Abstract
Summary
To evaluate bone mineral density (BMD) and morphometric vertebral fractures (MVF) in chronic obstructive pulmonary disease (COPD) patients in comparison with two control groups. BMD was lower in the disease group (DG) and was associated with the worst disease severity and prognosis. The prevalence of MVF was high and greater in the DG than in the control groups.
Introduction
Chronic obstructive pulmonary disease (COPD) is associated with osteoporosis and vertebral fractures. It is still unclear whether the presence of fractures and changes in bone mineral density (BMD) are associated with disease severity and prognosis. The aim of this study was to evaluate BMD and morphometric vertebral fractures (MVF) in COPD patients in comparison with two control groups and to correlate these parameters with indices of COPD severity (VEF1 and GOLD) and prognosis (BODE).
Methods
This was a cross-sectional study in COPD patients (disease group, DG) who underwent BMD and vertebral fracture assessment (VFA). Two control groups were used: smokers without COPD (smoker group, SG) and healthy never-smoker individuals (never-smoker group, NSG).
Results
The DG comprised 121 patients (65 women, mean age 67.9 ± 8.6 years). Altered BMD was observed in 88.4% of the patients in the DG, which was more prevalent when compared with the control groups (p < 0.001). The BMD values were lower in the DG than in the control groups (p < 0.05). BMD was associated with the worst disease severity and prognosis (p < 0.05). The prevalence of MVF was high (57.8%) and greater than that in the SG (23.8%) and the NSG (14.8%; p < 0.001). The prevalence of fractures was not associated with disease severity and prognosis.
Conclusions
COPD patients have a higher prevalence of MVF and low BMD, and the latter was associated with the severity and poor prognosis of the disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chronic obstructive pulmonary disease (COPD) is a condition characterized by progressive and not fully reversible airflow limitation [1]. Respiratory exacerbations, along with the presence of comorbidities, contribute to the overall severity of the disease [2]. COPD has a high prevalence, affecting up to 10% of individuals above age of 40 years and is associated with high morbidity and mortality rates [3]. Despite being primarily a lung disease, COPD is associated with several extrapulmonary disorders, including cardiovascular disease, sleep apnea, depression, anemia, chronic kidney disease, osteoporosis, cachexia, and skeletal muscle weakness [4].
Low bone mineral density (BMD) that leads to osteoporosis is common in COPD patients, and previous studies have reported rates of osteoporosis of 9–69% in this population [5,6,7]. The etiology of low BMD in these patients is likely multifactorial and includes smoking, vitamin D deficiency, hypercapnia, hypoxia, poor nutrition, low body mass index (BMI), inflammatory cytokines, and decreased lean mass [8,9,10]. In addition to low BMD, COPD patients also present altered bone quality and microstructure, leading to a high prevalence of fractures. Indeed, 30–63% of COPD patients present vertebral fractures [11]. In this population, thoracic vertebral fractures are particularly concerning because each vertebral fracture is estimated to lead to a 9% decline in the patient’s forced vital capacity (FVC) [12].
It is still unclear whether the presence of fractures and changes in BMD in COPD patients is associated with disease severity and prognosis [13], and data from the literature have shown controversial results in this regard [14, 15]. In contrast, evidence suggests that newly diagnosed COPD patients already have a high prevalence of osteoporosis and fractures [16].
The main aim of this study was to evaluate BMD and morphometric vertebral fractures (MVF) in COPD patients compared with two control groups, one consisting of smokers without COPD and the other consisting of healthy never-smoker individuals. Secondary aims included correlating these parameters with indices of COPD severity and prognosis.
Patients and methods
Study groups
This was a cross-sectional study performed on a group of COPD patients treated at the Pulmonary Outpatient Clinic of the Hospital de Clinicas at Universidade Federal do Parana, Curitiba, South Brazil. All patients signed an informed consent form, and the study was approved by the Ethics Committee on Human Research of our institution.
