Abstract
Artemisia roxburghiana Wall. ex Besser, belongs to the family Asteraceae (Compositae), is widely used in traditional medicine in Asia to treat various ailments like diabetes, rheumatism, malaria, hepatitis and helminths. In the present study, the relevant literature collected from various offline and online sources including Scopus, Web of Science and PubMed, was thoroughly reviewed for Phytochemistry, Ethnobotany and Pharmacology of A. roxburghiana. The literature survey revealed that A. roxburghiana contains a wide range of secondary metabolites like flavonoids, terpenoids, coumarins, steroids and fatty acids. The plant has been reported to be an important source of many bioactive compounds such as artemisinin, scopoletin and taraxeryl acetate. It showed anti-protozoal, anthelmintic, anti-inflammatory and antidiabetic activities in various experimental models. The present review concludes that the plant can be used to treat many human ailments; however, further research is warranted to develop it as a medicine.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Himalayas, a mountain range in Asia covering mainly China, India, Nepal, Pakistan and Tibet is also known as a treasure house of the medicinal plants with around 8000 known species in which 1748 species are known to have medicinal value (Singh and Hajra 1996). The lower Himalayan region has a rich diversity of medicinal plants which are well-used by the indigenous people for their healthcare (Joshi et al. 2016). In addition to the dense forest, this region is known for the cultivation of various crops and medicinal plants such as Centella asiatica, Boerhavia diffusa, Solanum nigrum, Eclipta alba, Desmodium gangeticum, Withania somnifera, Uraria picta, Sida cordifolia, Cissampelos pareira, Sphaeranthus indicus, Ocimum sactum, Acorus calamus, Mentha piperta, Cymbopogon citratus, Vetiveria zizanioides, Cassia angustifolia and Artemisia nilagirica. Many of these species are exotic in the region while others are endemic (Chitale et al. 2014). A majority of the rural population in India, China, Nepal and Tibet is mainly depending on traditional medicine such as Ayurveda in India and Chinese traditional medicine in China. The major sources of the traditional medicine are plants but due to increasing population, deforestation, increasing demand of traditional medicine and uncontrolled harvesting, the sources for the herbal medicines are exhausting very fast (Ekor 2013). Hence, there is an urgent need to either cultivate in higher scale or to discover the substitutes for such plants which are either near to threatened or having limited source in comparison of market demand.
The genus Artemisia belongs to the family Asteraceae (Compositae) and mainly consists of herbs and shrubs. Most of these plants have volatile oils with strong aromas and bitter tastes due to the presence of alcohols, terpenes, and sesquiterpene lactones (Wright 2003). According to The Plant List (2013), the genus Artemisia contains 481 accepted species whereas 288 species are still unassessed. This is a valuable genus of economic importance because of its use in pharmaceutical and food industries (Phan et al. 2012). Although most of these plants are used as folk medicine, food, forage, ornamentals and soil stabilizers in disturbed habitats, few species are recorded to be toxic or allergenic while some others are invasive weeds which can adversely affect the crop yield (Ashraf et al. 2010a). The plants of this genus are also known for their anthelmintic property in addition to its other therapeutical importance (Khan et al. 2015). Interestingly, because of the high therapeutical index, selected species such as A. absinthium L., A. annua L. and A. vulgaris L. have been incorporated into the pharmacopoeias of several European and Asian countries (Ashraf et al. 2010a).
Many plants of the genus Artemisia (like A. indica, A. roxburghiana, A. vulgaris, etc.) are very close in terms of their physical appearance, which makes their identification rather difficult. Hence, without studying the detailed taxonomic characteristics, the identification of Artemisia species is certainly a big challenge (Hayat et al. 2009). Sometimes, these plants can be identified with the help of some common taxonomical features such as densely hairy plant body, pinnatifid to pinnatisect leaves with variable dimensions, capitulum inflorescence with a paniculate-raceme arrangement, yellow or green corolla, bisexual disk florets and obovoid to oblong cypselas (Wright 2003). The leaves of many species are covered with white hairs. Most of the members of this genus, such as A. capillaris and A. nilagirica contain volatile constituents including linalool, capillene, ocimene, limonene, pinene, thujone, myricene and citral (Badoni et al. 2010; Semwal et al. 2015). These plants also showed variation in their constituents when collected from different altitude, season and geography (Badoni et al. 2009).
The plant Artemisia roxburghiana Wall. ex Besser, also known as Roxburgh’s Wormwood is one of the least explored folk medicinal plants of the genus Artemisia. Vernacularly, the plant is also called Kuranja in Garhwal Himalayas (Dua et al. 2011), Chota Afsantin, Garrotra, Jangli Ajwain in Pakistan (Adnan et al. 2015), Ngải rừng in Vietnam (Giang et al. 2013) and Hui bao hao in China (Zhang 2011). The creeping rootstock, simple stem, deeply dissected woolly leaves and purple flowers are some characteristics of the plant (Bicchi et al. 1998). The plant grows in the lower or mid-altitude of lower Himalayas from 500 to 4000 m. It is used as a folk medicine in various Asian countries for the treatment of malarial fever, skin disorders and intestinal worms. However, many of its traditional properties are not yet validated scientifically. A critical review based on the geographical distribution, phytochemical analysis and pharmacological importance of A. roxburghiana is presented here for the first time.
