Abstract
Purpose
The use of plant extracts obtained from plants that are highly tolerant to heavy metal toxicity has been beneficial in improving the growth of plants grown under metal toxicity conditions. A lab experiment was performed to elucidate the alleviating role of foliar applied cadmium (Cd)-hyperaccumulator Solanum nigrum (S. nigrum) extract on Datura innoxia (D. innoxia) plants grown under Cd stress (0, 50, and 100 mg Cd kg-1 soil).
Methods
Growth parameters, photosynthetic pigment content, osmo-metabolic compounds, reduced glutathione and phytochelatins content, oxidative damage, and lignin content and its related enzyme (cell wall-bound peroxidase, POX) were determined.
Results
Apart from the foliar application response of S. nigrum leaf extract (SNE) in either Cd exposed or non-Cd exposed plants, growth parameters of D. innoxia plant grown under both Cd concentrations (50 and 100 mg Cd kg-1 soil) in terms of root and shoot fresh, dry weight, length, and leaf area were noticeably diminished by 29 and 51%, 29 and 54%, 35 and 70%, 40 and 53%, 30 and 69%, 40 and 60%, and 11 and 23%, respectively, compared with untreated control plants. Foliar delivered SNE secured the photosynthetic pigment, free amino acids, soluble proteins, and soluble sugar content. Additionally, it lessened the adverse effects of Cd stress on D. innoxia plants by curtailing the content of hydrogen peroxide (H2O2) and malondialdehyde (MDA) by 28 and 27%, and 21 and 23%, respectively, compared with the plants subjected to 50 and 100 mg Cd kg-1 only. The findings herein indicated that the plant water extract and their interactions in the investigated Cd rates significantly augmented phenolics, alkaloids, reduced glutathione and phytochelatins content. Cell wall stiffening in D. innoxia indicated that lignin content and POX were significantly higher in plants exposed to 100 mg Cd kg-1 soil displaying increase values of 275 and 300%, respectively, against non-Cd treated control. The magnitude of increment imposed by Cd stress was lessened by using SNE that reflects on adequate cell growth advocated by limited lignification, in terms of lignin content, and downregulated POX activity. Owing to SNE application, root and leaves Cd contents were efficiently reduced reflecting apparent plant liveliness compared with the SNE non-treated Cd-stressed plants.
Conclusions
The outcomes of this study designate that foliar application of the Cd-hyperaccumulator S. nigrum leaf extract can be counted as an unconventional and innovative approach in the alleviation of Cd stress and can be employed as integrated practice when Cd-contaminated regions were exploited for sustainable agriculture of the multipurpose plants.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Toxic metals have been added to agricultural soils due to an increased reliance on sewage wastewater irrigation and chemical fertilizers, as well as quick industrialization, producing harmful impacts on plant and human health. Cd is one of the metal contaminants; it ranks seventh on a list of the most harmful toxins to crops in Egypt and around the world, and its long residence time in soil makes it a common environmental distress for the agricultural system (Ibrahim et al. 2022). Nowadays, plants may encounter more Cd due to the use of phosphorites and phosphatic fertilizers which contain threshold levels of Cd (4.77 mg kg-1). In plants, Cd exposure affects a wide range of physiological and biochemical processes, resulting in stunted growth, chlorosis, and oxidative stress initiated by the generated of reactive oxygen species (ROS) (Gutsch et al. 2018). The photosynthetic apparatus and water balance are also affected by stomatal closure in plants (Perfus-Barbeoch et al. 2002). Cd stress can be mitigated in living cell by minimizing the toxic effects, such as detoxification through compartmentation, chelation, and sequestration in extra cytoplasmatic compartments such as the cell wall (Jha and Bohra 2016).
Toxic metal tolerance comprises several adaptive scenarios at the organelle, cellular, and plant levels to cope up with metal toxicity including overproduction of soluble sugars, free amino acids, soluble proteins and medicinally active alkaloids (Saad-Allah and Elhaak 2017), osmoprotectants and antioxidant molecules (proline, betaine, glycine, phenolic compounds, flavonoids) (Khalid et al. 2019) in all plant tissues. Accordingly, it is very imperative to explore the mechanisms of Cd tolerance to ascertain which plants have ability to elicit physiological and biochemical mechanisms in response to Cd exposure. Furthermore, the practice of sustainable agricultural approaches to recuperate plant growth and productivity even under environmental stress can maintain the gaining of plants’ yield under predicted climatic fluctuations (Li et al. 2021).
Cd-hyperaccumulators are the only species that can survive and reproduce in soil solutions containing Cd concentrations above 35 μM (Dobrikova et al. 2021). The Cd tolerance in hyperaccumulators plants mostly relays on the complexation of metalloids in vacuoles and osmotic adjustment (Xu et al. 2009). Moreover, the mechanism of tolerance of hyperaccumulators plants is accomplished through the manufacturing of low molecular weight components viz. vitamins E and C, glutathione, and phytochelatins, as well as phenolic compounds that quash ROS (Xu et al. 2009). Cd-hyperaccumulators are focused owing to their photosynthesis, physiological properties, and Cd resistance along with their responding to oxidative stress (ur Rehman et al. 2019). It can also be used in sustainable agriculture under conditions of heavy metal stress and integral ultrastructure and anatomy (Tao et al. 2020). Some Cd-accumulator plants have various potentials benefits viz. potential source of a Cd-resistant gene (Kumar et al. 2021) as well as being an perfect source of generating several medicinal components (Pandey et al. 2019). Recently, utilizing plant extracts derived from plants that are highly tolerant to heavy metal toxicity has been beneficial in improving the growth of plants grown under metal toxicity conditions (Yang et al. 2021). Several studies has declared that hyperaccumulator intercropping (Lin et al. 2018) or applying hyperaccumulator straw or extracts to soil significantly enhance growth and Cd tolerance capacity of the target plants (Han et al. 2020). Also, foliar application of hyperaccumulator extracts can be more feasible and efficient as absorption of extract's nutrients by leaves is vastly diverse from absorption of the same nutrients by roots. Roots are bathed in a soil solution that is usually relatively dilute. The concentration of the nutrients is controlled to some extent by the surrounding soil. In foliar applications, the aqueous state evaporates rapidly after application and absorption must happen from concentrated solution (Gray and Akin 1984).
Solanum nigrum L. named as black nightshade was for the first time recognized as a Cd hyperaccumulator by Wei et al. (2005). Further investigations verified S. nigrum to be a Cd hyperaccumulator with Cd concentrations of above 100 mg kg-1 leaf DW (> 0.01%) and bio-concentration factor (BF) for its roots higher than 1 (Wang et al. 2015). The ability to uptake and accumulate bulky amounts of Cd in its leaves (124.6 mg kg-1 DW) assigns this plant an effective phytoremediator (Wei et al. 2005) as well. The tolerance mechanisms of Cd-mediated stress in S. nigrum are ascribed to the following: (1) release of organic acids that involved in coordinated and complexed with metal in vacuole away from active cellular components; heavy metal complexation by organic acids may play an important role in metal detoxification, transportation, and storage (Sun et al. 2006). (2) Activation of natural resistance-associated macrophage proteins (NRAMPs) (Song et al. 2014). (3) Antioxidants; phytochemical analysis of SNE revealed the presence of various bioactive compounds such as tannins, flavonoids, phenol, glycosides, steroids, coumarins, and quinones which shoulder an important role in Cd-binding (Kumar et al. 2020).
Datura innoxia belongs to solanaceae family has received high awareness all over the world (Jamshidi-Kia et al. 2018). It is cultivated for the production of secondary metabolites with commercial importance due to their extensive use in medicine, which comprise torpane alkaloids such as atropine, scopolamine, and hyoscyamine. They are exploited as parasympathicolitics for their ability to suppress parasympathetic nerve activity (Maheshwari et al. 2013). On the other hand, after alkaloid extraction, the remaining biomass can be also used for industrial applications such as bioethanol production. The less digestible stems counted more than 50% of its biomass, with high-yielded cell wall material (Wiart 2006).
