Abstract
Irrigation of Salicornia europaea with heavy metal–polluted wastewater is a promising alternative method for risk mitigation of the Urmia Lake ecosystem from uncontrolled sewage. The objective of the study was to evaluate morphological and physicochemical responses of Salicornia europaea under wastewater irrigation at different growth stages. A field experiment was conducted in a split-plot experiment based on randomized complete block design with four replications. Treatments included control and wastewater irrigation (containing zinc (Zn), iron (Fe), copper (Cu), cadmium (Cd), lead (Pb), and nickel (Ni)) at three stages (vegetative, flowering, and reproductive) of plant growth and two times (two and 4 days in each stage). The result showed that the wastewater application at reproductive stage resulted in higher biomass production than that of the control plants. Wastewater irrigation at the flowering stage caused a significant increase in the amount of total chlorophyll and chlorophyll-a, while chlorophyll-b content was decreased at both flowering and reproductive stages. The amount of the total soluble protein was also affected, with wastewater irrigation showing the most significant increase at the reproductive stage. There was significant enhancement of osmolytes in leaves of plant under heavy metal stress, and the increased rate of proline was higher than soluble sugar at the flowering stage. Relative water content in Salicornia was not duration- and time-dependent. A 154% increase in catalase activity, 32% increase in peroxidase activity, and 57% increase in polyphenol oxidase activity were observed in the plant exposed to long-term wastewater duration. Based on the observed positive effect of wastewater on shoot length and weight, total soluble protein, proline, soluble sugar, enzyme activities, and plant biomass of Salicornia europaea, long-term effect of heavy metal–polluted wastewater irrigation can be approved for Salicornia crops in coastal areas.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Pollution of irrigation water may affect both the biotic and abiotic components of the ecosystem (Qadir et al. 2010). Farmers in developing countries who require water for irrigation often have no other choice than using wastewater (Keraita et al. 2008; Liu et al. 2005). The wastewater irrigation provides a valuable source of plant nutrients and organic matter needed for maintaining fertility and productivity levels of the soil (Qadir et al. 2010). High concentrations of heavy metals inhibit plant growth and development and cause various morphological, physiological, and biochemical responses (Song et al. 2014; Ahmed and Slima 2018). The degree of plant damage depends on the plant species, the concentration of toxic ion, its stage of growth, climate, and soil conditions (Qadir et al. 2010). It was reported that high concentrations of heavy metals in wastewater affect many physiological and biochemical processes and inhibit plant growth and development (John et al. 2009).
Most of the peri-urban lands are contaminated with pollutants, including copper (Cu), zinc (Zn), lead (Pb), cadmium (Cd), nickel (Ni), and mercury (Hg) (Singh and Kumar 2006) especially in the arid and semi-arid regions (Mojid et al. 2019). The discharges of wastewater is the most hazardous when the heavy metals exceed allowable concentration limits (Barakat 2011). The toxic effects, environmental stability (Esmaeilzadeh et al. 2016), and high potential for being absorbed and bio-accumulated in many aquatic species are important aspects of heavy metals (Esmaeilzadeh et al. 2017). Long-term irrigation with sewage effluent was shown to increase the amount and availability of heavy metals in soils (Liu et al. 2005). Soil fertility and plant growth can be enhanced with properly managed wastewater irrigation through increasing levels of soil organic matter and plant nutrients which may eliminate the need for commercial fertilizers in cropland (Mojid et al. 2019; Eid 2019). To alleviate the problems of land salinization and the contamination of soils with heavy metals, a group of plants has been identified such as halophytes, salt-tolerant plants, and heavy metal hyperaccumulators which can extract heavy metals from the soil (Moray et al. 2015). In coastal saline areas, heavy metal pollution could only be remediated by using halophytes that are probably the only candidate for phytoremediation of heavy metal–polluted saline soils (Sharma et al. 2010; Kumari et al. 2019). Halophytes and hyperaccumulators are found in a diverse range of angiosperm families and use similar mechanisms to combat the salt and heavy metal–borne stresses (Moray et al. 2015).
Salicornia europaea L. is an annual halophyte from the Chenopodiaceae family (Piernik et al. 2017) and one of the plants that has the highest salt tolerance (greater than 1 M NaCl) in the world (Rozema and Schat 2013; Zare-Maivan et al. 2015) and therefore well adapted in an environment where the level of soil salinity is as high as half-strength of seawater (Ebadi et al. 2018). It is widespread in Urmia Lake, Iran (Zare-Maivan et al. 2015), Europe, South Africa, South Asia, and North America and prevalent in salt-affected areas near coastlines, tidal floodways, salt lakes, and brine springs (Piernik et al. 2017). Since S. europaea dominates arid and saline soils of retreated Urmia Lake, its cell membrane integrity and antioxidant potential to acclimated to such conditions (Zare-Maivan et al. 2015). These species can survive in saline environments due to physiological and biochemical adaptations (Smillie 2015). Enzymatic and non-enzymatic antioxidants are important in plant defense under heavy metal stress (Kandziora-Ciupa et al. 2013). Previous reports have shown that the content of free proline depends on the type of metal and its concentration (Sharma and Dietz 2006) and plant species and varies between organs (Kandziora-Ciupa et al. 2013).
Long-term heavy metal exposure can enhance ethylene production and induce oxidative stress in plants (Thao et al. 2015). Higher ethylene production is due to the peaked expression of ethylene-responsive genes (Sharma et al. 2020). Higher production of reactive oxygen species (ROS), induced by some heavy metals, causes oxidative stress (Mittler 2002; Sytar et al. 2018). Heavy metals lead to damaging effects such as photochemical reactions, chlorophyll degradation, enzyme inhibition, disruption of membranes integrity, and reduction in metabolic efficiency and carbon fixation (Liu et al. 2005; Seregin and Ivanov 2001). Antioxidant enzymes like superoxide dismutase (SOD), catalase (CAT), peroxidase (POD), and polyphenol oxidase (PPO) along with the non-enzymatic antioxidant are the major components of ROS-scavenging system (Babaei et al. 2017; Seyed Sharifi et al. 2017). In non-hyperaccumulator plants, heavy metals at high concentration like Zn in wheat (Li et al. 2013) and Cu in Solanum nigrum (Fidalgo et al. 2013) led to an inhibition of the antioxidant enzyme. Halophytes are expected to be more capable to cope with heavy metal stress than common plants (Wu et al. 2017) due to a more efficient antioxidant system (Perez-Romero et al. 2016). For example, halophyte Salicornia brachiata can upregulate the activity of CAT and SOD and have lower damage to lipid membrane from ROS in response to heavy metals such as Cd, Ni, and arsenic (As) (Sharma et al. 2010; Wang et al. 2014).
