Abstract
Anthropic activities usually affect the diversity, structure, and functionality of communities. The tropical rain forests from Chocó Biogeographic in Colombia are one of the world's biodiversity hotspots. However, very few studies evaluate the impact of anthropic activities on areas surrounding human populations. The structure of dung beetle assemblages was studied to compare a forest exposed to hunting activities (FH) and a forest intervened by cutting and thinning trees (FC). Two samplings were carried out in March and October 2009 using 64 pitfall traps distributed in four transects. We captured 678 individuals belonging to 18 species and nine genera. FH had 227 individuals and 12 species, and FC had 451 individuals and 16 species. The diversity index did not show statistical differences in the dung beetle assemblages between forests. Also, diversity profiles did not present differences between forests by richness (q0) and abundance (q1 and q2). Both forests shared the same species (ANOSIM: p < 0.01), while FH showed a lower dissimilarity in the species composition (ANOVA: F = 9.83). FH showed an absence of several species and the loss of some functional traits. FC has three times more roller beetles and almost twice as many small-medium tunneller beetles than FH, while there was a critical absence of large species in both forests. In conclusion, our results showed that hunting and cutting disturbances in these forests could similarly impact dung beetles' diversity and functional structure. However, it is important to continue evaluating the anthropogenic effect on nearby forests to establish conservation strategies for dung beetles.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biodiversity is affected negatively by anthropic disturbances such as agricultural expansion, livestock, or mining frontiers, generating a reduction in species richness, abundance, biomass, functional groups, and their composition (Braga et al. 2013; Kenyon et al. 2016; Cardoso et al. 2020). The Chocó Biogeographic in Colombia is one of the most important hotspots worldwide, with the highest species richness per square meter, making it relevant to conservation measures and policies (Myers et al. 2000; Jha and Bawa 2006; Hrdina and Romporti 2017). Chocó has a high affinity with Central America, sharing several species and becoming a biological corridor into South America (Hernández et al. 1992). Still, Chocó is one of the most under-sampled areas with very few published works (Neita et al. 2003; Pardo-Locarno 2007; Arias-Buriticá et al. 2011; Neita-Moreno 2011; Neita and Escobar 2012; Davies et al. 2020).
Chocó Biogeographic has been affected by anthropic disturbance for a long time, but the hotspot has few evaluations of the impact of the anthropic disturbances on its fauna and flora assemblages (Mittermeier et al. 2011; Davies et al. 2020). Forest areas have two critical disturbances: i) cutting down and thinning of trees, and ii) hunting of large mammals (Nichols et al. 2007; Andresen and Laurence 2007; Gardner et al. 2008; Culot et al. 2013; França et al. 2018). Deforestation affects the function and structure of the ecosystems and causes the loss of their services (Mosquera-Andrade 2014; Noriega et al. 2021). Chocó lost 29.951 hectares of forest at a loss rate of 0,003% per year between 1990 and 2010 (González et al. 2011). In 20 years, it went from having 3.412.441 hectares to 3.382.490 hectares. Then, Chocó has the largest deforested area corresponding to 18% of the national total. For example, Acandí, a municipality in the Chocó area, lost 1.240 hectares of humid primary forest cover from 2000 to 2019, equivalent to a 2,9% decrease in tree cover (Global Forest Watch 2020). Likewise, hunting large mammals could have a negative effect in food webs and their ecosystems because mammal disappearance could change the abundance and distribution of other species (Barbar and Lambertucci 2018).