To cope up with global growing demands of the multipurpose plants including D. innoxia, farmers and breeders in arid and semi-arid regions are prompted to often engage with the cultivation in low-grade soils such of those are heavy metal-contaminated ones. Consequently, plant will instantly combat the heavy metal stress. Since the plant chemical composition and the structure and composition of cell wall are likely affected by modified environmental conditions, this may influence plant potential value and quality as well. Thus studying the changes in lignin content and its mediated enzyme (cell wall-bound peroxidase, POX) under HMs stress may stipulate information on the effect of on cell wall structure as a mechanical barrier against Cd experience and a valuable criterion that imparts the plant quality.
To the best of the authors’ knowledge, this is the first study figuring the impact of foliar spraying with SNE on Cd-stressed D. innoxia. Therefore, the purpose herein is to examine the attenuating effect of foliar applied extract of Cd-hyperaccumulator S. nigrum on Cd-stressed D. innoxia performances that might be evaluated through some physio-biochemical indices viz. growth traits, photosynthetic pigment content, osmo-metabolic compounds, phenolics, alkaloids, phytochelatins and reduced glutathione content, oxidative damage, and lignin content and its mediated enzyme, POX, in Cd-stressed D. innoxia plant.
2 Materials and Methods
2.1 S. nigrum Collection and Extract Preparation
The current species were gathered from its natural habitat at Wadi Al-Assiuty (31°18′ and 31°48′ E and 27°10′ and 27°45′ N), Assiut Governate, Egypt. The Flora of Egypt (Hepper and Boulos 2001) was used for authentication of the plant. Samples were air-dried and ground to a grainy powder. The powder was then mixed with bidistilled water in ratio of a weight: volume (W/V) of 1:10 and put in a water bath at 80 °C for 25 min. The fresh extract was filtered through a Watman filter paper and left to cool at room temperature (Abeed et al. 2021). The resultant filterate was handled as 100% S. nigrum water leaves extracts and diluted to 50% for the usage as foliar spraying. The fresh prepared water extract was immediately used for foliar spraying. This concentration was chosen based on a previous preliminary experiment including five concentrations, i.e., 10, 20, 30, 40, 50%. The plants received 50% S. nigrum water leaves extract recorded a significant positive response based on biomass weight and chlorophyll concentration. S. nigrum leaf extract was analyzed and its biochemical constituents were as illustrated in Table 1.
2.2 Plant Materials, Growth Conditions, and Treatments
Two-week-old uniform D. innoxia plantlets were collected on summer 2021 from botanical garden of faculty of agriculture, Assiut University, and were transplanted in plastic pots (35 cm diameter) filled with 5 kg of sandy-clay soil in rate of 4 plantlets/pot. The pots were arranged in a completely random arrangement with four replicates. Seven days after transferring, three Cd concentrations (0, 50, and 100 mg Cd kg-1 soil) were delivered to the soil in a form of water solution. Cd application was introduced as cadmium dichloride (CdCl2). The developed plants of both control (0 mg Cd kg-1 soil ) and Cd (50 and 100 mg Cd kg-1 soil) treated pots were sprayed with SNE after one week of treatment with Cd, while the untreated control plants were sprayed with distilled water only. Two foliar spray treatments (water, SNE) were applied using hand pump trigger sprayers. Each replicate was sprayed by 250 ml of sprays per pot and the soil surface was covered by polyethylene bag to avoid foliar treatment reached soil. Plants were received sprays for two consecutive days. The addition of cadmium was based on our previous studies (Eissa and Abeed 2019); also, a preliminary experiment has revealed a high threshold of D. innoxia against Cd stress. Plant samples were harvested after 14 days of Cd exposure for analyzing the following parameters.
2.3 Analysis of Plant Growth Parameters
Plants were randomly selected from each treatment to estimate shoot and root length, fresh and dry weight of shoot and root, and leaf area at the end of the experiment. The length of shoot and root of D. innoxia plants were manually determined using a measuring tape. Fresh weight of the harvested plants was measured immediately then oven dried at 60 °C for 48 h to a constant weight to assess dry weight. Leaf area (cm2) was estimated via the product of the maximum length and maximum width of the leaf. The length and the width in cm were measured using a measuring tape.
2.4 Analysis of Photosynthetic Pigment Content
The photosynthetic pigments (chlorophyll a, chlorophyll b and carotenoids) in dry leaves were determined by suspending 0.25 g leaf sample in 10 ml ethyl alcohol (95%) in water bath at 60–70 °C. The absorbance of the extract was measured using a Unico UV-2100 spectrophotometer at wavelengths 663 and 644. Lichtenthaler (1987) equations were used for chlorophyll determinations as mg g-1 DW.
2.5 Analysis of Osmo-metabolic Compounds
Proline content was quantified in the fresh leaves according to Bates et al. (1973). Total free amino acids content was measured in the fresh leaves by the method of Moore and Stein 1948). The method of Lowry et al. (1951) was used to estimate soluble proteins content of fresh leaves. Soluble sugar content of the fresh leaves was determined by adopted the method of Fales (1951).
2.6 Quantification of Phenolics, Alkaloids, Reduced Glutathione, and Phytochelatins Content
Phenolic content was estimated according to Kofalvi and Nassuth (1995) using the Folin-Ciocalteu’s phenol reagent. A routine quantification method for analysis of the total alkaloidal content spectrophotometrically based on Dragendorff’s reagent (DR) as described by Sreevidya and Mehrotra (2003) was carried out. Non-enzymatic antioxidant as reduced glutathione (GSH): the supernatant of grinding fresh leaves in trichloroacetic acid was utilized for the quantification of reduced glutathione (GSH) by protocols of Ellman (1959). Phytochelatins (PCs) evaluated by subtracting the amount of GSH from non-protein thiols as mentioned by Nahar et al. (2016) which attained by mixing supernatant of leaves grounded in sulfosalicylic acid with Ellman’s reaction mixture (Ellman 1959).
2.7 Analysis of Oxidative Stress Markers; Hydrogen Peroxide, and Malondialdehyde Content
Hydrogen peroxide (H2O2) content of fresh leaves was spectrophotometrically estimated as described by Mukherjee and Choudhuri (1983). Malondialdehyde (MDA) content in the fresh leaves of D. innoxia plant was measured following to Zhang and Qu (2004).
2.8 Analysis of Lignin Content and Its Mediated Enzyme (Cell Wall-Bound Peroxidase, POX)
Lignin content was quantified via the thioglycolic acid reaction adopted by Kováčik and Klejdus (2008). The activity of peroxidases (POX, μmol mg-1 protein g-1 FW min-1) was measured after the extraction of the enzymes from leaves according to cited by Ghanati et al. (2002). The activity peroxidases (PO) was evaluated based on the increase in the absorbance at 470 nm using 168 mM guaiacol in 100 Mm phosphate buffer and 30 mM H2O2. The change in absorbance was modified to units (U) utilizing an extinction coefficient of 26.6 mM−1 cm−1.
2.9 Analysis of Cadmium Content of Root and Leaf of D. innoxia Plants
Cadmium (Cd) content was determined by following the method adopted by Eissa and Abeed (2019) in which dried leaf and root samples (0.5g) were ground and exposed to the acid-digestion using HNO3:HClO4 mixture (2,1). Cd concentrations were detected by atomic absorption spectrophotometer (PerkinElmer A Analyst 200) which has a detection limit of 0.001 mg L-1 for cadmium. The translocation factor (TF) of Cd was estimated by applying the following equation: TF = Cd leaves content/Cd root content (Li et al. 2021)
2.10 Statistical Analysis
A randomized complete design (RCD) with four replicates was applied in the present experiment. Duncan’s multiple range tests and one -ANOVA were proceed by SPSS 17.0 package (SPSS, Chicago, IL, USA) at probability level of 5%.
3 Results
3.1 S. nigrum Leaf Extract Improved D. innoxia Growth Under Cd Stress
To investigate how SNE application aided D. innoxia to combat Cd-induced adverse effect on the growth of D. innoxia plants, several growth-conjugated attributes were analyzed, including root and shoot fresh, dry weight, length, and leaf area, under both unstressed and stressed conditions with and without SNE foliar application (Fig. 1). In comparison with the control (0 mg Cd kg-1 soil), 50 and 100 mg Cd kg-1 soil decreased root FW (by 29 and 51%) and root DW (by 35 and 70%), shoot FW (by 29 and 54%), shoot DW (by 40 and 53%), root length (by 30 and 69%), shoot length (by 40 and 60%), area of leaves (by 11 and 23%) , respectively (Fig. 1). Foliar spray of D. innoxia plants with SNE improved root FW (by 25 and 26%) and root DW (by 23 and 100%), shoot FW (by 20 and 38), shoot DW (by 22 and 29%), root length (by 45 and 54%), shoot length (by 33 and 31%), area of leaves (by 6 and 7%), at 50 and 100 mg Cd kg-1 soil levels, respectively, versus plants subjected to Cd alone (Fig. 1). Furthermore, D. innoxia plants sprayed with SNE only exhibited increment in root FW, root DW, shoot FW, shoot DW, root length, shoot length, and leaf area by 3, 20, 6, 7, 11, 12, and 1%, respectively, over the control plants (Fig. 1).
