Abstract
Joint laxity should be distinguished from instability and its objective assessment is a determinant of safety and effectiveness clinical management of anterior cruciate ligament (ACL) tear and other often injured structures through injury mechanisms as knee sprains. Clinical evaluation is mandatory for all clinicians but requires the addition of medical devices for diagnosis or follow-up workflow objective assessment. Magnetic resonance imaging (MRI) instrumented-assessment of knee joint laxity combines the visual inspection of structural injury and multiplanar joint laxity quantification. The Porto knee Testing Device (PKTD) is an MRI-safe knee arthrometer that provides for accurate measurement of multiplanar joint laxity. Restoration of passive knee sagittal and transversal knee stability is the main objective of surgical interventions addressing ligaments reconstruction as ACL and pre- and post-operative measurements should be systematically performed and support precision orthopedic medicine.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Joint laxity
- Anterior cruciate ligament
- Knee instability
- Porto Knee Testing Device
- Objective measurement
- Magnetic resonance imaging
8.1 Laxity Versus Instability
Joint laxity is an objective and measurable parameter. Within human joints, we may have physiological laxity (normal laxity) or pathological laxity (abnormal laxity). Common language and definitions are crucial to enable a clear and constructive communication and scientific discussion. Back in 2006, during the works of the Anterior Cruciate Ligament (ACL) Study Group, John Feagin addressed the audience making a simplified but pretty clear distinction between instability and joint laxity, often used in an interchangeable manner. He stated that “instability is a symptom described by a patient, whereas laxity is an objective finding” [1]. Instability is present when the individual describes the joint as unstable when moving, walking, running, jumping, or twisting. Frequently, patients will refer that the joint “gives way”. Biomechanically joint laxity is the passive response of a joint to an externally applied force or torque [2]. The presence of abnormal laxity may or may not exist along with instability. The joint laxity profile varies among individuals. Differences in joint laxity have been reported related to sex [3,4,5], bone morphology and morphometrics [6,7,8,9], in presence of ligament or menisci injury [10,11,12,13,14], and outcomes of surgery [15,16,17,18], among others.
8.2 Measurement of Joint Laxity
When measuring any parameter, or you do it or you do not within an acceptable range. Resolution, precision, and accuracy of the measuring device are critical to enable the development of a screening system with clinical usefulness supported by its sensitivity and specificity. It should not be uncritically accepted the existence of important disparities in measurements outcomes (both under arthrokinematics and clinical views) that are obtained by different professionals, techniques, or devices [19] since they may mislead inappropriate interventions or absence of it. Both arthrokinematic and clinical outcomes may hinder the safety and efficacy that should support, by default, the clinical interventions. Precision and accuracy of joint laxity measurements should fit within physics conformity frames and not in general practices frames. Once it is considered a specific parameter, testing significance and setting, for the same person within a determined anatomofunctional status and period of human development, the outcomes expected must be reproducible and accurate. Methodological rigor is indispensable for research validity, usefulness of joint laxity measurements, and especially safety and effectiveness. We acknowledge though that majority of tests and testing principles yield value. Even facing different outcomes when assessing a same parameter (e.g., knee sagittal joint laxity as a quantity under observation) with different techniques or instruments, they may yield clinical useful information if the resolution and precision are suitable, and the accuracy is sufficiently close to its actual value. This may however be deceptive if the outcomes do not comprehend magnitudes indexed to normal and abnormal ranges. Take as an example that we acquired a thermometer which maximum scaling is 37 degrees Celsius (98.6 degrees Fahrenheit), it may yield an outstanding reliability, but there is no room for validity.
Knee joint laxity assessment has several angles that are worthy of research. We may allocate factors that interfere with knee joint multiplanar laxity envelope, to the individual intrinsic factors and the interplay of these with the choices and actions taken in case of need (e.g., treatment option in case of ACL ligament tear). Existing knee joint laxity may differ between uninjured and injured knees [20,21,22], either among different people in the same clinical condition [23], ontogenetic status of development [3, 24], biological circumstances [24, 25], sex [3,4,5], and different patterns of ACL tears [19].
There is great clinical and preventive potential to characterize and quantify the multiplanar knee joint laxity envelope. We are in need of studies that accurately assess joint laxity within different biological, pathological, or clinical conditions. This should be accomplished using laximetry—i.e., objective measurement of joint laxity—and, eventually, combining tests and/or equipment either for screening and to improve diagnosis [26]. The two main categories of laximetry are stress imaging and arthrometry. These two techniques classically aim to describe and quantify the displacement of the tibia in relation to the femur within the sagittal and transversal planes. These techniques often use cut-off values as dichotomic screening tools to elicit one of two diagnostic results: ruptured or not ruptured. The joint laxity data derive from an applied external force that aims to quantify bony displacement, either in unilateral or by side-to-side difference (SSD). Beyond the dichotomous application, laximetry can become an important diagnosis and profiling tool of different patterns of ACL tears (partial or total rupture) [19] and their interference in knee arthrokinematics, treatment decision, and surgical planning, prognostic purposes, or to quantify post-operative joint laxity.
8.3 Clinical Examination Combined with Laximetry and Imaging
While manual clinical examination is paramount for diagnosis, it is subjective both in the technique and interpretation [27]. The sensitivity and specificity of instrumented joint laxity measurements seem to increase with the combination of standard clinical examination in a two-step assessment process. Increased accuracy is found when combining clinical examination with Telos™ stress radiography [28]. The exposure to radiation and lack of ability to provide imaging evidence of soft tissue injury of stress radiography make the combined use of magnetic imaging resonance (MRI) and instrumented joint laxity testing the obvious next step to accurately measure knee joint laxity [22, 29].
The MRI alone does not provide for biomechanical competence data and joint laxity underestimation associated with some laximeters can mislead algorithm of treatment. Accurate laximetry combined with MRI will overcome barriers in anatomical and biomechanical competence assessment of apparent remaining intact ACL fibers or bundles or inserted grafts. When combining MRI with instrumented joint laxity, we sum up the visualization of anatomical structural evidence of injury and the ligament functional competence. The devices that are compatible with MRI use intrinsic anatomical landmarks [30] as references to measure the bony displacement and calculate the knee joint laxity.
