Abstract
This paper aims to the investigation of the global threshold dynamics of an infection age-space structured HIV infection model. The model is formulated in a bounded domain involving two infection routes (virus-to-cell and cell-to-cell) and Neumann boundary conditions. We first transform the original model to a hybrid system containing two partial differential equations and a Volterra integral equation. By appealing to the theory of fixed point problem together with Picard sequences, the well-posedness of the model is shown by verifying that the solution exists globally and the solution is ultimately bounded. Under the Neumann boundary condition, we establish the explicit expression of the basic reproduction number. By analyzing the distribution of characteristic roots of the associated characteristic equation in terms of the basic reproduction number, we achieve the local asymptotic stability of the steady states. The global asymptotic stability of the steady states is established by the technique of Lyapunov functionals, respectively. Numerical simulations are performed to validate our theoretical results.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
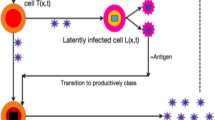
Inspired by the classical works of Ho et al. [17] and Perelson et al. [30], the dynamical properties of differential equations modelling the with-in host viral dynamics have obtained much attention (see, e.g., [6, 13, 19, 23, 24, 33, 39, 45, 49]). It has been widely recognized that some typical features of viral dynamics, such as, time delays, infection age structure, and spatial heterogeneity should be taken into account in studying with-in host dynamics, which may give us new insights into the interactions among uninfected target T cells, infected T cells, and free virus particles. Let T(t), u(t, a) and V(t) be the concentrations of uninfected target T cells, infected T cells of infection age a and the free virus particles at time t, respectively. Here the infection age is defined by the time since infection began and the infection-age structure is used to demonstrate the mechanisms that the death rate and virus production rate of infected T cells should be infection-age-dependent, denoted by \(\theta (a)\) and p(a), respectively. The following initial-boundary-value problem was studied in Nelson et al. [28],
where h and d are the constant recruitment rate and the natural death rate of uninfected cells; \(\beta _1\) is the infection rate; c is the clearance rate of virions. In [28], the local dynamics of the model was achieved for some special cases and the time to reach the peak viral level are illustrated by numerical simulations. To evaluate the roles of a distinct combination of therapies, Rong et al. [13, 33] took the model (1.1) as a basis and adapted it incorporating three different classes of drugs. The global dynamics of model (1.1) was completely solved in Huang et al. [19] by the technique of Lyapunov functionals. Model (1.1) also be used as a basic framework to investigate the dynamics of hepatitis B or C virus in Qesmi et al. [32].
As pointed in [3, 7, 31, 48], the variant infectivity in different ages may arise from the heterogeneous structure of the infected T cells. In recent years, more and more works have been devoted to investigating the effects of cell-to-cell infection routes in lymphoid tissues (via formation of virological synapses) on viral dynamics [11, 18, 37, 38]. The cell-to-cell infection routes have been recognized as important factors in virus spread (see, e.g., [6, 23, 39, 45, 49]). Lai and Zou [23] formulated the distributed delay differential equations, and investigated the global stability of equilibrium of the model. Wang et al. [40] further extended the model (1.1) to the following model:
where \(\beta _2\) and q(a) measure the infection rate and the infectivity between uninfected target T cells and infected T cells, respectively. The global threshold dynamics of (1.2) in terms of the basic reproduction number was achieved by solid analysis. Subsequently, by incorporating the logistic growth for target T cells, the oscillations via local Hopf bifurcation was studied in [45, 46, 49]. Shu et al. [39] gave the complete analysis on HIV infection model involving nonlinear target-cell dynamics and nonlinear incidences, and studied global dynamics in the aspect of global threshold dynamics and oscillations via global Hopf bifurcation. Cheng et al. [6] proposed an age-structured HIV infection model in the form of a two-compartment model and analyzed the global attractivity of the equilibria by the perturbation theory. Zhang and Liu [50] investigated the Hopf bifurcation of a delayed infection-age structured HIV infection model by appealing to the theory of integrated semigroup with the non-dense domain. Wu and Zhao [43] studied the effects of drug resistance by formulating an infection-age HIV model involving a drug-sensitive strain and a drug-sensitive strain, and revealed that the efficacy of antiretroviral drug treatments becomes weaker arising from the presence of cell-to-cell route.
However, ordinary differential equations modeling of viral infection assumed that the intracellular reaction occurs simultaneously. As argued in [12], the HIV spread and replication in lymphoid tissues are affected by the tissue architecture and composition. The results in [25] revealed that the dynamics of HIV in vivo may mainly be affected by different physiological environments, especially, in the early stage of infection.
Due to the complexity of the physiological environment and the tissue architectures of lymphoid tissues, the spatial aspects of the tissues should be taken into account on viral dynamics. Very recently, Ren et al. [34] considered a reaction-diffusion within-host HIV model in a heterogeneous environment. Let t and x be the time and location variables, respectively. We denote by T(t, x), \(T^*(t, x)\), and V(t, x) the densities of uninfected target T cells, infected T cells, and the free virus particles, associated with diffusion rates \(D_1(x)\), \(D_2(x)\), and \(D_3(x)\), respectively. The model formulated in [34] is the following form,
where \(\Omega \) is the spatial domain. \(\nu \) is the outward normal vector to \(\partial \Omega \). The space-dependent parameters h(x), d(x), \(\beta _i(x)\ (i=1,2)\), r(x), N(x) and c(x) (the biological meaning can be found in [34]) are strictly positive, and uniformly bounded functions on \(\overline{\Omega }\). In the bounded domain, the authors obtained the global threshold type result in terms of the basic reproduction number, while in the unbounded domain, the existence of traveling wave solutions and the minimum wave speed was established. Furthermore, the authors also found that the minimum wave speed and the asymptotic spreading speed are affected by the diffusion of cells and cell-to-cell infection route.
Recently, the infection age-space structured models have attracted wide attentions, which are spent on understanding the effects of the time since infection and the spatial heterogeneity on the transmission of infectious diseases. In 2009, Ducrot and Magal [8] studied the traveling wave problem for a diffusive SIR model with infection age. As a continuous study of [8], Ducrot and Magal [9] further considered the external supplies in the age-space structured SIR model, and they found that the model admits a traveling wave connecting the two different steady states. Until recently, Chekroun and Kuniya [4] proposed an infection age-space structured SIR model on a bounded domain. After reformulating the model into a hybrid system of one diffusive equation and one Volterra integral equation, the threshold-type results for the disease extinction and persistence in one-dimensional domain were studied. Later on, Yang et al. [47] made an attempt to extend the methods and ideas used in [4], and proposed a spatial spreading of brucellosis model in a continuous bounded domain. Some basic mathematical arguments, including the existence, uniqueness of the solution and threshold dynamics were successfully addressed.
The stability analysis of infection-free and infection steady state has witnessed an important and fundamental approach for understanding viral dynamics. We also mention that the global asymptotic stability of the constant equilibrium of (1.3) was achieved in the spatially homogeneous environment without considering the infection-age structure, and the infection-age structured model (1.2) was investigated with two infection routes but without considering the spatial aspects of the lymphoid tissues. Thus, we adopt the features of (1.3) and (1.2), and continue to consider the global threshold-type results of the model involving the following aspects:
-
In the early stage of infection, uninfected target T cells, infected T cells, and the free virus particles disperse at the target tissues (bounded domain) \(\Omega \subset \mathbb {R}^n\) according to the Fickian diffusion or Brownian motion associated with the Neumann boundary condition and constant diffusion coefficients \(d_1>0\), \(d_2>0\) and \(d_3>0\), respectively.
