Abstract
Heavy metal pollution has emerged as a severe threat to the environment as well as global food security. Exposure of plants to the heavy metals could cause perturbations in various physiological, biochemical, and metabolic processes including nitrogen (N) uptake and assimilation. Here, we discussed the effects of metal toxicity on N uptake, N forms, mechanism of metal toxicity, and nitrogen assimilation in plants. We provided a detailed description on the behavior of various enzymes including nitrate reductase (NR), nitrite reductase (NiR), glutamine synthetase (GS), glutamate synthase (GOGAT), and glutamate dehydrogenase (GDH) under metal toxicity. We highlighted the response of various nitrogenous compounds and their special role under metal toxicity. In addition, we discussed the effects of excess metals on N fixation in plants and provided the guidelines for further studies.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Heavy metal toxicity has become a serious environmental threat because of rapid industrialization, technological advancement, and disturbance of natural ecosystem due to a rapid increase in human population (Sarwar et al. 2010; Anjum et al. 2017; Ayangbenro and Babalola 2017). Heavy metals enter the agroecosystem by natural (such as erosion, volcanic eruptions, weathering of minerals, comets, etc.) and anthropogenic (such as coating, biosolids, alloy production, atmospheric depositions, pesticides, etc.) processes (Ayangbenro and Babalola 2017; Sarwar et al. 2017; Meena et al. 2017). Some types of soils inherit these heavy metals from parent material they are being originated having a high concentration of some of these metals which are hazardous to plants. Anthropogenic sources include sewage sludge, phosphate fertilizers, urban traffic, anthropogenic emissions from power stations, battery production, explosive manufacturing, cement industries, smelting, improper stacking of industrial solid waste, etc. (Wu et al. 2004; Sarwar et al. 2017; Ayangbenro and Babalola 2017; Marfo et al. 2015; Lojkova et al. 2015). These sources result in the buildup of higher concentrations of heavy metals in the agricultural soils.
Keeping in view their functions, heavy metals are categorized into two groups, essential and nonessential. Essential metals include zinc (Zn), manganese (Mn), copper (Cu), iron (Fe), and nickel (Ni) which have important roles in physiochemical and metabolic processes of living organism, such as in electron-transferring proteins and functioning as cofactors for some enzymes (Fageria et al. 2009; Chaffai and Koyama 2011; Fashola et al. 2016), while nonessential metals (like cadmium (Cd), mercury (Hg), and lead (Pb)) are not needed by plants for any biological functions (Dabonne et al. 2010). Plants exposed to high concentrations of heavy metals respond by altering the cellular metabolic process and gene expression (Hussain et al. 2004; Chaffai and Koyama 2011; Choppala et al. 2014). Heavy metals can cause the growth reduction in plants by decreasing photosynthetic activity and hindering the chlorophyll synthesis (Fig. 1; Gumpu et al. 2015). Heavy metals may cause hazardous effects in plants by disturbing essential groups of enzymes, blocking of essential functional groups and damaging the stability of important biomolecules, and disrupting the antioxidative defense mechanism as a result of higher generations of reactive oxygen species (ROS) (Chaffai and Koyama 2011; Choppala et al. 2014; Sarwar et al. 2015; Anjum et al. 2016; Ashraf et al. 2017).
Nitrogen (N) is among the most abundant essential macronutrients on earth (Cesco et al. 2010), and it is the critical limiting element for plant growth due to its unavailability (Graham and Vance 2000; Hussain et al. 2016). Plants can acquire N from different molecules (like nitrate, ammonium, urea, and amino acids) and utilized for different metabolic purposes, including the production of proteins, nucleic acids, as well as storage and signaling molecules (McAllister et al. 2012). Nitrate (NO3 −) and ammonium (NH4 +) are the major N sources for plants, and optimum conditions for activities of enzymes involved in the conversion of inorganic N into organic N are pivotal to plant biomass accumulation, growth, and final productivity. In plants, NO3 − being readily mobile can be stored in the vacuole and is the main source of N under well-aerated aerobic soil conditions. However, it must be reduced to NH4 + for the synthesis of proteins and other organic compounds in plants (Garnett et al. 2009). Nitrate reductase (NR) converts NO3 − into nitrite in the non-organelle portions of the cytoplasm. All the living plant cells have the ability to reduce NO3 − to nitrite, using the energy and reductant (NADH, NADPH) of photosynthesis and/or respiration in green tissues and of respiration in nongreen tissues and roots. Ammonium is the only reduced form of N available to plant for assimilation into N-carrying amino acids such as glutamate (Glu), glutamine (Gln), aspartate, and asparagine (Ireland and Lea 1999; Antunes et al. 2008; Dadhich and Meena 2014). For the biosynthesis of these N-containing amino acids, various enzymes such as Glu synthase, Gln synthetase (GS), glutamate dehydrogenase (GDH), alanine amino transferase, aspartate amino transferase, and asparagine synthetase are important (Garnett et al. 2009).
Heavy metals severely hamper the N metabolism by reducing the NO3 − uptake and altering the enzyme activity. In the past, the effect of Cd, Pb, Hg, Cu (copper), Ni (nickel), Mo (molybdenum), and V (vanadium) on N metabolism in different plant species has been reported; however, the effects of heavy metals on N assimilatory enzymes vary with the sensitivity level of enzymes and their localization in the cells/organs, mobility of metals, metal concentration in soil, and time of plant exposure to metal toxicity. Heavy metals can alter the activity of various N assimilatory enzymes by binding to the vital sulfhydryl (SH) groups (Prasad and Strzalka 2013). The metal-induced decrease in N assimilation process can also reveal the disruption of the general homeostasis of metabolic activities in plants. Heavy metals that induce the alterations in plant water status (Belimov et al. 2015); uptake, transfer, and supply of essential nutrients (Zia-ur-Rehman et al. 2015; Sarwar et al. 2015); photosynthesis (Emamverdian et al. 2018); and generation of ROS (Malar et al. 2016; Riaz et al. 2018b) ultimately affect the N metabolism in plant.
In the present article, we synthesized the effects of heavy metal on N uptake, N forms, mechanism of metal toxicity, and nitrogen assimilation. We provided a detailed description on the behavior of various enzymes including NR, nitrite reductase, Glu synthase, GS, and GDH under heavy metal stresses. We also highlighted the response of various nitrogenous compounds and their special role under metal toxicity. In addition, we discussed the effects of excess metals on N fixation in plants and provided future guidelines for further studies.
2 Heavy Metals in the Environment: Challenge for Plants
Heavy metals are pollutants in the environment, and their higher concentration creates the problems for ecological, evolutionary, nutritional, and environmental reasons. Although some metals (Zn, Cu, Fe, and Ni) are essential micronutrients and are required for normal growth and development of plants (Verkleij et al. 2009; Fageria et al. 2009; Imran et al. 2015), nevertheless, these metals are harmful to plants at higher levels and may lead to soil quality deterioration, plant growth inhibition, significant yield reduction, and poor quality of food (Ayangbenro and Babalola 2017), while some metals and metalloids (intermediate between metals and nonmetals) such as Cd, Cr, Pb, Se, As, and Hg have no known biological functions in living organisms (Seth 2012; Ayangbenro and Babalola 2017; Datta et al. 2017b).
Metal uptake from soil is carried out by cortical root tissues because of their resemblance with some essential micronutrients and transfer to the aerial plant parts via xylem vessels (Ali et al. 2013; Sarwar et al. 2017). Heavy metals disrupt several physiological, biochemical, and molecular processes in plant and could inhibit plant growth, disrupt the cellular functions, and ultimately cause cell death (Fig. 1; Xu et al. 2009; Fashola et al. 2016; Ashraf et al. 2017; Riaz et al. 2018a, b, c). Metal-induced growth inhibition might be explained on the basis of damage cell membrane, decreased synthesis of metabolites, photosynthetic rate, and chlorophyll content (Chibuike and Obiora 2014; Anjum et al. 2016; Ashraf et al. 2017). In some plants, metal toxicity may also cause drought stress because of significant decreases in stomatal conductivity, transpiration, and leaf relative water contents owing to less number of xylem vessels and poor cell enlargement (Saifullah et al. 2014). Metal-induced toxicity can cause cell membrane damage and destruction of biomolecules (like proteins and lipids) and cellular organelles in plants due to increase in the production of reactive oxygen species (ROS) under stress (Ekmekci et al. 2008; Ashraf et al. 2017). Moreover, nonessential heavy metals inhibit various biological mechanisms by altering the structure of biomolecules and important stress regulatory proteins (Sarwar et al. 2010).
3 Nitrogen Nutrition in Plants: No Alternative Lies
Even though N is among the most abundant elements on earth (about 80% of the earth’s atmosphere), it is the critical limiting element for the growth of plants (Greenwood and Earnshaw 2012; Wang et al. 2017; Li et al. 2018). In plants, N is required in huge amount, as it constitutes 1.5–2.0% total dry matter of plants (Frink et al. 1999; Xiong et al. 2006a). In most agricultural soils, NO3 − is the major form of the inorganic N available to the plants (Xiong et al. 2006a). Uptake of N followed by N reduction and N assimilation in plants is the only way through which plants may convert inorganic N into organic N form. The available N forms vary with organic (urea, amino acids, etc.) and inorganic (NH4 +, NO3 −, dinitrogen) compounds and the habitat of the plant. Nitrogen is a key constituent of proteins, amino acids, enzymes, alkaloids, vitamins, and some growth hormones. Increased NO3 − concentration in plants has a beneficial impact as it regulates the synthesis of proteins and amino acids, while the excess NH4 + content can be toxic as it promotes the formation of amides (Britto and Kronzucker 2002; Ram and Meena 2014). Geiger et al. (1999) studied the effects of different concentrations of ammonium nitrate and potassium nitrate on tobacco and reported that alanine, aspartate, Gln, arginine, and serine were increased considerably at higher N inputs. Terce´-Laforgue et al. (2004) also examined the influence of N fertilization (NO3 −, NH4 +, and low nitrogen) on the amino acid profile in tobacco leaves and concluded that Glu and total amino acids were increased from the old to young leaves under low N supply.
4 Interplay of Nitrogen Uptake and Heavy Metal Stress
Nitrogen (N) is mainly taken up by the plants in the organic form (NO3 − and NH4 +) from the soil solution. The plant’s preference to uptake either N form depends on various factors, including the plant species, the concentrations of NO3 − and NH4 + in soil, and the soil pH (Perkins et al. 2011; Miller 2016; Aziz et al. 2018). The soil N pool is driven mainly through mineralization, nitrification, and ammonification, which are believed to be influenced by soil microbiota. However, among the various abiotic stresses affecting the overall N cycle in the soil, heavy metal stress is considered to influence the nitrification and ammonification processes through modulating the activities of microbiota (Kapoor et al. 2015). Soil moisture, aeration, temperature, and the pH are the key drivers controlling these nitrifying agents and the respective processes, and therefore the applied N source in the soil fluctuates widely the pH of soil which in turn regulates the net proton extrusion rates. Generally, the NO3 − fertilizer source is preferred over the NH4 + due to its readily available form for direct adsorption by plant roots (no need to undergo any further conservation) and due to its less volatile nature. Therefore, the soil pH is crucial in determining the availability of heavy metals for plant uptake (Bravo et al. 2017; Datta et al. 2017a). NO3 − source decreases the soil acidification, and it prevents the accumulation and uptake of heavy metals by plants, while this case is opposite for NH4 + nutrition. Here we discussed the synergistic effects of both the N source and the heavy metal stress influencing the plant uptake mechanism.
