Abstract
Objectives
To evaluate the changes in the mean T2 values of articular cartilage on serial follow-up images up to 1 year in patients who underwent lateral meniscus allograft transplantation (MAT).
Methods
Fifty-two patients who underwent lateral MAT surgery at our hospital were evaluated preoperatively and at 2 days, 6 weeks, 3 months, 6 months, and 1 year after MAT using 3.0-T magnetic resonance imaging (MRI) that included T2 mapping. T2 value changes according to the arthroscopic grading of chondromalacia were evaluated in the lateral and medial compartment. Lysholm scores were obtained pre- and postoperatively.
Results
The T2 values of cartilage were significantly increased 2 days after operation, and then gradually reduced to the baseline level after 1 year in both compartments. In morphologic assessment performed after 1 year, most areas (92.9 %) showed no interval change of chondromalacia grade. Lyshom knee scores increased significantly from the mean preoperative value of 62.5 (range, 23–95) to 89.7 (range, 64–100) at 1 year (p < 0.001).
Conclusion
Mean T2 values of cartilage following MAT exhibited a return to baseline level after 1 year. T2 measurement can be a useful tool for quantitative evaluation of postoperative cartilage changes compared to conventional MRI.
Key Points
• T2 mapping provides objective data for longitudinal monitoring following surgery.
• Increased cartilage T2 values post-MAT returned to baseline in one year.
• Further studies are required to predict the chondroprotective effect of MAT.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Meniscus allograft transplantation (MAT) is now considered the preferred treatment option for symptomatic subtotal/total meniscectomized knees in young patients [1, 2]. The purpose of MAT is to provide pain relief and improve functions in the meniscectomized knees. Ultimately, MAT enables a long-term delay in articular cartilage degeneration by restoring normal load distribution [3, 4]. To evaluate the outcomes of MAT, many clinical scoring systems have been used. However, the diagnostic accuracy of clinical assessment does not accurately and objectively reflect the postoperative graft condition and joint preservation effect [5, 6].
Conventional magnetic resonance imaging (MRI) techniques are useful objective diagnostic tools for the detection of definitive morphologic alterations, including transplanted allograft signal change, shrinkage, extrusion, and re-tears as well as cartilage degeneration after MAT [3, 7–12]. However, the main disadvantage of morphologic cartilage imaging is its low sensitivity for detecting early degenerative cartilage changes that precede gross morphologic changes, emphasizing the need for a more sensitive MRI technique [13–16]. In the case of articular cartilage, quantitative T2 mapping provides additional objective data for longitudinal monitoring following surgery or for the evaluating therapeutic treatments [17–21]. Using the quantitative T2 value, alternations in water content and collagen structure in cartilage have been non-invasively evaluated in a number of published studies [19, 22–25]. Therefore, T2 mapping, by assessing the biochemical characteristics of cartilage, could potentially be a useful imaging technique for the follow-up of cartilage-preserving therapies.
To our knowledge, there are no published longitudinal studies on postoperative cartilage monitoring following MAT using T2 mapping. Therefore, the objective of our current study was to evaluate the postoperative changes in the T2 values of articular cartilage following MAT over a 1-year period and compare them with morphological changes and clinical scores.
Materials and methods
Study group
The retrospective study protocol was approved by our institutional review board. The requirement to obtain informed consent was waived. Since August 2012, T2 mapping has been performed with routine knee MRI protocols for patients who underwent MAT at our institution. Patients were eligible for MAT if they were not old enough to be considered for knee arthroplasty, had a history of subtotal or total meniscectomy, and had degenerative changes and associated localized pain in the involved compartment. Lateral MAT was performed using a fresh-frozen allograft with keyhole technique. All patients who underwent lateral MAT were evaluated preoperatively and again at 2 days, 6 weeks, 3 months, 6 months, and 1 year using 3.0-Tesla (T) MRI including T2 mapping of the medial and lateral tibiofemoral condyle.