-
With infection age a, we use u(t, a, x) to denote the concentration of infected T cells. Based on model (1.2), the dynamics of infected cells is governed by
$$\begin{aligned} \left( \frac{\partial }{\partial t}+\frac{\partial }{\partial a}\right) u(t,a,x)=d_2\Delta u(t,a,x)-\theta (a)u(t,a,x), \end{aligned}$$(1.4)where \(\theta (a)\in L_+^\infty (0,+\infty )\) is the natural mortality of infected cells. Further, we assume that \(\theta (a)>\theta _{\min }\) for some positive number \(\theta _{\min }\). The free virus particles are produced at the rate \(\int _0^{+\infty }p(a)u(t,a,x)\mathrm{d}a\), where \(p(a)\in L_+^\infty (0,+\infty )\) is the age-specific per capita viral production rate of infected cells. In fact, the functional form of the viral production kernel, p(a), is unknown and remains to be determined experimentally. Here we give an example that capture features of the biology:
$$\begin{aligned} p(a) = \left\{ \begin{array}{ll} \displaystyle P_{\text {max}}(1-e^{-\beta (a-a_1)}),\ a>a_1,\\ \displaystyle 0,\ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \ \text {else}, \end{array}\right. \end{aligned}$$where \(P_{\text {max}}\) is the maximum production rate, because cellular resources will ultimately limit how rapidly virions can be produced. \(\beta \) controls how rapidly the saturation level is reached. \(a_1\) is the delay in viral production. This kernel can mimic either a very rapid increase to maximal production or a slow increase to maximal production depending on the value of \(\beta \). We refer the readers to [28] for more details.
-
We measure the virus interference during infection as saturation effect. Thus, uninfected target T cells are contacted by free virus particles at the rate \(\beta _1T\frac{V}{1+\alpha V}\), where \(\alpha \) is a half-saturation constant. As to the cell-to-cell transmission mechanism, we use the bilinear mechanism
$$\begin{aligned} \beta _2T\int _0^{+\infty }\ q(a)u(t,a,x)\mathrm{d}a \end{aligned}$$to account for the sustaining interactions between uninfected target T cells and infected T cells through the formation of the virological synapses, as it accounts for about 60% of viral infection [22]. Here, \(q(a)\in L_+^\infty (0,+\infty )\). Hence, we adopt
$$\begin{aligned} u(t,0,x)=\beta _1T\frac{V}{1+\alpha V}+\beta _2T\int _0^{+\infty }q(a)u(t,a,x)\mathrm{d}a. \end{aligned}$$(1.5) -
To make things not too complicated (as model involving the two infection routes and spatial diffusion have already made the problem very challenging), we adopt the recruitment rate h, natural death rates of cells (d and b) and the clearance rate of virus particles c as constant. Biologically, there exist \(0<a_1<a_2<+\infty \) such that \(q(a)>0\) and \(p(a)>0\), for all \(a\in (a_1,a_2)\). Moreover, we give the following hypothesis: (H) Assume that \(\lim _{a\rightarrow \infty }u(t,a,x) = 0\), which means that all the biological individuals cannot survive all the time.
Therefore, we arrive at the following reaction-diffusion and infection-age structured HIV infection model,
with the following boundary condition
For mathematical considerations, denote by \(\mathbb {X}:=C(\overline{\Omega },\mathbb {R})\) the continuous functions space equipped with the norm \(\left| \cdot \right| _\mathbb {X}\). Denote by \(\mathbb {Y}:=L^1(\mathbb {R}_+,\mathbb {X})\) the integrable functions space equipped with the norm \(\left| \varphi \right| _\mathbb {Y}:=\int _0^{+\infty }\left| \varphi (a)\right| _\mathbb {X}\mathrm{d}a,~\varphi \in \mathbb {Y}\). Let \(\mathbb {X}^+\) and \(\mathbb {Y}^+\) be the positive cones of \(\mathbb {X}\) and \(\mathbb {Y}\), respectively.
Suppose that \(T_i(t)\ (i=1,2,3):\ \mathbb {X} \rightarrow \mathbb {X},\ t \ge 0\), are the strongly continuous semigroups corresponding to the operators, \(d_i\Delta \ (i=1,2,3)\) with Neumann boundary condition. It is well-known that
where \(\Gamma _i(t, x, y)\ (i = 1,2,3)\) is the Green function of \(d_i\Delta \ (i=1,2,3)\) subject to the Neumann boundary condition. By the arguments as those in [36, Corollary 7.2.3] and [29, Theorem 1.5], \(T_i(t)\ (i=1,2,3):\ \mathbb {X}\rightarrow \mathbb {X},\ t \ge 0\), is strongly positive and compact. Further, \(T(t) = (T_1(t),T_2(t),T_3(t)):\ \mathbb {X}^3 \rightarrow \mathbb {X}^3,\ t \ge 0\), forms a strongly continuous semigroup.
System (1.6) can be reformulated by the method of characteristics (see, e.g., [14, 15, 44]). In the following, we give the details for this issue. Define \(U_c(x,t) = u(t,t+c,x)\) with \(c\in \mathbb {R}\), one has that
with Neumann boundary condition
where \(t_c = \max \{0,c\}\). It follows from [15] that
If \(a>t\), then \(t_c=0\). Hence we have
where \(\pi (a)=e^{-\int _0^a\theta (\sigma )\mathrm{d}\sigma }\).
If \(t>a\), then \(t_c=t-a\). With a similar argument as above, we can obtain that
Hence, u(t, a, x) can be solved as
Note that
Substituting (1.8) into (1.6) gives the following hybrid system containing two reaction-diffusion equations (T and V) and a Volterra integral equation (for simplicity, we denote u(t, 0, x) as u(t, x)),
where
The main goal of the current paper is to rigorously investigate the global threshold type results of (1.6). In Sect. 2, Theorem 2.2 tell us that (1.10) has a unique nonnegative solution defined on \([0,\infty )\times \overline{\Omega }\), and the solution is ultimately bounded in \(\mathbb {X}^+\times \mathbb {Y}^+\times \mathbb {X}^+\). Thus it comes naturally to investigate system (1.10) in a bounded domain. Section 3 is spent on defining the basic reproduction number (BRN). Our result in Lemma 3.1 indicates that the next generation operator (NGO) \(\mathcal {L}\) is strictly positive, bounded, and compact, which is proved by the Ascoli-Arzela theorem. Thus one can get the specific expression of BRN \(\mathfrak {R}_0\) by appealing to Krein-Rutman theorem, where \(\mathfrak {R}_0\) is the only positive eigenvalue of \(\mathcal {L}\), corresponding to which, there is a positive eigenvector. Further, once \(\mathfrak {R}_0 > 1\), (1.6) has a unique space-independent infection equilibrium \(\hat{E}=(\hat{T}, \hat{u}(a), \hat{V})\) (see Lemma 3.2). In Sect. 4, Theorem 4.1 below indicates that \(\mathfrak {R}_0\) works perfectly in determining the local dynamics for infection-free steady state \(E_0\) and space-independent infection equilibrium \(\hat{E}\) by checking the distribution of characteristic root of Eq. (4.5). More specifically, if \(\mathfrak {R}_0 < 1\), \(E_0\) is locally asymptotically stable (LAS), while \(\hat{E}\) is LAS if \(\mathfrak {R}_0 > 1\). Section 5 is devoted to the study of the persistence of infection in the system (1.10) for \(\mathfrak {R}_0> 1\), where the strong persistence is implied by the weak persistence. In Sect. 6, the global attractivity of \(E_0\) and \(\hat{E}\) are obtained by the technique of Lyapunov functionals. Lastly, the numerical simulations are performed to reinforce the theoretical findings.
