Abstract
Key message
A novel locus on Agropyron cristatum chromosome 6P that increases grain number and spikelet number was identified in wheat–A. cristatum derivatives and across 3 years.
Abstract
Agropyron cristatum (2n = 4x = 28, PPPP), which has the characteristics of high yield with multiple flowers and spikelets, is a promising gene donor for wheat high-yield improvement. Identifying the genetic loci and genes that regulate yield could elucidate the genetic variations in yield-related traits and provide novel gene sources and insights for high-yield wheat breeding. In this study, cytological analysis and molecular marker analysis revealed that del10a and del31a were wheat–A. cristatum chromosome 6P deletion lines. Notably, del10a carried a segment of the full 6PS and 6PL bin (1–13), while del31a carried a segment of the full 6PS and 6PL bin (1–8). The agronomic characterization and genetic population analysis confirmed that the 6PL bin (9–13) brought about an increase in grain number per spike (average increase of 10.43 grains) and spikelet number per spike (average increase of 3.67) over the three growing seasons. Furthermore, through resequencing, a multiple grain number locus was mapped to the physical interval of 593.03–713.89 Mb on chromosome 6P of A. cristatum Z559. The RNA-seq analysis revealed the expression of 537 genes in the del10a young spike tissue, with the annotation indicating that 16 of these genes were associated with grain number and spikelet number. Finally, a total of ten A. cristatum-specific molecular markers were developed for this interval. In summary, this study presents novel genetic material that is useful for high-yield wheat breeding initiatives to meet the challenge of global food security through enhanced agricultural production.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Common wheat (Triticum aestivum, 2n = 6x = 42, AABBDD) is a major crop that feeds approximately 40% of the world’s population and provides 20% of all human calories and protein (Calderini et al. 2020; Xiao et al. 2022). Increasing grain number per spike (GNS) is an important way to increase grain yield while facing the challenges of increasing population and shrinking farmland area (Duan et al. 2018; Miransari and Smith 2019). GNS can be physically and genetically divided into two subcomponents: the spikelet number per spike (Boden et al. 2015; Glenn et al. 2023; Li et al. 2021) and the fertile floret number per spike (Sakuma et al. 2019; Zhang et al. 2022). However, there have been limitations to improvements in wheat, as there is a lack of novel genes and germplasms exhibiting high-yield potential.
The transfer of excellent genetic variation from wheat relatives to common wheat through distant hybridization is an effective method for enriching the genetic diversity of wheat (Li et al. 2008). For example, the wheat–rye (2n = 2x = 14, RR) T1RS·1BL translocation line is widely used in high-yield and disease-resistant wheat breeding (Hackauf et al. 2022; Mago et al. 2005; Mater et al. 2004), and approximately 1050 wheat cultivars that carry the T1RS·1BL translocation inherit the chromosome arm 1RS of the rye cultivar Petkus (Schneider et al. 2016). In addition, the powdery mildew resistance gene Pm21 from Dasypyrum villosum (2n = 2x = 14, VV), which is widely used in wheat disease resistance breeding in China, was cloned by map-based cloning and next-generation sequencing strategies (He et al. 2018; Xing et al. 2018). Therefore, identification of the ideal alien donor and introduction of high-yield genes into wheat are feasible for high-yield breeding.
Agropyron cristatum (2n = 4x = 28, PPPP) is an alien donor involved in wheat improvement. It has a great number of spikelets and flowers, and has the ability to confer resistance to various biotic and abiotic stresses (Farashi and Karimian 2021; Jiang et al. 2018; Li et al. 2016; Lin et al. 2023). In recent studies, we demonstrated that chromosome 6P was responsible for significant increases in the GNS, spikelet number per spike (SNS) and fertile floret number per spikelet (FFNS) (Wu et al. 2006), and the transfer of chromosome 6P into wheat has potential application value for increasing wheat yield. To narrow the physical interval of genes that regulate the grain number and to develop novel germplasms for wheat breeding, many translocation and deletion lines have been created by radiation with different segments from the chromosome 6P addition line 4844-12 (Song et al. 2013). In recent years, we confirmed that the genetic effect on increasing grain number was derived from chromosome 6PL (Zhang et al. 2019).
To further pinpoint the genetic loci regulating grain number and explore potential candidate genes, two novel deletion lines del10a (6PS·6PL bin 1–13) and del31a (6PS·6PL bin 1–8) were developed in this study. Through cytological, molecular marker and genetic analyses of agronomic traits, the enhanced-GNS locus was mapped to the 6PL bin (9–13). In this interval, the annotations of 16 genes were associated with grain number and were expressed in young spike of del10a. Additionally, we have developed ten specific molecular markers for A. cristatum that can be utilized in marker-assisted selection for this multi-grain genetic locus. In conclusion, we found a novel locus for increasing grain number and spikelet number in the 6PL bin (9–13), laying a foundation for further mapping, cloning and using A. cristatum genes for wheat breeding.
