665. https://doi.org/10.1093/aob/mct229
Khan MIR, Khan NA (2014) Ethylene reverses photosynthetic inhibition by nickel and zinc in mustard through changes in PSII activity, photosynthetic nitrogen use efficiency, and antioxidant metabolism. Protoplasma 251:1007–1019
Khan MIR, Nazir F, Asgher M, Per TS, Khan NA (2015) Selenium and sulfur influence ethylene formation and alleviate cadmium-induced oxidative stress by improving proline and glutathione production in wheat. J Plant Physiol 173:9–18
Khan NA, Asgher M, Per TS, Masood A, Fatma M, Khan MIR (2016) Ethylene potentiates sulfur-mediated reversal of cadmium inhibited photosynthetic responses in mustard. Front Plant Sci 7:1628
Kolewe ME, Gaurav V, Roberts SC (2008) Pharmaceutically active natural product synthesis and supply via plant cell culture technology. Mol Pharm 5:243–256. https://doi.org/10.1021/mp7001494
Kovacik J, Klejdus B, Hedbavny J, Stork F, Backor M (2009a) Comparison of cadmium and copper effect on phenolic metabolism, mineral nutrients and stress-related parameters in Matricaria chamomilla plants. Plant and Soil 320:231–242
Kovacik J, Klejdus B, Backor M (2009b) Phenolic metabolism of Matricaria chamomilla plants exposed to nickel. J Plant Physiol 166:1460–1464
Krupa Z, Baranowska M, Orzol D (1996) Can anthocyanins be considered as heavy metal stress indicator in higher plants. Acta Physiol Plant 18:147–151
Lake JA, Field KJ, Davey MP, Beerling DJ, Lomax BH (2009) Metabolomic and physiological responses reveal multi-phasic acclimation of Arabidopsis thaliana to chronic UV radiation. Plant Cell Environ 32:1377–1389. https://doi.org/10.1111/j.1365-3040.2009.02005.x
Li QS, Cai SS, Mo CH, Chu B, Peng LH, Yang FB (2010) Toxic effects of heavy metals and their accumulation in vegetables grown in a saline soil. Ecotoxicol Environ Saf 73:84–88. https://doi.org/10.1016/j.ecoenv.2009.09.002
Lin CC, Kao CH (1999) Excess copper induces an accumulation of putrescine in rice leaves. Bot Bull Acad Sin 40:213–218
Lucera A, Costa C, Conte A, Del Nobile MA (2012) Food applications of natural antimicrobial compounds. Front Microbiol 3:287. https://doi.org/10.3389/fmicb.2012.00287
Maggio A, Barbieri G, Raimondi G, De Pascale S (2009) Contrasting effects of GA3 treatments on tomato plants exposed to increasing salinity. J Plant Growth Regul 29:63–72. https://doi.org/10.1007/s00344-009-9114-7
Mahajan S, Tuteja N (2005) Cold, salinity and drought stresses: an overview. Arch Biochem Biophys 444:139–158. https://doi.org/10.1016/j.abb.2005.10.018
Maleki M, Ghorbanpour M, Kariman K (2017) Physiological and antioxidative responses of medicinal plants exposed to heavy metals stress. Plant Gene 11:247–254
Mazid, M., Khan, T.A. and Mohammad, F., 2011. Role of secondary metabolites in defense mechanisms of plants. Biology and medicine, 3(2), pp.232-249.