2 Well-Posedness of the Model
This section aims to verify that the solution of (1.10) exists globally. We first prove the following result.
Theorem 2.1
For any \((\phi _1, \phi _2, \phi _3)\in \mathbb {X}^+\times \mathbb {Y}^+\times \mathbb {X}^+\), system (1.10) with (1.11) admits a unique nonnegative solution (T, u, V) on \([0,t_{\text {max}})\), where \(t_{\text {max}}\in \mathbb {R}_+^*\).
Proof
Let \(\mathbb {Y}_{t_{\text {max}}}:=C([0,t_{\text {max}}],\mathbb {X})\), associated with the following norm in \(\mathbb {Y}_{t_{\text {max}}}\),
For \((t,x)\in [0,t_{\text {max}})\times \overline{\Omega }\), solving T and V from (1.10) yields that
where \(\breve{\mathbf{F }}=e^{-d t}\int _\Omega \Gamma _1(t,x,y)\phi _1(y)\mathrm{d}y\) and \(\tilde{\mathbf{F }}=e^{-c t}\int _\Omega \Gamma _3(t,x,y)\phi _3(y)\mathrm{d}y\). Putting T and V into u equation, we get that for \((t,x)\in [0,t_{\text {max}})\times \overline{\Omega }\),
In what follows, we shall utilize the Banach-Picard fixed point theorem to verify that the operator \(\mathcal {F}:\ \mathbb {Y}_{t_{\text {max}}}\rightarrow \mathbb {Y}_{t_{\text {max}}}\) admits a fixed point, that is, system (1.10) admits a unique local solution. For the simplicity of notations, we denote
In these settings, \(\mathcal {F}\) can be rewritten as
Hence, by selecting two functions \(u_1\) and \(u_2\) in \(\mathbb {Y}_{t_{\text {max}}}\) and set \(\tilde{u}:=u_1-u_2\), we have
where
Let
We can choose sufficiently small \(0<t_{\text {max}}\ll 1\) such that \(\tilde{L}(t_{\text {max}})<1\). Hence, we can get the following inequality,
which means that \(\mathcal {F}\) is a strict contraction in \(\mathbb {Y}_{t_{\text {max}}}\). This confirms the assertion that the system (1.10) admit a unique local solution on \([0,t_{\text {max}})\).\(\square \)
We next establish the positivity of the solution.
Proposition 2.1
For any \((\phi _1, \phi _2, \phi _3)\in \mathbb {X}^+\times \mathbb {Y}^+\times \mathbb {X}^+\) and \((t,x)\in [0,t_{\text {max}})\times \overline{\Omega }\), we have
Proof
By (1.9), we know that if \(\phi _2\in \mathbb {Y}^+\), then
Define the following positive linear operator \(\Phi _i\ (i=1,2): \mathbb {Y}\rightarrow \mathbb {Y}\) as
in the sense that \(\Phi _i(\mathbb {Y}^+)\subset \mathbb {Y}^+\) as \(\Gamma _2(a,x,y)>0\). It follows that
Due to the fact that for any \((t,x)\in [0,t_{\text {max}})\times \overline{\Omega }\), \(\beta _2(\Phi _1(u)+F_2)+\frac{\beta _1}{\alpha }+d\) is continuous and bounded, we have that \(T(t,x)>0\), by the standard strong maximum principle.
Next we shall show the positivity of u(t, x). If there exist \((t_1,x_1)\in [0,t_{\text {max}})\times \overline{\Omega }\) such that
Thus,
Moreover, it follows from the third equation of (1.10) that
Hence,
Note that
for small enough \(\varepsilon \). Together with \(\mathbf{F} _1\ge 0\), \(\mathbf{F} _2\ge 0\), we have \(u(t_1+\varepsilon ,x_1)\ge 0\) for small enough \(\varepsilon \), which results in a contradiction. By some similar arguments as above, we can conclude that \(V(t,x)\ge 0\). This completes the proof.\(\square \)
We next show the solution of (1.10) exists globally.
Theorem 2.2
For any \((\phi _1, \phi _2, \phi _3)\in \mathbb {X}^+\times \mathbb {Y}^+\times \mathbb {X}^+\), then (1.10) admits a unique nonnegative solution (T, u, V) on \([0,\infty )\times \overline{\Omega }\). Furthermore, the solution of (1.10) is ultimately bounded.
Proof
In fact, by Proposition 2.1, we know that T equation of system (1.10) satisfies
which implies that \(\frac{h}{d}\) is the upper solution of T(t, x). We confirm that for any \((t,x)\in [0,t_{\text {max}})\times \overline{\Omega }\), \(u(t,x)<+\infty \). If it is not true, we suppose that there exists \((t^*, x^*)\in [0,t_{\text {max}})\times \Omega \) satisfying \(\lim _{t\rightarrow {t^*-0}}u(t,x^*)=+\infty \). Hence, \(\lim _{t\rightarrow {t^*-0}}\partial _t T(t,x^*)=-\infty \), which results in the contradiction to the positivity of T (see in Proposition 2.1). Hence, \(u(t, x) <\infty \). We are now ready to confirm the boundedness of V(t, x). Let \(p^+=\text {ess.sup}_{a\in \mathbb {R}_+}p(a)<+\infty \). Since
it follows that

Hence, V never blow up in \(t\in [0,t_{\text {max}}),\ x\in \Omega \). Consequently, we arrive at the assertion that the solution of (1.10) exists globally in \(\mathbb {X}^+\times \mathbb {Y}^+\times \mathbb {X}^+\). After passing to some similar arguments as before if necessary, we can confirm that for any \((\phi _1, \phi _2, \phi _3)\in \mathbb {X}^+\times \mathbb {Y}^+\times \mathbb {X}^+\) and a sufficiently large positive number \(\mathbf{M} _\infty \),
In fact, solving the T-equation of (2.1) gets
By taking the limit \(t\rightarrow \infty \) in (2.6) and (2.7), we immediately get
This together with \(u(t,x)<+\infty \) immediately gives the ultimate boundedness of the solution in \(\mathbb {X}^+\times \mathbb {Y}^+\times \mathbb {X}^+\).
Since c is a constant, we have
By the standard strong maximum principle, we get that \(V(t,x)>0\). This completes the proof.\(\square \)
3 Basic Reproduction Number
Following the classical theory in [10, 42], in this section, we shall define the basic reproduction number \(\mathfrak {R}_0\) of model (1.10). Obviously, (1.10) always exists an infection-free steady state \(E_0=(T_0,0,0)\) with \(T_0=\frac{h}{d}\). We are now ready to define the next generation operator for model (1.10) on \(\mathbb {X}^+\times \mathbb {Y}^+\times \mathbb {X}^+\). We consider the following linear sub-system of model (1.10) at disease-free equilibrium \(E_0\).
Firstly, we have
where
Recall that \(u(t,x): = u(t,0,x)\). This combines with (1.8) gives that
where
and
Similar to [47], the next generation operator \(\mathcal {L}\) can be evaluated as follows,
for any \(\varphi \in \mathbb {X}\). Following the classical theory in [10, 42], the basic reproduction number \(\mathfrak {R}_0\) is defined by the spectral radius of \(\mathcal {L}\), i.e., \(\mathfrak {R}_0:=r(\mathcal {L})\).
