Abstract
In this paper, we will revisit the model studied in Lou and Zhao (J Math Biol 62:543–568, 2011), where the model takes the form of a nonlocal and time-delayed reaction–diffusion model arising from the fixed incubation period. We consider the infection age to be a continuous variable but without the limitation of the fixed incubation period, leading to an age-space structured malaria model in a bounded domain. By performing the elementary analysis, we investigate the well-posedness of the model by proving the global existence of the solution, define the explicit formula of basic reproduction number when all parameters remain constant. By analyzing the characteristic equations and designing suitable Lyapunov functions, we also establish the threshold dynamics of the constant disease-free and positive equilibria. Our theoretical results are also validated by numerical simulations for 1-dimensional and 2-dimensional domains.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Human malaria, caused by the genus Plasmodium, belongs to a mosquito-borne disease. The main intermediary vector is female anopheles mosquitoes. Rapid spread and global distribution (especially in Africa, Asia and South America) of human malaria causes public health problems and kills over a million people a year [20]. Since the classical work of [19], a lot of mathematical models based on vector-borne diseases framework (see, e.g., [3, 7, 9, 13,14,15,16,17, 27, 30, 31]) have been devoted to investigating the temporal and spatial patterns of disease burden and control strategies, which provides useful insights into the malaria transmission dynamics. In recent years, more and more biologically factors affecting vector-borne diseases are incorporated into mathematical models, such as immunity and clinical death [1, 21], spatial heterogeneity [3, 14, 15, 27, 31], the mobility of human and mosquito populations, extrinsic incubation period (EIP), vector-bias mechanism and seasonality (see, e.g., [3, 9, 14, 15, 27, 30, 31]). Here, EIP is a time interval during which mosquitoes could not transmit the malaria parasite to humans, which varies from 10 to 14 days [12] and significantly affect the number of infected mosquitoes. Spatial heterogeneity reflects the distinct contact patterns in distinct geographic regions, demonstrating the diversity in habitats. It is widely accepted and well known that the environmental conditions vary spatially, affecting the biting patterns, so setting the disease transmission parameters depending the location variable is biologically reasonable. The reaction–diffusion model is one of the most common tool in describing the spatial evolution of an epidemic, generalizing the classical models [16, 17, 19].
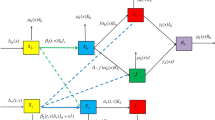
Let \(\Omega \subset \mathbb {R}^n\ (n\ge 1)\) be a bounded domain equipped with a smooth boundary \(\partial \Omega \). For \(x\in \Omega \), we introduce the Laplacian operator \(\Delta =\partial ^2/\partial x^2\) to represent the random mobility of human and mosquito populations in the domain. At time t and location x, we denote by \(S_m:=S_m(t,x)\), \(I_m:=I_m(t,x)\) and \(I_h:=I_h(t,x)\) the density of susceptible mosquitoes, infected mosquitoes and infected humans, whose diffusion rates are given by \(D_m\), \(D_m\) and \(D_h\), respectively. The model studied in [14] takes the following form,
for \(x\in \Omega , t>0\) and
Here, the density of total human population is assumed to be remained at H(x), \(H(x) \in C^2(\Omega , (0, \infty )) \cap C^1(\overline{\Omega },(0,\infty ))\) and H(x) satisfies
where \(K(x) \in C(\overline{\Omega }, (0,\infty ))\) is the carrying capacity dependent of location x and \(d_h\) the birth rate of humans, so the density of susceptible humans is given by \(H(x)-I_h\); The force of infection for human and mosquito populations is, respectively, characterized by \(\frac{b\beta (x)}{H(x)}S_mI_h\) and \(\frac{c\beta (x)}{H(x)}(H(x)-I_h)I_m\); \(\mu (x)\) and \(\beta (x)\), respectively, depict the space-dependent recruitment rate of adult female mosquitoes emerged from larval and biting rate; \(d_m\) and \(d_h\), respectively, stand for the natural death rate of mosquito and human populations; b and c describe the transmission probabilities per bite from infected humans to susceptible female mosquitoes and from infected female mosquitoes to susceptible humans; \(\rho \) is the recovery rate of humans; \(\Gamma (D_m\tau , x, y)\) is the Green function with respect to Laplace operator \(D_m\Delta \) subject to (1.2); \(\tau \) is a positive constant representing the fixed incubation period; \(\frac{\partial }{\partial n}\) denotes the differentiation along the outward normal n to \(\partial \Omega \). The basic assumptions on the parameters are as follows: \(D_m, D_h, d_m, \rho , b, c \in (0,\infty )\); \(\beta , \mu \in C(\overline{\Omega }, [0, \infty ) )\); \(\beta _1 \in L^\infty (\mathbb {R}_+, [0, \infty ))\).
The main feature of (1.1) is the nonlocal and time-delayed term appeared in \(I_m\) equation, which is obtained by the assumption that the incubation period is fixed at \(\tau >0\) and the standard method on characterizing age structured population with spatial diffusion [10]. Let \(i_m:=i_m(t,a,x)\) the density of the mosquitoes with infection age a at time t and location x, and \(i_m(t,0,x)=\frac{b\beta (x)}{H(x)}S_mI_h\) be the newly infected mosquitoes, which comes from the contact of susceptible mosquitoes and infectious humans. Then, \(i_m(t,a,x)\) fulfills
By the integration along characteristics, the nonlocal and time-delayed term in (1.1) is given by \(i_m(t,\tau ,x)\).