Materials and methods
Plant materials
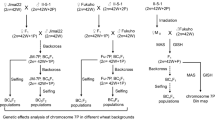
The common wheat–A. cristatum (Z559, PPPP, 2n = 4x = 28; from Xinjiang) deletion lines del10c (6PL bin 1–13) (Song et al. 2016a), del31a (6PS·6PL bin 1–8) and del10a (6PS·6PL bin 1–13) were derived from the chromosome 6P disomic addition line 4844-12 through radiation induction. Specifically, del10c (6PL bin 1–13) and del31a (6PS·6PL bin 1–8) were generated via five consecutive backcrosses with Fukuhokomugi (Fukuho; AABBDD, 2n = 6x = 42; sourced from Japan). In parallel, del10a was developed through five consecutive backcrosses with Gaocheng8901 (AABBDD, 2n = 6x = 42; from China). The BC5F2 and BC6F2 populations of del10c were subjected to genetic analysis, and the positive plants characterized by the presence of A. cristatum chromatin and negative plants lacking A. cristatum chromatin within the populations were distinguished by A. cristatum-specific repeat sequence markers, namely Acpr3a and Acpr7, which can be used to detect the chromatin of A. cristatum in wheat (Han et al. 2016). Del19b and del21 are also deletion lines; del19b has only chromosome 6PL, and del21 has only chromosome 6PS (Zhang et al. 2019). The details of all the materials are presented in Table S1. All materials were maintained at Prof. Lihui Li’s laboratory at the Institute of Crop Science, Chinese Academy of Agricultural Sciences.
GISH and FISH analysis
Genomic in situ hybridization (GISH) was performed to detect the chromosomes of A. cristatum (Z559) in del10a and del31a. The chromosomes of the root tip cells were analyzed according to the methods of Chen and Armstrong (1994). Z559 genomic DNA (gDNA) was labeled with Texas-Red-5-dCTP (red) as a GISH probe, while Fukuho gDNA was used as a block, and the ratio of probe to block was 1:40. The root tip cells were counterstained with 4′,6-diamidino-2-phenylindole (DAPI). Fluorescence in situ hybridization (FISH) was performed with the oligonucleotide probes Oligo-pSc119.2-1 (green) and Oligo-pTa535-1 (red) to determine the integrity of the alien chromosomes and the variation in the wheat chromosomes (Tang et al. 2014). A similar procedure was performed according to the methods of Han et al. (2006, 2009), and all probes were synthesized by Sangon Biotech (Shanghai). Fluorescent signals were observed via an Olympus AX80 fluorescence microscope (Olympus Corporation, Tokyo, Japan), imaged with a charge-coupled device (CCD) camera (Diagnostic Institute, Inc. Sterling Height, MI, USA) and modified with Photoshop CS6.
Chromosome 6P-specific molecular marker genotyping for del10a and del31a
Previously, Zhang et al. (2015) designed the chromosome 6P-specific expressed sequence tag-sequence tagged site (EST-STS) marker according to the transcriptome of A. cristatum, and Song et al. (2016b) used deletion and translocation lines to divide the chromosome 6P into 31 regions (6PS 14 regions, 6PL 17 regions) and mapped 255 6P-specific EST-STS markers to 31 regions. Among them, a total of thirty-one 6P-specific markers from 31 bins (one marker from each of the bins) were selected for detecting the segments of chromosome 6P in del10a and del31a. Details of the molecular markers used are shown in Table S2. All primers were synthesized by Shanghai Sangon Biotech Co., Ltd. (Shanghai, China).
A modified cetyltrimethyl ammonium bromide (CTAB) method was used to extract genomic DNA from all plant materials (Carra et al. 2007). A total of 20 μl of reaction mixture was used for PCR, which consisted of 10 μl of Green Master Mix (Nanjing Vazyme Biotech Co., Ltd.), 1.2 μl of DNA template (100 ng/μl), 1 μl of each primer (2 μM) and 6.8 μl of ddH2O. Each PCR was performed as follows: one cycle at 94 °C for 5 min for denaturation; 35 cycles of 94 °C for 30 s, 56–58 °C (depending on the annealing temperature for each marker) for 30 s and 72 °C for 30 s for extension; one cycle at 72 °C for 10 min for final extension; and maintenance at 4 °C. The PCR products were assessed by 1% agarose gel electrophoresis.
Analysis of agronomic characteristics
All materials were planted at the Xinxiang Experimental Base of Henan, Institute of Crop Science, Chinese Academy of Agricultural Sciences, during three growing seasons in 2020–2021, 2021–2022 and 2022–2023. Each row was 2 m long and was planted with 20 seeds; and the seeds were approximately 10 cm apart, and the row spacing was 25 cm. The deletion lines del10a, del10c and del31a and the recurrent parent Fukuho and Gaocheng8901 were randomly planted in the field over three growing seasons. Additionally, both positive and negative plants in the BC5F2 and BC6F2 genetic populations of del10c were also randomly planted. The plant height (PH, cm), spike length (SL, cm), effective tillering (ET), SNS, FFNS, GNS and thousand-grain weight (TGW, g) were determined after harvest. The measurement methods for each trait were as follows: PH is the height of the plant, calculated from the base of the stem to the top spikelet, not including the root length; SL indicates the length of the main spike, measured by the distance from the base spikelet to the top spikelet, not including the awn length; SNS represents the total number of spikelets in the main spike, including sterile spikelets and fertile spikelets; ET is the number of spikes per plant; FFNS is the largest kernel number in a spikelet from the middle region; GNS is the total grain number in the main spikelet; and TGW was measured by an SC-G seed detector (Hangzhou WSeen Detection Technology Co., Ltd.). The significant differences in the agronomic characteristics of Fukuho, Gaocheng8901, del10a, del31a and del10c were determined using one-way ANOVA at a probability level of p = 0.05. The significant differences in positive and negative plants of the del10c genetic populations were determined using Student’s t test at the probability levels of p < 0.05 (*) and p < 0.01 (**).
