Abstract
The monumental work of Olov Hedberg provided deep insights into the spectacular and fragmented tropical alpine flora of the African sky islands. Here we review recent molecular and niche modelling studies and re-examine Hedberg’s hypotheses and conclusions. Colonisation started when mountain uplift established the harsh diurnal climate with nightly frosts, accelerated throughout the last 5 Myr (Plio-Pleistocene), and resulted in a flora rich in local endemics. Recruitment was dominated by long-distance dispersals (LDDs) from seasonally cold, remote areas, mainly in Eurasia. Colonisation was only rarely followed by substantial diversification. Instead, most of the larger genera and even species colonised the afroalpine habitat multiple times independently. Conspicuous parallel evolution occurred among mountains, e.g., of gigantism in Lobelia and Dendrosenecio and dwarf shrubs in Alchemilla. Although the alpine habitat was ~ 8 times larger and the treeline was ~ 1000 m lower than today during the Last Glacial Maximum, genetic data suggest that the flora was shaped by strong intermountain isolation interrupted by rare LDDs rather than ecological connectivity. The new evidence points to a much younger and more dynamic island scenario than envisioned by Hedberg: the afroalpine flora is unsaturated and fragile, it was repeatedly disrupted by the Pleistocene climate oscillations, and it harbours taxonomic and genetic diversity that is unique but severely depauperated by frequent bottlenecks and cycles of colonisation, extinction, and recolonisation. The level of intrapopulation genetic variation is alarmingly low, and many afroalpine species may be vulnerable to extinction because of climate warming and increasing human impact.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The spectacular flora occurring on the tops of the tropical African sky islands has attracted the attention of generations of biogeographers because of its extreme fragmentation and iconic examples of gigantism and convergent evolution: the impressive plants of Lobelia and Dendrosenecio can rise up to 10 m above the otherwise low-growing alpine vegetation. These mountains provide an excellent example of a highly isolated system, where interaction between historical and contemporary processes has formed a flora rich in local endemics (Gehrke and Linder 2014; Gizaw et al. 2016a). The uppermost, afroalpine habitat is home to no more than ~ 520 species of vascular plants that are able to endure a peculiar and harsh climate: while the days can be very warm, the nights are often freezing cold, all through the year (Hedberg 1961; Gehrke and Linder 2014). This striking diurnal cycle has a great impact on the local biota, and stands in stark contrast to the seasonal climates at higher latitudes and to the warm climates of the adjacent tropical lowlands. Most species of the afroalpine flora are endemic to Africa (78%; Hedberg 1961), but almost as many (67%) are exclusive to these mountains (Gehrke and Linder 2014). Many of them also extend below the treeline into the species-rich afromontane region, one of the 36 global biodiversity hotspots (Mittermeier et al. 2011; CEPF 2016), and together, the afroalpine and afromontane floras can be referred to as the afrotemperate flora (White 1983; see Carbutt and Edwards 2015 for concept discussion and delimitation).
Most of the mountains that are high enough to support an afroalpine flora are found in Ethiopia and East Africa (here referred to as eastern Africa), but a few small patches with a depauperate afroalpine flora are also found in West Africa (Fig. 1; Gehrke and Linder 2014). In eastern Africa, these high mountains occur along one of the most prominent rift systems on earth, the East African Rift System (EARS), which is more than 3000 km long and transects the Ethiopian and Kenyan plateaus (Ring et al. 2018). It is divided into one older, volcanically active Eastern Branch and a younger, less volcanic Western Branch (Ring et al. 2018). The onset of mountain uplift is poorly dated, but it may have begun 45 Ma (Ebinger et al. 2000; Trauth et al. 2005; Ring et al. 2018). The associated volcanism resulted in the formation of the extensive mountain ranges in the Ethiopian highlands, where huge Miocene shield volcanoes are superimposed on much older flood basalts, and the scattered high mountains in East Africa, with origins ranging from the Miocene to the late Pliocene (Griffiths 1993). Most of the mountains appear to have been uplifted to sufficient elevation for developing alpine vegetation as late as the Pliocene or even Pleistocene, i.e., mainly during the last 5 Myr (reviewed in Gehrke and Linder 2014).
Major mountains and mountain ranges that are sufficiently high to support an alpine habitat in eastern Africa under the current climate (the Eastern Arc Mts are also indicated although these do not harbour alpine vegetation). Fault lines of the Great Rift Valley are shown as stippled lines. The small West African afroalpine habitat patches are indicated with the white circle on the small map in the left top corner
The exploration of the afroalpine flora started in the early 1800s (see, e.g., Richard 1847–1851; the term afroalpine was used for the first time by Hauman 1933). Many mountains were still unexplored when Engler (1892) published an early account of the East African mountain flora. Since then, many expeditions resulted in new knowledge of certain plant groups and individual mountain floras, such as the series of papers by Fries and Fries (e.g., 1922a, b, 1923, 1948). In 1948, the Swedish botanist Olov Hedberg participated in an East Africa Expedition and made numerous new collections from the afroalpine flora. In the following years, he studied all available herbarium material and published his monumental book ‘Afroalpine Vascular Plants. A Taxonomic Revision’ in 1957 (Hedberg 1957), the first complete account since that of Engler (1892). This book provided a state-of-the-art taxonomy of the entire afroalpine flora in East Africa (but did not include Ethiopia or Cameroon), as well as a comprehensive appendix with detailed discussions and morphological measurements. Building on this solid taxonomic foundation, Olov Hedberg, partly together with his wife Inga Hedberg, continued to explore these mountains and published several insightful papers on the origin, evolution, taxonomy, ecology and biogeography of the afroalpine flora in the following decades. They also carried out or initiated taxonomic revisions of the entire Ethiopian flora (Hedberg 1983), leading to the publication of the Flora of Ethiopia and Eritrea in 1980–2009 (Demissew et al. 2011; Hedberg and Persson 2011). The Hedbergs also performed extensive cytological investigations, documenting the impact of polyploid evolution in the afroalpine flora (49% polyploidy; Hedberg and Hedberg 1977; vs 33% globally; Rice et al. 2019; and > 80% in polar desert; Brochmann et al. 2004).
Hedberg (1961, 1965, 1969, 1970, 1986, 1997) emphasised the importance of the harsh climate and recurring frosts in defining the afroalpine flora. He postulated that whereas some species, such as the giant senecios (Fig. 2) and giant lobelias, were differentiated from less specialized forms found at lower elevations, a large proportion of the flora instead has strong phytogeographic affinities to cold-adapted lineages in remote temperate areas of the world. Hedberg (1961) stated that ‘[the afroalpine flora] seems to have existed in tropical East Africa before the formation of the mountains now harbouring it, and to have been strongly isolated from other high-mountain (or temperate) floras for a long time’. In his taxonomic work, he stressed that earlier authors typically had exaggerated the level of intermountain allopatric (‘vicarious’) speciation, reducing many described single-mountain endemics into synonymy (e.g., Hedberg 1955, 1957). He hypothesized the existence of habitat bridges between certain mountains under past climates, at least for plants that are able to grow in the montane zone. However, he thought that the mountain top populations must have remained isolated from each other even during glacial periods, as supported by his finding that the proportion of endemic species increased with their ascending lower altitude limit. He, therefore, emphasised the importance of long-distance dispersal (LDD), in particular by cyclones. He noted that direct habitat connection among mountains is inconsistent with the striking differences in species composition of ecologically similar plant communities in different mountains, such as the Dendrosenecio forests and Alchemilla shrublands, suggesting that the communities were synthesized on each mountain separately after independent LDD of the constituent species.

Adopted from Tusiime et al. (2020)
The afroalpine flagship genus Dendrosenecio shows conspicuous and intricate variation in growth form and morphology, resulting from parallel altitude/habitat adaptation on different mountains combined with occasional long-distance dispersal and hybridization (Tusiime et al. 2020; Gizaw et al. in press; photos: Abel Gizaw). a–d: Growth form differentiation in high-altitude habitats on Mt Kenya. Dendrosenecio keniensis a, b is a low-growing (< 1.5 m), procumbent plant that branches close to the ground and occurs on constantly water-saturated soils, whereas D. keniodendron c, d is an erect giant with tall stems (up to 7 m) that branch high above the ground and occurs on well-drained soils. e–h: Plants at lower altitudes tend to have thin stems with only few old leaves kept for insulation, exemplified by plants of D. battiscombei on Mt Kenya (e, f) and D. erici–rosenii on Mt Ruwenzori (g, h). i–o Plants at higher altitudes tend to have thick stems with a prominent layer of insulating old leaves, exemplified by plants of D. adnivalis in the Ruwenzori Mts (i, j), D. kilimanjari on Mt Kilimanjaro (k, l) and D. elgonensis on Mt Elgon (m–o).
In this review, we revisit Hedberg’s views on the history and evolution of the afroalpine flora and review recent work using molecular and niche modelling tools that were unavailable to him. We concentrate on the afroalpine flora, but also touch upon plant groups and themes representing the wider afrotemperate flora. We put particular emphasis on dated phylogenies and phylogeographic results based on range-wide sampling, and place these in the context of the geological and climatic history of the region. The new insights and large body of evidence accumulated since Hedberg’s era point to an emerging scenario of a young, unsaturated, fragile and vulnerable island flora, with unique taxonomic and genetic diversity that seems to be severely depauperated by frequent bottlenecks and cycles of colonisation, local extinction, and recolonisation.
Plio-Pleistocene LDDs prevail in colonisation of the emerging sky islands
Botanists from temperate regions often feel somewhat lost in tropical forests, but more happily at home if they take on the laborious task of climbing beyond the treeline. Hedberg (1961, 1965, 1986) analysed the phytogeographic connections of the afroalpine flora and estimated that an astonishing proportion of the species (48%) belong to temperate elements, 10% to a South African element, and 3% to a Himalayan element (data from Hedberg 1986). He assigned only one third of the afroalpine species to an endemic afromontane element, among them the giant senecios and lobelias, which he thought originated from montane forest ancestors. Because of the high level of endemism, Hedberg (e.g., 1961, 1986) believed that the afroalpine flora was old, and that even the afroalpine populations of widespread species such as Arabis alpina had been isolated from their northern counterparts for at least some millions of years.
A series of studies have now tested Hedberg’s hypotheses on the origin of the flora using molecular phylogenetic and phylogeographic tools to infer the ancestral lineages and ages of afroalpine species. The general patterns that begin to emerge might partly have surprised but certainly also excited Hedberg. Many afromontane clades of plants and animals are indeed old and many of them have their closest relatives in tropical lowland forests, with long-term climatic stability being a major factor generating and maintaining species diversity (e.g., Fjeldså and Lovett 1997; Lovett et al. 1988; Couvreur et al. 2008; Voje et al. 2009; Tolley et al. 2011; Dimitrov et al. 2012; Loader et al. 2014; Mairal et al. 2015). Afromontane clades, therefore, typically demonstrate niche convergence in their adaptation to higher elevations, consistent with the Out of the Tropical Lowlands (OTL) hypothesis (Qian and Ricklefs 2016; cf. also Merckx et al. 2015). Afroalpine species and clades, in contrast, tend to be much younger (Plio-Pleistocene) than believed by Hedberg, but in accordance with his view many of them originated after LDD from various remote temperate regions (cf. Linder 2014 and references below). Thus, many afroalpine lineages demonstrate niche conservatism in the sense that they were already adapted to cold climates, but they demonstrate niche convergence in the sense that their establishment in the afroalpine region required an adaptive shift from seasonal to diurnal cold climates (Nürk et al. 2018). It is possible, therefore, that many of the afroalpine lineages that arrived from temperate regions first established at lower, montane elevations (where many of them still occur today) before entering the true alpine zone with its nightly frosts (Gehrke 2018). Compared to temperate alpine floras, tropical alpine floras show stronger phylogenetic clustering relative to their surrounding floras; that is, on average, tropical alpine species are more closely related to each other than are species at lower elevations (Qian et al. 2021). This is probably caused by strong environmental filtering in combination with phylogenetic niche conservatism as suggested by Qian et al. (2021), but may also reflect that a high proportion of the species in tropical alpine floras are derived from remote rather than local species pools.