Botanical description of Artemisia roxburghiana
A. roxburghiana is a perennial herb or subshrub of about 45–100 cm tall (Giang et al. 2013). Leaves are abaxially densely arachnoid tomentose, adaxially sparsely pubescent and gland-dotted, or glabrescent; twice cut with linear pointed segments; 2 mm broad. The leaf blade is ovate to oblong-elliptic in shape, 6–10 cm × 4–6 cm, 2-pinnatisect; segments are elliptic to oblong in shape. Lateral lobes have 1–3 pairs of lobules which are lanceolate to linear-lanceolate, 0.5–1.5 mm × 2–2.5 mm with narrowly winged rachis. The uppermost leaves and leafy bracts are pinnatisect with 3–4 lobes. Lobes and entire bracts are linear-lanceolate to lanceolate. The branches are simple, to 4 cm, erect, uniform in length; involucral bracts are purple and hairy. Ovoid to subglobose capitula are in the broad paniculate arrangement, 2–3 mm in diameter. The flowers are borne in spikes occurring on branches and at their ends. Ray florets are 4–8 in number and disc florets are 8–15 in number; bisexual; corolla filiform; corolla limb yellow or purple; the flowering season is between June and September (Hayat et al. 2009; Zhang 2011).
Geographical distribution of Artemisia roxburghiana
The members of the genus Artemisia are wind pollinated cosmopolitans, mainly distributed in temperate areas of mid to high latitudes of the northern hemisphere, colonizing in arid and semiarid environmental landscape, with only a few representatives in the southern hemisphere such as A. vulgaris, A. tridentata, A. annua, A. absinthium, A. dracunculus and A. abrotanum. Central Asia is its centre of diversification, while the Mediterranean region and North West America are two secondary speciation areas. A few species are also reported in Africa (such as A. afra) and Europe (such as A. vulgaris) (Hayat et al. 2009).
The plant A. roxburghiana is found on the open slopes of the Himalayas, from Pakistan to Central Nepal, at altitudes of 1000–4300 m. It has also been cultivated in Trento, Italy at an altitude of 800 m and in Padua, Italy at an altitude of 200 m (Bicchi et al. 1998). In Nepal, it is well distributed at altitudes between 2600 and 4600 m in the Kumbu valley. In China, the plant is found in roadsides, slopes, dry canyons, grasslands, waste areas, terraces at an altitude between 700 and 3900 m in South Gansu, Guizhou, West Hubei, Qinghai, South Shaanxi, West Sichuan, Xizang and Yunnan. Galiat, Ladakh, Kurram Agency and Rawalpindi are the main distribution places in Pakistan (Ashraf et al. 2010b). The species is also recorded in North Thailand, North-East Afghanistan and Vietnam (Giang et al. 2013). In India, it is well distributed as wild population located on shady rocky humid soils, dry open slopes and wasteland on the foothills of Indian Himalaya at altitudes 1000–4300 m (Mathela et al. 1994). The country-wise distribution of A. roxburghiana is given in Fig. 1.
Ethnobotanical uses of Artemisia roxburghiana
The plant A. roxburghiana is used in traditional medicine for the treatment of various human ailments. The people of Vietnam used the aerial parts of the plant to treat fever and intestinal worms in the form of a decoction (Giang et al. 2013). It has been found as one of the most used traditional medicinal plants in Pakistan. The water extract obtained from the leaves is used by the natives of the Himalayan region against various health problems such as malarial fever, rheumatism, dysentery and hepatitis (Shah et al. 2016). In addition, the decoction obtained from the aerial parts of the plant either alone or in combination with the dried fruits of Zizyphus jujube are used to treat diabetes in Pakistan. The local community of northern Pakistan uses the extract of the whole plant for treating malarial fever (Ashraf et al. 2010b) while the people from Rawalpindi used the plant powder against intestinal worms (Adnan et al. 2015). The decoction of the whole plant powder is used against hyperglycemia and helminths in Pakistan (Hayat et al. 2009). A decoction from the leaves and other aerial parts of the plant has been a popular folk medicine in various parts of India mainly against protozoal infection, malarial fever and skin disorders (Dua et al. 2011). In Garhwal region of North West Himalayas, the powder of the whole plant is used as a tonic as well as an anti-allergic medicine (Nehra 2014). The fresh leaves of the plant are used for skin allergy in Uttarakhand, India by rubbing them on the affected area (Bhat et al. 2013).