Impact of Cd stress, foliar application with Solanum nigrum extract and their interactions on Fresh weight of shoots (g/plant) (a), dry weight of shoot (g/plant) (b), fresh weight of root (g/plant) (c), dry weight of root (g/plant) (d), shoot length (cm) (e), root length (cm) (f), and area of leaves (cm2) (g) in Datura innoxia plants. Water and Solanum nigrum leaf extract (SNE) indicate the plants sprayed with distilled water, Solanum nigrum extract, respectively. Bars donate averages and standard errors of four independent replicates (n = 4). Different alphabetical letters designate significant differences among the treatments at P < 0.05, based on LSD test.
3.2 S. nigrum Leaf Extract Protected Photosynthetic Pigments in D. innoxia Leaves Under Cd Stress
To assess the protective impact of exogenous SNE on the photosynthetic pigments under Cd toxicity, the levels of photosynthetic pigments (chlorophyll a, b and carotenoids) in Cd-delivered D. innoxia leaves were evaluated (Fig. 2). In comparison to the untreated control plants, there was a drastic decrement in the content of chlorophyll a by 46 and 69%, chlorophyll b by 63 and 88%, and carotenoids by 56 and 78% in the plants subjected to 50 and 100 mg Cd kg-1 stress, respectively (Fig. 2). Contrary, spraying the extract of S. nigrum protected photosynthetic pigments from Cd-mediated injurious effects, as witnessed by the considerable increased contents of chlorophyll a (57 and 100%), chlorophyll b (67 and 200%), and carotenoids (200 and 50%) as response to 50 and 100 mg Cd kg-1 rates, respectively, in comparison with only Cd-treated plants (Fig. 2). Non-stressed D. innoxia plants delivered SNE also exhibited enhanced contents of chlorophyll a (by 15%), chlorophyll b (by 13%), and carotenoids (by 11%), in comparison with water-sprayed non-Cd treated control (Fig. 2).
Impact of Cd stress, foliar application with Solanum nigrum extract and their interactions on chlorophyll a (mg g-1 DW) (a), chlorophyll b (mg g-1 DW) (b), and carotenoids (mg g-1 DW) (c) in Datura innoxia plants. Water and Solanum nigrum leaf extract (SNE) designate the plants sprayed with distilled water, Solanum nigrum extract, respectively. Bars donate averages and standard errors of four independent replicates (n = 4). Different alphabetical letters designate significant differences among the treatments at P < 0.05, based on LSD test. FW: fresh weight
3.3 S. nigrum Leaf Extract Modulated Osmo-metabolic Compound Accumulation in Cd-Exposed D. innoxia Plants
The imposition of 50 and 100 mg Cd kg-1 Cd led to an augmentation in proline content by 147 and 293% and total free amino acids by 26 and 23%, whilst it resulted in a decrement in soluble proteins content by 20 and 35% and soluble sugar content by 11 and 18%, respectively, compared with that of non-Cd treated control plants (Table 2). On the other hand, application of SNE to the plants exposed to 50 and 100 mg Cd kg-1 resulted in an exacerbation of proline accumulation by 29 and 56%, and total free amino acids by 4 and 4%, and an increase in soluble proteins by 9 and 15%, and soluble sugars by 11 and 12%, respectively, when compared with the plants subjected to the same levels of Cd stress without SNE spraying (Table 2). SNE spraying treatment to non-Cd treated plants increased the contents of proline, total free amino acids, soluble proteins, and soluble sugars by 10, 18, 40, and 2%, respectively, versus control plants (Table 2).
3.4 S. nigrum Leaf Extract Enhanced Phenolics, Alkaloids, Reduced Glutathione, and Phytochelatins Content Under Cd Stress Conditions
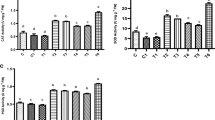
D. innoxia plants that are grown in 50 mg Cd kg-1 level exhibited a considerable increment in phenolics and alkaloid content (by 88 and 48%), respectively (Fig. 3a, b) versus the untreated control sample. Whereas plants grown in 100 mg Cd kg-1 level displayed drastic depletion in phenolics and alkaloid content (by 36 and 37%), respectively (Fig. 3a) versus the unstressed control sample. Obviously, supplementation of SNE to Cd-treated plants remarkably augmented phenolics (by 26 and 175%) and alkaloid content (by 25 and 68%), respectively, in comparison with the plants subjected to the same levels of Cd stress without SNE spraying (Fig. 3). In addition, SNE foliar spraying to non-stressed plants insignificantly enriched the phenolics content (by 12%) and alkaloid content (by 6%) when compared with that of water-sprayed non-stressed control (Fig. 3). D. innoxia plants that were grown under 50 and 100 mg Cd kg-1 levels displayed an augmented content of GSH (by 67 and 133%), and PCs (by 80 and 200%), respectively, in correspondence with non-stressed control (Fig. 3c, d). Supplement of SNE to Cd-stressed plants displayed a vital role in further exacerbation of GSH content (by 32 and 29%) and PCs (by 50 and 33%), respectively, compared with the plants subjected to 50 and 100 mg Cd kg-1 only (Fig. 3c, d). SNE spraying treatment to unstressed plants insignificantly decreased the contents of GSH and PCs by 20 and 10%, respectively, against control plants (Fig. 3).
Impact of Cd stress, foliar application with Solanum nigrum extract and their interactions on phenolics content (mg g-1 DW) (a), alkaloid content (mg g-1 DW) (b), reduced glutathione content (GSH; μmol g-1FW) (c), and phytochelatins (PCs; μmol g -1FW) (d) in Datura innoxia plants. Water and Solanum nigrum leaf extract (SNE) indicate the plants sprayed with distilled water, Solanum nigrum extract, respectively. Bars donate averages and standard errors of four independent replicates (n = 4). Different alphabetical letters designate significant differences among the treatments at P < 0.05, based on LSD test. DW: dry weight
3.5 S. nigrum Leaf Extract Reduced Cd Stress-Induced Oxidative Injury in D. innoxia Plants
D. innoxia plants grown under 50 and 100 mg Cd kg-1 levels exhibited an increased level of H2O2 by 66 and 98%, and MDA by 85 and 131%, respectively, when compared with non-stressed control (Fig. 4a). Application of SNE to Cd-stressed plants had a pivotal role in curtailing H2O2 level (by 28 and 27%, respectively) and MDA (by 21 and 23%, respectively) compared with the plants subjected to 50 and 100 mg Cd kg-1 only (Fig. 4a, b). Under normal conditions D. innoxia plants sprayed with SNE displayed decreasing in the level of MDA and H2O2 by 13 and 15%, respectively, compared with that of untreated control (Fig. 4).
Impact of Cd stress, foliar application with Solanum nigrum extract and their interactions on MDA (μmol g-1 FW) (a) and H2O2 (μmol g-1 FW) (b) in Datura innoxia plants. Water and Solanum nigrum leaf extract (SNE) indicate the plants sprayed with distilled water, Solanum nigrum extract, respectively. Bars donate averages and standard errors of four independent replicates (n = 4). Different alphabetical letters designate significant differences among the treatments at P < 0.05, based on LSD test. FW: fresh weight
3.6 S. nigrum Leaf Extract Altered Lignin Content and Its Mediated Enzyme (Peroxidase, POX) Activity of Cd-Exposed D. innoxia Plants
D. innoxia plants that were grown under 50 and 100 mg Cd kg-1 levels displayed an augmented content of lignin by 100 and 257%, respectively, in correspondence with non-Cd treated control (Fig. 5a). Application of SNE to Cd-stressed plants shouldered a pivotal role in minimizing the level of lignin (by 36 and 40%, respectively) compared with the plants subjected to 50 and 100 mg Cd kg-1 only (Fig. 5a). Besides, D. innoxia plants exposed to 50 and 100 mg Cd kg-1 levels elevated the activity of POX by 250 and 300%, respectively, against non-Cd treated control (Fig. 4b). However, SNE application downregulated the activity of POX by 29 and 31% in Cd-exposed plants (50 and 100 mg Cd kg-1, respectively) versus stressed plants only (Fig. 5b). Under normal conditions, D. innoxia plants delivered SNE decreased lignin content insignificantly and POX activity significantly by 6 and 35%, respectively, in comparison with that of untreated control (Fig. 5).