Resequencing analysis and molecular marker development
The leaves of del10a and del31a were collected at the seedling stage for resequencing to determine the physical interval of the 6PL bin (9–13) at Huazhi Biotech Co., Ltd. (Changsha, China). The DNA libraries were generated using MGIEasy DNA library preparation kits following the manufacturer’s recommendations, and the libraries were sequenced using the BGISEQ-500 platform with a paired-end read length of 150 bp (Lin et al. 2022). The sequencing depth was approximately 1X. The raw data were filtered by “fastp” tools (Chen et al. 2018), and the clean reads were mapped to the integrated A. cristatum Z559 reference sequences (unpublished) and the Chinese Spring (CS) RefSeqv1.0 (https://urgi.versailles.inra.fr/download/iwgsc/IWGSC_RefSeq_Assemblies/v1.0/) using the BWA tool (Houtgast et al. 2018). BEDTools was used to construct windows for the genome and calculate the sequencing coverage of reads in each window, for which the window size was 1 Mb (Quinlan and Hall 2010). The ggplot2 package of R was used to visualize read coverage (Gustavsson et al. 2022). The genes associated with grain number and spikelet number were obtained via trait annotation and domain annotation. SAMtools was used to extract gene sequences (Li et al. 2009), the BLASTN tool was used to identify SNP differences between A. cristatum and wheat CS (Ye et al. 2012), and primers were designed for SNP loci and synthesized by Shanghai Sangon Biotech Co., Ltd. PCR was the same as that used for the EST-STS markers. The bands present in 4844-12 and del10a but absent in Fukuho, Gaocheng8901 and del31a were identified as specific markers.
RNA-seq analysis
RNA was extracted from the young spike of del10a at the double ridge stage using TRIzol reagent (Thermo Fisher Scientific Inc., Shanghai, China) according to the manufacturer’s instructions. The RNA libraries were sequenced on the Illumina sequencing platform by Smartgenomics Technology Institute (Tianjin, China). One biological replicate was used for young spike tissue, and the sequencing volume was 10 G for each sample. The clean data were obtained by removing the reads containing adaptors, poly-N sequences and low-quality reads using the “fastp” tool (Chen et al. 2018). The clean data from each sample were separately aligned to the wheat CS reference genome (IWGSC RefSeq v1.0) and the reference genome of Z559 by the HISAT2 tool (Kim et al. 2019). Then the count of each gene was calculated by the featureCounts tool (Liao et al. 2014).
Real-time reverse transcription PCR (qRT-PCR)
The total RNA from the leaf, sheath, stem and young spike of del10a were extracted and reverse transcribed into cDNA for qRT–PCR using a Reverse Transcriptase Kit (ZOMANBIO, Beijing, China). The spike development genes from 6PL bin (9–13) were selected for qRT–PCR, and the qRT–PCR primers were designed using Primer 5 software. The qRT–PCR was performed using a TB Green®® Premix Ex Taq™ II (Takara, Osaka, Japan) kit in a StepOnePlus Real-Time PCR System (Applied Biosystems, Carlsbad, CA, USA). Three technical replicates were set for each biological sample, and the relative quantification of gene expression was calculated by the 2−∆∆Ct method. The wheat Actin (TraesCS1B02G024500) gene was used as the housekeeping gene to calibrate the expression levels of genes.
Results
Chromosome composition analysis of del10a and del31a
Tracking of the chromatin of A. cristatum in wheat was enabled with GISH by using gDNA from A. cristatum as a probe, and FISH was used to detect chromosome variation by using the oligonucleotide probes Oligo-pSc119.2-1 and Oligo-pTa535-1. The results revealed that both del10a and del31a had forty-two wheat chromosomes with blue 4’,6-diamidino-2-phenylindole (DAPI) signals and two 6P chromosomes from A. cristatum with red signals (Fig. 1), and compared with the chromosome 6P in the addition line 4844-12, the chromosome 6P in del10a and del31a was incomplete (Fig. 2a–c). The FISH analysis indicated that chromosome 6P in del10a and del31a lacked segments of the chromosome 6PL terminus, and the segment of chromosome 6P in del10a was larger than that in del31a (Fig. 2d). Therefore, del10a and del31a are wheat–A. cristatum chromosome 6P deletion lines with different segments.
Cytogenetic analysis of del10a and del31a by in situ hybridization. GISH (a) and FISH (b) analysis of del10a; GISH (c) and FISH (d) analysis of del31a. Chromosomes were counterstained with DAPI and visualized with blue fluorescence. The red signal in a and c represents the chromosome of Agropyron cristatum, the FISH probe Oligo-pTa535-1 is shown in red, and Oligo-pSc119.2-1 is shown in green in b and d. Scale bar = 10 μm
Chromosome length analysis of del10a and del31a. GISH (a) and FISH (b) analysis of 4844-12; (c) and (d) comparison of fluorescence signals between del10a, del31a and 4844-12. The FISH probes Oligo-pTa535-1 (red) and Oligo-pSc119.2-1 (green) are shown in b and d, and the GISH probes (red) are shown in a and c. Scale bar = 10 μm (color figure online)
To further determine the segments in del10a and del31a, a total of thirty-one 6P-specific EST-STS molecular markers were used for genotyping. The results demonstrated that 14 markers from 6PS had specific bands amplified in 4844-12 and del21; 17 markers from 6PL had specific bands amplified in 4844-12 and del19b; all 31 markers were absent in the recurrent parents Fukuho and Gaocheng8901; four markers (Agc10192, Agc33878, Agc26468 and Agc8139) from the 6PL bin (14–17) were missing the specific bands in del10a and del31a; and five markers (Agc5532, Agc2453, Agc32015, Agc9467 and Agc3960) from the 6PL bin (9–13) were also missing the specific bands in del31a (Table S3, Fig. 3). Therefore, we confirmed that del10a had a segment with the full 6PS and 6PL bin (1–13), while del31a carried a segment with the full 6PS and 6PL bin (1–8).
