Abstract
The present book chapter is about the phytochemistry, pharmacological properties, and ethonobotanical, safety, and toxicological aspects of the species, Artemisia indica. This genus Artemisia belonging to Asteraceae family comprises of more than 400 species, among which A. indica (also called mugwort) is a perennial medicinal herb found majorly in cold temperate zones of Asia including Pakistan, China, India, Thailand, Korea, Japan, etc. It is extensively utilized by the traditional people in many countries for treating Malaria, chronic fever, dyspepsia, ringworms, hepatic diseases, and diarrhea-like health problems. Artemisinin is one of the prime phytochemical components accountable for the potential antimalarial activity, found in this species. Besides this, the plant comprises chiefly sesquiterpenes and davanone in its volatile oil. Modern pharmacological studies have revealed that its crude extracts exhibit a wide range of pharmacological activities such as anti-inflammatory, anthelmintic, anticancer, antidiabetic, antimicrobial, antidepressant, and many more as a result of the existence of several bioactive components in it. Few toxicological evaluation have documented on this species but in depth experiments, safety, and toxicological evaluation are needed to explore medicinal properties of this species more.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Artemisia indica
- Asteraceae
- Traditional uses
- Phytochemical components
- Artemisinin
- Pharmacological properties
3.1 Introduction
Humans have depended on plant kingdom for their daily need which is as old as the humans’ existence in the world. The plant kingdom includes all the important medicinal flora, and these are used for ages to treat various diseases. Almost 87% human diseases or problems are cured with naturally occurring compounds, derived from medicinal herbs, and their allied medicines (Hussain 2020). One of such important medicinal plant is the genus Artemisia, commonly known as “worm wood.” This genus is a member of Asteraceae family comprising more than 400 species (Adewumi et al. 2020). One of the mostly known species of this genus is Artemisia indica, commonly called “mugwort” or Indian wormwood. This common perennial herb generally spreads in forest edges, grasslands, abandoned lands, roadside, fallow fields, etc. very quickly. This species is found in India, China, Nepal, Pakistan, Thailand, Japan, Korea, and many more cold-temperate regions of Asia; and in most of the regions, this plant is commonly called as “Titepati” (Jassal et al. 2019; Shimono et al. 2013). It is suitable for medium loamy and light sandy soils with well drainage system. This plant can be sometimes annual or woody subshrub that spreads through rhizomatous root-stock. The stem ranges from 80–150 cm tall, with many branches. Leaves are either stalkless or short – stalked and ovate, elliptic, or oblong-ovate shaped. Flowers are erect, ovoid or oblong-ovoid, bisexual, and borne in conical. Fruit is achene type and brown obovoid or oblong shaped. These plants are hardier and aromatic. It is generally propagated by seeds, also by stolon (Ken Fern 2022; Flowers of India 2021). This species had been widely utilized traditionally in many countries to treat many diseases and health problems like dyspepsia, hepatic diseases, chronic fever, diarrhea, ringworms, wounds, Malaria, and many more (Adewumi et al. 2020; Nigam et al. 2019; Bhattarai 2020). This plant primarily comprises of sterols, terpenoids, acetylenes, coumarins, flavonoids etc.; among these oxygenated sesquiterpenes and davanone are the main compounds found in its volatile oil (Adewumi et al. 2020; Haider et al. 2014). Because of its various biologically active components, it shows numerous pharmacological activities like antimalaria (main activity because of its component artemisinin), anticancer, anthelmintic, antiseptic, inflammatory, antioxidant, and many more; and these activities can be shown at specific safe doses of its extracts. So, the purpose of this study is to discover the traditional and folk medicinal uses, phytoconstituents, pharmacological activity, clinical studies, and safety and toxicity related to this study of A. indica in more details.
3.2 Traditional Uses
One of the most necessary health sources is traditional medicinal system in the entire world (especially the developing countries), derived from beneficial medical plants or herbs. A. indica is one of those beneficial perennial herbs, found in the different regions of Western Himalayas with the familiar name “Titepati.” Local people of this region used this herb for ages to heal dyspepsia, hepatic diseases, and also for chronic fevers (Adewumi et al. 2020). This plant had majorly utilized by the local peoples of China, Nepal, Pakistan, India, Thailand (especially for Malaria), and several other countries or areas of southeast Asia in medicines to treat various ailments (Nahid et al. 2017). In Nepal, the juice of this plant had been utilized in treatments of abdominal pain, diarrhea, ringworms, wounds, cuts, leech infestation, and dysentery. Different parts of this plant had been eaten here such as cooked young leaves were consumed with Barley, also with rice to add some color and flavors. Here, the dried leaf and flowers were used as insecticides and the juice of the leaf were used to heal the skin ailments (Rashid et al. 2013; Nigam et al. 2019). Local peoples of Eastern Nepal, this species along with some other plant species had been found in a survey to treat many infections or infestations such as for helminths, fever, diphtheria, malaria, scabies, gonorrhea, etc. (Bhattarai 2020). In Okinawa, a famous island of Japan in whole world for the presence of long-lived individuals in abundance, it was utilized as an important food plant with some other plants (Niwano et al. 2009). In Xishuangbanna of China, Dai people uses various plants as mosquito repellent but A. indica was found to be the most effective mosquito repellent, especially in the cases of Dengue (Gou et al. 2020). In South Uganda, this plant was used as pesticide or for pest control by the local peoples (Mwine et al. 2011). In northeast areas of Pakistan, leaves of this species had been used medicinally by their indigenous peoples, especially for ear problems (Afzal et al. 2009). In Assam, also it had been found to be used by the local people of Tejpur as antimalarial drug to treat malaria and associated symptoms such as fever, headache, sweating, joint pains, weakness, vomiting, and shivering (Namsa et al. 2011). Fried tender shoots of this plant were also reported to eat as a vegetable food by the indigenous people of Garo in Norkek-Biosphere Reserve of Meghalaya state of India, through a survey (Singh et al. 2012). In another state of India, Darjeeling, it was used to cure asthma, amoebic dysentery, helminthic infections, several skin, and stomach problems (Tiwary et al. 2015). Young leaves had also been consumed as vegetable by the indigenous people of different areas (like Arunachal Pradesh) of India (Joram et al. 2021). The flowering shoots and leaves had been documented to show several pharmacological efficacies such as anthelmintic, antispasmodic, anti-septic, cytotoxic, antimicrobial, and anticancer (Rashid et al. 2013). Usage of A. indica traditionally in different places is given below in Table 3.1.