Quite a few of the afroalpine species that Hedberg (1986) attributed to the endemic afromontane element seem rather to have originated after recent dispersal from temperate regions (e.g., all Alchemilla spp., Gehrke et al. 2016; some Carex, Box 1, Gehrke 2011; Gizaw et al. 2016b). Helichrysum, on the other hand, one of the most species-rich genera in the afroalpine flora, colonised the tropical alpine zone at least five times independently from local ancestors at lower elevations (Galbany-Casals et al. 2014). This genus originated in Southern Africa and spread via the Drakensberg Mountains into the montane and alpine regions of eastern Africa. In Lobelia, the tropical alpine zone was occupied independently by montane ancestors in the Western and Eastern Rifts (Knox and Palmer 1998). The giant lobelias arrived in East Africa from eastern Asia in the Miocene, and spread from there across the Congo basin in the early Pliocene to West Africa, across the deserts of northern Kenya at the Pliocene–Pleistocene transition to Ethiopia, and even to South America (Brazil; Knox and Li 2017; Perez and Yu 2021). The lineage radiated throughout the Pliocene to the late Pleistocene (Knox and Li 2017). In Dendrosenecio, the situation is not yet fully clarified, but a recent study based on high-throughput sequencing (Hyb-Seq) suggests that the lineage diversified from the Late Miocene and well into the Pleistocene (Gizaw et al. in press), and the current species diversity seems to result from parallel altitude/habitat adaptation in four mountain groups (Tusiime et al. 2020). Knox and Palmer (1995) suggested, contrary to Hedberg (1986) and others that the giant senecios originated at high altitudes on Mt Kilimanjaro. This is, however, highly unlikely given the young (Pleistocene) age of Mt Kilimanjaro and the new dating results of Gizaw et al. (in press). The genus Hypericum seems to have a complex history in Africa. It most likely originated there and dispersed to Eurasia and elsewhere, preferring temperate and often mountain habitats. It re-established in Africa multiple times independently in lower montane areas, from where it colonised the afroalpine habitat at least twice (Nürk et al. 2015).
Many studies have now documented temperate origins of afroalpine species. The widespread Arabis alpina colonized the afroalpine region twice during the middle and late Pleistocene, in both cases from the Balkans/Middle East (Box 2, Koch et al. 2006; Assefa et al. 2007). Western Eurasian origins have been inferred for Anthoxanthum (Box 3; late Pliocene; Pimentel et al. 2013), Lychnis (Box 4; late Miocene–early Pliocene; Popp et al. 2008; Gizaw et al. 2016a), and most likely Alchemilla (probably Pleistocene; Gehrke et al. 2016). The eastern African Swertia species belong to two different clades, both originating from the Himalayas (Chassot et al. 2001). Eurasian origins have also been inferred for some Carex and Ranunculus (Gehrke and Linder 2009). Southern African mountain origins have been inferred for most afroalpine lineages of Senecio (Box 5, Kandziora et al. 2016), and, astonishingly, the afroalpine fine-leaved Festuca clade was inferred to have originated from northern South America (Box 6; Messinian–Pliocene; Minaya et al. 2017).
The model plant Arabidopsis thaliana has an intriguing history. Hedberg considered this species to be indigenous to the afroalpine flora in his 1957 book (Hedberg 1957), and assigned it to the northern temperate element (Hedberg 1986). This was long disregarded by the scientific community, who assumed that this otherwise intensively studied plant is native only to western Eurasia. Arabidopsis thaliana diverged from its primarily western Eurasian sister clade in the early Pliocene according to Hohmann et al. (2015). It was surprising, therefore, when Durvasula et al. (2017) and Fulgione and Hancock (2018) found that the African populations are older and more diverse than the Eurasian ones. They inferred that the species originated in Africa, possibly somewhere in northern Africa, in the middle Pleistocene (1.2–0.8 Ma) associated with a transition from outcrossing to selfing. Colonisation of the eastern African mountains occurred during the last glaciation (90 ka), and the current lineages in Eurasia arrived from Africa as recently as 45 ka.
Some afroalpine lineages have been inferred to have connections both to western Eurasia and to southern Africa. Eastern African Erica are related to the western European and Mediterranean species as well as to those in the Drakensberg Mountains in southern Africa (Box 7, Pirie et al. 2019). In the Late Pliocene, sub-Saharan Africa was colonized by two divergent lineages of Anthoxanthum, one from southeast Asia to southern Africa, and one from western Eurasia to eastern Africa (Pimentel et al. 2013). Intriguingly, these currently allopatric lineages were inferred to have met in East Africa during the Pleistocene and produced an allopolyploid (Box 3, Tusiime et al. 2017).

Adopted from Chala et al. (2017)
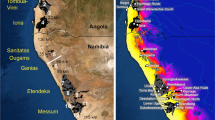
Extent of the afroalpine habitat in eastern Africa under a current and b LGM (Last Glacial Maximum, ~ 21 ka) climate; alpine habitat is indicated in black. c Spatial extent of the alpine habitat in different regions: AA—Yemen (araboalpine), EA—East Africa (Kenya, Tanzania and Uganda), ETH—Ethiopia. d Extent of alpine habitat at different elevations; the y-axis represents 1 km × 1 km grid cell count.
Ongoing work based on high-throughput sequencing of ~ 100 afroalpine species points to continuous and in fact accelerating establishment of lineages arriving from temperate areas through the last 5 Myr (Kandziora, Gehrke, Popp, Gizaw, Brochmann and Pirie, unpubl.). Thus, the eastern African sky islands can be viewed as a strongly isolated archipelago that is still young and experiencing repeated disturbance. It is characterized by open and unsaturated environments that continue to diversify topographically and ecologically and that increasingly recruit more cold-adapted immigrants arriving with the occasional ‘seed rain’ from temperate areas. The Pleistocene climate cycles probably induced high community turnover and extinction rates, opening up niches for colonisation by new lineages. The general dynamic model of oceanic island biogeography predicts that immigration increases during the first part of an island’s life (Borregaard et al. 2016; Whittaker et al. 2017), and this might apply to volcanic continental islands as well (see also Vidal and Clark 2019; Flantua et al. 2020). The glacial periods potentially had even more dramatic and disturbing impact on continental islands than on typical oceanic islands, where the climate is thermally stabilized by the ocean (Cronk 1997) although heavily influenced by sea level changes (Fernández-Palacios et al. 2016). In the sky islands, the propagule pool arriving from temperate regions has probably been at its maximum during the glacials, because these lasted much longer than the interglacials, because of reduced distances to source regions, and because of considerably larger size of the target area (Fig. 3; the afroalpine habitat was estimated to be eight times larger during the LGM; Chala et al. 2017; see also van der Hammen and Cleef 1986 for similar inference for the Andean Páramo flora). Successful establishment after immigration may also have been more likely during glacial periods because of increased availability of empty niches following extinctions, altitudinal shifts, and accompanying disruption of entire communities such as bogs, which need thousands of years to re-establish (cf. van der Hammen and Cleef 1986; Smith 1986). The climate oscillations in eastern Africa were, however, particularly complex because of a lack of synchrony between changes in temperature and rainfall. Major drought periods may have caused massive extinctions on mountains harbouring a flora adapted to constantly humid conditions, such as present-day Ruwenzori.

Reproduced from Gizaw et al. (2016b)
AFLP-based phylogeographic analysis of the afroalpine endemics a Carex runssoroensis (29 plants from 7 populations; white symbols) and b C. monostachya (84 plants from 19 populations; black and grey symbols). c Geographic distribution of the three main genetic groups inferred from Bayesian clustering. d Graphical representation of the assignment pattern. e Principal Coordinates Analysis (PCoA) based on Dice’s coefficient of similarity between genotypes. Photos: a Abel Gizaw (spike) and Berit Gehrke (tussock); b Berit Gehrke. c–e:
Frequent immigration, limited diversification
The level of endemism at the species level is high in the afroalpine flora: 67% of the 520 native species that regularly occur above 3200 m are endemic to these sky islands (Gehrke and Linder 2014). Many of the endemics are widespread (Hedberg 1957); according to the updated species list of Gehrke and Linder (2014), only 15% are endemic to a single mountain (or a group of closely adjacent mountains). In many cases, the evolution of endemic species was probably linked to adaptive shifts from seasonally cold climates (or, more rarely, from the milder climate at lower elevations) to a strongly diurnal climate with recurrent night frosts (Gehrke 2018). There are only four endemic genera (Dendrosenecio, Haplosciadium, Hedbergia (cf. Scheunert et al. 2012), and Oreophyton; the former endemic genus Uebelinia is embedded in Lychnis; Popp et al. 2008).
The high level of species endemism is, however, not associated with substantial diversification of lineages establishing in the afroalpine region. There is much less diversification than implied by the early explorers who tended to over-describe new taxa (cf. Hedberg 1957), and much less than might be inferred solely from the number of species per genus. More than half of the 191 genera in the afroalpine flora are represented by a single species, and just 14% of them are represented by more than five species (Gehrke and Linder 2014). Phylogenetic studies show that many congeneric species result from independent migrations into the afroalpine region rather than diversification. Multiple immigrations have been inferred for Carex (at least 13 times; Gehrke and Linder 2009, 2011), Ranunculus (4 times; Gehrke and Linder 2009, 2011; Hörandl and Emadzade 2011), Senecio (4–14 times; Kandziora et al. 2016), Helichrysum (at least 5 times; Galbany-Casals et al. 2014), Cardamine (4 times; Carlsen et al. 2009), Hypericum (at least 2 times; Nürk et al. 2015), Swertia (2 times; Chassot et al. 2001), and Anthoxanthum (2 times; Tusiime et al. 2017).
Notable exceptions are Alchemilla, the giant Lobelia lineage, Dendrosenecio, and Senecio. In Alchemilla, a single immigration resulted in 17 afroalpine species, most of them with restricted distributions; these species probably evolved by allopatric speciation in different mountains or mountain groups after initial adaptation to the diurnal climate (Gehrke et al. 2016). The giant Lobelia lineage in eastern Africa diversified into 21 species to occupy a range of habitats in montane forests and the afroalpine zone (Knox and Palmer 1998), representing a combination of adaptive radiation and intermountain allopatric speciation. In Dendrosenecio, parallel altitudinal adaptation in different mountain groups, in a few cases combined with adaptation to different habitats within individual mountains, seem to have resulted in 5–10 species (Tusiime et al. 2020; Gizaw et al., in press; note, however, that Knox 2005 and in particular Hedberg 1957, 1969 accepted more species). In Senecio, the most species-rich genus in the afroalpine flora, one of the independent immigrations resulted in seven species (Kandziora et al. 2016). In most cases, diversification was more limited and typically resulted in only two or a few allopatric species (e.g., Carex monostachya and C. runssoroensis; Gizaw et al. 2016b; Fig. 4; Box 1).
Realized and potential distribution of one exclusively afroalpine species (Carex monostachya) and one non-exclusively afroalpine species (Carduus schimperi) and geographic structuring of their genetic (AFLP) diversity. The first column shows the modelled potential distribution under the current climate and the occurrence points used in the models, reflecting realized current distribution (most of the occurrence points are based on own field work and herbarium material verified by us (green and blue); a few added from GBIF (black)). The second column shows the modelled distribution under the Last Glacial Maximum (LGM) climate. The third column shows genetic structuring (genetic groups inferred from Bayesian analyses) mapped on the LGM distribution. Map colors indicate habitat suitability based on agreement among pixels from three binary maps constructed using different algorithms (0.0–0.3—yellow, habitat unsuitable with high certainty; 0.3–0.6—orange, habitat suitability uncertain; > 0.6—red, habitat suitable with high certainty). In both species, the southeastern Ethiopian populations belong to the same genetic group as East African populations, in spite of lack of habitat connectivity. In C. schimperi, putative ecological connectivity across the Rift Valley both in Ethiopia and East Africa during the LGM did not result in genetic connectivity. Genetic data: Gizaw et al. 2016b and Wondimu et al. 2014; modelling: Chala, unpublished data)
In addition to limited time since colonisation, it is possible that the level of intermountain gene flow is sufficiently high to prevent allopatric divergence into distinct species, rendering the afroalpine flora poor in single-mountain endemics. Phylogeographic studies show that although many afroalpine species are represented by distinct genetic groups in different mountains, there is often only subtle morphological differentiation between them or none at all, and there is increasing evidence for intermountain LDD and hybridisation (see below). This might be surprising in an island system with such strong geographic isolation, especially among the distant peaks in East Africa. Limited adaptive radiation within individual mountains is, however, expected given the limited size of their afroalpine habitat. This is probably the main reason why the tropical alpine Andean Páramo with its much larger and more connected areas of alpine habitat shows vastly greater species richness and many lineages showing extreme and rapid radiation (Madriñán et al. 2013; Sklenář et al. 2014; Hughes and Atchison 2015; Kolář et al. 2016a, b; Lagomarsino et al. 2016). Mountain ruggedness combined with degree and frequency of past connectivity are central concepts in the ‘flickering connectivity system (FCS)’ proposed by Flantua and Hooghiemstra (2018) as a major driver of species richness and endemism in mountains. The combination of extreme fragmentation and small extent of the alpine patches has probably resulted in much less expression of the FCS system in eastern Africa than in the Andes.
Parallel adaptation: giants, dwarfs and cushions
Hedberg was fascinated by the classical examples of convergent (or parallel) evolution provided by the enigmatic giant rosette plants of tropical alpine environments, such as the giant Lobelia and giant Dendrosenecio in Africa and Puya and Espeletia in South America. These lineages are distantly related but have independently developed strikingly similar morphological, physiological and life history traits in response to the tropical alpine climate (Hedberg 1964, 1969; Nagy and Grabherr 2009; Givnish 2010). They have large leaf rosettes which often fold up during the night to protect the buds, and they retain old leaves for insulation, accumulate large amounts of water to counteract temperature shocks, and grow taller with increasing altitude to escape the low temperatures close to the ground (Hedberg 1964; Smith and Young 1987; Fetene et al. 1998).