Impact of Cd stress, foliar application with Solanum nigrum extract and their interactions on Lignin content (mg g-1 DW) (a) and Activity of peroxidase (POX; μmol mg-1 protein g-1 FW min-1) (b) in Datura innoxia plants. Water and Solanum nigrum leaf extract (SNE) indicate the plants sprayed with distilled water, Solanum nigrum extract, respectively. Bars donate averages and standard errors of four independent replicates (n = 4). Different alphabetical letters designate significant differences among the treatments at P < 0.05, based on LSD test.
3.7 S. nigrum Leaf Extract Altered Cadmium Content and Its Translocation Factor of Cd-Exposed D. innoxia Plants
Data presented in Table 3 showed that the increase in the soil cadmium level caused an increase in cadmium content of root and leaves tissues. Lower amount of cadmium was recorded in leaves than root. Application of SNE significantly decreased cadmium content of root and leaves tissues in plants exposed to 50 and 100 mg Cd kg-1 by 34.5 and 56.9%, and 11.5 and 67.9%, respectively, when compared with the plants subjected to the same levels of Cd stress without SNE spraying. The translocation of Cd from roots to leaves D. innoxia plants was expressed by the translocation factor (TF) (Table 3). In plants subjected to 50 and 100 mg Cd kg-1, translocation factor increased from 0.651 to 0.795. Surprisingly, plants received SNE exhibited the lowest TF.
4 Discussion
Heavy metal tolerance is established as a complicated process, so looking for another approach encompassing the exploiting of metal-hyperaccumulators extracts as naturally pervasive to metalliferous soils species (Hagemeyer 2004) may provide some promising perceptions to a higher potential of detoxification capacity of plants (Shanmugaraj et al. 2019). The detrimental influence of high Cd level in D. innoxia performance can be cleared to decrease the area of leaves, fresh, dry weight, and length of shoot and root of D. innoxia plants grown under Cd stress. The growth reduction influence of Cd on D. innoxia plants may be due to different factors; among them are oxidative burst, nutrients deficiency, and water balance disturbance (Sarwar et al. 2010). Moreover, cell elongation, cell division, and an amplification may be repressed by Cd stress as demonstrated by El Rasafi et al. (2020).
Utilizing natural plant extracts has been registered in the literature to underpin the growth of various plants such as wheat (Tomar et al. 2015), and venca (Abeed et al. 2021). Stimulating impacts of SNE were noticeable owing to the existence of citric acid (CA), ascorbic acid (AsA) and proline (as antioxidants) and some micro and macronutrients (Fe, iron; Zn, zinc; Mg, magnesium) in the plant extract (Table 1). Study of Zeng et al. (2021) on castor bean proved that foliar sprayed citric acid alleviated lead (Pb) stress via improving growth, as well as fresh and dry biomass. Also, a study of Zhang et al. (2019) on Cd-stressed maize plants revealed that AsA application induced growth improvement via stimulation in photosynthetic pigments, amino acids, and protein, which might have enhanced cell enlargement and growth of plant (Aziz et al. 2018). Study of Zouari et al. (2016) indicated that proline enhanced growth rate of date palm (Phoenix dactylifera L.) under Cd stress. In addition to Mahdieh et al. (2018) who pointed that Zn as a micronutrient caused an increment in the growth parameters (root and shoot lengths, dry and fresh weights, and number of leaves) of Phaseolus vulgaris plants. Wu et al. (2020) stated that Zn foliar sprayed enhanced the photosynthesis, tissue biomass of wheat under Cd stress.
Photosynthetic pigments and carotenoids are fundamental for photosynthesis in the plant (Abeed et al. 2020; Ding et al. 2021). The drop in photosynthetic pigments may be ascribed to reduction in leaf area which shoulders light capturing and photosynthesis achieving (Xu et al. 2009). Moreover, diminutions in photosynthetic pigments resulted from chlorophyll destruction by triggering activity of chlorophyllase and chlorophyll degrading enzymes under environmental stress circumstances (Vernon and Selly 1966; Sayyari et al. 2013). The protective effect SNE on photosynthetic pigment and carotenoids could be owing to SNE with a high incidence of AsA, Mg, Ca (calcium), Zn and Fe ions (Table 1), which directly influence photosynthetic pigments since Fe and Ca activate the chlorophyll biosynthesis, whereas ascorbic acid acts as an antioxidant. It was also established to be conjugated with several biological activities in the plant as an enzyme catalyst and as a donor/receptor in electron transport (Abdel-Hafeez et al. 2019). Zhang et al. (2019) also continued the results of AsA-mediated protection of chlorophyll versus environmental stresses. Hence, it is shown that foliar spraying with AsA withstand the negative effects of Cd on photosynthesis and retained pigments biosynthesis and related components.
Among the diverse plant responses to Cd stress is generation of compatible osmoprotectants. This aids cells to decline oxidative injury produced by ROS in response to high Cd stress level via protecting sub-cellular cells and also displaying osmotic adjustment (Abeed and Dawood 2020). Documented drops in soluble sugars are linked to a decrease in leaves photosynthetic pigments that resulted in suppression of photosynthetic activity and employment of carbohydrates into another sink.
In this concern, Yaghoubian et al. (2016) who documented that Cd stress declines photosynthetic pigment and total carbohydrates due to photosystem II higher sensitivity. The decrease of soluble proteins in D. innoxia plants was demonstrated by Dawood and Abeed (2020) which was inhibited by the buildup of proline and total amino–N. Moreover, Hussain et al. (2020) deduced this reduction as a result of the disturbance in nitrogen metabolism or nitrate absorption inhibition. On the other hand, spraying with SNE enhances the ability of Cd-stressed D. innoxia plants to intensify levels of soluble sugars and soluble proteins. These findings designated that SNE may have participated in osmo-tolerance of Cd-exposed D. innoxia plants by stimulation of carbohydrates production in response to Zn, which has an important role in activation of the enzymes that are responsible for carbohydrates biosynthesis and transformation (Gheshlaghi et al. 2019). The existence of Ca in the Cd-hyperaccumulator extract may increases protein synthesis or decrease protein degradation in Cd-treated plants.
There is a close relationship between the accumulation of amino acids, proline and potency of plants to survive against Cd stress. Proline acts for osmotic adjustment and donates the stability of the subcellular structures to scavenge ROS and to neutralize cellular redox potential in addition to its role as a signaling molecule and source of energy (Szabados and Savouré 2010). Results of this study witnessed highly significant increases in total free amino acids and proline contents in Cd-exposed plants. Comparable results were detected by Yılmaz and Parlak (2011). The rehabilitation from the noxious effect of Cd stress has manifested as a result of the existence of enriched content of ascorbic acid and proline in the extract of S. nigrum plant and this increase in amino acids could be ascribed to ascorbic acid suppressed the rise of the ROS (Shalata and Neumann 2001). Ascorbic acid plays as a detoxification agent to destructive damage induced by free radicals (Asada 1999). Perveen et al. (2016) documented that alfalfa leaf extract conferred Cd resistance of wheat plant by increasing leaf free proline and total free amino acids thus increased growth and yield of plants under Cd stress.
