Abstract
Background
Open parenchymal-preserving resection is the current standard of care for lesions in the posterosuperior liver segments. Laparoscopy and robot-assisted surgery are emergent surgical approaches for liver resections, even in posteriorly located lesions. The objective of this study was to compare robot-assisted to laparoscopic parenchymal-preserving liver resections for lesions located in segments 7, 8, 4a, and 1.
Methods
Demographics, comorbidities, clinicopathologic characteristics, surgical treatments, and outcomes from patients who underwent laparoscopic and robot-assisted liver resection in two centers for lesions in the posterosuperior segments between June 2008 and February 2014 were reviewed. A 1:2 matched propensity score analysis was performed by individually matching patients in the robotic cohort to patients in the laparoscopic cohort based on demographics, comorbidities, performance status, tumor stage, location, and type of resection.
Results
Thirty-six patients who underwent robot-assisted liver resection were matched with 72 patients undergoing laparoscopic liver resection. Matched patients displayed no significant differences in postoperative outcomes as measured by blood loss, hospital stay, R0 negative margin rate, and mortality. The overall morbidity according to the comprehensive complication index was also similar (34.6 ± 33 vs. 18.4 ± 11.3, respectively, for robotic and laparoscopic approach, p = 0.11). Patients undergoing robotic liver surgery had significantly longer inflow occlusion time (77 vs. 25 min, p = 0.001) as compared with their laparoscopic counterparts.
Conclusions
Although number and severity of complications in the robotic group appears to be higher, robotic and laparoscopic parenchymal-preserving liver resections in the posterosuperior segments display similar safety and feasibility.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Laparoscopic liver resections (LLR) of posterosuperior (PS) segments (Segments 1, 4a, 7, and 8) are considered to be challenging tasks due to difficult bleeding control and liver mobilization [1–9]. Many authors do not consider lesions in PS segments to be amenable to a pure laparoscopic approach, while others obviate the poor maneuverability of laparoscopic tools with “hand assistance” [10]. Different devices and approaches have been used to improve parenchymal transection and bleeding control in this area of the liver.
Better visualization can be achieved with left lateral patient position and flexible laparoscopes. Deep parenchymal transection can be improved with the routine use of laparoscopic Cavitron Ultrasonic Surgical Aspirator (CUSA) [4].
Over the last decade, robot-assisted laparoscopy has been included in the surgical armamentarium to manage complex abdominal scenarios, including those encountered in liver surgery [11–13]. The first report of robotic liver resection (ROBR) dates back to 2003, 10 years after the report of the first LLR [14, 15].
The robotic platform Da Vinci S and the newer versions (Intuitive Surgical Inc., Sunnyvale, CA) can overcome many of the limitations of conventional laparoscopic liver surgery: two-dimensional imaging and tremor amplification, fulcrum effect, limited degrees of freedom of laparoscopic tools, and awkward ergonomics. Augmented dexterity given by endowristed instruments, filtration of the surgeon’s movements, and high-definition three-dimensional vision are integrated to allow steady and careful dissection [16]. Due to the technical features of the robotic surgical system, facilitation of minimally invasive hepatectomies in the PS segments is also expected.
Reduction in the major hepatectomy rate according to the principles of parenchymal-sparing surgery and better early outcome are the main goals in this investigation of the effectiveness of the robotic surgical system, with respect to traditional laparoscopy [17].
Some studies comparing robotic and laparoscopic techniques have been published recently, but no one has provided conclusive results in favor of one of two approaches [18–24]. Two of these studies are case–control match but without the propensity score analysis. Our data coming from a retrospective study have shown an advantage of robotics over traditional laparoscopy through reduction in major hepatectomies [20].
The aim of the present study was to compare minor robotic liver resections to laparoscopic liver resections for tumors located in PS segments in a 2:1 case–control study matched by propensity score analysis in order to investigate intra- and postoperative outcomes of the two techniques in conditions where parenchymal-preserving resections were performed.
Materials and methods
Study design
Between June 2008 and February 2014, 824 liver resections performed at two different institutions were retrospectively analyzed from a prospective kept database. A total of 697 resections were performed at Ghent University Hospital Medical School, Belgium (center 1), and 127 at Spoleto General Hospital (center 2). In 371 (45 %) cases, liver resection involved PS segments, of which 141 (38 %) were performed laparoscopically and 230 (62 %) were performed in a standard open fashion. Among 141 laparoscopic cases, 132 patients (eligible for both approach) received parenchymal-preserving resections, of which 96 were fully laparoscopic (n = 80 in center 1 and 16 in center 2) and 36 were performed with robot assistance (n = 10 in center 1 and n = 26 in center 2). Moreover, a mutual exchange of surgeons between the two centers has been adopted since 2012 and for a total period of 4 weeks before starting the study in order to verify that indications and surgical strategies were similar. Contraindications for laparoscopic or robotic approach in both centers included a tumor close to the hilum and the inferior vena cava, bulky tumors, and patients with cirrhosis Child-Pugh categories B and C.