Attempts also have been made, within a single-step assessment process, coupling measuring devices with manual elicited testing maneuvers, as the pivot shift, to confer objectivity and quantification [27]. Yet, the subjective variability in the testing technique persists due to disparities among professionals when eliciting the motion and determining which parameters use. These disparities are predominantly dependent on the assessor skills, training and experience, being present even in the often-used clinical tests as the pivot-shift [31].
The quest for accuracy and clinical usefulness should be the main goal of researchers and health care providers. The type of information is not so important—whereas from static or dynamic sagittal and/or rotatory testing, instrumented or not, separately or coupled with imaging assessment, under anesthesia or unanesthetized—but the validity, reliability, and accuracy should remain our focus. There are however premises learned from decades of research that pinpoint that settings and parameters are critical in joint laxity objective assessment and quantification.
8.4 Joint Laxity After Single ACL or Combined with Other Anterolateral Structures Injury
The diagnosis of different patterns of ACL tear is important for precision health care. New knowledge and evidence gathered in different domains as anatomy, biomechanics, pathomechanics, reinjury rates, and surgical techniques related to ACL injury and treatment should support customized risk management interventions and surgical planning.
Observed anterior translation and internal rotation of the tibia varies due to different ACL injury patterns. Partial or total tears, partial tears involving either the anteromedial or the posterolateral bundles and, it is also believed, that part of the abnormal rotatory joint laxity originates from additional injury to the anterolateral soft tissue structures. This is well documented in studies of biomechanical testing of cadaveric specimens. Lagae et al. [32] have recently reported different patterns of knee joint laxity after sectioning different anterolateral soft tissue structures which potentially mimic injuries subsequent to knee trauma, as the anterolateral ligament (ALL) and the deep fibers of the iliotibial band (ITB). Cutting the ACL did not significantly increase tibial internal rotation laxity significantly compared to the intact knee at any flexion angle. In the ACL-deficient knee, sectioning the ALL significantly increased the anterior laxity only at 20° to 30° of knee flexion, and only significantly increased internal rotation at 50° of knee flexion. A large increase in internal rotatory laxity is found however between 20° and 100° of flexion after sectioning the deep fibers of the ITB (including the Kaplan fibers), specifically the proximal and distal bundles [33] and the condylar strap [34]. This goes in line with the findings of Godin et al. [33] that support the role of the proximal and distal Kaplan fibrous bundles in rotational knee stability. The proximal and distal (Kaplan) bundles are 22.5 mm apart at the distal femur and revealed a mean maximum load during pull-to-failure testing of 71.3 N and 170.2 N, respectively. Later, Landreau et al. [34] identified a third and more distal bundle of deep ITB fibers attaching to the femur between the distal Kaplan fibers and the epicondyle, which they named as “condylar strap”. Even lacking biomechanical analysis, the qualitative evaluation of behavior in internal rotation revealed a tenodesis effect of the ITB which may add to anterolateral knee stability.
Several legit questions and concerns of translation to the clinical practice arise when interpreting these valuable anatomic and biomechanical studies. While biomechanical evidence suggests an important role of ITB in anterolateral instability control, injury frequency of deep ITB fibers in the setting of acute ACL tear [35, 36] is low compared to that of the ALL [37]. Yet, in presence of Segond fractures [38], where the ITB seems to be attached approximately in half of the cases and even in the absence of a Segond fracture [39], an ITB injury is a good marker for ACL injury. In fact, Lagae et al. [32] have shown that an isolated ACL anatomic reconstruction restored anterior tibial translation, but the remaining and significant internal rotatory laxity was only normalized after adding an extra-articular lateral tenodesis. Inderhaug et al. [40] also showed us that isolated ACL reconstruction does not restore normal kinematics, ACL combined with ALL reconstruction resulted in abnormal rotational joint laxity and that adding a lateral extra-articular tenodesis (MacIntosh or Lemaire) restored the knee internal rotation laxity to its native values. Other studies have also highlighted the importance of deep fibers of the ITB in controlling rotational joint laxity, but with a minimal influence of the ALL [41,42,43]. This makes us think of a potential overlooking behavior in MRI patterns in the setting of ACL injury and of the utility to combine the assessment of the ligament structural integrity and its functional competence within the same examination.
Correlational studies involving MRI and surgical exploration of the anterolateral complex (ALC) have shown high incidence in the setting of acute ACL-injured knees. However, MRI alone has low sensitivity, specificity, and accuracy for the diagnosis of ITB injury. The ITB was considered abnormal in approximately 31% of the cases [36]. Giving the number of cases, low diagnostic values of MRI alone, and relevance of ITB injury on rotatory joint laxity [32], the PKTD can play a role in functional diagnosis workflow of these additional injuries through joint laxity profiling. Rotatory joint laxity assessment within MRI may also be of particular importance in presence of Segond fractures since different structures of the ALC can be detached along with the bone avulsed fragment. The ITB often detaches along with the fragment with frequency depending on the dimensions and volume of the fragment as distance sparing it from the center of Gerdy’s tubercle [38, 44]. It is important to identify the patients with injury of the anterolateral structures, that if combined with increased rotatory joint laxity, are candidates to concomitant procedures such as lateral extra-articular tenodesis to better control the tibial internal rotation [45] and decrease the risk of graft failure [46].