Evaluation of agronomic traits and location of multiple grain number locus
The agronomic performance of del10a, del31a, del10c (6PL bin 1–13), Fukuho and Gaocheng8901 were investigated during the three growing seasons of 2020–2021, 2021–2022 and 2022–2023 in Xinxiang, Henan Province. The results indicated that, compared with the GNS, SNS and PH of Gaocheng8901, those of del10a increased significantly by 11.00, 10.90 and 9.40; by 3.50, 3.70 and 3.80; and by 27.20 cm, 21.75 cm and 22.50 cm, respectively, during the three growing seasons (p value < 0.01); and compared with the spike length of Gaocheng8901 (recurrent parent), that of del10a decreased significantly by 1.05, 0.59 and 1.47 cm, respectively (p value < 0.05). Compared with the TGW of Gaocheng8901, that of del10a increased significantly by 3.95 g and 2.55 g in the 2021–2022 and 2022–2023 growing seasons, respectively, while FFNS and ET showed no significant differences. In addition, compared with Fukuho (a recurrent parent), del31a did not exhibit significant increases in GNS, FFNS, TGW and SNS during the three growing seasons, and the del10c exhibited significant increases in GNS, FFNS and SNS (p value < 0.01); moreover, TGW was not significantly different between the 2021–2022 and 2022–2023 growing seasons. In addition, compared with del31a, del10a exhibited significant increases in GNS, FFNS and SNS by 12.90, 21.00 and 19.70; by 1.00, 0.80 and 0.40; and by 6.40, 8.30 and 8.40, respectively, in the three years (p value < 0.05) (Table 1, Figs. 4, 5a–c). Therefore, we speculate that the 6PL bin (9–13) in del10a has genetic benefits that steadily increase GNS and SNS (Fig. 6a).
Agronomic traits of del10a, Gaocheng8901, del31a and Fukuho in the field. 1: del10a; 2: Gaocheng8901; 3: del31a; 4: Fukuho. a and f Plants; b and g Front view of the spike; c and h Side view of a stalk of the spike; d, e, i and j Spikelet. The spikes were chosen from the primary stem, while the spikelets were specifically selected from the central region of the spike. Scale bar in a and f = 10 cm; scale bar in b, c, g and h = 1 cm; scale bar in d, e, i and j = 0.5 cm
Significance analysis of agronomic traits. a Significance analysis of Fukuho, del31a, Gaocheng8901 and del10a regarding SNS, FFNS, GNS and TGW in 2020–2021; b Significance analysis of Fukuho, del10c, del31a, Gaocheng8901 and del10a regarding SNS, FFNS, GNS and TGW in 2021–2022; c Significance analysis of Fukuho, del10c, del31a, Gaocheng8901 and del10a regarding SNS, FFNS, GNS and TGW in 2022–2023; d Significance analysis of BC5F2 population of del10c regarding SNS, FFNS and GNS in 2021–2022; e Significance analysis of BC6F2 population of del10c regarding SNS, FFNS and GNS in 2022–2023
Multiple GNS locus in the 6PL bin (9–13). a On the left are 31 chromosome bins of chromosome 6P. Pink represents A. cristatum chromatin. b Physical interval analysis of the 6PL bin (9–13) by resequencing. The horizontal coordinate represents the position of the 6P chromosome, and the ordinate represents read coverage at the corresponding location. The red dots represent the chromosome 6P of del10a and del31a, and the black dots represent missing parts of the chromosome
To further confirm the genetic effects of the 6PL bin (9–13) in the Fukuho background, the agronomic traits of the BC5F2 and BC6F2 genetic populations of the deletion line del10c (carrying 6PL bin 1–13) were analyzed during the 2021–2022 and 2022–2023 growing seasons. A total of 67 positive plants and 120 negative plants in the BC5F2 population and 39 positive plants and 89 negative plants in the BC6F2 population were identified by the A. cristatum repeat sequence markers Acpr3a and Acpr7. Compared with the SNS, FFNS and GNS in the negative plants, those in the BC5F2 population significantly increased by 0.64, 0.49 and 8.49, representing increases of 3.19%, 10.84% and 13.24%, respectively (p value < 0.01). Similarly, compared with those in the negative plants, the SNS, FFNS and GNS in the BC6F2 population significantly increased by 0.66, 0.41 and 6.20, respectively, representing increases of 3.15%, 9.58% and 10.09%, respectively (p value < 0.01) (Table 1, Fig. 5d, e). Therefore, these results indicated that the 6PL bin (9–13) is a novel locus that is mainly associated with enhanced GNS and SNS.