The 36 patients who underwent robotic liver resection (ROB group) were matched in a case–control approach with the propensity score analysis with 72 patients undergoing laparoscopic liver resection (LAP group). The group had a 1:2 ratio according to age, sex, year of operation, type of operation, number of lesions, total mean size of lesions, previous abdominal surgery, and neoadjuvant chemotherapy. Inclusion criteria for enrollment in the study were laparoscopic or liver resections involving one or more PS segments; exclusion criteria were major hepatectomy and palliative resections.
Blood loss was evaluated by taking into account fluid balance and drop in hemoglobin levels [25]. Preoperative investigations consisted of a clinical biochemistry panel, ultrasonography, triphasic spiral CT, magnetic resonance imaging, and positron emission tomography CT when required. Indications for surgical treatment were determined during a multidisciplinary conference including hepatobiliary surgeons, oncologists, gastroenterologists, radiologists, radiotherapists, and pathologists in each institution. During the study period, in both centers, non-anatomical resection and anatomical segmentectomy were the preferred strategies for the treatment for colorectal liver metastases (CRLM) and hepatocellular carcinoma (HCC), respectively, according to the principles of parenchymal preservation surgery [26–28].
Histological margin was defined R1 when microscopically described as inferior to 1 mm. The terminology for liver anatomy and resection is based on the Brisbane classification [29]. The postoperative management was similar between the two centers; the enhanced recovery after surgery (fast-track) protocol [30] was adopted from 2011 onwards in center 1 and from 2012 in center 2.
The Clavien–Dindo classification [31] and the comprehensive complication index [32] systems were used to describe complications in both groups.
Surgical technique for robotic liver resections
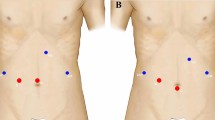
All the procedures were performed using the Da Vinci S and Si Surgical System (Intuitive Surgical, Sunnyvale, CA, USA), and the intraoperative ultrasound assessment was conducted with the Aloka Prosound Alpha 7 (Aloka, Tokyo, Japan). The patient was rotated onto the left flank to facilitate liver mobilization and inferior vena cava dissection, with the right arm hanging over the patient’s forehead. The camera port and the left robotic trocars were placed at the level of the right costal margin, whereas the right robotic trocar was inserted in the intercostal space between the 10th and 11th ribs along the scapular line, as previously described [33]. After pneumoperitoneum induction (10–12 mmHg), a camera port was inserted at the level of the umbilicus. Two 8-mm robotic trocars were placed along the left and right mid-clavicular lines and five fingerbreadths from the umbilicus. Two accessory trocars were placed along the left and right mammillary lines below the robotic trocars. For lesions in segment 4a, trocars were positioned with the same disposition, but more cranially.
Two accessory trocars can be placed along the midline and the anterior axillary line for suction and retraction. To control the liver inflow, an extracorporeal tourniquet was used to encircle the liver pedicle and perform the Pringle maneuver [34]. Parenchyma was usually transected with a harmonic scalpel for straight-line resections. The Kelly clamp crushing technique using endowristed bipolar precise forceps (Intuitive Surgical, Sunnyvale, CA, USA) was preferred for curved and angulated section lines and tumor dissection close to major liver vessels (Fig. 1).
Hemostasis of small vessels was obtained using a monopolar or bipolar cautery. To secure larger vessels on the transection line, we used Hem-o-lock® clips (TFX Medical Ltd, RTP Durham, NC, USA) or ligatures with Vicryl® or Prolene®. The hepatic veins (HVs) were usually divided with a laparoscopic linear stapler (EndoGIA, Ethicon, Cincinnati, OH, USA) or sutured. A suction drainage was always left in place. The specimen was generally extracted through the umbilical port, as previously described [35].