8.5 Partial ACL Tears: MRI Diagnosis, Instrumented Joint Laxity Discrimination and Assessment of Biomechanical Competence
The MRI has high diagnostic accuracy for complete ACL tears [47]. Even the novel fully automated deep learning MRI techniques show high accuracy in identifying ACL tears [48]. However, when used to diagnose partial tears, the MRI is not capable to reliably detect partial tears [49] showing a high rate of false positives [47], even when using 3-Tesla MRI machines [50, 51]. Indeed, the MRI has low correlation with arthroscopic findings in cases of partial ACL tears [28, 52] and does not assess the functional competence of the intact ACL bundle. The instrumented joint laxity assessment is able to discriminate and document significant differences in mean SSD anterior tibial displacement in partial ACL tears [28]. Near one-third of patients treated arthroscopically for ACL injuries display a partial tear, being 14.1% classified as intact posterolateral bundle, 4.0% as intact anteromedial bundle, and 12.4% as posterior cruciate ligament healing. The SSD tibial displacement between ACL complete tear and all types of partial tears was significantly greater with Telos (mean 7.4 mm in total vs. 4.0 mm in partial ACL tears) than with the Rolimiter (mean 5.3 mm in total vs. 2.6 mm in partial ACL tears) [19]. The underestimation of joint laxity using the Rolimiter can hamper the desired accuracy for treatment decision and follow-up. The GeNouRoB, also comparing to Telos device, has showed a reasonably high diagnostic accuracy for ACL partial tears using a 2.5 mm cut-off (sensitivity of 84% and specificity of 81%) [53].
The ability to discriminate total from partial tears can be decisive for the surgical planning because the preservation of the ACL remnants enables anatomical landmarks for tunnel positioning [54] and provides vascular and mechanical benefits to the graft [55,56,57]. A selected group of patients with partial ACL tears may also respond well to conservative treatment [58,59,60,61] and in these cases it is crucial to assess the intact bundle competence. In cases of suspected partial tears, we use MRI instrumented-assessment to evaluate if there is any associated abnormal joint laxity [62].
8.6 Post-operative Knee Joint Laxity
Residual sagittal [63] and rotatory joint laxity [30, 64] as well as abnormal rotational motion [65,66,67,68] often persist after ACLR and are a common cause of poor long-term outcomes [69,70,71]. Residual knee joint laxity may disclose differences after ACL reconstruction procedures that might be related to the surgical technique [72,73,74], graft choice [75], concomitant procedures [16], graft tension or fixation angle [76, 77], and healing [78]. Residual anterior knee joint laxity 6 months following primary ACL reconstruction is associated with younger age (<30 years old), preoperative anterior laxity (SSD >5 mm), hamstring tendon graft, and resection of the medial meniscus [79]. Residual rotatory joint laxity measured by the pivot shift at 1 year after ACL reconstruction is associated with knee hyperextension and greater preoperative pivot shift under anesthesia. Age, gender, Lachman test, KT-1000 measurement, single-bundle vs. double-bundle, meniscus injury sites, and meniscus surgery were not predictors of residual rotational joint laxity [80].
Despite the evolution of surgical techniques, residual joint laxity should be a concern because it increases the ACL peak strain and has a fourfold increased risk for ACL injury for every 1.3 mm increase in SSD in anterior-posterior tibial displacement [81]. When athletes display residual joint laxity that is combined with neuromuscular deficits common in patients who tear the ACL [82]—such as weakness of hip external rotators—they will be exposed to a higher risk of reinjury during sport-specific tasks that involve pivoting or landing where the strain applied to graft is increased.
The use of accurate multiplanar laximetry techniques is important to monitor the post-operative outcomes. Restoration of knee stability is the main goal of surgical reconstruction and post-operative joint laxity evaluation should therefore always take part of a complete follow-up assessment. Despite the current literature on the importance of knee joint laxity on the treatment outcomes [83], but only 6% of studies use laxity-based assessment as a criterion for the return to sport decision [84]. In our experience, we use the MRI instrumented-assessment [85] that, in addition to the other often reported clinical and physical impairment-based objective criteria, supports our decision on when the athlete is ready to return to unrestricted sports.
8.7 MRI Instrumented-Assessment of Knee Joint Laxity
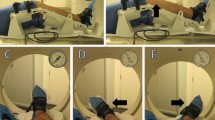
The Porto Knee Testing Device (PKTD) is an MRI-safe knee joint laxity testing device, made of polyurethane-based mixed resins, for the measurement of sagittal and rotatory knee joint laxity (Fig. 8.1). The PKTD operates through two movable platforms that are activated by plunger mechanisms. One platform induces an antero-posterior translation and the other internal or external rotation of the leg. These two movable platforms can operate isolated or in combination, allowing to measure isolated sagittal and rotatory joint laxity, or the two simultaneously combined. The operator can control the magnitude of load transmission and adjust for different degrees of knee flexion.
We combine the PKTD assessment with MRI visualization to objectively assess the knee joint laxity. After applying postero-anterior and/or rotatory stress, we measure the tibial displacement in the medial and lateral plateaus relative to the resting baseline position (Fig. 8.2). The tibial displacement is used as an isolated measure—i.e., the total amount of displacement—and is also compared with the contralateral knee.
PKTD exam of an ACL total rupture. MP medial plateau, LP lateral plateau, PA posteroanterior translation, ER external tibial rotation, IR internal tibial rotation. Blue line indicates tangent line to the posterior tibial plateau and orange line indicates tangent line to the posterior femoral condyle
The PKTD is a valid tool to assess ACL complete tears. The sagittal joint laxity is correlated with the KT-1000 and the rotational joint laxity is correlated with the pivot shift results [29]. While combining the anterior tibial displacement in both the medial and lateral plateaus, we obtain the most specific measure (94%); when combining the tibial internal and external rotation in the lateral plateau, we obtain the most sensitive measure (93%) [22].