There is now convincing molecular evidence for parallel evolution also among mountains in eastern Africa, not only of gigantism in Lobelia (Knox and Palmer 1998; Knox and Li 2017) and Dendrosenecio (Knox and Palmer 1995; Tusiime et al. 2020; Gizaw et al. in press) but also of dwarf shrubs and perennial cushions, life forms typical of harsh arctic and alpine environments. After the single dispersal of Alchemilla to the eastern African mountains, herbaceous ancestors developed secondary woodiness independently in Ethiopia and East Africa (Gehrke et al. 2016). The widespread perennial, cushion-formed high-altitude plants described as Swertia lugardae evolved independently in five Ethiopian mountains from annual, erect low-altitude plants described as S. abyssinica; their genotypes grouped according to mountain, not to the described species (Bekele 2006).
Recent, parallel adaptive evolution (Pleistocene, and even Holocene) has now been convincingly documented in a number of animal and plant groups in other parts of the world, in particular along steep environmental clines which drive repeated and rapid evolutionary diversification, for example along altitudinal gradients in mountains (e.g., Kolář et al. 2016a, b; Bertel et al. 2018; Konečná et al. 2019) and development of secondary woodiness on continental and oceanic islands (Nürk et al. 2019). Recent genomic studies show that there are several molecular mechanisms that can facilitate adaptation to extreme cold and other abiotic stresses, and that many genes can be involved (Birkeland et al. 2020; Nowak et al. 2020). Three plant lineages of the Brassicaceae, which colonized the Arctic independently, have evolved similar suites of adaptations by modifying different components of similar stress response pathways, implying that there are many genetic trajectories for adaptation to extremely cold environments (Birkeland et al. 2020). In Dendrosenecio, high-altitude plants typically have larger stems and leaves than low-altitude plants, and show stronger physiological adaptations ensuring frost tolerance and avoidance (Hedberg 1964; Mabberley 1986; Beck 1986). Whereas high-alpine and low-alpine plants on the same mountain tend to be conspicuously different in morphology, they are genetically more similar to each other than they are to plants on other mountains (Knox and Palmer 1995; Tusiime et al. 2020; Gizaw et al. in press). Many of the numerous taxa of Dendrosenecio described over the years are only poorly differentiated genetically, and the genetic variation within mountains and mountain groups is often more or less continuous, consistent with recent ecoclinal or ecotypic differentiation in the presence of gene flow (Tusiime et al. 2020). It seems that only in a few cases, ecotypes may be recognised as morphologically distinct subspecies, and only on one or possibly two mountains as distinct species.
The time is obviously ripe to re-address the timing, mechanisms and patterns of parallel adaptation in the afroalpine flora using genomic approaches. Recent genomic studies of the Andean lineages Espeletia and Lupinus show that diversification often has occurred exceptionally fast via ecological speciation during the last glacial cycles of the Pleistocene, and that small‐scale ecological divergence may have occurred along multiple niche dimensions (Nevado et al. 2018, 2019; Pouchon et al. 2020).
Migration corridors among the sky islands: do they, or did they exist?
As noted above, many members of the afroalpine flora are widespread, occurring on more than one mountain or mountain group, in spite of the extreme fragmentation of this system. How, and how often, did or do they spread among the mountains? Based on topographic, floristic and ecological analyses, Hedberg (1969) predicted that the montane forest expanded to form corridors among some adjacent mountains during the warm and humid interglacials. Studies of several montane forest plants and animals have provided evidence consistent with this prediction, some of them even for montane forest corridors between East and West Africa (e.g., Kadu et al. 2011, 2013; Mihretie et al. 2015; Mairal et al. 2015, 2017; Chartier et al. 2016; Gichira et al. 2017; Migliore et al. 2020), and in some cases found that such forest bridges disappeared only recently due to anthropogenic impact (e.g., Kebede et al. 2007; Hemp and Hemp 2018). Hedberg (e.g., 1970) did not believe, however, that past climates had enabled high alpine species to migrate gradually among mountains, and assumed that their ranges must have been established solely based on LDD. He found that whereas adaptation to endozoochorous dispersal with birds was virtually non-existent in the afroalpine flora, one third of the species were adapted to wind-dispersal and two thirds to ectozoochorous dispersal, but believed that cyclones in any case might be crucial for their long-distance dispersal.
Paleoecological, niche modelling, and phylogeographic studies have provided new insights into these questions raised repeatedly by Hedberg. Geological and pollen analytical work has shown that glaciers were present on the highest peaks during the LGM (Coetzee 1964; Hamilton 1982; Messerli and Winiger 1992; Umer et al. 2007). The glacial climates led to a considerable downward shift of afroalpine plant communities, and they oscillated along the elevational gradient in response to the frequent Pleistocene cycles. The paleoecological evidence suggests that the extent of the alpine habitat increased during the glacials, but not enough to create direct connections between the main mountains (Sepulchre et al. 2006; Bonnefille 2010; Hendrickx et al. 2014).
Using niche modelling, Chala et al. (2017) addressed whether suitable habitats shifted far enough downslope under past colder climates to form bridges enabling gradual migration, taking into account that only one third of the species in the afroalpine flora are exclusively alpine, and that the other species extend to varying degrees into grasslands and open forests at lower elevations. They mapped the extent of the current alpine habitat and projected it to the LGM climate to assess whether gradual migration was possible for exclusively alpine taxa during this glacial period, and thus potentially also during earlier glaciations. The LGM treeline was estimated to be about 1000 m lower, which is consistent with estimates from pollen records (Coetzee 1964; Maitima 1991; Zech 2006; Schüler et al. 2012), and the alpine habitat to be eight times larger than today (Fig. 3). At the LGM, most of the alpine habitat of the Ethiopian highlands was interconnected except across the Rift Valley, whereas the solitary mountains of East Africa remained isolated for exclusively alpine species. Chala et al. (2017) also modelled land cover under current and LGM climates to assess whether grassland and open forests could have served as migration corridors for alpine taxa that today extend into lower vegetation belts. Notably, they found that for drought-tolerant species, gradual migration may have been possible during the dry glacial periods, and possibly also under the current climate before agriculture transformed the low-lying landscapes.
In contrast, molecular data, partly in combination with niche modelling for individual species, do not provide support for gradual migration of any afroalpine species among mountains (although the possibility for gradual migration among closely adjacent mountains has not yet been analysed in detail). The molecular studies rather identify many LDD episodes across vast lowland barriers. This implies that although some potential migration corridors may have existed (Chala et al. 2017), they do not seem to have resulted in effective intermountain gene flow. It turns out that geographic distance is often a poor predictor of genetic distance in the afroalpine flora. In accordance with Hedberg’s (1970) view, morphological adaptations to bird and wind dispersal may play a subordinate role in LDD relative to truly unpredictable 'chance dispersal' with non-standard vectors (Berg 1983; Alsos et al. 2007, 2015; Nathan et al. 2008). Wondimu et al. (2014) used AFLPs to infer the history of two species with different dispersal adaptations, but similar geographic ranges and habitats: the wind-dispersed Carduus schimperi and the supposedly short-distance-dispersed Trifolium cryptopodium. In both species, populations from opposite sides of the narrow Rift Valley barrier in Ethiopia were genetically more distant than those from opposite sides of the vast Ethiopia–East Africa barrier. The surprising pattern of high genetic similarity between populations from the Bale Mts in Ethiopia and the distant East African mountains has also been identified in several other species (Ehrich et al. 2007; Gizaw et al. 2013, 2016b; Masao et al. 2013; Chen et al. 2015; Wondimu et al. 2017). It is still a mystery why the vast Ethiopia–East Africa barrier seems to be more permeable—at least for some species—than the narrow Rift Valley barrier within Ethiopia, which is found to be a major biogeographic barrier for both animals (reviewed in Komarova et al. 2021) and plants.
Based on dated phylogenies, extreme Pleistocene LDD episodes have been inferred between the afroalpine habitats on the opposite sides of tropical Africa for Lychnis abyssinica (< 300 ka; Gizaw et al. 2016a) and Festuca abyssinica (two LDDs, 860 ka and 520 ka; Mairal et al. 2021). For Festuca, three small patches of suitable LGM habitat were identified between East and West Africa, potentially acting as stepping stones. In Carex, allopatric speciation resulting in one eastern (C. monostachya) and one western species (C. runssoroensis) in East Africa was followed by LDD of the western species across the vast Ugandan lowland gap, resulting in secondary contact and admixture (Gizaw et al. 2016b).
Geographic distance among mountain groups appears to be a better predictor of floristic distance than intraspecific genetic distance. Gehrke and Linder (2014) analysed similarities among the mountain floras and identified four geographic groups: West Africa, Ethiopia/Sudan (with eastern and western subgroups, separated by the Rift Valley), western East Africa and eastern East Africa. They showed that this pattern is better explained by geographic isolation than by environmental filtering. They further showed (Gehrke and Linder 2011, 2014) that species richness—as in oceanic islands—can be explained by a model including age, area size, elevation and isolation, whereas levels of endemism are best explained by species richness in combination with area and isolation.
In ongoing work, we address the relative roles of LDD and past habitat connectivity based on genotypes of 28 afroalpine species (a subset of the species in Table 1, i.e., those occurring on at least two mountains) and modelling individual species ranges during the LGM and for 1 kyr time slice intervals until today (Gizaw, Chala, Popp, Zimmermann, Brochmann et al., unpublished data). Most species show distinct, but highly variable genetic structuring among mountains, reflected by large variation in median pairwise population differentiation both among (Φst 0.24–0.47) and within species (Φst 0.03–0.78). Many of the species had considerably larger potential ranges during colder climates (e.g., the non-exclusively alpine Carduus schimperi; Fig. 5). However, the correlation between ecological and genetic distances was virtually constant through time in all species and lower than that between geographic and genetic distances, suggesting that LDD has always been the main driver shaping genetic structure. Geographic distance and species characteristics such as dispersal adaptations only explained part of the variation, suggesting a major role of stochasticity associated with LDDs. Genetic and floristic distances were, however, correlated, suggesting that initial colonisation of mountains and subsequent intermountain gene flow are both driven by similar LDD events. Additional evidence for high levels of stochasticity in community assembly in the afroalpine flora is provided by our ongoing work assessing genetic diversity of 38 species (Table 1) versus total species diversity in 100 m × 100 m plots analysed in five major habitat types (rock outcrop, bog, Alchemilla shrubland, grassland, and Dendrosenecio woodland; Dimitrov, Gizaw, Chala, Brochmann et al., unpublished data). Whereas the meta-analysis of Vellend et al. (2014) found that correlations between species diversity and genetic diversity are generally positive (but see, e.g., Taberlet et al. 2012), we find no correlation in these afroalpine habitats, probably reflecting LDD stochasticity combined with frequent habitat disturbance caused by the Pleistocene climate oscillations.

Adopted from Gizaw et al. (2016a). Photo: Lychnis abyssinica by Abel Gizaw
Single-locus tree for Lychnis inferred from sequences of a combined rps16/psbE-petL pDNA data matrix. Node bars represent 95% highest posterior density (HPD) age intervals. Time scale in million years. The star denotes the afromontane/afroalpine clade. Lychnis abyssinica LGS and L. abyssinica SGS denote large-genome-size and small-genome-size variants, respectively. Blue fonts depict the West African accessions.
Afroalpine plant populations are genetically depauperate and prone to extinction
It is of considerable conservation concern that most afroalpine plant species studied so far harbour very little intrapopulation genetic variation, most likely because of repeated bottlenecks induced by frequent climatic and vegetational shifts and long-distance colonisation by few propagules. Table 1 presents average within-population Nei’s gene diversity (D) based on published and unpublished AFLP data sets for 3756 plants of 38 afroalpine species. Average diversity over all 38 species is 0.087, ranging from 0.042 to 0.204 for individual species. Thus, afroalpine plant species tend to contain much less within-population genetic diversity than reported for plant species occurring elsewhere by Nybom (2004), who found averages ranging from 0.27 for wind-dispersed and outcrossing species to 0.12 for selfing species. In our set of species, both wind-dispersed and supposedly short-distance-dispersed species have low diversity (0.068–0.099). Sunbird- and wind-pollinated species, which are likely to be outcrossing, have slightly higher genetic diversity (0.105–0.116) than species with insect-pollination syndromes (0.077), of which some are likely to be primarily selfing. Extremely little within-population genetic diversity have also been reported for some high alpine species in the Andes, attributed to genetic drift caused by founder effects, lack of gene flow, and/or autogamy (Vásquez et al. 2016; Sgorbati et al. 2004).