Inclusion criteria were as follows: (a) patients who underwent lateral MAT and have serial follow-up MRI up to 1 year; (b) patients had no history of trauma or previous knee surgery; (c) patients had no history of inflammatory or infectious arthritis. The exclusion criteria included a history of medial meniscus transplantation, any loss of follow-up MRI images, and limited evaluation of T2 map due to misregistration between images by consensus of two musculoskeletal radiologists (S-Y.P, and S.H.L). Finally, we selected 52 consecutive patients (26 males, 26 females) from August 2012 to July 2014.
Image acquisition
MRI was performed on an Achieva or Ingenia 3.0-T MR scanner (Philips Medical System, Best, The Netherlands) using a dedicated, 16-channel knee coil. The knee MRI protocol included T2 mapping, which was performed after routine conventional MRI for 30 min in the resting position. For the morphological evaluation of cartilage, we used the imaging sequence summarized in Table 1. The T2 map consisted of a sagittal, multi-echo spin-echo (SE), T2-weighted sequence performed with the following parameters: a repetition time (TR) of 3500 ms and six echo times (TE) of 13, 26, 39, 52, 65, and 78 ms; slice thickness, 3 mm; field of view, 160 x 160 mm; pixel matrix, 304 x 304; and a total acquisition time of 7 min 56 s. The T2 relaxation time was obtained from T2 maps reconstructed using a multi-echo measurement. T2 maps of the articular cartilage of the knee joint, with a colour scale ranging between 1 and 81 msec, were created from the T2 mapping source data.
Data analysis
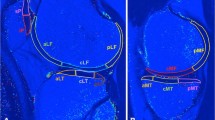
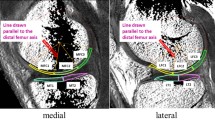
T2 maps were analyzed using an advanced cartilage analysis application (IntelliSpace Portal, Philips Healthcare, The Netherlands). In this program, a least-squares regression weighted by the variance of the estimates of the signal intensity was used [26]. Two separate sagittal T2 maps were selected for analysis at 6 mm medial and 6 mm lateral from the central point of articulation of the lateral tibiofemoral joint (Fig. 1a). The same measurements were taken from the non-operated medial compartment as a control. The region of interest (ROI) analyses were performed at the main weight-bearing area of the articular cartilage, which was divided into three portions, the anterior and posterior meniscal coverage areas, and the middle weight-bearing portion (Fig. 1b). Each ROI included the entire thickness of the articular cartilage and was equally divided into superficial and deep layers for zonal analysis (Fig. 1c).
Segmentation method for quantifying the articular cartilage of the femoral condyle. (a) Two, separate, sagittal T2 maps were selected for analysis 6 mm medially and 6 mm laterally (black line) from the central point of articulation (dotted line) of the lateral tibiofemoral joint. (b) ROI analyses were performed at the main weight-bearing area of the articular cartilage divided into three portions including the anterior and posterior meniscal coverage areas, and the middle weight-bearing portion. (c) Matched with sagittal T2-weighted imaging, T2 measurement was performed using a colour-scale T2 map. Two enlarged sections of sagittal cartilage T2 map were selected. Each subregion included the entire thickness of the articular cartilage and was equally divided into two equal-sized deep and superficial layers
Efforts were made to measure the same ROI at each of the six time points in each patient. Care was taken to avoid a partial volume effect due to the joint fluid and the subchondral bone plate during the ROI evaluation. The T2 maps from the 52 patients were reviewed by two, musculoskeletal radiologists (S-Y.P and S.H.L., with three and fifteen years of clinical experience, respectively) and ROI measurement was performed by one musculoskeletal radiologist (S-Y.P) at first. ROI analyses were manually performed for image evaluation. The readers were blind with regard to the arthroscopic findings of chondromalacia.
The T2 mapping from 10 randomly chosen patients that contained 1440 ROIs were reviewed a second time independently by the two musculoskeletal radiologists for qualitative study. The second review of the T2 mapping was performed in the same manner as the initial review and was used to assess interobserver agreement.
In order to evaluate the morphological change before and after MAT for each ROI, we used conventional MR sequences using sagittal and coronal proton density (PD) fast spine-echo (FSE), and 3D fat-saturated intermediate-weighted FSE images.