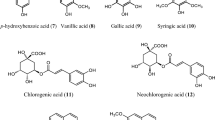
3.3 Chemical Constituents (Fig. 3.1)
A. indica represents a rich source of numerous biologically active components, responsible for various pharmacological activities. Quantitative and qualitative composition of these compounds differ hugely that might be linked with species variation; climatic, geographic, genetic, and environmental conditions; vegetation phase; anatomical part and age of plant; soil; method and season of harvesting; etc. (Nigam et al. 2019). This plant chiefly comprises of sterols, terpenoids, acetylenes, coumarins, flavonoids, etc. Oxygenated terpenes (among all terpenes) and hydrocarbons are present in this plant in abundance out of all the components in its volatile oil (Rather et al. 2017; Adewumi et al. 2020). In its essential oil, total 32 compounds had been reported to be found from Uttarakhand Himalaya, isolated and evaluated by GC and GC/MS method, among which oxygenated sesquiterpenes in 33.83% and davanone in 30.8% were the main compound. Besides this two, monoterpene hydrocarbons in 25.90%, sesquiterpene hydrocarbons in 20.54%, and oxygenated monoterpenes in 15.15% were found. Other major components found were β-elemene, β-pinene, β-myrcene, δ-cadinene, τ-muurolol, germacrene-D, trans-caryophyllene, cymene, limonene, linalool, 1,8-cineol, sabinene, etc. (Haider et al. 2014). Some previous studies in this region had shown that essential oil of A. indica were lacking the component davanone; even not present in traces (Shah and Rawat 2008). Besides these components, Artemisia ketone, ascaridole, borneol, transverbenol, alpha thujone, p-cymene, chrysanthenyl acetate, cubebene, caryophyllene oxide, terpineol, eucalyptol, isoascaridole, camphor, etc. were also found to be present in its volatile oil (Satyal et al. 2012; Rashid et al. 2013). Diverse solvent extracts had been isolated from the leaf of this species and assessed by HPLC-MS and HPLC-DAD method which showed flavonoids like casticin, cirsilineol, eupatin, and chrysoplenetin in it. These are all polymethoxy flavonoids (Tasdemir et al. 2015). In A. indica, exiguaflavanone-A, exiguaflavanone-B, maacklain, 2-(2,4-dihydroxyphenyl)-5,6-methylenedioxybenzofuran, and artemisinin were isolated and discovered to show the antimalarial activity as major compound responsible for its antimalarial activity (Chanphen et al. 1998; Mannan et al. 2011). From the root and shoot, various compounds had been isolated and identified through bioassay-guided fractionation method such as 5-hydroxy-3,7,4′-trimethoxyflavone, 6-methoxy-7,8-methylenedioxy coumarin, cis-matricaria ester, lupeol, maackiain, ludartin, cis-matricaria ester, and trans-matricaria ester (Zeng et al. 2015). Various components, isolated from various parts of the plant, are shown below with its pharmacological attributes in Table 3.2.
3.4 Pharmacological Activities
This medicinal plant is industrially very crucial as it exhibits diverse pharmacological properties from its different parts. Some of these activities are mentioned below in brief (Fig. 3.2).
3.4.1 Antimalarial Activity
It is used majorly in malaria, caused by protozoa Plasmodium sp., mostly by Plasmodium falciparum (Saxena et al. 2003), as antipyretic to reduce the fevers. An effective antimalarial drug artemisinin, derived from the leaves of its species which is an endoperoxide sesquiterpene lactone (Mannan et al. 2011). This artemisinin had been produced through different cultures such as callus, hairy shoot, and root cultures. One of these cultures had been experimentally proven to show increased amount of artemisinin. In such experiments with hairy root cultures of A. indica in liquid medium, it was found that artemisinin content increased (0.042%) in its transformed roots after transformed by T- DNA of Agrobacterium rhizogenes strain (Mannan et al. 2008). Another research on A. indica showed other two components—exiguaflavanone A and B—to have in vitro antimalarial effect. These components were isolated and purified by the crude MeOH extracts, obtained from the air-dried stem. Then their antimalarial activities were evaluated by their effective concentration (EC50) values (Chanphen et al. 1998).