These data imply that afroalpine plants may be extremely vulnerable and prone to extinction, in particular because of the compounding effects of ongoing habitat destruction (e.g., overgrazing, human-induced fires) and climate change. The plants inhabit a narrow band of suitable conditions that already is limited to the upper extremes of the mountains. Chala et al. (2016) modelled the habitat suitability of the Ethiopian endemic Lobelia rhynchopetalum and assessed how the range and genetic diversity of this enigmatic giant is affected under two climate models and four emission scenarios. They found that only 3.4% of its habitat may remain suitable by 2080, resulting in loss of 82% of its genetic diversity. Thus, specialized high-alpine giant rosette plants are likely to face very high risk of extinction following the ongoing climate change. These results also suggest that historical climate oscillations probably had dramatic effects on genetically depauperate afroalpine species and likely caused extinctions. There are similar concerns for afroalpine animals. The impressive Walia ibex (Capra walie) is restricted to a single population in the Simen Mountains in Ethiopia, harbours the lowest genetic diversity ever recorded in a mammal, and competes with domestic goats for food plants, as shown by DNA metabarcoding of faeces using a DNA reference library constructed for the afroalpine flora (Gebremedhin et al. 2009, 2016). The peculiar and endangered Ethiopian wolf (Canis simensis) shows very little mtDNA diversity, attributed to its origin from Eurasian grey wolf ancestors as late as during the last glaciation and thus testifying extremely rapid adaptation to the afroalpine environment (Gottelli et al. 2004).
There is also some phylogenetic evidence suggesting that afroalpine plant populations are particularly prone to extinction. The most clear-cut evidence of extinction was found for Anthoxanthum; the currently allopatric southern and northern lineages once met in East Africa and produced an allopolyploid, but the southern lineage is absent from East Africa today (Fig. 8, Box 3;Tusiime et al. 2017). In the Lychnis phylogeny, the species are distinctly differentiated, but the terminal branches within species are typically very short and suggest not only recent colonisation of individual mountains, but also frequent local extinctions (Fig. 6, Box 4; Gizaw et al. 2016a). Similar findings of relatively old species that show recent interpopulational divergence and little intrapopulation genetic diversity are made in afromontane Canarina (Mairal et al. 2015, 2018). Thus, the afroalpine flora seems to represent a highly dynamic system characterized by cycles of colonisation, extinction, and recolonisation of individual sky islands during the Pleistocene climatic oscillations. A similar scenario has been suggested for the entire flora of the Cape Verde Islands (Brochmann et al. 1997), recently also for the flora of the easternmost islands in the Canarian archipelago (García-Verdugo et al. 2019); these are oceanic islands that are particularly prone to aridification. A promising avenue for further research into these questions is DNA metabarcoding of afroalpine lake sediments. Boessenkool et al. (2014) showed that this approach allows identification of past occurrences of taxa using a DNA reference library of the afroalpine flora.
Conclusions
We have shown that several of Hedberg’s insightful inferences and hypotheses on the origin and evolution of the afroalpine flora now have been tested with abundant molecular data and niche modelling tools, in many cases providing evidence fully consistent with his views. In particular, his emphasis on LDD as a major factor both for the initial establishment of the flora and for intermountain colonisation and gene flow has been confirmed in many studies. Hedberg held his ground concerning this question at a time when the importance of LDD was heatedly debated and strongly believed to be negligible by a major part of the scientific community in Scandinavia (summarized in Brochmann et al. 2003 and Abbott and Brochmann 2003). It should also be highlighted that Hedberg (1970) felt ‘inclined to believe that this [intermountain] dispersal may be due, not only to regular transport by the vectors mentioned above, but also to occasional catastrophic events, such as cyclones, which might easily carry away even those diaspores without any apparent adaptation for air flotation’. He thus anticipated what later became known as one of Berg’s principles of plant dispersal (Berg 1983; Nathan et al. 2008): morphological adaptations to dispersal play a subordinate role in LDD relative to truly unpredictable 'chance dispersal' with non-standard vectors.
However, the new insights and large body of new evidence point to a much younger and more dynamic island scenario than envisioned by Hedberg. The afroalpine flora appears to be unsaturated, fragile and vulnerable, harbouring unique taxonomic and genetic diversity that is severely depauperated by frequent bottlenecks and cycles of colonisation, extinction, and recolonisation. The tropical African sky islands can be viewed as a strongly isolated archipelago that still is in its infancy, repeatedly disturbed by the Pleistocene climatic oscillations, and with accelerating recruitment of immigrants arriving from remote temperate areas. Most of the effective dispersal into this system as well as within it has probably occurred during the glacials, because they were long-lasting, the source regions were closer, and the target areas were larger. The major shifts in temperature and rainfall must have induced extinctions, elevational oscillations of species and disruption of plant communities, repeatedly opening up new niches available for colonisation. Today, it is of particular conservation concern that many afroalpine plant species harbour little intrapopulation genetic variation, much less than reported from elsewhere. Thus, afroalpine plants may be extremely vulnerable and prone to extinction, not only because climate warming forces them to move upwards beyond the highest summits, but perhaps more seriously by the accelerating and destructive impact of human activities at high altitudes in these spectacular sky islands.
TEXT BOXES
Box 1
Carex—multiple Eurasian origins, limited diversification, LDDs and hybridisation (Figs. 4, 5). Gehrke and Linder (2009) used phylogenetic evidence to show that Carex colonized Africa 9–13 times independently. The genus originated in East Asia in the late Eocene, from where several migration events took place (e.g., Martin-Bravo et al. 2019). Several regions in the Northern Hemisphere have acted both as source and sink, and Africa and the Southern Hemisphere were colonized several times independently from Eurasia. All major lineages in Carex except the more warm-climate adapted subgenus Vigneastra colonized the afroalpine region, most of them probably during the Miocene, but they resulted in limited diversification (Gehrke and Linder 2009). Eight species reach afroalpine habitats, including the unusually tall C. bequaertii (Míguez 2018), several species of sect. Spirostachyae (Escudero and Luceño 2011), and the tussock-forming C. monostachya and C. runssoroensis (Gizaw et al. 2016b). The latter two sedges can form impressive tussocks up to 1.5 m high and dominate extensive afroalpine bogs, and are most closely related to the circumarctic/circumboreal C. capitata (Starr and Ford 2009; Gehrke 2011). Whereas C. monostachya is restricted to Ethiopia and eastern East Africa, C. runssoroensis occurs mainly in western East Africa but also in some eastern mountains, where the two species co-occur (Mt. Elgon, Mt. Kenya and the Aberdares). Gizaw et al. (2016b) addressed previous hybridisation hypotheses using AFLPs. The two species were distinctly differentiated, also in one of the mountains, where they co-occur, but the plants from Mt. Elgon were admixed. The results suggest initial allopatric divergence into one northern/eastern and one western lineage, which later spread across the vast Ugandan lowland gap. Surprisingly, one genetic group in C. monostachya was shared between the Ethiopian Bale Mountains and the distant East African mountains, testifying high dispersal capacity, but gene flow has nevertheless not prevented strong divergence in this species across the narrow Rift Valley in Ethiopia.
Box 2
Arabis alpina—double Eurasian origin, secondary contacts, local range shifts during glacial cycles. This arctic-alpine and afroalpine species is widespread in the eastern African mountains, where it reaches very high altitudes. Hedberg (1957) measured its morphological variation and concluded that the afroalpine populations belong to the same species as the arctic-alpine ones. He also performed artificial crosses between Kenyan and Swedish plants and found strong reduction in F1 fertility, but almost full recovery in F2 hybrids (Hedberg 1962). He thought that evolutionary diversification is slow in afroalpine Brassicaceae relative to other groups, and that A. alpina is a Tertiary relict in Africa (Hedberg 1965, 1969, 1970). Phylogeographic studies showed, however, that the species colonized the African mountains much more recently, and not only once. Based on synonymous mutation rates, Koch et al. (2006) found that two lineages of A. alpina migrated from the Balkans/Middle East into the eastern African mountains during the middle to late Pleistocene. The first migration probably occurred several glacial cycles ago; the second was more recent and resulted in secondary contact. Inferred from plastid DNA variation, the early-arriving lineage spread all over the eastern African mountains, whereas the late-arriving one seemed to be confined to Ethiopian mountains close to the Arabian Peninsula (Assefa et al. 2007). Ehrich et al. (2007) used AFLP data from 236 populations of A. alpina to test the prediction that the genetic consequences of range shifts in response to climate change during the Pleistocene differ among geographic areas. They found virtually no genetic diversity in the entire North Atlantic region, implying that plants from a single source population had recolonized vast areas after the last ice age. In contrast, genetic diversity was high and distinctly structured in the European Alps and the eastern African mountains, suggesting that range shifts in response to the glacial cycles were more local and served to preserve diversity. In the Alps, the genetic groups had patchy and partly overlapping distributions, whereas the genetic groups in the more fragmented African mountain system were clearly structured, although some LDDs were inferred. The AFLP data showed that the late-arriving lineage also dispersed from Ethiopia to the East African Mt Elgon, forming another secondary contact zone.
Box 3
Anthoxanthum—into Africa both from the north and southeast, limited diversification, secondary contact, polyploidization, local extinction (Fig. 8). In the Late Pliocene, sub-Saharan Africa was colonized by two divergent lineages of sweet vernal grasses, one from southeast Asia to southern Africa, and one from western Eurasia to eastern Africa (Pimentel et al. 2013). The nuclear phylogeny of Tusiime et al. (2017) shows that the two eastern African taxa originated after a single immigration from the north, confirming Hedberg’s (1976) hypothesis of a close relationship to the A. alpinum/odoratum complex. The eastern African nuclear sequences formed three subclades, one with the Ethiopian A. aethiopicum, one with eastern East African A. nivale, and one with western East African A. nivale. Surprisingly, however, the plastid phylogeny placed eastern A. nivale together with the southern African taxa, and coalescent simulations rejected lineage sorting as explanation for the incongruence. Thus, although currently allopatric, the southern and eastern African taxa must have met and hybridized in the middle to late Pleistocene. It is likely that the nuclear genome of eastern A. nivale also contains DNA derived from the southern African lineage; the nuclear phylogeny of Tusiime et al. (2017) was inferred from ribosomal sequences, and southern African rDNA may have been eliminated via concerted evolution. This may resolve the conflicting morphology-based opinions of whether A. nivale has northern (Hedberg 1976; Phillips 1995) or southern affinities (Clayton 1970). It has both. Notably, mountains such as Mt Kilimanjaro and Mt Kenya are now quite well-explored, so it is likely that the southern African lineage went extinct there after the hybridisation event. Tusiime et al. (2017) also found western A. nivale to be low-ploid and eastern A. nivale to be high-ploid (cf. Hedberg 1976), suggesting that the latter is an allopolyploid in which the southern African lineage left its footprint. This Pleistocene hybridisation event is particularly intriguing given the deeper history of the two lineages. The southern African lineage belongs to a mainly southeast Asian, tropical-alpine group which itself originated via Miocene hybridisation between Anthoxanthum and Hierochloë (Pimentel et al. 2013). The East African allopolyploid thus has a double hybrid background spanning millions of years and widely different geographic affinities.
Box 4
Lychnis—single Eurasian origin, polyploidization, moderate diversification, recent LDD to West Africa (Fig. 6). The sky island taxa of Lychnis (formerly referred to the endemic genus Uebelinia) were considered as typical examples of vicarious speciation by Fries (1923), but Ousted (1985) rather recognised one very widespread, highly disjunct species (L. abyssinica), as well as five other, partly disjunct species. They originated after a single colonisation from Eurasia in the late Miocene or early Pliocene (5.2 Ma), followed by differentiation into one diploid and one tetraploid lineage (Popp et al. 2008; Gizaw et al. 2016a). Most of the 95% HPD of the estimated time of origin and initial diversification corresponds to a warm and moist period (Bonnefille 1995), consistent with the preference of these species for swampy areas and bogs. Subsequent speciation events occurred sporadically from the late Pliocene to the middle Pleistocene. Habitat fragmentation triggered by aridification and subsequent allopatric speciation may explain the diversification pattern, as the time estimates roughly fit with known arid periods. Notably, whereas the sky island species of Lychnis are distinctly differentiated, the terminal branches within species are typically very short and suggest not only recent colonisation of individual mountains, but also frequent local extinctions. Colonisations seem, for example, to have occurred during the last 0.3 Myr when the Ethiopian L. kiwuensis and L. kigesiensis dispersed to mountains in Uganda, and, most strikingly, when the Ethiopian L. abyssinica spread to the remote mountains in Cameroon and Nigeria.