The ability to visualize soft tissues concomitantly with accurate objective joint laxity measurement [30] allows to correlate the structural integrity of the ligament with its functional competence. Eventually, we can establish multiplanar knee joint laxity cluster profiles that may be associated with specific injury patterns [37, 86,87,88], time between injury and surgery [89], different ACL reconstruction surgical techniques outcomes [68, 90], or anatomic features such as bone morphology or morphometrics [91,92,93,94,95]. The PKTD can have an important role in establishing these multiplanar knee joint laxity cluster profiles as it combines the assessment of both “anatomy” and “function” [96]. For instance, the MRI visualization might identify a partial ACL tear with an intact bundle, that after the PKTD assessment can reveal incompetent to provide stability to the knee (Fig. 8.3) [62]. We may find also injury of the anterolateral structures of the knee that, if combined with abnormal rotational joint laxity, may require the addition of a lateral extra-articular tenodesis. When examining external tibial rotation laxity at 30 degrees of flexion, it may identify cases with posterolateral corner injury that may have been undetected during the dial test (Fig. 8.4) [97]. Using the PKTD, we can identify these subclinical groups that may require differentiated or additional surgical intervention and thus refine our surgical indications and individualize the treatment. In the follow-up of conservative or surgical approaches, the PKTD also plays an important role in the prospective monitoring of knee joint laxity and identify those with residual joint laxity (Fig. 8.5) [98]. It will provide useful information for the decision to clear the athletes to unrestricted sporting activities or those that may require further rehabilitation or surgical reintervention [85].
PKTD exam of two cases of ACL partial rupture. (a) Partial ACL rupture with an intact, but non-functional bundle; (b) partial ACL rupture with an intact and functional bundle. MP medial plateau, LP lateral plateau, PA posteroanterior translation, ER external tibial rotation, IR internal tibial rotation. Blue line indicates tangent line to the posterior tibial plateau and orange line indicates tangent line to the posterior femoral condyle
PKTD exam showing increased external rotation that was undetected under the dial test. LP lateral plateau, PA posteroanterior translation, ER external tibial rotation. Blue line indicates tangent line to the posterior tibial plateau and orange line indicates tangent line to the posterior femoral condyle
PKTD exam comparing an ACL-reconstructed knee (a) with its contralateral healthy knee (b). From the PKTD examination, we can observe that there is still residual laxity that requires reintervention. MP medial plateau, LP lateral plateau, PA posteroanterior translation, ER external tibial rotation, IR internal tibial rotation. Blue line indicates tangent line to the posterior tibial plateau and orange line indicates tangent line to the posterior femoral condyle
8.8 Conclusions
The PKTD is an MRI-safe knee joint laxity testing device which enables assessment of isolated or combined anteroposterior and rotatory joint laxity. Accurate assessment of multiplanar tibial displacement with imaging visualization can establish joint laxity cluster profiles that may correlate with specific injury patterns. Joint laxity can vary in quantity and in quality if there is an isolated ACL injury or there is additionally injury to peripheral structures, such as the ALC (especially the ALL and deep fibers of the ITB) or the menisci. Combining the MRI visual inspection of anatomical injury with the mechanical capability using the PKTD, we are able to accurately assess and characterize the knee joint multiplanar laxity and thus support treatment decisions and customized interventions while aiming for superior outcomes. The restoration of passive sagittal and transversal knee stability is the main purpose of surgical interventions addressing ACL reconstruction and pre- and post-operative measurements should therefore be systematically performed to support orthopedic precision medicine.
References
Feagin J. Personal communication. Hawaii: ACL Study Group; 2006.
Musahl V, Hoshino Y, Becker R, Karlsson J. Rotatory knee laxity and the pivot shift. Knee Surg Sports Traumatol Arthrosc. 2012;20:601–2.
Tagesson S, Witvrouw E, Kvist J. Differences in knee joint stabilization between children and adults and between the sexes. Am J Sports Med. 2013;41:678–83.
Beynnon BD, Bernstein IM, Belisle A, Brattbakk B, Devanny P, Risinger R, Durant D. The effect of estradiol and progesterone on knee and ankle joint laxity. Am J Sports Med. 2005;33:1298–304.
Pfeiffer TR, Kanakamedala AC, Herbst E, Nagai K, Murphy C, Burnham JM, Popchak A, Debski RE, Musahl V. Female sex is associated with greater rotatory knee laxity in collegiate athletes. Knee Surg Sports Traumatol Arthrosc. 2018;26:1319–25.
Andrade R, Vasta S, Sevivas N, Pereira R, Leal A, Papalia R, Pereira H, Espregueira-Mendes J. Notch morphology is a risk factor for ACL injury: a systematic review and meta-analysis. J ISAKOS. 2016;1:70–81.
Saita Y, Schoenhuber H, Thiébat G, et al. Knee hyperextension and a small lateral condyle are associated with greater quantified antero-lateral rotatory instability in the patients with a complete anterior cruciate ligament (ACL) rupture. Knee Surg Sports Traumatol Arthrosc. 2019;27:868–74.
Branch T, Stinton S, Sharma A, Lavoie F, Guier C, Neyret P. The impact of bone morphology on the outcome of the pivot shift test: a cohort study. BMC Musculoskelet Disord. 2017;18:463.
Rahnemai-Azar AA, Abebe ES, Johnson P, Labrum J, Fu FH, Irrgang JJ, Samuelsson K, Musahl V. Increased lateral tibial slope predicts high-grade rotatory knee laxity pre-operatively in ACL reconstruction. Knee Surg Sports Traumatol Arthrosc. 2017;25:1170–6.
Grassi A, Di Paolo S, Lucidi GA, Macchiarola L, Raggi F, Zaffagnini S. The contribution of partial Meniscectomy to preoperative laxity and laxity after anatomic single-bundle anterior cruciate ligament reconstruction: in vivo kinematics with navigation. Am J Sports Med. 2019;47:3203–11.
Dejour D, Pungitore M, Valluy J, Nover L, Saffarini M, Demey G. Preoperative laxity in ACL-deficient knees increases with posterior tibial slope and medial meniscal tears. Knee Surg Sports Traumatol Arthrosc. 2019;27:564–72.
Cristiani R, Rönnblad E, Engström B, Forssblad M, Stålman A. Medial meniscus resection increases and medial meniscus repair preserves anterior knee laxity: a cohort study of 4497 patients with primary anterior cruciate ligament reconstruction. Am J Sports Med. 2018;46:357–62.