4.1 Nitrate Uptake
Soil type (structure and texture) is the key determinant of the N fertilizer source to be applied, which is readily available for plant uptake in the rhizosphere (Li et al. 2018; Aziz et al. 2018). Nitrate is believed to be the best source in well-aerated agricultural soils as compared to NH4 +. Nitrification is the process in which ammonia is converted to NO3 − through biological oxidation, and these conversion steps are mediated by aerobic nitrifying bacteria requiring sufficient soil aeration and oxygen for the oxidation process. Therefore, the oxidation of NH4 + is restricted in waterlogged soils (Ma et al. 2016). Established reports have indicated that the NO3 − absorption process by the plant is the active process, as the NO3 − influx to plant roots is proton-coupled depending upon the H+ pumping (Miller and Smith 1996; Zhou et al. 2016). The presence of the excessive heavy metals in soil affects the NO3 − uptake by root cells (Fig. 1). Among these heavy metals, Cd is considered the most effective NO3 − inhibitor which is reported to repress the NO3 − absorption process very sharply even at very low Cd level (Boussama et al. 1999a; Gouia et al. 2000b; Huang and Xiong 2009). It was observed that excluding Cd from the uptake solution lasted its impact of NO3 − inhibition till 96 h (Gouia et al. 2000b). However, this inhibitory effect was observed for several plant species. One vital reason for the reduced NO3 − uptake is the damaged plasma membrane in roots due to excessive membrane leakage and the MDA contents (Huang and Xiong 2009). Such reduced NO3 − contents were observed more for roots versus shoot. Wang et al. (2008) observed better shoot growth under high Cd stress, but the NO3 − concentrations were much decreased for Solanum nigrum. Similarly, besides the Cd stress, nitrate uptake is also limited by Pb excess in plant uptake solution (Singh et al. 2017). Aluminum (Al) is the third most abundant metal known to cause the inhibitory effects upon NO3 − uptake by plant roots (Schmitt et al. 2016; Riaz et al. 2018a, b). But the extent of Al toxicity depends upon the ionic form, its concentration, and the plant exposure to stress. Acceleratory effect of short-term Al exposure to plants (barley, cucumber) had been observed with increased nitrate uptake (Nichol et al. 1993; Jerzykiewicz 2001). However, higher external Al inputs had decreased dramatically the net NO3 − uptake in Vigna unguiculata upon prolonged plant exposure to imposed stress (Cruz et al. 2014).
4.2 Ammonium Uptake
Ammonium ion is an intermediary during the N metabolism in plants, which is produced several times during NO3 − assimilation, deamination of amino acids, and photorespiration (Andrews et al. 2013). NH4 + may accumulate in the soil, when this conversion process of N is limited or completely stopped due to any of the soil conditions, viz., low soil pH, lack of soil oxygen due to waterlogging, lack of organic matter for carbon stick, dry soils, and low soil temperature, affecting microbiota activity (Mengel and Kirkby 1987; Cheng et al. 2013; Ashoka et al. 2017). Therefore, all these features may affect the soil microclimate to enhance or depress the soil microorganism activities determining the NH4 + concentration in soil varying from micromolar to hundreds of millimolar. The optimum level nitrification process is attained at 20–40 °C, while this is 50–70 °C for optimum ammonification (Myers 1975; Yao et al. 2013). Hence, in tropical region soils, NH4 + may accumulate in the soil even under neutral pH conditions (Mengel and Kirkby 1987). As the NH4 + transport in plants is considered to be passive as well as active uptake depending upon the NH4 + availability in the soil solution (Wang et al. 1993; Cheng et al. 2018), studies on the restricted NH4 + uptake due to heavy metals stress suggested significantly decreased uptake of NH4 + by plant roots in various crops, mainly due to the perturbations in active uptake process of ATPase (Weber et al. 1991). Relatively decreased or suppressive NH4 + uptake was seen in Silene vulgaris at the presence of Cu (Weber et al. 1991). Toxicity of other heavy metals (Cd2+, Pb2+, and Fe2+) was also observed with the reduced or diminished uptake of NH4 + in cucumber seedlings (Burzynski and Buczek 1998). Several other stress agents including Pb, Cd, Ni, Zn, and Mn are also reported to influence the uptake of NH4 + in different plant species. However, Cu-dependent alterations are known to affect the NH4 + uptake to the greatest extent as compared to other metal ions (Kubik-Dobosz et al. 2001).
4.3 Mechanisms of Metal Action on the N Uptake
Heavy metal stress is considered to induce several alterations, either direct or indirect, during the active as well as passive uptake of both inorganic forms of N. Physiological studies suggest that metal toxicity affects the NO3 − and NH4 + uptake through disrupting constitutive and the inductive components of NO3 − transportation system (Burzynski and Buczek 1994; Shruti and Dubey 2006). This effect was observed for metal toxicity with Cd, Pb, Cu, and Ni in cucumber seedlings which inhibited the NO3 − transporters with higher affinities (Burzynski and Buczek 1994; Ashraf et al. 2015). The similar effect was also observed for inhibition of high-affinity NH4 + transporters (Burzynski and Buczek 1998). The explanations for restricted N absorption could be the direct interaction of metal ions and the NH4 + and NO3 − transporter proteins of both low and high affinities (Shruti and Dubey 2006). Besides this direct interaction of metals and proteins, heavy metal alterations in these transporter regions are due to the downregulation of NRT and AMTl genes (Quesada et al. 1997, Krapp et al. 1998; Singh et al. 2008). This diminishing effect is attributed due to the lowered intercellular nitrates and the enhanced amino acids. Plants treated with external Cd inputs showed fluctuations in the NO3 − as well as NH4 + and amino acid tissue content (Hernandez et al. 1996, 1997; Gouia et al. 2000b). Other effects of metal stress include the Cu and Fe bonding with phosphate residues of nucleic acids thus damaging DNA (Lloyd and Phillips 1999). Furthermore, heavy metals including Cu, Cd, and Ni are reported to accelerate the free radical generation, thus oxidizing several vital organic molecules (Shaw et al. 2004; Michalak 2006).
Among the indirect effects of heavy metal stress on ion uptake in plants, some key metal ions, viz., Cu, Pb, Hg, Cd, Ni, and Zn, are believed to interact with membrane components thus affecting the net ion transport system in plant body (Devi and Prasad 1999; Michalak 2006; Riaz et al. 2018c). This membrane interaction includes altered membrane lipids, total lipid amount, and their composition and saturation; in this process, the main damage is associated with the lipid peroxidation (Demidchik et al. 1997; Hernandez et al. 1997; Singh et al. 2008; Meena et al. 2018). Besides this, heavy metals especially Cu, Hg, Cd, Zn, and Al also induce the potassium leakage leading to increased cell membrane permeability (Demidchik et al. 1997; Hall 2002; Riaz et al. 2018c). Therefore, it is evident that the indirect influence of heavy metal stress during the NO3 − and NH4 + uptake is the alteration in membrane permeability. In addition, metal ions also interact with the plasma membrane proton pump by affecting the activity of H+ ATPase activity (Rengel et al. 2016).
5 Mineral Nitrogen Assimilation and Heavy Metals
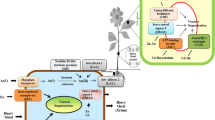
Nitrogen assimilation is an important plant metabolic process, which not only controls plant growth and development but also plays an important role in plant survival under stress conditions. For plants, NH4 + and NO3 − are major nitrogen sources and are required during different metabolic processes (Burger and Jackson 2003). Nitrate is converted into NH4 + via two-step process; during the first step, NO3 − is converted into nitrite with the action of nitrate reductase, and, in the second step, nitrite is converted into NH4 + with the action of nitrite reductase. Nitrate reductase is located in the cytoplasm, while nitrite reductase is located in chloroplast and uses energy and some reductants such as NADH or NADPH from photosynthesis or respiration process to carry out such NO3 − to NH4 + conversion reaction (Pérez-Tienda et al. 2014). Moreover, the first step of this reaction occurs in the cytosol, while the second step takes place in the plastid. Following NH4 + production, it has to be incorporated in carbon skeleton, and this process takes place primarily via GOGAT cycle. Moreover, there are two isoforms of GOGAT and GS, and their localization has been found in a tissue-specific manner, e.g., in roots, NADH-GOGAT and GS1 are primarily involved in nitrogen assimilation, while in leaves, reduced ferredoxin GOGAT and GS2 are predominantly involved in nitrogen assimilation (Mevel and Prieur 2000). Heavy metal toxicity significantly reduces nitrogen assimilation process (Fig. 2). However, the level of reduction depends on the localization and sensitivity of enzymes to heavy metal toxicity. Moreover, duration, concentration, and mobility of heavy metal ions in growth medium further aggravate alterations in nitrogen assimilation process. In this section, heavy metal-induced alterations in mineral nitrogen assimilation and associated enzymes have been discussed.
5.1 Nitrate Reductase Activity
Nitrate reductase (NR) enzyme is a very important enzyme and controls the initiation of NO3 − assimilation process in plants (Silveira et al. 2001). NR enzyme activity is severely affected by numerous environmental factors including hyperaccumulation of heavy metal ions in soil or in plant tissues (Silveira et al. 2001; Sharma and Dubey 2005). It has been shown that heavy metal stress reduces NR activity by altering numerous associated physiological processes. Sharma and Dubey (2005) showed that inhibition of NR activity under Al toxicity was due to the direct interaction of Al with functional SH groups present in the active sites of NR. Contrarily, Huang and Xiong (2009) noted that the reduction in NR activity under Cd stress was due to alteration in sugar metabolism and declined photosynthetic activity. According to Barroso et al. (2006), reduction in photosynthesis can induce indirect effects of Cd toxicity on NR activity. However, Vajpayee et al. (2000) reported that under Cr stress, NR activity was decreased because of reduced chlorophyll biosynthesis, which led to lower photosynthesis and poor supply of photosynthates. Similarly, in pea plants, Cd toxicity not only inhibited NO3 − uptake and transportation but also detrimentally affected the activity of NR and increased endogenous NH4 + level through deamination of free amino acids and other N forms (Hernandez et al. 1997; Datta et al. 2017c). An in vitro examination of Cd toxicity on NR activity showed that decline in NR activity was associated with the reduction of 80% in the production of NR protein and alteration in the molybdenum cofactor-binding domain of NR (Gouia et al. 2000a). In tomato, Chaffei et al. (2004) also reported similar results that Cd toxicity significantly reduced NR activity. Wang et al. (2008) found that NR activity could also be influenced by Cd-induced reduction in NO3 − concentration.