Surgical technique for laparoscopic liver resections
Laparoscopic resection was performed with the patient in supine and 30° anti-Trendelenburg position and the surgeon between the patient’s legs. The patient was turned two-thirds to his left side, with the right arm alongside the body whenever possible. Four to six port sites (5 mm, 10 mm, and two to four 12 mm ports) were inserted in the upper abdominal quadrant: The 12-mm ports were placed to allow insertion of a 30° optical device and the linear stapler, the 10-mm port for the surgical aspirator or harmonic scissors, and the 5-mm port was used mainly to allow irrigation and aspiration during surgery and to hang the liver when necessary.
Parenchyma division was almost exclusively performed using the surgical aspirator (CUSA Excell Integra, Plainsboro, NJ, USA; or Olympus SonoSurg, Olympus USA, Center Valley, PA, USA) and a harmonic scalpel (Ethicon, Cincinnati, OH, USA) for the Glissonian approach. The Pringle maneuver was applied selectively in case of bleeding. Bipolar coagulation was used for minor bleeding or oozing (Fig. 2). Larger vascular/biliary structures were controlled with endoclips (Hem-o-lock clips, TFX Medical, RTP Durham, NC, USA) or vascular staplers (EndoGIA, Ethicon; Tri-stapler technology® Covidien). Finally, the specimen was extracted using a plastic bag through the Pfannestiel incision, additional port site enlargement, or by partial opening of a previous abdominal scar.
Statistical analysis
Continuous data were reported as mean ± SD (ranges) and were compared using the two-sided Student’s t test. Comparisons between groups for categorical variables were performed by means of the χ 2 test with Yates’ correction or the Fisher’s exact test when appropriate.
To overcome biases from the different distribution of covariates among patients in the two study groups, a propensity score analysis was performed. The model was used to obtain a one-to-two match by using the nearest-neighbor matching method. Patients in whom the propensity score was not applicable were excluded from further analysis. The matching algorithm was based on logistic regression, without replacement, until all possible matches had been formed.
The following covariates were matched for age, sex, date of operation, number of liver metastases, total mean size of the lesions, previous abdominal surgery, underlying hepatopathy (e.g., cirrhosis), and neoadjuvant chemotherapy. The matched model was tested by a histogram of the propensity score and the dot plot standardized mean differences.
Propensity score analysis and matching was performed with the psmatching program [36] that performs all analyses in R through the SPSS R-Plugin (SPSS R Essentials) and utilizes newly written R code, as described by Thoemmes [37]. The last version of extension bundles for SPSS PSMATCHING3.03.spe was utilized. We used single nearest-neighbor matching with no replacement (a single participant could not be selected multiple times) to match patients.
Statistical significance was set at p < 0.05. Statistical analysis was performed using the IBM SPSS Statistics for Windows, version 20.0. Armonk, NY, IBM Corp.
Results
Preoperative
Patient demographics, indications for liver surgery, and preoperative data are shown in Table 1. There were no significant differences in all main patients and disease characteristics.
Operative procedure
Intra-operative characteristics are shown in Table 2. Types of resections were similar in the two study groups (wedge, monosegmentectomy, bisegmentectomy, or mixed resections). ROB was characterized by a more intensive use of the Pringle maneuver (55.6 vs. 22.2 %, p = 0.001) that results in a significantly longer inflow occlusion time (76.7 ± 41.3 vs. 24.6 ± 16.5, p < 0.001) as compared with their laparoscopic counterparts. The mean surgical time and the amount of bleeding were similar in the two groups. The conversion rate was not significantly different between the two groups (13.9 vs. 9.7 %, p = 0.53), as well as the causes of conversion, which were mainly bleeding, oncological concerns, and adhesions (Table 3).
Histology assessment
Histology assessment is described in Table 2. Mean lesion number and total mean size of lesions were similar between the two study groups. More patients with multiple lesions were present in the robotic group, even though the difference was not statistically significant (47.2 vs. 35.2 %, p = 0.29). The rate of R1 resection was similar between the two study groups as well (11.1 vs. 12.5 %, p = 1).
Morbidity and mortality
In the ROB group, three patients experienced a grades III–V morbidity: the first with a solitary HCC on cirrhosis that developed postoperative liver failure requiring liver transplantation; the second died on the POD 4 following a myocardial infarction; the third developed vena cava inferior syndrome after 7–8 bisegmentectomy for a huge and symptomatic adenoma requiring redo laparotomy and opening of the diaphragm. Postoperative complications were identical between the two study groups (19.4 %, p = 1). The comprehensive complication index was higher in ROB group (34.6 ± 33 vs. 18.4 ± 11.3), although statistically not significant (p = 0.11). The main complications that occurred in the ROB group were pleural effusion, fluid collection, and transient renal failure (5.5 % each). In the LAP group, the complications were infections and pneumothorax (5.5 % each). The rate of biliary leaks was overall very low (2.8 and 1.4 %, respectively, for ROB and LAP).