As to \(\mathcal {L}\), we have the following result.
Lemma 3.1
Let \(\mathcal {L}\) be defined by (3.2). The next generation operator \(\mathcal {L}\) is strictly positive, bounded, and compact.
Proof
Obviously, the next generation operator \(\mathcal {L}\) is positive. Due to the properties of \(\Gamma _2\) and \(\Gamma _3\), we denote by \(\{\varphi _n\}_{n\in \mathbb {N}}\) the bounded sequence in \(\mathbb {X}\), which satisfies \(|\varphi _n|_\mathbb {X}\le \mathbb {K}\), for some \(\mathbb {K}>0\). Let \(\{\psi _n\}_{n\in \mathbb {N}}=\mathcal {L}\varphi _n\). It then follows that
for all \(x\in \Omega \), which gives the uniform boundedness of \(\{\psi _n\}_{n\in \mathbb {N}}\). By the Ascoli-Arzela theorem, we are now ready to verify that \(\{\psi _n\}_{n\in \mathbb {N}}\) is equi-continuous. Taking any \(x, \tilde{x}\in \Omega \) with \(|x-\tilde{x}|\le \delta \). Direct calculation gives
Denote \(q^+=\text {ess.sup}_{a\in \mathbb {R}_+}q(a)<+\infty \) and \(p^+=\text {ess.sup}_{a\in \mathbb {R}_+}p(a)<+\infty \). From the properties of \(\Gamma _i(a, x, y),\ (i=2,3)\), we can select \(\epsilon >0\) such that \(|\Gamma _2(a,x,y)-\Gamma _2(a,\tilde{x},y)|\le \frac{\epsilon }{T_0\mathbb {K}\mid \Omega \mid q^+\beta _2}\) and \(|\Gamma _3(a,x,y)-\Gamma _3(a,\tilde{x},y)|\le \frac{c\epsilon }{ T_0\mathbb {K}\mid \Omega \mid p^+\beta _1}\) for all \(y\in \Omega \), where \(|\Omega |\) is the volume of \(\Omega \). For such \(\epsilon \) and \(\delta \), we immediately have
that is, \(\{\psi _n(x)\}_{n\in \mathbb {N}}\) is equi-continuous. Thus, the next generation operator \(\mathcal {L}\) is compact. This proves Lemma 3.1.\(\square \)
Generally speaking, it is not east to get the spectral radius of the next generation operator \(\mathcal {L}\), if not impossible, so that we can not get further information on dynamical properties of (1.10). By Lemma 3.1 combined with the Krein-Rutman theorem [1, Theorem 3.2], we know that the basic reproduction number is the only positive eigenvalue of \(\mathcal {L}\), corresponding to which, there is a positive eigenvector. Substituting \(\varphi (x) \equiv \phi ^* > 0\) (\(\phi ^*\) is a constant) into (3.2) and using \(\int _\Omega \Gamma _i(\cdot , x, y)\mathrm{d}y = 1,\ (i=2,3)\),
Hence, \(\mathfrak {R}_0=r(\mathcal {L})\) is given by
where
We should mention that with the assumption that all parameters of (1.10) are spatially homogeneous, \(E_0\) is constant equilibrium. It is crucial to (3.2) that the next generation operator \(\mathcal {L}\) does have a positive constant eigenvector, which in turn implies that \(\mathfrak {R}_0\) can be explicitly characterized by a positive constant (see also in [4, 5]).
Denote by \(\hat{E}\) the space-independent infection equilibrium of (1.10), if it exists. Then it satisfies
From the second equation of (3.4), one has that
Furthermore, from the first and forth equations of (3.4), we have
Now, we can see that \(\hat{T}\), \(\hat{V}\) and \(\hat{u}(0)\) can be written as terms of \(\hat{u}(0)\). Putting \(\hat{T}\) and \(\hat{V}\) into the third equation of (3.4) gives
where \(a_0=\alpha \beta _2PQ\), \(a_1=c\beta _2Q+\beta _1P+\alpha dP-\alpha \beta _2hQP\) and \(a_2=cd-\beta _1hQ-c\beta _2hQ=cd[1-\mathfrak {R}_0]\). Since \(a_0>0\), it has \(\Upsilon (\pm \infty )=+\infty \). In the case that \(\mathfrak {R}_0\le 1\), we know that \(\Upsilon (0)\ge 0\) and
Since \(\mathfrak {R}_0\le 1\), that is, \(c\beta _2hQ + \beta _1hP\le cd\), thus we can conclude that
we have that \(\Upsilon '(\hat{u}(0))>0\) for any \(\hat{u}(0)\ge 0\) when \(\mathfrak {R}_0\le 1\). It follows that Eq. (3.6) has no positive root, which in turn implies that (1.10) has no space-independent infection equilibrium \(\hat{E}\). In the case that \(\mathfrak {R}_0\ge 1\), we know that \(\Upsilon (0) = a_2 < 0\). From the properties of the quadratic function \(\Upsilon (\hat{u}(0))\), (3.6) admits a unique positive root \(\hat{u}(0)\).
In summary, we have the following result.
Lemma 3.2
If \(\mathfrak {R}_0 > 1\), (1.6) has a unique space-independent infection equilibrium \(\hat{E}=(\hat{T}, \hat{u}(a), \hat{V})\), which is unique and defined by (3.5).
4 Dynamics for the System
This section is paid to the local and global asymptotic stability of \(E_0\) and \(\hat{E}\).
4.1 Local Dynamics
We are now ready to establish the local asymptotic stability of \(E_0\) and \(\hat{E}\). Let \(E^*=(T^*,u^*(a),V^*)\) be \(E_0\) or \(\hat{E}\) of (1.6), we linearize (1.6) around \(E^*\) yields
By a classical parabolic theory [2], we denote by \(\zeta _j (j = 1, 2, \cdots )\) with \(0 = \zeta _0< \zeta _1< \zeta _2 <\cdots \) the eigenvalues of \(-\Delta \) subject to (1.7). Assume that the following parabolic problem with the homogeneous Neumann boundary condition
has the exponential solution in the form of \(U(t, x) = e^{\eta t}z(x), ~z(x) \in X_i\). Further from the exponential Ansatz (see, e.g., [27, Theorem 3.1]), we have that \(\Delta z(x) = -\zeta _iz(x)\). We substitute \(T = e^{\eta t}\phi (x)\), \(u(t, a, x) = e^{\eta t}\varphi (a, x)\), \(V =e^{\eta t}\psi (x)\) into (4.1), obtaining that
Combined with the second and fourth equation of (4.2), we get that
We next claim that \(\eta \ne -(d_1\zeta _i+d)\) and \(\eta \ne -(d_3\zeta _i+c)\). In fact, if \(\eta =-(d_1\zeta _i+d)\), together with the first equation of (4.2), imply that \(\varphi (0, x) = 0\). Hence, by the third equation of (4.2), \(\eta =-(d_3\zeta _i+c)\), which results in a contradiction. \(\eta \ne -(d_3\zeta _i+c)<0\) can be proved in a similar way. This claim together with the first and third equation of (4.2) imply that
where \(\widehat{P}(\eta ):=\int _0^\infty p(a)\hat{\pi }(a)e^{-\eta a}\mathrm{d}a\). Plugging (4.3) into the fourth equation of (4.2) yields
where \(\widehat{K}(\eta ):=\int _0^\infty q(a)\hat{\pi }(a)e^{-\eta a}\mathrm{d}a\). Canceling \(\varphi (0,x)\) on both sides of (4.4), we conclude that
In what follows, we pay attention to analyze the characteristic roots of (4.5).