Unlike in [14] where spatial movements in EIP will cause nonlocal infection, here we plan to ignore the fixed incubation period and view the infection age as a continuous variable. In this paper, adopting the same notations used in [14], we directly investigate the following malaria model with age and space structure:
where the initial condition for (1.5) is given by, for all \(x \in \overline{\Omega }\) and \(a \ge 0\),
where \(\phi _1,\phi _3 \in C_+(\Omega )\) and \(\phi _2 \in L^1_+(\mathbb {R}_+, C(\Omega ))\). The boundary condition for (1.5) is the homogeneous Neumann condition, that is, for all \(x \in \partial \Omega \), \(t>0\) and \(a>0\),
We also note that some studies on spatial Zika models [9, 15, 27] could be viewed as an extension of the classical models in [16, 17, 19]. Specifically, in a recent paper [33], the authors implemented the global attractivity of a positive constant equilibrium of model (1.1) in a homogeneous case by designing a suitable Lyapunov functional, where the same problem was partially solved in [14], but requiring a sufficient condition through the fluctuation method. Very recently, Wang and Wang in [28] attempted to solve the global threshold dynamics of the problem (1.5)–(1.7) with mass-action mechanism and a stabilized density of susceptible humans H(x), which is not altered by the epidemics as in [15].
Our main goal of this paper is to provide a rigorous analysis of (1.5), where (1.5) can be viewed as the one for the generalization version of model (1.1). Here, we use an age structured population with spatial diffusion reflecting the diffusion of the latent individuals. Following the main idea in [3, 14] but using different analysis method, we address in Sect. 3, the basic questions on the existence, uniqueness and positivity of solutions to problem (1.5)–(1.7). We first treat the local existence of solution on \([0,T] \times \overline{\Omega }\) for small \(T>0\), where the method is different to that of [5, 6, 28, 29, 32]. The main reason is that we cannot construct a fixed point problem with one equation. To overcome this issue, we construct a fixed point problem with vector-valued functions. We also confirm that the solution never blows up in finite time and globally exists in a positive invariant set \(\mathcal {D}\) for all \(t > 0\). In Sect. 4, we derive the next-generation operator aiming to define the basic reproduction number \(\Re _0\) through renewal equations. In general, \(\Re _0\) cannot be directly calculated. However, in a spatially homogeneous case, the next-generation operator is compact. Thus, the Krein–Rutman theorem can be directly applied to get the explicit formula of \(\Re _0\). Section 4 is devoted to investigating the local and global dynamics of the disease-free and positive steady states in a spatially homogeneous case. It should be highlighted here that it is not easy work to design suitable Lyapunov functions. The main results obtained in Sect. 4 are validated by numerical simulations in Sect. 5 for 1-dimensional and 2-dimensional domain.
2 Preliminaries
Throughout of the paper, for ease of notations, we set
where \(f \in \{ \mu , \beta , H, \Lambda \}\).
Let \(\mathbb {Y}:= C(\overline{\Omega },\mathbb {R})\) and \(\mathbb {X}:=L^1(\mathbb {R}_+,\mathbb {Y})\) equipped with norms
, respectively. Denote the positive cones of \(\mathbb {X}\) and \(\mathbb {Y}\) by \(\mathbb {X}_+\) and \(\mathbb {Y}_+\), respectively. It is a classical fact that the diffusion operators \(D_m \Delta \) and \(D_h\Delta \) with (1.7) generate the strongly continuous semigroups \(\{ T_i(t)\}_{t\ge 0}:\mathbb {Y}_+ \rightarrow \mathbb {Y}_+\) (\(i=1,2\)) defined by, for \(t > 0\),
where \(\Gamma _i(t, x, y)\) (\(i=1,2\)) denote the associated Green functions. Note that, for any \(\varphi \in \mathbb {Y}_+\), \(i=1,2\) and \(t>0\),
because \(\int \limits _\Omega \Gamma _i(t,x,y)\textrm{d}y = 1\).
Let \(\mathbb {\overline{X}}=\mathbb {Y}\times \mathbb {X}\times \mathbb {Y}\) and \(\mathbb {\overline{X}}_+=\mathbb {Y}_+\times \mathbb {X}_+\times \mathbb {Y}_+\), equipped with norm
The state space for our system is as follows:
Our main result of this section reads as follows.
Theorem 2.1
There exists a solution semiflow \(\{ \Phi (t) \}_{t\ge 0}: \overline{\mathbb {X}}_+ \rightarrow \overline{\mathbb {X}}_+\) such that, for any \(\phi :=(\phi _1, \phi _2, \phi _3) \in \mathcal {D}\), \(\Phi (0)\phi = \phi \) and
gives a unique global solution to problem (1.5)–(1.7).
Before proving Theorem 2.1, we first introduce a lemma. For convenience, let us denote the newly infected mosquitoes by
By appealing to the method of characteristics, one can easily get that, for all \(x \in \Omega \),
where \(\Pi (a):=e^{-d_m a}\). Hence, we directly have
We now show the local existence of the solution.
Lemma 2.2
For each \(\phi \in \mathbb {\overline{X}}\), there exists a \(T>0\) such that problem (1.5)–(1.7) has a unique solution for all \(t \in (0,T)\).