The overall survival in patients with colorectal liver metastases was 92.3, 64.6, and 40.4 % versus 96.4, 70.8, and 62.9 % (p = 0.24) at 1, 3, and 5 years in ROB versus LAP group, respectively. Accordingly, RFS was 73.3, 46.2, and 46.2 % versus 63.7, 37.1, and 32.5 % (p = 0.56) in ROB versus LAP group, respectively.
Discussion
According to the Louisville consensus statement, indications for laparoscopic liver resection are patients with solitary lesions, 5 cm or less, located in liver segments 2–6 [1]. Instead, liver resections for lesions located in the PS segments (the so-called difficult segments) are mostly reserved for open surgery. The degree of difficulty of resection in segments 7 and 8 is considered to be similar to that of a major hepatectomies [9, 38]. Yoon et al. [8] demonstrated that patients who underwent pure LLR for HCC located in the PS segments had a longer operative time and longer postoperative hospital stay, in addition to a tendency toward a higher rate of open conversion and greater blood loss compared to anterolateral segments Even in our series, only 38 % of patients with lesions in PS segments received a minimally invasive resection.
Technical struggles for a minimally invasive resection in PS segments are due to poor visibility and scarce degrees of freedom for laparoscopic tools. Multiple port sites, including intercostal ports, are necessary to reach the posterior aspect of the right liver, and outflow vascular control and inferior vena cava dissection remain as demanding and potentially harmful steps [39]. Moreover, the complex lines of parenchymal transection hamper bleeding control and require meticulous ultrasound monitoring to perform margin-free resections [40].
Maneuverability of the standard 10-mm laparoscopic ultrasound probe is limited by the trocars’ position, the narrow space of the subdiaphragmatic area, and the convexity of the liver dome. While the vertical demarcation of the transection line is an easy task, more difficult is the identification of the horizontal border of the transection line.
Therefore, LLRs are still limited to a few specialized hepatobiliary units. Intrinsic limitations of conventional laparoscopic surgery—including reduced freedom of movement and a two-dimensional view—can be overcome by the robotic surgical systems cleared for use.
The robot has several advantages compared to the traditional laparoscopic tool, although it has as yet some important limitations (Table 4). Well-known advantages of the robotic system are improved vision via three-dimensional view, magnification, tremor suppression, and the endowristed instruments. However, to date, the benefits of robot assistance in liver surgery have not yet been clearly defined.
Accurate real-time imaging of liver anatomy and a well thought out operative resection plan are necessary, and today, this is accomplished with the use of intraoperative ultrasound image [41].
The available robotic ultrasound probes are merely laparoscopic probes that can be handled by a robotic Prograsp (Intuitive Surgical Inc., Sunnyvale, CA). Prototypes with three-dimensional movements were designed to facilitate visualization of the lesions in the segments PS, but they are not ready for the market [42].
However, the three-dimensional movements of the robotic instruments allow multiplanar parenchymal transections that are arduously reproducible with laparoscopy. Only a few authors have reported minor laparoscopic resections in segments 7, 8, and 1 with results comparable to open surgery using specific technical tricks for right posterior lesions, including left lateral decubitus position and transthoracic port placement [40, 43–47]. The hand-assisted (HA) or hybrid procedures have been advocated to reduce the technical difficulty of laparoscopy in PS segments. It seems likely that the HA procedures may have inferior aesthetic results and, perhaps, cause more postoperative pain than the fully laparoscopic or robotic surgery. However, it seems rather to be a surgeon preference than a clinical relevant issue, and to date, no data are available comparing the benefits of HA procedures to a pure laparoscopic or robotic surgery.
Few studies in the literature have compared LLRs to robotic liver resections, and none of them were specific on PS segments [18, 21–24, 48, 49].
In the present study, we compared laparoscopic and robotic minor liver resections in PS segments in a bi-institutional analysis. We selected the laparoscopic group utilizing the propensity score analysis that was performed to adjust for potential bias and is used often in observational studies because of non-randomized group assignment. Major liver resection involving PS segments was excluded in the analysis because the transection line for these types of operations is considered to be easier than in minor liver resections.