source to destination across a resistance cost surface. b Spatial extent of suitable habitats for this species under current and the three LGM climate models. c Schematic representation of the distribution of F. abyssinica during the LGM (in blue), showing the supported transcontinental dispersal events (continuous lines) and a potential stepping-stone pathway through Central Africa—Sudan (dashed line) (adopted from Mairal et al. 2021). d F. abyssinica grassland on Mt Kenya. Photo: Abel Gizaw
Likelihood analysis of dispersal routes in Festuca abyssinica. a Least-cost paths between western and eastern African high mountains under current and three LGM climate models [MIROC-ESM (mr), MPI-ESM-P (me), and CCSM (cc)]. The paths are plotted on a habitat suitability map produced by averaging predicted probability values from nine LGM habitat suitability maps (three algorithms *three climate models), with increasing suitability indicated as increasing darkness of grey to black. The cost raster was produced by assigning different cost values for the ensemble habitat suitability classes. Pixels which were unsuitable with high certainty were assigned a resistance cost of 10, pixels with uncertain suitability a cost of 5, and pixels which were suitable with high certainty a cost of 1. The accumulated cost to migrate between Cameroon and Gahinga/Muhavura is shown in green columns, and the accumulated cost to migrate between Cameroon and Simen is shown in yellow columns. Cost distance refers to the accumulation of these costs to migrate from
Box 5
Senecio—multiple diverse origins, moderate diversification. Senecio (or Senecioneae; ragweeds) with its > 1250 species is possibly the largest genus of flowering plants (depending on generic concept; Pelser et al. 2007). This clade has an almost worldwide distribution despite its relatively young age (mid to late Miocene; Kandziora et al. 2017). The highest species diversity is found in Southern Africa and South America. Ragweeds display a wide range of growth forms and life histories, including small trees, shrubs, vines, succulents, epiphytes and herbaceous annuals, and occupy a multitude of habitats. Many species and lineages have hybridized, undergone polyploidization, and colonized diverse environments, often through rapid phenotypic divergence and adaptive radiation (Walter et al. 2020). There are at least five clades with afroalpine species, resulting from 4 to 14 independent colonisations (Kandziora et al. 2016). Most afroalpine lineages probably originated from southern African mountains, and some possibly from the Palearctic and South America. Colonisation of the afroalpine region often resulted in moderate in situ diversification. The most species-rich afroalpine clade includes seven species, much less than some clades of this genus in the tropical-alpine Andean Páramo. This difference is probably associated with the different spatial extents of the alpine habitat (Kandziora et al. 2016).
Box 6
Fine-leaved Festuca—single South American origin, moderate diversification, double Pleistocene dispersal between East and West Africa (Fig. 7). The fine-leaved fescues form conspicuous tussocks in afromontane and afroalpine grasslands. They belong to a single clade that seem to have originated at the Messinian–Pliocene transition, remarkably from northern South America (Minaya et al. 2017). Most taxa are reported as narrow endemics (note, however, that a critical taxonomic revision is needed), but F. abyssinica s. lat. is widespread on both sides of tropical Africa (Hutchinson et al. 1968; Clayton 1970). Mairal et al. (2021) studied these fescues by combining phylogenetic, phylogeographic and niche modelling approaches. Surprisingly, the western East African populations of F. abyssinica were genetically more similar to the West African than to the eastern East African populations. Dispersal models and ecological niche predictions supported multiple LDDs in eastern Africa and two LDDs between the two sides of Africa in the Pleistocene (0.86 Ma and 0.52 Ma), possibly facilitated by stepping stone(s) along a Central Africa–Sudan pathway during the coldest periods. This is supported by scenarios using a coalescence approach (pDNA) and correlations between genetic and geographic distances (AFLP), and by ecological niche modelling showing patches of LGM habitat suitability in the Marrah Mountains, Bongos Massif, and Adamawa Plateau, resulting in reduced least-cost path distances.

Adopted from Tusiime et al. (2017). The map shows the double migration scenario inferred by Pimentel et al. (2013) and Tusiime et al. (2017): a single migration from Eastern Asia resulted in the southern African lineage, and a single migration from western Eurasia resulted in the eastern African lineage (A. nivale and A. aethiopicum). These lineages are currently allopatric, but they met and hybridized in the eastern East African mountains in the late Pleistocene. Photo: A. nivale by Abel Gizaw
Multi-labelled maximum clade credibility species tree of Anthoxanthum in sub-Saharan Africa obtained from a *BEAST analysis based on two nuclear (ITS and ETS) and two plastid (trnLF and trnTL) DNA regions. In A. nivale, the eastern mountain (EM) group is inferred to be hybridogeneous (hence represented twice in the tree). Dashed lines indicate branches with PP < 0.8. Bars show the 95% confidence interval for the age of the divergence.
Box 7
Erica—connections both to the south and north. This genus comprises > 800 species, of which ~ 700 represent a single radiation in the Cape Floristic Region (Pirie et al. 2016). By contrast, just 23 species occur across the African high mountains, but these have a somewhat longer history, with occasional exchange with the ancestral range of the Ericeae clade to the north (western Europe and the Mediterranean) and with the younger Drakensberg Mountains lineage to the south (Pirie et al. 2019). Dispersal has typically occurred between adjacent, ecologically similar areas, but the pattern of regional endemic clades and near 100% regional species endemism implies that most of the African high mountain species evolved within the region. Most of them belong to a single clade that also includes the substantial Madagascan diversity. Two of them, Erica arborea and E. trimera, are unusual: both are phylogenetically isolated, relict lineages stemming back to the initial establishment of Erica in the afrotemperate region around 15 Ma. Both species are shrubs or small trees with tiny seeds, potentially capable of dispersal by wind over long distances, and both are widespread in the subalpine ericaceous zone, but with E. trimera restricted to higher altitudes than E. arborea. Gizaw et al. (2013) used pDNA and AFLP data to test the prediction that the more high-altitudinal E. trimera shows more distinct genetic structuring than the lower-altitudinal E. arborea, because intermountain migration of E. arborea may have been facilitated by habitat bridges under former climates. They found limited within-species diversity, but the genetic structuring was according to the prediction: E. trimera consisted of several distinct genetic groups with different pDNA haplotypes in different mountains, suggesting long-term isolation, whereas E. arborea showed virtually no geographic structuring at AFLP loci and only a single, widespread pDNA haplotype.
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Abbott RJ, Brochmann C (2003) History and evolution of the arctic flora: in the footsteps of Eric Hultén. Mol Ecol 12:299–313. https://doi.org/10.1046/j.1365-294X.2003.01731.x
Alsos IG, Ehrich D, Eidesen PB, Solstad H, Westergaard KB, Schönswetter P, Tribsch A, Birkeland S, Elven R, Brochmann C (2015) Long-distance plant dispersal to North Atlantic islands: colonization routes and founder effect. AoB PLANTS 7. plv036, https://doi.org/10.1093/aobpla/plv036
Alsos IG, Eidesen PB, Ehrich D, Skrede I, Westergaard K, Jacobsen GH, Landvik JY, Taberlet P, Brochmann C (2007) Frequent long-distance plant colonization in the changing Arctic. Science 316:1606–1609. https://doi.org/10.1126/science.1139178
Assefa A, Ehrich D, Taberlet P, Nemomissa S, Brochmann C (2007) Pleistocene colonization of afro-alpine “sky islands” by the arctic-alpine Arabis alpina. Heredity 99:133–142. https://doi.org/10.1038/sj.hdy.6800974
Beck E (1986) Biology of afroalpine Dendrosenecio (Asteraceae). Plant Syst Evol 152:123–131. https://doi.org/10.1007/BF00985353
Bekele B, (2006) The Swertia abyssinica complex in afroalpine mountain systems: parallel ecoclinal evolution or secondary contact between differentiated glacial gene pools? MSc Thesis. Addis Ababa University, Ethiopia.
Berg RY (1983) Plant distribution as seen from plant dispersal: General principles and basic modes of plant dispersal. In: Kubitzki, K (Ed.) Dispersal and distribution. Paul Parey, pp. 13–36.
Bertel C, Rešetnik I, Frajman B, Erschbamer B, Hülber K, Schönswetter P (2018) Natural selection drives parallel divergence in the mountain plant Heliosperma pusillum s.l. Oikos 127:1355–1367. https://doi.org/10.1111/oik.05364
Birkeland S, Gustafsson ALS, Brysting AK, Brochmann C, Nowak MD (2020) Multiple genetic trajectories to extreme abiotic stress adaptation in Arctic Brassicaceae. Mol Biol Evol 37:2052–2068. https://doi.org/10.1093/molbev/msaa068
Boessenkool S, McGlynn G, Epp LS, Taylor D, Pimentel M, Gizaw A, Nemomissa S, Brochmann C, Popp M (2014) Ancient sedimentary DNA from a biodiversity hotspot in the humid tropics introduces a novel tool in conserving high-altitude tropical biodiversity. Conserv Biol 28:446–455. https://doi.org/10.1111/cobi.12195
Bonnefille R (1995) A reassessment of the Plio-Pleistocene pollen record of East Africa. In: Vrba, ES, Denton, GH, Partridge, TC, Burckle, LH (Eds.) Paleoclimate and evolution with emphasis on human origins. Yale University Press, pp. 299–310.
Bonnefille R (2010) Cenozoic vegetation, climate changes and hominid evolution in tropical Africa. Glob Planet Change 72:390–411. https://doi.org/10.1016/j.gloplacha.2010.01.015
Borregaard MK, Matthews TJ, Whittaker RJ (2016) The general dynamic model: towards a unified theory of island biogeography? Glob Ecol Biogeogr 25:805–816. https://doi.org/10.1111/geb.12348
Brochmann C, Brysting AK, Alsos IG, Borgen L, Grundt HH, Scheen A-C, Elven R (2004) Polyploidy in arctic plants. Biol J Linn Soc 82:521–536. https://doi.org/10.1111/j.1095-8312.2004.00337.x
Brochmann C, Gabrielsen TM, Nordal I, Landvik JY, Elven R (2003) Glacial survival or tabula rasa? The history of North Atlantic biota revisited. Taxon 52:417–450
Brochmann C, Rustan ØH, Lobin W, Kilian N (1997) The endemic vascular plants of the Cape Verde Islands, W Africa. Sommerfeltia 24:1–356. https://doi.org/10.2307/3647381
Carbutt C, Edwards TJ (2015) Reconciling ecological and phytogeographical spatial boundaries to clarify the limits of the montane and alpine regions of sub-Sahelian Africa. S Afr J Bot 98:64–75. https://doi.org/10.1016/j.sajb.2015.01.014
Carlsen T, Bleeker W, Hurka H, Elven R, Brochmann C (2009) Biogeography and phylogeny of Cardamine (Brassicaceae). Ann Missouri Bot Gard 96:215–236. https://doi.org/10.3417/2007047
Chala D, Brochmann C, Psomas A, Ehrich D, Gizaw A, Masao CA, Bakkestuen V, Zimmermann NE (2016) Good-bye to tropical alpine plant giants under warmer climates? Loss of range and genetic diversity in Lobelia rhynchopetalum. Ecol Evol 6:8931–8941
Chala D, Zimmermann NE, Brochmann C, Bakkestuen V (2017) Migration corridors for alpine plants among the ‘sky islands’ of eastern Africa: do they, or did they exist? Alp Bot 127:133–144. https://doi.org/10.1002/ece3.2603
Chartier M, Dressler S, Schönenberger J, Mora AR, Sarthou C, Wang W, Jabbour F (2016) The evolution of afro-montane Delphinium (Ranunculaceae): Morphospecies, phylogenetics and biogeography. Taxon 65:1313–1327. https://doi.org/10.12705/656.6
Chassot P, Nemomissa S, Yuan Y-M, Küpfer P (2001) High paraphyly of Swertia L. (Gentianaceae) in the Gentianella-lineage as revealed by nuclear and chloroplast DNA sequence variation. Plant Syst Evol 229:1–21. https://doi.org/10.1007/s006060170015
Chen L-Y, Muchuku JK, Yan X, Hu G-W, Wang Q-F (2015) Phylogeography of Haplocarpha rueppelii (Asteraceae) suggests a potential geographic barrier for plant dispersal and gene flow in East Africa. Sci Bull 60:1184–1192. https://doi.org/10.1007/s11434-015-0832-x
Clayton WD (1970) Gramineae I. In: Milne-Redhead, CE, Polhill, RM (Eds.) Flora of tropical East Africa. Crown Agents, pp. 1–176.
Coetzee JA (1964) Evidence for a considerable depression of the vegetation belts during the upper Pleistocene on the East African mountains. Nature 204:564–566. https://doi.org/10.1038/204564a0
Couvreur TLP, Chatrou LW, Sosef MSM, Richardson JE (2008) Molecular phylogenetics reveal multiple tertiary vicariance origins of the African rain forest trees. BMC Evol Biol 6:54. https://doi.org/10.1186/1741-7007-6-54
CEPF - Critical Ecosystem Partnership Fund (2016). Biodiversity Hotspots Defined. https://www.cepf.net/our-work/biodiversity-hotspots/hotspots-defined. Accessed 26 Feb. 2021.