There has been a continuing interest in searching for tolerant native plants to heavy metals. It remains to be further investigated how these metal ions affect plant growth at different developmental stages. Recently, the responses of plants to individual metal were extensively studied (Pedro et al. 2013; Perez-Romero et al. 2016). However, in natural soil-plant systems, they often have to face multiple metal stresses, and the interactive effects of more elements should be studied (An et al. 2004). Consequently, the present study aimed to evaluate the effects of time and duration of heavy metal–polluted wastewater application on the growth parameters (nodes on the main stem, number of pairs of side branches, diameter of the branched area, the plant biomass), enzyme activities, proline, soluble sugars, chlorophyll, and protein contents of Salicornia europaea in coastal areas of Urmia Lake. We hypothesized that the native S. europaea exhibits optimum growth and physiological parameters in urban and industrialized areas with respect to effectively reduce pollution of irrigation water.
2 Materials and Methods
2.1 Study Area
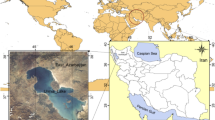
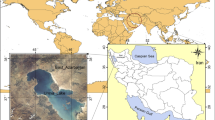
The study site is in the Urmia region (37°42′ N, 45° 19′ E), located in the north west of Iran (Fig. 1). The area is characterized by average annual rainfall of 300–700 mm; average annual temperature is −5 °C to 10 °C and has cold winters and hot summers. This study was conducted at coastal areas of Urmia Lake irrigated with the water contaminated by domestic and industrial effluent.
2.2 Treatments and Experimental Design
The experiment was conducted in a split-plot design with four replications for each treatment. The main-plot factor of the experiment includes control (freshwater irrigation) and wastewater irrigation, and sub-plot factors include the irrigation with sewage at three stages, i.e., short term (20 days after transplanting, up to the vegetative growth stage in 14 August 2019), average term (30 days after transplanting, up to the flowering stage in 24 August 2019), and long term (40 days after sowing up to the reproductive stage), and two sampling times, i.e., 2 days and 4 days after irrigation with sewage. Indeed, wastewater quality parameters change over time due to floating/settling according to the type of media (Sirianuntapiboon and Kongchum 2006; Cortes-esquivel et al. 2012). Each plot consisted of 3 rows with 100-m long. Seeds were sown in 10 July and transplanted in 24 July 2019. Irrigation was done to maintain soil moisture above 80% field capacity. The amount of irrigation water was obtained based on Benami and Ofen (1984).
2.3 Soil and Water Analysis
The soil of the site was clay loam soil (21% sand, 48% silt, and 31% clay) with EC 33.8 dS m−1, pH 8.81, phosphorus 3.7 mg kg−1, and nitrogen 0.01%. Minimum and maximum values of heavy metals in sewage water during the 2019 are shown in Table 1. Farmers in these areas who require water for irrigation often use sewage, and the process is going on for about 30 years.
2.4 Plant Analysis
The number of pairs of side branches, nodes on the main stem, stem elongation, diameter of the branched area, and root length was recorded during the growth period. The concentrations of chlorophyll and carotenoids were calculated using the Arnon (1949) method. The total soluble protein content was determined by the method of Bradford (1976). To extract protein, 0.2 g of the plant fresh tissue was crushed by using liquid nitrogen, followed by adding 1 mL of buffer Tris-HCl (0.05 M, pH = 7.5). The obtained mixture was centrifuged for 20 min at 16000×g (13,000 rpm) at 4 °C, and then, the supernatant was utilized for enzyme activity measurements. Further, CAT, POD, and PPO activities were assayed according to Kar and Mishra (1976). Soluble sugars were measured according to Dubois et al. (1956). Leaf proline content was determined based on Bates et al. (1973). Relative water content (RWC) was calculated based on Tambussi et al. (2005). Electrical conductivity was estimated by the method of Jodeh et al. (2015).
2.5 Statistical Analysis
Analysis of variance (ANOVA) and means comparison on data were performed using the SAS 9.1 statistical package program. The least significant difference (LSD) method was used to test the significant differences between means comparison of main effects and interactions.
3 Results
3.1 Morphological Traits
Increasing the time exposure to wastewater improves the diameter of the branched area, but did not present a significant change in response to heavy metals at flowering and reproductive stages (Table 2). The nodes on the main stem and the number of pairs were lower in the reproductive stage than the flowering stage. Time had a significant effect on a number of pairs and the diameter of the branched area.
3.2 Biomass Components
As presented in Table 3, with wastewater irrigation, up to 16% increase in shoot length is observed, and the longest shoot is recorded at the reproductive stage. Root length was not significantly affected by wastewater irrigation, whereas it significantly decreased at reproductive stage. The highest plant biomass is found in plants irrigated with sewage at the reproductive stage (Table 3). Under the long-term duration of wastewater, aboveground dry biomass of S. europaea reached to 780 mg per plant. At the reproductive growth stage, root biomass of Salicornia reached to the highest weight per plant.
3.3 Photosynthetic Pigments and Total Soluble Protein
This study shows that the application of wastewater caused a significant increase in chlorophyll-a, chlorophyll-b, and total chlorophyll compared with the control but 40 days after sowing (reproductive stage) produced a severe reduction in chlorophyll-a, total chlorophyll, and carotenoid by 49%, 44%, and 153%, respectively, than the flowering stage (Table 4). Chlorophyll-a and total chlorophyll increased significantly at 30 days after sowing in plants exposed to wastewater but, at the reproductive stage, 19 to 37% and 14 to 43% reduction was noted in chlorophyll-a and total chlorophyll, respectively, in comparison with the control. The reduction of total chlorophyll was less pronounced in chlorophyll-b than in chlorophyll-a. Chlorophyll-b was 37% lower than the control for longer time (40 days after sowing) of wastewater exposure. A significant increase in leaf total soluble protein was obtained from wastewater-irrigated Salicornia at the reproductive stage. The content of soluble protein was the lowest in untreated control plants after 20 days of sowing (vegetative growth stage).
3.4 Soil Electrical Conductivity
In the present study, although there was no significant effect of time on electrical conductivity (EC), a significant interaction between time and irrigation is observed when wastewater treatment is applied (Table 4). During a growth period of plants, the level of lipid peroxides improves with an increase in the time of exposure to wastewater (Table 4). Long-term expose to wastewater led to 11% increase in EC level compared with the control.
3.5 Proline and Soluble Sugars
The results presented in Fig. 2 show that proline and soluble sugars were significantly increased in leaves of Salicornia plants exposed to wastewater, and they were higher in the reproductive stage within 4 days after irrigation with sewage. Under wastewater irrigation, proline content at the flowering and reproductive stage increases by 33% and 59%, respectively, compared with the control, but at vegetative stage, proline content increases to 5% compared with the control (Fig. 2A).
Proline (A) and soluble sugars (B) of Salicornia europaea as affected by time and duration of wastewater application. Fast, 20 days after sowing (at vegetative stage); average, 30 days after sowing (at flowering stage); long term, 40 days after sowing (at reproductive stage). Each bar indicates mean (n = 4) ± standard error. The different alphabets on bars indicate significant difference (P ≤ 0.05) using LSD
In control plants, the level of soluble sugars is markedly decreased, in 4 days after irrigation (Fig. 2B). The accumulation of soluble sugars in the leaves was observed as a response to the long-term effect of wastewater, and maximum level was reached at 4 days after irrigation which was 63% higher than at control.