Similarly, the detrimental effects of other heavy metals on NR activity have also been reported elsewhere. Xiong et al. (2006b) showed that NR activity was reduced by 106% due to Pb stress, and this was mainly due to Pb-induced reduction in the shoot NO3 − concentration (71–80%) and free amino acid (81–82%). Similarly, Cu stress-induced decline in NR activity was suggested as a direct effect of Cu on NR by attaching Cu2+ to SH groups, with subsequent inactivation of NR enzyme (Xiong et al. 2006a). Similarly, Kevrešan et al. (2001) studied the effects of Ni, Cd, Pb, and Mo (applied at various concentrations) on N metabolism in pea plants. They found that all the concentrations of Ni, Cd, and Pb caused a significant decrease in the activity of NR, and the highest decrease in NR activity was observed in the presence of Cd followed by Pb and Ni. However, the presence of Mo results in increased activity of the NR. The NR activity was less dependent on NO3 − contents present in the cell and to a greater extent on the intensity of uptake and transport of NO3 − to the sites of their reduction (Carillo et al. 2005; Varma et al. 2017). Moreover, a high concentration of heavy metals in the growth medium causes dehydration in plant tissues thus reduces NR synthesis process (Chaffei et al. 2004).
Heavy metal-induced oxidative stress (Khan et al. 2018; Shahzad et al. 2018) and enhanced lipid peroxidation may also influence the NR activity in plants. In a study, Jha and Dubey (2004) showed that As decreased NR activity by decreasing the enzyme affinity toward their substrate, thus resulting in considerable reduction in NR activity and N assimilation rate in rice. Likewise, Gajewska and Skłodowska (2009) substantiated that under Ni stress, NO3 − contents were decreased with the subsequent reduction of 40% in NR activity in wheat seedlings. Rai et al. (2004) investigated the toxic effects of Cr on Ocimum tenuiflorum and found that excess Cr in the growing media severely reduced the NR activity through impaired substrate utilization. Hg stress has been reported as the most toxic stress in reducing NR activity as Hg can inactivate NR enzyme by binding to a thiol group (Sharma and Subhadra 2010; Meena and Meena 2017). Similar results have been found in maize under Hg stress that Hg not only competes with thiol group but also disturbs the production of cysteine (Pandey and Srivastava 1993). Thus it can be inferred from above results that heavy metal stress reduces NR activity by altering cysteine production, by increasing ROS production, by disturbing the balance between the substrate for NR reaction, and by affecting sugar metabolism and photosynthesis.
5.2 Nitrite Reductase Activity
Nitrite enters into plastids or chloroplast and reduced to NH4 + by the action of NiR—a nuclear-encoded enzyme (Shah and Dubey 2003). In general, NiR is more resistant to stress conditions than NR. As this enzyme is localized in plastids, therefore, metals have less access to NiR, compared with cytoplasmic NR. The NiR has not been reported to limit the N assimilation in plants. Therefore, the studies regarding the effect of heavy metals on NiR activity are rare. However, studies showed that NiR decreases under heavy metals stress. Ghosh et al. (2013) showed that exposure of arsenate to wheat seedling significantly reduced NiR activity, and this was associated with a reduction in NR activity and total nitrite contents, which is the first primary reason (Llorens et al. 2000). Heavy metals can also influence the NO3 − uptake and NO3 − homeostasis in the plant cell. Therefore, NO3 − also induces the expression of NiR genes along with NR genes (Stitt 1999). Therefore, the regulation of NiR genes’ expression by NO3 − could be the main reason of NiR inhibition by heavy metals. In a study, Dinakar et al. (2009) showed that Cd stress reduced NiR activity up to 75%, and this was due to a significant reduction in NR activity.
5.3 Glutamine Synthetase and Glutamate Synthase Activities
In higher plants, NH4 + is assimilated into nontoxic glutamine and glutamate, and this reaction is carried out by glutamine synthetase (GS) and glutamate synthase (GOGAT) (Lam et al. 1996). The effects of metal toxicity on GS and GOGAT are often regarded as a mean of the effect of metal on all isoenzymes of GS or GOGAT localized either in the root or shoot. The decrease of one form of the enzyme may trigger the other. Therefore, the variations recorded in the activities of enzymes after metal exposure are different and sometimes difficult to infer. In the past, reductions in activities of GS and GOGAT were found after the exposure of plants to Cu (Yu et al. 2005), Cd (Chaffei et al. 2004), Al and Se (Ruiz et al. 2007), and As (Jha and Dubey 2004) toxicities by different researchers.
Heavy metals are known to disturb the NO3 − absorption by the plants. Intracellular NO3 − level affects the gene expression of GS and GOGAT along with NR and NiR (Oaks 1994; Stitt 1999). Possibly, this dependence of GS and GOGAT genes on NO3 − is the cause of metal-induced inhibitions in the activities of these enzymes along with the direct metal action on the SH groups of enzyme proteins. Huang and Xiong (2009) reported that NH4 + content in shoot and root of rice seedlings was significantly increased, while GS activity was decreased after Cd stress. Chien and Kao (2000) also reported that the NH4 + accumulation in shoot and root was ascribed to the decline in GS and GOGAT activities in rice. Likewise, Cd reduced the activities of GS and GOGAT and consequently increased the level of NH4 + in maize seedlings (Boussama et al. 1999a). Chugh et al. (1992) studied the effect of Cd on GS and GOGAT activities in pea seedlings and reported that GS activity in leaves was practically unchanged; however, it was markedly reduced in the roots. Both GOGAT and GS were suppressed by the Cd toxicity in leaves of barley (Boussama et al. 1999b) and bean seedlings (Gouia et al. 2000a). In cucumber, Cd and Pb treatment for 24 or 48 h decreased the GS activity in roots but did not alter GS activity in the cotyledons (Burzynski 1990). He further reported considerable increases in the activity of GOGAT-NADH activity in the cotyledons under Pb as well as Cd toxicity (Burzynski 1990). Application of 100 μM Cu/Cd/Fe and 500 μM Pb to cucumber seedlings for 1 h decreased the activity of GS in the root (Burzynski and Buczek 1998). In the in vitro experiments, metals only reduced the enzyme activity at high concentrations, Cu at 1000 μM, Cd at 500 μM, and Fe at 1000 μM, while Pb was even detrimental at 10 μM (Burzynski and Buczek 1997), and the minor effect of Cu, Cd, and Fe on GS activity indicated the indirect action on enzyme activity. Correspondingly, high concentrations (500–1000 μM) of Co, Ni, Cd, Zn, and Cu repressed both GS forms in triticale seedlings (Bielawski 1994). A study on Chlamydomonas reinhardtii by Domínguez et al. (2003) demonstrated that the 24 h exposure to 150 μM Cd did not inhibit the NO3 − uptake but reduced the GS activity by 45%, which enhanced the intracellular accumulation of NH4 +.
Kevrešan et al. (2001) stated that the exposure of Cd, Ni, Mo, and Pb to young pea plants significantly decreased the GS activity at all the concentrations used, especially at the highest level, and the metal-induced reductions were in the order of Cd > Pb > Ni > Mo. The presence of Cd in the medium strongly inhibited (about 78%) the GS activity in young pea plants than the other metals (Kevrešan et al. 2001). Likewise, Popović et al. (1996) reported a complete inhibition of GS enzyme in the young sugar beet plants under Cd toxicity. Gajewska and Skłodowska (2009) found that Ni application did not significantly change the GS activity in wheat shoots. However, a significant decline in GS activity was reported in the leaves of Ni-stressed sugar beet (Kevrešan et al. 1998). Miflin et al. (1980) stated that the presence of Cu, Hg, Cd, Zn, Ni, and Fe resulted in a pronounced inhibition of GS in rice. However, Hg was more toxic for GS even at 10 μM. Orzechowski and Bielawski (1997) studied the effects of Cd, Zn, and Pb toxicity on NH4 + assimilation in Triticale crop and reported that the metal sensitivity of GS varied in root and shoot. Cd and Zn toxicity reduced the GS activity in shoots but increased in root compared with control. Gajewska and Skłodowska (2009) concluded that a decrease in the activities of both GOGAT and GS in metal-stressed plants was because of alterations in the oxidative metabolism of plants (Balestrasse et al. 2006). As metal toxicity leads to the overproduction of ROS in a plant cell, thus decrease in both GS and GOGAT activities might be related, at least partly, to oxidative destruction of these enzymes.
5.4 Glutamate Dehydrogenase Activity
In higher plants, GS/GOGAT is the main pathway of NH4 + assimilation under normal conditions; nevertheless, with the increase in endogenous NH4 + concentration, an alternate pathway, controlled by glutamate dehydrogenase (GDH), contributes in decreasing this internal NH4 + concentration. The exact role of alternate GDH pathway is still poorly understood in plants. The up- or downregulation of enzyme often varies depending on the species, cultivars, tested tissue, and intensity and duration of metal exposure in plants (Miflin and Habash 2002). Masclaux-Daubresse et al. (2002) stated that Glu is synthesized by the combined action of GOGAT and GS in both young and old leaves of tobacco, while GDH is responsible for deamination of Glu. GDH is mainly located in mitochondria, occasionally in the cytoplasm, within the phloem companion cells of the shoot (Terce´-Laforgue et al. 2004; Fontaine et al. 2006). Till date, controversy exists as to the role of the enzyme GDH in higher plants. It has been suggested that GDH has a role in NH4 + reassimilation under stress conditions (Gouia et al. 2000a, 2003). Skopelitis et al. (2006) reported that the formation of ROS under stress induced the synthesis of the a-subunit of GDH, when GS was inhibited. For instance, it has been reported that the GDH was induced under Cd stress (Miflin and Habash 2002; Astolfi et al. 2004; Yadav et al. 2017). Chaffei et al. (2003) also stated that GDH has a major role in cellular NH4 + detoxification. They noted that deaminating activity of GDH was inhibited by Cd (due to a direct action of Cd on the enzymatic protein or to a retroinhibitory effect of ammonia), while the reducing aminating activity of GDH was enhanced. Lee et al. (1976) found the decreased activity of GDH and low NO3 – contents in soybean plants under Pb toxicity, whereas, malate dehydrogenase activity remained unaffected by Pb. Mittal and Sawhney (1990) reported that Pb retarded the utilization of N reserves from cotyledons and decreased the activity of GDH in germinating pea seeds compared with control, which disturbed the respiratory activity because of restricted generation of organic acids from amino acids. Gouia et al. (2000a) reported that under Cd stress, GDH fulfills the Glu pool required for synthesis of Cd-binding peptides and detoxify the recycling of the high NH4 + originating from a decline in the GS/GOGAT activities. Higher GDH-NADH activity in root and shoot of triticale under Cd, Zn, and Pb toxicity was also recorded by Orzechowski et al. (1997). A short-term (1 h) exposure of cucumber plants to Cd, Cu, Pb, and Fe inhibited the NADH-GDH activity in root because NH4 + in tissues was in excess (Burzynski and Buczek 1997, 1998). However, Cd or Pb toxicity significantly increased the root NADH-GDH activity after 24 and 48 h. Gouia et al. (2003) reported that Cd stress triggered the activity of GDH in bean seedling concomitant with the dramatic buildup of an NH4 + pool. The Cd-induced enhancement in GDH activity has also been reported in maize (Boussama et al. 1999a) and pea (Chugh et al. 1992), which were attributed to de novo synthesis and/or activation of specific isoenzymes that eliminate excess NH4 + (Syntichaki et al. 1996; Loulakakis and Loulakakis-Roubelakis 1996). Boussama et al. (1999a) have demonstrated that in Cd-stressed maize plants, Cd treatment triggered the NADH-GDH activity. Even though the role of GDH in higher plants is controversial, GDH isoenzymes are known to remove, in part, the excess of NH4 + under Cd toxicity.