Proof
Solving the equations of \(S_m\) and \(I_h\) in (1.5), we directly obtain: for \(t>0\),
where
and
For \(T>0\), we set
Let \(\mathcal {F}\) be a nonlinear operator defined on \(\mathbb {W}_T\) to itself,
Next, we show that \(\mathcal {F}\) has a fixed point on \(\mathbb {W}_T\), i.e., (1.5)–(1.7) has a unique solution on \([0,T] \times \overline{\Omega }\). For any \((S_m',I_h'), (S_m'',I_h'') \in \mathbb {W}_T\), we have
Hence, by virtue of (2.1), we obtain
where
Note that for any \(0<T_*<T\), we can regard \((S_m',I_h'), (S_m'',I_h'')\) as functions in \(\mathbb {W}_{T_*}\), and
and thus, \(h_1(T_*) \rightarrow 0\) as \(T_* \rightarrow + 0\). Hence, we let T being sufficiently small that \(h_1(T)<1\) (regarding \(T_*\) such that \(h(T_*)<1\) as a new T). Similarly, we obtain
and hence,
where
Similar to the case of \(h_1\), we let T being sufficiently small that \(h_2(T)<1\). Consequently, we obtain
As \(\max (h_1(T),h_2(T)) < 1\), the operator \(\mathcal {F}\) is a strict contraction in \(\mathbb {W}_T\). Consequently, \(\mathcal {F}\) has a unique fixed point in \(\mathbb {W}_T\). Hence, the local existence of \(S_m\) and \(I_h\) follows. The local existence of \(i_m\) then follows from (2.2) and (2.3). The regularity of the solution directly follows because the right-hand sides of (2.4) and (2.5) are continuously differentiable with respect to t and twice continuously differentiable with respect to x by virtue of the Green functions in \(\{ T_i(t) \}_{t\ge 0}\), \(i=1,2\). This proves Lemma 2.2. \(\square \)
Using Lemma 2.2, we continue to show Theorem 2.1.
Proof of Theorem 2.1
Let \(\phi =(\phi _1,\phi _2,\phi _3) \in \mathcal {D}\) and \(\tilde{T} \in (0,T)\). We first show the positivity of \(S_m\) on \((0,\tilde{T}) \times \overline{\Omega }\). Clearly, for \(x \in \Omega , \ t \in (0,\tilde{T})\),
As \(b\beta (x)I_h/H(x)+d_m\) is bounded and continuous on \((0,\tilde{T}) \times \overline{\Omega }\), a standard result for PDEs ensures that \(S_m>0\) for all \((t,x) \in (0,\tilde{T}) \times \overline{\Omega }\).
We next show that, for all \((t,x) \in (0, \tilde{T}) \times \overline{\Omega }\),
One can then easily see from the maximum principle that the last inequality in (2.7) holds. In addition, we have
Hence, similar to the above, one can easily see that \(M(t,x) > 0\) for all \((t,x) \in (0,\tilde{T}) \times \overline{\Omega }\).
We then show that \(I_h < H(x)\) for all \((t,x) \in (0,\tilde{T}) \times \overline{\Omega }\). Let \(Y_h:= H-I_h\). It then follows from (1.5)–(1.7) and (1.3) that
and
Similar to the above, as \(c\beta (x)\overline{\beta }_1\int \limits _0^\infty i_m \textrm{d}a/H(x) + d_h + \rho \) is bounded and continuous on \((0,\tilde{T}) \times \overline{\Omega }\), the standard result for PDEs yields that \(Y_h > 0\), \((t,x) \in (0,\tilde{T}) \times \overline{\Omega }\). We then directly have \(I_h < H(x)\) for all \((t,x) \in (0,\tilde{T}) \times \overline{\Omega }\).
We continue to prove that \(I_h \ge 0\) for all \((t,x) \in (0,\tilde{T}) \times \overline{\Omega }\). The abstract equation (2.5) in \(\mathbb {Y}\) can be rewritten as follows: for \(t \in (0,\tilde{T})\),
where \(\mathbb {F}_2\) is given as in (2.6) and
By (2.2), we get
This is a renewal equation and the solution can be written as \(\mathcal {B}=\sum _{n=0}^\infty \mathcal {B}_n\), where
Since \(S_m\) and \(H-I_h\) are positive, one can see that \(\mathcal {B}_n\) is nonnegative for all \(n \ge 0\). Hence, \(\mathcal {B}=\sum _{n=0}^\infty \mathcal {B}_n\) is also nonnegative. From (2.8), one knows that \(I_h\ge 0\) for all \((t,x) \in (0,\tilde{T}) \times \overline{\Omega }\). In addition, the nonnegativitiy of \(i_m\) also follows from (2.3).
In conclusion, the solution remains in the bounded set \(\mathcal {D}\) for all \(t \in (0,\tilde{T})\), that is, \(\mathcal {D}\) is positively invariant for system (1.5)–(1.7). Thus, the solution never blows up in finite time and globally exists in \(\mathcal {D}\) for all \(t > 0\). The existence of the solution semiflow \(\{ \Phi (t) \}_{t\ge 0}\) is a simple consequence. This proves Theorem 2.1. \(\square \)
3 The basic reproduction number
The disease-free equilibrium of (1.5) with boundary condition (1.7) can be written as \(E_0:= (S^0_m(x),0,0) \in \mathcal {D}\), where \(S_m^0(x)\) satisfies
More precisely, using the Green function \(\Gamma _1\), we can obtain the following explicit formulation of \(S_m^0(x)\):
Note that, if \(\mu (x) \equiv \mu \), then \(S_m^0 \equiv \mu /d_m\).