McMurry JE (2014) Organic chemistry with biological applications. Cengage Learning
Megateli S, Semsari S, Couderchet M (2009) Toxicity and removal of heavy metals (cadmium, copper, and zinc) by Lemna gibba. Ecotoxicol Environ Saf 72:1774–1780. https://doi.org/10.1016/j.ecoenv.2009.05.004
Metwally A, Finkemeier I, Georgi M, Dietz K-J (2003) Salicylic acid alleviates the cadmium toxicity in barley seedlings. Plant Physiol 132:272–281. https://doi.org/10.1104/pp.102.018457
Michalak A (2006) Phenolic compounds and their antioxidant activity in plants growing under heavy metal stress. Polish J Environ Stud 15:523–530
Milic BL, Djilas SM, Čanadanović-Brunet JM (1998) Antioxidative activity of phenolic compounds on the metal-ion breakdown of lipid peroxidation system. Food Chem 61:443–447. https://doi.org/10.1016/s0308-8146(97)00126-x
Mizukami H, Konoshima M, Tabata M (1977) Effect of nutritional factors on shikonin derivative formation in Lithospermum callus cultures. Phytochemistry 16:1183–1186. https://doi.org/10.1016/s0031-9422(00)94356-5
Moran JF, Klucas RV, Grayer RJ, Abian J, Becana M (1997) Complexes of iron with phenolic compounds from soybean nodules and other legume tissues: prooxidant and antioxidant properties. Free Radic Biol Med 22:861–870. https://doi.org/10.1016/s0891-5849(96)00426-1
Mostofa MG, Hossain MA, Fujita M, Tran LSP (2015) Physiological and biochemical mechanisms associated with trehalose-induced copper-stress tolerance in rice. Sci Rep 5:11433. https://doi.org/10.1038/srep11433
Murthy HN, Lee E-J, Paek K-Y (2014) Production of secondary metabolites from cell and organ cultures: strategies and approaches for biomass improvement and metabolite accumulation. Plant Cell Tissue Organ Cult 118:1–16. https://doi.org/10.1007/s11240-014-0467-7
Muthukumarasamy M, Gupta SD, Pannerselvam R (2000) Enhancement of peroxidase, polyphenol oxidase and superoxide dismutase activities by triadimefon in NaCl stressed Raphanus sativus L. Biol Plant 43:317–320
Mutlu F, Bozcuk S (2007) Salinity-induced changes of free and bound polyamine levels in sunflower (Helianthus annuus L.) roots differing in salt tolerance. Pak J Bot 39:1097–1102
Nagegowda DA (2010) Plant volatile terpenoid metabolism: biosynthetic genes, transcriptional regulation and subcellular compartmentation. FEBS Lett 584:2965–2973. https://doi.org/10.1016/j.febslet.2010.05.045
Nagegowda DA, Gupta P (2020) Advances in biosynthesis, regulation, and metabolic engineering of plant specialized terpenoids. Plant Sci 294:110457
Nakashima K, Ito Y, Yamaguchi-Shinozaki K (2009) Transcriptional regulatory networks in response to abiotic stresses in Arabidopsis and grasses. Plant Physiol 149:88–95
Nasim SA, Dhir B (2009) Heavy metals alter the potency of medicinal plants. Rev Environ Contam Toxicol:139–149
Navarro J, Flores P, Garrido C, Martinez V (2006) Changes in the contents of antioxidant compounds in pepper fruits at different ripening stages, as affected by salinity. Food Chem 96:66–73. https://doi.org/10.1016/j.foodchem.2005.01.057
Ncube B, Staden JV (2015) Tilting plant metabolism for improved metabolite biosynthesis and enhanced human benefit. Molecules 20:12698–12731
Nocito FF, Pirovano L, Cocucci M, Sacchi GA (2002) Cadmium-induced sulfate uptake in maize roots. Plant Physiol 129:1872–1879. https://doi.org/10.1104/pp.002659
Noctor G (2002) Interactions between biosynthesis, compartmentation and transport in the control of glutathione homeostasis and signalling. J Exp Bot 53:1283–1304. https://doi.org/10.1093/jexbot/53.372.1283