Exposure of Ni or Cd enhanced the activity of GDH in the shoot of Silene italica (Mattioni et al. 1997). Van Assche et al. (1988) reported that toxicity of Zn or Cd beyond threshold level increased the GDH activity in Phaseolus vulgaris leaves, while Domínguez et al. (2003) reported that aminating GDH activity was increased by 75% in Chlamydomonas reinhardtii after 24 h exposure to 150 μM Cd and suggested that aminating GDH activity could support the biosynthesis rate of the Glu in Cd-treated plants.
While studying the effects of Cu on N metabolism in grapevines, Llorens et al. (2000) observed dramatic changes especially in the root system and found that NADH-GDH was active to keep the root function. Gajewska and Skłodowskahe (2009) found that activity of NADH- and NAD-GDH in wheat shoots increased after 7 days of Ni exposure to wheat seedlings. They further stated that under unfavorable environmental conditions like Ni toxicity, GDH could play an important role in detoxification of NH4 + released in response to stress as well as in the replenishment of Glu pool (Jha and Dubey 2004). Jha and Dubey (2004) reported that the activities of aminating and deaminating GDH increased at moderately toxic level (25 μM) of As2O3, whereas a higher As level of 50 μM was inhibitory to the enzymes. They suggested that under As toxicity, GDHs play an important role by helping in NH4 + assimilation and sustaining general C metabolism.
6 Accumulation of Nitrogenous Compounds in Metal-Stressed Plants
As an adaptive mechanism, plant accumulates numerous nitrogenous compounds such as proline or polyamines to protect the plant from detrimental effects of heavy metals (Alcázar et al. 2006; Anjum et al. 2017). These compounds exhibit a specific role in numerous metabolic processes and also vary in response to plant tissue, heavy metal concentration, and plant species. There are different soluble nitrogenous compounds which can significantly accumulate in the plant under heavy metal stress to alleviate heavy metal stress-induced adversities in plants. These soluble compounds include amino acids, polyamines, proline, or glycine betaine. Generally, these compounds regulate osmotic potential in plant cells and thus improve heavy metal stress tolerance (Wang et al. 2008). The following sections provide a brief discussion on the behavior of some important nitrogenous compounds in response to heavy metals.
6.1 Proline and Glycine Betaine Accumulation Under Heavy Metal Stress
Proline is a very important amino acid with multiple roles in different metabolic processes. Generally, under heavy metal stress, proline accumulation increases, thus improving stress tolerance in plants (Szabados and Savoure 2010). Higher accumulation of proline and its role in stress alleviation can be partially explained by its unique chemical properties as compared with other amino acids: proline being a water-soluble amino acid exists as in a zwitterionic state having both positive charge and weak negative charge in N groups and carboxylic acid, respectively (Lehmann et al. 2010). Proline also plays a key role as an osmolyte, electron sink, radical scavenger, component of the cell wall, and stabilizer of macromolecules (Matysik et al. 2002; Yancey 2005; Kumar et al. 2017). Significant accumulation of proline was noted under Cd stress in brassica (Singh and Tewari 2003). Similarly, Dinakar et al. (2008) showed that under Cd stress, proline accumulation increased by 160%, and this was accompanied with high glutathione reductase and NR activity. This can be supported with the findings of Sharma et al. (1998) who reported that proline protects NR and glucose-6-phosphate dehydrogenase against the Zn and Cd toxicity, and such protection occurs due to a reduction of the free metal ion activity owing to the formation of proline-metal complexes. Under Pb stress, Yang et al. (2011) showed that Pb tolerance in two wheat cultivars was associated with higher proline contents. Likewise, detrimental effects of Ni toxicity were mediated by higher proline accumulation in wheat (Parlak 2016). Under Cd toxicity, upregulation of proline biosynthesis resulted in higher Cd tolerance associated with higher GSH/GSSG ratio and higher glutathione S-transferase accumulation.
Glycine betaine (GB, the trimethylglycine) is a common osmolyte and N-based compound that accrues in plants under drought and salinity stresses (Sharma and Dietz 2006). It not only protects membranes and proteins but also reduces stress-induced alteration in enzymes (Ashraf and Foolad 2007). Moreover, GB helps in ROS scavenging, thus regulating redox regulation under stress conditions (Chen et al. 2006). Betaine has been regarded as the most promising osmolyte in protecting plant metabolism from Cd toxicity (Islam et al. 2010). Bharwana et al. (2014) showed that exogenous GB application protected cotton seedling from Pb-induced oxidative damage by increasing antioxidant defense system.
6.2 Polyamine Accumulation and Heavy Metal Tolerance
Besides the protective role of proline and glycine betaine, some other nitrogenous compounds such as amino acids and polyamines also play an important role in heavy metal stress alleviation and tolerance in plants. Polyamines such as spermine or spermidine or putrescine reduce heavy metal-induced adversities in the plant (especially N metabolism) by playing a multifaceted role in different physiological and biochemical processes (Kakkar and Sawhney 2002). In a study, it was found that spermine application increased Cd stress tolerance in mung bean by increasing glutathione S-transferase, glutathione reductase, and glutathione contents. Generally, levels of polyamines increase in different plant tissues under heavy metals stress; however, it has also been seen that different polyamines showed different responses under different heavy meal stresses in different plant species. For instance, Groppa et al. (2007) also showed that spermidine content was not affected by Cd and Cu, while spermine was significantly reduced. They also noted that putrescene was increased under Cd and Cu stress in wheat. Similarly, it has been seen that spermidine increased Al toxicity by increasing photosystem II D1 protein gene and glutathione reductase activity, thus improving photosynthesis and N metabolism (Sen et al. 2014). In another study, Mandal et al. (2013) documented that putrescene can reduce Al toxicity by downregulating H+-ATPase activity and by increasing flavonoid production.
Under metal stress, the specific role of polyamines in plants is poorly known. However, these may effectively protect and stabilize the membranes against the toxicity of metal ions especially the redox active metals. Shi and Chan (2014) reported that polyamines enhance the plant stress tolerance by improving antioxidant production and osmolyte production (especially amino acids). In a study, it was found that spermidine improves Al stress tolerance by increasing GSH pool and Gly II activity. Besides activating defense system in plants, polyamines also increase NR activity. Rosales et al. (2012) showed that putrescence could increase NR activity by 63%, while spermine or spermidine can increase NR activity by 114%. Thus these results are clearly highlighting the protective role of polyamines in improving N metabolism under heavy metal stress conditions.
7 Nitrogen Fixation and Heavy Metal Toxicity
Nitrogen fixation is a very important process in plants (especially legumes), which helps in fixing molecular nitrogen into organic nitrogen via symbiosis process. Biological nitrogen fixation accounts for about 70% in fixing elemental N and is very crucial for plant growth (Matiru and Dakora 2004; Meena and Yadav 2015). Heavy metals considerably reduce the nitrogen fixation process. However some metalloids are beneficial in low concentration and promote growth and activity of different Rhizobium and Bradyrhizobium strains. The presence of high concentrations of heavy metal ions in soil restricts nitrogen fixation by reducing infection process, growth of Rhizobium, nodule development, or host plant growth (Broos et al. 2005); however it was found that the extent of reduction depends on heavy metal and plant species. For instance, Athar and Ahmad (2002b) found that Cd is more toxic to Bradyrhizobium and Rhizobium as compared with Ni, Zn, or Co. Similarly, Kalyanaraman and Sivagurunathan (1993) also noted that Cd is more toxic than Zn and reduces nitrogen fixation process significantly. In another study, it was found that a number of free-living Azotobacter chroococcum cells were severely influenced by Cd as compared with Zn, Ni, Cr, and Pb (Athar and Ahmad 2002a). Moreover, Paudyal et al. (2007) tested the growth and N2 fixation process by two strains of Rhizobium in response to Al, Fe, and Mo toxicity and found that Al was the most lethal heavy metal for them, while Fe promoted the growth of both strains. Some other studies also reported toxic effects on different heavy metals such as Th, Ni, As, Cd, and Cu on the growth and morphology of numerous symbiotic N2-fixing organisms including R. leguminosarum, Sinorhizobium, and Mesorhizobium ciceri (Santamarı’a et al. 2003; Wani 2008; Arora et al. 2010; Stan et al. 2011; Bianucci et al. 2011). All these studies suggested that differential toxicity levels of different heavy metals on nitrogen-fixing organisms were due to experimental conditions, heavy metal ion concentration, plant species, and solubility of heavy metals ions in the growth medium.
Heavy metal stress significantly alters nitrogen fixation in both symbiotic and nonsymbiotic system (Giller et al. 1998; Selosse et al. 2004; Molaei et al. 2017a, b). It has been reported in Wani et al. (2007) that Cd stress decreased symbiosis process and root N content by 34%, compared with the control. They also reported that such decline in nitrogen fixation and nitrogen contents in root was associated with a decline in a number of nodules per plant and dry mass of nodules. Moreover, Balestrasse et al. (2001) reported that Cd toxicity causes oxidative damage to nodule by increasing ROS production in nodules, thus reducing nitrogen fixation in nodules. Broos et al. (2005) showed that sludge treatment in white clover reduced nitrogen fixation up to 50%, and this was associated with dysfunctional nodules. In another study, Balestrasse et al. (2004) showed that Cd stress considerably reduces leghemoglobin levels, nitrogenase activity, and protease activity in nodules, and most importantly Cd also reduces the number of bacteroides per symbiosome and total effective area for N2 fixation in soybean. In white lupin, Cd stress decreases N-amino compounds, malate, succinate, and soluble protein in nodule (Carpena et al. 2003). Arsenic (As) is another detrimental heavy metal, which reduces nitrogen fixation in nodules and activity of different nitrogen-fixing microorganism significantly. Pajuelo et al. (2008) studied the effects of As toxicity on the symbiotic interaction between Sinorhizobium and Medicago sativa and reported that As toxicity reduced the total number of nodules by 75% and a number of rhizobial infections by 90%. These reductions were attributed to damage of root hair and shorter infective root zone. Exposure of As toxicity in black gram reduced the nitrogenase activity in nodule by twofolds and restricted the rate of nitrogen fixation (Mandal et al. 2011; Verma et al. 2015). In soybean, though B. japonicum E109 stain showed tolerance to arsenic, however, there was still reduction in nodulation, and this was associated with reduced motility (swarming and swimming) of the microorganism in the presence of arsenic (Talano et al. 2013). In pea, chromium application decreased the ability of pea plants to fix atmospheric nitrogen symbiotically, and this was associated with a decline in total nitrogenase activity and nodule formation (Bishnoi et al. 1993).