Theorem 4.1
If \(\mathfrak {R}_0 < 1\), \(E_0\) is locally asymptotically stable, while \(\hat{E}\) is locally asymptotically stable if \(\mathfrak {R}_0 > 1\).
Proof
Let us first prove the local stability of \(E_0\). In this case, (4.5) can be simplified to
Suppose by contrary that (4.6) admits a real positive root \(\eta >0\). We directly have
If \(\mathfrak {R}_0<1\), the above inequality leads to a contradiction to (4.6). Hence, all the real roots of (4.6) are negative.
Let \(\eta = m \pm ni\) (with \(m \ge 0\) and \(n > 0\)) be a pair of complex roots of (4.6). After passing elementary analysis, we directly have
This contradicts with \(\mathfrak {R}_0 < 1\). This proves the assertion that \(E_0\) is LAS.
We next prove the local stability of \(\hat{E}\). If \(\eta = m+ni\) with \(m \ge 0\), the left-hand side of (4.5) satisfies
where \(\Xi =\beta _1\hat{V}+\beta _2\int _0^{+\infty }q(a)\hat{u}(a)\mathrm{d}a(1+\alpha \hat{V})\). However, the right-hand side of (4.5) satisfies
Comparing (4.7) and (4.8), we conclude that all roots of (4.5) have negative real parts if \(\mathfrak {R}_0>1\). This proves the assertion that \(\hat{E}\) is LAS if \(\mathfrak {R}_0>1\).\(\square \)
4.2 Persistence of Infection When \(\mathfrak {R}_0>1\)
In this subsection, we are concerned with the uniform persistence of (1.10) for \(\mathfrak {R}_0> 1\). Considering a semiflow associated with system (1.10), and replacing \(u(t-a, 0, y)\) in (1.8) by \(u(t-a, y)\) for short, we have the following result (see also in [35, Section 9.4]).
Lemma 4.1
Let \((\phi _1, \phi _2, \phi _3)\in \mathbb {X}^+ \times \mathbb {Y}^+\times \mathbb {X}^+\). For all \(t\ge 0\) and \(x\in \Omega \), system (1.10) admits a continuous semiflow defined by \(\Theta (t, \phi _1, \phi _2, \phi _3):=(T(t,\cdot ), u(t,\cdot ,\cdot ), V(t,\cdot ))\in \mathbb {X}^+ \times \mathbb {Y}^+\times \mathbb {X}^+\).
Proof
For any \(r, t, a\ge 0\) and \(x\in \Omega \), let
Hence, we have
with \(T_r(0,x)=T(r ,x)\) and \( V_r(0,x)= V(r,x)\), and
This together with (1.8) implies that for \(x\in \Omega \),
After passing elementary calculation, we have
On the other hand,
Combined with (4.11), we directly have
It then follows from (4.9), (4.10) and (4.12) that for all \(r\ge 0\) and \(t\ge 0\),
This completes the proof.\(\square \)
Let
The following result implies that the solution of (1.10) is uniformly weak \(|\cdot |_\mathbb {X}\)-persistence (see also in [5, Lemma 6.1] and [4, Lemma 5.2]).
Lemma 4.2
If \(\mathfrak {R}_0 > 1\) and \((\phi _1, \phi _2, \phi _3) \in \mathbb {D}\), then
holds for a sufficiently small constant number \(\varepsilon _1> 0\).
Proof
Since \(\mathfrak {R}_0 > 1\), choosing a sufficiently small number \(\varepsilon > 0\) such that
If there exists \(t_1>0\) such that
then, by (4.13), we can choose a small \(\lambda >0\) and \(t_2>t_1\) such that \(\tilde{\mathfrak {R}}>1\), where
and \(\tilde{s}=t_2-t_1\). By using this \(\varepsilon \),
Hence,
Similarly,
holds for all \(t>t_2,\ x\in \Omega \). Not that the incidence function \(f(V)=\beta _1\frac{V}{1+\alpha V}\) satisfies \(f(0)=0\), \(f'(V)=\frac{\beta _1}{(1+\alpha V)^2}>0\) and \(f''(V)=\frac{-2\alpha \beta _1}{(1+\alpha V)^3}<0\). Due to the fact that \(\lim \limits _{V\rightarrow 0} \frac{f(V)}{V}=f'(0)\), there exists a \(\hat{\varrho }\) such that
Using the fact \(\varepsilon _1=\min \{\hat{\varrho },\varepsilon \}\), together with Lemma 4.1, we take \(t_2=0\) (and thus, \(t_1=-\tilde{s}\)) by taking \(T(t_2,x)\), \(V(t_2,x)\) and \(u(t_2,a,x)\) as a new initial condition. Hence,
Obviously, for all \(x\in \Omega \), \(\int _0^\infty e^{-\lambda t}u(t,x)\mathrm{d}t<\infty \). Define \(u(t,\hat{x})=\min _{x\in \Omega } u(t,x)\). Taking Laplace transform on both sides of (4.15), we get
After passing elementary calculations, we obtain
Consequently, we obtain that
which is a contradiction with (4.14). This proves Lemma 4.2.\(\square \)
By the arguments similar to those in [4, Proposition 5.3] and [16, Theorem 1], we arrive at the below assertion on the strong \(|\cdot |_X\)-persistence.
Proposition 4.1
Suppose that \(\mathfrak {R}_0 > 1\). For any \((\phi _1, \phi _2, \phi _3) \in \mathbb {D}\), there exists a sufficiently small number \(\epsilon _2 > 0\) such that
With the help of Proposition 4.1, we show the following result.
Proposition 4.2
If \(\mathfrak {R}_0>1\), system (1.6) is uniformly strongly persistent, namely, there exists a positive value \(\varepsilon \) such that for any solution with the initial condition in \(\mathbb {D}\)
for \((a, x)\in \mathbb {R}_+\times \Omega \).
Proof
By Proposition 4.1, there exists positive constants \(\eta \) and \(T_0\) such that \(u(t,a,x)\ge \eta \pi (a)\) for \(t\ge T_0\). Then there exists a sufficiently small constant \(\eta _0\) such that \(u(t,a,x)\ge \eta \pi (a) - \eta _0\). It follows from the third equation of (1.6) that
where \(H = \int _0^\infty p(a) (\eta \pi (a) - \eta _0) \text {d} a\). Hence,
Thus, there exists \(\eta _1\) and \(T_2>T_1\) such that \(V(t,x)\ge \varepsilon _1\). Lastly, by the positivity of T(t, x) and choose \(\eta = \min \{\eta _0,\eta _1\}\), we finish the proof.\(\square \)
4.3 Global Attractivity of Steady States
This subsection is spent on the global asymptotic stability of \(E_0\) and \(\hat{E}\). Combined with local asymptotic stability and global attractivity of equilibria, we shall confirm that both \(E_0\) and \(\hat{E}\) are globally asymptotically stable. The global attractivity of \(E_0\) and \(\hat{E}\) is achieved by the technique of Lyapunov functionals.
Theorem 4.2
Suppose that \(\mathfrak {R}_0 < 1\), then \(E_0\) is globally asymptotically stable.