Phytochemical description of Artemisia roxburghiana
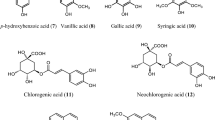
A. roxburghiana possesses essential oil and other volatile constituents as well as alcohols, coumarins, fatty acids, flavones, sterols and terpenes (Shah et al. 2014). The leaves contain sesquiterpene lactones (roxbughianin A and B, achillin), fatty acids (eicosanoic acid, palmitic acid) and fatty alcohol (1-octacosanol) as key constituents (Giang et al. 2013). The main constituents reported from the essential oil obtained from the aerial parts of A. roxburghiana are nerolidol, tibetin spiroether, ocimene, cineole, curcumene, borneol, camphene, camphor, carvone, eugenol, limonene, linalool, elemene, and terpinene (Bicchi et al. 1998). In addition, various other bioactive compounds such as betulin, betulinic acid, scopoletin, taraxerol acetate (a pentacyclic triterpene) and artemisinin were also recorded from the aerial parts (Mannan et al. 2010; Shah et al. 2016; Rahman et al. 2016a). Shahwar et al. (2013) reported the yield of artemisinin as 0.005% (dry weight) in the whole parts of the plant collected from Muree (Pakistan) whereas Rahman et al. (2016b) reported the yield of taraxerol acetate as 0.01% (dry weight) in the aerial parts of the plant collected from Mansehra (Pakistan). Shah et al. (2016) isolated apigenin-7,4′-dimethyl ether (48.4 mg), taraxeryl acetate (271 mg), betulin (9 mg), betulinic acid (9.3 mg), scopoletin (42 mg) and 6,7-dimethoxy-coumarin (5 mg) from 17 kg dry powder of aerial parts collected from Mansehra (Pakistan).
A study conducted by Haider et al. (2009) revealed that the plants found in high altitude have comparatively lower contents of the volatile oil with borneol, linalyl acetate and α-humulene as the major compounds. In addition to various volatile and non-volatile constituents, many trace elements such as cadmium, calcium, chromium, cobalt, copper, iron, lead, magnesium, manganese, nickel, potassium, sodium and zinc were also detected from the plant (Ashraf et al. 2010a). The phytochemicals isolated from A. roxburghiana are given in Table 1 and the chemical structures of the isolated constituents are shown in Fig. 2.
Pharmacological importance of Artemisia roxburghiana
Based on the occurrence of the plant i.e. in Asia, various studies have been conducted in the region to evaluate its biological efficacy against a variety of diseases. The pharmacological studies done with various parts of the plant are mainly based on the traditional uses of the plant such as antimalarial, anthelmintic and antidiabetic activities. The crude extract as well as some purified compounds isolated from A. roxburghiana also showed anti-inflammatory, antimicrobial and protease inhibitory activities. Artemisinin, isolated from the aerial parts of A. roxburghiana, inhibited trypsin in vitro with an IC50 value of 0.29 mM (Shahwar et al. 2013). Leaf extracts have been found active against conidial germination of the powdery mildew fungus Erysiphe cichoracearum, a plant pathogen that causes powdery mildew disease of cucurbits (Mir et al. 2012). Detailed pharmacological activities of the plant are given below.
Anti-inflammatory and antipyretic activities
Inflammation occurs if the body tissues are injured by any of the causes, including a bacterial infection, wounds, trauma, heat, etc. The damaged cells release various chemicals like histamine, bradykinin, prostaglandins, etc. that cause blood vessels to discharge fluid into the tissues which causes inflammation in the damaged area. The mechanism of inflammation, based on carrageenan-induced oedema (Rahman et al. 2016a), is given in Fig. 3. Changes in blood flow, destruction of tissues, increased vascular permeability and the synthesis of pro-inflammatory mediators are involved in this process. The injured cells, lymphocytes, phagocytes, mast cells and blood proteins are the sources of inflammatory mediators including bradykinins, serotonins, histamine, IL-6, leukotrienes, TNF-α, IL-1β, phospholipase A2, NO, lipoxygenases and COX-2 (Bian et al. 2012). This is usually a defence process of the body which helps in destroying the foreign substance from the body. Inflammation is characterised by an increase in permeability of blood vessels, extravasation of fluid and proteins and accumulation of WBCs for a short time (Markiewski and Lambris 2007).