8 Conclusions and Perspectives
Heavy metal toxicity has become a serious environmental threat worldwide. Heavy metals severely hamper the N metabolism by reducing the NO3 − uptake and altering the enzyme activity; however, the effects of heavy metals on N assimilatory enzymes vary with the sensitivity level of enzymes and their localization in the cells/organs, mobility of metals, metal concentration in soil, and time of plant exposure to metal toxicity. Heavy metals can alter the activity of various N assimilatory enzymes (e.g., NR, GS, GOGAT) by binding to the vital SH groups. Heavy metals that induce the alterations in plant water status; sugar metabolism; reductions in uptake and supply of essential nutrients; decrease in photosynthesis; generation of ROS; and membrane damage ultimately affect the N metabolism in the plant (Fig. 2).
In higher plants, GS/GOGAT is the main pathway of NH4 + assimilation under normal conditions, nevertheless, with the increase in endogenous NH4 + concentration, an alternate pathway, controlled by GDH contributes in decreasing this internal NH4 + concentration. Therefore, the activity of GDH is generally linked with the internal NH4 + concentration in plants. Heavy metals, particularly at higher concentrations, hamper the rate of nitrogen fixation in plants, by reducing infection process, growth of Rhizobium, nodule development, or host plant growth; however, such effects depend on heavy metal and plant species. Different soluble nitrogenous compounds such as amino acids, polyamines, proline, or glycine betaine can significantly accumulate in the plant under heavy metal stress to alleviate metal-induced adversities in plants. Generally, these compounds regulate osmotic potential in plant cells and thus improve heavy metal stress tolerance.
In the past, the majority of the research work was concerned only with the effect of metal toxicity on the uptake of N or activities of enzymes involved in N assimilation. Future studies should focus on the possible interferences between N transport and its acquisition under metal toxicity. Moreover, use of recent genomic techniques at transcriptomic or proteomic level for expression of specific genes encoding the N assimilatory enzyme proteins and N transporters will allow exploring the molecular intricacies of metal action. This will also lead to the development of strategies for improving plant resistance against metal toxicity. Efforts should also be made to unravel the mechanisms of possible antagonisms/synergisms among metal ions and determine the interaction of one metal ion in the transport and/or homeostasis of the other(s) and their effects on the N uptake and assimilation. The responses of N metabolism to multi-metal stress should also be examined in future studies because single metal toxicity is virtually nonexistent in the environment.
Abbreviations
- As:
-
Arsenic
- Cd:
-
Cadmium
- Cu:
-
Copper
- Fe:
-
Iron
- GB:
-
Glycine betaine
- GDH:
-
Glutamate dehydrogenase
- Gln:
-
Glutamine
- Glu:
-
Glutamate
- GOGAT:
-
Glutamate synthase
- GS:
-
Glutamine synthetase
- Hg:
-
Mercury
- MDA:
-
Malondialdehyde
- Mn:
-
Manganese
- Mo:
-
Molybdenum
- N:
-
Nitrogen
- NH4 + :
-
Ammonium
- Ni:
-
Nickel
- NiR:
-
Nitrite reductase
- NO3 − :
-
Nitrate
- NR:
-
Nitrate reductase
- Pb:
-
Lead
- ROS:
-
Reactive oxygen species
- SH:
-
Sulfhydryl
- V:
-
Vanadium
- Zn:
-
Zinc
References
Alcázar R, Marco F, Cuevas JC et al (2006) Involvement of polyamines in plant response to abiotic stress. Biotechnol Lett 28(23):1867–1876
Ali H, Khan E, Sajad MA (2013) Phytoremediation of heavy metals, concepts and applications. Chemosphere 91(7):869–881
Andrews M, Raven JA, Lea PJ (2013) Do plants need nitrate? The mechanisms by which nitrogen form affects plants. Ann Appl Biol 163(2):174–199
Anjum SA, Tanveer M, Hussain S et al (2016) Osmoregulation and antioxidant production in maize under combined cadmium and arsenic stress. Environ Sci Pollut Res 23(12):11864–11875
Anjum SA, Tanveer M, Hussain S et al (2017) Alteration in growth, leaf gas exchange, and photosynthetic pigments of maize plants under combined cadmium and arsenic stress. Water Air Soil Pollut 228(1):13
Antunes F, Aguilar M, Pineda M et al (2008) Nitrogen stress and the expression of asparagine synthetase in roots and nodules of soybean (Glycine max). Physiol Plant 133:736–743
Arora NK, Khare E, Singh S et al (2010) Effect of Al and heavy metals on enzymes of nitrogen metabolism of fast and slow growing rhizobia under explanta conditions. World J Microbiol Biotechnol 26:811–816
Ashoka P, Meena RS, Kumar S, Yadav GS, Layek J (2017) Green nanotechnology is a key for eco-friendly agriculture. J Clean Prod 142:4440–4441
Ashraf M, Foolad MR (2007) Roles of glycinebetaine and proline in improving plant abiotic resistance. Environ Exp Bot 59:206–216
Ashraf U, Kanu AS, Mo Z et al (2015) Lead toxicity in rice: effects, mechanisms, and mitigation strategies-a mini review. Environ Sci Pollut Res 22(23):18318–18332
Ashraf U, Hussain S, Anjum SA et al (2017) Alterations in growth, oxidative damage, and metal uptake of five aromatic rice cultivars under lead toxicity. Plant Physiol Biochem 115:461–471
Astolfi S, Zuchi S, Passera C (2004) Role of sulphur availability on cadmium induced changes of nitrogen and sulphur metabolism in maize (Zea mays L.) leaves. J Plant Physiol 161:795–802
Athar R, Ahmad M (2002a) Heavy metal toxicity: effect on plant growth and metal uptake by wheat, and on free living Azotobacter. Water Air Soil Pollut 138(1–4):165–180
Athar R, Ahmad M (2002b) Heavy metal toxicity in legume-microsymbiont system. J Plant Nutr 25(2):369–386
Ayangbenro AS, Babalola OO (2017) A new strategy for heavy metal polluted environments: a review of microbial biosorbents. Int J Environ Res Public Health 14:1–94
Aziz O, Hussain S, Rizwan M et al (2018) Increasing water productivity, nitrogen economy, and grain yield of rice by water saving irrigation and fertilizer-N management. Environ Sci Pollut Res 25:1–15. https://doi.org/10.1007/s11356-018-1855-z
Balestrasse KB, Gardey L, Gallego SM et al (2001) Response of antioxidant defence system in soybean nodules and roots subjected to cadmium stress. Funct Plant Biol 28(6):497–504
Balestrasse KB, Gallego SM, Tomaro ML (2004) Cadmium-induced senescence in nodules of soybean (Glycine max L.) plants. Plant Soil 262(1–2):373–381
Balestrasse KB, Gallego SM, Tomaro ML (2006) Oxidation of the enzymes involved in nitrogen assimilation plays an important role in the cadmium-induced toxicity in soybean plants. Plant Soil 284(1–2):187–194
Barroso JB, Francisco JC, Alfonso C et al (2006) Localization of S-nitrosoglutathione and expression of S-nitrosoglutathione reductase in pea plants under cadmium stress. J Exp Bot 57(8):1785–1793
Belimov AA, Dodd IC, Safronova VI et al (2015) The cadmium-tolerant pea (Pisum sativum L.) mutant SGECdt is more sensitive to mercury: assessing plant water relations. J Exp Bot 66(8):2359–2369
Bharwana SA, Ali S, Farooq MA et al (2014) Glycine betaine-induced lead toxicity tolerance related to elevated photosynthesis, antioxidant enzymes suppressed lead uptake and oxidative stress in cotton. Turk J Bot 38(2):281–292
Bianucci E, Fabra A, Castro S (2011) Cadmium accumulation and tolerance in Bradyrhizobium spp. (Peanut Microsymbionts). Curr Microbiol 62:96–100
Bielawski W (1994) Effect of some compounds on glutamine synthetase isoform activity from Triticale seedling leaves. Acta Physiol Plant 16:303–308
Bishnoi NR, Chugh LK, Sawhney SK (1993) Effect of chromium on photosynthesis, respiration and nitrogen fixation in pea (Pisum sativum L.) seedlings. J Plant Physiol 142(1):25–30
Boussama N, Ouariti O, Suzuki A (1999a) Cd-stress on nitrogen assimilation. J Plant Physiol 159:310–317
Boussama N, Ouariti O, Ghorbal MH (1999b) Changes in growth and nitrogen assimilation in barley seedlings under cadmium stress. J Plant Nutr 22:731–752
Bravo S, Amorós JA, Pérez-de-los-Reyes C et al (2017) Influence of the soil pH in the uptake and bioaccumulation of heavy metals (Fe, Zn, Cu, Pb and Mn) and other elements (Ca, K, Al, Sr and Ba) in vine leaves, Castilla-La Mancha (Spain). J Geochem Explor 174:79–83
Britto DT, Kronzucker HJ (2002) NH4+ toxicity in higher plants: a critical review. J Plant Physiol 159(6):567–584
Broos K, Beyens H, Smolders E (2005) Survival of rhizobia in soil is sensitive to elevated zinc in the absence of the host plant. Soil Biol Biochem 37:573–579
Burger M, Jackson LE (2003) Microbial immobilization of ammonium and nitrate in relation to ammonification and nitrification rates in organic and conventional cropping systems. Soil Biol Biochem 35(1):29–36
Burzynski M (1990) Activity of some enzymes involved in N03’ assimilation in cucumber seedlings treated with lead or cadmium. Acta Physiol Plant 12:105–110
Burzynski M, Buczek J (1994) The influence of Cd, Pb, Cu and Ni on NO-uptake by cucumber seedlings. II. In vivo and in vitro effects of Cd, Pb, Cu and Ni on the plasmalemma ATPase and oxidoreductase from cucumber seedling roots. Acta Physiol Plant 16:297–302
Burzynski M, Buczek J (1997) The effect of Cu2+ on uptake and assimilation of ammonium by cucumber seedlings. Acta Physiol Plant 19:3–8
Burzynski M, Buczek J (1998) Uptake and assimilation of ammonium ions by cucumber seedlings from solutions with different pH and addition of heavy metals. Acta Soc Bot Polon 67:197–200
Carillo P, Mastrolonardo G, Nacca F et al (2005) Nitrate reductase in durum wheat seedlings as affected by nitrate nutrition and salinity. Funct Plant Biol 32(3):209–219
Carpena RO, Vázquez S, Esteban E et al (2003) Cadmium-stress in white lupin: effects on nodule structure and functioning. Plant Physiol Biochem 41(10):911–919