Proof
Let \(g(\alpha ) = \alpha -1 -\ln \alpha ,\ \alpha \in \mathbb {R}^+\). Then \(g(\alpha )\ge 0\) for all \(\alpha \in \mathbb {R}^+\) and the equality holds if and only if \(\alpha =1\).
Define a Lyapunov function \(L_{E_0}(t):\ \mathbb {D}\rightarrow \mathbb {R}\):
where \(L_T=T_0g(\frac{T}{T_0})\), \(L_u=\int _0^\infty \Psi (a)u(t,a,x)\mathrm{d}a\) and \(L_V=\frac{\beta _1T_0}{c}V\). The function \(\Psi (a)\) is nonnegative and integrable. We define \(\Psi (a)\) as
Obviously, we have the following properties for \(\Psi (a)\),
We first calculate the derivative of \(L_T(t, x)\) along the solution of system (1.6), obtaining that
By (1.8), we rewrite \(L_u(t, x)\) as
It then follows that
It follows from [21] that the Green function \(\Gamma _2\) satisfies \(\int _\Omega \Gamma _2(0,x,y)u(t,0,y)\mathrm{d}y = u(t,0,x)\) and \(\frac{\partial \Gamma _2}{\partial t} = d_2\Delta u(t,a,x)\). On the other hand, it follows from (1.8) that
where \(\Psi _t(t-a): = \frac{\partial \Psi (t-a)}{\partial t}\) and \(\Psi '(a):=\frac{\partial \Psi (a)}{\partial a}\). Same arguments with the other terms of (4.18), we have
Further, we calculate the derivative of \(L_V\), obtaining that
Finally, we integrate the equation (4.17), (4.19) and (4.20) over \(\Omega \), obtaining that
Here we have used \(\int _\Omega \Delta T \text {d}x=0\) and \(\int _\Omega \frac{\Delta T}{T} \text {d}x= \int _\Omega \frac{\Vert \nabla T\Vert ^2}{T^2}\text {d}x\). With the help of (4.16), one arrives at
As a result, with the help of [41, Theorem 4.2], \(E_0\) is globally attractive in \(\mathbb {D}\) if \(\mathfrak {R}_0 < 1\).\(\square \)
Now we are ready to confirm that \(\hat{E}\) is globally attractive in \(\mathbb {D}\), where \(\hat{E}\) is the space-independent infection equilibrium of (1.10). We first show the following lemma.
Lemma 4.3
The infection steady state \((\hat{T}, \hat{u}(a), \hat{V})\) satisfies
Proof
By the forth equation of (3.4), we have
On the other hand, using the fact that
we have
This finishes the proof. \(\square \)
Theorem 4.3
Suppose that \(\mathfrak {R}_0 >1\) and initial data \((\phi _1, \phi _2, \phi _3) \in \mathbb {D}\), then \(\hat{E}\) is globally asymptotically stable.
Proof
In this proof, we first give some notations,
where
and
Define a Lyapunov functional as
We calculate the derivative of \(\bar{L}_T\), together with \(h= d\hat{T}+\hat{u}(0)\), obtaining that
Next, we deal with \(\bar{L}_u\), clearly,
Note that
here we have used the fact that \(\hat{u}_a(a) = -\theta (a) \hat{u}(a)\). Further, direct calculation yields
Using integration by parts, one has
where g(x) was defined in the proof of Theorem 4.2. By hypothesis (H), we know that
Hence,
We further have the derivative of \(\bar{L}_V\) as follows:
By denoting \(\hat{L}=\bar{L}_T+\bar{L}_u+\bar{L}_V\), we directly have
where
Recall that
Furthermore, by the forth equation of (3.4), we have
Moreover, since
and recall that \(\hat{u}(a) = \hat{u}(0)\pi (a)\), we have
It then follows that
Note that
and using the zero trick in Lemma 4.3, one has that
Clearly, the last two terms of the above equation is zero, where we have used the fact that \(\ln \frac{a}{b} + \ln \frac{b}{a} = 0\). We then have
Consequently, we integrate (4.21) over \(\Omega \), obtaining that
where \(g(\alpha ) = \alpha -1 -\ln \alpha \ge 0\), for \(\alpha \in \mathbb {R}^+\). Note that each term of \(\frac{\text {d} L_{\hat{E}}(t)}{\text {d} t}\) is non-negative, Hence, due to the terms contain \((T-\hat{T})^2\) and \((V-\hat{V})^2\), we must have \(\frac{\text {d} L_{\hat{E}}(t)}{\text {d} t} =0\) holds if and only if \(T = \hat{T}\), \(V =\hat{V}\). Moreover, since \(g(\alpha )=0\) if and only if \(\alpha =1\), then \(\frac{\text {d} L_{\hat{E}}(t)}{\text {d} t} =0\) means that
Inserting \(T = \hat{T}\) and \(V =\hat{V}\) into the above relation, give us \(u(t, a, x) = \hat{u}(a)\). With the help of [41, Theorem 4.2], together with Theorem 4.1, we confirm that \(\hat{E}\) is globally asymptotically stable if \(\mathfrak {R}_0>1\). This proves Theorem 4.3. \(\square \)
5 Numerical Simulation
To support and validate the global threshold type result of (1.10), we perform numerical simulations in the cases of the 1-dimensional and 2-dimensional domain. We first consider the spatially 1-dimensional case and fix \(\Omega = (0, 1)\). We artificially set
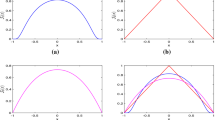
If we take \(\beta _1 = \beta _2=0.0026\), then \(\mathfrak {R}_0 = 0.95335 < 1\). From Theorem 4.2, \(E_0\) is globally attractive. Figure 1a–c demonstrate that the density of uninfected target T cells approaches a positive level, the densities of infected T cells and the free virus particles decay to zeros as time evolves. The spatial distributions of infected T cells gradually enlarge with higher prevalence but decays as time evolves (see Fig. 1d).
The time evolution of the densities of uninfected target T cells, infected T cells (with \(U(t,x)=\int _0^{\infty }u(t,a,x)\text {d}a\)) and the free virus particles of system (1.6) with (5.1) and \(\beta _1 = \beta _2=0.0026\) (\(\mathfrak {R}_0=0.95335 < 1\)). The initial data is \(\phi _1(x)=10,\ \phi _2(a,x)=e^{-b\times a-\int _0^a \theta (s)\text {d}s} (x-0.3)(0.7-x)\), and \(\phi _3(x)=0\)
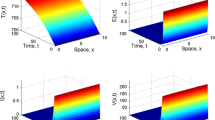
If we take \(\beta _1 = \beta _2=0.003\), then \(\mathfrak {R}_0 = 1.100021\). Theorem 4.3 ensures that \(\hat{E}\) is globally attractive. From Fig. 2a–c, the densities of uninfected target T cells, the densities of infected T cells and the free virus particles go towards some positive distributions as time evolves. We have seen from Fig. 2d that the spatial distributions of infected T cells gradually enlarge with higher prevalence as time evolves.