By appealing to the standard procedures as those in [8, 25], let us define the basic reproduction number \(\Re _0\) of (1.5)–(1.7). The linearized system of (1.5)–(1.7) around \(E_0\) is given by
for \(x\in \Omega , \ t> 0, \ a> 0\) and
By integrating the equations of \(I_h\) and \(i_m\) in (3.1), we obtain the following abstract equations in \(\mathbb {Y}\):
and
Hence, we get the following abstract equation in \(\mathbb {Y}\): for \(t>0\),
where
Hence, the generational expression \(\widetilde{\mathcal {B}} = \sum _{n=0}^\infty \widetilde{\mathcal {B}}_n\) can be obtained, where
Note that \(\widetilde{\mathcal {B}}_n\) denotes the newly infected population in the n-th generation. Let \(\widehat{\mathcal {B}}_n:= \int \limits _0^\infty \widetilde{\mathcal {B}}_n(t,\cdot )\textrm{d}t\). We then have, by changing the order of integration,
Thus, the next-generation operator \(\mathcal {K}:\mathbb {Y}_+ \rightarrow \mathbb {Y}_+\) can be defined by
More precisely, for \(\psi \in \mathbb {Y}_+\) and \(x \in \overline{\Omega }\),
One can easily see that \(\mathcal {K}\) is strictly positive, i.e., if \(\psi \in \mathbb {Y}_+ \setminus \{ 0 \}\), then \(\mathcal {K}\psi (x)>0\) for all \(x \in \overline{\Omega }\). According to [8, 25], \(\Re _0:= r(\mathcal {K})\), the spectral radius of \(\mathcal {K}\). In general, \(\Re _0\) cannot be explicitly calculated. However, in a spatially homogeneous case that
we can get that \(S_m^0(x) \equiv \mu /d_m\) and \(\mathcal {K}\) is compact. The Krein–Rutman theorem [2, Theorem 3.2] guarantees that \(\Re _0\) is the only positive eigenvalue of \(\mathcal {K}\) associated with a positive eigenvector. More precisely, we obtain
4 Dynamical analysis in the spatially homogeneous case
In the spatially homogeneous case, problem (1.5)–(1.7) reduces to
with the same initial and boundary conditions (1.6) and (1.7).
Corollary 4.1
The solution semiflow \(\{ \Phi \}_{t\ge 0}\) of (4.1) admits a global attractor in \(\mathcal {D}\).
Proof
With the help of Theorem 2.1, one knows that \(\Phi \) is point dissipative and eventually bounded on bounded sets of \(\mathcal {D}\). Moreover, one can easily confirm that \(\Phi \) is asymptotically smooth in the spatially homogeneous case by using the method as in [18]. An application of [22, Theorem 2.33] confirms that (4.1) admits a global attractor. This proves Corollary 4.1. \(\square \)
System (4.1) has constant equilibria which are solutions to the following equations:
Obviously, there exists a constant disease-free equilibrium \(\tilde{E}_0:= (S_m^0,0,0) \in \mathcal {D}\), where \(S_m^0=\mu /d_m\). Moreover, rearranging (4.2), we have
where \(K:= \int \limits _0^\infty \beta _1(a) \Pi (a) \textrm{d}a\). By the equation of \(i_m(0)\) in (4.2), we have
Thus, we have the following proposition on the existence of constant equilibrium.
Proposition 4.2
Let \([\Re _0]\) is defined in (3.4). If \([\Re _0] > 1\), then (4.1) admits a constant equilibrium \(E^*=(S^*_m, i^*_m(a), I_h^*) \in \mathcal {D}\), where
and
4.1 Local asymptotic stability of equilibria
We shall prove that both \(\tilde{E}_0\) and \(E^*\) are locally asymptotically stable (LAS).
Theorem 4.3
Let \([\Re _0]\) be defined by (3.4).
-
(i)
If \([\Re _0]<1\), then \(\tilde{E}_0\) is LAS;
-
(ii)
If \([\Re _0]>1\), then \(E^*\) is LAS.
Proof
We first prove (i). The linearized system of (4.1) around \(\tilde{E}_0\) is as follows:
with boundary condition (1.7). Let \(\mu _j \ (j= 1,2,\ldots )\) be the eigenvalues of linear operator \(-\Delta \) on \(\Omega \) with homogeneous Neumann boundary condition corresponding to the eigenvectors \(v_j\in C^2(\Omega ) \cap C^1(\overline{\Omega })\):
From a well-known fact, we can assume that \(0=\mu _0<\mu _1<\mu _2< \cdots \). Substituting \((S_m, i_m, I_h)=e^{\eta t} v_i(x) (u_1, u_2 (a), u_3) \ (\eta \in \mathbb {C})\) into (4.3) and dividing each side by \(e^{\eta t}v_i(x)\), we have
It is easy checked from the second and fourth equations of (4.4) that
where \(\tilde{\Pi }(a)=e^{-D_m\mu _ia}\Pi (a)\). Rewriting (4.4) in terms of \((u_1,u_3)\), we obtain the following characteristic equation:
where \(\mathcal {K(\eta )}=\eta +D_h\mu _i+d_h+\rho -\frac{bc\beta ^{2}}{H}S_{m}^{0}\int \limits _0^\infty \beta _1(a)e^{-\eta a}\tilde{\Pi }(a)\textrm{d}a\). To show that \(\tilde{E}_0\) is LAS, we suppose on the contrary that \(\eta = m + ni \ (m,n \in \mathbb {R}, \ i^2=-1)\) with \(m \ge 0\). We then see that \(\eta +D_m\mu _i+d_m\ne 0\), and thus, we can only pay attention to the roots of \(\mathcal {K(\eta )}=0\). This equation can be rewritten as
Taking the absolute value of both sides, we have
which leads to a contradiction with \([\Re _0]<1\). Hence, \(m \le 0\). This proves (i).