3.4.2 Anticancer Activity
Cancer is the serious worldwide public health issue, mostly because of its lacking in early detection methods. So anticancer activities generally help to prevent or suppress the carcinogenic development (Chanda and Nagani 2013). Study on 50 μg/mL ethanol extract of leaf of A. indica in MTT, trypan blue exclusion assays, and morphological assessment showed that these extracts had inhibited more than 50% of human breast adenocarcinoma (MCF7) growth, human cervix adenocarcinoma (HeLa), and human hepatocarcinoma (HepG2) cell lines (Tiwary et al. 2015). Another study on aerial parts of A. indica had been done by sulforhodamine-B assay to know its cytotoxic effects on leukemia (THP1), lung (A-549), liver (HEP-2), and colon (Caco-2) cancer cell lines. This study revealed that essential oil, extracted from these extracts, had significantly inhibited these cell lines’ growth in the dilution range of 10–100 μg/mL with 19.5 μg/mL IC50 value for Caco-2, 10 μg/mL IC50 value for THP-1, 15.5 μg/mL IC50 value for HEP-2, and 25 μg/mL IC50 value for A-549 (Rashid et al. 2013). Other reports suggest that ethyl acetate extract of this species showed anticancer activity by decreasing the mitochondrial membrane potential and by inducing the DNA damage against lung (A-549), colon (Colo-205), human breast adenocarcinoma (MCF-7), and BHY and Miapaca cancer cell lines. Ludartin and lupeol had been found in this study to be the major component, responsible for this activity (Zeng et al. 2015).
3.4.3 Antidiabetic and Antihyperlipidemic Activity
Diabetes is another major issue (a metabolic disorder) in the world now as insufficient blood sugar regulation creates serious health concern. It is generally distinguished by chronic hyperglycemia, i.e., high blood sugar, because of the disabilities in insulin action and secretion (Salehi et al. 2019). An evaluation study on these activities of A. indica in 50 mg/kg diabetic Sprague-Dawley rats induced with streptozotocin had been performed. This study showed that 200 and 400 mg/kg b.w. hydroethanolic crude extracts and 200 mg/kg b.w. chloroform extracts of aerial parts of the plant had reduced the blood glucose levels considerably. It showed antihyperlipidemic (high lipid levels in blood) activities by reducing the cholesterol levels, serum creatinine, lipoproteins, and triglyceride levels (Ahmad et al. 2014). In reports of Jassal et al. (2019), oleanolic acid and carnosol had been found to have antidiabetic activities, whereas caryophyllene had found to treat hyperlipidemia, i.e., high lipid in blood (Jassal et al. 2019). 1–100 mg/kg b.w. carnosol, present in chloroform extracts of A. indica, was found in recent studies to lower the blood glucose levels, cholesterol, triglycerides, low-density lipoproteins, and creatinine levels in diabetes-induced rats (Khan et al. 2017).
3.4.4 Anthelmintic Activity
Helminths, the parasitic worms, create the most common infections (helminthic infections) in men. So anthelmintic drugs destroy or expel these parasitic worms which are present in large numbers, majorly in gastrointestinal (GI) tract (Das et al. 2011). Many experiments had been performed to show this activity in A. indica plant. One such investigation on crude aqueous, chloroform, and methanolic extracts of this species had been done against the healthy Indian earthworm, Pheretima posthuma where 2.5 mg/mL chloroform and methanol extract had shown efficient anthelmintic effect against the earthworm. The former one showed paralysis and death time of 9.67 and 20.67 min respectively, whereas the later one showed 19.67 and 25.33 min for paralysis and death respectively (Sarnim et al. 2013). Another study was done to scientifically prove its activity with 3.75, 6.25, 12.5, 25, and 50 mg/mL concentrations, accompanied by positive control with 10% albendazole and negative control with PBS. This resulted in the highest anthelmintic activity of A. indica at 50 mg/mL concentration of ethanolic extracts by different assays such as egg hatch inhibition, larval mortality, and adult worm mortality (Khan et al. 2015).
3.4.5 Antidepressant and Anxiolytic Activity
Antidepressant drugs have been used to decrease the symptoms of psychological disorders, especially the depressive disorders, by rectifying the chemical imbalances of vital neurotransmitters (common link of communication between nerve cells) in the brain that can improve the behavior and mood during these disorders. They generally increase the levels of neurotransmitters (e.g., selective serotonin reuptake inhibitors) around the nerves (Ogbru 2021). An anxiolytic drug has been used to treat anxiety disorders almost in the same way. Some investigations on nonvolatile components of A. indica had been done in mouse models to understand its efficacy on the function of central nervous system (CNS). The outcome of these investigations showed that oleanolic acid, carnosol, and ursolic acid, isolated from this species, had induced the anxiolytic (through light or dark box paradigms and elevated plus maze tests) and antidepressant (through forced swim and tail suspension tests) activities in mouse without any signs of toxicity (Khan et al. 2016).