Noctor G, Mhamdi A, Foyer CH (2014) The roles of reactive oxygen metabolism in drought: not so cut and dried. Plant Physiol 164:1636–1648. https://doi.org/10.1104/pp.113.233478
Novak K, Lisa L, Skrdleta V (2004) Rhizobial nod gene-inducing activity in pea nodulation mutants: dissociation of nodulation and flavonoid response. Physiol Plant 120:546–555. https://doi.org/10.1111/j.0031-9317.2004.0278.x
O’Brien JA, Benková E (2013) Cytokinin cross-talking during biotic and abiotic stress responses. Front Plant Sci 4:451. https://doi.org/10.3389/fpls.2013.00451
Ochoa-Villarreal M, Howat S, Hong SM, Jang MO, Jin YW, Lee EK, Loake GJ (2016) Plant cell culture strategies for the production of natural products. BMB Rep 49:149–158. https://doi.org/10.5483/bmbrep.2016.49.3.264
Oksman-Caldentey K-M, Inzé D (2004) Plant cell factories in the post-genomic era: new ways to produce designer secondary metabolites. Trends Plant Sci 9:433–440. https://doi.org/10.1016/j.tplants.2004.07.006
Pagare S, Bhatia M, Tripathi N, Pagare S, Bansal YK (2015) Secondary metabolites of plants and their role: overview. Curr Trends Biotechnol Pharm 9:293–304
Pandey S, Gupta K, Mukherjee AK (2007) Impact of cadmium and lead on Catharanthus roseus – a phytoremediation study. J Environ Biol 28:655–662
Pandey P, Irulappan V, Bagavathiannan MV, Senthil-Kumar M (2017) Impact of combined abiotic and biotic stresses on plant growth and avenues for crop improvement by exploiting physio-morphological traits. Front Plant Sci 8:537
Parida AK, Das AB (2005) Salt tolerance and salinity effects on plants: a review. Ecotoxicol Environ Saf 60:324–349. https://doi.org/10.1016/j.ecoenv.2004.06.010
Patil RA, Lenka SK, Normanly J, Walker EL, Roberts SC (2014) Methyl jasmonate represses growth and affects cell cycle progression in cultured Taxus cells. Plant Cell Rep 33:1479–1492. https://doi.org/10.1007/s00299-014-1632-5
Pedranzani H, Racagni G, Alemano S, Miersch O, Ramirez I, Pena-Cortes H, Taleisnik E, Machado-Domenech E, Abdala G (2003) Salt tolerant tomato plants show increased levels of jasmonic acid. Plant Growth Regul 412:149–158
Perfus-Barbeoch L, Leonhardt N, Vavasseur A, Forestier C (2002) Heavy metal toxicity: cadmium permeates through calcium channels and disturbs the plant water status. Plant J 32:539–548. https://doi.org/10.1046/j.1365-313X.2002.01442.x
Peto A, Lehotai N, Lozano-Juste J, León J, Tari I, Erdei L, Kolbert Z (2011) Involvement of nitric oxide and auxin in signal transduction of copper-induced morphological responses in Arabidopsis seedlings. Ann Bot 108:449–457. https://doi.org/10.1093/aob/mcr176
Petrusa LM, Winicov I (1997) Proline status in salt tolerant and salt sensitive alfalfa cell lines and plants in response to NaCl. Plant Physiol Biochem 35:303–310
Popović BM, Štajner D, Ždero-Pavlović R, Tumbas-Šaponjac V, Čanadanović-Brunet J, Orlović S (2016) Water stress induces changes in polyphenol profile and antioxidant capacity in poplar plants (Populus spp.). Plant Physiol Biochem 105:242–250
Posmyk MM, Kontek R, Janas KM (2009) Antioxidant enzymes activity and phenolic compounds content in red cabbage seedlings exposed to copper stress. Ecotoxicol Environ Saf 72:596–602. https://doi.org/10.1016/j.ecoenv.2008.04.024
Rady MM, Osman AS (2012) Response of growth and antioxidant system of heavy metal-contaminated tomato plants to 24-epibrassinolide. Afr J Agric Res 7:3249–3254. https://doi.org/10.5897/ajar12.079
Ramakrishna A, Ravishankar GA (2011) Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal Behav 6:1720–1731. https://doi.org/10.4161/psb.6.11.17613