Musahl V, Rahnemai-Azar AA, Costello J, Arner JW, Fu FH, Hoshino Y, Lopomo N, Samuelsson K, Irrgang JJ. The influence of meniscal and anterolateral capsular injury on knee laxity in patients with anterior cruciate ligament injuries. Am J Sports Med. 2016;44:3126–31.
van Lieshout WAM, Martijn CD, van Ginneken BTJ, van Heerwaarden RJ. Medial collateral ligament laxity in valgus knee deformity before and after medial closing wedge high tibial osteotomy measured with instrumented laxity measurements and patient reported outcome. J Exp Orthop. 2018;5:49.
Samuelsen BT, Webster KE, Johnson NR, Hewett TE, Krych AJ. Hamstring autograft versus patellar tendon autograft for ACL reconstruction: is there a difference in graft failure rate? A meta-analysis of 47,613 patients. Clin Orthop Relat Res. 2017;475:2459–68.
Grassi A, Signorelli C, Lucidi GA, Raggi F, Macchiarola L, Roberti Di Sarsina T, Marcheggiani Muccioli GM, Filardo G, Zaffagnini S. ACL reconstruction with lateral plasty reduces translational and rotatory laxity compared to anatomical single bundle and non-anatomical double bundle surgery: an in vivo kinematic evaluation with navigation system. Clin Biomech (Bristol, Avon). 2019;69:1–8. https://doi.org/10.1016/j.clinbiomech.2019.06.012.
Kim SJ, Choi CH, Kim SH, Lee SK, Lee W, Kim T, Jung M. Bone-patellar tendon-bone autograft could be recommended as a superior graft to hamstring autograft for ACL reconstruction in patients with generalized joint laxity: 2- and 5-year follow-up study. Knee Surg Sports Traumatol Arthrosc. 2018;26:2568–79.
Nyland J, Collis P, Huffstutler A, Sachdeva S, Spears JR, Greene J, Caborn DNM. Quadriceps tendon autograft ACL reconstruction has less pivot shift laxity and lower failure rates than hamstring tendon autografts. Knee Surg Sports Traumatol Arthrosc. 2020;28:509–18.
Panisset JC, Ntagiopoulos PG, Saggin PR, Dejour D. A comparison of Telos™ stress radiography versus Rolimeter™ in the diagnosis of different patterns of anterior cruciate ligament tears. Orthop Traumatol Surg Res. 2012;98:751–8.
Imbert P, Belvedere C, Leardini A. Human knee laxity in ACL-deficient and physiological contralateral joints: intra-operative measurements using a navigation system. Biomed Eng Online. 2014;13:86.
Mouton C, Theisen D, Meyer T, Agostinis H, Nührenbörger C, Pape D, Seil R. Noninjured knees of patients with noncontact acl injuries display higher average anterior and internal rotational knee laxity compared with healthy knees of a noninjured population. Am J Sports Med. 2015;43:1918–23.
Espregueira-Mendes J, Andrade R, Leal A, Pereira H, Skaf A, Rodrigues-Gomes S, Oliveira JM, Reis RL, Pereira R. Global rotation has high sensitivity in ACL lesions within stress MRI. Knee Surg Sports Traumatol Arthrosc. 2017;25:2993–3003.
Sernert N, Kartus JT, Ejerhed L, Karlsson J. Right and left knee laxity measurements: a prospective study of patients with anterior cruciate ligament injuries and normal control subjects. Arthroscopy. 2004;20:564–71.
Shultz SJ, Sander TC, Kirk SE, Perrin DH. Sex differences in knee joint laxity change across the female menstrual cycle. J Sports Med Phys Fitness. 2005;45:594–603.
Charlton WP, Coslett-Charlton LM, Ciccotti MG. Correlation of estradiol in pregnancy and anterior cruciate ligament laxity. Clin Orthop Relat Res. 2001;(387):165–70.
Hegedus EJ, Cook C, Lewis J, Wright A, Park JY. Combining orthopedic special tests to improve diagnosis of shoulder pathology. Phys Ther Sport. 2015;16:87–92.
Ahldén M, Hoshino Y, Samuelsson K, Araujo P, Musahl V, Karlsson J. Dynamic knee laxity measurement devices. Knee Surg Sports Traumatol Arthrosc. 2012;20:621–32.
Dejour D, Ntagiopoulos PG, Saggin PR, Panisset JC. The diagnostic value of clinical tests, magnetic resonance imaging, and instrumented laxity in the differentiation of complete versus partial anterior cruciate ligament tears. Arthroscopy. 2013;29:491–9.
Espregueira-Mendes J, Pereira H, Sevivas N, Passos C, Vasconcelos JC, Monteiro A, Oliveira JM, Reis RL. Assessment of rotatory laxity in anterior cruciate ligament-deficient knees using magnetic resonance imaging with Porto-knee testing device. Knee Surg Sports Traumatol Arthrosc. 2012;20:671–8.
Tashiro Y, Okazaki K, Miura H, Matsuda S, Yasunaga T, Hashizume M, Nakanishi Y, Iwamoto Y. Quantitative assessment of rotatory instability after anterior cruciate ligament reconstruction. Am J Sports Med. 2009;37:909–16.
Musahl V, Hoshino Y, Ahlden M, Araujo P, Irrgang JJ, Zaffagnini S, Karlsson J, Fu FH. The pivot shift: a global user guide. Knee Surg Sports Traumatol Arthrosc. 2012;20:724–31.
Lagae KC, Robberecht J, Athwal KK, Verdonk PCM, Amis AA. ACL reconstruction combined with lateral monoloop tenodesis can restore intact knee laxity. Knee Surg Sports Traumatol Arthrosc. 2020;28:1159–68.
Godin JA, Chahla J, Moatshe G, Kruckeberg BM, Muckenhirn KJ, Vap AR, Geeslin AG, LaPrade RF. A comprehensive reanalysis of the distal iliotibial band: quantitative anatomy, radiographic markers, and biomechanical properties. Am J Sports Med. 2017;45:2595–603.
Landreau P, Catteeuw A, Hamie F, Saithna A, Sonnery-Cottet B, Smigielski R. Anatomic study and reanalysis of the nomenclature of the anterolateral complex of the knee focusing on the distal iliotibial band: identification and description of the condylar strap. Orthop J Sports Med. 2019;7:2325967118818064.