3.4.6 Antimicrobial Activity
Antimicrobial activity is activity of all the active agents that can cease the microbial growth or can inhibit or destroy their colonies or may kill them directly (Elmogahzy 2020). This species has thus shown effectively this antimicrobial activity, especially antibacterial and antifungal activity. Study on antimicrobial activities of A. indica, Tecoma stans, and Medicago falcata in vitro had shown that chloroform, ethyl acetate, and butanol extracts of A. indica have 15–20 mm inhibitory effects towards Pseudomonas aeruginosa, Staphylococcus aureus, and Escherichia coli bacteria, whereas it showed 12–14 mm inhibitory activities against bacterium Salmonella typhi. Chloroform and n-hexane extracts of A. indica had been shown antifungal activities against Fusarium solani and Aspergillus flavus, respectively, by completely ceasing their growth (Javid et al. 2015). Another investigation to understand the antimicrobial activity of A. indica was done on the essential oil, extracted from the aerial parts of the plant. Total 43 phytoconstituents had been found through GC-MS and GC-FID analysis, among which 42.1% artemisia ketone, 8.6% germacrene, 6.1% borneol, and 4.8% cis-chrysanthenyl acetate were the major components to show notable antibacterial activities, mostly against gram-negative bacteria like Pseudomonas aeruginosa and klebsiella pneumonia and antifungal activities against fungal strains such as Pseudomonas chrysogenum and Aspergillus niger (Rashid et al. 2013). Leaves of A. indica had also been reported to show antileishmanial activity against the protozoan parasite Leishmania sp. (which creates leishmaniasis), due to the presence of artemisinin compound in it (Ganguly et al. 2006).
3.4.7 Anti-Inflammatory Activity
Many reports suggested that this species has ability to reduce inflammation. Inflammation generally occurs in response to the cell death, degeneration, cancer, ischemia, and tissue injury or due to the invasion of microbes in the body (Azab et al. 2016). Research on anti-inflammatory action of methanol extract of aerial parts of A. indica by carrageenin-induced rat paw edema, a well-established model of assessing chronic inflammation, was done. This research had revealed that methanolic (100, 200, and 400 mg/kg) extracts had notably inhibited the carrageenin-induced paw edema in rats. Flavonoids had been reported to be responsible for this anti-inflammatory activity as it can inhibit the prostaglandin synthesizing enzymes (Mansouri et al. 2015; Sagar et al. 2010). Other reports suggested that saponins, tannins, casticin, and terpenoids (like carnosol, caryophyllene), present in A. indica extracts, showed anti-inflammatory activity because of its effect of inhibition on inflammation (Ruwali et al. 2015; Jassal et al. 2019).
3.4.8 Immunomodulatory Activity
Modulation of immune system (defense system of our body) denotes any change in the immune response such as expression, stimulation, inhibition, or amplification of any phase of the immune response. So immunomodulators are used to modify to alter this immune response of the system (Abood et al. 2014). Major information is reported about its immunomodulatory action but in vitro experiments are few. One experiment on methanolic fresh aerial parts of this plant had been performed through B and T lymphocyte proliferation assay in lymphocyte culture of chicken. The results suggested that 200 μg/mL extracts had significantly increased the regulation of B cell proliferation at 11.76% and T cell proliferation at 12.018% in the presence of LPS (B cell mitogen) and Con A, respectively (Ruwali et al. 2015). Casticin had been reported as one of the most responsible phyto-constituent for this activity in A. indica (Jassal et al. 2019).
3.4.9 Antioxidant Activity
Prevention of developing reactive oxygen species and their reactions, thereby inhibition or limitation of oxidation of nutrients like proteins, lipids, is called antioxidant activity (Guclu et al. 2021). These reactive oxygen species can also bring about necrosis, apoptosis, and oxidative stress. Thus, antioxidants can prevent all these distress from these harmful substances. Methanolic extracts of leaves of A. indica had been evaluated in vitro by DPPH free radical scavenging assay and revealed to consist antioxidant activity because of the existence of phenol and flavonoid in significant quantity (Nahid et al. 2017). Essential oil, collected from its aerial parts, was also reportedly found to have antioxidant activities, after studying through hydroxyl and DPPH radical scavenging assays (Rashid et al. 2013).
3.4.10 Anticonvulsant Activity
Anticonvulsant drugs, also known as anti-epileptic or anti-seizure drugs, have been used to prevent the convulsions or seizures by either decreasing the excitation or enhancing the inhibition. A. indica was investigated for its anticonvulsant activity and found as an effective drug for it. Few studies had been done on non-volatile compounds such as ursolic acid, oleanolic acid, and carnosol which had been assessed for this effect on GABA-A receptors in mouse models of convulsions, induced by pentylenetetrazole (PTZ). The result of this assessment showed that 10 mg/kg carnosol, 30 mg/kg ursolic acid, and 100 mg/kg oleanolic acid had effectively increased the span of onset and reduced the span of clonic-tonic seizures and also gave 100%, 83.37%, and 66.7% protection, respectively. In this assessment, 1 mg/kg diazepam had been used as reference drug (Khan et al. 2016).