In a previous study, we compared the results of robotic and laparoscopic liver resections for lesions located in all segments. The study showed similar results in terms of safety and efficacy, but we found out a higher rate of minor liver resection in PS segments with the robotic technique (55 vs. 34.1 %, p = 0.019) [20]. These data showed that robot-assisted surgery could promote the liver parenchymal-preserving resections in the PS segments. We speculated that robotic assistance could increase the rate of minimally invasive resections, especially in cases of lesions that required several and multiplanar transection lines. In the present study, no significant differences were observed in terms of efficacy and safety between the two methods concerning the type of surgery, duration of surgery, bleeding, conversion rate, and morbidity. The rate of complication was similar between the two groups, but the comprehensive complication index was higher in the ROB group The severity of complications in the robotic groups appeared to be higher as measured by the comprehensive complication index, although not reaching statistical significance (34.6 ± 33 vs. 18.4 ± 11.3, p = 0.11). Similarly, the rate of R1 resection was not significantly different between the two techniques. A lack of precision in the positioning of the stapler may have caused the VCI syndrome in the patient with the giant adenoma. In fact, the positioning of the stapler is currently performed by the assistant surgeon at the operating table and not by the first surgeon sitting at the console. This situation also highlights a major flaw of the robotic approach: the possible need for two trained surgeons, one at the console and the second one beside to the patient, to obtain the best results, as recently stated at the second international consensus conferences on laparoscopic liver surgery (2ICCLLS) [50]. The technical limitations will certainly be overcome in a near future (i.e., robot-driven endoscopic staplers, suction-irrigation, and robotic ultrasound systems). The length of hospital stay was significantly lower in the laparoscopic group likely due to the implemented enhanced recovery after surgery (ERAS) protocol. The other substantial difference was evident in the use of the Pringle maneuver that was utilized much more frequently in the robotic group. The pedicle clamping is often necessary during robotic resection, because the robotic transection is performed with the crush technique that often requires an intermittent Pringle maneuver. In laparoscopy, liver transection is based on the use of CUSA, and such a device is not currently available for the da robotic surgical system. The ultrasonic aspirator is aspirating cells and promoting a necrosis of the cutting edge due to the thermal energy delivered. This means that the true resection margins are wider than those measured on the specimen by the pathologist. In our series, the frequent use of pedicle clamping in patients with chronic liver disease who underwent robotic resections caused two postoperative liver failures, requiring a successful urgent liver transplant in one case and causing a death in the other.
The need to extend inflow occlusion duration with the robotic technique is one of the major limitations of this approach. Prolonged inflow occlusion can promote postoperative liver failure especially in patients with chronic liver disease or parenchymal alterations due to toxicity resulting from neoadjuvant chemotherapy. However, a reduction in the use and duration of the Pringle maneuver can be appreciated along the surgeon learning curve. Moreover, both liver failures developed after long-lasting surgeries due to deep location and proximity to the hepatocaval confluence of the lesions. These events could be considered also the result of long operating times on poor liver function questioning the safety of minimally invasive surgery for complex resections in the PS segments in the presence of chronic liver disease.
It has been demonstrated that the learning curve of laparoscopic liver resection is completed when 60 procedures are achieved [51]. There are no studies in the literature that describe the necessary learning curve for robotic liver resections, but based on our experience and those presented during the 2ICCLLS [50], it seems likely that it is shorter.
On these equal terms, the main drawback of advanced robotic surgery is the associated cost. In our internal cost analysis, we prospectively collected the number and type of disposable materials used for LLR such as trocars, vascular staplers and refills, clips, stitches, sutures, irrigation suction, disposable materials for CUSA and Sonosurg, argon beamer, and endobags. The mean instrumentation costs of a LLR were 1406€. Although a case-by-case cost analysis was not performed, the robotic instrumentation in general adds 500$ per case to the laparoscopic equipment cost [18]. The cost of the equipment purchase and annual service fees are an additional financial burden of robotic procedures. These can be reduced with heavy utilization of the robot by the other surgical specialties at the same institution. It was demonstrated that in left lateral sectionectomy, which is considered to be a gold standard in laparoscopy, robotic assistance does not improve the operative outcomes but involves significantly higher costs [49].
The main advantages that we found utilizing the robotic technique were three-dimensional viewing, the wide range of movements of the surgical instruments and eventually the 3D movements of ultrasound and the greater surgical precision of instruments movement. Conversely, the benefits we have seen with laparoscopic approach included easier mobility in the management of surgical instruments, the availability of CUSA, and the lower costs.