Daggett M, Claes S, Helito CP, Imbert P, Monaco E, Lutz C, Sonnery-Cottet B. The role of the anterolateral structures and the acl in controlling laxity of the intact and acl-deficient knee: letter to the editor. Am J Sports Med. 2016;44:14–5.
Monaco E, Helito CP, Redler A, Argento G, De Carli A, Saithna A, Helito PVP, Ferretti A. Correlation between magnetic resonance imaging and surgical exploration of the anterolateral structures of the acute anterior cruciate ligament-injured knee. Am J Sports Med. 2019;47:1186–93.
Ferretti A, Monaco E, Redler A, Argento G, De Carli A, Saithna A, Helito PVP, Helito CP. High prevalence of anterolateral ligament abnormalities on MRI in knees with acute anterior cruciate ligament injuries: a case-control series from the SANTI study group. Orthop J Sports Med. 2019;7:2325967119852916.
Helito PVP, Bartholomeeusen S, Claes S, Rodrigues MB, Helito CP. Magnetic resonance imaging evaluation of the anterolateral ligament and the iliotibial band in acute anterior cruciate ligament injuries associated with segond fractures. Arthroscopy. 2020; https://doi.org/10.1016/j.arthro.2020.02.005.
Khanna M, Gupte C, Dodds A, Williams A, Walker M. Magnetic resonance imaging appearances of the capsulo-osseous layer of the iliotibial band and femoral attachments of the iliotibial band in the normal and pivot-shift ACL injured knee. Skelet Radiol. 2019;48:729–40.
Inderhaug E, Stephen JM, Williams A, Amis AA. Biomechanical comparison of anterolateral procedures combined with anterior cruciate ligament reconstruction. Am J Sports Med. 2017;45:347–54.
Stentz-Olesen K, Nielsen ET, de Raedt S, Jørgensen PB, Sørensen OG, Kaptein B, Søballe K, Stilling M. Reconstructing the anterolateral ligament does not decrease rotational knee laxity in ACL-reconstructed knees. Knee Surg Sports Traumatol Arthrosc. 2017;25:1125–31.
Jenny JY, Puliero B, Schockmel G, Harnoist S, Clavert P. Minimal influence of the anterolateral knee ligament on anterior and rotational laxity of the knee: a cadaveric study. Eur J Orthop Surg Traumatol. 2018;28:955–8.
Smith PA, Thomas DM, Pomajzl RJ, Bley JA, Pfeiffer FM, Cook JL. A biomechanical study of the role of the anterolateral ligament and the deep iliotibial band for control of a simulated pivot shift with comparison of minimally invasive extra-articular anterolateral tendon graft reconstruction versus modified lemaire reconstruction after anterior cruciate ligament reconstruction. Arthroscopy. 2019;35:1473–83.
Shaikh H, Herbst E, Rahnemai-Azar AA, Bottene Villa Albers M, Naendrup JH, Musahl V, Irrgang JJ, Fu FH. The segond fracture is an avulsion of the anterolateral complex. Am J Sports Med. 2017;45:2247–52.
Slette EL, Mikula JD, Schon JM, Marchetti DC, Kheir MM, Turnbull TL, LaPrade RF. Biomechanical results of lateral extra-articular tenodesis procedures of the knee: a systematic review. Arthroscopy. 2016;32:2592–611.
Getgood AMJ, Bryant DM, Litchfield R, et al. Lateral extra-articular tenodesis reduces failure of hamstring tendon autograft anterior cruciate ligament reconstruction: 2-year outcomes from the STABILITY study randomized clinical trial. Am J Sports Med. 2020;48:285–97.
Ghasem Hanafi M, Momen Gharibvand M, Jaffari Gharibvand R, Sadoni H. Diagnostic value of oblique coronal and oblique sagittal magnetic resonance imaging (MRI) in diagnosis of anterior cruciate ligament (ACL) tears. J Med Life. 2018;11:281–5.
Liu F, Guan B, Zhou Z, et al. Fully automated diagnosis of anterior cruciate ligament tears on knee MR images by using deep learning. Radiol Artif Intell. 2019;1:180091.
Lawrance JA, Ostlere SJ, Dodd CA. MRI diagnosis of partial tears of the anterior cruciate ligament. Injury. 1996;27:153–5.
Van Dyck P, De Smet E, Veryser J, Lambrecht V, Gielen JL, Vanhoenacker FM, Dossche L, Parizel PM. Partial tear of the anterior cruciate ligament of the knee: injury patterns on MR imaging. Knee Surg Sports Traumatol Arthrosc. 2012;20:256–61.
Van Dyck P, Vanhoenacker FM, Gielen JL, Dossche L, Van Gestel J, Wouters K, Parizel PM. Three tesla magnetic resonance imaging of the anterior cruciate ligament of the knee: can we differentiate complete from partial tears? Skelet Radiol. 2011;40:701–7.
Jog AV, Smith TJ, Pipitone PS, Toorkey BC, Morgan CD, Bartolozzi AR. Is a partial anterior cruciate ligament tear truly partial? A clinical, arthroscopic, and histologic investigation. Arthroscopy. 2020; https://doi.org/10.1016/j.arthro.2020.02.037.
Lefevre N, Bohu Y, Naouri JF, Klouche S, Herman S. Validity of GNRB® arthrometer compared to Telos™ in the assessment of partial anterior cruciate ligament tears. Knee Surg Sports Traumatol Arthrosc. 2014;22:285–90.
Shimodaira H, Tensho K, Akaoka Y, Takanashi S, Kato H, Saito N. Remnant-preserving tibial tunnel positioning using anatomic landmarks in double-bundle anterior cruciate ligament reconstruction. Arthroscopy. 2016;32:1822–30.
Georgoulis AD, Pappa L, Moebius U, Malamou-Mitsi V, Pappa S, Papageorgiou CO, Agnantis NJ, Soucacos PN. The presence of proprioceptive mechanoreceptors in the remnants of the ruptured ACL as a possible source of re-innervation of the ACL autograft. Knee Surg Sports Traumatol Arthrosc. 2001;9:364–8.