We next proceed to prove (ii). The linearized system of (4.1) around \(E^{*}\) is as follows:
with boundary condition (1.7). Substituting \((S_m, i_m, I_h)=e^{\eta t} v_i(x) (u_1, u_2(a), u_3)\) into (4.5) and dividing each side by \(e^{\eta t}v_i(x)\), we have
It is easily checked that
Hence, rewriting (4.6) in terms of \((u_1,u_3)\), we obtain the following characteristic equation:
where \(P=\int \limits _0^\infty \beta _1(a)e^{-\eta a}\tilde{\Pi }(a)\textrm{d}a\) and \(Q=\int \limits _0^\infty \beta _1(a)i_{m}^{*}(a)\textrm{d}a\). Rearranging this equation, we obtain
To show that \(E^*\) is LAS, we suppose on the contrary that \(\eta = m + ni \ (m,n \in \mathbb {R}, \ i^2=-1)\) with \(m \ge 0\). By taking the absolute value of both sides of (4.7), we obtain
It then follows from the equilibrium equations that
a contradiction. Hence, \(m \le 0\). This proves (ii). \(\square \)
4.2 Global dynamics
We shall investigate the threshold dynamics of (4.1) in terms of \([\Re _0]\), that is, both \(\tilde{E}_0\) and \(E^*\) are globally attractive. This together with the related results in above subsection tells us that both \(\tilde{E}_0\) and \(E^*\) are globally asymptotically stable (GAS).
Theorem 4.4
Suppose that \([\Re _0]<1\). Then, \(\tilde{E}_0\) is GAS in \(\mathcal {D}\).
Proof
Note that, in \(\mathcal {D}\), \(S_m\le \mu /d_m = S_m^0\) for all \(t>0\) and \(x \in \overline{\Omega }\). Hence, an application of the comparison principle gives \(0\le i_m \le \overline{i}_m\) and \(0\le I_h \le \overline{I}_h\), where \((\overline{i}_m, \overline{I}_h)\) is the solution to the following auxiliary system:
for \(x\in \Omega ,\ t>0,\ a > 0\) and
It then suffices to show that \((\overline{i}_m, \overline{I}_h)\) converges to (0, 0) as time goes to infinity, which implies that \((i_m,I_h)\) convereges to (0, 0), and thus, \(S_m\) converges to \(S_m^0\) as time goes to infinity.
Let
One can then easily check that
Let \(V(t):=V_1(t) + V_2(t)\) be a Lyapunov function, where
We then have that
and
Hence, the derivative of V gives
Consequently, \(\tilde{E}_0\) is globally attractive in \(\mathcal {D}\) when \([\Re _0] < 1\) (see, for instance, [26, Theorem 4.2]). Combined with the results in Theorem 4.3, one knows that \(\tilde{E}_0\) is GAS. This completes the proof of Theorem 4.4. \(\square \)
To define a Lyapunov function for \(E^*\) when \([\Re _0] > 1\), we need a uniform persistence result. The following estimation for \(S_m\) immediately follows.
Proposition 4.5
There exists an \(\epsilon _0>0\) such that, for any \(\phi \in \mathcal {D}\) and \(x\in \overline{\Omega }\),
Proof
By Theorem 2.1, and \(I_h \le H\), one can get that \(\frac{\partial S_m}{\partial t}\ge \ D_m\Delta S_m+\mu -( b\beta + d_m)S_m\). Again from the comparison principle, one can get that for any \(x\in \overline{\Omega }\),
This proves Proposition 4.5. \(\square \)
We next define the following subset of \(\mathcal {D}\):
Epidemiologically, \(\mathcal {D}_0\) is the set where the disease persists. The forthcoming lemma immediately follows.
Lemma 4.6
If \(\phi \in \mathcal {D}_0\), then \(I_h > 0\) for all \(t>0\) and \(x \in \overline{\Omega }\).
The following result indicates that \(\{ \tilde{E}_0 \}\) is a uniform weak repeller in \(\mathcal {D}\).
Lemma 4.7
If \([\Re _0] > 1\), then there exists an \(\epsilon _1>0\) such that, for any \(\phi \in \mathcal {D}_0\),
Proof
We proceed it indirectly and assume that for any \(\epsilon _1>0\), there exists a \(\phi \in \mathcal {D}_0\) that
This inequality implies that there exists a \(t_1>0\) such that, for any \(t > t_1\) and \(x \in \overline{\Omega }\),
Without loss of generality, taking \(\Phi (t_1)\phi \) be the new initial condition, we can assume that inequalities (4.10) hold for all \(t>0\) and \(x \in \overline{\Omega }\).
For simplicity, we write \(\mathcal {B}(t)=\mathcal {B}(S_m,I_h)(t)\). By (2.9), we obtain the following abstract inequality in \(\mathbb {Y}\):
For any \(\lambda > 0\), let \(\hat{\mathcal {B}}(\lambda ): = \int \limits _0^\infty e^{-\lambda t} \int \limits _\Omega \mathcal {B}(t,x) \textrm{d}x \textrm{d}t\). By Lemma 4.6 and (2.2), we can easily confirm that \(0< \hat{\mathcal {B}}(\lambda ) < +\infty \). Moreover, from (4.11), we have
where
One can easily see that \([\Re _{\epsilon _1,\lambda }] \rightarrow [\Re _0] > 1\) as \((\epsilon _1,\lambda )\rightarrow (0,0)\), which allow us to choose \(\epsilon _1>0\) and \(\lambda >0\) small enough such that \([\Re _{\epsilon _1,\lambda }] > 1\). We then have from (4.12) that \(\hat{\mathcal {B}}(\lambda ) > \hat{\mathcal {B}}(\lambda ) \), a contradiction. This proves Lemma 4.7. \(\square \)
Using Lemma 4.7, we now prove the following result.