The patients included in this study had been invited to undergo BMD and vertebral fracture assessment (VFA) between January 2010 and December 2014. The inclusion criteria were men and women older than 50 years with previous diagnosis of tobacco-induced COPD. COPD was evaluated by spirometry (KoKo PFT Spirometer, Occupational Health Dynamics, Hoover, AL, USA) with a post-bronchodilator (FEV1/FVC < 0.70) as defined by the Global Initiative for Chronic Obstructive Lung Disease (GOLD) [1]. Patients were excluded if one of the required tests was unavailable or if the patient was taking medications (including continuous oral glucocorticoid) or had another disease known to interfere with bone mass. Two control groups matched by age and sex were used for comparison with the COPD group (disease group, DG): smokers without COPD (smoker group, SG) and healthy never-smoker individuals (never-smoker group, NSG). Individuals in the SG were invited to participate during their first visit to a smoking cessation clinic of the same hospital, and individuals in the NSG were volunteers of both sexes from a random population invited to participate in the study. All individuals in the control groups were older than 50 years and had undergone evaluation of BMD and VFA with the same equipment as those in the DG. Exclusion criteria were the use of medications or the presence of diseases known to interfere with bone mass. All individuals in the SG underwent spirometry using the same spirometer used for the patients in the DG and were excluded from the study if diagnosed with COPD.
Weight (kilograms) was measured on a digital electronic scale while the participants wore light clothes. Height (meters) was measured while the individuals kept their back straight, heels together, and arms extended alongside their body. Their BMI was determined by dividing the weight (in kilograms) by the squared height (in square meter) [17].
All participants underwent a BMD assessment and VFA using dual-energy X-ray absorptiometry (DXA) on a lunar prodigy whole-body scanner (GE Medical Systems, Madison, WI, USA). The regions evaluated in the DXA were the lumbar spine (LS), total femur (TF), and femoral neck (FN). The BMD results were expressed as grams per centimeter and as scores in relation to reference values determined by the International Society for Clinical Densitometry (ISCD) [18]. In our center, the least significant change (LSC) for the lumbar spine was 0.010 g/cm2 and for the proximal femur was 0.012 g/cm2. The VFA was performed simultaneously and was analyzed by a single experienced physician using the Genant semiquantitative method [19].
COPD severity and prognosis
We collected the patients’ clinical data, including information about their lifetime smoking exposure (quantified in pack-years) [20] and history of exacerbations, using post-bronchodilator forced expiratory volume in the first second (FEV1), the modified Medical Research Council (mMRC) dyspnea scale [21], the COPD Assessment Test (CAT) [22], and a 6-min walk test [23].
Patients in the DG were classified according to the degree of airflow obstruction (FEV1) into categories 1 (≥ 80%), 2 (50–79%), 3 (30–49%), and 4 (< 30%) [20]. The COPD severity was determined according to the GOLD index—which is based on the post-bronchodilator FEV1, history of exacerbations in the previous year, and symptoms such as dyspnea (measured with the mMRC or CAT)—and the study groups were classified into A, B, C, and D. The COPD prognosis was evaluated with the body mass index, airflow obstruction, dyspnea, and exercise (BODE) index [24]—which integrates the patient’s BMI, the degree of airflow obstruction, grade of dyspnea, and exercise capacity—with total criteria point count varying from 0 to 10. Additionally, the patients were classified into four quartiles, with the first being the least severe and the fourth being the most severe.
Statistical analysis
Data are presented as the mean ± standard deviation (SD). All analyses were performed using IBM SPSS Statistics, v.20.0 (IBM Corp, Armonk, NY, USA). The normality of the distribution of the variables was evaluated by the Kolmogorov-Smirnov test. The comparison between two groups of quantitative variables was performed with Student’s t test for independent samples or using the nonparametric Mann-Whitney test. When comparing more than two groups, we used the analysis of variance (ANOVA) with one factor and the least significant difference (LSD) test for multiple comparisons or the nonparametric Kruskal-Wallis test. For the preliminary statistical analysis, we used Fisher’s exact test and the chi-square test to assess the association between two qualitative variables. P values below 0.05 were considered statistically significant.
Results
Figure 1 is the flow chart of the participants.