3.6 The Activity of Catalase, Peroxidase, and Polyphenol Oxidase Enzymes
Activities of antioxidant enzymes are determined over an experimental period in control and wastewater-treated leaves (Figs. 3 and 4). In control plants, the tendency of PPO activity ascended at flowering stage and then declined in 4 days after irrigation of reproductive stage, while POD activity fluctuated in different time and duration, and CAT activity was dropped slightly. In plants growing in the wastewater with excessive heavy metals, the lowest level of CAT, POD, and PPO was observed for the first time in the vegetative stage, but they were still higher than in controls. The data showed that CAT activity increased between 5.5 and 155% in plants treated with wastewaters. The highest CAT activity was observed at the reproductive stage. At this time, CAT activity is approximately 154% higher than in the control plants (Fig. 3A).
Catalase (CAT, A) and peroxidase (POD, B) of Salicornia europaea as affected by time and duration of wastewater application. Fast, 20 days after sowing (at vegetative stage); average, 30 days after sowing (at flowering stage); long term, 40 days after sowing (at reproductive stage). Each bar indicates mean (n = 4) ± standard error. The different alphabets on bars indicate significant difference (P ≤ 0.05) using LSD
Polyphenol oxidase enzymes (PPO, A) and RWC (B) of Salicornia europaea as affected by time and duration of wastewater application. Fast, 20 days after sowing (at vegetative stage); average, 30 days after sowing (at flowering stage); long term, 40 days after sowing (at reproductive stage). Each bar indicates mean (n = 4) ± standard error. The different alphabets on bars indicate significant difference (P ≤ 0.05) using LSD
Plants exposed to short- and long-term duration did not show significant changes in POD, indicating that the high metal concentrations did not affect the activity of POD at any time (Fig. 3B). At 4 days after irrigation of the flowering and reproductive stages, a 15–56% higher than control of POD was obtained. The maximum increase in POD activity was 32.4% during 4 days after irrigation at the reproductive stage, 16.7% during 4 days after irrigation, and 16.2% during 2 days after irrigation at the flowering stage. The activity of PPO was increased with the wastewater irrigation, but significant changes were not observed at the vegetative stages (Fig. 4A). The enzymatic activity observed at 4 days of irrigation was higher than 2 days of irrigation.
3.7 Relative Water Content
Irrigation with wastewater results in considerable negative water potential within 4 days after irrigation of flowering stage when compared with the untreated control (Fig. 4B). In control plants, RWC was high and then remained unaltered when the plants were irrigated with wastewater for the first time. Long-term exposure to wastewater caused only minor alterations in RWC. RWC in Salicornia was not duration- and time-dependent in wastewater irrigated plants, and there is a gradual decrease in RWC in plant exposed to wastewater at the vegetative and flowering stage. At the 4 days after irrigation of the reproductive stage, RWC considerably decreased by 5.4% in comparison with the control.
4 Discussions
Salicornia plants exposed to time and different duration of heavy metals exhibited morphological, physiological, and biochemical changes. The metal iron (Fe), Cu, Ni, and Zn are essential for plants but are also toxic when found in high concentrations (Kranner and Colville 2011; Yang et al. 2012). The presence of unbalance nutrients can disturb the uptake of necessary elements and consequently plant growth (Mallick 2004). Wastewater-treated plants show a healthy morphology, indicating the non-toxic concentrations of the metals. Based on observed results in response to higher concentrations of Cu-, Cd-, Pb-, and Ni-treated plant, the higher diameter of the branched area, shoot length, and shoot weight may be due to the sufficiency of Fe and Zn in the wastewater. In general, the root-shoot transfer of Zn and Cu was higher than that of Cd, Pb, and Ni (Eissa and Negim 2018). The gradual supply of plants with Zn, Fe, and other heavy metals occurred when plants exposed to the average term duration of wastewater at the flowering stage. Reduction in dry matter yield of Salicornia plants at a higher concentration of heavy metals was observed by Sharma et al. (2011) due to Ni, Ozawa et al. (2009) due to Cd, and by Sharma et al. (2010) due to Cd and Ni. Hatata and Abdel-aal (2008) stated that a high ratio of dry mass to fresh mass is a criterion for stress response, which is indicative of the whole-plant level. Some researchers have shown that biomass estimations have been confined to aboveground biomass and the root system biomass can comprise 30–80% of the total plant biomass (Eltaher et al. 2019). It is interesting to note that higher shoot to root growth ratio occurred in plants that had been treated with Pb (Nicholls and Mal 2003). With the development of plant roots during the growing season, Pb and Cd were uptake much more by Salicornia plants. These above adaptions and improvements make the soil suitable for the growth of healthy plants (Farid et al. 2013). The inhibitory effect of heavy metals on root length seems mainly due to the reduced water uptake (Farid et al. 2013) and reduction in mitotic cells in the meristematic zone of the root (Kabir et al. 2010). Also, it may be correlated with the metal-induced inhibition of the photosynthetic process (Hatata and Abdel-aal 2008) and the respiration in the shoot system and protein synthesis in the root (Drazkiewicz and Baszyński 2005). Due to the presence of large quantities of Ni and Cd in the wastewater (Table 1), only minor amounts of Fe and Zn are translocated to the shoots, and the excess of Fe in roots of plants hinders the development of cells and causes higher plant biomass (Adamski et al. 2012). Nicholls and Mal (2003) also observed that the cells of roots are affected more severely by the exposure to Cu than are other parts of the plant. Thus, if Cu damaged the roots of the treated plants more severely, the roots may not have been able to support the growth of the shoot.