Lee BI, Min KD, Choi HS, Kim JB, Kim ST. Arthroscopic anterior cruciate ligament reconstruction with the tibial-remnant preserving technique using a hamstring graft. Arthroscopy. 2006;22:340.e1–7.
Gohil S, Annear PO, Breidahl W. Anterior cruciate ligament reconstruction using autologous double hamstrings: a comparison of standard versus minimal debridement techniques using MRI to assess revascularisation. A randomised prospective study with a one-year follow-up. J Bone Joint Surg Br. 2007;89:1165–71.
Ekås GR, Moksnes H, Grindem H, Risberg MA, Engebretsen L. Coping with anterior cruciate ligament injury from childhood to maturation: a prospective case series of 44 patients with mean 8 years’ follow-up. Am J Sports Med. 2019;47:22–30.
Thoma LM, Grindem H, Logerstedt D, Axe M, Engebretsen L, Risberg MA, Snyder-Mackler L. Coper classification early after anterior cruciate ligament rupture changes with progressive neuromuscular and strength training and is associated with 2-year success: the delaware-Oslo ACL cohort study. Am J Sports Med. 2019;47:807–14.
Wellsandt E, Failla MJ, Axe MJ, Snyder-Mackler L. Does anterior cruciate ligament reconstruction improve functional and radiographic outcomes over nonoperative management 5 years after injury? Am J Sports Med. 2018;46:2103–12.
Colombet P, Dejour D, Panisset JC, Siebold R. Current concept of partial anterior cruciate ligament ruptures. Orthop Traumatol Surg Res. 2010;96:S109–18.
Ohashi B, Ward J, Araujo P, Kfuri M, Pereira H, Espregueira-Mendes J, Musahl V. Partial anterior cruciate ligament ruptures: knee laxity measurements and pivot shift. In: Doral MM, Karlsson J, editors. Sports injuries: prevention. Treatment and Rehabilitation: Diagnosis; 2015. p. 1245–58.
Nakanishi Y, Matsushita T, Nagai K, Araki D, Kanzaki N, Hoshino Y, Matsumoto T, Niikura T, Kuroda R. Greater knee joint laxity remains in teenagers after anatomical double-bundle anterior cruciate ligament reconstruction compared to young adults. Knee Surg Sports Traumatol Arthrosc. 2020; https://doi.org/10.1007/s00167-020-05910-z.
Izawa T, Okazaki K, Tashiro Y, Matsubara H, Miura H, Matsuda S, Hashizume M, Iwamoto Y. Comparison of rotatory stability after anterior cruciate ligament reconstruction between single-bundle and double-bundle techniques. Am J Sports Med. 2011;39:1470–7.
Tagesson S, Öberg B, Kvist J. Static and dynamic tibial translation before, 5 weeks after, and 5 years after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2015;23:3691–7.
Tashman S, Collon D, Anderson K, Kolowich P, Anderst W. Abnormal rotational knee motion during running after anterior cruciate ligament reconstruction. Am J Sports Med. 2004;32:975–83.
Tashman S, Kolowich P, Collon D, Anderson K, Anderst W. Dynamic function of the ACL-reconstructed knee during running. Clin Orthop Relat Res. 2007;454:66–73.
Georgoulis AD, Ristanis S, Chouliaras V, Moraiti C, Stergiou N. Tibial rotation is not restored after ACL reconstruction with a hamstring graft. Clin Orthop Relat Res. 2007;454:89–94.
Kamath GV, Redfern JC, Greis PE, Burks RT. Revision anterior cruciate ligament reconstruction. Am J Sports Med. 2011;39:199–217.
Corsetti JR, Jackson DW. Failure of anterior cruciate ligament reconstruction: the biologic basis. Clin Orthop Relat Res. 1996:42–9.
Vergis A, Gillquist J. Graft failure in intra-articular anterior cruciate ligament reconstructions: a review of the literature. Arthroscopy. 1995;11:312–21.
Lee S, Kim H, Jang J, Seong SC, Lee MC. Comparison of anterior and rotatory laxity using navigation between single- and double-bundle ACL reconstruction: prospective randomized trial. Knee Surg Sports Traumatol Arthrosc. 2012;20:752–61.
Li YL, Ning GZ, Wu Q, Wu QL, Li Y, Hao Y, Feng SQ. Single-bundle or double-bundle for anterior cruciate ligament reconstruction: a meta-analysis. Knee. 2014;21:28–37.
Kilinc BE, Kara A, Oc Y, Celik H, Camur S, Bilgin E, Erten YT, Sahinkaya T, Eren OT. Transtibial vs anatomical single bundle technique for anterior cruciate ligament reconstruction: a Retrospective Cohort Study. Int J Surg. 2016;29:62–9.
Cristiani R, Sarakatsianos V, Engström B, Samuelsson K, Forssblad M, Stålman A. Increased knee laxity with hamstring tendon autograft compared to patellar tendon autograft: a cohort study of 5462 patients with primary anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2019;27:381–8.
Koga H, Muneta T, Yagishita K, Ju YJ, Sekiya I. The effect of graft fixation angles on anteroposterior and rotational knee laxity in double-bundle anterior cruciate ligament reconstruction: evaluation using computerized navigation. Am J Sports Med. 2012;40:615–23.
Dhaher YY, Salehghaffari S, Adouni M. Anterior laxity, graft-tunnel interaction and surgical design variations during anterior cruciate ligament reconstruction: a probabilistic simulation of the surgery. J Biomech. 2016;49:3009–16.
Hofbauer M, Soldati F, Szomolanyi P, Trattnig S, Bartolucci F, Fu F, Denti M. Hamstring tendon autografts do not show complete graft maturity 6 months postoperatively after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2019;27:130–6.
Cristiani R, Forssblad M, Engström B, Edman G, Stålman A. Risk factors for abnormal anteroposterior knee laxity after primary anterior cruciate ligament reconstruction. Arthroscopy. 2018;34:2478–84.