Cronk Q (1997) Islands: stability, diversity, conservation. Biodivers Conserv 6:477–493. https://doi.org/10.1023/A:1018372910025
Demissew S, Brochmann C, Kelbessa E, Friis I, Nordal I, Nemomissa S, Woldemariam T, Bekele T, Asfaw Z, Woldu Z (2011) The Ethiopian Flora project: a springboard to other projects. Symb Bot Ups 35:189–215
Dimitrov D, Nogués-Bravo D, Scharff N (2012) Why do tropical mountains support exceptionally high biodiversity? The Eastern Arc mountains and the drivers of Saintpaulia diversity. PLoS ONE 7:e48908. https://doi.org/10.1371/journal.pone.0048908
de Deus Vidal Jr J, Clark VR, (2020) Afro-alpine plant diversity in the tropical mountains of Africa. Encyclopedia of the World’s Biomes. https://doi.org/10.1016/B978-0-12-409548-9.11885-8
Durvasula A, Fulgione A, Gutaker RM, Alacakaptan SI, Flood PJ, Neto C, Tsuchimatsu T, Burbano HA, Picó FX, Alonso-Blanco C (2017) African genomes illuminate the early history and transition to selfing in Arabidopsis thaliana. PNAS 114:5213–5218. https://doi.org/10.1073/pnas.1616736114
Ebinger C, Yemane T, Harding D, Tesfaye S, Kelley S, Rex D (2000) Rift deflection, migration, and propagation: Linkage of the Ethiopian and Eastern rifts, Africa. Geol Soc Am Bull 112:163–176
Ehrich D, Gaudeul M, Assefa A, Koch MA, Mummenhoff K, Nemomissa S, Brochmann C (2007) Genetic consequences of Pleistocene range shifts: contrast between the Arctic, the Alps and the East African mountains. Mol Ecol 16:2542–2559. https://doi.org/10.1111/j.1365-294X.2007.03299.x
Emadzade K, Gehrke B, Linder HP, Hörandl E (2011) The biogeographical history of the cosmopolitan genus Ranunculus L. (Ranunculaceae) in the temperate to meridional zones. Mol Phylogenet Evol 58:4–21. https://doi.org/10.1016/j.ympev.2010.11.002
Engler A (1892) Über die Hochgebirgsflora des tropischen Afrika. Verlag der Königl. Akademie der Wissenschaften in Commission bei G, Reimer, Berlin
Escudero M, Luceño M (2011) Taxonomic revision of the tropical African group of Carex subsect. Elatae (sect. Spirostachyae, Cyperaceae). An Jard Bot Madr 68:225–247. https://doi.org/10.3989/ajbm.2256
Fernández-Palacios JM, Rijsdijk KF, Norder SJ, Otto R, de Nascimento L, Fernández-Lugo S, Tjørve E, Whittaker RJ (2016) Towards a glacial-sensitive model of island biogeography. Glob Ecol Biogeogr 25:817–830. https://doi.org/10.1111/geb.12320
Fetene M, Gashaw M, Nauke P, Beck E (1998) Microclimate and ecophysiological significance of the tree-like life-form of Lobelia rhynchopetalum in a tropical alpine environment. Oecologia 113:332–340. https://doi.org/10.1007/s004420050384
Fjeldså J, Lovett JC (1997) Geographical patterns of old and young species in African forest biota: The significance of specific montane areas as evolutionary centres. Biodivers Conserv 6:325–346. https://doi.org/10.1023/A:1018356506390
Flantua SG, Hooghiemstra H (2018) Historical connectivity and mountain biodiversity. In: Hoorn C, Perrigo A, Antonelli A (eds) Mountains, climate and biodiversity. Wiley, Blackwell, pp 171–185
Flantua SG, Payne D, Borregaard MK, Beierkuhnlein C, Steinbauer MJ, Dullinger S, Essl F, Irl SD, Kienle D, Kreft H (2020) Snapshot isolation and isolation history challenge the analogy between mountains and islands used to understand endemism. Glob Ecol Biogeogr 29:1651–1673. https://doi.org/10.1111/geb.13155
Fries RE, Fries TCE (1922a) Die Riesen-Lobelien Afrikas Sven Bot Tidskr 16:383–416
Fries RE, Fries TCE (1922b) Ober die Riesen-Senecionen der afrikanischen Hochgebirge. Sven Bot Tidskr 16:321–340
Fries TCE (1923) Beiträge zur Kenntnis der Flora des Kenia, Mt. Aberdare und Mt. Elgon Notizbl Bot Gart Berlin-Dahlem 8:389–423. https://doi.org/10.2307/3994426
Fries RE, Fries TCE (1948) Phytogeographical researches on Mt. Kenya and Mt. Aberdare, British East Africa. Kongl Vetensk Acad Handl 25:1–83
Fulgione A, Hancock AM (2018) Archaic lineages broaden our view on the history of Arabidopsis thaliana. New Phytol 219:1194–1198. https://doi.org/10.1111/nph.15244
Galbany-Casals M, Unwin M, Garcia-Jacas N, Smissen RD, Susanna A, Bayer RJ (2014) Phylogenetic relationships in Helichrysum (Compositae: Gnaphalieae) and related genera: Incongruence between nuclear and plastid phylogenies, biogeographic and morphological patterns, and implications for generic delimitation. Taxon 63:608–624. https://doi.org/10.12705/633.8
García-Verdugo C, Caujapé-Castells J, Illera JC, Mairal M, Patiño J, Reyes-Betancort A, Scholz S (2019) Pleistocene extinctions as drivers of biogeographical patterns on the easternmost Canary Islands. J Biogeogr 46:845–859. https://doi.org/10.1111/jbi.13563
Gebremedhin B, Ficetola GF, Naderi S, Rezaei HR, Maudet C, Rioux D, Luikart G, Flagstad O, Thuiller W, Taberlet P (2009) Combining genetic and ecological data to assess the conservation status of the endangered Ethiopian walia ibex. Anim Conserv 12:89–100. https://doi.org/10.1111/j.1469-1795.2009.00238.x
Gebremedhin B, Flagstad O, Bekele A, Chala D, Bakkestuen V, Boessenkool S, Popp M, Gussarova G, Schroder-Nielsen A, Nemomissa S, Brochmann C, Stenseth NC, Epp LS (2016) DNA metabarcoding reveals diet overlap between the endangered Walia Ibex and domestic goats - Implications for conservation. PLoS ONE. https://doi.org/10.1371/journal.pone.0159133
Gehrke B (2011) Synopsis of Carex (Cyperaceae) from sub-Saharan Africa and Madagascar. Bot J Linn Soc 166:51–59. https://doi.org/10.1111/j.1095-8339.2011.01116.x
Gehrke B, Kandziora M, Pirie MD (2016) The evolution of dwarf shrubs in alpine environments: a case study of Alchemilla in Africa. Ann Bot 117:121–131
Gehrke B, Linder HP (2009) The scramble for Africa: pan-temperate elements on the African high mountains. Proc Royal Soc B 276:2657–2665. https://doi.org/10.1093/aob/mcv159
Gehrke B, Linder HP (2011) Time, space and ecology: why some clades have more species than others. J Biogeogr 38:1948–1962. https://doi.org/10.1111/j.1365-2699.2011.02544.x
Gehrke B, Linder HP (2014) Species richness, endemism and species composition in the tropical Afroalpine flora. Alp Bot 124:165–177. https://doi.org/10.1007/s00035-014-0132-0
Gehrke B (2018) Staying cool: preadaptation to temperate climates required for colonising tropical alpine-like environments. PhytoKeys 96:111–125. https://doi.org/10.3897/phytokeys.96.13353
Gichira AW, Li Z-Z, Saina JK, Hu G-W, Gituru RW, Wang Q-F, Chen J-M (2017) Demographic history and population genetic structure of Hagenia abyssinica (Rosaceae), a tropical tree endemic to the Ethiopian highlands and eastern African mountains. Tree Genet Genomes 13:1–11. https://doi.org/10.1007/s11295-017-1156-6
Givnish TJ (2010) Giant lobelias exemplify convergent evolution. BMC Biol 8:3. https://doi.org/10.1186/1741-7007-8-3
Gizaw A, Brochmann C, Nemomissa S, Wondimu T, Masao CA, Tusiime FM, Abdi AA, Oxelman B, Popp M, Dimitrov D (2016a) Colonization and diversification in the African “sky islands”: insights from fossil-calibrated molecular dating of Lychnis (Caryophyllaceae). New Phytol 211:719–734. https://doi.org/10.1111/nph.13937
Gizaw A, Gorospe JM, Kandziora M, Chala D, Gustafsson L, Zinaw A, Salomón L, Eilu G, Brochmann C, Kolář F, Schmickl R. Is lack of phylogenetic resolution the hallmark of alpine radiation? Insights from the afro-alpine giants in Dendrosenecio (Senecioneae, Asteraceae). Alpine Botany
Gizaw A, Kebede M, Nemomissa S, Ehrich D, Bekele B, Mirré V, Popp M, Brochmann C (2013) Phylogeography of the heathers Erica arborea and E. trimera in the afro-alpine ‘sky islands’ inferred from AFLPs and plastid DNA sequences. Flora 208:453–463. https://doi.org/10.1016/j.flora.2013.07.007
Gizaw A, Wondimu T, Mugizi TF, Masao CA, Abdi AA, Popp M, Ehrich D, Nemomissa S, Brochmann C (2016b) Vicariance, dispersal, and hybridization in a naturally fragmented system: the afro-alpine endemics Carex monostachya and C. runssoroensis (Cyperaceae). Alp Bot 126:59–71. https://doi.org/10.1007/s00035-015-0162-2
Gottelli D, Marino J, Sillero-Zubiri C, Funk SM (2004) The effect of the last glacial age on speciation and population genetic structure of the endangered Ethiopian wolf (Canis simensis). Mol Ecol 13:2275–2286. https://doi.org/10.1111/j.1365-294X.2004.02226.x
Griffiths CJ (1993) The geological evolution of East Africa. In: Lovett JC, Wasser SK (eds) Biogeography and ecology of the rain forests of Eastern Africa. Cambridge University Press, Cambridge, pp 19–23
Hamilton AC (1982) Environmental history of East Africa: A study of the quaternary. Academic Press, London
Hauman L (1933) Esquisse de la végétation des hautes altitudes sur le Ruwenzori. Bull Acad r Belg 19:602–616
Hedberg I (1976) A cytotaxonomic reconnaissance of tropical African Anthoxanthum L. (Gramineae). 129:85–90
Hedberg I, Hedberg O (1977) Chromosome numbers of afroalpine and afromontane angiosperms. Bot Not 130:1–24
Hedberg I, Persson E (2011) The Ethiopian Flora Project 1980–2009: Exploration, Collaboration, Inspiration. Dedicated to Olov Hedberg. In: Hedberg, I, Persson, E (Eds.) Proceedings of the Ethiopian Flora Symposium. Uppsala universitet, Uppsala, pp. 1–250.
Hedberg O (1955) Altitudinal zonation of the vegetation on the East African Mountains. J Proc Linn Soc Lond 165:134–136
Hedberg O (1956) Some taxonomic problems concerning the afro-alpine flora. Webbia 11:471–487. https://doi.org/10.1080/00837792.1956.10669645
Hedberg O (1957) Afroalpine vascular plants. A Taxonomic Revision Symb Bot Ups 15:1–411
Hedberg O (1961) The phytogeographical position of the afroalpine flora. Rec Adv Bot 1:914–919
Hedberg O (1962) Intercontinental crosses in Arabis alpina L. Caryologia 15:253–260. https://doi.org/10.1080/00087114.1962.10796058
Hedberg O (1964) Features of afroalpine plant ecology. Acta Phytogeogr Suec 49:1–144
Hedberg O (1965) Afroalpine flora elements. Webbia 19:519–529
Hedberg O (1969) Evolution and speciation in a tropical high mountain flora. Biol J Linn Soc 1:135–148. https://doi.org/10.1111/j.1095-8312.1969.tb01816.x
Hedberg O (1970) Evolution of the afroalpine flora. Biotropica 2:16–23. https://doi.org/10.2307/2989783
Hedberg O (1983) Ethiopian flora project. Bothalia 14:571–574
Hedberg O (1986) Origin of the afroalpine flora. In: Vuilleumier, F, Monastero, M (Eds.) High altitude tropical biogeography. Oxford University Press, pp. 443–468.
Hedberg O (1997) High-Mountain areas of Tropical Africa. In: Wielgolaski, FE (Ed.) Ecosystems of the world 3: Polar and alpine tundra. Elsevier, pp. 185–197.