Heavy metals observed in irrigation treatments (Table 1), as micronutrients essential for the plant growth and development, could lead to inhibition of photosynthesis when they were greater in the reproductive stage. It seems that during the flowering stage in Salicornia, the optimum of the metal uptake occurs, causing increased chlorophyll content. During the reproductive stage, where the heavy metals were the strongest inducer, a content of chlorophyll-a, chlorophyll-b, and total chlorophyll was decreased in the leaves of plants. However, the negative effect of heavy metals on chlorophyll-a fluorescence, electron transport, and light-dependent reactions of photosynthesis depend on metal concentrations and exposure time (Hajihashemi et al. 2020). Moreover, the long-term exposure of heavy metals inhibited the activity of PSI (Sharma et al. 2020). In this study, a high concentration of heavy metals at the end of growing stage inhibits uptake and transportation of other metal elements such as Zn and Fe by antagonistic effects, and therefore, the leaves lose the capacity of synthesis of pigments (Zengin and Kirbag 2007; Hatata and Abdel-aal 2008). The higher concentrations of Ni caused serious damage to chloroplasts, carotenoids, and chlorophyll and caused CO2 deficiency due to reduced enzyme activity in the Calvin cycle, as well as changes in the thylakoid membrane that affected the photosynthesis process (Pandey and Sharma 2002). Many studies have shown the influences of a positive effect of Zn and Cd metals on chlorophyll content, but high concentration of Zn in wastewater could be one of the main reasons for the decline in chlorophyll (Hajihashemi et al. 2020). Farid et al. (2013) stated that in Cd-treated wheat plants, increased leaf photosynthesis was observed due to the smaller leaf area. However, the results of this study could be interpreted as the effect of strong oxidation due to the metabolic perturbations and accelerated senescence (Zhou and Qiu, 2018), presence of Cd on the photochemical apparatus (Hatata and Abdel-aal 2008; Singh et al. 2012), and replaced Zn in chlorophylls (Mukhopadhyay et al. 2013) and probably due to the effect of interference of Cu with Fe uptake (Zengin and Kirbag 2007) and interaction of Pb to –SH group of enzymes of chlorophyll biosynthesis (Shu et al. 2012). Zengin and Kirbag (2007) reported that 0.4 mg L−1 of Cu resulted in a significant decrease of chlorophyll, and the minimum value of chlorophyll was observed when plants were exposed to 0.6 mg L−1 copper. The severe effects of Cu on the chloroplast in Salicornia were also reported from other plants (Andosch et al. 2012; Bazihizina et al. 2015). Moreover, numerous studies have reported a direct effect of Cu on the photosynthetic electron transport chain (Li et al. 2015), which may be associated with substantial stress response. Bibi and Hussain (2005) reported that Pb despite of low concentration proved more toxic in effecting photosynthetic pigments compared with Cu- and Cd-treated plants.
Visible symptoms of excessive heavy metals were accompanied by an increased total soluble protein, as an important indicator of the biochemical status of plants (Kandziora-Ciupa et al. 2013). This observation does not exclude the possibility of increased degradation of protein under excess heavy metals. Long-term exposure to wastewater irrigation showed significant increasing either enzyme activity or protein as well as inhibiting the production of H2O2 (El-Amier et al. 2019) to acclimate heavy metal toxicity through metal binding (Kandziora-Ciupa et al. 2013). In this respect, increasing the soluble protein contents was correlated with Zn concentration in Melia azedarach (Doganlar and Atmaca 2011), Pb concentrations in wheat and lentil (Mesmar and Jaber 1991), and Cd concentration in Oryza sativa (Singh et al. 2006).
The treatment with heavy metals can lead to the interruption of activities of several essential enzymes, various aspects of the photosynthetic processes, and water content of cells (Kabir et al. 2010; Verma and Dubey 2003). Heavy metal at high concentrations is a major factor that enhances membrane damage and cell structures (Ekmekçi et al. 2008), which in turn could explain a higher value of EC (Table 1). Deleterious effects of Cu (Nicholls and Mal 2003; Demirevska-kepova et al. 2004), Cd (Hatata and Abdel-aal 2008), and Pb (Kabir et al. 2010) on membrane permeability have been reported. Hatata and Abdel-aal (2008) reported that Cd treatment caused a declined in the efflux of sodium (Na), potassium (K), calcium (Ca), magnesium (Mg), and Fe from roots and leaves of plants. In this respect, Nicholls and Mal (2003) stated that Cu could damage the permeability of plasma membranes, which can lead to the leakage of potassium ions and other substances dissolved in the cell. These damaging membrane effects could explain, in part, the reduction of water content in sewage-treated plants affecting cellular turgor; it seems that such damages could be mitigated and repaired by antioxidative enzymes like CAT, POD, and PPO and after that increasing cell enlargement and plant growth. To impose environmental stresses, it is a general strategy to overcome oxidative stress (Verma and Dubey 2003).
The accumulation of proline and total soluble sugars is a well-known biochemical response in plant to HMs-induced stress conditions (Moghaieb et al. 2004; Li et al. 2013). The Cd may bind to the carbohydrates of cell walls and the thiol groups of enzymes that exchanges metal ion concentrations such as Mg, Zn, and Fe (Iannelli et al. 2002). Proline increases the tolerance of plants to heavy metals through metal chelation in the cytoplasm (Zengin and Kirbag 2007), scavenging hydroxyl radicals (Xing et al. 2010), inhibit lipid peroxidation (Li et al. 2013), maintaining the water balance (Mallick 2004), and the protection and stabilization of protein synthesis (El-Amier et al. 2019). According to Tripathi and Gaur (2004), proline reduced the uptake of Cu by plant cells, and probably stabilized the plasma membrane, thus mitigating the toxic effects of Cu to the plant. Induction or activation of proline biosynthesis enzymes along with decreasing proline oxidation under stress conditions may be caused by decreased utilization of proline in protein synthesis and enhanced protein turnover (Li et al. 2013; Xing et al. 2010). Protein degradation contributes to Ni- or Cu-induced proline accumulation (Lin and Kao 2007). Mehta and Gaur (1999) reported that the hierarchy of metal toxicity to Chlorella vulgaris was Cu > Cr > Ni > Zn. They suggested that the intracellular concentration of proline followed by a slow and steady increase with increasing concentration of Zn, Cu andchromium (Cr). The increase in proline in plants may be due to a decrease in the electron transport system activity leading to the accumulation of NADH and H+ (Sharma et al. 2010). Proline accumulation is related to the activation of the gene encoding the OAT in response to stress (ornithine d-aminotransferase) (Thompson et al. 1997), which is catalyzed by the PDH (proline dehydrogenase) in plant cells (Kavi Kishor et al. 2005), and it may be caused by diminished consumption of proline in protein synthesis and higher protein turnover (Delauney and Verma 1993). A previous investigation suggests that the OAT activity, rather than the PDH activity, is associated with the proline accumulation in Ni- (Lin and Kao 2007), Cu- (Ku et al. 2011), and Zn-treated (Li et al. 2013) plants.