General mechanism of inflammation based on carrageenan-induced oedema. PMN polymorphonuclear leukocytes (neutrophil), TNF tumour necrosis factor, IL1 interleukin-1, cNOS constitutive nitric oxide synthase, iNOS inducible nitric oxide synthase, COX2 cyclooxygenase-2, NO nitric oxide; PG prostaglandins
The pentacyclic triterpene derivative taraxerol acetate, isolated from the ethyl acetate fraction of a methanol extract of the aerial parts showed in vitro inhibitory activity against both COX1 and COX2 with IC50 values of 116.3 and 94.7 μM, respectively (Rehman et al. 2013). The molecular docking study showed the interactions of this molecule with some key amino acids surrounding the inhibitor in the binding pocket of COX2. In addition, taraxerol acetate was also found effective in vivo against carrageenan-induced paw oedema in rats. It significantly reduced oedema in 3 h at an oral dose of 60 mg/kg body weight. Indomethacin, a nonsteroidal anti-inflammatory drug at a dose of 5 mg/kg body weight was used as a positive control (Rahman et al. 2016a). An in vitro study conducted by Shah et al. (2014) revealed that scopoletin, a coumarin isolated from the methanol extract of the whole plant showed significant inhibition of nitric oxide (NO) production in the mouse macrophage J774 cells by 78% at a concentration of 25 µg/mL. This study also found that scopoletin (25 µg/mL) capped with gold nanoparticles inhibited the NO production by 21.6%. NG-Monomethyl-l-arginine acetate was used as a positive control which showed inhibition by 65%. This study revealed that conjugation to Au nanoparticles does not affect the anti-inflammatory activity of scopoletin. The study also found that scopoletin conjugation to Au nanoparticles was 3 times more efficient than scopletin itself in preventing the oxidative burst induced by ROS in phagocytes and isolated neutrophils. The activity was compared with ibuprofen, a positive control, which showed activity with an IC50 of 11.2 µg/mL against whole blood phagocytes and 2.5 µg/mL against isolated neutrophils.
The mechanism for the anti-inflammatory activity of scopoletin was earlier demonstrated by Ding et al. (2008). This compound significantly inhibited croton oil-induced mouse ear oedema and also decreased the vascular dye leakage induced by topical application of croton oil, consistent with the reduced myeloperoxidase activity and polymorphonuclear infiltration. Besides, scopoletin was found to decrease the overproduction of PGE2 and TNF-α. Scopoletin also reduced the carrageenan-induced paw oedema in mice and lowered the myeloperoxidase activity and malondialdehyde level in paw tissues (Ding et al. 2008).
In addition to the anti-inflammatory activity, taraxerol acetate showed antipyretic activity by decreasing the body temperature of experimental rats. Its initial action was recorded as similar as aspirin (Rahman et al. 2016b). The compound at the doses of 30 and 60 mg/kg was found effective against yeast-induced pyrexia up to 3 h, although the clear mechanism of this activity is not known.
Antiparasitic activity
Parasitic diseases such as malaria, African trypanosomiasis and visceral leishmaniasis are one of the major health problems and causes of death worldwide. Malaria is the most common parasitic disease caused by Plasmodium parasites spread to people through the bites of infected female Anopheles mosquitoes. According to the recent report of WHO (World malaria report 2017), about 237 million, 211 million and 216 million cases of malaria were estimated worldwide in 2010, 2015 and 2016, respectively. In 2016, there were estimated 445,000 deaths from malaria globally. Leishmania donovani, a human blood parasite causes visceral leishmaniasis, also known as kala-azar which transmitted to humans by sand flies. This parasite infects the mononuclear phagocyte system which includes spleen, liver and bone marrow) (Stuart et al. 2008). After malaria, visceral leishmaniasis is the second most important parasitic killer in the world, responsible for about two to four hundred thousand cases each year worldwide (WHO Fact sheet 2017). Trypanosoma brucei, a pathogenic parasite has three subspecies i.e. brucei, gambiense and rhodesiense. T. brucei gambiense and T. brucei rhodesiense are the causative pathogens for African trypanosomiasis, also known as sleeping sickness whereas T. brucei brucei is responsible for the trypanosomiasis or nagana in animals (Baker 1995).
Antiprotozoal activity
A. roxburghiana is used in traditional medicine for the treatment of malaria which was validated by an in vitro study against Plasmodium falciparum, L. donovani, Trypanosoma brucei rhodesiense and Trypanosoma cruzi. The chloroform extract of A. roxburghiana showed strong activity against P. falciparum, L. donovani, T. brucei rhodesiense and T. cruzi with the IC50 values of 0.42, > 16, > 8 and > 8 μg/mL, respectively (Dua et al. 2011). In addition, the petroleum ether extract showed activity against the above-mentioned parasites with IC50 values of 3.03, > 16, 6 and > 8 μg/mL, respectively, whereas the methanol extract showed activity with IC50 values of > 5, > 16, > 8 and > 8 μg/mL, respectively. The drugs chloroquine (IC50 0.051 µg/mL), melarsoprol (IC50 0.005 µg/mL), benznidazole (IC50 0.427 µg/mL) and miltefosine (IC50 0.142 µg/mL) were used as positive controls against P. falciparum, T. brucei rhodesiense, T. cruzi and L. donovani, respectively. It can be suggested that the activity showed by the extracts of A. roxburghiana, mainly by the chloroform extract, is due to the presence of artemisinin among other similar constituents which are already proven to be strong antimalarial agents.