Rastgoo L, Alemzadeh A, Afsharifar A (2011) Isolation of two novel isoforms encoding zinc- and copper-transporting P1B-ATPase from Gouan (Aeluropus littoralis). Plant Omics J 4:377–383
Rivas-San Vicente M, Plasencia J (2011) Salicylic acid beyond defence: its role in plant growth and development. J Exp Bot 62:3321–3338. https://doi.org/10.1093/jxb/err031
Roy S (2016) Function of MYB domain transcription factors in abiotic stress and epigenetic control of stress response in plant genome. Plant Signal Behav 11:e1117723
Ruiz JM, Rivero RM, Boron LR (2006) Increases synthesis of glutathione in sunflower plants subjected to aluminum stress. Plant Soil 279:25–30
Saha JK, Selladurai R, Coumar MV, Dotaniya ML, Kundu S, Patra AK (2017) Major inorganic pollutants affecting soil and crop quality. In: Soil pollution - an emerging threat to agriculture. Environmental Chemistry for a Sustainable World. Springer, pp 75–104
Saito K (2004) Sulfur assimilatory metabolism. The long and smelling road. Plant Physiol 136:2443–2450. https://doi.org/10.1104/pp.104.046755
Sakai Y, Watanabe T, Wasaki J, Senoura T, Shinano T, Osaki M (2010) Influence of arsenic stress on synthesis and localization of low-molecular-weight thiols in Pteris vittata. Environ Pollut 158:3663–3669
Sarma H, Deka S, Deka H, Saikia RR (2011) Accumulation of heavy metals in selected medicinal plants. Rev Environ Contam Toxicol:63–86
Saviranta NMM, Julkunen-Tiitto R, Oksanen E, Karjalainen RO (2010) Leaf phenolic compounds in red clover (Trifolium pratense L.) induced by exposure to moderately elevated ozone. Environ Pollut 158:440–446. https://doi.org/10.1016/j.envpol.2009.08.029
Schafer H, Wink M (2009) Medicinally important secondary metabolites in recombinant microorganisms or plants: progress in alkaloid biosynthesis. Biotechnol J 4:1684–1703. https://doi.org/10.1002/biot.200900229
Schreck E, Foucault Y, Sarret G, Sobanska S, Cécillon L, Castrec-Rouelle M, Uzu G, Dumat C (2012) Metal and metalloid foliar uptake by various plant species exposed to atmospheric industrial fallout: mechanisms involved for lead. Sci Total Environ 427–428:253–262. https://doi.org/10.1016/j.scitotenv.2012.03.051
Sgherri C, Cosi E, Navari-Izzo F (2003) Phenols and antioxidative status of Raphanus sativus grown in copper excess. Physiol Plant 118:21–28. https://doi.org/10.1034/j.1399-3054.2003.00068.x
Shahid M, Dumat C, Khalid S, Schreck E, Xiong T, Niazi NK (2017) Foliar heavy metal uptake, toxicity and detoxification in plants: a comparison of foliar and root metal uptake. J Hazard Mater 325:36–58. https://doi.org/10.1016/j.jhazmat.2016.11.063
Sharma SS, Dietz K-J (2009) The relationship between metal toxicity and cellular redox imbalance. Trends Plant Sci 14:43–50. https://doi.org/10.1016/j.tplants.2008.10.007
Sharma K, Zafar R (2016) Optimization of methyl jasmonate and β-cyclodextrin for enhanced production of taraxerol and taraxasterol in (Taraxacum officinale Weber) cultures. Plant Physiol Biochem 103:24–30. https://doi.org/10.1016/j.plaphy.2016.02.029
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 2012:1–26. https://doi.org/10.1155/2012/217037
Siemens DH, Garner SH, Mitchell-Olds T, Callaway RM (2002) Cost of defense in the context of plant competition: Brassica rapa may grow and defend. Ecology 83:505. https://doi.org/10.2307/2680031
Singh A, Dwivedi P (2018) Methyl-jasmonate and salicylic acid as potent elicitors for secondary metabolite production in medicinal plants: a review. J Pharmacogn Phytochem 7:750–757
Smirnoff N (2000) Ascorbic acid: metabolism and functions of a multi-facetted molecule. Curr Opin Plant Biol 3:229–235. https://doi.org/10.1016/s1369-5266(00)00069-8