3.5 Studies Related to Safety and Toxicity
Medicinal plants or natural herbs have been the chief remedy for almost 100 years ago to treat numerous diseases, and 25% of the medicines (those are recently used/modern medicines) are primarily utilized by these herbs. Generally, these herbs have been regarded safe because of its long history of use in traditional medicines, but still nowadays this safety has been a major concern for both of the general public and the national health authorities. On the other hand, toxicity is the capability of substances to cause toxic or harmful effects in living beings. It can be acute, chronic, subacute, and subchronic type (Mensah et al. 2019). So besides of safety, toxic doses of these herbs must have assessed and are also the major concern. Extracts of A. indica have also specific safe and toxic doses of showing particular pharmacological activities. No such toxicity of this plant was found, but in some cases dermatitis or different allergies can occur in people (Foster and Duke 2020). Some reports suggested that crude hydro-methanolic extracts at 200 mg/kg body weight and 400 mg/kg body weight; chloroform extracts at 200 mg/kg body weight of A. indica had shown to decrease the blood glucose levels. So, these doses were safe and did not produce any harmful changes or effects in the diabetic rats induced by streptozotocin, but the extract at 2000 mg/kg body weight dose had shown few irritations and escape behavior in rats (Ahmad et al. 2014). Other studies on anthelmintic activity of this species, 6.25 mg/L methanol and chloroform extracts and 2.5 mg/L chloroform extracts were found to show significant positive effect against Pheretima posthuma, but the toxic doses were not evaluated that much (Sarnim et al. 2013). In an in vitro study, 0.3–1.6 mg/mL methanolic extracts had also shown 54.72% cytotoxicity in chicken lymphocytes, and 0.2 mg/mL extracts had shown complete cell viability (Ruwali et al. 2015).
3.6 Conclusions and Future Recommendations
Humans rely on medicinal plants for their daily need for years and investigations on these plants have been in attention more for scientists in recent days. Artemisia is one of such genera that is very beneficial medicinal plant, but the species, A. indica, is not that much explored till now. So, this chapter covers its history of traditional uses in different places for different diseases and its photochemistry, vast pharmacological activities, and clinical studies with safety and toxicity. Various phytochemical components have been isolated from the plant and identified which has enormous health benefits. Several studies on the extracts of A. indica have shown that it has numerous pharmacological properties such as antimalarial, antitumor, anticancer, anthelmintic, antihyperglycemic, anti-inflammatory, anticonvulsant, antidepressant, and many more. Many in vitro and in vivo experiments are mentioned regarding to its different pharmacological properties. This plant is generally is considered only as a weed and has been neglected but many more medicinal uses, and experiments should be performed and established on it. In recent times, research interests have been shifted towards the exploration of biologically active components or natural components for the health benefits so various phytochemical components from different plant parts of A. Indica should be assessed more to know and understand it’s health benefits. To use it more efficiently and before the clinical applications, the toxicity or safety levels or doses should also need to be evaluated. When this plant has already gained attention in the world because of its traditional uses to combat malaria, this information summarized in this chapter thereby is proposed to act as a reference tool to the people interested in this plant and to others for understand the importance of this beneficial plant, also for performing several experiments and research on it to understand it more.
References
Abood WN, Fahmi I, Abdulla MA, Ismail S (2014) Immunomodulatory effect of an isolated fraction from Tinospora crispa on intracellular expression of INF-γ, IL-6 and IL-8. BMC Complement Altern Med 14(205). https://doi.org/10.1186/1472-6882-14-205
Adewumi OA, Singh V, Singh G (2020) Chemical composition, traditional uses and biological activities of artemisia species. J Pharmacogn Phytochem 9(5):1124–1140
Afzal S, Afzal N, Awan MR, Khan TS, Gilani A, Khanum R, Tariq S (2009) Ethno-botanical studies from Northern Pakistan. J Ayub Med Coll Abbottabad 21(1):52–57
Ahmad N, Anwar S, Fazal H, Abbasi BH (2013) Medicinal plants used in indigenous therapy by people of Madyan Valley in district Swat, Pakistan. Int J Med Aromat Plants 3(1):47–54