One of the most relevant insect groups to evaluate the ecological services and human impacts is the dung beetles (Coleoptera: Scarabaeidae: Scarabaeinae), which feed mainly on vertebrate excrements (omnivores and herbivores) and play a crucial role in ecosystems as recyclers of manure, nutrient cycling processes, secondary dispersal of seeds, control of fly and nematode populations, and improving soil conditions (Nichols et al. 2007; Slade et al. 2011; Nervo et al. 2017; Noriega et al. 2021). Usually, dung beetles are used to carry out biodiversity, conservation, and environmental impact studies due to their well-known biology, presence in a wide variety of habitats, well-known taxonomy, short generational cycles, and high sensitivity to anthropogenic disturbances due to high specificity of habitat (Halffter and Favila 1993; McGeoch et al. 2002; Spector 2006; Nichols et al. 2007; Noriega et al. 2007; Nichols et al. 2009; Otavo et al. 2013; Manning et al. 2016). Globally, there are approximately 8000 species and 300 genera, while in Colombia, there are 305 species of 40 genera (Noriega et al. 2015; Schoolmeesters 2020). However, the state of knowledge of this group in Colombia is not complete, and several regions of the country need local and regional studies, such as the Chocó Biogeographic (Noriega et al. 2015).
The disturbance of forests affects the diversity of dung beetle, which could severely affect their ecological processes. Deforestation affects the canopy cover that regulates extreme temperatures and humidity conditions, which could modulate the flight of dung beetles and change the dung paths altering the beetle’s behaviour (Gómez et al. 2018). Likewise, the microclimatic conditions could affect the dung beetle activity because they are highly sensitive to physiological restrictions (Verdú and Lobo 2008; Dortel et al. 2013). For example, tree shade reduces excrement desiccation, keeping the interior fresh for long periods and thus improving its quality (Lobo et al. 1988; Gill 1991; Louzada and Carvalho e Silva 2009; Cardinale et al. 2012; Laurance et al. 2014). Another forest disturbance is hunting some mammals because the human population considers them dangerous for them or their subsistence resources (Rodríguez-Mahecha et al. 2008). The changes in their composition could affect the trophic guild of dung beetles because they are closely associated with these vertebrates by feeding on their faeces (Rodríguez-Mahecha et al. 2008).
Based on the above, we evaluate the anthropic disturbance effect on the taxonomic and functional diversity of dung beetle assemblages in the Chocó Biogeographic of Colombia. This study assessed patterns of taxonomic and functional diversity of dung beetles among two forests in Chocó, providing information on their diversity, functional groups, traits, and the anthropic perturbation impact on dung beetles. We compared a forest with cutting and thinning trees and a forest with hunting activities. We hypothesize that the cutting and thinning of trees disturbance affects the diversity and functional structure of dung beetles more than a forest with hunting activities. This first disturbance modifies the vegetation cover, niches, habitats, microclimates, and fauna diversity on a higher level compared to hunting activities.
Materials and methods
Study area
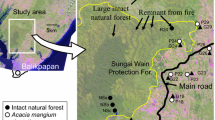
The fieldwork was carried out in Capurganá in the municipality of Acandí, department of Chocó, Colombia, located in the Gulf of Urabá near the Atrato River and the Caribbean Sea (8º37'N–77º21'W, 90 m a.s.l.; Fig. 1). Capurganá has a mean annual temperature of 27º C and 2296 mm of annual precipitation. The primary ecosystem is a tropical moist forest with bimodal weather (April and October are the wettest months) (García-Valencia 2007). Soil types are inceptisol, entisol, alfisol, and vertisol with clay-dominated texture, deep and well-drained (IGAC 2004). We selected two forests with different anthropic interventions: i) a forest in a regeneration process after an effect of cutting and thinning trees in the last 20 years (FC) and ii) a forest used as a source of illegal hunting activities (FH) to analyse the anthropic effect on dung beetle assemblages. We tried to sample a primary forest as a control in our design, but it was impossible to find a forest without any anthropic disturbance. All the forest in the region has suffered from anthropogenic disturbance processes at different scales, from ancestral hunting and thinning to total forest clearing.