Flow chart describing patients distribution in the different COPD classifications. COPD chronic obstructive pulmonary disease, FEV1 post-bronchodilator forced expiratory volume in the first second, BODE: B body mass index, O airway obstruction, D dyspnea, E exercise capacity, GOLD Global Initiative for Chronic Obstructive Lung Disease
From 758 COPD patients in treatment at the pneumology clinic, 150 consecutive COPD patients were invited to participate in the study; 127 accepted the invitation, and from these, six were excluded for not having all required tests in their records. The final DG comprised 121 patients (65 women, mean age 67.9 ± 8.6 years, mean BMI 26.5 ± 6.2 kg/m2). The mean tobacco consumption was 58.9 ± 40.8 pack-years. Overall, 23 patients (19.1%) were still smoking at the time of the cross-sectional evaluation. According to the degree of obstruction evaluated by the FEV1, 21 patients (17.3%) were classified as group 1, 48 (39.6%) were classified as group 2, 39 (32.2%) were classified as group 3, and 13 (10.7%) were classified as group 4. Based on the GOLD classification, the DG was subdivided as follows: GOLD A: 29 patients (23.9%), GOLD B: 29 patients (23.9%), GOLD C: 34 patients (28%), and GOLD D: 29 patients (23.9%). According to the BODE index, 55 patients (45.4%) were in the first quartile, 37 patients (30.5%) were in the second quartile, 18 patients (14.8%) were in the third quartile, and 11 (9%) were in the fourth quartile.
There were 29 patients (22.8%, GOLD A) taking short action beta 2 agonists, 92 patients (72.4%, GOLD B plus C and D) taking long action beta 2 agonists, and 63 patients (49.6%, GOLD C plus D) taking inhaled corticosteroids.
The SG comprised 63 individuals (29 women) with a mean age of 65.5 ± 8.9 years and a mean BMI 27.6 ± 3.6 kg/m2. All individuals were smokers at the time of the study evaluation. The mean tobacco consumption was 38 ± 28.2 pack-years. The NSG comprised 81 individuals (47 women) with a mean age of 66 ± 8.5 years and a mean BMI of 26.1 ± 2.6 kg/m2, and in this group, none of the participants ever smoked. There were no differences in gender, age, or BMI among the groups, although the mean tobacco consumption in the DG was significantly higher than that in the SG (Table 1).
Abnormal BMD was observed in 88.4% of the patients in the DG; 49 (40.5%) had osteopenia [24 (36.9%) and 25 (44.6%) of women and men, respectively] and 58 (47.9%) had osteoporosis [33 (50.7%) and 25 (44.6%) of women and men, respectively]. Abnormal BMD was more prevalent in the DG than in the SG (68.2%, p < 0.001) and the NSG (72.8%, p < 0.001, Table 1). The distribution of BMD classifications was similar in both control groups: in the SG, 31 patients (49.2%) had osteopenia [11 (31.4%) and 21 (60%) of women and men, respectively], 12 (19%) had osteoporosis [10 (28.5%) and 2 (5.7%) of women and men, respectively], and 20 (31.7%) had normal BMD [14 (40%) and 12 (34.2%) of women and men, respectively]. In the NSG, 47 patients (58%) had osteopenia [28 (59.7%) and 19 (55.8%) of women and men, respectively], 12 (14.8%) had osteoporosis [9 (19.1%) and 3 (8.8%) of women and men, respectively], and 22 (27.1%) had normal BMD [10 (21.2%) and 12 (35.2%) of women and men, respectively]. The BMD (grams per square centimeter) values in all three sites evaluated were lower in the DG than in the control groups (Fig. 2). BMD was associated with the degree of obstruction (FEV1), GOLD, and BODE (p < 0.05, Fig. 3).