Exposure to HMs has been shown to induce the displacement of nutritional elements (Rocha et al. 2014) and oxidative stress in plant cells due to the enhancement in ROS production, such as singlet oxygen (1O2), hydroxyl radical (HO•), hydrogen peroxide (H2O2), and superoxide radical (\( {\mathrm{O}}_2^{-} \)). Increased enzyme activity, as observed in our studies, was either due to the protective measure adopted by Salicornia plants against oxidative damage and/or could be an increased production of ROS (Yan et al. 2008). The change in the activity of antioxidative enzymes is depended on the plant species, age, and duration of the stress (Demirevska-kepova et al. 2004). At the reproductive stage, the activity of the CAT that was approximately four-fold increases than the controls (Fig. 3A). It can be explained by an increase in its substrate to maintain the level of H2O2 as an adaptive mechanism of the plants (Shu et al. 2012) and was related to increased synthesis of glucocorticoid receptor (GR protein) after Cd exposure (Hatata and Abdel-aal 2008). The increase in CAT activity due to Fe (Xing et al. 2010), Pb (Shu et al. 2012), Cd (Singh et al. 2012), Ni, and Cd (Pandey and Sharma 2002) stresses has been observed in other plant species. Higher activities of enzymes in Zn and Fe supplemented wastewater-irrigated plants indicate the specific role of Zn-Fe in promoting ROS detoxification through CAT and PPO enzymes. Cadmium toxicity is moderated trough Zn antioxidant properties (Aravind and Prasad 2005). Induction in peroxidase activity, as stress enzymes (Verma and Dubey 2003), has been documented under toxic levels of Pb, Cu, Cd, Zn, and Fe (John et al. 2009; Jucoski et al. 2013; Shu et al. 2012). Enhancement in the enzyme activity of CAT and PPO suggests that these enzymes serve as an essential defense tool to resist heavy metal–induced oxidative damage in Salicornia plants. Therefore, this also indicates that Salicornia may be more efficient in avoiding damage from heavy metals (Shu et al. 2012). So, under metal toxicity, the level of peroxidase activity has been used as the potential biomarker to evaluate the intensity of stress (Verma and Dubey 2003). The effects on CAT and PPO were similar. CAT eliminates H2O2 by breaking it down directly to form water and oxygen, however, less efficient than POD in H2O2 scavenging because of its low substrate affinity. Therefore, as long as the stress is not too intense for the plant’s defense capacity, the main response to heavy metals is an increase in POD activity (Zhang et al. 2007). On the contrary, the non-significant increase in POD activity in this study might be due to the increasing rate of ROS scavenging by other antioxidative enzymes (CAT, PPO) and the increased H2O2 due to the inactivation of POD. The binding of Cd metal with thiol groups could inactivate these enzymes (Hatata and Abdel-aal 2008). Delay in the elimination of H2O2 and toxic peroxides is mediated by POD and in turn an enhancement in the free radical-mediated lipid peroxidation under Pb toxicity (Verma and Dubey 2003). Enhancement of POD activity under Ni stress was explained by its role in building up a physical barrier against toxic metals entering the cell (Hegedüs et al. 2001) as well as in scavenging H2O2 (Yan et al. 2008). Therefore, PODs serve as a parameter of metabolism activity against metal toxicity.
This study suggested that heavy metals could disturb the plant-water relationships in Salicornia europaea at flowering stage. When proline and soluble sugar contents in the plant leaf reached their highest level, the water content of leaves decreased by 5.12% compared with the control. According to Pedro et al. (2013), Cd decreases the turgor potential and cell wall elasticity, which might result in a smaller of leaf cells with smaller intercellular spaces (Ghnaya et al. 2005). The reduction of xylem vessel area can lead to the reduction of water flow to the leaves (Hajihashemi et al. 2020). The decrease of weight observed at reproductive stage may support the depressive action of Cd on cellular turgor.
5 Conclusions
Based on these results, the tolerance of Salicornia europaea to heavy metal stress relies on physio-chemical mechanisms. Increasing heavy metal exposure at the reproductive stage significantly increases the development of plant biomass of plants, which experience destruction of photosynthetic apparatus. Additionally, the accumulation of total soluble protein, proline, and soluble sugar was also observed at the reproductive stage. The increased activities of antioxidant enzymes and proline may be attributed to the adaptive defense system of S. europaea against the toxic effect imposed by cadmium, copper, nickel, and lead. However, the protection of Salicornia against reactive oxygen species was insufficient, especially at the reproductive stage. The level of electrical conductivity and relative water content explained the greater tolerance of plants to heavy metal stress at the flowering than the reproductive stage. In S. europaea, catalase activity was much higher indicating that it is the most important reactive oxygen species (ROS)-scavenging enzyme. It can be concluded that proper management of wastewater irrigation and quality parameters is required to ensure the successful and long-term use of wastewater for irrigation. These plants grown in sewage-irrigated soils would be an environmentally friendly solution for disposal problems and effectively reduce food chain contamination and risks to human health.
Since roots are the first organ for the hyperaccumulation of different heavy metal ions, therefore, the changes of enzyme activities and metal accumulation in root system need to be determined, and intermittent use of clean water between wastewater irrigation will not only reduce the heavy metal load but could also enhance the soil fertility.
References
Adamski JM, Danieloski R, Deuner S, Braga EJB, de Castro LAS, Peters JA (2012) Responses to excess iron in sweet potato: impacts on growth, enzyme activities, mineral concentrations, and anatomy. Acta Physiol Plant 34:1827–1836. https://doi.org/10.1007/s11738-012-0981-3
Ahmed DA, Slima DF (2018) Heavy metal accumulation by Corchorus olitorius L. irrigated with wastewater. Environ Sci Pollut Res Int 25:14996–15005. https://doi.org/10.1007/s11356-018-1675-1
An YJ, Kim YM, Kwon TI, Jeong SW (2004) Combined effect of copper, cadmium, and lead upon Cucumis sativus growth and bioaccumulation. Sci Total Environ 326:85–93. https://doi.org/10.1016/j.scitotenv.2004.01.002