Proposition 4.8
If \([\Re _0] > 1\), then there exists an \(\epsilon _2>0\) such that, for any \(\phi \in \mathcal {D}_0\) and \(x \in \overline{\Omega }\),
Before the proof, we prepare some notations:
-
\(\partial \mathcal {D}_0: = \mathcal {D} \setminus \mathcal {D}_0 = \left\{ \varphi = (\varphi _1, \varphi _2, \varphi _3) \in \mathcal {D}: \varphi _3 \equiv 0 \right\} \).
-
\(M_\partial := \{ \varphi \in \partial \mathcal {D}_0: \Phi (t)\varphi \in \partial \mathcal {D}_0 \ \mathrm {for \ all} \ t > 0 \}\).
-
\(\omega (\varphi ):= \cap _{t\ge 0}\overline{\cup _{s\ge t} \Phi (s)\varphi }\): the omega limit set.
-
\(\delta (\varphi ):= \inf _{x\in \Omega } \varphi _3(x)\), \(\rho :\mathcal {D}\rightarrow \mathbb {R}_+\): a generalized distance function.
-
\(W^s(\tilde{E}_0):= \{ \varphi \in \mathcal {D}: \lim _{t\rightarrow \infty } \Vert \Phi (t)\varphi - \tilde{E}_0 \Vert _{\overline{\mathbb {X}}} = 0 \}\): the stable set of \(\tilde{E}_0\).
Proof
One can easily confirm that
-
1.
\(\cup _{\varphi \in M_\partial } \omega (\varphi ) = \{ \tilde{E}_0 \}\).
-
2.
No subset of \(\{ \tilde{E}_0\}\) forms a cycle in \(\partial \mathcal {D}_0\).
-
3.
\(\{ \tilde{E}_0 \}\) is isolated in \(\mathcal {D}\).
-
4.
\(W^s(\tilde{E}_0) \cap \delta ^{-1}(0,\infty ) = \emptyset \).
Moreover, by Corollary 4.1, \(\Phi \) has a global attractor in \(\mathcal {D}\). Thus, conditions in [23, Theorem 3] are satisfied and there exists an \(\epsilon _2>0\) such that
where \(\mathcal {L}\) is an arbitrary compact chain transitive set in \(\mathcal {D} \setminus \{ \tilde{E}_0 \}\). Hence, for any \(\phi \in \mathcal {D}_0\) and \(x \in \overline{\Omega }\), (4.13) holds. This proves Proposition 4.8. \(\square \)
By Propositions 4.5 and 4.8, there exists \(c_i>0, i=1,2\), such that, for any total trajectory in a persistence attractor (see, e.g., [18, Theorem 8.3]), the following inequalities hold:
Thus, for any total trajectory in a persistence attractor, the following functions are finite for all \(t \in \mathbb {R}\):
where \(g(u):= u-1-\ln u\) and
Clearly, \(g(u) > 0\) for each \(u \in (0,\infty ) \setminus \{1 \}\) and \(g(1)=0\). Using a Lyapunov function \(W:=\kappa W_1+W_2+W_3\) with \(\kappa >0\) to be determined below, we can obtain the following result.
Theorem 4.9
Suppose that \([\Re _0] > 1\). Then, \(E^*\) is GAS in \(\mathcal {D}_0\).
Proof
By appealing to [18, Theorem 9.5], we consider a total trajectory in the persistence attractor. Then, \(W(t)=\kappa W_1(t)+W_2(t)+W_3(t)\) is finite for all \(t \in \mathbb {R}\). Direct calculation gives
and
\(\Omega =(0,1)\). The time evolution of susceptible mosquitoes \(S_m(t,x)\), infected mosquitoes with \(I(t,x)=\int \limits _0^{\infty }i_m(t,a,x)\textrm{d}a\)) and infected humans \(I_h(t,x)\) of system (4.1) with (5.1), \(\beta _ =0.026\) and \(\Omega =(0,1)\). The initial data is \(\phi _1(x)=100,\ \phi _2(a,x)=e^{-d_m*a} (x-0.3)(0.7-x)\) and \(\phi _3(x)=0.\)
The time evolution of susceptible mosquitoes \(S_m(t,x)\), infected mosquitoes with \(I(t,x)=\int \limits \limits _0^{\infty }i_m(t,a,x)\textrm{d}a\)) and infected humans \(I_h(t,x)\) of system (4.1) with (5.1), \(\beta =0.036\) and \(\Omega =(0,1)\). The initial data is \(\phi _1(x)=100,\ \phi _2(a,x)=e^{-d_m*a} (x-0.3)(0.7-x)\) and \(\phi _3(x)=0.\)
\(t=280\). The time evolution of susceptible mosquitoes \(S_m(t,x)\), infected mosquitoes with \(I(t,x)=\int \limits _0^{\infty }i_m(t,a,x)\textrm{d}a\)) and infected humans \(I_h(t,x)\) of system (4.1) with (5.1), \(\beta =0.26\) and \(\Omega = (0, 1)\times (0,1)\). The initial data is \(\phi _1(x,y)=100,\ \phi _2(a,x,y)=e^{-d_m*a}(x-0.3)(0.7-x)(y-0.3)(0.7-y)\) and \(\phi _3(x,y)=0.\)
\(t=350\). The time evolution of susceptible mosquitoes \(S_m(t,x)\), infected mosquitoes with \(I(t,x)=\int \limits _0^{\infty }i_m(t,a,x)\textrm{d}a\)) and infected humans \(I_h(t,x)\) of system (4.1) with (5.1), \(\beta =0.36\) and \(\Omega = (0, 1)\times (0,1)\). The initial data is \(\phi _1(x,y)=100,\ \phi _2(a,x,y)=e^{-d_m*a}(x-0.3)(0.7-x)(y-0.3)(0.7-y)\) and \(\phi _3(x,y)=0.\)
Here, note that
Thus, we have
Hence, letting \(\kappa := c\beta (1-I_h^*/H) K\), we obtain
One can easily see that \(W'(t)=0\) iff \((S_m,i_m,I_h)=E^*\). As in the proof of [18, Theorem 9.5], we see that the singleton \(\{E^*\}\) is indeed the persistence attractor. This gives the global attractivity of \(E^*\). Together with Theorem 4.3, one can get \(E^*\) is GAS. This proves Theorem 4.9. \(\square \)
5 Numerical simulations
5.1 Dynamical behaviors of system (4.1)
We perform numerical simulations to support the main results obtained in Sect. 4. Specifically, we shall carry out the simulations for 1-dimensional and 2-dimensional domain to validate Theorems 4.4 and 4.9, that is, both \(\tilde{E}_0\) and \(E^*\) are GAS.