Cesco S, Neumann G, Tomasi N et al (2010) Release of plant-borne flavonoids into the rhizosphere and their role in plant nutrition. Plant Soil 329(1–2):1–25
Chaffai R, Koyama H (2011) Heavy metal tolerance in Arabidopsis thaliana. In: Advances in botanical research, vol 60. Academic, Dordrecht, pp 1–49
Chaffei C, Gouia H, Ghorbel MH (2003) Nitrogen metabolism in tomato plants under cadmium stress. J Plant Nutr 26:1617–1634
Chaffei C, Pageau K, Suzuki A et al (2004) Cadmium toxicity induced changes in nitrogen management in Lycopersicon esculentum leading to a metabolic safeguard through an amino acid storage strategy. Plant Cell Physiol 45:1681–1693
Chen C, Wabduragala S, Becker DF et al (2006) Tomato QM-like protein protects Saccromyces cerevisiae cells against oxidative stress by regulation intracellular proline levels. Appl Environ Microbiol 72:4001–4006
Cheng Y, Wang J, Mary B et al (2013) Soil pH has contrasting effects on gross and net nitrogen mineralizations in adjacent forest and grassland soils in central Alberta, Canada. Soil Biol Biochem 57:848–857
Cheng Y, Wang C, Chai S et al (2018) Ammonium N influences the uptakes, translocations, subcellular distributions and chemical forms of Cd and Zn to mediate the Cd/Zn interactions in dwarf polish wheat (Triticum polonicum L.) seedlings. Chemosphere 193:1164–1171
Chibuike G, Obiora S (2014) Heavy metal polluted soils: effect on plants and bioremediation methods. Appl Environ Soil Sci 2014:1–12
Chien HF, Kao CH (2000) Accumulation of ammonium in rice leaves in response to excess cadmium. Plant Sci 156:111–115
Choppala G, Saifullah BN et al (2014) Cellular mechanisms in higher plants governing tolerance to cadmium toxicity. Crit Rev Plant Sci 33:374–391
Chugh LK, Gupta VK, Sawhney SK (1992) Effect of cadmium on enzymes of nitrogen metabolism in pea seedlings. Phytochemistry 31:395–400
Cruz FJR, de Almeida HJ, dos Santos et al (2014) Growth, nutritional status and nitrogen metabolism in ‘Vigna unguiculata’ (L.) Walp is affected by aluminum. Aust J Crop Sci 8(7):1132
Dabonne S, Koffi B, Kouadio E et al (2010) Traditional utensils: potential sources of poisoning by heavy metals. Br J Pharmacol Toxicol 151(2):90–92
Dadhich RK, Meena RS (2014) Performance of Indian mustard (Brassica juncea L.) in response to foliar spray of thiourea and thioglycolic acid under different irrigation levels. Indian J Ecol 41(2):376–378
Datta R, Anand S, Moulick A, Baraniya D, Pathan SI, Rejsek K, Vranova V, Sharma M, Sharma D, Kelkar A (2017a) How enzymes are adsorbed on soil solid phase and factors limiting its activity: a review. Int Agrophys 31(2):287–302
Datta R, Kelkar A, Baraniya D, Molaei A, Moulick A, Meena RS, Formanek P (2017b) Enzymatic degradation of lignin in soil: a review. Sustain MDPI 1163(9):1–18
Datta R, Baraniya D, Wang Y-F, Kelkar A, Meena RS, Yadav GS, Teresa Ceccherini M, Formanek P (2017c) Amino acid: its dual role as nutrient and scavenger of free radicals in soil. Sustainability 9(8):1402
Demidchik V, Sokolik A, Yurin V (1997) The effect of Cu2+ on ion transport systems of the plant cell plasmalemma. Plant Physiol 114:1313–1325
Devi SR, Prasad MNV (1999) Membrane lipid alterations in heavy metal exposed plants. In: Prasad MNV, Hagemeyer J (eds) Heavy metal stress in plants, from molecules to ecosystems. Springer, New York, pp 99–116
Dinakar N, Nagajyothi PC, Suresh S et al (2008) Phytotoxicity of cadmium on protein, proline and antioxidant enzyme activities in growing Arachis hypogaea L. seedlings. J Environ Sci 20(2):199–206
Dinakar N, Nagajyothi PC, Suresh S et al (2009) Cadmium induced changes on proline, antioxidant enzymes, nitrate and nitrite reductases in Arachis hypogaea L. J Environ Biol 30(2):289–294
Domínguez MJ, Gutiérrez F, León R (2003) Cadmium increases the activity levels of glutamate dehydrogenase and cysteine synthase in Chlamydomonas reinhardtii. Plant Physiol Biochem 41:828–832
Ekmekci Y, Tanyolac D, Ayhan B (2008) Effects of cadmium on antioxidant enzyme and photosynthetic activities in leaves of two maize cultivars. J Plant Physiol 165:600–611
Emamverdian A, Ding Y, Mokhberdoran F et al (2018) Growth responses and photosynthetic indices of bamboo plant (Indocalamus latifolius) under heavy metal stress. Sci World J 2018:1
Fageria NK, Filho MPB, Moreira A et al (2009) Foliar fertilization of crop plants. J Plant Nutr 32:1044–1064
Fashola M, Ngole-Jeme V, Babalola O (2016) Heavy metal pollution from gold mines: environmental effects and bacterial strategies for resistance. Int J Environ Res Public Health 13:1047
Fontaine JX, Saladino F, Agrimonti C et al (2006) Control of the synthesis and subcellular targeting of the two GDH gene products in leaves and stems of Nicotiana plumbaginifolia and Arabidopsis thaliana. Plant Cell Physiol 47:410–418
Frink CR, Waggoner PE, Ausubel JH (1999) Nitrogen fertilizer: retrospect and prospect. Proc Natl Acad Sci 96(4):1175–1180
Gajewska E, Skłodowska M (2009) Nickel-induced changes in nitrogen metabolism in wheat shoots. J Plant Physiol 166:1034–1044
Garnett T, Conn V, Kaiser BN (2009) Root based approaches to improving nitrogen use efficiency in plants. Plant Cell Environ 32(9):1272–1283
Geiger M, Haake V, Ludewig F (1999) The nitrate and ammonium nitrate supply have a major influence on the response of photosynthesis, carbon metabolism, nitrogen metabolism and growth to elevated carbon dioxide in tobacco. Plant Cell Environ 22(10):1177–1199
Ghosh S, Saha J, Biswas AK (2013) Interactive influence of arsenate and selenate on growth and nitrogen metabolism in wheat (Triticum aestivum L.) seedlings. Acta Physiol Plant 35(6):1873–1885
Giller KE, Witter E, McGrath SP (1998) Toxicity of heavy metals to microorganisms and microbial process in agricultural soils. A review. Soil Biol Biochem 30:1389–1414
Gouia H, Ghorbal MH, Meyer C (2000a) Effects of cadmium on activity of nitrate reductase and on other enzymes of the nitrate assimilation pathway in bean. Plant Physiol Biochem 38:629–638
Gouia H, Suzuki A, Brulfert J et al (2000b) Effects of cadmium on the co-ordination of nitrogen and carbon metabolism in bean seedlings. J Plant Physiol 160:367–376
Gouia H, Suzuki A, Brulfert J et al (2003) Effects of cadmium on the co-ordination of nitrogen and carbon metabolism in bean seedlings. J Plant Physiol 160(4):367–376
Graham PH, Vance CP (2000) Nitrogen fixation in perspective: an overview of research and extension needs. Field Crops Res 65(2–3):93–106
Greenwood NN, Earnshaw A (2012) Chemistry of the elements. Elsevier, Amsterdam/London/New York
Groppa MD, Tomaro ML, Benavides MP (2007) Polyamines and heavy metal stress: the antioxidant behavior of spermine in cadmium-and copper-treated wheat leaves. Biometals 20(2):185–195
Gumpu MB, Sethuraman S, Krishnan UM et al (2015) A review on detection of heavy metal ions in water? An electrochemical approach. Sens Actuators B Chem 213:515–533
Hall JL (2002) Cellular mechanisms for heavy metal detoxification and tolerance. J Exp Bot 53(366):1–11
Hernandez LE, Giirate A, Carpenta-Ruiz R (1996) Influence of cadmium on the assimilation of two cultivars of Zea mays. In: Cleemput OV, Hofman G, Vermoesen A (eds) Progress in nitrogen cycling studies. Kluwer Academic Publishers, Dordrecht, pp 115–132
Hernandez LE, Garate A, Carpena-Ruiz R (1997) Effects of cadmium on the uptake, distribution and assimilation of nitrate in Pisum sativum. Plant Soil 189:97–106
Huang H, Xiong ZT (2009) Toxic effects of cadmium, acetochlor and bensulfuron-methyl on nitrogen metabolism and plant growth in rice seedlings. Pest Biochem Physiol 94:64–67
Hussain D, Haydon MJ, Wang Y et al (2004) P-type ATPase heavy metal transporters with roles in essential zinc homeostasis in Arabidopsis. Plant Cell 16:1327–1339
Hussain S, Khan F, Cao W et al (2016) Seed priming alters the production and detoxification of reactive oxygen intermediates in rice seedlings grown under sub-optimal temperature and nutrient supply. Front Plant Sci 7. https://doi.org/10.3389/fpls.2016.00439
Imran M, Kanwal S, Hussain S et al (2015) Efficacy of zinc application methods for concentration and estimated bioavailability of zinc in grains of rice grown on a calcareous soil. Pak J Agric Sci 52(1):169–175
Ireland RJ, Lea PJ (1999) The enzymes ofglu-nine, glutamate, asparagine and aspartate metabolism. In: Singh BK (ed) Plant amino acids: biochemistry and biotechnology, vol 109. Marcel Dekker, New York
Islam MM, Hoque MA, Okuma E et al (2010) Exogenous proline and glycinebetaine increase antioxidant enzyme activities and confer tolerance to cadmium stress in cultured tobacco cells. J Plant Physiol 166:1587–1159
Jerzykiewicz J (2001) Aluminium effect on the nitrate assimilation in cucumber (Cucumis sativus L.) roots. Acta Physiol Plant 23:213–219
Jha AB, Dubey RS (2004) Arsenic exposure alters activity behavior of key nitrogen assimilatory enzymes in growing rice plants. Plant Growth Regul 43:259–268
Kakkar RK, Sawhney VK (2002) Polyamine research in plants–a changing perspective. Physiol Plant 116(3):281–292
Kalyanaraman SB, Sivagurunathan P (1993) Effect of cadmium, copper and zinc on the growth of blackgram. J Plant Nutr 16:2029–2042
Kapoor V, Li X, Elk M et al (2015) Impact of heavy metals on transcriptional and physiological activity of nitrifying bacteria. Environ Sci Technol 49(22):13454–13462
Kevrešan S, Petrović N, Popović M et al (1998) Effect of heavy metals on nitrate and protein metabolism in sugar beet. Biol Plant 41(2):235–240
Kevrešan S, Petrović N, Popović M et al (2001) Nitrogen and protein metabolism in young pea plants as affected by different concentrations of nickel, cadmium, lead, and molybdenum. J Plant Nutr 24(10):1633–1644