The time evolution of the densities of uninfected target T cells, infected T cells (with \(U(t,x)=\int _0^{\infty }u(t,a,x)\text {d}a\)) and the free virus particles of system (1.6) with (5.1) and \(\beta _1 = \beta _2=0.003\) (\(\mathfrak {R}_0=1.100021>1\)). The initial data is \(\phi _1(x)=10,\ \phi _2(a,x)=e^{-b\times a-\int _0^a \theta (s)\text {d}s} (x-0.3)(0.7-x)\), and \(\phi _3(x)=0\)
We next consider the spatially 2-dimensional case and fix \(\Omega = (0, 1)\times (0,1)\). We artificially set the parameters the same as in (5.1). In Fig. 3, we see from Theorem 4.2 that \(E_0\) is globally attractive. Figure 3a demonstrates that the density of uninfected target T cells approaches a positive level. Figure 3b, c demonstrate that the densities of infected T cells and the free virus particles decay to zeros as time evolves. In Fig. 4, we see from Theorem 4.3 that \(\hat{E}\) is globally attractive, that is, the densities of uninfected target T cells, the densities of infected T cells and the free virus particles converges to some positive distributions as time evolves.
The time evolution of the densities of uninfected target T cells, infected T cells (with \(U(t,x)=\int _0^{\infty }u(t,a,x)\text {d}a\)) and the free virus particles of system (1.6) with (5.1), \(\Omega = (0, 1)\times (0,1)\) and \(\beta _1 = \beta _2=0.0026\) (\(\mathfrak {R}_0=0.95335 < 1\)). The initial data is \(\phi _1(x,y)=10,\ \phi _2(a,x,y)=e^{-b\times a-\int _0^a \theta (s)\text {d}s}(x-0.3)(0.7-x)(y-0.3)(0.7-y)\), and \(\phi _3(x,y)=0\)
The time evolution of the densities of uninfected target T cells, infected T cells (with \(U(t,x)=\int _0^{\infty }u(t,a,x)\text {d}a\)) and the free virus particles of system (1.6) with (5.1), \(\Omega = (0, 1)\times (0,1)\) and \(\beta _1 = \beta _2=0.003\) (\(\mathfrak {R}_0=1.100021>1\)). The initial data is \(\phi _1(x,y)=10,\ \phi _2(a,x,y)=e^{-b\times a-\int _0^a \theta (s)\text {d}s}(x-0.3)(0.7-x)(y-0.3)(0.7-y)\), and \(\phi _3(x,y)=0\)
6 Discussion
The stability analysis of infection-free and infection steady state has witnessed an important and fundamental approach for understanding viral dynamics. This paper is spent on the global threshold type dynamics of an infection age-space structured HIV infection model involving two infection routes. The formulated model is inspired from previous models (1.3) and (1.2), where global threshold dynamics of (1.3) is obtained in a spatially homogeneous case and global threshold dynamics of (1.2) is obtained without considering the spatial aspects of the lymphoid tissues. The formulated model also extend models in [19, 23, 28, 40] in spatial aspects. In a bounded domain, we investigated the model (1.6) under the Neumann boundary condition. We first transform the system into a hybrid system containing two reaction-diffusion equations and a Volterra integral equation. By appealing to the Banach-Picard fixed point theorem, we have proved the well-posedness of the system (1.10), that is, the solution of (1.10) exists globally, and it is ultimately bounded. Following the classical theory in [10, 42], the basic reproduction number \(\mathfrak {R}_0\) is defined by the spectral radius of \(\mathcal {L}\). We should mention that with the assumption that all parameters of (1.10) are spatially homogeneous, \(E_0\) is constant. It is crucial to (3.2) that the next generation operator \(\mathcal {L}\) does have a positive constant eigenvector, which in turn implies that basic reproduction number \(\mathfrak {R}_0\) can be explicitly characterized by a positive constant (see also in [4, 5]).
The global threshold dynamics in terms of basic reproduction number \(\mathfrak {R}_0\) is investigated by determining the local and global asymptotic stability of \(E_0\) and \(\hat{E}\) (see Theorems 4.2 and 4.3). The methods used here are standard but not trivial. We also proved the strong \(|\cdot |_\mathbb {X}\)-persistence of (1.10) with \(\mathfrak {R}_0>1\), which is implied by the uniformly weak \(|\cdot |_\mathbb {X}\)-persistence (see (Proposition 4.1)). The global attractivity of \(E_0\) and \(\hat{E}\) are achieved by the technique of Lyapunov functional. Biologically, the HIV infection can be controlled with eradication and persistence in terms of basic reproduction number \(\mathfrak {R}_0\) as time evolves. Finally, numerical simulations in the 1-dimensional and 2-dimensional domain are carried out to validate our main results.
References
Amann, H.: Fixed point equations and nonlinear eigenvalue problems in ordered banach spaces. SIAM Rev. 18, 620–709 (1976)
Cantrell, R.S., Cosner, C.: Spatial Ecology via Reaction-Diffusion Equations. Wiley Series in Mathematical and Computational Biology, Wiley, Chichester (2003)
Coombs, D., Hyman, J.M., Perelson, A.S., Nelson, P.W., Gilchrist, M.A.: An age-structured model of hiv infection that allows for variations in the production rate of viral particles and the death rate of productively infected cells. Math. Biosci. Eng. 9, 267–288 (2004)
Chekroun, A., Kuniya, T.: An infection age-space structured SIR epidemic model with Neumann boundary condition. Appl. Anal. 99, 1972–1985 (2020)
Chekroun, A., Kuniya, T.: Global threshold dynamics of an infection age-structured SIR epidemic model with diffusion under the Dirichlet boundary condition. J. Differ. Equ. 269, 117–148 (2020)
Cheng, C., Dong, Y., Takeuchi, Y.: An age-structured virus model with two routes of infection in heterogeneous environments. Nonlinear Anal. RWA 39, 464–91 (2018)
Ducrot, A., Demasse, R.D.: An age-structured within-host model for multistrain malaria infections. SIAM J. Appl. Math. 73, 572–593 (2013)
Ducrot, A., Magal, P.: Travelling wave solutions for an infection-age structured model with diffusion. Proc. R. Soc. Edinb. A 139, 459–482 (2009)
Ducrot, A., Magal, P.: Travelling wave solutions for an infection-age structured epidemic model with external supplies. Nonlinearity 24, 2891–2911 (2011)
Diekmann, O., Heesterbeek, J.A.P., Metz, J.A.J.: On the definition and the computation of the basic reproduction ratio \(R_0\) in models for infectious diseases in heterogeneous populations. J. Math. Biol. 28, 365–382 (1990)
Dimitrov, D.S., Willey, R.L., Sato, H., et al.: Quantitation of human immunodeficiency virus type 1 infection kinetics. J. Virol. 67, 2182–2190 (1993)
Fackler, O.T., Murooka, T.T., Imle, A., et al.: Towards an integrative understanding of HIV-1 spread. Nat. Rev. Microbiol. 12, 563–574 (2014)
Feng, Z., Rong, L.: The influence of anti-viral drug therapy on the evolution of HIV-1 pathogens, DIMACS Ser. Discr. Math. Theo. Comp. Sci. 71, 261–279 (2006)
Fitzgibbon, W.E., Parrott, M.E., Webb, G.F.: Diffusive epidemic models with spatial and age dependent heterogeneity. Discrete Contin. Dyn. Syst. 1, 35–57 (1995)
Fitzgibbon, W.E., Morgana, J.J., Webb, G.F., Wu, Y.: A vector-host epidemic model with spatial structure and age of infection. Nonlinear Anal. RWA 41, 692–705 (2018)