In conclusion, robot-assisted resections in the PS segments are comparable in safety and feasibility to pure LLR in a high-volume center, although a trend to a higher degree of complications was recorded with the robotic technique. However, based on our results using propensity matching, the claimed superiority of robotics over the laparoscopic approach has not been proven for PS segments. This situation also suggests that a more in-depth comparative cost analysis between the two techniques is necessary to verify the actual cost–benefit rate, which is higher with the robot. It is also important to emphasize that the frequent need of pedicle clamping in robotic surgery exposes patients with diseased liver parenchyma to an increased risk of postoperative liver failure. The possibility of having in the future a wider range of miniaturized liver-specific instruments (such as the surgical aspirator) combined to an haptic feedback will possibly improve the results of robotic liver resections, whereas the availability of 3D cameras in pure laparoscopy could further ameliorate the results of the laparoscopic approach.
References
Buell JF, Cherqui D, Geller DA, O’Rourke N, Iannitti D, Dagher I, Koffron AJ, Thomas M, Gayet B, Han HS, Wakabayashi G, Belli G, Kaneko H, Ker CG, Scatton O, Laurent A, Abdalla EK, Chaudhury P, Dutson E, Gamblin C, D’Angelica M, Nagorney D, Testa G, Labow D, Manas D, Poon RT, Nelson H, Martin R, Clary B, Pinson WC, Martinie J, Vauthey JN, Goldstein R, Roayaie S, Barlet D, Espat J, Abecassis M, Rees M, Fong Y, McMasters KM, Broelsch C, Busuttil R, Belghiti J, Strasberg S, Chari RS, World Consensus Conference on Laparoscopic S (2009) The international position on laparoscopic liver surgery: the Louisville Statement, 2008. Ann Surg 250:825-830
Cherqui D, Husson E, Hammoud R, Malassagne B, Stephan F, Bensaid S, Rotman N, Fagniez PL (2000) Laparoscopic liver resections: a feasibility study in 30 patients. Ann Surg 232:753–762
Dulucq JL, Wintringer P, Stabilini C, Berticelli J, Mahajna A (2005) Laparoscopic liver resections: a single center experience. Surg Endosc 19:886–891
Cho JY, Han HS, Yoon YS, Shin SH (2008) Feasibility of laparoscopic liver resection for tumors located in the posterosuperior segments of the liver, with a special reference to overcoming current limitations on tumor location. Surgery 144:32–38
Laurent A, Cherqui D, Lesurtel M, Brunetti F, Tayar C, Fagniez PL (2003) Laparoscopic liver resection for subcapsular hepatocellular carcinoma complicating chronic liver disease. Arch Surg 138(3):763–769 (discussion 769)
Kazaryan AM, Rosok BI, Marangos IP, Rosseland AR, Edwin B (2011) Comparative evaluation of laparoscopic liver resection for posterosuperior and anterolateral segments. Surg Endosc 25:3881–3889
Xiang L, Xiao L, Li J, Chen J, Fan Y, Zheng S (2015) Safety and feasibility of laparoscopic hepatectomy for hepatocellular carcinoma in the posterosuperior liver segments. World J Surg 39(5):1202–1209
Yoon YS, Han HS, Cho JY, Ahn KS (2010) Total laparoscopic liver resection for hepatocellular carcinoma located in all segments of the liver. Surg Endosc 24:1630–1637
Dagher I, Proske JM, Carloni A, Richa H, Tranchart H, Franco D (2007) Laparoscopic liver resection: results for 70 patients. Surg Endosc 21:619–624
Nitta H, Sasaki A, Fujita T, Itabashi H, Hoshikawa K, Takahara T, Takahashi M, Nishizuka S, Wakabayashi G (2010) Laparoscopy-assisted major liver resections employing a hanging technique: the original procedure. Ann Surg 251:450–453
Desai PH, Lin JF, Slomovitz BM (2014) Milestones to optimal adoption of robotic technology in gynecology. Obstet Gynecol 123:13–20
Antoniou SA, Antoniou GA, Koch OO, Pointner R, Granderath FA (2012) Robot-assisted laparoscopic surgery of the colon and rectum. Surg Endosc 26:1–11
Autorino R, Zargar H, Kaouk JH (2014) Robotic-assisted laparoscopic surgery: recent advances in urology. Fertil steril 102(4):939–949