Alkaloids and Phenolics act as non- enzymatic antioxidants that enhance the antioxidant capacity of a cell to scavenge ROS liberated under Cd stress (Gill and Tuteja 2010). Alkaloids metabolites are not essential for the completion of life cycle of the plant; rather frequently function as signal molecules, or chemical defenses against stress conditions (Vernay et al. 2008). Also, phenols serve as a substrate for many antioxidants enzymes, so, it mitigates Cd stress injuries. Evaluation the content of alkaloids and total phenolic compounds in this investigation denotes the non-enzymatic level of ROS scavenging, and we noticed that the levels of both alkaloids and phenolic compounds were increased in the shoots of D. innoxia plants supplied with moderate Cd level (50 mg kg-1) confirming the hypothesis of the external constraints (e.g. HMs) which restrict the rate of dry matter accumulation may elicit the alkaloids manufacture (Vernay et al. 2008). It has also been postulated that secondary metabolism may be an integral part of the plant capacity to adjust metabolic processes to survive and reproduce in abiotic stress involving HMs existence. Our findings are parallel with the finding in Narcissus tazetta (Soleimani et al. 2020), Papaver somniferum (Lachman et al. 2006) and Brassica juncea (Kaur et al. 2018). Furthermore, Manquián-Cerda et al. (2016) who reported that the content of phenolic compounds significantly increased in 100 μM Cd-stressed blueberry (Vaccinium corymbosum L.) leaves. While under high Cd level (100 mg kg-1), D. innoxia plant showed considerable depletion in the content of alkaloids and phenolics indicating that Cd contamination can alter the chemical composition of D. innoxia leaves, thus, affecting the quality, efficacy, and safety of natural plant derivatives produced by medicinal species. The lack of capacity to produce these molecules (alkaloids and phenolics) in leaves under high Cd level suggests the loss of biosynthetic components that may be due to an inactivation of enzymes or a redirection of metabolic functions to maintain growth (Vernay et al. 2008). Supplementation of SNE exacerbated content of alkaloids and phenolics significantly in moderate Cd-stressed plants and re-nourished alkaloids and phenolics content of leaves in highly Cd-stressed plants, thereby participating adjustment of osmotic status or an increase in plant hormone activities. Thus, the overaccumulation of plant secondary metabolite acts as a plant adaptive mechanism in response to Cd stress (Ashraf et al. 2018). These results are in harmony with that of Howladar (2014) and Khalofah et al. (2020) who indicated that foliar application with moringa leaf extract significantly augmented the content of total phenols and the antioxidant potential in shoots of Phaseolus vulgaris and Lepidium sativum plants when compared to plants grown under Cd stress.
One of mechanisms to metal detoxification is by conjugation or chelation and compartmentalizes them in the vacuole away from the cytosolic environment. In plant cell, GSH and PCs shoulder the coordinate of Cd by binding it with the thiol group. In the current study, GSH and PCs display similar responses to Cd treatments (both increased), probably due to their co-regulation. High glutathione levels facilitate phytochelatins synthesis by the activation of the enzyme phytochelatin synthase thereby sequestration of heavy metal phytochelatin conjugates in the vacuole. Several investigations indicated that exacerbated GSH content is associated with capability of plants to counter Cd-induced oxidative stress (Hossain et al. 2010). SNE enhanced their content for efficient Cd-detoxification, and the subsequent tolerance of Cd toxicity. These results may be ascribed to biochemical components denoted in the extract such as proline and citric acid. Similar results of Xu et al. (2009) proved that proline application serves in attenuating Cd toxicity by detoxifying ROS, boosting the glutathione level and protecting the activity of antioxidative enzymes in Cd-stressed plants. Al Mahmud et al. (2018) showed that citric acid confer Cd stress tolerance via enhancing the pool of ascorbate (AsA) and glutathione (GSH) and increasing the phytochelatins (PCs) content. Furthermore, GSH can play a key role in ROS detoxification and altering cellular redox status of protein via the AsA-GSH pathway in plants this pathway was activated by natural amended ascorbic in SNE (Table 1).
In the current study, D. innoxia plants grown under different Cd levels displayed higher content of hydrogen peroxide and malonaldehyde. Production of malonaldehyde in plant amplified under elevated Cd stress levels as a result of the plant losing its ability to hamper ROS whilst hydrogen peroxide posed membrane injury by the generation of hydroxyl radical (OH-) and lipid peroxidation (Das and Roychoudhury 2014). Addition of SNE to Cd-stressed D. innoxia plant registered the ability to counteract the noxious impacts mediated by Cd stress through lessening MDA and H2O2 hyperaccumulation. These results may be ascribed to biochemical components denoted in the extract such as ascorbic acid, proline and organic acids viz. citric acid (Table 1) that shoulders an important role in declining ROS levels in the Cd stressed plants via activation of antioxidant enzymes (Eissa and Abeed 2019). These findings are in line with Weijie et al. (2021). AsA was linked to ROS metabolism protecting plant tissues from detrimental oxidative injury by serving as reductant thus decreased free radicals level in cell (Zhang et al. 2019). Citric acid has been reported in iron-generated stress, aluminum detoxification, and tolerance towards heavy metal (Gao et al. 2010; Faraz et al. 2020).
We found lignin content was highly abundant and its biosynthetic peroxidase activity was triggered in Cd-exposed plants. Cd is recognized to motivate oxidative stress as a signaling molecule; H2O2 elicits secondary reactions, such as an induced peroxidase activity, which contributes to boosted lignification (Finger-Teixeira et al. 2010). Lignification results in decrement of the cell wall extensibility which might limit cell enlargement as evidenced by reduced growth and stunted D. innoxia plants grown under Cd stress.
Lignin is cell wall material that highly deposited as a mechanical barrier against external stressor, e.g. HMs, preventing metal entrance (stress lignin) (Bruce and West 1989). Addition of SNE to Cd-stressed D. innoxia plants showed the ability to reduce POX activity and subsequent adequate lignification regulated from high inducible rate (restricting cell growth) to relatively low or moderate level to the extent that permits cell elongation and growth. Normal lignin deposition accompanied with reduced POX activity under SNE application may be mostly attributed to the represented ascorbic acid in the extract (Table 1). Peroxidase has been proposed to play an vital role in the lignification of cell walls which can oxidize phenolics to phenoxy radicals, whereas AsA has a regulatory role in the oxidation of phenolics. The oxidation is completely suppressed by a low AsA concentration. AsA compete phenolics as a substrate result in oxidation of AsA into dehydroascorbic acid rather than production of phenoxy radicals (Takahama 1993). These radicals, formed via phenolics oxidation, can bind to cell wall leading to lignification or formation of cross-links in walls, but this would be impossible as long as any AsA was present in cell walls (Takahama 1993).
The lower content of Cd in leaves than roots jointed with absence of toxicity symptoms in SNE treated Cd-stressed plants could rationalize the importance of Cd root-retention mechanism established by D. innoxia plants in order to protect aerial parts versus the toxic influence of Cd. Similarly, Guo et al. (2015) reported that Cd ions were mainly retained in the roots of Thuya plants (Platycladus orientalis) and juniper (Juniperus chinensis) submitted to Cd stress, and therefore, small amounts of Cd were transported to the leaves.
Also, the current study revealed that SNE application reduced the Cd content in roots and leaves of Cd-stressed D. innoxia plants that witnessed by the apparent plant liveliness compared with the SNE non-treated Cd-stressed plants. These results may be maily ascribed to proline denoted in the extract. Hence, similar results of Zouari et al. (2016) proved that proline application not only serves in reduced Cd uptake by date palm roots but also reinforced its exclusion, and therefore, exogenous proline could constitute a barrier against Cd absorption the matter that emphasized herein by the decrease of Cd translocation recorded in SNE-treated plants.
5 Conclusions
Using plant extracts derived from plants that are highly tolerant to heavy metal toxicity can be accounted as a satisfactory approach for a healthy future. Solanum nigrum leaf extract herein accomplished beneficial role in enhancing the performance of plants grown under cadmium toxicity via improving morphological attributes, photosynthetic pigments, osmo-metabolic compounds, cell extensibility accomplished by adequate lignification in terms of low lignin content and downregulated pexoidase activity, and non-enzymatic antioxidants that imparted its medicinal properties/quality as well as curtailed oxidative stress and reduced foliar cadmium content. Accordingly, it could be employed in the agricultural sector under cadmium stress conditions and as an opportune way in cadmium stress green mitigation in plants. To the best of the authors’ knowledge, this is the first investigation clarifying the impact of Solanum nigrum leaf extract on physio-biochemical performance of Datura innoxia under Cd stress and additional proteomic reports can provide information on the influence of Solanum nigrum leaf extract on plant metabolism under cadmium stress to ensure the safety and sustainability; thus, it can be utilized Solanum nigrum leaf extract as integrated practice when metal-contaminated regions were exploited for the agriculture of the multipurpose plants.