For the case that \(\Omega =(0,1)\), we set the following parameters:
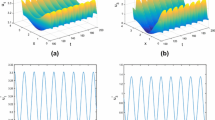
If we take \(\beta =0.26\), we can compute \([\Re _0] = 0.844669\). From Theorem 4.4, we know that \(\tilde{E}_0\) is GAS in \(\mathcal {D}\). Figure 1a, b and c illustrates that the density of susceptible mosquitoes will attain a positive level and infected mosquitoes and infected humans decay to zero. We also know from Fig. 1d that the spatial distribution of infected mosquitoes gradually enlarges with higher prevalence but decays to zero.
If we take \(\beta =0.36\) and the other parameters remain the same as in (5.1), then \([\Re _0] = 1.619366\). It is known from Theorem 4.9 that \(E^*\) is GAS in \(\mathcal {D}_0\). Figure 2a, b and c illustrates that the densities of susceptible mosquitoes, infected mosquitoes and infected humans will attain a positive level as time evolves. Figure 2d illustrates that the spatial distribution of infected mosquitoes gradually enlarge with higher prevalence.
For the case that \(\Omega = (0, 1)\times (0,1)\). We set the same parameters as in Fig. 1 and 2. Figure 3a illustrates that the density of susceptible mosquitoes will attain a positive level. We can see from Fig. 3b, c that the densities of infected mosquitoes and infected humans decay to zero. Figure 4 demonstrates the densities of susceptible mosquitoes, infected mosquitoes and infected humans will attain a positive level.
5.2 The influence of parameters on [\(\Re _0\)]
To analyze the effects of the parameter values on \([\Re _0]\), we perform sensitivity analysis to check the effects of the parameter values on [\(\Re _0\)] by Latin Hypercube Sampling and partial rank correlation coefficient (PRCC) method (see, for example, [4, 11]). Under the setting that \(\mu \), \(d_m\), \(\rho \) and \(\beta _1\) are changed concomitantly, we can observe the dependence of \([\Re _0]\) on parameters \(\mu \), \(d_m\), \(\rho \) and \(\beta _1\), respectively. Specifically, numeric plots in Fig. 5 indicate that \([\Re _0]\) is a monotonically increasing function with respect to \(\mu \) and \(\beta _1\), while \([\Re _0]\) is a monotonically decreasing function of \(d_m\) and \(\rho \), respectively. Figure 6 demonstrates that \([\Re _0]\) is more sensitive to \(\mu \) and \(\beta _1\).
We next investigate the influence of \(\beta _1(a)\) on \([\Re _0]\). As pointed in [24], the smaller the age of infection, the smaller transmission rate \(\beta _1(a)\) of the disease. The rate of infection increases along with the infectious age. When the age of infection is very large, the infection rate is reduced to zero due to the loss of infectivity. Therefore, we artificially select the following form of \(\beta _1\),
It can be observed from Fig. 7 that \([\Re _0]\) decreases monotonically as n increases.
Data Availability
The datasets generated and/or analyzed during the current study are not publicly available but are available from the authors on reasonable request.