Freedman, H.I., Moson, P.: Persistence definitions and their connections. Proc. Am. Math. Soc. 109, 1025–1033 (1990)
Ho, D.D., Neumann, A.U., Perelson, A.S., Chen, W., Leonard, J.M., Markowitz, M.: Rapid turnover of plasma virions and CD4 lymphocytes in HIV-1 infection. Nature 373, 123–126 (1995)
Hübner, W., McNerney, G.P., Chen, P., et al.: Quantitative 3D video microscopy of HIV transfer across T cell virological synapses. Science 323, 1743–1747 (2009)
Huang, G., Liu, X., Takeuchi, Y.: Lyapunov functions and global stability for age-structured HIV infection model. SIAM J. Appl. Math. 72, 25–38 (2012)
Hsu, S.B., Wang, F.-B., Zhao, X.-Q.: Dynamics of a periodically pulsed bio-reactor model with a hydraulic storage zone. J. Dyn. Differ. Equ. 23, 817–842 (2011)
It\(\hat{{\rm o}}\), S.: Diffusion Equations, Translations of Mathematical Monographs, vol. 114. American Mathematical Society, Providence (1992)
Iwami, S., Takeuchi, J.S., Nakaoka, S., et al.: Cell-to-cell infection by HIV contributes over half of virus infection. Elife 4, e08150 (2015)
Lai, X., Zou, X.: Modeling HIV-1 virus dynamics with both virus-to-cell infection and cell-to-cell transmission. SIAM J. Appl. Math. 74(3), 898–917 (2014)
Liu, S., Zhang, R.: On an age-structured hepatitis B virus infection model with HBV DNA-containing capsids. Bull. Malays. Math. Sci. Soc. 44, 1345–1370 (2021)
Lorenzo-Redondo, R., Fryer, H.R., Bedford, T., et al.: Persistent HIV-1 replication maintains the tissue reservoir during therapy. Nature 530, 51–56 (2016)
Magal, P., McCluskey, C.C., Webb, G.F.: Lyapunov functional and global asymptotic stability for an infection-age model. Appl. Anal. 89, 1109–1140 (2010)
McCluskey, C.C., Yang, Y.: Global stability of a diffusive virus dynamics model with general incidence functions and time delay. Nonlinear Anal. RWA 25, 64–78 (2015)
Nelson, P.W., Gilchris, M.A., Coombs, D., Hyman, J.M., Perelson, A.S.: An age-structured model of HIV infection that allow for variations in the production rate of viral particles and the death rate of productively infected cells. Math. Biosci. Eng. 1, 267–288 (2004)
Pazy, A.: Semigroups of Linear Operators and Application to Partial Differential Equations, Applied Mathematical Sciences, vol. 44. Springer, New York (1983)
Perelson, A.S., Neumann, A.U., Markowitz, M., Leonard, J.M., Ho, D.D.: HIV-1 dynamics in vivo: Virion clearance rate, infected cell life-span, and viral generation time. Science 271, 1582–1586 (1996)
Perelson, A.S., Rong, L., Feng, Z.: Mathematical analysis of age-structured HIV-1 dynamics with combination antiviral therapy. SIAM J. Appl. Math. 67, 731–756 (2007)
Qesmi, R., Elsaadany, S., Heffernan, J.M., Wu, J.: A hepatitis B and C virus model with age since infection that exhibits backward bifurcation. SIAM J. Appl. Math. 71, 1509–1530 (2011)
Rong, L., Feng, Z., Perelson, A.S.: Mathematical analysis of age-structured HIV-1 dynamics with combination antiretroviral therapy. SIAM J. Appl. Math. 67, 731–756 (2007)
Ren, X., Tian, Y., Liu, L., Liu, X.: A reaction-diffusion within-host HIV model with cell-to-cell transmission. J. Math. Biol. 76, 1831–1872 (2018)
Smith, H.L., Thieme, H.R.: Dynamical Systems and Population Persistence, Graduate Studies in Mathematics, vol. 118. American Mathematical Society, Providence (2011)
Smith, H.L.: Monotone Dynamical Systems: An Introduction to the Theory of Competitive and Cooperative Systems, Mathematical Surveys and Monographs, vol. 41. American Mathematical Society, Providence (1995)
Sigal, A., Kim, J.T., Balazs, A.B., Dekel, E., Mayo, A., Milo, R., Baltimore, D.: Cell-to-cell spread of HIV permits ongoing replication despite antiretroviral therapy. Nature 477, 95–98 (2011)
Sattentau, Q.: The direct passage of animal viruses between cells. Curr. Opin. Virol. 1, 396–402 (2011)
Shu, H., Chen, Y., Wang, L.: Impacts of the cell-free and cell-to-cell infection modes on viral dynamics. J. Dyn. Differ. Equ. 30, 1817–1836 (2018)
Wang, J., Lang, J., Zou, X.: Analysis of an age structured HIV infection model with virus-to-cell infection and cell-to-cell transmission. Nonlinear Anal. RWA 34, 75–96 (2017)
Walker, J.A.: Dynamical Systems and Evolution Equations: Theory and Applications, Mathematical Concepts and Methods in Science and Engineering, vol. 20. Springer, New York (1980)
Wang, W., Zhao, X.-Q.: Basic reproduction numbers for reaction-diffusion epidemic models. SIAM J. Appl. Dyn. Syst. 11, 1652–1673 (2012)
Wu, P., Zhao, H.: Dynamics of an HIV infection model with two infection routes and evolutionary competition between two viral strains. Appl. Math. Model. 84, 240–264 (2020)
Wu, P., Zhao, H.: Mathematical analysis of an age-structured HIV/AIDS epidemic model with HAART and spatial diffusion. Nonlinear Anal. RWA. 60, 103289 (2021)
Yan, D., Fu, X.: Asymptotic analysis of an age-structured HIV infection model with logistic target-cell growth and two infecting routes. Int. J. Bifurcat. Chaos 30, 2050059 (2020)
Yan, D., Fu, X.: Analysis of an age-structured HIV infection model with logistic target-cell growth and antiretroviral therapy. IMA J. Appl. Math. 83, 1037–1065 (2018)
Yang, J., Xu, R., Li, J.: Threshold dynamics of an age-space structured brucellosis disease model with Neumann boundary condition. Nonlinear Anal. RWA. 50, 192–217 (2019)
Zhang, W., Zou, L., Ruan, S.: An age-structured model for the transmission dynamics of hepatitis B. SIAM J. Appl. Math. 70, 3121–3139 (2010)
Zhang, X., Liu, Z.: Bifurcation analysis of an age structured HIV infection model with both virus-to-cell and cell-to-cell transmissions. Int. J. Bifurcat. Chaos 28, 1850109 (2018)
Zhang, X., Liu, Z.: Periodic oscillations in HIV transmission model with intracellular time delay and infection-age structure. Commun. Nonlinear Sci. Numer. Simul. 91, 105463 (2020)
Acknowledgements
The authors would like to thank the anonymous reviewers for their helpful comments to the previous version of our manuscript. J. Wang was supported by National Natural Science Foundation of China (Nos. 12071115, 11871179), and Heilongjiang Provincial Key Laboratory of the Theory and Computation of Complex Systems. R. Zhang was supported by the National Natural Science Foundation of China (No. 12101309), the China Postdoctoral Science Foundation (No. 2021M691577) and the Postdoctoral Fundation of Jiangsu Province.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, J., Zhang, R. & Gao, Y. Global Threshold Dynamics of an Infection Age-Space Structured HIV Infection Model with Neumann Boundary Condition. J Dyn Diff Equat 35, 2279–2311 (2023). https://doi.org/10.1007/s10884-021-10086-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10884-021-10086-2
Keywords
- HIV infection model
- Age-space structure
- Basic reproduction number
- Global stability
- Lyapunov functional
- Uniform persistence