Andosch A, Affenzeller MJ, Lütz C, Lütz-meindl U (2012) A freshwater green alga under cadmium stress: ameliorating calcium effects on ultrastructure and photosynthesis in the unicellular model Micrasterias. J Plant Physiol 169:1489–1500. https://doi.org/10.1016/j.jplph.2012.06.002
Aravind P, Prasad MN (2005) Modulation of cadmium-induced oxidative stress in Ceratophyllum demersum by zinc involves ascorbate– glutathione cycle and glutathione metabolism. Plant Physiol Biochem 43:107–116. https://doi.org/10.1016/j.plaphy.2005.01.002
Arnon DI (1949) Copper enzymes in isolated chloroplast polyphenol oxidase in Beta vulgaris. Plant Physiol 24:1–15
Babaei K, Sharifi RS, Pirzad A, Khalilzadeh R (2017) Effects of bio fertilizer and nano zn-fe oxide on physiological traits, antioxidant enzymes activity and yield of wheat (Triticum aestivum L.) under salinity stress. J Plant Interact 12:381–389. https://doi.org/10.1080/17429145.2017.1371798
Barakat MA (2011) New trends in removing heavy metals from industrial wastewater. Arab J Chem 4:361–377. https://doi.org/10.1016/j.arabjc.2010.07.019
Bates I, Waldren R, Teare J (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207
Bazihizina N, Colzi I, Giorni E, Mancuso S, Gonnelli C (2015) Photosynthesizing on metal excess: Copper differently induced changes in various photosynthetic parameters in copper tolerant and sensitive Silene paradoxa L. populations. Plant Sci 232:67–76
Benami A, Ofen A (1984) Irrigation engineering: sprinkle, trickle, surface irrigation, principles, design and agricultural practices. Engineering Scientific Publications
Bibi M, Hussain M (2005) Effect of copper and lead on photosynthesis and plant pigments in black gram [Vigna mungo (L.) Hepper]. Bull Environ Contam Toxicol 74:1126–1133. https://doi.org/10.1007/s00128-005-0698-8
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Cortes-esquivel JA, Giácoman-vallejos G, Barceló-quintal ID (2012) Heavy metals removal from swine wastewater using constructed wetlands with horizontal sub-surface flow. J Environ Prot 3:871–877. https://doi.org/10.4236/jep.2012.328102
Delauney AJ, Verma DPS (1993) Proline biosynthesis and osmoregulation in plants. Plant J 4(2):215–223. https://doi.org/10.1046/j.1365-313X.1993.04020215.x
Demirevska-kepova K, Simova-stoilova L, Stoyanova Z et al (2004) Biochemical changes in barley plants after excessive supply of copper and manganese. Environ Exp Bot 52:253–266. https://doi.org/10.1016/j.envexpbot.2004.02.004
Doganlar ZB, Atmaca M (2011) Influence of airborne pollution on Cd, Zn, Pb, Cu, and Al accumulation and physiological parameters of plant leaves in Antakya (Turkey). Water Air Soil Pollut 214:509–523. https://doi.org/10.1007/s11270-010-0442-9
Drazkiewicz M, Baszyński T (2005) Growth parameters and photosynthetic pigments in leaf segments of Zea mays exposed to cadmium, as related to protection mechanisms. J Plant Physiol 162:1013–1021. https://doi.org/10.1016/j.jplph.2004.10.010
Dubois M, Gilles K, Hamilton I et al (1956) Colorimetric method for determination of sugars and related substances. Chem Anal 28:350–356
Ebadi A, Azam N, Sima K et al (2018) Remediation of saline soils contaminated with crude oil using the halophyte Salicornia persica in conjunction with hydrocarbon- degrading bacteria. J Environ Manag 219:260–268. https://doi.org/10.1016/j.jenvman.2018.04.115
Eid EM (2019) Sewage sludge application enhances the growth of corchorus olitorius plants and provides a sustainable practice for nutrient recirculation in agricultural soils. J Soil Sci Plant Nutr 20:149–159. https://doi.org/10.1007/s42729-019-00113-z
Eissa MA, Negim OE (2018) Heavy metals uptake and translocation by lettuce and spinach grown on a metal-contaminated soil. J Soil Sci Plant Nutr 18:1097–1107. https://doi.org/10.4067/S0718-95162018005003101
Ekmekçi Y, Tanyolaç D, Ayhan B (2008) Effects of cadmium on antioxidant enzyme and photosynthetic activities in leaves of two maize cultivars. J Plant Physiol 165:600–611. https://doi.org/10.1016/j.jplph.2007.01.017
El-Amier Y, Elhindi K, El-Hendawy S et al (2019) Antioxidant system and biomolecules alteration in Pisum sativum under heavy metal stress and possible alleviation by 5-aminolevulinic acid. Molecules 24. https://doi.org/10.3390/molecules24224194
Eltaher GT, Ahmed DA, El-Beheiry M, Sharaf El-Din A (2019) Biomass estimation and heavy metal accumulation by Pluchea dioscoridis (L.) DC. in the Middle Nile Delta, (Egypt): perspectives for phytoremediation. S Afr J Bot 127:153–166. https://doi.org/10.1016/j.sajb.2019.08.053
Esmaeilzadeh M, Karbassi A, Moattar F (2016) Heavy metals in sediments and their bioaccumulation in Phragmites australis in the Anzali wetland of Iran. Chin J Oceanol Limnol 34:810–820. https://doi.org/10.1007/s00343-016-5128-8
Esmaeilzadeh M, Karbassi A, Bastami K (2017) Antioxidant response to metal pollution in Phragmites australis from Anzali wetland. Mar Pollut Bull 119:376–380. https://doi.org/10.1016/j.marpolbul.2017.03.030
Farid M, Shakoor MB, Ehsan S et al (2013) Morphological, physiological and biochemical responses of different plant species to Cd stress. Int J Chem Biochem Sci 3:53.60
Fidalgo F, Azenha M, Silva AF, de Sousa A, Santiago A, Ferraz P, Teixeira J (2013) Copper-induced stress in Solanum nigrum L. and antioxidant defense system responses. Food Energy Secur 2:70–80. https://doi.org/10.1002/fes3.20
Ghnaya T, Nouairi I, Slama I, Messedi D, Grignon C, Abdelly C, Ghorbel MH (2005) Cadmium effects on growth and mineral nutrition of two halophytes: Sesuvium portulacastrum and Mesembryanthemum crystallinum. J Plant Physiol 162:1133–1140. https://doi.org/10.1016/j.jplph.2004.11.011
Hajihashemi S, Mbarki S, Skalicky M, Noedoost F, Raeisi M, Brestic M (2020) Effect of wastewater irrigation on photosynthesis, growth, and anatomical features of two wheat cultivars (Triticum aestivum L.). Water 12:1–16. https://doi.org/10.3390/w12020607
Hatata MM, Abdel-Aal EA (2008) Oxidative stress and antioxidant defense mechanisms in response to cadmium treatments. Am-Euras J Agric Environ Sci 4:655–669
Hegedüs A, Erdei S, Horváth G (2001) Comparative studies of H2O2 detoxifying enzymes in green and greening barley seedlings under cadmium stress. Plant Sci 160:1085–1093. https://doi.org/10.1016/S0168-9452(01)00330-2
Iannelli MA, Pietrini F, Fiore L, Petrilli L, Massacci A (2002) Antioxidant response to cadmium in Phragmites australis plants. Plant Physiol Biochem 40:977–982. https://doi.org/10.1016/S0981-9428(02)01455-9
Jodeh S, Alkowni R, Hamed R, Samhan S (2015) The study of electrolyte leakage from barley (Hordeum vulgare L) and Pearl millet using plant growth promotion (PGPR) and reverse osmosis. J Food Nutr Res 3:422–429. https://doi.org/10.12691/jfnr-3-7-3