Hemp A, Hemp C (2018) Broken bridges: The isolation of Kilimanjaro’s ecosystem. Glob Change Biol 24:3499–3507. https://doi.org/10.1111/gcb.14078
Hendrickx H, Jacob M, Frankl A, Guyassa E, Nyssen J (2014) Quaternary glacial and periglacial processes in the Ethiopian Highlands in relation to the current afro-alpine vegetation. Z Geomorphol 58:37–57
Hohmann N, Wolf EM, Lysak MA, Koch MA (2015) A time-calibrated road map of Brassicaceae species radiation and evolutionary history. Plant Cell 27:2770–2784
Hörandl E, Emadzade K (2011) The evolution and biogeography of alpine species in Ranunculus (Ranunculaceae): a global comparison. Taxon 60:415–426. https://doi.org/10.1105/tpc.15.00482
Hughes CE, Atchison GW (2015) The ubiquity of alpine plant radiations: from the Andes to the Hengduan Mountains. New Phytol 207:275–282. https://doi.org/10.1111/nph.13230
Hutchinson J, Dalziel J, Hepper F (1968) Phylogenetic sequence of orders and families. Published by Crown Agents for Overseas Governments and Administrations, Flora of West Tropical Africa, p 574
Kadu CA, Konrad H, Schueler S, Muluvi GM, Eyog-Matig O, Muchugi A, Williams VL, Ramamonjisoa L, Kapinga C, Foahom B, Katsvanga C, Hafashimana D, Obama C, Geburek T (2013) Divergent pattern of nuclear genetic diversity across the range of the Afromontane Prunus africana mirrors variable climate of African highlands. Ann Bot 111:47–60. https://doi.org/10.1093/aob/mcs235
Kadu CA, Schueler S, Konrad H, Muluvi GM, Eyog-Matig O, Muchugi A, Williams VL, Ramamonjisoa L, Kapinga C, Foahom B, Katsvanga C, Hafashimana D, Obama C, Geburek T (2011) Phylogeography of the Afromontane Prunus africana reveals a former migration corridor between East and West African highlands. Mol Ecol 20:165–178. https://doi.org/10.1111/j.1365-294X.2010.04931.x
Kandziora M, Kadereit JW, Gehrke B (2016) Frequent colonization and little in situ speciation in Senecio in the tropical alpine-like islands of eastern Africa. Am J Bot 103:1483–1498. https://doi.org/10.3732/ajb.1600210
Kandziora M, Kadereit JW, Gehrke B (2017) Dual colonization of the Palaearctic from different regions in the Afrotropics by Senecio. J Biogeogr 44:147–157. https://doi.org/10.1111/jbi.12837
Kebede M, Ehrich D, Taberlet P, Nemomissa S, Brochmann C (2007) Phylogeography and conservation genetics of a giant lobelia (Lobelia giberroa) in Ethiopian and Tropical East African mountains. Mol Ecol 16:1233–1243. https://doi.org/10.1111/j.1365-294X.2007.03232.x
Knox E, Palmer J (1995) Chloroplast DNA variation and the recent radiation of the giant senecios (Asteraceae) on the tall mountains of eastern Africa. PNAS 92:10349–10353. https://doi.org/10.1073/pnas.92.22.10349
Knox E, Palmer J (1998) Chloroplast DNA evidence on the origin and radiation of the giant lobelias in eastern Africa. Syst Bot 23:109–149. https://doi.org/10.2307/2419583
Knox EB (2005) Dendrosenecio. In: Beentje H, GS (Ed.) Flora of tropical East Africa, Compositae (part 3). Royal Botanical Gardens Kew, pp. 548–563.
Knox EB, Li C (2017) The East Asian origin of the giant lobelias. Am J Bot 104:924–938. https://doi.org/10.3732/ajb.1700025
Koch MA, Kiefer C, Ehrich D, Vogel J, Brochmann C, Mummenhoff K (2006) Three times out of Asia Minor: the phylogeography of Arabis alpina L. (Brassicaceae). Mol Ecol 15:825–839. https://doi.org/10.1111/j.1365-294X.2005.02848.x
Kolář F, Dušková E, Sklenář P (2016a) Niche shifts and range expansions along cordilleras drove diversification in a high-elevation endemic plant genus in the tropical Andes. Mol Ecol 25:4593–4610. https://doi.org/10.1111/mec.13788
Kolář F, Fuxová G, Záveská E, Nagano AJ, Hyklová L, Lučanová M, Kudoh H, Marhold K (2016b) Northern glacial refugia and altitudinal niche divergence shape genome-wide differentiation in the emerging plant model Arabidopsis arenosa. Mol Ecol 25:3929–3949. https://doi.org/10.1111/mec.13721
Komarova VA, Kostin DS, Bryja J, Mikula O, Bryjová A, Čížková D, Šumbera R, Meheretu Y, Lavrenchenko LA (2021) Complex reticulate evolution of speckled brush-furred rats (Lophuromys) in the Ethiopian centre of endemism. Mol Ecol. https://doi.org/10.1111/mec.15891
Konečná V, Nowak MD, Kolář F (2019) Parallel colonization of subalpine habitats in the central European mountains by Primula elatior. Sci Rep 9:1–12. https://doi.org/10.1038/s41598-019-39669-2
Kosman E (2003) Nei’s gene diversity and the index of average differences are identical measures of diversity within populations. Plant Pathol 52:533–535
Lagomarsino LP, Condamine FL, Antonelli A, Mulch A, Davis CC (2016) The abiotic and biotic drivers of rapid diversification in Andean bellflowers (Campanulaceae). New Phytol 210:1430–1442. https://doi.org/10.1111/nph.13920
Linder HP (2014) The evolution of African plant diversity. Front Ecol Evol 2:1–14. https://doi.org/10.3389/fevo.2014.00038
Loader SP, Sara Ceccarelli F, Menegon M, Howell KM, Kassahun R, Mengistu AA, Saber SA, Gebresenbet F, de Sá R, Davenport TRB, Larson JG, Müller H, Wilkinson M, Gower DJ (2014) Persistence and stability of Eastern Afromontane forests: evidence from brevicipitid frogs. J Biogeogr 41:1781–1792. https://doi.org/10.1111/jbi.12331
Lovett JC, Bridson DM, Thomas DW (1988) A preliminary list of the moist forest angiosperm flora of the Mwanihana Forest Reserve, Tanzania. Ann Missouri Bot Gard 75:874–888. https://doi.org/10.2307/2399376
Mabberley DJ (1986) Adaptive syndromes of the afroalpine species of Dendrosenecio. In: Vuillemuier, F, Monasterio, M (Eds.) High altitude tropical biogeography. Oxford University Press, pp. 81–102.
Madriñán S, Cortés AJ, Richardson JE (2013) Páramo is the world’s fastest evolving and coolest biodiversity hotspot. Front Genet 4:192. https://doi.org/10.3389/fgene.2013.00192
Mairal M, Caujapé-Castells J, Pellissier L, Jaén-Molina R, Álvarez N, Heuertz M, Sanmartín I (2018) A tale of two forests: ongoing aridification drives population decline and genetic diversity loss at continental scale in Afro-Macaronesian evergreen-forest archipelago endemics. Ann Bot 122:1005–1017. https://doi.org/10.1093/aob/mcy107
Mairal M, Pokorny L, Aldasoro JJ, Alarcón M, Sanmartín I (2015) Ancient vicariance and climate-driven extinction continental-wide disjunctions in Africa: the case of the Rand Flora genus Canarina (Campanulaceae). Mol Ecol 24:1335–1354. https://doi.org/10.1111/mec.13114
Mairal M, Sanmartín I, Herrero A, Pokorny L, Vargas P, Aldasoro JJ, Alarcón M (2017) Geographic barriers and Pleistocene climate change shaped patterns of genetic variation in the Eastern Afromontane biodiversity hotspot. Sci Rep 7:1–13. https://doi.org/10.1038/srep45749
Mairal M, Namaganda M, Gizaw A, Chala D, Brochmann C, Catalán P (2021) Multiple mountain-hopping colonization of sky-islands on the two sides of Tropical Africa during the Pleistocene: the afroalpine Festuca grasses. J Biogeogr. https://doi.org/10.1111/jbi.14117
Maitima JM (1991) Vegetation response to climatic change in Central Rift Valley, Kenya. Quat Res 35:234–245. https://doi.org/10.1016/0033-5894(91)90070-L
Martín-Bravo S, Jiménez-Mejías P, Villaverde T, Escudero M, Hahn M, Spalink D, Roalson EH, Hipp AL, Global Carex Group, Benítez-Benítez C (2019) A tale of worldwide success: Behind the scenes of Carex (Cyperaceae) biogeography and diversification. J Syst Evol 57:695–718. https://doi.org/10.1111/jse.12549
Masao CA, Gizaw A, Piñeiro R, Tusiime FM, Wondimu T, Abdi AA, Popp M, Gussarova G, Lye KA, Munishi P, Nemomissa S, Brochmann C (2013) Phylogeographic history and taxonomy of some afro-alpine grasses assessed based on AFLPs and morphometry: Deschampsia cespitosa, D. angusta and Koeleria capensis. Alp Bot 123:107–122. https://doi.org/10.1007/s00035-013-0119-2
Merckx VSFT, Hendriks KP, Beentjes KK et al (2015) Evolution of endemism on a young tropical mountain. Nature 524:347–350. https://doi.org/10.1038/nature14949
Messerli B, Winiger M (1992) Climate, environmental change, and resources of the African mountains from the Mediterranean to the equator. Mt Res Dev 12:315–336. https://doi.org/10.2307/3673683
Migliore J, Lézine A-M, Hardy OJ (2020) The recent colonization history of the most widespread Podocarpus tree species in Afromontane forests. Ann Bot 126:73–83. https://doi.org/10.1093/aob/mcaa049
Míguez M, Martín-Bravo S, Jiménez-Mejías P (2018) Reconciling morphology and phylogeny allows an integrative taxonomic revision of the giant sedges of Carex section Rhynchocystis (Cyperaceae). Bot J Linn Soc 188:34–58. https://doi.org/10.1093/botlinnean/boy040
Mihretie Z, Schueler S, Konrad H, Bekele E, Geburek T (2015) Patterns of genetic diversity of Prunus africana in Ethiopia: hot spot but not point of origin for range-wide diversity. Tree Genet Genomes 11:1–13. https://doi.org/10.1007/s11295-015-0945-z
Minaya M, Hackel J, Namaganda M, Brochmann C, Vorontsova MS, Besnard G, Catalán P (2017) Contrasting dispersal histories of broad- and fine-leaved temperate Loliinae grasses: range expansion, founder events, and the roles of distance and barriers. J Biogeogr 44:1980–1993. https://doi.org/10.1111/jbi.13116
Mittermeier RA, Turner WR, Larsen FW, Brooks TM, Gascon C (2011) Global Biodiversity Conservation: The Critical Role of Hotspots. In: Zachos FE, Habel JC (eds) Biodiversity hotspots: distribution and protection of conservation priority areas. Springer, Berlin Heidelberg, pp 3–22
Nagy L, Grabherr G (2009) The biology of alpine habitats. Oxford University Press, Oxford
Nathan R, Schurr FM, Spiegel O, Steinitz O, Trakhtenbrot A, Tsoar A (2008) Mechanisms of long-distance seed dispersal. Trends Ecol Evol 23:638–647. https://doi.org/10.1016/j.tree.2008.08.003
Nevado B, Contreras-Ortiz N, Hughes C, Filatov DA (2018) Pleistocene glacial cycles drive isolation, gene flow and speciation in the high-elevation Andes. New Phytol 219:779–793. https://doi.org/10.1111/nph.15243
Nevado B, Wong EL, Osborne OG, Filatov DA (2019) Adaptive evolution is common in rapid evolutionary radiations. Curr Biol 29(3081–3086):e3085. https://doi.org/10.1016/j.cub.2019.07.059
Nordenstam B (1978) Taxonomic studies in the tribe Senecioneae (Compositae). Opera Bot 44:1–84
Nowak MD, Birkeland S, Mandáková T, Roy Choudhury R, Guo X, Gustafsson ALS, Gizaw A, Schrøder-Nielsen A, Fracassetti M, Brysting AK, Rieseberg L, Slotte T, Parisod C, Lysak MA, Brochmann C (2020) The genome of Draba nivalis shows signatures of adaptation to the extreme environmental stresses of the Arctic. Mol Ecol Resour 21:661–676. https://doi.org/10.1111/1755-0998.13280
Nürk NM, Uribe-Convers S, Gehrke B, Tank DC, Blattner FR (2015) Oligocene niche shift, Miocene diversification - cold tolerance and accelerated speciation rates in the St. John’s Worts (Hypericum, Hypericaceae). BMC Evol Biol 15:1–13. https://doi.org/10.1186/s12862-015-0359-4
Nürk NM, Michling F, Linder HP (2018) Are the radiations of temperate lineages in tropical alpine ecosystems pre-adapted? Glob Ecol Biogeogr 27:334–345. https://doi.org/10.1111/geb.12699
Nürk NM, Atchison GW, Hughes CE (2019) Island woodiness underpins accelerated disparification in plant radiations. New Phytol 224:518–531. https://doi.org/10.1111/nph.15797
Nybom H (2004) Comparison of different nuclear DNA markers for estimating intraspecific genetic diversity in plants. Mol Ecol 13:1143–1155. https://doi.org/10.1111/j.1365-294X.2004.02141.x
Ousted S (1985) A taxonomic revision of the genus Uebelinia Hochst. (Caryophyllaceae). Bull Jard Bot Natl Belg 55:421–459. https://doi.org/10.2307/3667962
Pelser PB, Nordenstam B, Kadereit JW, Watson LE (2007) An ITS phylogeny of tribe Senecioneae (Asteraceae) and a new delimitation of Senecio L. Taxon 56:1077–1104. https://doi.org/10.2307/25065905
Perez MP, Yu W-B (2021) Pleistocene origin and colonization history of Lobelia columnaris Hook. f.(Campanulaceae: Lobelioideae) across sky islands of West Central Africa. Authorea Preprints. https://doi.org/10.22541/au.161201731.10223625/v1
Phillips S (1995) Poaceae (Gramineae). In: Hedberg I, Edwards S (eds) Flora of Ethiopia and Eritrea. Addis Ababa University and Department of Systematic Botany, Uppsala University, The National Herbarium, pp 1–420
Pimentel M, Sahuquillo E, Torrecilla Z, Popp M, Catalán P, Brochmann C (2013) Hybridization and long-distance colonization at different time scales: towards resolution of long-term controversies in the sweet vernal grasses (Anthoxanthum). Ann Bot 112:1015–1030. https://doi.org/10.1093/aob/mct170
Pirie MD, Kandziora M, Nürk NM, Le Maitre NC, de Kuppler AM, Gehrke B, Oliver EG, Bellstedt DU (2019) Leaps and bounds: geographical and ecological distance constrained the colonisation of the Afrotemperate by Erica. BMC Evol Biol 19:222. https://doi.org/10.1186/s12862-019-1545-6
Pirie MD, Oliver EGH, de Kuppler AM, Gehrke B, Le Maitre NC, Kandziora M, Bellstedt DU (2016) The biodiversity hotspot as evolutionary hot-bed: spectacular radiation of Erica in the Cape Floristic Region. BMC Evol Biol 16:190. https://doi.org/10.1186/s12862-016-0764-3
Popp M, Gizaw A, Nemomissa S, Suda J, Brochmann C (2008) Colonization and diversification in the African “sky islands” by Eurasian Lychnis L. (Caryophyllaceae). J Biogeogr 35:1016–1029. https://doi.org/10.1111/j.1365-2699.2008.01902.x
Pouchon C, Lavergne S, Fernández Á, Alberti A, Aubert S, Mavárez J (2020) Phylogenetic signatures of ecological divergence and leapfrog adaptive radiation in Espeletia. Am J Bot 108:113–128. https://doi.org/10.1002/ajb2.1591
Qian H, Ricklefs RE (2016) Out of the tropical lowlands: latitude versus elevation. Trends Ecol Evol 31:738–741. https://doi.org/10.1016/j.tree.2016.07.012
Qian H, Ricklefs RE, Thuiller W (2021) Evolutionary assembly of flowering plants into sky islands. Nat Ecol Evol. https://doi.org/10.1038/s41559-021-01423-1
Rice A, Šmarda P, Novosolov M, Drori M, Glick L, Sabath N, Meiri S, Belmaker J, Mayrose I (2019) The global biogeography of polyploid plants. Nat Ecol Evol 3:265–273. https://doi.org/10.1038/s41559-018-0787-9
Richard A (1847–1851) Tentamen Florae Abyssinicae. Paris: Arthus Bertrand (Reprinted by Uppsala University, 1982).