Sreevidya V (2006) Metabolic engineering of rice with soybean isoflavone synthase for promoting nodulation gene expression in rhizobia. J Exp Bot 57:1957–1969. https://doi.org/10.1093/jxb/erj143
Srivastava S, Srivastava AK, Suprasanna P, D’Souza SF (2012) Identification and profiling of arsenic stress-induced microRNAs in Brassica juncea. J Exp Bot 64:303–315. https://doi.org/10.1093/jxb/ers333
Stevenson D, Szczeklik A (2006) Clinical and pathologic perspectives on aspirin sensitivity and asthma. J Allergy Clin Immunol 118:773–786. https://doi.org/10.1016/j.jaci.2006.07.024
Street RA (2012) Heavy metals in medicinal plant products — an African perspective. S Afr J Bot 82:67–74. https://doi.org/10.1016/j.sajb.2012.07.013
Sytar O, Kumari P, Yadav S, Brestic M, Rastogi A (2018) Phytohormone priming: regulator for heavy metal stress in plants. J Plant Growth Regul 38:739–752. https://doi.org/10.1007/s00344-018-9886-8
Taiz L, Zeiger E (2006) Plant physiology, vol 82, 4th edn. Sinauer Associates Publishers, Sunderland, pp 278–279. https://doi.org/10.1086/523151
Tamás L, Mistrík I, Alemayehu A, Zelinová V, Bočová B, Huttová J (2015) Salicylic acid alleviates cadmium-induced stress responses through the inhibition of Cd-induced auxin-mediated reactive oxygen species production in barley root tips. J Plant Physiol 173:1–8. https://doi.org/10.1016/j.jplph.2014.08.018
Ten Hoopen HJG, Vinke JL, Moreno PRH, Verpoorte R, Heijnen JJ (2002) Influence of temperature on growth and ajmalicine production by Catharanthus roseus suspension cultures. Enzym Microb Technol 30:56–65. https://doi.org/10.1016/s0141-0229(01)00456-2
Tian N, Liu F, Wang P, Zhang X, Li X, Wu G (2017) The molecular basis of glandular trichome development and secondary metabolism in plants. Plant Gene 12:1–12
Torre-González AD, Montesinos-pereira D, Blasco B, Ruiz JM (2018) Influence of the proline metabolism and glycine betaine on tolerance to salt stress in tomato (Solanum lycopersicum L.) commercial genotypes. J Plant Physiol 231:329–336
Trejo-Tapia G, Jimenez-Aparicio A, Rodriguez-Monroy M, De Jesus-Sanchez A, Gutierrez-Lopez G (2001) Influence of cobalt and other microelements on the production of betalains and the growth of suspension cultures of Beta vulgaris. Plant Cell Tissue Organ Cult 67:19–23. https://doi.org/10.1023/a:1011684619614
Truong HA, Lee WJ, Jeong CY, Trịnh CS, Lee S, Kang CS, Cheong YK, Hong SW, Lee H (2018) Enhanced anthocyanin accumulation confers increased growth performance in plants under low nitrate and high salt stress conditions owing to active modulation of nitrate metabolism. J Plant Physiol 231:1–48
Tuteja N (2007) Mechanisms of high salinity tolerance in plants. Methods Enzymol:419–438
Tuteja N, Sopory SK (2008) Chemical signaling under abiotic stress environment in plants. Plant Signal Behav 3:525–536. https://doi.org/10.4161/psb.3.8.6186
Verpoorte R, Contin A, Memelink J (2002) Biotechnology for the production of plant secondary metabolites. Phytochem Rev 1:13–25. https://doi.org/10.1023/a:1015871916833
Verstraeten SV, Keen CL, Schmitz HH, Fraga CG, Oteiza PI (2003) Flavan-3-ols and procyanidins protect liposomes against lipid oxidation and disruption of the bilayer structure. Free Radic Biol Med 34:84–92. https://doi.org/10.1016/s0891-5849(02)01185-1
Wani SH, Kumar V, Shriram V, Sah SK (2016) Phytohormones and their metabolic engineering for abiotic stress tolerance in crop plants. Crop J 4:162–176. https://doi.org/10.1016/j.cj.2016.01.010
Wink M (1999) Functions of plant secondary metabolites and their exploitation in biotechnology. In: Annual plant reviews, vol 3. CRC Press, Boca Raton