Artemisinin, a sesquiterpene lactone endoperoxide, has firstly been isolated by Tu from A. annua in 1972 as the main antiplasmodial compound from A. annua, used for millennia in Asia to cure malaria fevers (among other diseases). It has been found to be a promising agent for malaria treatment including chloroquine resistant parasites. Artemisinin isolated from A. roxburghiana was found effective against both simple and complicated malaria (Cumming et al. 1997). It contains a peroxide bridge which is believed to be the key to its mode of action against Plasmodium (Klayman 1985). The journey of this molecule from its discovery to development as an anti-malarial drug is very interesting (Tu 2011). The mechanism-based studies suggested that the molecule generates free radicals after reacting with haem and iron (II) oxide, and damages susceptible proteins, which ultimately causes the death of the parasite (Winzeler and Manary 2014). Artemisinin derivatives including artesunate (a first line treatment for severe malaria) are now recommended by WHO as reference treatment for malaria (Bhattarai et al. 2007). In 2015, Youyou Tu has been awarded the Nobel Prize in Medicine for her discoveries concerning a novel therapy against malaria.
Anthelmintic activity
Helminth infection is one of the most common infections in man which affects a large proportion of world’s population by causing malnutrition, anaemia, eosinophilia, and pneumonia (Das et al. 2011). A methanol extract from the aerial parts of A. roxburghiana showed in vitro anthelmintic activity against the mixed infection of gastrointestinal nematodes (Haemonchus contortus and Telodorsagia spp.). The extract at a concentration of 50 mg/mL showed egg hatch inhibition, larval mortality and adult worm mortality by 80, 17 and 8%, respectively. However, these results were found statistically insignificant (p > 0.05) when compared to the negative control (phosphate buffer saline). Doses lower than 50 mg/mL (i.e., 25, 12.5, 6.25 and 3.75 mg/mL) were found ineffective against both nematodes. Albendazole (10%) was used as a positive control (Khan et al. 2015). This study was conducted with the crude extract; no pure compound was used to evaluate the activity.
Antidiabetic activity
Diabetes is one of the most deadly metabolic syndromes in today’s life due to the use of junk food, lack of exercise and too much stress. Nowadays, hundreds of medicines are available in the market to manage the sugar level in type 2 diabetic patients, not a single medicine is used to cure diabetes completely i.e. to revive beta cells to secrete adequate insulin and normalise the body function for the glucose utilisation. Available medicines, particularly the synthetic medicines, show numerous side effects including heartburn, stomach ache, nausea, bloating, diarrhoea, constipation and obesity (Cefalu 2007). Recent studies revealed that there are many genes, hormones, proteins or enzymes are involved in the mechanism of diabetes that cause insulin resistance or the damage of pancreatic beta cells (Berlanga-Acosta et al. 2013).
The methanol extract from the aerial parts of A. roxburghiana was found to inhibit the activity of protein tyrosine phosphatase 1B (PTP1B), a therapeutic target for type 2 diabetes mellitus, obesity and related states of insulin resistance, by 59.2% at a concentration of 100 µg/mL. An ethyl acetate fraction of the methanol extract showed PTP1B inhibitory activity by 68.2% at a similar concentration. Two compounds, betulinic acid and betulin displayed PTP1B inhibitory activity with IC50 values of 3.49 and 4.17 µM, respectively, whereas ursolic acid, used as a positive control, showed activity with an IC50 value of 3.21 µM. The structure–activity relationship study revealed that C-3 and C-17 positions of betulinic acid and betulin are the possible centres for the activity (Shah et al. 2016). The ethanol extract of the leaves at 10, 20 and 40 μg/mL showed in vitro insulin secretagogue activity on an insulinoma cell line (INS-1 cells) in the presence of glucose using rat insulin as a standard, (mono-1251-Tyr A14)-porcine as a labelled compound and anti-insulin antibodies. Glibenclamide, an antidiabetic drug was used as a positive control which showed insulin secretagogue activity at 1 μg/mL (Hussain et al. 2004).
An in vivo study revealed that betulin, a key compound of this plant has potential to inhibit the maturation of sterol regulatory element-binding proteins, which required for cholesterol and fatty acids biosynthesis. The study found a decrease in the biosynthesis of cholesterol and fatty acids (Tang et al. 2011). Besides, this molecule ameliorated fat diet-induced obesity, decreased the lipid contents in serum and tissues, increased insulin sensitivity and reduced the size and improved the stability of atherosclerotic plaques.
Anticancer activity
Various preclinical studies suggested that betulin is effective against a variety of tumours (Alakurtti et al. 2006). It has been found that betulin causes some types of tumour cells to start apoptosis and slow down the growth of tumour cells. Likewise, taraxerol, a parent compound of taraxerol acetate was found effective in inhibiting the AGS cell proliferation. It arrested the human gastric carcinoma AGS cells at G(2)/M stage at 110 μmol/L and elevated the population of AGS cells arrested in G(2)/M phase. The molecule also promoted early cell apoptosis rate from 4.45 to 10.29% in AGS cells at a similar concentration. Taraxerol acetate showed lower activity than taraxerol (Tan et al. 2011).