Giulianotti PC, Coratti A, Angelini M, Sbrana F, Cecconi S, Balestracci T, Caravaglios G (2003) Robotics in general surgery: personal experience in a large community hospital. Arch Surg 138:777–784
Gagner M, Rheault M, Dubuc J (1992) Laparoscopic partial hepatectomy for liver tumor. Surg Endosc 6:99
Kitisin K, Packiam V, Bartlett DL, Tsung A (2011) A current update on the evolution of robotic liver surgery. Minerva Chir 66:281–293
Fisher SB, Kneuertz PJ, Dodson RM, Patel SH, Maithel SK, Sarmiento JM, Russell MC, Cardona K, Choti MA, Staley CA 3rd, Pawlik TM, Kooby DA (2013) A comparison of right posterior sectorectomy with formal right hepatectomy: a dual-institution study. HPB 15:753–762
Berber E, Akyildiz HY, Aucejo F, Gunasekaran G, Chalikonda S, Fung J (2010) Robotic versus laparoscopic resection of liver tumours. HPB 12:583–586
Ji WB, Wang HG, Zhao ZM, Duan WD, Lu F, Dong JH (2011) Robotic-assisted laparoscopic anatomic hepatectomy in China: initial experience. Ann Surg 253:342–348
Troisi RI, Patriti A, Montalti R, Casciola L (2013) Robot assistance in liver surgery: a real advantage over a fully laparoscopic approach? results of a comparative bi-institutional analysis. MRCAS 9:160–166
Lai EC, Yang GP, Tang CN (2013) Robot-assisted laparoscopic liver resection for hepatocellular carcinoma: short-term outcome. Am J Surg 205:697–702
Wu YM, Hu RH, Lai HS, Lee PH (2014) Robotic-assisted minimally invasive liver resection. Asian J Surg 37:53–57
Tsung A, Geller DA, Sukato DC, Sabbaghian S, Tohme S, Steel J, Marsh W, Reddy SK, Bartlett DL (2014) Robotic versus laparoscopic hepatectomy: a matched comparison. Ann Surg 259:549–555
Spampinato MG, Coratti A, Bianco L, Caniglia F, Laurenzi A, Puleo F, Ettorre GM, Boggi U (2014) Perioperative outcomes of laparoscopic and robot-assisted major hepatectomies: an Italian multi-institutional comparative study. Surg endosc 28(10):2973–2979
Meunier A, Petersson A, Good L, Berlin G (2008) Validation of a haemoglobin dilution method for estimation of blood loss. Vox Sang 95:120–124
Gold JS, Are C, Kornprat P, Jarnagin WR, Gonen M, Fong Y, DeMatteo RP, Blumgart LH, D’Angelica M (2008) Increased use of parenchymal-sparing surgery for bilateral liver metastases from colorectal cancer is associated with improved mortality without change in oncologic outcome: trends in treatment over time in 440 patients. Ann Surg 247:109–117
Montalti R, Berardi G, Laurent S, Sebastiani S, Ferdinande L, Libbrecht LJ, Smeets P, Brescia A, Rogiers X, de Hemptinne B, Geboes K, Troisi RI (2014) Laparoscopic liver resection compared to open approach in patients with colorectal liver metastases improves further resectability: oncological outcomes of a case–control matched-pairs analysis. Eur J Surg Oncol 40:536–544
von Heesen M, Schuld J, Sperling J, Grunhage F, Lammert F, Richter S, Schilling MK, Kollmar O (2012) Parenchyma-preserving hepatic resection for colorectal liver metastases. Langenbecks Arch Surg 397:383–395
Strasberg SM (2005) Nomenclature of hepatic anatomy and resections: a review of the Brisbane 2000 system. J Hepatobiliary Pancreat Surg 12(5):351–355
Schultz NA, Larsen PN, Klarskov B, Plum LM, Frederiksen HJ, Christensen BM, Kehlet H, Hillingso JG (2013) Evaluation of a fast-track programme for patients undergoing liver resection. Br J surg 100:138–143
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Slankamenac K, Graf R, Barkun J, Puhan MA, Clavien PA (2013) The comprehensive complication index: a novel continuous scale to measure surgical morbidity. Ann Surg 258:1–7
Casciola L, Patriti A, Ceccarelli G, Bartoli A, Ceribelli C, Spaziani A (2011) Robot-assisted parenchymal-sparing liver surgery including lesions located in the posterosuperior segments. Surg Endosc 25:3815–3824
Patriti A, Ceccarelli G, Bartoli A, Casciola L (2011) Extracorporeal Pringle maneuver in robot-assisted liver surgery. Surg Laparosc Endosc Percutan Tech 21:e242–e244