Grading system
The morphological MRI classification system for grading of a chondral abnormality is based on modified Outerbridge’s classification. The chondromalacia was graded at each location with reference to the arthroscopic grading taking into account the depth and size of the most severe chondral lesions [27, 28]. In our present study, the modified Outerbridge’s classification was simplified and re-grouped into three groups as follows: group 1 (corresponding to grades 0 and I); group 2 (corresponding to grade II); and group 3 (corresponding to grades III and IV). Arthroscopic examination was performed during MAT.
Clinical evaluation
All patients were evaluated by an orthopaedic surgeon to determine the clinical score at baseline and at 1 year after MAT. The modified Lysholm scoring system was used for the assessment of clinical outcomes of knee surgeries involving patients with chondral damage. The Lysholm knee scale is an 8-item questionnaire scored on a 0–100 weighted scale measuring pain, instability, locking, swelling, limp, stair-climbing, squatting and use of support [29].
Statistical analysis
A linear mixed model [30] was used to analyze the mean T2 value changes of articular cartilage during 1-year follow-up. The mean T2 values of medial and lateral compartments, the deep and superficial cartilage layers of the articular cartilage, and the chondromalacia groups were also evaluated on serial follow-up images by a linear mixed model. The paired Student’s t test was used to compare the clinical scores before MAT surgery and at 1-year follow-up. Spearman correlation analysis was used to assess if the percentage change of the mean T2 value from baseline to 1 year was associated with that of the clinical outcome scales. Interobserver agreement between the two readers was by assessed using ICCs and the 95 % Bland-Altman limits of agreement. An intraclass correlation coefficient (ICC) greater than 0.75 was considered to represent good agreement [31]. Values of p < 0.05 were considered to indicate statistical significance. Data handling and statistical analyses were performed using SAS® version 9.2 (SAS Institute, Inc., Cary, NC, USA), SPSS version 20.0 (SPSS Institute, Chicago, IL), and MedCalc version 16.2.1 (MedCalc Software, Mariakerke, Belgium).
Results
Study subject characteristics
Fifty-two patients who underwent lateral MAT surgery at our hospital were included in the study. Our study group consisted of 26 men and 26 women with an average of age 32.0 ± 11 years (age range, 15–55 years) at the time of meniscus transplantation. T2 maps from the 2496 allocated regions in the 52 patients were measured at all 6 MRI time points, that is, preoperatively and 2 days, 6 weeks, 3 months, 6 months, and 1 year following the surgery. A total of 14,976 ROIs were measured for T2 value evaluation.
Morphologic evaluation
Table 2 shows the subject characteristics of the number of allocated regions of cartilage segmentation and those of the chondromalacia grade in each segment during operation. There were 72 group 1 lesions, 177 group 2 lesions, and 63 group 3 lesions in the lateral femoral condyles, and there were 84 group 1 lesions, 173 group 2 lesions, and 55 group 3 lesions in the lateral tibial plateau. In follow-up cartilage assessment performed after one-year, most areas (92.9 %) showed no interval change of the chondromalacia grade. Sixteen cartilage lesions (2.6 %) showed aggravation of the chondromalacia grade after MAT. Twenty-eight cartilage lesions (4.5 %), all of which belonged to group 3, showed improvement of the chondromalacia grade on the follow-up images.
T2 value changes of articular cartilage on serial MRI
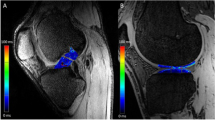
There was a trend of decrease in the mean T2 value following MAT in both femoral condyles and tibial plateaus at each time point over the one year (Table 3 and Figs. 2 and 3). The mean T2 values of both femoral condyles and tibial plateaus rapidly increased 2 days following MAT surgery compared with the preoperative T2 values (p < 0.001), and then gradually reduced to the baseline level after 1 year in both tibiofemoral compartments. The decrease of T2 value from the 2 days postoperative was most prominent during the first 6 weeks following the surgery in both femoral condyles and tibial plateaus.