Khan F, Hussain S, Tanveer M et al (2018) Coordinated effects of lead toxicity and nutrient deprivation on growth, oxidative status, and elemental composition of primed and non-primed rice seedlings. Environ Sci Pollut Res 19:1–10
Krapp A, Fraisier V, Schleible WR et al (1998) Expression studies ofNrt2: INp, a putative high affinity nitrate transporter: evidence for its role in nitrate uptake. Plant J 6:723–732
Kubik-Dobosz G, Halajko T, Gbrska A (2001) Expression of amtl gene in the presence of some toxic ions. Acta Physiol Plant 23:187–192
Kumar S, Meena RS, Yadav GS, Pandey A (2017) Response of sesame (Sesamum indicum L.) to sulphur and lime application under soil acidity. Int J Plant Soil Sci 14(4):1–9
Lam HM, Coschigano KT, Melo-Oliveira O et al (1996) The molecular genetics of nitrogen assimilation into amino acids in higher plants. Annu Rev Plant Physiol Plant Mol Biol 47:569–593
Lee KC, Cunningham BA, Paulsen GM et al (1976) Effects of cadmium on respiration rate and activities of several enzymes in soybean seedlings. Physiol Plant 36(1):4–6
Lehmann S, Funck D, Szabados L et al (2010) Proline metabolism and transport in plant development. Amino Acids 39(4):949–962
Li P, Lu J, Wang Y et al (2018) Nitrogen losses, use efficiency, and productivity of early rice under controlled-release urea. Agric Ecosyst Environ 251:78–87
Llorens N, Arola L, Blade C et al (2000) Effects of copper exposure upon nitrogen metabolism in tissue cultured Vitis vinifera. Plant Sci 160:159–163
Lloyd DR, Phillips DH (1999) Oxidative DNA damage mediated by copper (II), iron (II) and nickel (II) Fenton reactions: evidence for site-specific mechanisms in the formation of double-strand breaks, 8-hydroxydeoxyguanosine and putative intrastrand cross-links. Mutat Res/Fundam Mol Mech Mutagen 424(1):23–36
Lojkova L, Datta R, Sajna M, Marfo TD, Janous D, Pavelka M, Formanek P (2015) Limitation of proteolysis in soils of forests and other types of ecosystems by diffusion of substrate. In: Amino acids, vol 8. Springer, Wien, pp 1690–1691
Loulakakis KA, Loulakakis-Roubelakis KA (1996) The seven NAD(H)-glutamate dehydrogenase isoenzymes exhibit similar anabolic and catabolic activities. Physiol Plant 96:29–35
Ma B, Wang S, Cao S et al (2016) Biological nitrogen removal from sewage via anammox: recent advances. Bioresour Technol 200:981–990
Malar S, Vikram SS, Favas PJ et al (2016) Lead heavy metal toxicity induced changes on growth and antioxidative enzymes level in water hyacinths [Eichhornia crassipes (Mart.)]. Bot Stud 55(1):54
Mandal SM, Gouri SS, De D et al (2011) Effect of arsenic on nodulation and nitrogen fixation of blackgram (Vigna mungo). Indian J Microbiol 51(1):44–47
Mandal C, Ghosh N, Dey N et al (2013) Physiological responses of Salvinia natans L to aluminium stress and its interaction with putrescine. J Stress Physiol Biochem 9(4):1
Marfo TD, Datta R, Lojkova L, Janous D, Pavelka M, Formanek P (2015) Limitation of activity of acid phosphomonoesterase in soils. In: Amino acids, vol 8. Springer, Wien, pp 1690–1691
Masclaux-Daubresse C, Valadier MH, Carrayol E et al (2002) Diurnal changes in the expression of glutamate dehydrogenase and nitrate reductase are involved in the C/N balance of tobacco source leaves. Plant Cell Environ 25:1451–1462
Matiru VN, Dakora FD (2004) Potential use of rhizobial bacteria as promoters of plant growth for increased yield in landraces of African cereal crops. Afr J Biotechnol 3(1):1–7
Mattioni C, Gabbrielli R, Vangronsveld J et al (1997) Nickel and cadmium toxicity and enzymatic activity in intolerant and non-tolerant populations of Silene italica Pers. J Plant Physiol 150(1–2):173–177
Matysik J, Alia BB et al (2002) Molecular mechanisms of quenching of reactive oxygen species by proline under stress in plants. Curr Sci 10:525–532
McAllister CH, Beatty PH, Good AG (2012) Engineering nitrogen use efficient crop plants: the current status. Plant Biotechnol J 10(9):1011–1025
Meena H, Meena RS (2017) Assessment of sowing environments and bio-regulators as adaptation choice for clusterbean productivity in response to current climatic scenario. Bangladesh J Bot 46(1):241–244
Meena RS, Yadav RS (2015) Yield and profitability of groundnut (Arachis hypogaea L) as influenced by sowing dates and nutrient levels with different varieties. Leg Res 38(6):791–797
Meena RS, Kumar S, Pandey A (2017) Response of sulfur and lime levels on productivity, nutrient content and uptake of sesame under guava (Psidium guajava L.) based Agri-horti system in an acidic soil of eastern Uttar Pradesh, India. J Crop Weed 13(2):222–227
Meena RS, Kumar V, Yadav GS, Mitran T (2018) Response and interaction of Bradyrhizobium japonicum and Arbuscular mycorrhizal fungi in the soybean rhizosphere: a review. Plant Growth Regul 84:207–223
Mengel, Kirkby (1987) Further elements of importance. Principle of plant nutrition, 4th edn. IPI Bern, Switzerland, pp 577–582
Mevel G, Prieur D (2000) Heterotrophic nitrification by a thermophilic Bacillus species as influenced by different culture conditions. Can J Microbiol 46(5):465–473
Michalak A (2006) Phenolic compounds and their antioxidant activity in plants growing under heavy metal stress. Pol J Environ Stud 15(4):523–530
Miflin BJ, Habash DZ (2002) The role of glutamine synthetase and glutamate dehydrogenase in nitrogen assimilation and possibilities for improvement in the nitrogen utilization of crops. J Exp Bot 53:979–987
Miflin BJ, Lea PJ, Wallsgrove RM (1980) The role of glutamine in ammonia assimilation and reassimilation in plants. In: Glutamine: metabolism, enzymology, and regulation. Academic Press, New York, pp 213–234
Miller JO (2016) Soil PH and nutrient availability. http://hdl.handle.net/1903/18519
Miller AJ, Smith SJ (1996) Nitrate transport and compartmentation in cereal root cells. J Exp Bot 47:1455–1463
Mittal S, Sawhney SK (1990) Influence of lead on enzymes of nitrogen metabolism in germinating pea seeds. Plant Physiol Biochem 17:73
Molaei A, Lakzian A, Datta R, Haghnia G, Astaraei A, Rasouli-Sadaghiani M, Ceccherini MT (2017a) Impact of chlortetracycline and sulfapyridine antibiotics on soil enzyme activities. Int Agrophys 31(4):499–505
Molaei A, Lakzian A, Haghnia G, Astaraei A, Rasouli-Sadaghiani M, Ceccherini MT, Datta R (2017b) Assessment of some cultural experimental methods to study the effects of antibiotics on microbial activities in a soil: an incubation study. PLoS One 12(7):e0180663
Myers RJK (1975) Temperature effects on ammonification and nitrification in a tropical soil. Soil Biol Biochem 7:83–86
Nichol BE, Oliveira LA, Glass ADM (1993) The effects of aluminum on the influx of calcium, potassium, ammonium, nitrate and phosphate in an aluminum-sensitive cultivar of barley (Hordeum vulgare L.). Plant Physiol 101:1263–1266
Oaks A (1994) Primary nitrogen assimilation in higher plants and its regulation. Can J Bot 72(6):739–750
Orzechowski S, Bielawski W (1997) Heavy metals and ammonium assimilation in triticale. J Appl Genet 38:265–270
Orzechowski S, Kwinta J, Gworek B et al (1997) Biochemical indicators of environmental contamination with heavy metals. Pol J Environ Stud 6:47–50
Pajuelo E, Rodríguez-Llorente ID, Dary M et al (2008) Toxic effects of arsenic on Sinorhizobium–Medicago sativa symbiotic interaction. Environ Pollut 154(2):203–211
Pandey M, Srivastava HS (1993) Inhibition of nitrate reductase activity and nitrate accumulation by mercury in maize leaf segments. Indian J Environ Health 39:473–481
Parlak KU (2016) Effect of nickel on growth and biochemical characteristics of wheat (Triticum aestivum L.) seedlings. NJAS-Wageningen J Life Sci 76:1–5
Paudyal SP, Aryal RR, Chauhan SVS et al (2007) Effect of heavy metals on growth of rhizobium strains and symbiotic efficiency of two species of tropical legumes. Sci World 5:27–32
Pérez-Tienda J, Corrêa A, Azcón-Aguilar C et al (2014) Transcriptional regulation of host NH4+ transporters and GS/GOGAT pathway in arbuscular mycorrhizal rice roots. Plant Physiol Biochem 75:1–8
Perkins LB, Johnson DW, Nowak RS (2011) Plant-induced changes in soil nutrient dynamics by native and invasive grass species. Plant Soil 345(1–2):365–374
Popović M, Kevrešan S, Kandrač J et al (1996) The role of sulphur in detoxification of cadmium in young sugar beet plants. Biol Plant 38(2):281–287
Prasad MN, Strzalka K (eds) (2013) Physiology and biochemistry of metal toxicity and tolerance in plants. Springer, Dordrecht
Quesada A, Hidalgo J, Femandez E (1997) Three nrt genes are differentially regulated in Chlamydomonas. Mol Genet 258:373–377
Rai V, Vajpayee P, Singh SN et al (2004) Effect of chromium accumulation on photosynthetic pigments, oxidative stress defense system, nitrate reduction, proline level and eugenol content of Ocimum tenuiflorum L. Plant Sci 167(5):1159–1169
Ram K, Meena RS (2014) Evaluation of pearl millet and mungbean intercropping systems in arid region of Rajasthan (India). Bangladesh J Bot 43(3):367–370
Rengel Z, Bose J, Chen Q et al (2016) Magnesium alleviates plant toxicity of aluminium and heavy metals. Crop Pasture Sci 66(12):1298–1307
Riaz M, Yan L, Wu X et al (2018a) Boron alleviates the aluminum toxicity in trifoliate orange by regulating antioxidant defense system and reducing root cell injury. J Environ Manag 208:149–158
Riaz M, Yan L, Wu X et al (2018b) Boron reduces aluminum-induced growth inhibition, oxidative damage and alterations in the cell wall components in the roots of trifoliate orange. Ecotoxicol Environ Saf 153:107–115
Riaz M, Yan L, Wu X et al (2018c) Mechanisms of organic acids and boron induced tolerance of aluminum toxicity: a review. Ecotoxicol Environ Saf 165:25–35
Rosales EP, Iannone MF, Groppa MD et al (2012) Polyamines modulate nitrate reductase activity in wheat leaves: involvement of nitric oxide. Amino Acids 42(2–3):857–865