Bone mineral density at lumbar spine (LS), femoral neck (FN), and total femur (TF) in the disease group (DG) comprising 121 patients with chronic obstructive pulmonary disease (COPD), 63 smokers without COPD (SG), and 81 never-smoker healthy individuals (NSG). Horizontal lines in the box plots indicate median and 5, 25, 75, and 95 percentiles. BMD bone mineral density, LS lumbar spine, FN femoral neck, TF total femur. *Comparison between the DG and the SG. **Comparison between the DG and the NSG, p < 0.05
Bone mineral density according to the severity and prognosis of COPD. LS lumbar spine, FN femoral neck, TF total femur, FEV1 post-bronchodilator forced expiratory volume in the first second, BODE: B body mass index, O airway obstruction, D dyspnea, E exercise capacity, GOLD Global Initiative for Chronic Obstructive Lung Disease; significance = p < 0.05
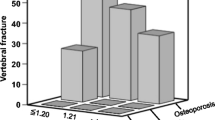
One hundred eighty-four vertebral fractures were found in 70 COPD patients. The prevalence of MVF was significantly higher in the DG than in the SG (57.8% vs. 23.8%, p < 0.001) and the NSG (14.8%, p < 0.001, Figs. 3 and 4). There was no difference in the prevalence of MVF between the SG and the NSG.
The thoracic region was the location mostly affected with MVF in all groups, with the prevalence of 87.7%, 93.3%, and 100% in the DG, SG, and NSG, respectively. In the DG, the prevalence of fractures was associated with low BMD in FN (p = 0.02), but not in the LS and TF. In the control groups, there was no association between low BMD and MVF. Moreover, we did not find an association between the prevalence of fractures and FEV1, GOLD, or BODE in any of the study groups.
Discussion
This is the first study comparing COPD patients with two control groups to evaluate simultaneously BMD and MVF and their association with the severity and prognosis of the disease. The smoking load was higher in the DG when compared with the SG, which was expected considering that the number of pack-years is related to the development of COPD, although other factors are also involved in this process [25, 26].
The high prevalence of altered BMD in the DG (88.4%), in which approximately 50% of the patients showed osteoporosis, is consistent with rates described in the literature (9–69%) [6] and is different from the prevalence of altered BMD in both control groups. Several factors may contribute to a reduction in bone mass with COPD, including inflammatory cytokines, hypercapnia, hypoxia, impaired nutrition, immobilization, hypogonadism, vitamin D deficiency, low BMI, lean mass reduction, and decreased exercise capacity [8, 9]. We observed no differences in the prevalence of osteopenia or osteoporosis among smokers and never-smokers, which contradicts some studies in the literature that have found smoking to be an independent risk factor for osteoporosis [27,28,29]. Decreased BMD was associated with the degree of obstruction, clinical staging, and COPD’s BODE prognostic index results that confirm others in the literature [13, 28]. The possible explanations for the higher BMD in GOLD 4 COPD patients than in the GOLD 3 COPD patients may be the low number of patients in each GOLD stage; the severity of the disease in GOLD 3 and GOLD 4 COPD patients may be equally as bad for the bone as shown by Watanabe R et al. [30].
The prevalence of MVF was increased in COPD patients (57.8%), similar to the literature [31, 32], and was higher than that in both control groups. Despite the high prevalence of fractures, they were not associated with disease severity or prognosis in COPD. The literature has controversial data regarding the association of fractures with disease severity in COPD [14, 32]. A recent study with 99 COPD patients also could not significantly correlate the number of vertebral deformities with FEV1 for men or women with COPD, and increasing age was the only factor significantly associated with vertebral fractures [33]. Majumdar et al. [32] suggested that patients with lower lung function had more severe vertebral fractures. The possible explanations for this lack of association may be the existence of decreases in bone quality starting at early stages of the disease [34, 35], limited sample size [33], or, in part, related to how “severity” is defined [32].
The limitations of the study were that no laboratory evaluation related to the bone metabolism of these patients was performed and that a spine X-ray also was not done to corroborate the diagnosis of vertebral fractures.
In conclusion, this was the first study that compared COPD patients with two control groups and evaluated simultaneously the BMD and MVF with the severity and prognosis of the disease. This study showed a high prevalence of low BMD and vertebral fractures in COPD patients, serving as an alert for physicians to weigh in on potential skeletal changes that occur in these patients, in addition to the changes related to aging. These results may provide evidence for the need to implement interventions for prevention of osteoporosis and fractures, thus improving the quality of life of COPD patients.