Toxicity studies
Cytotoxicity assays of petroleum ether, chloroform and methanol extracts of the leaves of A. roxburghiana were performed against rat skeletal myoblasts L6-cells (normal cells). All the extracts showed low toxicity with IC50 values of 23.7, 32.6 and > 90 µg/mL, respectively, while the positive control, podophyllotoxin showed activity against L6-cells with an IC50 of 0.004 µg/mL (Dua et al. 2011). An analytical study on the heavy metal contents by Ashraf et al. (2010a) revealed that the plant A. roxburghiana, collected from the northern area of Pakistan contained lead concentrations above the permissible limit. However, according to international safety standards, high concentrations of heavy metals in herbal medicine can cause metal poisoning in humans. Lead is considered a poisonous metal, its higher concentration may cause both acute and chronic poisoning on brain, kidney, liver and lungs, and even damage the immune system (Wani et al. 2015).
Conclusion and perspectives
Artemisia roxburghiana has been used for the treatment of fever; however, this activity is not yet authenticated scientifically. The earlier literature showed that all parts of the plant are effective against skin disorders which should be evaluated clinically. These plants showed variation in their constituents when collected from different altitude, season and geography; hence, there should be an optimum time, temperature and habitation for the harvesting of A. roxburghiana for medicinal purpose. In view of its medicinal utility, Banerjee et al. (2010) used an apical meristem culture to regenerate the phytoplasma-free A. roxburghiana in vitro. This technique can be used to cultivate this wild medicinal plant in high scale in some cultivation sites.
Various experimental studies confirmed the antitumour activity of betulin, a key compound of A. roxburghiana. It was also found effective against hyperglycemia and hyperlipidemia; it reduces cholesterol and fatty acids biosynthesis in selected experimental models. Besides, this compound ameliorated fat diet-induced obesity, decreased the lipid contents in serum and tissues and increased insulin sensitivity in animal models. Hence, on the basis of above studies, A. roxburghiana can be developed as a new drug in future mainly against malaria, obesity and diabetes due to presence of various bioactive compounds such as artemisinin, betulinic acid, betulin, scopoletin, β-sitosterol, apigenin and quercetin derivatives.
The thorough literature analysis revealed that although the plant is used in the traditional medicine in various Asian countries for a variety of human ailments, its scientific validation by clinical trials is still an assignment of future research. The most striking traditional uses of the plant A. roxburghiana are its anti-inflammatory, anti-parasitic and antidiabetic activities which are validated scientifically through preliminary studies i.e. in vitro studies. Although few in vivo studies were also conducted to validate its traditional uses, the mechanism of action is still unknown.
The plant was found to be a rich source of essential oil which contains a variety of bioactive compounds such as curcumene, ocimene, limonene and terpinene. This oil can be used commercially for the perfumery and soap industries as well as for the Panchakarma therapy and body massage.
Abbreviations
- IL-1β:
-
Interleukin 1β
- IL-6:
-
Interleukin 6
- TNF-α:
-
Tumor necrosis factor α
- NO:
-
Nitric oxide
- COX-1:
-
Cyclooxygenase 1
- COX-2:
-
Cyclooxygenase 2
- PGE2:
-
Prostaglandin E2
- IC50 :
-
Half maximal inhibitory concentration
- AGS:
-
Adenocarcinoma gastric cell line
- WBCs:
-
White blood cells
References
Adnan M, Bibi R, Azizullah A, Andaleeb R, Mussarat S, Tariq A, Ullah R, AbdElsalam NM, Khan AL, Begum S (2015) Ethnomedicinal plants used against common digestive problems. Afr J Tradit Complement Altern Med 12:99–117
Alakurtti S, Makela T, Koskimies S, Yli-Kauhaluoma J (2006) Pharmacological properties of the ubiquitous natural product betulin. Eur J Pharm Sci 29:1–13
Ashraf M, Hayat MQ, Jabeen S, Shaheen N, Khan MA, Yasmin G (2010a) Artemisia L. species recognized by the local community of northern areas of Pakistan as folk therapeutic plants. J Med Plants Res 4:112–119
Ashraf M, Hayat MQ, Mumtaz AS (2010b) A study on elemental contents of medicinally important species of Artemisia L. (Asteraceae) found in Pakistan. J Med Plants Res 4:2256–2263