Casciola L, Codacci-Pisanelli M, Ceccarelli G, Bartoli A, Di Zitti L, Patriti A (2008) A modified umbilical incision for specimen extraction after laparoscopic abdominal surgery. Surg Endosc 22:784–786
PS Matching in SPSS. http://sourceforge.net/projects/psmspss/
Thoemmes F (2012) Propensity score matching in SPSS. http://arxiv.org/ftp/arxiv/papers/1201/1201.6385.pdf
Vibert E, Perniceni T, Levard H, Denet C, Shahri NK, Gayet B (2006) Laparoscopic liver resection. Br J surg 93:67–72
Makuuchi M, Yamamoto J, Takayama T, Kosuge T, Gunven P, Yamazaki S, Hasegawa H (1991) Extrahepatic division of the right hepatic vein in hepatectomy. Hepatogastroenterology 38:176–179
Ishizawa T, Gumbs AA, Kokudo N, Gayet B (2012) Laparoscopic segmentectomy of the liver: from segment I–VIII. Ann Surg 256:959–964
Araki K, Conrad C, Ogiso S, Kuwano H, Gayet B (2014) Intraoperative ultrasonography of laparoscopic hepatectomy: key technique for safe liver transection. J Am Coll Surg 218:e37–e41
Schneider CM, Peng PD, Taylor RH, Dachs GW 2nd, Hasser CJ, DiMaio SP, Choti MA (2012) Robot-assisted laparoscopic ultrasonography for hepatic surgery. Surgery 151:756–762
Yoon YS, Han HS, Cho JY, Kim JH, Kwon Y (2013) Laparoscopic liver resection for centrally located tumors close to the hilum, major hepatic veins, or inferior vena cava. Surgery 153:502–509
Nguyen KT, Marsh JW, Tsung A, Steel JJ, Gamblin TC, Geller DA (2011) Comparative benefits of laparoscopic vs open hepatic resection: a critical appraisal. Arch Surg 146:348–356
Kim HH, Park EK, Seoung JS, Hur YH, Koh YS, Kim JC, Cho CK, Kim HJ (2011) Liver resection for hepatocellular carcinoma: case-matched analysis of laparoscopic versus open resection. J Korean Surg Soc 80:412–419
Abu Hilal M, Underwood T, Zuccaro M, Primrose J, Pearce N (2010) Short- and medium-term results of totally laparoscopic resection for colorectal liver metastases. Br J surg 97:927–933
Ikeda T, Mano Y, Morita K, Hashimoto N, Kayashima H, Masuda A, Ikegami T, Yoshizumi T, Shirabe K, Maehara Y (2013) Pure laparoscopic hepatectomy in semiprone position for right hepatic major resection. J Hepato-Biliary-Pancreat Sci 20:145–150
Lai EC, Tang CN, Yang GP, Li MK (2011) Multimodality laparoscopic liver resection for hepatic malignancy—from conventional total laparoscopic approach to robot-assisted laparoscopic approach. Int J surg 9:324–328
Packiam V, Bartlett DL, Tohme S, Reddy S, Marsh JW, Geller DA, Tsung A (2012) Minimally invasive liver resection: robotic versus laparoscopic left lateral sectionectomy. J Gastrointest Surg 16:2233–2238
Wakabayashi G, Cherqui D, Geller DA, Buell JF, Kaneko H, Seong Han H, Asbun H, O’Rourke N, Tanabe M, Koffron AJ, Tsung A, Soubrane O, Machado MA, Gayet B, Troisi RI, Pessaux P, Van Dam RM, Scatton O, Abu Hilal M, Belli G, Kwon CHD, Edwin B, Choi GH, Aldrighetti LA, Cai X, Cleary S, Chen K-H, Schön MR, Sugioka A, Tang C-N, Herman P, Pekolj J, Chen X-P, Dagher I, Jarnagin W, Yamamoto M, Strong R, Jagannath P, Lo C-M, Clavien P-A, Kokudo N, Barkun J, Strasberg SM (2015) Current status and recommendations for laparoscopic liver resection: a report from the 2nd international consensus conference held in IWATE. Ann Surg (In press)
Vigano L, Laurent A, Tayar C, Tomatis M, Ponti A, Cherqui D (2009) The learning curve in laparoscopic liver resection: improved feasibility and reproducibility. Ann Surg 250:772–782
Disclosures
Roberto Montalti, Vincenzo Scuderi, Alberto Patriti, Marco Vivarelli, and Roberto I. Troisi declared that have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Montalti, R., Scuderi, V., Patriti, A. et al. Robotic versus laparoscopic resections of posterosuperior segments of the liver: a propensity score-matched comparison. Surg Endosc 30, 1004–1013 (2016). https://doi.org/10.1007/s00464-015-4284-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-015-4284-9