References
Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease. National Heart, Lung and Blood Institutes. Revised 2011. www.goldcopd.com
Cielen N, Maes K, Gayan-Ramirez G (2014) Musculoskeletal disorders in chronic obstructive pulmonary disease. Biomed Res Int 2014:96576
Eagan TM, Aukrust P, Upland T, Hardier JA, Johannessen A, Mollnes TE et al (2010) Body composition and plasma levels of inflammatory biomarkers in COPD. Eur Respir J 36(5):1027–1033
Decramer M, Janssens W, Miravitlles M (2012) Chronic obstructive pulmonary disease. Lancet 379(9823):1341–1351
Miller J, Edwards LD, Agustí A, Bakke P, Calverley PM, Celli B et al (2013) Evaluation of COPD longitudinally to identify predictive surrogate endpoints (ECLIPSE) investigators. Comorbidity, systemic inflammation and outcomes in the ECLIPSE cohort. Respir Med 107(9):1376–1384
Franco CB, Paz-Filho G, Gomes PE, Nascimento VB, Kulak CA, Boguszewski CL et al (2009) Chronic obstructive pulmonary disease is associated with osteoporosis and low levels of vitamin D. Osteoporos Int 20(11):1881–1887
Graat-Verboom L, Wouters EF, Smeenk FW, van den Borne BE, Lunde R, Spruit MA (2009) Current status of research on osteoporosis in COPD: a systematic review. Eur Respir J 34:209–218
Yamamoto Y, Yoshikawa M, Tomoda K, Fujita Y, Yamauchi M, Fukuoka A et al (2014) Distribution of bone mineral content is associated with body weight and exercise capacity in patients with chronic obstructive pulmonary disease. Respiration 87(2):158–164
Fouda MA, Alhamad EH, Al-Hajjaj MS, Shaik SA, Alboukai AA, Al-Kassimi FA (2017) A study of chronic obstructive pulmonary disease-specific causes of osteoporosis with emphasis on the emphysema phenotype. Ann Thorac Med 12(2):101–106
Hamdy RC (2016) Bone mineral density and fractures. J Clin Densitom 19(2):125–126
Nuti R, Siviero P, Maggi S, Guglielmi G, Caffarelli C, Crepaldi G et al (2009) Vertebral fractures in patients with chronic obstructive pulmonary disease: the EOLO Study. Osteoporos Int 20(6):989–998
Schlaich C, Minne HW, Bruckner T (1998) Reduced pulmonary function in patients with spinal osteoporotic fractures. Osteoporos Int 8:261–267
Silva DR, Coelho AC, Dumke A, Valentini JD, de Nunes JN, Stefani CL et al (2011) Osteoporosis prevalence and associated factors in patients with COPD: a cross-sectional study. Respir Care 56(7):961–968
Ogura-Tomomatsu H, Asano K, Tomomatsu K, Miyata J, Ohmori N, Kodama M et al (2012) Predictors of osteoporosis and vertebral fractures in patients presenting with moderate-to-severe chronic obstructive lung disease. COPD 9(4):332–337
Koo HK, Park JH, Park HK, Jung H, Lee SS (2014) Conflicting role of sarcopenia and obesity in male patients with chronic obstructive pulmonary disease: Korean National Health and Nutrition Examination Survey. PLoS One 9(10):e110448
Soriano JB, Visick GT, Muellerova H, Payvandi N, Hansell AL (2005) Patterns of comorbidities in newly diagnosed COPD and asthma in primary care. Chest 128(4):2099–2107
World Health Organ (2000) Obesity: preventing and managing the global epidemic. Report of a WHO consultation. Tech Rep Ser 894:i–xii 1–253
(2015) 6th ISCD position development conference (adult). www.iscd.org
Genant HK, Jergas M, Palermo L, Nevitt M, Valentin RS, Black D et al (1996) Comparison of semiquantitative visual and quantitative morphometric assessment of prevalent and incident vertebral fractures in osteoporosis The Study of Osteoporotic Fractures Research Group. J Bone Miner Res 11(7):984–996
Prignot J (1987) Quantification and chemical markers of tobacco-exposure. Eur J Respir Dis 70(1):1–7