References
Abdel-Hafeez AA, Abd El-Mageed TA, Rady MM (2019) Impact of ascorbic acid foliar spray and seed treatment with cyanobacteria on growth and yield component of sunflower plants under saline soil conditions. Intern Lett Natu Sci 76:136–146. https://doi.org/10.18052/www.scipress.com/ILNS.76.136
Abeed A, Dawood M (2020) Comparative impact of different isoosmotic solutions on osmotic adjustment in Gossypium barbadense. J Global Nest 22:75–84. https://doi.org/10.30955/gnj.003106
Abeed AHA, Eissa MA, Abdel-Wahab DA (2020) Effect of exogenously applied jasmonic acid and kinetin on drought tolerance of wheat cultivars based on morpho-physiological evaluation. J Soil Sci Plant Nutr 21(1):131–144. https://doi.org/10.1007/s42729-020-00348-1
Abeed AHA, Ali M, Ali EF, Majrashi A, Eissa MA (2021) Induction of Catharanthus roseus secondary metabolites when Calotropis procera was used as bio-stimulant. Plants 10:1623. https://doi.org/10.3390/plants10081623
Al Mahmud J, Hasanuzzaman M, Nahar K, Bhuyan MHMB, Fujita M (2018) Insights into citric acid-induced cadmium tolerance and phytoremediation in Brassica juncea L.: coordinated functions of metal chelation, antioxidant defense and glyoxalase systems. Ecotox Environ Safe 147:990–1001. https://doi.org/10.1016/j.ecoenv.2017.09.045
Asada K (1999) The water-water cycle in chloroplast, scavenging of active oxygens and dissipation of excess photons. Ann Rev Plant Physiol Mol Biol 50:601–639. https://doi.org/10.1146/annurev.arplant.50.1.601
Ashraf MA, Iqbal M, Rasheed R, Hussain I, Riaz M, Arif MS (2018) Environmental stress and secondary metabolites in plants: an overview. Plant Metab Regul under Environ Stress:153–167. https://doi.org/10.1016/B978-0-12-812689-9.00008-X
Aziz A, Akram NA, Ashraf M (2018) Influence of natural and synthetic vitamin c (ascorbic acid) on primary and secondary metabolites and associated metabolism in quinoa (Chenopodium quinoa Willd.) plants under water deficit regimes. Plant Physiol Biochem 123:192–203. https://doi.org/10.1016/j.plaphy.2017.12.004
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Bruce RJ, West CA (1989) Elicitation of lignin biosynthesis and isoperoxidase activity by pectic fragments in suspension cultures of castor bean. Plant Physiol 91(3):889–897. https://doi.org/10.1104/pp.91.3.889
Das K, Roychoudhury A (2014) Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front Environ Sci 2:53. https://doi.org/10.3389/fenvs.2014.00053
Dawood MF, Abeed AH (2020) Spermine-priming restrained water relations and biochemical deteriorations prompted by water deficit on two soybean cultivars. Heliyon 6:e04038. https://doi.org/10.1016/j.heliyon.2020.e04038
Ding Z, Ali EF, Almaroai YA, Eissa MA, Abeed AHA (2021) Effect of potassium solubilizing bacteria and humic acid on faba bean (Vicia faba L.) plants grown on sandy loam soils. J Soil Sci Plant Nutr 21(1):791–800. https://doi.org/10.1007/s42729-020-00401-z
Dobrikova AG, Apostolova EL, Hanć A, Yotsova E, Borisova P, Sperdouli I, Adamakis I-DS, Moustakas M (2021) Cadmium toxicity in Salvia sclarea L.: an integrative response of element uptake, oxidative stress markers, leaf structure and photosynthesis. Ecotox Environ Safe 209:111851. https://doi.org/10.1016/j.ecoenv.2020.111851
Eissa MA, Abeed AH (2019) Growth and biochemical changes in quail bush (Atriplex lentiformis (Torr.) S. Wats) under Cd stress. Environ Sci Pollut Res 26(1):628–635. https://doi.org/10.1007/s11356-018-3627-1
El Rasafi T, Oukarroum A, Haddioui A, Song H, Kwon EE, Bolan N et al (2020) Cadmium stress in plants: a critical review of the effects, mechanisms, and tolerance strategies. Crit Rev Environ Sci Technol:1–52. https://doi.org/10.1080/10643389.2020.1835435
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82(1):70–77. https://doi.org/10.1016/0003-9861(59)90090-6
Fales F (1951) The assimilation and degradation of carbohydrates by yeast cells. J Biol Chem 193:113–124
Faraz A, Faizan M, Sami F et al (2020) Supplementation of salicylic acid and citric acid for alleviation of cadmium toxicity to Brassica juncea. J Plant Growth Regul 39:641–655. https://doi.org/10.1007/s00344-019-10007-0
Finger-Teixeira A, Ferrarese MDLL, Soares AR, da Silva D, Ferrarese-Filho O (2010) Cadmium-induced lignification restricts soybean root growth. Ecotox Environ Safe 73(8):1959–1964. https://doi.org/10.1016/j.ecoenv.2010.08.021
Gao Y, Miao C, Mao L et al (2010) Improvement of phytoextraction and antioxidative defense in Solanum nigrum L. under cadmium stress by application of cadmium-resistant strain and citric acid. J Hazard Mater 181:771–777. https://doi.org/10.1016/j.jhazmat.2010.05.080
Ghanati F, Morita A, Yokota H (2002) Induction of suberin and increase of lignin content by excess boron in Tabacco cells. Soil Sci Plant Nutr 48(3):357–364. https://doi.org/10.1080/00380768.2002.10409212
Gheshlaghi MZ, Pasari M, Shams B, Rokhzadi K, Mohammadi K (2019) The effect of micronutrient foliar application on yield, seed quality and some biochemical traits of soybean cultivars under drought stress. J Plant Nutr 42(20):2715–2730. https://doi.org/10.1080/01904167.2019.1655034
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930. https://doi.org/10.1016/j.plaphy.2010.08.016
Gray RC, Akin GW (1984) Foliar fertilization. In: Hauck RD (ed) Nitrogen in crop production. ASA, Madison, pp 579–584. Chapter 39. https://doi.org/10.2134/1990.nitrogenincropproduction.c39
Guo B, Liu C, Ding N, Fu Q, Lin Y (2015) Silicon alleviates cadmium toxicity in two cypress varieties by strengthening the exodermis tissues and stimulating phenolic exudation of roots. J Plant Growth Regul 1–10. https://doi.org/10.1007/s00344-015-9549-y
Gutsch A, Keunen E, Guerriero G, Renaut J, Cuypers A, Hausman J-F, Sergeant K (2018) Long-term cadmium exposure influences the abundance of proteins that impact the cell wall structure in Medicago sativa stems. Plant Biol 20(6):1023–1035. https://doi.org/10.1111/plb.12865
Hagemeyer J (2004) Ecophysiology of plant growth under heavy metal stress. In Heavy metal stress in plants. Springer, Berlin, pp 201–222. https://doi.org/10.1007/978-3-662-07743-6_8
Han R, Dai H, Twardowska I, Zhan J, Wei S (2020) Aqueous extracts from the selected hyperaccumulators used as soil additives significantly improve accumulation capacity of Solanum nigrum L. for Cd and Pb. J Hazard Mater 394(122553). https://doi.org/10.1016/j.jhazmat.2020.122553
Hepper FN, Boulos L (2001) Flora of Egypt Volume 1 (Azollaceae-Oxalidaceae). Kew Bull 56(4):944. https://doi.org/10.2307/4119305
Hossain MA, Hasanuzzaman M, Fujita M (2010) Up-regulation of antioxidant and glyoxalase systems by exogenous glycinebetaine and proline in mung bean confer tolerance to cadmium stress. Physiol Mol Biol Plants 16(3):259–272. https://doi.org/10.1007/s12298-010-0028-4
Howladar SM (2014) A novel Moringa oleifera leaf extract can mitigate the stress effects of salinity and cadmium in bean (Phaseolus vulgaris L.) plants. Ecotox Environ Safe 100:69–75. https://doi.org/10.1016/j.ecoenv.2013.11.022