Ueki H, Nakagawa Y, Ohara T, et al. Risk factors for residual pivot shift after anterior cruciate ligament reconstruction: data from the MAKS group. Knee Surg Sports Traumatol Arthrosc. 2018;26:3724–30.
Myer GD, Ford KR, Paterno MV, Nick TG, Hewett TE. The effects of generalized joint laxity on risk of anterior cruciate ligament injury in young female athletes. Am J Sports Med. 2008;36:1073–80.
Paterno MV, Schmitt LC, Ford KR, Rauh MJ, Myer GD, Huang B, Hewett TE. Biomechanical measures during landing and postural stability predict second anterior cruciate ligament injury after anterior cruciate ligament reconstruction and return to sport. Am J Sports Med. 2010;38:1968–78.
Rohman EM, Macalena JA. Anterior cruciate ligament assessment using arthrometry and stress imaging. Curr Rev Musculoskelet Med. 2016;9:130–8.
Burgi CR, Peters S, Ardern CL, Magill JR, Gomez CD, Sylvain J, Reiman MP. Which criteria are used to clear patients to return to sport after primary ACL reconstruction? A scoping review. Br J Sports Med. 2019;53:1154–61.
Andrade R, Pereira R, Bastos R, Duarte H, Pereira H, Rodrigues-Gomes S, Espregueira-Mendes J. MRI-based laxity measurement for return to play. In: Musahl V, Karlsson J, Krutsch W, Mandelbaum B, Espregueira-Mendes J, d’Hooghe P, editors. Return to play in football. Berlin, Heidelberg: Springer; 2018. p. 205–15.
Zaffagnini S, Signorelli C, Bonanzinga T, Grassi A, Galán H, Akkawi I, Bragonzoni L, Cataldi F, Marcacci M. Does meniscus removal affect ACL-deficient knee laxity? An in vivo study. Knee Surg Sports Traumatol Arthrosc. 2016;24:3599–604.
Andrade R, Rebelo-Marques A, Bastos R, Zaffagnini S, Seil R, Ayeni OR, Espregueira-Mendes J. Identification of normal and injured anterolateral ligaments of the knee: a systematic review of magnetic resonance imaging studies. Arthroscopy. 2019;35:1594–1613.e1.
Bastos R, Andrade R, Vasta S, Pereira R, Papalia R, van der Merwe W, Rodeo S, Espregueira-Mendes J. Tibiofemoral bone bruise volume is not associated with meniscal injury and knee laxity in patients with anterior cruciate ligament rupture. Knee Surg Sports Traumatol Arthrosc. 2019;27:3318–26.
Yanagisawa S, Kimura M, Hagiwara K, Ogoshi A, Nakagawa T, Shiozawa H, Ohsawa T. Factors affecting knee laxity following anterior cruciate ligament reconstruction using a hamstring tendon. Knee. 2017;24:1075–82.
Rupp S, Müller B, Seil R. Knee laxity after ACL reconstruction with a BPTB graft. Knee Surg Sports Traumatol Arthrosc. 2001;9:72–6.
Vasta S, Andrade R, Pereira R, Bastos R, Battaglia AG, Papalia R, Espregueira-Mendes J. Bone morphology and morphometry of the lateral femoral condyle is a risk factor for ACL injury. Knee Surg Sports Traumatol Arthrosc. 2018;26:2817–25.
Fernandes MS, Pereira R, Andrade R, Vasta S, Pereira H, Pinheiro JP, Espregueira-Mendes J. Is the femoral lateral condyle’s bone morphology the trochlea of the ACL? Knee Surg Sports Traumatol Arthrosc. 2017;25:207–14.
Napier RJ, Garcia E, Devitt BM, Feller JA, Webster KE. Increased radiographic posterior tibial slope is associated with subsequent injury following revision anterior cruciate ligament reconstruction. Orthop J Sports Med. 2019;7:2325967119879373.
Cooper JD, Wang W, Prentice HA, Funahashi TT, Maletis GB. The association between tibial slope and revision anterior cruciate ligament reconstruction in patients ≤21 years old: a matched case-control study including 317 revisions. Am J Sports Med. 2019;47:3330–8.
Dejour D, Saffarini M, Demey G, Baverel L. Tibial slope correction combined with second revision ACL produces good knee stability and prevents graft rupture. Knee Surg Sports Traumatol Arthrosc. 2015;23:2846–52.
Pereira H, Gomes S, Vasconcelos JC, Soares L, Pereira R, Oliveira JM, Reis RL, Espregueira-Mendes J. MRI laxity assessment. In: Musahl V, Karlsson J, Kuroda R, Zaffagnini S, editors. Rotatory knee instability. Cham: Springer; 2017. p. 49–61.
Andrade R, Duarte H, Pereira R, Arias C, Bastos R, Rodrigues-Gomes S, Oliveira JM, Reis RL, Espregueira-Mendes J. An advanced device for multiplanar instability assessment in MRI. In: Margheritini F, Espregueira-Mendes J, Gobbi A, editors. Complex knee ligament injuries. Springer; 2019. p. 27–33.
Andrade R, Pereira H, Espregueira-Mendes J. ACL treatment in 2016–controversy and consensus. Asian J Arthroscopy. 2016;1:3–10.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 ISAKOS
About this chapter
Cite this chapter
Pereira, R., Andrade, R., Florim, S., Duarte, J.A., Espregueira-Mendes, J. (2022). Laxity Objective Measurement Within MRI of ACL Lesions. In: Nakamura, N., Marx, R.G., Musahl, V., Getgood, A., Sherman, S.L., Verdonk, P. (eds) Advances in Knee Ligament and Knee Preservation Surgery. Springer, Cham. https://doi.org/10.1007/978-3-030-84748-7_8
Download citation
DOI: https://doi.org/10.1007/978-3-030-84748-7_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-84747-0
Online ISBN: 978-3-030-84748-7
eBook Packages: MedicineMedicine (R0)