Ferris BG (1978) Epidemiology standardization project (American Thoracic Society). Am Rev Respir Dis 118(6 Pt 2):1–120
Jones PW, Harding G, Berry P (2009) Development and first validation of the COPD assessment test. Eur Respir J 34(3):648–654
Balke B (1963) A simple field test for the assessment of physical fitness. Rep 63–6. Rep Civ Aeromed Res Inst US
Celli B, Cote C, Marin J (2004) The body mass index, airflow obstruction, dyspnea, and exercise capacity index in chronic obstructive pulmonary disease. N Engl J Med 350:1005–1012
Stern DA, Morgan WJ, Wright AL, Guerra S, Martinez FD (2007) Poor airway function in early infancy and lung function by age 22 years: a non-selective longitudinal cohort study. Lancet 370(9589):758–764
Hunninghake GM, Cho MH, Tesfaigzi Y, Soto-Quiros ME, Avila L, Lasky-Su J, Stidley C et al (2009) MMP12, lung function, and COPD in high-risk populations. N Engl J Med 361(27):2599–2608
Pinheiro MM, Ciconelli RM, Martini LA, Ferraz MB (2009) Clinical risk factors for osteoporotic fractures in Brazilian women and men: the Brazilian Osteoporosis Study (BRAZOS). Osteoporos Int 20(3):399–408
Sakurai-Iesato Y, Kawata N, Tada Y, Iesato K, Matsuura Y, Yahaba M et al (2017) The relationship of bone mineral density in men with chronic obstructive pulmonary disease classified according to the global initiative for chronic obstructive lung disease (GOLD) combined chronic obstructive pulmonary disease (COPD) assessment system. Intern Med 56(14):1781–1790
Romme EA, Murchison JT, Edwards LD, van Beek E Jr, Murchison DM, Rutten EP et al (2013) Evaluation of COPD longitudinally to identify predictive surrogate endpoints (ECLIPSE) study investigators. CT-measured bone attenuation in patients with chronic obstructive pulmonary disease: relation to clinical features and outcomes. J Bone Miner Res 28(6):1369–1377
Watanabe R, Tanaka T, Aita K, Hagiya M, Homma T, Yokosuka K et al (2015) Osteoporosis is highly prevalent in Japanese males with chronic obstructive pulmonary disease and is associated with deteriorated pulmonary function. J Bone Miner Metab 33(4):392–400
Kjensli A, Mowinckel P, Ryg MS, Falch JA (2007) Low bone mineral density is related to severity of chronic obstructive pulmonary disease. Bone 40:493–497
Majumdar SR, Villa-Roel C, Lyons KJ, Rowe BH (2010) Prevalence and predictors of vertebral fracture in patients with chronic obstructive pulmonary disease. Respir Med 104(2):260–266
Graumam RQ, Pinheiro MM, Nery LE, Castro CHM (2018) Increased rate of osteoporosis, low lean mass, and fragility fractures in COPD patients: association with disease severity. Osteoporos Int 29:1457–1468. https://doi.org/10.1007/s00198-018-4483-z
Kulak CA, Borba VC, Jorgetti V, Dos Reis LM, Liu XS, Kimmel DB et al (2010) Skeletal microstructural abnormalities in postmenopausal women with chronic obstructive pulmonary disease. J Bone Miner Res 25(9):1931–1940
Jaramillo JD, Wilson C, Stinson DS, Lynch DA, Bowler RP, Lutz S et al (2015) Reduced bone density and vertebral fractures in smokers. Men and COPD patients at increased risk. Ann Am Thorac Soc 12(5):648–654
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflicts of interest
None.
Rights and permissions
About this article
Cite this article
Munhoz da Rocha Lemos Costa, T., Costa, F.M., Hoffman Jonasson, T. et al. Bone mineral density and vertebral fractures and their relationship with pulmonary dysfunction in patients with chronic obstructive pulmonary disease. Osteoporos Int 29, 2537–2543 (2018). https://doi.org/10.1007/s00198-018-4643-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-018-4643-1