Sagittal colour-scaled T2 maps of group three chondromalacia in the lateral tibial plateau anterior aspect, performed preoperatively, and at 2 days, 6 weeks, 3 months, 6 months and 1 year after MAT. The T2 maps show initial increase of T2 value on a 2-day image following MAT, and then gradually decreased up to one year (circles)
Interobserver agreement of ROI measurement of T2 maps was good for both readers with ICCs ranging from 0.76 to 0.92, with a mean of 0.85. The coefficient of repeatability for measurement of interobserver agreement ranged from 7.2 % to 13.2 % of the mean T2 values (Table 4). Bland-Altman plots for T2 measurement of lateral femoral condyles and lateral tibial plateaus are shown in the supplementary material.
T2 value changes according to the chondromalacia
In all three chondromalacia groups, there were significant differences in the mean T2 values in both the superficial and deep cartilage layers within the lateral tibiofemoral joint (p < 0.001, Table 5). The mean T2 values differed significantly among the chondromalacia groups, with the highest values in group 3 and the lowest in group 1 at the two layers of both the femoral and tibial cartilages. Furthermore, zonal variations with the characteristic pattern of high T2 values in superficial cartilage layers were noted in both medial and lateral tibiofemoral compartment (p < 0.001).
Correlation between mean T2 value changes and clinical scores
Regarding the postoperative outcome, most of the patients except two patients showed success of the operation. The Lyshom knee score increased significantly from the mean preoperative value of 62.5 (range, 23–95) to 89.7 (range, 64–100) at 1 year after MAT (p < 0.001). Spearman correlation analysis showed weak negative correlation in the lateral tibial plateaus between the percentage change in the preoperative values to 1-year mean T2 values and clinical scores (r = -0.283, p = 0.049), but no relationship was evident in lateral femoral condyles (p > 0.05; Fig. 4).
Scatter plots for the correlation analysis between the percentage changes in the preoperative values to 1-year mean T2 values and clinical scores in lateral femoral condyles (a) and lateral tibial plateaus (b). T2 values correlate significantly with clinical scores in lateral tibial plateaus (r = -0.283, p = 0.049)
Discussion
MAT has been shown to produce effective clinical outcome for patients with symptomatic meniscal deficient knees [32, 33]. The results of this study also revealed clinical improvement after MAT. In conventional MRI, there was no change of articular cartilage in 92.9 % and improvement was shown in 4.5 %. However, T2 mapping demonstrated dynamic changes of cartilage, showing an initial increase in mean cartilage T2 value after MAT followed by return to baseline level after 1 year, although there was no strong correlation between T2 value and clinical score.
The initial increase in T2 value after MAT at 2 days postoperative in this study could be explained by the restriction of motion during the patient’s hospitalization. Mamisch et al. reported that even after a 45-minute interval in which patients were supine during MRI, the late unloading T2 values were significantly higher than the early unloading values in both cartilage repair tissue and native cartilage after matrix-associated autologous chondrocyte transplantation [23]. This could represent an increase in the mean T2 value at 2 days postoperatively. Similarly, in a previous study using an animal model, immobilization of the knee joint increased the water content and subsequently decreased the proportional solid component of the cartilage [34].
Several studies involving quantitative cartilage T2 assessment have examined the loading condition in knee cartilage. Liess et al. found in an in vivo study that detection of changes in cartilage water content due to exercise and loading is feasible with T2 mapping. Rubenstein et al. reported a T2 decrease in bovine articular cartilage in response to compressive force that resulted from a combination of net water loss and an alternation in collagen orientational structure [35]. Subburaj et al. investigated the changes of T2 relaxation time in young healthy adults after 30 minute of running. A significantly large reduction in relaxation times is observed in the central weight-bearing region of the femoral condyle [36]. In agreement with these studies, we speculate that a possible explanation for the gradual decrease in the T2 value following MAT might be the effect of decreased joint effusion and cartilage loading during the postoperative period following rehabilitation or daily activity. After surgery, weight-bearing of the knee joints may cause water in cartilage to move through the extracellular matrix due to mechanical compression and the pressure gradient. This would then be revealed as a gradual decrease of the T2 value during the patient’s recovery period in both femoral condyles and tibial plateaus.