Badoni R, Semwal DK, Rawat U (2009) Altitudinal variation in the volatile constituents of Artemisia nilagirica. Int J Essent Oil Ther 3:66–68
Badoni R, Semwal DK, Rawat U (2010) Composition variation in essential oils of Artemisia nilagirica and Artemisia capillaris, growing in India. J Appl Nat Sci 2:30–33
Baker JR (1995) The subspecific taxonomy of Trypanosoma brucei. Parasite 2:3–12
Banerjee S, Haider F, Bagchi GD, Samad A (2010) Regeneration of phytoplasma-free Artemisia roxburghiana Besser var. purpurascens (Jacq.) Hook. plants using apical meristem culture. Plant Cell Tissue Organ Cult 103:189–196
Berlanga-Acosta J, López-Saura P, Guillen-Pérez I, Guillen-Nieto G, Acevedo-Castro B, Herrera-Martínez L (2013) Type 2 Diabetes Mellitus (T2DM): biological overview from pathways to organelles and its translation toward a torpid wound healing process. J Diabetes Metab 4:285
Bhat JA, Kumar M, Bussmann RW (2013) Ecological status and traditional knowledge of medicinal plants in Kedarnath Wildlife Sanctuary of Garhwal Himalaya, India. J Ethnobiol Ethnomed 9:1
Bhattarai A, Ali AS, Kachur SP, Mårtensson A, Abbas AK, Khatib R, Al-Mafazy AW, Ramsan M, Rotllant G, Gerstenmaier JF, Molteni F, Abdulla S, Montgomery SM, Kaneko A, Björkman A (2007) Impact of artemisinin-based combination therapy and insecticide-treated nets on malaria burden in Zanzibar. PLoS Med 4:e309
Bian Z, Guo Y, Ha B, Zen K, Liu Y (2012) Regulation of the inflammatory response: enhancing neutrophil infiltration under chronic inflammatory conditions. J Immunol 188:844–853
Bicchi C, Rubiolo P, Marschall H, Weyerstahl P, Laurent R (1998) Constituents of Artemisia roxburghi—ana Besser essential oil. Flavour Fragr J 13:40–46
Bicchi C, Binello A, D’Amato A, Rubiolo P (1999) Reliability of Van den Dool retention indices in the analysis of essential oils. J Chromatogr Sci 37:288–294
Cefalu WT (2007) Pharmacotherapy for the treatment of patients with type 2 diabetes mellitus: rationale and specific agents. Clin Pharmacol Ther 81:636–649
Chitale VS, Behera MD, Roy PS (2014) Future of endemic flora of biodiversity hotspots in India. PLoS ONE 9:e115264
Cumming JN, Ploypradith P, Posner GH (1997) Antimalarial activity of artemisinin (qinghaosu) and related trioxanes: mechanism(s) of action. Adv Pharmacol 37:253–297
Das SS, Dey M, Ghosh AK (2011) Determination of anthelmintic activity of the leaf and bark extract of Tamarindus indica Linn. Indian J Pharm Sci 73:104–107
Ding Z, Dai Y, Hao H, Pan R, Yao X, Wang Z (2008) Anti-inflammatory effects of scopoletin and underlying mechanisms. Pharm Biol 46:854–860
Dua VK, Verma G, Agarwal DD, Kaiser M, Brun R (2011) Antiprotozoal activities of traditional medicinal plants from the Garhwal region of North West Himalaya, India. J Ethnopharmacol 136:123–128
Ekor M (2013) The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol 4:177
Giang PM, Nhan TTT, Son PT, Matsunami K, Otsuka H (2013) A new guaianolide from Artemisia roxburghiana. Nat Prod Res 27:1856–1858
Haider F, Kumar N, Banerjee S, Naqvi AA, Bagchi GD (2009) Effect of altitude on the essential oil constituents of Artemisia roxburghiana besser var. Purpurascens (jacq.) Hook. J Essent Oil Res 21:303–304
Hayat MQ, Khan MA, Ashraf M, Jabeen S (2009) Ethnobotany of the genus Artemisia L. (Asteraceae) in Pakistan. Ethnobot Res Appl 7:147–162
Hussain Z, Waheed A, Qureshi RA, Burdi DK, Verspohl EJ, Khan N, Hasan M (2004) The effect of medicinal plants of Islamabad and Murree region of Pakistan on insulin secretion from INS-1 cells. Phytother Res 18:73–77
Joshi RK, Satyal P, Setzer WN (2016) Himalayan aromatic medicinal plants: a review of their ethnopharmacology, volatile phytochemistry, and biological activities. Medicines 3:6
Khan S, Afshan K, Mirza B, Miller JE, Manan A, Irum S, Rizvi SS, Qayyum M (2015) Anthelmintic properties of extracts from Artemisia plants against Nematodes. Trop Biomed 32:257–268
Klayman DL (1985) Qinghaosu (artemisinin): an antimalarial drug from China. Science 228:1049–1055
Li Y, Shi YP, Hu YH (1994) Chemical-constituents of Artemisia roxburghiana Bess. Indian J Chem B 33:302–304
Mannan A, Ahmed I, Arshad W, Asim MF, Qureshi RA, Hussain I, Mirza B (2010) Survey of artemisinin production by diverse Artemisia species in northern Pakistan. Malar J 9:310
Markiewski MM, Lambris JD (2007) The role of complement in inflammatory diseases from behind the scenes into the spotlight. Am J Pathol 171:715–727
Mathela CS, Kharkwal H, Shah GC (1994) Essential oil composition of some Himalayan Artemisia species. J Essent Oil Res 6:345–348