Ruiz JM, Rivero RM, Romero L (2007) Comparative effect of Al, Se, and Mo toxicity on NO3 − assimilation in sunflower (Helianthus annuus L.) plants. J Environ Manag 83(2):207–212
Saifullah N, Sarwar S, Bibi M et al (2014) Effectiveness of zinc application to minimize cadmium toxicity and accumulation in wheat (Triticum aestivum L). Environ Earth Sci 71:1663–1672
Santamarı’a MM, Marrero ARD, Herna’ndez J et al (2003) Effect of thorium on the growth and capsule morphology of Bradyrhizobium. Environ Microbiol 5:916–924
Sarwar N, Malhi SS, Zia MH et al (2010) Role of mineral nutrition in minimizing cadmium accumulation by plants. J Sci Food Agric 90(6):925–937
Sarwar N, Ishaq W, Farid G et al (2015) Zinc-cadmium interactions: impact on wheat physiology and mineral acquisition. Ecotoxicol Environ Saf 122:528–536
Sarwar N, Imran M, Shaheen MR et al (2017) Phytoremediation strategies for soils contaminated with heavy metals: modifications and future perspectives. Chemosphere 171:710–721
Schmitt M, Watanabe T, Jansen S (2016) The effects of aluminium on plant growth in a temperate and deciduous aluminium accumulating species. AoB Plants 8:plw065
Selosse MA, Baudoin E, Vandenkoornhuyse P (2004) Symbiotic microorganisms, a key for ecological success and protection of plants. C R Biol 327(7):639–648
Sen G, Eryilmaz IE, Ozakca D (2014) The effect of aluminium-stress and exogenous spermidine on chlorophyll degradation, glutathione reductase activity and the photosystem II D1 protein gene (psbA) transcript level in lichen Xanthoria parietina. Phytochemistry 98:54–59
Seth CS (2012) A review on mechanisms of plant tolerance and role of transgenic plants in environmental clean-up. Bot Rev 78(1):32–62
Shah K, Dubey RS (2003) Environmental stresses and their impact on nitrogen assimilation in higher plants. In: Hemantranjan A (ed) Advances in plant physiology, vol 5. Scientific Publishers, Jodhpur, pp 397–431
Shahzad B, Tanveer M, Rehman A (2018) Nickel; whether toxic or essential for plants and environment: a review. Plant Physiol Biochem 132:641. https://doi.org/10.1016/j.plaphy.2018.10.014
Sharma SS, Dietz KJ (2006) The significance of amino acids and amino acid-derived molecules in plant responses and adaptation to heavy metal stress. J Exp Bot 57(4):711–726
Sharma P, Dubey RS (2005) Modulation of nitrate reductase activity in rice seedlings under aluminium toxicity and water stress: role of osmolytes as enzyme protectant. J Plant Physiol 162(8):854–864
Sharma JOT, Subhadra AV (2010) The effect of mercury on nitrate reductase activity in bean leaf segments (Phaseolus vulgaris) and its chelation by phytochelatin synthesis. Life Sci Med Res 2010:1–8
Sharma SS, Schat H, Vooijs R (1998) In vitro alleviation of heavy metal-induced enzyme inhibition by proline. Phytochemistry 49(6):1531–1535
Shaw BP, Sahu SK, Mishra RK (2004) Heavy metal induced oxidative damage in terrestrial plants. In: Presad MNV (ed) Heavy metal stress in plants: from biomolecules to ecosystems, 2nd edn. Springer, Berlin, pp 84–126
Shi H, Chan Z (2014) Improvement of plant abiotic stress tolerance through modulation of the polyamine pathway. J Integr Plant Biol 56(2):114–121
Shruti M, Dubey RS (2006) Heavy metal uptake and detoxification mechanisms in plants. Int J Agric Res 1:122–141
Silveira JAG, Melo ARB, Viégas RA et al (2001) Salinity-induced effects on nitrogen assimilation related to growth in cowpea plants. Environ Exp Bot 46(2):171–179
Singh PK, Tewari RK (2003) Cadmium toxicity induced changes in plant water relations and oxidative metabolism of Brassica juncea L. plants. J Environ Biol 24(1):107–112
Singh RP, Singh DP, Jaiwal PK (2008) Nitrate and ammonium transporters in plants. Plant membrane and vacuolar transporters. CAB International, Wallingford, pp 83–103
Singh H, Singh A, Hussain I et al (2017) Oxidative stress induced by lead in Vigna radiata L. seedling attenuated by exogenous nitric oxide. Trop Plant Res 4(2):225–234
Skopelitis DS, Paranychianakis NV, Paschalidis KA et al (2006) Abiotic stress generates ROS that signal expression of anionic glutamate dehydrogenases to form glutamate for proline synthesis in tobacco and grapevine. Plant Cell 18:2767–2781
Stan V, Gament E, Corena CP et al (2011) Effects of heavy metal from polluted soils on the rhizobium diversity. Not Bot Hort Agrobot Cluj 39:88–95
Stitt M (1999) Nitrate regulation of metabolism and growth. Curr Opin Plant Biol 2:178–186
Syntichaki KM, Loulakakis KA, Loulakakis-Roubelakis KA (1996) The amino-acid sequence similarity of plant glutamate dehydrogenase to the extremophilic archaeal enzyme conforms to its stress-related function. Gene 68:87–92
Szabados L, Savoure A (2010) Proline: a multifunctional amino acid. Trends Plant Sci 15:89–97
Talano MA, Cejas RB, González PS et al (2013) Arsenic effect on the model crop symbiosis Bradyrhizobium–soybean. Plant Physiol Biochem 63:8–14
Terce-Laforgue´ T, Dubois F, Ferrario-Me’ry S et al (2004) Glutamate dehydrogenase of tobacco is mainly induced in the cytosol of phloem companion cells when ammonia is provided either externally or released during photorespiration. Plant Physiol 136:4308–4317
Vajpayee P, Tripathi RD, Rai UN et al (2000) Chromium (VI) accumulation reduces chlorophyll biosynthesis, nitrate reductase activity and protein content in Nymphaea alba L. Chemosphere 41:1075–1082
Van Assche F, Cardinaels C, Clijsters H (1988) Induction of enzyme capacity in plants as a result of heavy metal toxicity: dose-response relations in Phaseolus vulgaris L., treated with zinc and cadmium. Environ Pollut 52(2):103–115
Varma D, Meena RS, Kumar S (2017) Response of mungbean to fertility and lime levels under soil acidity in an alley cropping system in Vindhyan region, India. Int J Chem Stud 5(2):384–389
Verkleij JA, Golan-Goldhirsh A, Antosiewisz DM et al (2009) Dualities in plant tolerance to pollutants and their uptake and translocation to the upper plant parts. Environ Exp Bot 67(1):10–22
Verma SK, Singh SB, Prasad SK, Meena RN, Meena RS (2015) Influence of irrigation regimes and weed management practices on water use and nutrient uptake in wheat (Triticum aestivum L. Emend. Fiori and Paol.). Bangladesh J Bot 44(3):437–442
Wang MY, Siddiqi MY, Ruth TJ et al (1993) Ammonium uptake by rice roots. Plant Physiol 103:1259–1267
Wang L, Zhou Q, Ding L et al (2008) Effect of cadmium toxicity on nitrogen metabolism in leaves of Solanum nigrum L. as a newly found cadmium hyperaccumulator. J Hazard Mater 154:818–825
Wang Y, Lu J, Ren T et al (2017) Effects of nitrogen and tiller type on grain yield and physiological responses in rice. AoB Plants 9. https://doi.org/10.1093/aobpla/plx012
Wani PA (2008) Heavy metal toxicity to plant growth promoting rhizobacteria (PGPR) and certain legume crops. Ph.D. Thesis, Aligarh Muslim University, Aligarh, India
Wani PA, Khan MS, Zaidi A (2007) Impact of heavy metal toxicity on plant growth, symbiosis, seed yield and nitrogen and metal uptake in chickpea. Aust J Exp Agric 47(6):712–720
Weber MB, Schat H, Ten Bookum-Van Der Maarel WM (1991) The effect of copper toxicity on the contents of nitrogen compounds in Silvena vulgaris (Moench) Garcke. Plant Soil 133:101–109
Wu LH, Luo YM, Xing XR et al (2004) EDTA-enhanced phytoremediation of heavy metal contaminated soil with Indian mustard and associated potential leaching risk. Agric Ecosyst Environ 102(3):307–318
Xiong ZT, Liu C, Geng B (2006a) Phytotoxic effects of copper on nitrogen metabolism and plant growth in Brassica pekinensis Rupr. Ecotoxicol Environ Saf 64(3):273–280
Xiong ZT, Zhao F, Li MJ (2006b) Lead toxicity in Brassica pekinensis Rupr.: effect on nitrate assimilation and growth. Environ Toxicol Int J 21(2):147–153
Xu J, Yin H, Li X (2009) Protective effects of proline against cadmium toxicity in micropropagated hyperaccumulator, Solanum nigrum L. Plant Cell Rep 28(2):325–333
Yadav GS, Babu S, Meena RS, Debnath C, Saha P, Debbaram C, Datta M (2017) Effects of godawariphosgold and single supper phosphate on groundnut (Arachis hypogaea) productivity, phosphorus uptake, phosphorus use efficiency and economics. Indian J Agric Sci 87(9):1165–1169
Yancey PH (2005) Organic osmolytes as compatible, metabolic and counteracting cytoprotectants in high osmolarity and other stresses. J Exp Biol 208:2819–2830
Yang Y, Zhang Y, Wei X et al (2011) Comparative antioxidative responses and proline metabolism in two wheat cultivars under short term lead stress. Ecotoxicol Environ Saf 74(4):733–740
Yao S, Ni JR, Ma T et al (2013) Heterotrophic nitrification and aerobic denitrification at low temperature by a newly isolated bacterium, Acinetobacter sp. HA2. Bioresour Technol 139:80–86
Yu CC, Hung KT, Kao CH (2005) Nitric oxide reduces Cu toxicity and Cu-induced NH4+ accumulation in rice leaves. J Plant Physiol 162(12):1319–1330
Zhou X, Gu Z, Xu H et al (2016) The effects of exogenous ascorbic acid on the mechanism of physiological and biochemical responses to nitrate uptake in two rice cultivars (Oryza sativa L.) under aluminum stress. J Plant Growth Regul 35(4):1013–1024
Zia-ur-Rehman M, Sabir M, Nadeem M (2015) Remediating cadmium-contaminated soils by growing grain crops using inorganic amendments. In: Soil remediation and plants: prospects and challenges. Elsevier Inc/Academic, London, pp 367–396
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Hussain, S. et al. (2020). Metal Toxicity and Nitrogen Metabolism in Plants: An Overview. In: Datta, R., Meena, R., Pathan, S., Ceccherini, M. (eds) Carbon and Nitrogen Cycling in Soil. Springer, Singapore. https://doi.org/10.1007/978-981-13-7264-3_7
Download citation
DOI: https://doi.org/10.1007/978-981-13-7264-3_7
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-7263-6
Online ISBN: 978-981-13-7264-3
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)