Ahmad W, Khan I, Khan MA, Ahmad M, Subhan F, Karim N (2014) Evaluation of antidiabetic and antihyperlipidemic activity of Artemisia indica Linn (aeriel parts) in Streptozotocin induced diabetic rats. J Ethnopharmacol 151(1):618–623. https://doi.org/10.1016/j.jep.2013.11.012
Azab A, Nassar A, Azab AN (2016) Anti-inflammatory activity of natural products. Molecules 21(10):1321. https://doi.org/10.3390/molecules21101321
Baldissera MD, Souza CF, Grando TH, Doleski PH, Boligon AA, Stefani LM, Monteiro SG (2017) Hypolipidemic effect of β-caryophyllene to treat hyperlipidemic rats. Naunyn Schmiedeberg's Arch Pharmacol 390(2):215–223
Bhattarai KR (2020) Ethnobotanical survey on plants used in Mai municipality of Ilam district, eastern Nepal. Banko Janakari 30(2):11–35. https://doi.org/10.3126/banko.v30i2.33476
Chan EWC, Wong SK, Chan HT (2018) Casticin from Vitex species: a short review on its anticancer and anti-inflammatory properties. J Integr Med 16(3):147–152. https://doi.org/10.1016/j.joim.2018.03.001
Chanda S, Nagani K (2013) Vitro and in vivo methods for anticancer activity evaluation and some Indian medicinal plants possessing anticancer properties: an overview. J Pharmacogn Phytochem 2(2):140–152
Chanphen R, Thebtaranonth Y, Wanauppathamkul S, Yuthavong Y (1998) Antimalarial principles from Artemisia indica. J Nat Prod 61(9):1146–1147. https://doi.org/10.1021/np980041x
Das SS, Dey M, Ghosh AK (2011) Determination of anthelmintic activity of the leaf and bark extract of Tamarindus indica Linn. Indian J Pharm Sci 73(1):104–107. https://doi.org/10.4103/0250-474X.89768
Elmogahzy YE (2020) Finished fibrous assemblies. In: Engineering textiles, Integrating the design and manufacture of textile products. The Textile Institute book series, 2nd edn. Woodhead Publishing, Duxford, pp 275–298. https://doi.org/10.1016/B978-0-08-102488-1.00011-3
Ferreira JFS, Luthria DL, Sasaki T, Heyerick A (2010) Flavonoids from Artemisia annua L. as antioxidants and their potential synergism with artemisinin against malaria and cancer. Molecules 15(5):3135–3170. https://doi.org/10.3390/molecules15053135
Flowers of India (2021) Artemisia indica – Indian Wormwood. http://www.flowersofindia.net/catalog/slides/Indian%20Wormwood.html. Accessed 17 Nov 2021
Foster S, Duke JA (2020) A field guide to medicinal plants. Eastern and Central N. America. Houghton Mifflin Co. 1990 a concise book dealing with almost 500 species. A line drawing of each plant is included plus colour photographs of about 100 species. Very good as a field guide, it only gives brief details about the plants medicinal properties
Gallo MBC, Sarachine MJ (2009) Biological activities of Lupeol. Int j res pharm biomed sci 3(1):46–66
Ganguly S, Bandyopadhyay S, Bera A, Chatterjee M (2006) Antipromastigote activity of an ethanolic extract of leaves of Artemisia indica. Indian J Pharmacol 38(1):64–65
Gou Y, Li Z, Fan R, Guo C, Wang L, Sun H, Li J, Zhou C, Wang C, Wang Y (2020) Ethnobotanical survey and evaluation of traditional mosquito repellent plants of Dai people in Xishuangbanna, Yunnan Province, China. J Ethnopharmacol 262
Guclu G, Kelebek H, Selli S (2021) Chapter 26 - Antioxidant activity in olive oils. In: Preedy VR, Watson RR (eds) Olives and olive oil in health and disease prevention, 2nd edn. Academic Press, Amsterdam, pp 313–325. https://doi.org/10.1016/B978-0-12-819528-4.00031-6
Haider SZ, Mohan M, Andola HC (2014) Constituents of Artemisia indica Willd. from Uttarakhand Himalaya: a source of davanone. Pharm Res 6(3):257–259. https://doi.org/10.4103/0974-8490.132607
Hou T, Li X, Peng C (2017) Borneol enhances the antidepressant effects of asiaticoside by promoting its distribution into the brain. Neurosci Lett 646:56–61. https://doi.org/10.1016/j.neulet.2017.02.068
Hu J, Wang W, Dai J, Zhu L (2019) Chemical composition and biological activity against Tribolium castaneum (Coleoptera: Tenebrionidae) of Artemisia brachyloba essential oil. Ind Crop Prod 128:29–37
Hussain A (2019) Distribution and molecualr phylogeny of Artemisia plants from Gilgit-Baltistan, Pakistan. http://173.208.131.244:9060/xmlui/handle/123456789/4631
Hussain A (2020) The genus Artemisia (Asteraceae): a review on its Ethnomedicinal prominence and taxonomy with emphasis on foliar anatomy, morphology, and molecular phylogeny. Proc Pak Acad Sci B Life Environ Sci 57(1):1–28
Jaffal SM, Abbas MA (2019) Antinociceptive action of Achillea biebersteinii methanolic flower extract is mediated by interaction with cholinergic receptor in mouse pain models. Inflammopharmacology 27:961–968. https://doi.org/10.1007/s10787-018-0524-7
Jassal PS, Nahid A, Kumar N, Sharma M (2019) Pharmaceutical applications of Artemisia: an overview. Think India J 22(16):597–610