Apart from the trend toward a T2 decline, the individual T2 values differed significantly according to the chondromalacia grade; that is, they were the highest in group 3 and the lowest in group 1 in the two layers of both femoral and tibial cartilage. Our T2 values were similar to those already reported. Lusse et al. showed a positive correlation between increases in the T2 relaxation time and the cartilage water content in ex vivo cartilage obtained from patients with osteoarthritis who underwent total knee replacement surgery [37]. Dunn et al. reported a correlation between increased T2 values and the severity of osteoarthritis, with patients with osteoarthritis showing significantly higher cartilage T2 values than healthy patients [38]. Together with chondromalacia grades, zonal variations in the mean T2 measurements from the deep to superficial cartilage layers were observed in both femoral condyles and tibial plateaus at all time points. These earlier results are consistent with the findings of our present study, with a comparable zonal pattern for healthy control cartilage.
In most studies, MAT outcomes have been evaluated using clinical parameters, regardless of the symptomatic improvements or deterioration [5]. However, these clinical assessments might not accurately reflect the true status of the transplanted meniscus [6]. Quantitative T2 mapping of articular cartilage allows the non-invasive assessment of compositional changes in articular cartilage following meniscus transplantation. Little attempts have been made previously to correlate clinical outcomes with MRI or even T2 mapping [3, 8]. Although this study concerns relatively short-term data, we tried to determine if there was a correlation between the mean T2 value and the clinical score. Ideally, for clinically significant data, a controlled trial should be performed with a larger sample size and appropriate adjustment for confounding variables.
Our study had several limitations. Firstly, the T2 value was manually segmented for analysis, which is operator-dependent and time-consuming. Automated cartilage segmentation software might allow a larger dataset for cartilage evaluation, especially in a longitudinal study. Secondly, we used arthroscopy as a reference standard for the chondromalacia grade, not a histological evaluation. There is significant operator variability in arthroscopy, particularly regarding the chondromalacia location or grade. Published data suggest that arthroscopy is approximately 95 % accurate and some results may be misinterpreted when performed by an inexperienced operator [39]. In our institution, one experienced arthroscopic knee surgeon with a consistent chondromalacia grade record performed the MAT. Third, there is no control cohort in this study. The current study aimed to observe the T2 value changes following MAT over 1 year. Unfortunately, it was not possible to provide a control group as it would be unethical to carry out this surgical procedure on a healthy group. Therefore, the medial tibiofemoral compartment was chosen as a control in our current analyses to improve the reliability. Finally, two different MR instruments in the same vendor’s scanner were used for this single-center study. In further study, prior to scanning, an inter-scanner comparison study should be taken for feasibility.
The goals of MAT are to provide functional improvement following replacement of the meniscus cushion and to prevent further articular degeneration in a symptomatic meniscectomized knee. Furthermore, the ultimate aim is to achieve a long-term chondroprotective effect, which still has been actively researched. Only the postoperative reduction in T2 value could be observed over 1 year in this study; however, a further study with a longer period should be performed using quantitative MRI to establish the lasting chondroprotective effect.
This study demonstrates postoperative recovery of knee articular cartilage after MAT, quantified with T2 values of the cartilage’s return to baseline level after 1 year. T2 measurement can be a useful tool and a more sensitive biomarker for quantitative evaluation of postoperative cartilage changes compared to conventional MRI.