Physical mapping of the multiple GNS locus
To determine the accuracy of the physical interval of the 6PL bin (9–13), del10a and del31a were resequenced and mapped to the A. cristatum Z559 reference genome by the BWA tool. By calculating the chromosome coverage of the reads, we determined that del10a carried an approximately 0–713.89 Mb interval of chromosome 6P, while del31a carried an approximately 0–593.03 Mb interval of chromosome 6P. Therefore, the multiple grain number locus was located within the 593.03–713.89 Mb region of the 6P chromosome (Fig. 6b). According to the results of de novo prediction, homology searches and transcript-based assembly, a total of 993 protein-coding genes (Ac6P02G357100–Ac6P02G456300) were deposited in the locus of the 6PL bin (9–13).
RNA-seq for del10a and tissue-specific expression analysis for spike development genes
To further investigate the tissue expression of the 993 genes in the 6PL bin (9–13), the young spike of del10a were collected at the double ridge stage for RNA-seq. A total of 537 genes were expressed in young spike. Among these genes, sixteen were associated with spikelet number, spikelet fertility and filled grain number, including five AP2 genes and one MADS-box gene (Table S4). To further characterize these genes, their cDNA sequences were extracted from A. cristatum genome sequences and subsequently utilized for the development of A. cristatum-specific cDNA markers to facilitate tissue-specific expression analysis. Four genes, namely Ac6P02G384400, Ac6P02G393000, Ac6P02G398000 and Ac6P02G410600, successfully generated their specific cDNA markers, denoted as Ac-cDNA3844-1, Ac-cDNA3930-1, Ac-cDNA3980-1 and Ac-cDNA4106-2, respectively (Table S5, Fig. 7a). The result of qRT–PCR demonstrated that Ac6P02G384400, Ac6P02G393000 and Ac6P02G410600 exhibited expression in young spike, stem and sheath tissues, while their expression levels were extremely low in leaf tissue. And Ac6P02G398000 displayed high expression levels across all four tissues (Fig. 7b). However, further work is needed to verify whether these genes indeed function in young spike development of wheat.
A. cristatum-specific cDNA molecular marker development and tissue-specific expression analysis. a Development of A. cristatum-specific cDNA molecular marker for A. cristatum genes from 6PL bin (9–13); b The tissue-specific expression analysis of genes Ac6P02G384400, Ac6P02G393000, Ac6P02G398000 and Ac6P02G410600
Development of specific markers at the locus of multiple GNS
To efficiently trace the chromatin of the 6PL bin (9–13) in wheat, the sequences of ten genes from this interval were extracted from Z559 via SAMtools, the different SNP loci were determined via the BLASTN tool and compared against the wheat CS reference genome, and a total of ten dominant molecular markers were obtained (Table S6). PCR and agarose gel electrophoresis revealed that all the markers with specific bands were amplified from 4844-12 and del10a, while Fukuho, Gaocheng8901 and del31a had none (Fig. 8). In the future, these markers may enable accurate tracing of A. cristatum chromatin in wheat and provide a marker tool for subsequent mining and cloning of high-yield genes to increase grain number and spikelet number.
Discussion
A. cristatum provides germplasm and gene resources for high-yield wheat breeding
Creating new germplasms through distant hybridization with distant relatives and making those germplasms available to breeders are important methods for addressing the issue of the narrowing gene pools in wheat breeding. For example, in terms of providing germplasm and genetic resources for wheat resistance breeding, wheat–rye derivative lines from chromosomes 1R, 4R and 6R with powdery mildew resistance (Han et al. 2020; Ma et al. 2020; Perovic et al. 2015); wheat–Thinopyrum ponticum translocation line WTT80 with powdery mildew resistance; WTT34 with stem rust resistance (Yang et al. 2021a, 2022); wheat–Thinopyrum elongatum translocation lines Zhongke 1878, Zhongke 166 and Zhongke 545 with Fusarium head blight resistance (Guo et al. 2023); and wheat–Thinopyrum intermedium translocation line WTT11 with stripe rust resistance have been developed in wheat breeding (Yang et al. 2021b).
However, there are few reports about the specific genetic traits present in wheat relatives that contribute to an increase in the number of grains and spikelets. Fortunately, A. cristatum, which has multiple flowers and spikelets, is an ideal gene donor for high-yield wheat breeding. Li et al. (1998) achieved hybridization between common wheat and the distant relative A. cristatum in the 1990s, and the wheat–A. cristatum chromosome 6P addition line 4844-12, which exhibited significantly increased grain number, was developed (Wu et al. 2006). To map the genes that regulate grain number on chromosome 6P, many wheat–A. cristatum 6P translocation and deletion lines with different segments were created by radiation induction 4844-12 (Song et al. 2013). In this study, we produced two deletion lines, del10a and del10c, with high GNS and high SNS values and located a novel physical interval in the 6PL bin (9–13) that regulates grain number. These achievements have the potential to be used in wheat breeding to transfer the segment 6PL bin (9–13) into the wheat background in the form of small segment translocation lines by radiation induction. In summary, the deletion lines del10a and del10c presented in this study provide new germplasm and genetic resources for improving wheat yield.