Javid T, Adnan M, Tariq A, Akhtar B, Ullah R, Salam NMAE (2015) Antimicrobial activity of three medicinal plants (Artemisia indica, Medicago falcate and Tecoma stans). Afr J Tradit Complement Altern Med 12(3):91–96. https://doi.org/10.4314/ajtcam.v12i3.11
Joram A, Mahanta D, Das AK (2021) Documentation of wild edible plants of ethnobotanical significance associated with selected tribes of Arunachal Pradesh, India. East Himalayan Society for Spermatophyte Taxonomy. Pleione 15(2):127–140. https://doi.org/10.26679/Pleione.15.2.2021.123-140
Joshi AR, Joshi K (2004) Insecticidal plants of the Bagmati watershed, Nepal: ethnobotany and traditional uses. Bionotes 6(2):37–39
Ken Fern (2022) Tropical plants database. https://tropical.theferns.info/viewtropical.php?id=Artemisia+indica. Accessed 15 Oct 2022
Khan S, Afshan K, Mirza B, Miller JE, Manan A, Irum S, Rizvi SS, Qayyum M (2015) Anthelmintic properties of extracts from Artemisia plants against nematodes. Trop Biomed 32(2):257–268
Khan I, Karim N, Ahmad W, Abdelhalim A, Chebib M (2016) GABA-A receptor modulation and anticonvulsant, anxiolytic, and antidepressant activities of constituents from Artemisia indica Linn. Evid Based Complement Alternat Med 2016:1215393. https://doi.org/10.1155/2016/1215393
Khan I, Ahmad W, Karim N, Ahmad M, Khan M, Tariq SA, Sultana N, Shah R, Khan A, Abdelhalim A (2017) Antidiabetic activity and histopathological analysis of carnosol isolated from Artemisia indica Linn in streptozotocin-induced diabetic rats. Med Chem Res 26:335–343. https://doi.org/10.1007/s00044-016-1750-4
Lee JY, Park H, Lim W, Song G (2020) Therapeutic potential of α,β-thujone through metabolic reprogramming and caspase-dependent apoptosis in ovarian cancer cells. J Cell Physiol 236(2):1545–1558. https://doi.org/10.1002/jcp.30086
Mannan A, Shaheen N, Arshad W, Qureshi RA, Zia M, Mirza B (2008) Hairy roots induction and artemisinin analysis in Artemisia dubia and Artemisia indica. Afr J Biotechnol 7(18):3288–3292
Mannan A, Ahmed I, Arshad W, Hussain I, Mirza B (2011) Effects of vegetative and flowering stages on the biosynthesis of artemisinin in Artemisia species. Arch Pharm Res 34:1657–1661. https://doi.org/10.1007/s12272-011-1010-6
Mansouri MT, Hemmati AA, Naghizadeh B, Mard SA, Rezaie A, Ghorbanzadeh B (2015) A study of the mechanisms underlying the anti-inflammatory effect of ellagic acid in carrageenan-induced paw edema in rats. Indian J Pharmacol 47(3):292–298. https://doi.org/10.4103/0253-7613.157127
Mensah MLK, Komlaga G, Forkuo AD, Firempong C, Anning AK, Dickson RA (2019) Toxicity and safety implications of herbal medicines used in Africa. In: Builders PF (ed) Herbal medicines. Intech Open, London. https://doi.org/10.5772/intechopen.72437
Mwine JP, Damme PV, Gerard K, Charles K (2011) Ethnobotanical survey of pesticidal plants used in South Uganda: case study of Masaka district. J Med Plants Res 5(7):1155–1163
Nahid A, Neelabh C, Navneet K (2017) Antioxidant and antimicrobial potentials of Artemisia Indica collected from the Nepal region. J Pharm Sci Res 9(10):1822–1826
Namsa ND, Mandal M, Tangjang S (2011) Anti-malarial herbal remedies of Northeast India, Assam: an ethnobotanical survey. J Ethnopharmacol 133(2):1–572. https://doi.org/10.1016/j.jep.2010.10.036
Negreiros PDS, Costa DSD, Silva VGD, Lima IBDC, Nunes DB, Sousa FBDM, Araújo TDSL, Medeiros JVR, Santos RFD, Oliveira RDCM (2019) Antidiarrheal activity of α-terpineol in mice. Biomed Pharmacother 110:631–640. https://doi.org/10.1016/j.biopha.2018.11.131
Nigam M, Atanassova M, Mishra AP, Pezzani R, Devkota HP, Plygun S, Salehi B, Setzer WN, Sharifi-Rad J (2019) Bioactive compounds and health benefits of Artemisia species. Nat Prod Commun 14(7). https://doi.org/10.1177/1934578X19850354
Niwano Y, Beppu F, Shimada T, Kyan R, Yasura K, Tamaki M, Nishino M, Midorikawa Y, Hamada H (2009) Extensive screening for plant foodstuffs in Okinawa, Japan with anti-obese activity on adipocytes. Plant Foods Hum Nutr 64:6–10
Ogbru AG (2021) Adult ADHD Medications. 2021-6-9. https://www.rxlist.com/adhd_prescription_drugs/drugscondition.htm
Oliveira GB, Brito RG, Santos PL, Araújo-Filho HG, Quintans JSS, Menezes PP, Serafini MR, Carvalho YMBG, Silva JC, Almeida JRGS, Scotti L, Scotti MT, Shanmugam S, Thangaraj P, Araújo AAS, Quintans-Júnior LS (2016) α-Terpineol, a monoterpene alcohol, complexed with β-cyclodextrin exerts antihyperalgesic effect in animal model for fibromyalgia aided with docking study. Chem Biol Interact 254:54–62. https://doi.org/10.1016/j.cbi.2016.05.029
Rahmatullah M, Biswas KR (2012) Traditional medicinal practices of a Sardar healer of the Sardar (Dhangor) Community of Bangladesh. J Altern Complement Med 18(1):10–19. https://doi.org/10.1089/acm.2011.0395
Rashid S, Rather MA, Shah WA, Bhat BA (2013) Chemical composition, antimicrobial, cytotoxic and antioxidant activities of the essential oil of Artemisia indica Willd. Food Chem 138(1):693–700. https://doi.org/10.1016/j.foodchem.2012.10.102