Hussain S et al (2020) Metal toxicity and nitrogen metabolism in plants: an overview. In: Datta R, Meena R, Pathan S, Ceccherini M (eds) Carbon and Nitrogen Cycling in Soil. Springer, Singapore. https://doi.org/10.1007/978-981-13-7264-3_7
Ibrahim OHM, Ali EF, Eissa MA (2022) Jasmonic acid and EDTA-enhanced Cd and Pb phytoextraction by the halophytic plants quail bush [Atriplex lentiformis (Torr.) S. Wats]. J Soil Sci Plant Nutr. https://doi.org/10.1007/s42729-021-00743-2
Jamshidi-Kia F, Lorigooini Z, Amini-Khoei H (2018) Medicinal plants: past history and future perspective. J Herbmed Pharmacol 7(1):1–7. https://doi.org/10.15171/jhp.2018.01
Jha UC, Bohra A (2016) Genomics enabled breeding approaches for improving cadmium stress tolerance in plants. Euphytica 208:1–31. https://doi.org/10.1007/s10681-015-1580-3
Kaur R, Yadav P, Thukral AK et al (2018) Castasterone and citric acid supplementation alleviates cadmium toxicity by modifying antioxidants and organic acids in Brassica juncea. J Plant Growth Regul 37:286–299. https://doi.org/10.1007/s00344-017-9727-1
Khalid H, Zia-ur-Rehman M, Naeem A, Rizwan M, Ali S, Umair M, Sohail MI (2019) Solanum nigrum L.: a novel hyperaccumulator for the phyto-management of cadmium contaminated soils. In: Hasanuzzaman M, Prasad MNV, Fujito M (eds) Cadmium toxicity and tolerance in plants: from physiology to remediation. Academic Press, Elsevier Inc 451–477 Chapter 18. https://doi.org/10.1016/b978-0-12-814864-8.00018-8
Khalofah A, Bokhari NA, Migdadi HM, Alwahibi MS (2020) Antioxidant responses and the role of Moringa oleifera leaf extract for mitigation of cadmium stressed Lepidium sativum L. S Afr J Bot 129:341–346. https://doi.org/10.1016/j.sajb.2019.08.041
Kofalvi SA, Nassuth A (1995) Influence of wheat streak mosaic virus infection phenylpropanoid metabolism and the accumulation of phenolics and lignin in wheat. Physiol Mol Plant Pathol 47:365–377. https://doi.org/10.1006/pmpp.1995.1065
Kováčik J, Klejdus B (2008) Dynamics of phenolic acids and lignin accumulation in metal-treated Matricaria chamomilla roots. Plant Cell Rep 27:605–615. https://doi.org/10.1007/s00299-007-0490-9
Kumar J, Kaur A, Narang P (2020) Phytochemical screening and metal binding studies on floral extract of Solanum nigrum. Mater Today: Proceedings 26:3332–3336. https://doi.org/10.1016/j.matpr.2019.09.170
Kumar A, Subrahmanyam G, Mondal R, Cabral-Pinto MMS, Shabnam AA, Jigyasu DK, Malyan SK, Fagodiya RK, Khan SA, Yu Z-G (2021) Bio-remediation approaches for alleviation of cadmium contamination in natural resources. Chemosphere 268:128855. https://doi.org/10.1016/j.chemosphere.2020.128855
Lachman J, Hejtmankova A, Miholova D, Kolihová D, Tluka P (2006) Relations among alkaloids, cadmium and zinc contents in opium poppy (Papaver somniferum L.). Plant Soil Environ 52(6):282. https://doi.org/10.17221/3442-pse
Li J, Chang Y, Al-Huqail AA, Ding Z, Al-Harbi MS, Ali EF et al (2021) Effect of manure and compost on the phytostabilization potential of heavy metals by the halophytic plant wavy-leaved saltbush. Plants 10(10):2176. https://doi.org/10.3390/plants10102176
Lichtenthaler HK (1987) Chlorophyll and carotenoids pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382. https://doi.org/10.1016/0076-6879(87)48036-1
Lin L, Chen F, Wang J, Liao MA, Lv X, Wang Z, Li H, Deng Q, Xia H, Liang D, Tang Y, Wang X, Lai Y, Ren W (2018) Effects of living hyperaccumulator plants and their straws on the growth and cadmium accumulation of Cyphomand rabetacea seedlings. Ecotox Environ Safe 155:109–116. https://doi.org/10.1016/j.ecoenv.2018.02.072
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/s0021-9258(19)52451-6
Mahdieh M, Sangi MR, Bamdad F, Ghanem A (2018) Effect of seed and foliar application of nano-zinc oxide, zinc chelate, and zinc sulphate rates on yield and growth of pinto bean (Phaseolus vulgaris) cultivars. J Plant Nutr 41(18):2401–2412. https://doi.org/10.1080/01904167.2018.1510517
Maheshwari NO, Khan A, Chopade BA (2013) Rediscovering the medicinal properties of Datura sp.: a review. J Med Plant Res 7(39):2885–2897. https://doi.org/10.5897/JMPR11.1657
Manquián-Cerda K, Escudey M, Zúñiga G, Arancibia-Miranda N, Molina M, Cruces E (2016) Effect of cadmium on phenolic compounds, antioxidant enzyme activity and oxidative stress in blueberry (Vaccinium corymbosum L.) plantlets grown in vitro. Ecotox Environ Safe 133:316–326. https://doi.org/10.1016/j.ecoenv.2016.07.029
Moore S, Stein W (1948) Photometric ninhydrin method for use in the chromatography of amino acids. J Biol Chem 176(1):367–388. https://doi.org/10.1016/s0021-9258(18)51034-6
Mukherjee SP, Choudhuri MA (1983) Implications of water stress-induced changes in the level of endogenous ascorbic acid and hydrogen peroxide in Vigna seedlings. Physiol Plant 58:166–170. https://doi.org/10.1111/j.1399-3054.1983.tb04162.x
Nahar K, Hasanuzzaman M, Alam MM, Rahman A, Suzuki T, Fujita M (2016) Polyamine and nitric oxide crosstalk: antagonistic effects on cadmium toxicity in mung bean plants through upregulating the metal detoxification, antioxidant defense and methylglyoxal detoxification systems. Ecotox Environ Safe 126:245–255. https://doi.org/10.1016/j.ecoenv.2015.12.026
Pandey J, Verma RK, Singh S (2019) Suitability of aromatic plants for phytoremediation of heavy metal contaminated areas: a review Int. J Phytoremediat 21:405–418. https://doi.org/10.1080/15226514.2018.1540546
Perfus-Barbeoch L, Leonhardt N, Vavasseur A, Forestier C (2002) Heavy metal toxicity: cadmium permeates through calcium channels and disturbs the plant water status. Plant J 32:539–548. https://doi.org/10.1046/j.1365-313X.2002.01442.x
Perveen S, Shahbaz M, Iqbal M, Akram MS, Parveen A, Ali HMM (2016) Induction of cadmium stress tolerance in Triticum aestivum L. by alfalfa leaf extract. Appl Ecol. Environ Res 14(5):121–136. https://doi.org/10.15666/aeer/1405_121136
Saad-Allah KM, Elhaak MA (2017) Hyperaccumulation activity and metabolic responses of Solanum nigrum in two differentially polluted growth habitats. J Saudi Soc Agric Sci 16:227–235. https://doi.org/10.1016/j.jssas.2015.08.001
Sarwar N, Malhi SS, Zia MH, Naeem A, Bibi S, Farid G (2010) Role of mineral nutrition in minimizing cadmium accumulation by plants. J Sci Food Agric 90(6):925–937. https://doi.org/10.1002/jsfa.3916
Sayyari M, Ghanbari F, Fathhi S, Bavandpour F (2013) Chilling tolerance improving of water melon seedling by salicylic acid seed and foliar application. Not Sci Biol 51, 67:–73. https://doi.org/10.15835/nsb518293
Shalata A, Neumann P (2001) Exogenous ascorbic acid (vitamin C) increases resistance to Cd stress and reduces lipid peroxidation. J Exp Bot 52(364):2207–2211. https://doi.org/10.1093/jexbot/52.364.2207
Shanmugaraj BM, Malla A, Ramalingam S (2019) Cadmium stress and toxicity in plants: an overview. In Hasanuzzaman M, Prasad MNV, Nahar K (eds) Cadmium toxicity and tolerance in plants (pp. 1–17). Elsevier Inc. https://doi.org/10.1016/B978-0-12-814864-8.00001-2