The advantages and disadvantages of chromosomal mapping
Many deletion lines and translocation lines harboring different chromosome segments can be created by radiation induction, which can narrow the physical range of excellent genes. For example, the leaf rust resistance gene in the 2PL bin (0.66–0.86) (Jiang et al. 2018) and the adult powdery mildew resistance gene were found to be located in the 6PL bin (3–7) by deletion and translocation lines mapping (Lin et al. 2022). In addition, the blue-grained gene and powdery mildew resistance gene from Thinopyrum ponticum were located in bin 4AgL-6 with FL 0.75–0.89 and in the 3.79–97.12 Mb region of the 4AgS according to a similar method (Liu et al. 2018; Yang et al. 2023). Therefore, deletion and translocation lines mapping can narrow the physical interval of candidate genes at the chromosomal level to a certain extent. However, due to the segment size of the translocation and deletion lines, the target interval was too large to determine, and candidate genes could not be accurately screened. In this study, a total of 993 genes in the 120.86 Mb interval were identified by resequencing the deletion lines del10a and del31a, and a total of 16 potential candidate genes were annotated. These genes were expressed in the young spike at the double ridge stage. However, we cannot yet determine which genes regulate the GNS.
Therefore, in our future research, we plan to perform radiation induction on del10c, create translocation lines and deletion lines with smaller segments and backcross them with wheat cultivars to create new germplasms for high-yield wheat production and promote the cloning of multiple GNS genes.
The development of molecular markers facilitates the tracing of A. cristatum chromatin and cloning of candidate genes
In hybridization with distant relatives, much alien chromatin can be introduced into the wheat background to produce addition lines, translocation lines and introgression lines. The analysis of agronomic traits and GISH techniques can help identify alien chromatin in wheat. However, GISH does not detect small segments of alien chromatin at the chromosomal level. Therefore, the development of specific molecular markers is critical for tracing alien chromatin in wheat and applying this information to molecular marker-assisted breeding. In this study, we developed 10 PCR-based primers based on the SNP sites between A. cristatum and CS. For example, the markers Ac3778-1 and Ac4024-2 amplified specific bands in 4844-12 and del10a but not in Fukuho, Gaocheng8901 or del31a. Therefore, these markers can be used as convenient and efficient tools for detecting chromatin in the 6PL bin (9–13) of A. cristatum in wheat. On the other hand, these molecular markers provide a tool for further identifying multiple GNS genes in A. cristatum via map-based cloning in wheat.
Abbreviations
- GNS:
-
Grain number per spike
- SNS:
-
Spikelet number per spike
- FFNS:
-
Fertile floret number per spikelet
- TGW:
-
Thousand-grain weight
- GISH:
-
Genomic in situ hybridization
- FISH:
-
Fluorescence in situ hybridization
References
An D, Zheng Q, Luo Q, Ma P, Zhang H, Li L, Han F, Xu H, Xu Y, Zhang X, Zhou Y (2015) Molecular cytogenetic identification of a new wheat–rye 6R chromosome disomic addition line with powdery mildew resistance. PLoS ONE 10:e0134534
Boden SA, Cavanagh C, Cullis BR, Ramm K, Greenwood J, Jean Finnegan E, Trevaskis B, Swain SM (2015) Ppd-1 is a key regulator of inflorescence architecture and paired spikelet development in wheat. Nat Plants 1:14016
Calderini DF, Castillo FM, Arenas-M A, Molero G, Reynolds MP, Craze M, Bowden S, Milner MJ, Wallington EJ, Dowle A, Gomez LD, McQueen-Mason SJ (2020) Overcoming the trade-off between grain weight and number in wheat by the ectopic expression of expansin in developing seeds leads to increased yield potential. New Phytol 230:629–640
Carra A, Gambino G, Schubert A (2007) A cetyltrimethylammonium bromide-based method to extract low-molecular-weight RNA from polysaccharide-rich plant tissues. Anal Biochem 360:318–320
Chen Q, Armstrong K (1994) Genomic in situ hybridization in Avena sativa. Genome 37:607–612
Chen S, Zhou Y, Chen Y, Gu J (2018) fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34:884–890
Duan J, Wu Y, Zhou Y, Ren X, Shao Y, Feng W, Zhu Y, Wang Y, Guo T (2018) Grain number responses to pre-anthesis dry matter and nitrogen in improving wheat yield in the Huang-Huai Plain. Sci Rep 8:7126
Farashi A, Karimian Z (2021) Assessing climate change risks to the geographical distribution of grass species. Plant Signal Behav 16:e1913311
Glenn P, Woods DP, Zhang J, Gabay G, Odle N, Dubcovsky J (2023) Wheat bZIPC1 interacts with FT2 and contributes to the regulation of spikelet number per spike. Theor Appl Genet 136:237
