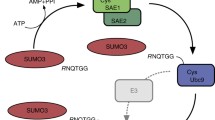
Abstract
Protein regulation by reversible attachment of SUMO (small ubiquitin-related modifier) plays an important role in several cellular processes such as transcriptional regulation, nucleo-cytoplasmic transport, cell-cycle progression, meiosis, and DNA repair. However, most sumoylated proteins are of marginal abundance at steady state levels, which is due to strict regulation and/or rapid turnover of modification and de-modification. Consequently, analysis of protein sumoylation in vivo is very challenging. Nonetheless, a novel method was established that allows detection of sumoylated proteins at endogenous levels from vertebrate cells and tissues. This approach involves the enrichment of sumoylated proteins by immunoprecipitation followed by peptide elution. After endogenous substrate sumoylation is verified, addressing its functional consequences is the next logical step. This requires SUMO site mapping that benefits from larger quantities of modified protein. Here, we shortly describe strategies to achieve efficient in vitro sumoylation of many substrates.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Droescher M, Chaugule VK, Pichler A (2013) SUMO rules: regulatory concepts and their implication in neurologic functions. Neuromolecular Med 15:639–660
Flotho A, Melchior F (2013) Sumoylation: a regulatory protein modification in health and disease. Annu Rev. Biochem 82:357–385
Hickey CM, Wilson NR, Hochstrasser M (2012) Function and regulation of SUMO proteases. Nat Rev. Mol Cell Biol 13:755–766
Schulz S, Chachami G, Kozaczkiewicz L et al (2012) Ubiquitin-specific protease-like 1 (USPL1) is a SUMO isopeptidase with essential, non-catalytic functions. EMBO Rep 13:930–938
Shin EJ, Shin HM, Nam E et al (2012) DeSUMOylating isopeptidase: a second class of SUMO protease. EMBO Rep 13:339–346
Pichler A (2008) Analysis of sumoylation. Methods Mol Biol 446:131–138
Pichler A, Knipscheer P, Oberhofer E et al (2005) SUMO modification of the ubiquitin-conjugating enzyme E2-25K. Nat Struct Mol Biol 12:264–269
Rodriguez MS, Desterro JM, Lain S et al (1999) SUMO-1 modification activates the transcriptional response of p53. EMBO J 18:6455–6461
Tirard M, Hsiao HH, Nikolov M et al (2012) In vivo localization and identification of SUMOylated proteins in the brain of His6-HA-SUMO1 knock-in mice. Proc Natl Acad Sci U S A 109:21122–21127
Becker J, Barysch SV, Karaca S et al (2013) Detecting endogenous SUMO targets in mammalian cells and tissues. Nat Struct Mol Biol 20:525–531
Matunis MJ, Coutavas E, Blobel G (1996) A novel ubiquitin-like modification modulates the partitioning of the Ran-GTPase-activating protein RanGAP1 between the cytosol and the nuclear pore complex. J Cell Biol 135:1457–1470
Zhang XD, Goeres J, Zhang H et al (2008) SUMO-2/3 modification and binding regulate the association of CENP-E with kinetochores and progression through mitosis. Mol Cell 29:729–741
Pichler A, Knipscheer P, Saitoh H et al (2004) The RanBP2 SUMO E3 ligase is neither HECT- nor RING-type. Nat Struct Mol Biol 11:984–991
Pichler A, Gast A, Seeler JS et al (2002) The nucleoporin RanBP2 has SUMO1 E3 ligase activity. Cell 108:109–120
Flotho A, Werner A, Winter T et al (2012) Recombinant reconstitution of sumoylation reactions in vitro. Methods Mol Biol 832:93–110
Acknowledgments
Our special thanks go to Pichler group members for discussions and sharing reagents, Dr. Mathias Droescher for critical comments on the manuscript, Dr. Frauke Melchior for sharing the protocol, Dr. Michael Matunis for making the SUMO antibodies available to the community and the Developmental Studies Hybridoma Bank at the University of Iowa for distribution of the SUMO antibody-producing hybridoma cells, and Dr. Ron Hay for providing PML antibodies.
The work in the Pichler lab is funded by the Max Planck Society, the IMPRS PhD program, the Deutsche Forschungsgemeinschaft (DFG, PI 917/1-1, DFG-SPP1365 PI 917/2-1), and the Fritz Thyssen Stiftung (10.11.1.210).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Breucker, J., Pichler, A. (2019). Analysis of Sumoylation. In: Kannicht, C. (eds) Post-Translational Modification of Proteins. Methods in Molecular Biology, vol 1934. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9055-9_14
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9055-9_14
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-9053-5
Online ISBN: 978-1-4939-9055-9
eBook Packages: Springer Protocols