John R, Ahmad P, Gadgil K, Sharma S (2009) Heavy metal toxicity: effect on plant growth , biochemical parameters and cadmium and lead-induced changes in lipid peroxidation , antioxidative enzymes and metal accumulation in Brassica juncea L. at three differen. Arch Agron Soil Sci 55(4):395–405. https://doi.org/10.1080/03650340802552395
Jucoski GO, Cambraia J, Ribeiro C et al (2013) Impact of iron toxicity on oxidative metabolism in young Eugenia uniflora L. plants. Acta Physiol Plant 35:1645–1657. https://doi.org/10.1007/s11738-012-1207-4
Kabir M, Iqbal MZ, Shafiq M, Farooqi ZR (2010) Effects of lead on seedling growth of Thespesia populnea L. Plant Soil Environ 56(4):194–199. https://doi.org/10.17221/147/2009-PSE
Kandziora-Ciupa M, Ciepał R, Nadgórska-Socha A, Barczyk G (2013) A comparative study of heavy metal accumulation and antioxidant responses in Vaccinium myrtillus L. leaves in polluted and non-polluted areas. Environ Sci Pollut Res 20:4920–4932. https://doi.org/10.1007/s11356-012-1461-4
Kar M, Mishra D (1976) Catalase, peroxidase, and Polyphenoloxidase activities during rice leaf senescence. Plant Physiol 57:315–319. https://doi.org/10.1104/pp.57.2.315
Kavi Kishor PB, Sangam S, Amrutha RN et al (2005) Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: its implications in plant growth and abiotic stress tolerance. Curr Sci 88:424–438
Keraita B, Jimenez B, Drechsel P (2008) Extent and implications of agricultural reuse of untreated, partly treated and diluted wastewater in developing countries. CAB Rev Perspect Agric Vet Sci Nutr Nat Resour 3. https://doi.org/10.1079/PAVSNNR20083058
Kranner I, Colville L (2011) Metals and seeds: biochemical and molecular implications and their significance for seed germination. Environ Exp Bot 72:93–105. https://doi.org/10.1016/j.envexpbot.2010.05.005
Ku HM, Tan CW, Su YS et al (2011) The effect of water deficit and excess copper on proline metabolism in Nicotiana benthamiana. Biol Plantarum 56(2):337–343. https://doi.org/10.1007/s10535-011-0098-3
Kumari A, Sheokand S, Pooja et al (2019) Halophyte growth and physiology under metal toxicity. In: ecophysiology, abiotic stress responses and utilization of halophytes. Springer Nature Singapore Pte Ltd., pp 83–113
Li X, Yang Y, Jia L, Chen H, Wei X (2013) Zinc-induced oxidative damage , antioxidant enzyme response and proline metabolism in roots and leaves of wheat plants. Ecotoxicol Environ Saf 89:150–157. https://doi.org/10.1016/j.ecoenv.2012.11.025
Li S, Yang W, Yang T, Chen Y, Ni W (2015) Effects of cadmium stress on leaf chlorophyll fluorescence and photosynthesis of Elsholtzia argyi — a cadmium accumulating plant. Int J Phytoremediation 17:85–92. https://doi.org/10.1080/15226514.2013.828020
Lin YC, Kao CH (2007) Proline accumulation induced by excess nickel in detached rice leaves. Biol Plant 51:351–354. https://doi.org/10.1007/s10535-007-0071-3
Liu W, Zhao J, Ouyang Z, So L (2005) Impacts of sewage irrigation on heavy metal distribution and contamination in Beijing, China. Environ Int 31:805–812. https://doi.org/10.1016/j.envint.2005.05.042
Mallick N (2004) Copper-induced oxidative stress in the chlorophycean microalga chlorella vulgaris: response of the antioxidant system. J Plant Physiol 161:591–597. https://doi.org/10.1078/0176-1617-01230
Mehta SK, Gaur JP (1999) Heavy metal-induced proline accumulation and its role in ameliorating metal toxicity in Chlorella vulgaris. New Phytol 143:253–259. https://doi.org/10.1046/j.1469-8137.1999.00447.x
Mesmar MN, Jaber K (1991) The toxic effect of lead on seed germination, growth, chlorophyll and protein contents of wheat and lens. Acta Biol Hung 42:331–344
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410. https://doi.org/10.1016/S1360-1385(02)02312-9
Moghaieb REA, Saneoka H, Fujita K (2004) Effect of salinity on osmotic adjustment, glycinebetaine accumulation and the betaine aldehyde dehydrogenase gene expression in two halophytic plants, Salicornia europaea and Suaeda maritima. Plant Sci 166:1345–1349. https://doi.org/10.1016/j.plantsci.2004.01.016
Mojid MA, Hossain ABMZ, Wyseure GCL (2019) Impacts of municipal wastewater on basic soil properties as evaluated by soil column leaching experiment in laboratory. J Soil Sci Plant Nutr:402–412. https://doi.org/10.1007/s42729-019-00042-x
Moray C, Goolsby EW, Bromham L (2015) The phylogenetic association between salt tolerance and heavy metal hyperaccumulation in angiosperms. Evol Biol 43:119–130. https://doi.org/10.1007/s11692-015-9355-2
Mukhopadhyay M, Das A, Subba P, Bantawa P, Sarkar B, Ghosh P, Mondal TK (2013) Structural, physiological, and biochemical profiling of tea plants under zinc stress. Biol Plant 57:474–480. https://doi.org/10.1007/s10535-012-0300-2
Nicholls AM, Mal TK (2003) Effects of lead and copper exposure on growth of an invasive weed, Lythrum salicaria L. (Purple Loosestrife). Ohio J Sci 103:129–133
Ozawa T, Miura M, Fukuda M, Kakuta S (2009) Cadmium tolerance and accumulation in a halophyte Salicornia europaea as a new candidate for phytoremediation of saline soils. Sci Rep Grad Sch Life Envi Sci Osaka Pref Univ 60:1–8
Pandey N, Sharma CP (2002) Effect of heavy metals Co2+, Ni2+ and Cd2+ on growth and metabolism of cabbage. Plant Sci 163:753–758
Pedro CA, Santos MSS, Ferreira SMF, Gonçalves SC (2013) The influence of cadmium contamination and salinity on the survival, growth and phytoremediation capacity of the saltmarsh plant Salicornia ramosissima. Mar Environ Res 92:197–205. https://doi.org/10.1016/j.marenvres.2013.09.018
Perez-Romero JA, Redondo-Gomez S, Mateos-Naranjo E (2016) Growth and photosynthetic limitation analysis of the cd-accumulator Salicornia ramosissima under excessive cadmium concentrations and optimum salinity conditions. Plant Physiol Biochem 109:103–113. https://doi.org/10.1016/j.plaphy.2016.09.011
Piernik A, Hrynkiewicz K, Wojciechowska A, Szymańska S, Lis MI, Muscolo A (2017) Effect of halotolerant endophytic bacteria isolated from Salicornia europaea L. on the growth of fodder beet (Beta vulgaris L.) under salt stress. Arch Agron Soil Sci 63:1404–1418. https://doi.org/10.1080/03650340.2017.1286329
Qadir M, Wichelns D, Raschid-sally L et al (2010) The challenges of wastewater irrigation in developing countries. Agric Water Manag 97:561–568. https://doi.org/10.1016/j.agwat.2008.11.004
Rocha ACS, Almeida CMR, Basto MCP, Vasconcelos MTSD (2014) Antioxidant response of Phragmites australis to Cu and Cd contamination. Ecotoxicol Environ Saf 109:152–160. https://doi.org/10.1016/j.ecoenv.2014.06.027