Guo X, Shi Q, Liu Y, Su H, Zhang J, Wang M, Wang C, Wang J, Zhang K, Fu S, Hu X, Jing D, Wang Z, Li J, Zhang P, Liu C, Han F (2023) Systemic development of wheat–Thinopyrum elongatum translocation lines and their deployment in wheat breeding for Fusarium head blight resistance. Plant J 114:1475–1489
Gustavsson EK, Zhang D, Reynolds RH, Garcia-Ruiz S, Ryten M, Mathelier A (2022) ggtranscript: an R package for the visualization and interpretation of transcript isoforms using ggplot2. Bioinformatics 38:3844–3846
Hackauf B, Siekmann D, Fromme FJ (2022) Improving yield and yield stability in winter rye by hybrid breeding. Plants 11:2666
Han F, Gao Z, Birchler JA (2009) Reactivation of an inactive centromere reveals epigenetic and structural components for centromere specification in maize. Plant Cell 21:1929–1939
Han F, Lamb JC, Birchler JA (2006) High frequency of centromere inactivation resulting in stable dicentric chromosomes of maize. Proc Natl Acad Sci 103:3238–3243
Han G, Liu S, Wang J, Jin Y, Zhou Y, Luo Q, Liu H, Zhao H, An D (2020) Identification of an elite wheat-rye T1RS·1BL translocation line conferring high resistance to powdery mildew and stripe rust. Plant Dis 104:2940–2948
Han H, Liu W, Lu Y, Zhang J, Yang X, Li X, Hu Z, Li L (2016) Isolation and application of P genome-specific DNA sequences of Agropyron Gaertn. in Triticeae. Planta 245:425–437
He H, Zhu S, Zhao R, Jiang Z, Ji Y, Ji J, Qiu D, Li H, Bie T (2018) Pm21, encoding a typical CC-NBS-LRR protein, confers broad-spectrum resistance to wheat powdery mildew disease. Mol Plant 11:879–882
Houtgast EJ, Sima V-M, Bertels K, Al-Ars Z (2018) Hardware acceleration of BWA-MEM genomic short read mapping for longer read lengths. Comput Biol Chem 75:54–64
Jiang B, Liu T, Li H, Han H, Li L, Zhang J, Yang X, Zhou S, Li X, Liu W (2018) Physical mapping of a novel locus conferring leaf rust resistance on the long arm of Agropyron cristatum chromosome 2P. Front Plant Sci 9:817
Kim D, Paggi JM, Park C, Bennett C, Salzberg SL (2019) Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat Biotechnol 37:907–915
Liao Y, Smyth GK, Shi W (2014) featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30:923–930
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25:2078–2079
Li H, Jiang B, Wang J, Lu Y, Zhang J, Pan C, Yang X, Li X, Liu W, Li L (2016) Mapping of novel powdery mildew resistance gene(s) from Agropyron cristatum chromosome 2P. Theor Appl Genet 130:109–121
Li L, Yang X, Li X, Dong Y, Chen X (1998) Introduction of desirable genes from Agropyron cristatum into common wheat by intergeneric hybridization. Sci Agricult Sin 31:1–6
Li T, Deng G, Tang Y, Su Y, Wang J, Cheng J, Yang Z, Qiu X, Pu X, Zhang H, Liang J, Yu M, Wei Y, Long H (2021) Identification and validation of a novel locus controlling spikelet number in bread wheat (Triticum aestivum L.). Front Plant Sci 12:611106
Li Z, Li B, Tong Y (2008) The contribution of distant hybridization with decaploid Agropyron elongatum to wheat improvement in China. J Genet Genom 35:451–456
Lin Y, Zhou S, Liang X, Guo B, Han B, Han H, Zhang J, Lu Y, Zhang Z, Yang X, Li X, Liu W, Li L (2022) Chromosomal mapping of a locus associated with adult-stage resistance to powdery mildew from Agropyron cristatum chromosome 6PL in wheat. Theor Appl Genet 135:2861–2873
Lin Y, Zhou S, Liang X, Han B, Yang J, Guo B, Zhang J, Han H, Liu W, Yang X, Li X, Li L (2023) Introgression of chromosome 6PL terminal segment from Agropyron cristatum to increase both grain number and grain weight in wheat. Crop J 11:878–886
Liu L, Luo Q, Li H, Li B, Li Z, Zheng Q (2018) Physical mapping of the blue-grained gene from Thinopyrum ponticum chromosome 4Ag and development of blue-grain-related molecular markers and a FISH probe based on SLAF-seq technology. Theor Appl Genet 131:2359–2370
Ma P, Han G, Zheng Q, Liu S, Han F, Wang J, Luo Q, An D (2020) Development of novel wheat–rye chromosome 4R translocations and assignment of their powdery mildew resistance. Plant Dis 104:260–268
Mago R, Miah H, Lawrence GJ, Wellings CR, Spielmeyer W, Bariana HS, McIntosh RA, Pryor AJ, Ellis JG (2005) High-resolution mapping and mutation analysis separate the rust resistance genes Sr31, Lr26 and Yr9 on the short arm of rye chromosome 1. Theor Appl Genet 112:41–50
Mater Y, Baenziger S, Gill K, Graybosch R, Whitcher L, Baker C, Specht J, Dweikat I (2004) Linkage mapping of powdery mildew and greenbug resistance genes on recombinant 1RS from ‘Amigo’ and ‘Kavkaz’ wheat–rye translocations of chromosome 1RS.1AL. Genome 47:292–298
Miransari M, Smith D (2019) Sustainable wheat (Triticum aestivum L.) production in saline fields: a review. Crit Rev Biotechnol 39:999–1014
Quinlan AR, Hall IM (2010) BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26:841–842