Mir RA, Wani AH, Pala SA (2012) Evaluation of in vitro inhibitory effect of Artemisia species on spore germination of powdery mildew fungus. Biopestic Int 8:168–171
Nehra S (2014) Herbal folk medicine. Pointer publishers, Jaipur
Pandey V, Verma RS, Chauhan A, Tiwari R (2015) Compositional characteristics of the volatile oils of three Artemisia spp. from western Himalaya. J Essent Oil Res 27:107–114
Phan MG, Tran TTN, Phan TS, Otsuka H, Matsunami K (2012) Two new sesquiterpene lactones and other chemical constituents of Artemisia roxburghiana. Biochem Syst Ecol 45:115–119
Rahman U, Ali S, Ubaidullah Khan I, Khan MA, Arif S, Wazir SR (2016a) Anti-inflammatory activity of taraxerol acetate. J Med Sci 24:216–219
Rahman U, Durrani S, Ubaidullah Ali S, Rahman S (2016b) Anti-pyretic activity of taraxerol acetate. Khyber J Med Sci 9:165–167
Rehman U, Shah J, Khan MA, Shah MR, Ishtiaq Khan I (2013) Molecular docking of taraxerol acetate as a new COX inhibitor. Bangladesh J Pharmacol 8:194–197
Semwal RB, Semwal DK, Mishra SP, Semwal R (2015) Chemical Composition and Antibacterial Potential of Essential Oils from Artemisia capillaris, Artemisia nilagirica, Citrus limon, Cymbopogon flexuosus, Hedychium spicatum and Ocimum tenuiflorum. Nat Prod J 5:199–205
Shah MR, Shamim A, White LS, Bertino MF, Mesaik MA, Soomro S (2014) The anti-inflammatory properties of Au-scopoletin nanoconjugates. New J Chem 38:5566–5572
Shah MR, Ishtiaq Hizbullah SM, Habtemariam S, Zarrelli A, Muhammad A, Collina S, Khan I (2016) Protein tyrosine phosphatase 1B inhibitors isolated from Artemisia roxburghiana. J Enzyme Inhib Med Chem 31:563–567
Shahwar D, Raza MA, Ahmad VU (2013) Trypsin inhibitory activity of artemisinin and its biotransformed product. J Chem Soc Pak 35:135–138
Singh DK, Hajra PK (1996) Floristic diversity. In: Gujral GS, Sharma V (eds) Changing perspective of biodiversity status in the Himalaya. British Council Division, British High Commission Publication, Wildlife Youth Services, New Delhi, pp 23–38
Stuart K, Brun R, Croft S, Fairlamb A, Gurtler RE, McKerrow J, Reed S, Tarleton R (2008) Kinetoplastids: related protozoan pathogens, different diseases. J Clin Invest 118:1301–1310
Tan B, Shi HL, Ji G, Xie JQ (2011) Effects of taraxerol and taraxerol acetate on cell cycle and apoptosis of human gastric epithelial cell line AGS. Zhong Xi Yi Jie He Xue Bao 9:638–642
Tang JJ, Li JG, Qi W, Qiu WW, Li PS, Li BL, Song BL (2011) Inhibition of SREBP by a small molecule, betulin, improves hyperlipidemia and insulin resistance and reduces atherosclerotic plaques. Cell Metab 13:44–56
The Plant List (2013) Version 1.1. Published on the Internet, http://www.theplantlist.org/. Accessed 1 Jan 2013
Tu Y (2011) The discovery of artemisinin (qinghaosu) and gifts from Chinese medicine. Nat Med 17:1217–1220
Wani AL, Ara A, Usmani JA (2015) Lead toxicity: a review. Interdiscip Toxicol 8:55–64
WHO Fact sheet (2017) Leishmaniasis. World Health Organization. Updated April 2017. http://www.who.int/mediacentre/factsheets/fs375/en. Accessed 23 Mar 2018
WHO World malaria report (2017) 29 November. Geneva: World Health Organization. http://www.who.int/malaria/publications/world-malaria-report-2017/en. Accessed 27 Mar 2018
Winzeler EA, Manary MJ (2014) Drug resistance genomics of the antimalarial drug artemisinin. Genome Biol 15:544
Wright CW (2003) Artemisia: medicinal and aromatic plants—industrial profiles. CRC Press, New York
Zhang L (2011) Flora of China, vol. 20–21, pp. 697, 701. http://www.efloras.org/florataxon.aspx?flora_id=2&taxon_id=200023316. Accessed 13 Apr 2018
Acknowledgements
This work was financially supported by National Medicinal Plants Board, Ministry of AYUSH, Govt. of India (Grant No. Z.18017/187/CSS/R&D/UK-01/2017-18-NMPB-IV A).
Author information
Authors and Affiliations
Contributions
Ankit Kumar, Sonali Aswal, and Ashutosh Chauhan contributed in the searching literature and writing this manuscript. Ruchi Badoni Semwal provided an effective analysis of the document. Deepak Kumar Semwal revised and proofread the paper.
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, A., Aswal, S., Semwal, R.B. et al. Insights on the pharmacological, phytochemical and ethnobotanical aspects of Artemisia roxburghiana: a rather less explored but therapeutically important species of lower Himalayas. Phytochem Rev 18, 199–214 (2019). https://doi.org/10.1007/s11101-018-9589-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11101-018-9589-6