References
Sekiya JK, Ellingson CI (2006) Meniscal allograft transplantation. J Am Acad Orthop Surg 14:164–174
Rijk PC (2004) Meniscal allograft transplantation—part I: background, results, graft selection and preservation, and surgical considerations. Arthroscopy 20:728–743
Verdonk PC, Verstraete KL, Almqvist KF et al (2006) Meniscal allograft transplantation: long-term clinical results with radiological and magnetic resonance imaging correlations. Knee Surg Sports Traumatol Arthrosc 14:694–706
Wirth CJ, Peters G, Milachowski KA, Weismeier KG, Kohn D (2002) Long-term results of meniscal allograft transplantation. Am J Sports Med 30:174–181
Lubowitz JH, Verdonk PCM, Reid JB III, Verdonk R (2007) Meniscus allograft transplantation: a current concepts review. Knee Surg Sports Traumatol Arthrosc 15:476–492
Lee B-S, Kim J-M, Sohn D-W, Bin S-I (2013) Review of meniscal allograft transplantation focusing on long-term results and evaluation methods. Knee Surg Relat Res 25:1–6
Alparslan L, Winalski CS, Boutin RD, Minas T (2001) Postoperative magnetic resonance imaging of articular cartilage repair. Semin Musculoskelet Radiol 5:345–363
Verstraete KL, Verdonk R, Lootens T, Verstraete P, De Rooy J, Kunnen M (1997) Current status and imaging of allograft meniscal transplantation. Eur J Radiol 26:16–22
Lee BS, Chung JW, Kim JM, Cho WJ, Kim KA, Bin SI (2012) Morphologic changes in fresh-frozen meniscus allografts over 1 year: a prospective magnetic resonance imaging study on the width and thickness of transplants. Am J Sports Med 40:1384–1391
Lee DH, Kim TH, Lee SH, Kim CW, Kim JM, Bin SI (2008) Evaluation of meniscus allograft transplantation with serial magnetic resonance imaging during the first postoperative year: focus on graft extrusion. Arthroscopy 24:1115–1121
Trattnig S, Millington SA, Szomolanyi P, Marlovits S (2007) MR imaging of osteochondral grafts and autologous chondrocyte implantation. Eur Radiol 17:103–118
Horng A, Raya JG, Stockinger M et al (2015) Topographic deformation patterns of knee cartilage after exercises with high knee flexion: an in vivo 3D MRI study using voxel-based analysis at 3T. Eur Radiol 25:1731–1741
Disler DG, McCauley TR, Wirth CR, Fuchs MD (1995) Detection of knee hyaline cartilage defects using fat-suppressed three-dimensional spoiled gradient-echo MR imaging: comparison with standard MR imaging and correlation with arthroscopy. AJR Am J Roentgenol 165:377–382
Kijowski R, Blankenbaker DG, Munoz Del Rio A, Baer GS, Graf BK (2013) Evaluation of the articular cartilage of the knee joint: value of adding a T2 mapping sequence to a routine MR imaging protocol. Radiology 267:503–513
Rubenstein JD, Li JG, Majumdar S, Henkelman RM (1997) Image resolution and signal-to-noise ratio requirements for MR imaging of degenerative cartilage. AJR Am J Roentgenol 169:1089–1096
Cha JG, Yi JS, Han JK, Lee YK (2015) Comparison of Quantitative Cartilage T2 Measurements and Qualitative MR Imaging between Professional Ballet Dancers and Healthy Volunteers. Radiology 276:199–206
Link TM, Stahl R, Woertler K (2007) Cartilage imaging: motivation, techniques, current and future significance. Eur Radiol 17:1135–1146
Kim HK, Laor T, Graham TB et al (2010) T2 relaxation time changes in distal femoral articular cartilage in children with juvenile idiopathic arthritis: a 3-year longitudinal study. AJR Am J Roentgenol 195:1021–1025
Mosher TJ, Dardzinski BJ (2004) Cartilage MRI T2 relaxation time mapping: overview and applications. Semin Musculoskelet Radiol 8:355–368
Welsch GH, Mamisch TC, Domayer SE et al (2008) Cartilage T2 assessment at 3-T MR imaging: in vivo differentiation of normal hyaline cartilage from reparative tissue after two cartilage repair procedures--initial experience. Radiology 247:154–161
Welsch GH, Mamisch TC, Quirbach S, Zak L, Marlovits S, Trattnig S (2009) Evaluation and comparison of cartilage repair tissue of the patella and medial femoral condyle by using morphological MRI and biochemical zonal T2 mapping. Eur Radiol 19:1253–1262