Ring U, Albrecht C, Schrenk F (2018) The east African rift system: tectonics, climate and biodiversity. In: Hoorn C, Perrigo A, Antonelli A (Eds.) Mountains, climate and biodiversity. Wiley Blackwell, pp. 391–406.
Scheunert A, Fleischmann A, Olano-Marín C, Bräuchler C, Heubl G (2012) Phylogeny of tribe Rhinantheae (Orobanchaceae) with a focus on biogeography, cytology and re-examination of generic concepts. Taxon 61:1269–1285. https://doi.org/10.1002/tax.616008
Schüler L, Hemp A, Zech W, Behling H (2012) Vegetation, climate and fire-dynamics in East Africa inferred from the Maundi crater pollen record from Mt Kilimanjaro during the last glacial–interglacial cycle. Quat Sci Rev 39:1–13. https://doi.org/10.1016/j.quascirev.2012.02.003
Sepulchre P, Ramstein G, Fluteau F, Schuster M, Tiercelin JJ, Brunet M (2006) Tectonic uplift and eastern Africa aridification. Science 313:1419–1423. https://doi.org/10.1126/science.1129158
Sgorbati S, Labra M, Grugni E, Barcaccia G, Galasso G, Boni U, Mucciarelli M, Citterio S, Benavides Iramátegui A, Venero Gonzales L (2004) A survey of genetic diversity and reproductive biology of Puya raimondii (Bromeliaceae), the endangered queen of the Andes. Plant Biol 6:222–230. https://doi.org/10.1055/s-2004-817802
Sklenář P, Hedberg I, Cleef AM (2014) Island biogeography of tropical alpine floras. J Biogeogr 41:287–297. https://doi.org/10.1111/jbi.12212
Smith AP, Young TP (1987) Tropical Alpine plant ecology. Annu Rev Ecol Evol Syst 18:137–158. https://doi.org/10.1146/annurev.es.18.110187.001033
Smith J (1986) Origins and history of the Malesian high mountain flora. In: Vuilleumier, F, Monastero, M (Eds.) High altitude tropical biogeography. Oxford University Press, pp. 469–477.
Starr JR, Ford BA (2009) Phylogeny and evolution in Cariceae (Cyperaceae): current knowledge and future directions. Bot Rev 75:110–137. https://doi.org/10.1007/s12229-008-9020-x
Taberlet P, Zimmermann NE, Englisch T, Tribsch A, Holderegger R, Alvarez N, Niklfeld H, Coldea G, Mirek Z, Moilanen A (2012) Genetic diversity in widespread species is not congruent with species richness in alpine plant communities. Ecol Lett 15:1439–1448. https://doi.org/10.1111/ele.12004
Tolley KA, Tilbury CR, Measey GJ, Menegon M, Branch WR, Matthee CA (2011) Ancient forest fragmentation or recent radiation? Testing refugial speciation models in chameleons within an African biodiversity hotspot. J Biogeogr 38:1–13. https://doi.org/10.1111/j.1365-2699.2011.02529.x
Trauth MH, Maslin MA, Deino A, Strecker MR (2005) Late cenozoic moisture history of East Africa. Science 309:2051–2053. https://doi.org/10.1126/science.1112964
Tusiime FM, Gizaw A, Gussarova G, Nemomissa S, Popp M, Masao CA, Wondimu T, Abdi AA, Mirré V, Muwanika V, Eilu G, Brochmann C (2020) Afro-alpine flagships revisited: Parallel adaptation, intermountain admixture and shallow genetic structuring in the giant senecios (Dendrosenecio). PLoS ONE 15:e0228979. https://doi.org/10.1371/journal.pone.0228979
Tusiime FM, Gizaw A, Wondimu T, Masao CA, Abdi AA, Muwanika V, Travnicek P, Nemomissa S, Popp M, Eilu G, Brochmann C, Pimentel M (2017) Sweet vernal grasses (Anthoxanthum) colonized African mountains along two fronts in the Late Pliocene, followed by secondary contact, polyploidization and local extinction in the Pleistocene. Mol Ecol 26:3513–3532. https://doi.org/10.1111/mec.14136
Umer M, Lamb HF, Bonnefille R, Lezine AM, Tiercelin JJ, Gibert E, Cazet JP, J. W, (2007) Late pleistocene and holocene vegetation history of the Bale mountains, Ethiopia. Quat Sci Rev 26:2229–2246. https://doi.org/10.1016/j.quascirev.2007.05.004
Van der Hammen T, Cleef AM (1986) Development of the high Andean páramo flora and vegetation. In: Vuilleumier, F, Monastero, M (Eds.) High altitude tropical biogeography. Oxford University Press, pp. 153–201.
Vásquez DL, Balslev H, Hansen MM, Sklenář P, Romoleroux K (2016) Low genetic variation and high differentiation across sky island populations of Lupinus alopecuroides (Fabaceae) in the northern Andes. Alp Bot 126:135–142. https://doi.org/10.1007/s00035-016-0165-7
Vellend M, Lajoie G, Bourret A, Múrria C, Kembel SW, Garant D (2014) Drawing ecological inferences from coincident patterns of population-and community-level biodiversity. Mol Ecol 23:2890–2901. https://doi.org/10.1111/mec.12756
Voje KL, Hemp C, Flagstad O, Saetre GP, Stenseth NC (2009) Climatic change as an engine for speciation in flightless Orthoptera species inhabiting African mountains. Mol Ecol 18:93–108. https://doi.org/10.1111/j.1365-294X.2008.04002.x
Walter GM, Abbott RJ, Brennan AC, Bridle JR, Chapman M, Clark J, Filatov D, Nevado B, Ortiz-Barrientos D, Hiscock SJ (2020) Senecio as a model system for integrating studies of genotype, phenotype and fitness. New Phytol 226:326–344. https://doi.org/10.1111/nph.16434
White F (1983) The vegetation of Africa, a descriptive memoir to accompany the UNESCO. AETFAT/UNSO Vegetation Map of Africa 20:1–356
Whittaker RJ, Fernández-Palacios JM, Matthews TJ, Borregaard MK, Triantis KA (2017) Island biogeography: Taking the long view of nature’s laboratories. Science. https://doi.org/10.1126/science.aam8326
Wondimu T, Gizaw A, Tusiime FM, Masao CA, Abdi AA, Gussarova G, Popp M, Nemomissa S, Brochmann C (2014) Crossing barriers in an extremely fragmented system: two case studies in the afro-alpine sky island flora. Plant Syst Evol 300:415–430. https://doi.org/10.1007/s00606-013-0892-9
Wondimu T, Gizaw A, Tusiime FM, Masao CA, Abdi AA, Hou Y, Nemomissa S, Brochmann C (2017) Genetic structuring, dispersal and taxonomy of the high-alpine populations of the Geranium arabicum/kilimandscharicum complex in tropical eastern Africa. PLoS ONE 12:e0178208. https://doi.org/10.1371/journal.pone.0178208
Zech M (2006) Evidence for Late Pleistocene climate changes from buried soils on the southern slopes of Mt. Kilimanjaro. Tanzania Palaeogeogr Palaeoclimatol Palaeoecol 242:303–312. https://doi.org/10.1016/j.palaeo.2006.06.008
Acknowledgements
Much of the work synthesized here was carried out in two large projects including Ethiopia, Uganda, Tanzania, Kenya and Norway, funded by the Norwegian Programme for Development, Research and Higher Education (NUFU) to Christian Brochmann and Sileshi Nemomissa: the AFROALP project (54/2003—Afroalpine ‘islands’ as natural laboratories: dynamics and units of plant biodiversity), and the AFROALP-II project (2007/1058—Afroalpine sky islands: genetic versus taxonomic biodiversity, climate change, and conservation). We thank Sileshi Nemomissa, Geoffrey Mwachala, and Pantaleo Munishi for contributing to these projects, and the students for their huge efforts during the many field expeditions to these fantastic mountains and for their enthusiasm during the following years devoted to data analyses and discussions: Ahmed A. Abdi, Adane Assefa, Biructawit Bekele, Mulugeta Kebede, Catherine A. Masao, Felly M. Tusiime, and Tigist Wondimu. CB, AG, DC and MP thank the other members of our research group for patient help with supervision and analyses, in particular Dorothee Ehrich, Galina Gussarova, Virginia Mirré and Manuel Pimentel; Peter Linder for support, inspiration, discussions, and comments on this manuscript; Inga Hedberg for discussions and for providing plant material and a huge box with books and reprints; and Pierre Taberlet and Nick Zimmermann for collaboration and support. BG acknowledges Peter Linder for the wonderful opportunity to work on the afroalpine, and Muthama Muasya for support and collaboration. We also thank two reviewers for helpful comments on the manuscript. The Ethiopian Wildlife Conservation Authority, Uganda National Council for Science and Technology, Uganda Wildlife Authority, Tanzanian Commission for Science and Technology, Tanzanian National Parks Authority, and National Museums of Kenya are acknowledged for permission to conduct fieldwork, and the staff at the ETH, O, EA, MHU, SUA and NHT herbaria for curation of our specimens. CB’s, AG’s and DC’s work with this review was funded by the Research Council of Norway (project 274607: SpeciationClock—How fast does the 'speciation clock' tick in selfing versus outcrossing lineages?).
Funding
Open access funding provided by University of Oslo (incl Oslo University Hospital). CB’s, AG’s and DC’s work with this review was funded by the Research Council of Norway (project 274607: SpeciationClock—How fast does the 'speciation clock' tick in selfing versus outcrossing lineages?).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest/competing interests
Not applicable.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Brochmann, C., Gizaw, A., Chala, D. et al. History and evolution of the afroalpine flora: in the footsteps of Olov Hedberg. Alp Botany 132, 65–87 (2022). https://doi.org/10.1007/s00035-021-00256-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00035-021-00256-9