Winkel-Shirley B (2001) Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol 126:485–493. https://doi.org/10.1104/pp.126.2.485
Winkel-Shirley B (2002) Biosynthesis of flavonoids and effects of stress. Curr Opin Plant Biol 5:218–223. https://doi.org/10.1016/s1369-5266(02)00256-x
Xiong T, Austruy A, Pierart A, Shahid M, Schreck E, Mombo S, Dumat C (2016a) Kinetic study of phytotoxicity induced by foliar lead uptake for vegetables exposed to fine particles and implications for sustainable urban agriculture. J Environ Sci 46:16–27. https://doi.org/10.1016/j.jes.2015.08.029
Xiong T, Dumat C, Pierart A, Shahid M, Kang Y, Li N, Bertoni G, Laplanche C (2016b) Measurement of metal bioaccessibility in vegetables to improve human exposure assessments: field study of soil–plant–atmosphere transfers in urban areas, South China. Environ Geochem Health 38:1283–1301. https://doi.org/10.1007/s10653-016-9796-2
Yang J, Pan X (2013) Root exudates from sunflower (Helianthus annuus L.) show a strong adsorption ability toward Cd (II). J Plant Interact 8:263–270
Yang L, Wen KS, Ruan X, Zhao YX, Wei F, Wang Q (2018) Response of plant secondary metabolites to environmental factors. Molecules 24:762
Yuan HM, Huang X (2015) Inhibition of root meristem growth by cadmium involves nitric oxide-mediated repression of auxin accumulation and signalling in Arabidopsis. Plant Cell Environ 39:120–135. https://doi.org/10.1111/pce.12597
Yuan HM, Xu HH, Liu WC, Lu YT (2013) Copper regulates primary root elongation through pin1-mediated auxin redistribution. Plant Cell Physiol 54:766–778. https://doi.org/10.1093/pcp/pct030
Yue W, Ming Q, Lin B, Rahman K, Zheng CJ, Han T, Qin LP (2014) Medicinal plant cell suspension cultures: pharmaceutical applications and high-yielding strategies for the desired secondary metabolites. Crit Rev Biotechnol 36:215–232. https://doi.org/10.3109/07388551.2014.923986
Zhang Y, Wang G, Li L, Li Y, Zhou B, Yan H (2020) Identification and expression analysis of BrTT8 during anthocyanin biosynthesis and exposure to abiotic stress in turnip (Brassica rapa subsp. rapa ‘Tsuda’). Sci Hortic 268:109332
Zheljazkov VD, Craker LE, Xing B (2006) Effects of Cd, Pb, and Cu on growth and essential oil contents in dill, peppermint, and basil. Environ Exp Bot 58:9–16. https://doi.org/10.1016/j.envexpbot.2005.06.008
Zheljazkov VD, Jeliazkova EA, Kovacheva N, Dzhurmanski A (2008a) Metal uptake by medicinal plant species grown in soils contaminated by a smelter. Environ Exp Bot 64:207–216. https://doi.org/10.1016/j.envexpbot.2008.07.003
Zheljazkov VD, Craker LE, Xing B, Nielsen NE, Wilcox A (2008b) Aromatic plant production on metal contaminated soils. Sci Total Environ 395:51–62. https://doi.org/10.1016/j.scitotenv.2008.01.041
Zheng T, Li Y, Lei W, Qiao K, Liu B, Zhang D, Lin H (2020) SUMO E3 ligase SIZ1 stabilizes MYB75 to regulate anthocyanin accumulation under high light conditions in Arabidopsis. Plant Sci 292:110355
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Editor(s) (if applicable) and The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Zia ur Rehman, M. et al. (2021). Synthesis and Regulation of Secondary Metabolites in Plants in Conferring Tolerance Against Pollutant Stresses. In: Hasanuzzaman, M. (eds) Approaches to the Remediation of Inorganic Pollutants. Springer, Singapore. https://doi.org/10.1007/978-981-15-6221-1_4
Download citation
DOI: https://doi.org/10.1007/978-981-15-6221-1_4
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-6220-4
Online ISBN: 978-981-15-6221-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)