Crema MD, Roemer FW, Marra MD et al (2011) Articular cartilage in the knee: current MR imaging techniques and applications in clinical practice and research. Radiographics 31:37–61
Mamisch TC, Trattnig S, Quirbach S, Marlovits S, White LM, Welsch GH (2010) Quantitative T2 mapping of knee cartilage: differentiation of healthy control cartilage and cartilage repair tissue in the knee with unloading--initial results. Radiology 254:818–826
Nardo L, Carballido-Gamio J, Tang S, Lai A, Krug R (2016) Quantitative assessment of morphology, T, and T of shoulder cartilage using MRI. Eur Radiol. doi:10.1007/s00330-016-4322-6
Juras V, Bohndorf K, Heule R et al (2015) A comparison of multi-echo spin-echo and triple-echo steady-state T2 mapping for in vivo evaluation of articular cartilage. Eur Radiol. doi:10.1007/s00330-015-3979-6
Raya JG, Dietrich O, Horng A, Weber J, Reiser MF, Glaser C (2010) T2 measurement in articular cartilage: impact of the fitting method on accuracy and precision at low SNR. Magn Reson Med 63:181–193
Curl WW, Krome J, Gordon ES, Rushing J, Smith BP, Poehling GG (1997) Cartilage injuries: a review of 31,516 knee arthroscopies. Arthroscopy 13:456–460
Mandelbaum BR, Browne JE, Fu F et al (1998) Articular cartilage lesions of the knee. Am J Sports Med 26:853–861
Wright RW (2009) Knee injury outcomes measures. J Am Acad Orthop Surg 17:31–39
Laird NM, Ware JH (1982) Random-effects models for longitudinal data. Biometrics 38:963–974
Büsing KA, Kilian AK, Schaible T, Debus A, Weiss C, Neff KW (2008) Reliability and Validity of MR Image Lung Volume Measurement in Fetuses with Congenital Diaphragmatic Hernia and in Vitro Lung Models 1. Radiology 246:553–561
Smith NA, MacKay N, Costa M, Spalding T (2015) Meniscal allograft transplantation in a symptomatic meniscal deficient knee: a systematic review. Knee Surg Sports Traumatol Arthrosc 23:270–279
Milachowski KA, Weismeier K, Wirth CJ (1989) Homologous meniscus transplantation. Int Orthop 13:1–11
Vanwanseele B, Lucchinetti E, Stussi E (2002) The effects of immobilization on the characteristics of articular cartilage: current concepts and future directions. Osteoarthritis Cartil 10:408–419
Rubenstein JD, Kim JK, Henkelman RM (1996) Effects of compression and recovery on bovine articular cartilage: appearance on MR images. Radiology 201:843–850
Subburaj K, Kumar D, Souza RB et al (2012) The acute effect of running on knee articular cartilage and meniscus magnetic resonance relaxation times in young healthy adults. Am J Sports Med 40:2134–2141
Lusse S, Claassen H, Gehrke T et al (2000) Evaluation of water content by spatially resolved transverse relaxation times of human articular cartilage. Magn Reson Imaging 18:423–430
Dunn TC, Lu Y, Jin H, Ries MD, Majumdar S (2004) T2 relaxation time of cartilage at MR imaging: comparison with severity of knee osteoarthritis. Radiology 232:592–598
Nickinson R, Darrah C, Donell S (2010) Accuracy of clinical diagnosis in patients undergoing knee arthroscopy. Int Orthop 34:39–44
Acknowledgments
The scientific guarantor of this publication is Sang Hoon Lee, professor at the Asan Medical Center. The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article. The authors state that this work has not received any funding. No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was waived by the Institutional Review Board.
Methodology: retrospective, observational study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 364 kb)
Rights and permissions
About this article
Cite this article
Park, SY., Lee, S.H., Lee, M.H. et al. Changes in the T2 value of cartilage after meniscus transplantation over 1 year. Eur Radiol 27, 1496–1504 (2017). https://doi.org/10.1007/s00330-016-4497-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-016-4497-x