Soleimani SH, Bernard F, Amini M et al (2020) Cadmium accumulation and alkaloid production of Narcissus tazetta plants grown under in vitro condition with cadmium stress. Plant Physiol Rep 25:51–57. https://doi.org/10.1007/s40502-019-00476-6
Song Y, Hudek L, Freestone D, Puhui J, Michalczyk AA, Senlin Z, Ackland ML (2014) Comparative analyses of cadmium and zinc uptake correlated with changes in natural resistance-associated macrophage protein (NRAMP) expression in Solanum nigrum L. and Brassica rapa. Environ Chem 11(6):653–660. https://doi.org/10.1071/EN14078
Sreevidya N, Mehrotra S (2003) Spectrophotometric method for estimation of alkaloids precipitable with Dragendorff’s reagent in plant materials. J AOAC Int 86:1124–1127. https://doi.org/10.1093/jaoac/86.6.1124
Sun RL, Qx Z, Cx J (2006) Cadmium accumulation in relation to organic acids in leaves of Solanum nigrum L. as a newly found cadmium hyperaccumulator. Plant Soil 285:125–134. https://doi.org/10.1007/s11104-006-0064-6
Szabados L, Savouré A (2010) Proline: a multifunctional amino acid. Trends Plant Sci 15:89–97. https://doi.org/10.1016/j.tplants.2009.11.009
Takahama U (1993) Regulation of peroxidase-dependent oxidation of phenolics by ascorbic acid: different effects of ascorbic acid on the oxidation of coniferyl alcohol by the apoplastic soluble and cell wall-bound peroxidases from epicotyls of Vigna angularis. Plant Cell Physiol 34(6):809–817. https://doi.org/10.1093/oxfordjournals.pcp.a078488
Tao Q, Liu Y, Li M, Li J, Luo J, Lux A, Kováč J et al (2020) Cd-induced difference in root characteristics along root apex contributes to variation in Cd uptake and accumulation between two contrasting ecotypes of Sedum alfredii. Chemosphere 243:125290. https://doi.org/10.1016/j.chemosphere.2019.125290
Tomar NS, Sharma M, Agarwal RM (2015) Phytochemical analysis of Jatropha curcas L. during different seasons and developmental stages and seedling growth of wheat (Triticum aestivum L.) as affected by extracts/leachates of Jatropha curcas L. Physiol Mol Biol Plants 21:83–92. https://doi.org/10.1007/s12298-014-0272-0
ur Rehman MZ, Rizwan M, Sohail MI, Ali S, Waris AA, Khalid H, Naeem A, Ahmad HR, Rauf A (2019) Opportunities and challenges in the remediation of metal-contaminated soils by using tobacco (Nicotiana tabacum L.): a critical review. Environ Sci Pollut Res 26(18):18053–18070. https://doi.org/10.1007/s11356-019-05391-9
Vernay P, Gauthier-Moussard C, Jean L, Bordas F, Faure O, Ledoigt G, Hitmi A (2008) Effect of chromium species on phytochemical and physiological parameters in Datura innoxia. Chemosphere 72(5):763–771. https://doi.org/10.1016/j.chemosphere.2008.03.018
Vernon LP, Selly GR (1966) The chlorophylls. Academic press, New York and London. https://doi.org/10.1016/S0021-9258(19)52433-4
Wang SQ, Wei SH, Ji DD, Bai JY (2015) Co-planting Cd contaminated field using hyperaccumulator Solanum nigrum L. through interplant with low accumulation welsh onion. Int J Phytoremediat 17:879–884. https://doi.org/10.1080/15226514.2014.981247
Wei S, Zhou Q, Wang X, Zhang K, Guo G, Ma LQ (2005) A newly-discovered Cd-hyperaccumulator Solanum nigrum L. Chin Sci Bull 50(1):33–38. https://doi.org/10.1360/982004-292
Weijie X, Wang P, Tang L, Zhang C, Wang C, Huang Y, Zhang X, Li Y, Zhao B, Liu Z (2021) Citric acid inhibits Cd uptake by improving the preferential transport of Mn and triggering the defense response of amino acids in grains. Ecotox Environ Safe 211:111921. https://doi.org/10.1016/j.ecoenv.2021.111921
Wiart C (2006) Medicinal plants classified in the family Solanaceae. In: Medicinal Plants of Asia and the Pacific. CRC Press, Washington, pp 269–277. https://doi.org/10.1201/9781420006803-36
Wu C, Dun Y, Zhang Z, Li M, Wu G (2020) Foliar application of selenium and zinc to alleviate wheat (Triticum aestivum L.) cadmium toxicity and uptake from cadmium-contaminated soil. Ecotox Environ Safe 190:110091. https://doi.org/10.1016/j.ecoenv.2019.110091
Xu J, Yin H, Li X (2009) Protective effects of proline against cadmium toxicity in micropropagated hyperaccumulator, Solanum nigrum L. Plant Cell Rep 28:325–333. https://doi.org/10.1007/s00299-008-0643-5
Yaghoubian Y, Siadat SA, Telavat MMR et al (2016) Quantify the response of purslane plant growth, photosynthesis pigments and photosystem II photochemistry to cadmium concentration gradients in the soil. Russ J Plant Physiol 63:77–84. https://doi.org/10.1134/S1021443716010180
Yang X, Qin J, Li J, Lai Z, Li H (2021) Upland rice intercropping with Solanum nigrum inoculated with arbuscular mycorrhizal fungi reduces grain Cd while promoting phytoremediation of Cd-contaminated soil. J Hazard Mater 406:124325. https://doi.org/10.1016/j.jhazmat.2020.124325
Yılmaz DD, Parlak KU (2011) Changes in proline accumulation and antioxidative enzyme activities in Groenlandia densa under cadmium stress. Ecol Indic 11(2):417–423. https://doi.org/10.1016/j.ecolind.2010.06.012
Zeng F, Mallhi ZI, Khan N, Rizwan M, Ali S, Ahmad A, Hussain A, Alsahli AA, Alyemeni MN (2021) Combined citric acid and glutathione augments lead (Pb) stress tolerance and phytoremediation of castorbean through antioxidant machinery and Pb uptake. Sustainability 13(7):4073. https://doi.org/10.3390/su13074073
Zhang ZL, Qu W (2004) Experimental guidance of plant physiology. High Education, Beijing
Zhang K, Wang G, Bao M et al (2019) Exogenous application of ascorbic acid mitigates cadmium toxicity and uptake in Maize (Zea mays L.). Environ Sci Pollut Res 26:19261–19271. https://doi.org/10.1007/s11356-019-05265-0
Zouari M, Ahmed CB, Zorrig W, Elloumi N, Rabhi M, Delmail D, Rouina BB, Labrousse P, Abdallah FB (2016) Exogenous proline mediates alleviation of cadmium stress by promoting photosynthetic activity, water status and antioxidative enzymes activities of young date palm (Phoenix dactylifera L.). Ecotoxicol Environ Saf 128:100–108. https://doi.org/10.1016/j.ecoenv.2016.02.015
Acknowledgements
Authors acknowledge Botany and Microbiology Department, Faculty of Science, Assiut University, for supporting this work. The authors are very grateful for Prof Dr Mamdouh Alsayed Eissa (Eissa MA) professor of Soils and Water, Department of Soils and Water, Faculty of Agriculture, Assiut University (mamdouhessa@gmail.com), for his helpful comments, critical reading, and revision of this research article. Also, the authors are very grateful for the insightful and helpful comments, constructive suggestions, and careful corrections made by the editor-in-chief and associate editor and the anonymous referees for further improvements of this manuscript.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abeed, A.H.A., Salama, F.M. Attenuating Effect of an Extract of Cd-Hyperaccumulator Solanum nigrum on the Growth and Physio-chemical Changes of Datura innoxia Under Cd Stress. J Soil Sci Plant Nutr 22, 4868–4882 (2022). https://doi.org/10.1007/s42729-022-00966-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-022-00966-x