References
Aron, J.L., May, R.M.: The Population Dynamics of Malaria. In: Anderson, R.M. (ed.) The Population Dynamics of Infectious Diseases: Theory and Applications, pp. 139–179. Chapman and Hall, London (1982)
Amann, H.: Fixed point equations and nonlinear eigenvalue problems in ordered Banach spaces. SIAM Rev. 18, 620–709 (1976)
Bai, Z., Peng, R., Zhao, X.-Q.: A reaction-diffusion malaria model with seasonality and incubation period. J. Math. Biol. 77, 201–228 (2018)
Blower, S.M., Dowlatabadi, H.: Sensitivity and uncertainty analysis of complex-models of disease transmission: an HIV model, as an example. Int. Stat. Rev. 62, 229–243 (1994)
Chekroun, A., Kuniya, T.: An infection age-space structured SIR epidemic model with Neumann boundary condition. Appl. Anal. 99, 1972–1985 (2020)
Chekroun, A., Kuniya, T.: Global threshold dynamics of aninfection age-structured SIR epidemic model with diffusion under the Dirichlet boundary condition. J. Differ. Equ. 269, 117–148 (2020)
Cantrell, R.S., Cosner, C.: Spatial Ecology via Reaction-Diffusion Equations. Mathematical and Computational Biology, John Wiley Sons Ltd, West Sussex (2003)
Diekmann, O., Heesterbeek, J.A.P., Metz, J.A.J.: On the definition and the computation of the basic reproduction ratio \(R_0\) in models for infectious diseases in heterogeneous populations. J. Math. Biol. 28, 365–382 (1990)
Fitzgibbon, W.E., Morgan, J.J., Webb, G.: An outbreak vector-host epidemic model with spatial structure: the 2015–2016 Zika outbreak in Rio De Janeiro. Theor. Biol. Med. Model. 14, 7 (2017)
Gourley, S.A., Wu, J.: Delayed non-local diffusive systems in biological invasion and disease spread. In: Nonlinear Dynamics and Evolution Equations. AMS, Providence (2006)
Hoare, A., Regan, D.P., Wilson, D.G.: Sampling and sensitivity analyses tools (SaSAT) for computational modelling. Theor. Biol. Med. Model. 5, 1–18 (2008)
Killeen, G.F., McKenzie, F.E., Foy, B.D., et al.: A simplified model for predicting malaria entomologic inoculation rates based on entomologic and parasitologic parameters relevant to control. Am. J. Trop. Med. Hyg. 62, 535–544 (2000)
Li, J., Zou, X.: Modeling spatial spread of infectious diseases with a fixed latent period in a spatially continuous domain. Bull. Math. Biol. 71, 2048–2079 (2009)
Lou, Y., Zhao, X.-Q.: A reaction-diffusion malaria model with incubation period in the vector population. J. Math. Biol. 62, 543–568 (2011)
Magal, P., Webb, G., Wu, Y.: On a vector-host epidemic model with spatial structure. Nonlinearity 31, 5589–5614 (2018)
Macdonald, G.: The analysis of equilibrium in malaria. Trop. Dis. Bull. 49, 813–829 (1952)
Macdonald, G.: The Epidemiology and Control of Malaria. Oxford University Press, London (1957)
McCluskey, C.C.: Global stability for an SEI epidemiological model with continuous age-structure in the exposed and infectious classes. Math. Biosci. Eng. 9, 819–841 (2012)
Ross, R.: The Prevention of Malaria, 2nd edn. Murray, London (1911)
Snow, R.W., Guerra, C.A., Noor, A.M., Myint, H.Y., Hay, S.I.: The global distribution of clinical episodes of Plasmodium falciparum malaria. Nature 434, 214–217 (2005)
Smith, D.L., Dushoff, J., McKenzie, F.E.: The risk of a mosquito-borne infection in a heterogeneous environment. PLoS Biol. 2, 1957–1964 (2004)
Smith, H.L., Thieme, H.R.: Dynamical Systems and Population Persistence. AMS, Providence (2011)
Smith, H.L., Zhao, X.-Q.: Robust persistence for semidynamical systems. Nonlinear Anal. TMA 47, 6169–6179 (2001)
Shi, Y., Zhao, H., Zhang, X.: Threshold dynamics of an age-space structure vector-borne disease model with multiple transmission pathways. Commun. Pure Appl. Anal. 22(5), 1477–1516 (2023)
Wang, W., Zhao, X.-Q.: Basic reproduction numbers for reaction-diffusion epidemic models. SIAM J. Appl. Dyn. Syst. 11, 1652–1673 (2012)
Walker, J.A.: Dynamical Systems and Evolution Equations: Theory and Applications. Plenum Press, New York (1980)
Wang, J., Chen, Y.: Threshold dynamics of a vector-borne disease model with spatial structure and vector-bias. Appl. Math. Lett. 100, 106052 (2020)
Wang, C., Wang, J.: Analysis of a malaria epidemic model with age structure and spatial diffusion. Z. Angew. Math. Phys. 72, 74 (2021)
Wang, J., Zhang, R., Gao, Y.: Global threshold dynamics of an infection age-space structured HIV infection model with Neumann boundary condition. J. Dyn. Differ. Equ. (2021). https://doi.org/10.1007/s10884-021-10086-2
Wang, X., Zhao, X.-Q.: A periodic vector-bias malaria model with incubation period. SIAM J. Appl. Math. 77, 181–201 (2017)
Xu, Z., Zhao, X.-Q.: A vector-bias malaria model with incubation period and diffusion. Discrete Contin. Dyn. Syst. Ser. B 17, 2615–2634 (2012)
Yang, J., Xu, R., Li, J.: Threshold dynamics of an age-space structured brucellosis disease model with Neumann boundary condition. Nonlinear Anal. RWA 50, 192–217 (2019)
Zhang, R., Wang, J.: On the global attractivity for a reaction-diffusion malaria model with incubation period in the vector population. J. Math. Biol. 84, 53 (2022)
Acknowledgements
The authors would like to thank the editor and the anonymous reviewers for his/her suggestions that have improved this paper.
Funding
Open access funding provided by Kobe University. J. Wang was funded by the National Natural Science Foundation of China (No. 12071115), the Heilongjiang Natural Science Funds for Distinguished Young Scholar (No. JQ2023A005), the Fundamental Research Funds for the Heilongjiang Education Department (no. 2021-KYYWF-0034), and the Heilongjiang Provincial Key Laboratory of the Theory and Computation of Complex Systems. T. Kuniya was funded by the Japan Society for the Promotion of Science (grant numbers 19K14594, 23K03214).
Author information
Authors and Affiliations
Contributions
All authors contributed equally.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, J., Cao, M. & Kuniya, T. Dynamical analysis of an age-space structured malaria epidemic model. Z. Angew. Math. Phys. 74, 214 (2023). https://doi.org/10.1007/s00033-023-02097-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00033-023-02097-4