Rather MA, Dar BA, Shah WA, Prabhakar A, Bindu K, Banday JA, Qurishi MA (2017) Comprehensive GC–FID, GC–MS and FT-IR spectroscopic analysis of the volatile aroma constituents of Artemisia indica and Artemisia vestita essential oils. Arab J Chem 10(2):S3798–S3803. https://doi.org/10.1016/j.arabjc.2014.05.017
Ruwali P, Ambwani TK, Gautam P, Thapliyal A (2015) Qualitative and quantitative phytochemical analysis of Artemisia indica Willd. J Chem Pharm Res 7(4):942–949
Sadraei H, Asghari G, Kasiri F (2015) Comparison of antispasmodic effects of Dracocephalum kotschyi essential oil, limonene and α-terpineol. Res Pharm Sci 10(2):109–116
Sagar MK, Ashok PK, Chopra H, Upadhyaya K (2010) Phytochemical and pharmacological potential of Artemisia indica in experimental animal models. Pharmacologyonline 2:1–4
Salehi B, Ata A, Kumar NVA, Sharopov F, Ramírez-Alarcón K, Ruiz-Ortega A, Ayatollahi SA, Fokou PVT, Kobarfard F, Zakaria ZA, Iriti M, Taheri Y, Martorell M, Sureda A, Setzer WN, Durazzo A, Lucarini M, Capasso ASR, Ostrander EA, Rahman AU, Choudhary MI, Cho WC, Sharifi-Rad J (2019) Antidiabetic potential of medicinal plants and their active components. Biomol Ther 9(10):551. https://doi.org/10.3390/biom9100551
Sarnim G, Sanjay ST, Roshan A, Vedamurthy AB, Joy HH (2013) Artemisia indica extracts as anthelminthic agent against Pheretima posthuma. Int J Pharm Pharm Sci 5(3):259–262
Satyal P, Paudel P, Kafle A, Pokharel SK, Lamichhane B, Dosoky NS, Moriarity DM, Setzer WN (2012) Bioactivities of volatile components from Nepalese Artemisia species. Nat Prod Commun 7(12):1651–1658
Saxena S, Pant N, Jain DC, Bhakuni RS (2003) Antimalarial agents from plant sources. Curr Sci 85(9):1314–1329. http://www.jstor.org/stable/24108135
Shah GC (2010) Terpenoid diversity in some Artemisia species of Uttarakhand Himalaya. Indian Perfumer 54:17–19
Shah GC, Rawat TS (2008) Chemical constituents of Artemisia indica Willd. Oil. Indian Perfumer 52:27–29
Shimono Y, Hayakawa H, Kurokawa S, Nishida T, Ikeda H, Futagami N (2013) Phylogeography of Mugwort (Artemisia indica), a native Pioneer herb in Japan. J Hered 104(6):830–841. https://doi.org/10.1093/jhered/est054
Sigdel SR, Rokaya MB, Timsina B (2013) Plant inventory and ethnobotanical study of Khimti Hydropower Project, Central Nepal. Sci World J 11(11):105–112
Singh B, Sinha BK, Phukan SJ, Borthakur SK, Singh VN (2012) Wild edible plants used by Garo tribes of Nokrek biosphere reserve in Meghalaya, India. Indian J Tradit Knowl 11:166–171
Souza R, Cardoso M, Menezes C, Silva J, De Sousa D, Batista J (2011) Gastroprotective activity of α-terpineol in two experimental models of gastric ulcer in rats. Daru 19(4):277–281
Tasdemir D, Tierney M, Sen R, Bergonzi MC, Demirci B, Bilia AR, Baser KH, Brun R, Chatterjee M (2015) Antiprotozoal effect of Artemisia indica extracts and essential oil. Planta Med 81(12–13):1029–1037
Tiwary BK, Bihani S, Kumar A, Chakraborty R, Ghosh R (2015) The in vitro cytotoxic activity of ethno-pharmacological important plants of Darjeeling district of West Bengal against different human cancer cell lines. BMC Complement Altern Med 15:22. https://doi.org/10.1186/s12906-015-0543-5
Torres Y, Vargas D, Uribe C, Carrasco C, Torres C, Rocha R, Oyarzún C, Martín RS, Quezada C (2016) Pro-apoptotic and anti-angiogenic properties of the α /β-thujone fraction from Thuja occidentalis on glioblastoma cells. J Neurooncol 128(1):9–19
Xu L, Wu YL, Zhao XY, Zhang W (2015) The study on biological and pharmacological activity of Coumarins. Asia-Pac Power Energy Eng Conf 135–138
Xu W, Miao S, Feng Y (2019) Ludartin exhibits therapeutic effect on spinal cord injury through inhibition of apoptosis and inflammation. Bangladesh J Pharmacol 14(1):54–60. https://doi.org/10.3329/bjp.v14i1.38725
Zeng YT, Jiang JM, Lao HY, Guo JW, Lun YN, Yang M (2015) Antitumor and apoptotic activities of the chemical constituents from the ethyl acetate extract of Artemisia indica. Mol Med Rep 11:2234–2240. https://doi.org/10.3892/mmr.2014.3012
Zhu QC, Wang Y, Liu YP, Zhang RQ, Li X, Su WH, Long F, Luo XD, Peng T (2011) Inhibition of enterovirus 71 replication by chrysosplenetin and penduletin. Eur J Pharm Sci 44(3):392–398
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Sanyal, R. et al. (2022). Artemisia indica Willd.: Ethnobotany, Phytochemistry, Pharmacological Attributes, and Safety Profile. In: Devkota, H.P., Aftab, T. (eds) Medicinal Plants of the Asteraceae Family. Springer, Singapore. https://doi.org/10.1007/978-981-19-6080-2_3
Download citation
DOI: https://doi.org/10.1007/978-981-19-6080-2_3
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-6079-6
Online ISBN: 978-981-19-6080-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)