Sakuma S, Golan G, Guo Z, Ogawa T, Tagiri A, Sugimoto K, Bernhardt N, Brassac J, Mascher M, Hensel G, Ohnishi S, Jinno H, Yamashita Y, Ayalon I, Peleg Z, Schnurbusch T, Komatsuda T (2019) Unleashing floret fertility in wheat through the mutation of a homeobox gene. Proc Natl Acad Sci 116:5182–5187
Schneider A, Rakszegi M, Molnár-Láng M, Szakács É (2016) Production and cytomolecular identification of new wheat-perennial rye (Secale cereanum) disomic addition lines with yellow rust resistance (6R) and increased arabinoxylan and protein content (1R, 4R, 6R). Theor Appl Genet 129:1045–1059
Song L, Jiang L, Han H, Gao A, Yang X, Li L, Liu W (2013) Efficient induction of Wheat-Agropyron cristatum 6P translocation lines and GISH detection. PLoS ONE 8:e69501
Song L, Lu Y, Zhang J, Pan C, Yang X, Li X, Liu W, Li L (2016a) Physical mapping of Agropyron cristatum chromosome 6P using deletion lines in common wheat background. Theor Appl Genet 129:1023–1034
Song L, Lu Y, Zhang J, Pan C, Yang X, Li X, Liu W, Li L (2016b) Cytological and molecular analysis of wheat–Agropyron cristatum translocation lines with 6P chromosome fragments conferring superior agronomic traits in common wheat. Genome 59:840–850
Tang Z, Yang Z, Fu S (2014) Oligonucleotides replacing the roles of repetitive sequences pAs1, pSc119.2, pTa-535, pTa71, CCS1, and pAWRC.1 for FISH analysis. J Appl Genet 55:313–318
Wu J, Yang X, Wang H, Li H, Li L, Li X, Liu W (2006) The introgression of chromosome 6P specifying for increased numbers of florets and kernels from Agropyron cristatum into wheat. Theor Appl Genet 114:13–20
Xiao J, Liu B, Yao Y, Guo Z, Jia H, Kong L, Zhang A, Ma W, Ni Z, Xu S, Lu F, Jiao Y, Yang W, Lin X, Sun S, Lu Z, Gao L, Zhao G, Cao S, Chen Q, Zhang K, Wang M, Wang M, Hu Z, Guo W, Li G, Ma X, Li J, Han F, Fu X, Ma Z, Wang D, Zhang X, Ling H-Q, Xia G, Tong Y, Liu Z, He Z, Jia J, Chong K (2022) Wheat genomic study for genetic improvement of traits in China. Sci China Life Sci 65:1718–1775
Xing L, Hu P, Liu J, Witek K, Zhou S, Xu J, Zhou W, Gao L, Huang Z, Zhang R, Wang X, Chen P, Wang H, Jones JDG, Karafiátová M, Vrána J, Bartoš J, Doležel J, Tian Y, Wu Y, Cao A (2018) Pm21 from Haynaldia villosa encodes a CC-NBS-LRR protein conferring powdery mildew resistance in Wheat. Mol Plant 11:874–878
Yang G, Boshoff WHP, Li H, Pretorius ZA, Luo Q, Li B, Li Z, Zheng Q (2021a) Chromosomal composition analysis and molecular marker development for the novel Ug99-resistant wheat–Thinopyrum ponticum translocation line WTT34. Theor Appl Genet 134:1587–1599
Yang G, Deng P, Ji W, Fu S, Li H, Li B, Li Z, Zheng Q (2023) Physical mapping of a new powdery mildew resistance locus from Thinopyrum ponticum chromosome 4AgS. Front Plant Sci 14:1131205
Yang G, Tong C, Li H, Li B, Li Z, Zheng Q (2022) Cytogenetic identification and molecular marker development of a novel wheat–Thinopyrum ponticum translocation line with powdery mildew resistance. Theor Appl Genet 135:2041–2057
Yang G, Zheng Q, Hu P, Li H, Luo Q, Li B, Li Z (2021b) Cytogenetic identification and molecular marker development for the novel stripe rust-resistant wheat–Thinopyrum intermedium translocation line WTT11. aBIOTECH 2:343–356
Ye J, Coulouris G, Zaretskaya I, Cutcutache I, Rozen S, Madden TL (2012) Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinform 13:134
Zhang X, Jia H, Li T, Wu J, Nagarajan R, Lei L, Powers C, Kan CC, Hua W, Liu Z, Chen C, Carver BF, Yan L (2022) TaCol-B5 modifies spike architecture and enhances grain yield in wheat. Science 376:180–183
Zhang Y, Zhang J, Huang L, Gao A, Zhang J, Yang X, Liu W, Li X, Li L (2015) A high-density genetic map for P genome of Agropyron Gaertn. Based on specific-locus amplified fragment sequencing (SLAF-seq). Planta 242:1335–1347
Zhang Z, Han H, Liu W, Song L, Zhang J, Zhou S, Yang X, Li X, Li L (2019) Deletion mapping and verification of an enhanced-grain number per spike locus from the 6PL chromosome arm of Agropyron cristatum in common wheat. Theor Appl Genet 132:2815–2827
Funding
This work was financially supported by the National Key R&D Program of China (2021YFD1200600).
Author information
Authors and Affiliations
Contributions
LHL conceived the research. YDL performed the research. YDL and SHZ wrote the paper. WJY modified some pictures. BH, XZL and YXZ participated in part of the data collection and cytology work. JPZ, HMH, BJG, XMY, XQL and WHL participated in the preparation of the reagents and materials used in this study.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Communicated by Peter Langridge.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, Y., Zhou, S., Yang, W. et al. Chromosomal mapping of a major genetic locus from Agropyron cristatum chromosome 6P that influences grain number and spikelet number in wheat. Theor Appl Genet 137, 82 (2024). https://doi.org/10.1007/s00122-024-04584-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00122-024-04584-2