Rozema J, Schat H (2013) Salt tolerance of halophytes, research questions reviewed in the perspective of saline agriculture. Environ Exp Bot 92:83–95. https://doi.org/10.1016/j.envexpbot.2012.08.004
Seregin IV, Ivanov VB (2001) Physiological aspects of cadmium and lead toxic effects on higher plants. Russ J Plant Physiol 48:523–544
Seyed Sharifi R, Khalilzadeh R, Jalilian J (2017) Effects of biofertilizers and cycocel on some physiological and biochemical traits of wheat (Triticum aestivum L.) under salinity stress. Arch Agron Soil Sci 63:308–318. https://doi.org/10.1080/03650340.2016.1207242
Sharma SS, Dietz KJ (2006) The significance of amino acids and amino acid-derived molecules in plant responses and adaptation to heavy metal stress. J Exp Bot 57:711–726. https://doi.org/10.1093/jxb/erj073
Sharma A, Gontia I, Agarwal PK, Jha B (2010) Accumulation of heavy metals and its biochemical responses in Salicornia brachiata, an extreme halophyte. Mar Biol Res 6:37–41. https://doi.org/10.1080/17451000903434064
Sharma A, Gontia-mishra I, Srivastava AK (2011) Toxicity of heavy metals on germination and seedling growth of salicornia brachiata. J Phytol 3:33–36
Sharma A, Sidhu GPS, Araniti F, Bali AS, Shahzad B, Tripathi DK, Brestic M, Skalicky M, Landi M (2020) The role of salicylic acid in plants exposed to heavy metals. Molecules 25:1–22. https://doi.org/10.3390/molecules25030540
Shu X, Yin L, Zhang QF, Wang W (2012) Effect of Pb toxicity on leaf growth, antioxidant enzyme activities, and photosynthesis in cuttings and seedlings of Jatropha curcas L. Environ Sci Pollut Res 19:893–902. https://doi.org/10.1007/s11356-011-0625-y
Singh S, Kumar M (2006) Heavy metal load of soil,water and vegetables in peri-urban Delhi. Environ Monit Assess 120:79–91. https://doi.org/10.1007/s10661-005-9050-3
Singh S, Eapen S, D’Souza SF (2006) Cadmium accumulation and its influence on lipid peroxidation and antioxidative system in an aquatic plant, Bacopa monnieri L. Chemosphere 62:233–246. https://doi.org/10.1016/j.chemosphere.2005.05.017
Singh SS, Khan NA, Tuteja N (2012) Plant Science Cadmium at high dose perturbs growth , photosynthesis and nitrogen metabolism while at low dose it up regulates sulfur assimilation and antioxidant machinery in garden cress (Lepidium sativum L.). Plant Sci 182:112–120. https://doi.org/10.1016/j.plantsci.2011.04.018
Sirianuntapiboon S, Kongchum M (2006) Effects of hydraulic retention time and media of constructed wetland for treatment of domestic wastewater. Afr J Agric Res 1:027–037
Smillie C (2015) Salicornia spp. as a biomonitor of Cu and Zn in salt marsh sediments. Ecol Indic 56:70–78. https://doi.org/10.1016/j.ecolind.2015.03.010
Song WY, Yang HC, Shao HB, Zheng AZ, Brestic M (2014) The alleviative effects of salicylic acid on the activities of catalase and superoxide dismutase in malting barley (Hordeum uhulgare L.) seedling leaves stressed by heavy metals. Clean - Soil Air Water 42:88–97. https://doi.org/10.1002/clen.201200310
Sytar O, Kumari P, Yadav S et al (2018) Phytohormone priming: regulator for heavy metal stress in plants. J Plant Growth Regul. https://doi.org/10.1007/s00344-018-9886-8
Tambussi EA, Nogués S, Araus JL (2005) Ear of durum wheat under water stress: water relations and photosynthetic metabolism. Planta 221:446–458. https://doi.org/10.1007/s00425-004-1455-7
Thao NP, Khan MIR, Thu NBA, Hoang XLT, Asgher M, Khan NA, Tran LSP (2015) Role of ethylene and its cross talk with other signaling molecules in plant responses to heavy metal stress. Plant Physiol 169:73–84. https://doi.org/10.1104/pp.15.00663
Thompson WF, Beven AF, Wells B, Shaw PJ (1997) Sites of rDNA transcription are widely dispersed through the nucleolus in Pisum sativum and can comprise single genes. Plant J 12:571–581. https://doi.org/10.1111/j.0960-7412.1997.00571.x
Tripathi BN, Gaur JP (2004) Relationship between copper- and zinc-induced oxidative stress and proline accumulation in Scenedesmus sp. Planta 219:397–404. https://doi.org/10.1007/s00425-004-1237-2
Verma S, Dubey RS (2003) Lead toxicity induces lipid peroxidation and alters the acti vities of antioxidant enzymes in growing rice plants. Plant Sci 164:645–655. https://doi.org/10.1016/S0168-9452(03)00022-0
Wang HL, Tian CY, Jiang L, Wang L (2014) Remediation of heavy metals contaminated saline soils: a halophyte choice? Environ Sci Technol 48:21–22. https://doi.org/10.1021/es405052j
Wu M, Luo Q, Zhao Y, Long Y, Liu S, Pan Y (2017) Physiological and biochemical mechanisms preventing Cd toxicity in the new hyperaccumulator Abelmoschus manihot. J Plant Growth Regul 37:709–718. https://doi.org/10.1007/s00344-017-9765-8
Xing W, Li D, Liu G (2010) Antioxidative responses of Elodea nuttallii (Planch.) H. St. John to short-term iron exposure. Plant Physiol Biochem 48:873–878. https://doi.org/10.1016/j.plaphy.2010.08.006
Yan R, Gao S, Yang W, Cao M, Wang S, Chen F (2008) Nickel toxicity induced antioxidant enzyme and phenylalanine ammonia-lyase activities in Jatropha curcas L. cotyledons. Plant Soil Environ 54(7):294–300
Yang CW, Zheng SS, Huang HL et al (2012) Comparison of osmotic adjustment and ion balance strategies in nineteen alkali-tolerant halophyte species during adaptation to salt-alkalinized habitats in northeast China. Aust J Crop Sci 6:141–148
Zare-Maivan H, Daviran M, Ghanati F, Sharifi M (2015) Salicornia europaea L. maintains its physiological integrity under high salinity stress in retreated sections of the Urmia Lake, Iran. J Persian Gulf 6:79–89
Zengin FK, Kirbag S (2007) Effects of copper on chlorophyll, proline, protein and abscisic acid level of sunflower (Helianthus annuus L.) seedlings. J Environ Biol 28:561–566
Zhang FQ, Wang YS, Lou ZP, Dong JD (2007) Effect of heavy metal stress on antioxidative enzymes and lipid peroxidation in leaves and roots of two mangrove plant seedlings (Kandelia candel and Bruguiera gymnorrhiza). Chemosphere 67:44–50. https://doi.org/10.1016/j.chemosphere.2006.10.007
Zhou W, Qiu B (2018) Effects of cadmium hyperaccumulation on physiological characteristics of effects of cadmium hyperaccumulation on physiological characteristics of Sedum alfredii Hance (Crassulaceae). Plant Sci 169:737–745. https://doi.org/10.1016/j.plantsci.2005.05.030
Acknowledgments
We thank “Iran’s National Elites Foundation (INEF)” for their help during the course of experimentation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khalilzadeh, R., Pirzad, A., Sepehr, E. et al. Long-Term Effect of Heavy Metal–Polluted Wastewater Irrigation on Physiological and Ecological Parameters of Salicornia europaea L.. J Soil Sci Plant Nutr 20, 1574–1587 (2020). https://doi.org/10.1007/s42729-020-00299-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-020-00299-7