Dung beetles sampling
Two samplings were carried out corresponding to March and October of 2009 to collect dung beetle species in dry (March) and wet (October) seasons. Each season, four parallel transects (separated by 150 m) were located (FC and FH). Transects (replicates) had four pitfalls installed 50 m from each other and baited with fresh human excrement (30 g) in each ecosystem to be the most effective resources to attract dung beetles in Colombia (Lobo et al. 1988; Gill 1991; Noriega and Fagua 2009; Bourg et al. 2016). Pitfall traps (64 total traps, sample unit) consisted of a plastic container (12 cm in diameter and depth) filled with a preservative solution (water, salt, and detergent) to prevent dung beetles from decomposing (Beiroz et al. 2018). After collecting the samples, this solution was changed by alcohol 75%. The traps remained active for 72 h. The collected specimens were identified at the species level using different taxonomic keys (Howden and Young 1981; Kohlmann 1997; Kohlmann and Solis 1997, 2001, 2006; González et al. 2009; Camero 2010; Vaz-de-Mello et al. 2011), the comparison with material from collections, and the help of experts in some groups. Specimens were deposited in the Pontificia Universidad Javeriana (Colombia)'s entomological collection with respective labels, mentioning the collection place, date, type of trap, and identified species.
Estimation of ecological and functional diversity
We performed sampling completeness analysis to evaluate the efficiency of the sampling effort and the representation of dung beetle assemblages in each forest (FH and FC). The analysis compared the sampling sites, considering the number of missing species, their average abundances, and detailed and “fair” inferences about the sampled assemblages (Moreno 2001). We carried out diversity profiles of order q, based on the Hill series, to describe and compare the taxonomic diversity of dung beetle between the hunting forest (FH) and the cutting forest (FC) (Hill 1973). Diversity profiles represent the number of species (q0, richness) and the number of equally common species weighted by their relative abundance (q1: rare and common species and q2: dominant species) (Moreno et al. 2011).
The functional diversity of dung beetles was estimated by combining different traits into functional groups related to several ecosystem services such as burial activity, secondary seed dispersal, and nutrient cycling. Those functional traits (Petchey et al. 2009) were the food relocation strategy (tunneller, roller, and dweller species) and the beetle size (small, medium, and large species) (Doube 1990). Food relocation strategies were obtained from previous studies (Halffter and Edmonds 1982; Doube 1990; Hanski and Cambefort 1991). The average beetle body size was measured as the total length from the external border of the clypeus to the pygidium for each species, and they were assigned to three size categories: small (< 10 mm), medium (10–18 mm), and large (> 18 mm).
Data analysis
Simpson (D) and Shannon–Wiener (H) indices were estimated in a matrix of species captured in each forest (FC and FH). The Simpson index established the probability that two individuals are drawn randomly from a sample to the same species, considering the best-represented species. The Shannon–Wiener index assumes that all species are in the samples and indicates the species distribution’s uniformity (evenness), number of species (richness), and the relative number of individuals (abundance) (Moreno et al. 2011; Majeed et al. 2020; Ramzan et al. 2021). Simpson and Shannon–Wiener indexes were compared through a chi-square test (X2). We used the BioDiversity Pro program for indices and EstimateS (Colwell 2013) to estimate each forest’s species accumulation curves.
The diversity profiles were compared through overlapping confidence intervals (5–95%) (Hill series: q0, q1 and q2). The C.hat and alpha diversity were estimated using the R-based interactive online version of the software iNEXT (Chao et al. 2016). Beta diversity components were evaluated using the indices βsim (Simpson dissimilarity–spatial turnover), βnes (nestedness dissimilarity), and βsor (Sorensen dissimilarity–total Beta diversity) performed within forest sites, employing pair.beta function of the betapart package (Baselga et al. 2018). The dissimilarity measures were additive fractions (βsor = βsim + βnes), and the indices values were compared through Anova and DGC (Di Rienzo, Guzmán, Casanoves). Non-metric multidimensional scaling analysis (NMDS) was used to explore the species composition, using the isoMDS function in the MASS package (Venables and Ripley 2002). We also use ANOSIM to test differences in species composition based on the NMDS analysis. Finally, the HDS.test function was used from the agricolae package for the posthoc comparisons because of controlled type I error (Di Rienzo et al. 2002; Mendiburu 2017).
Functional diversity was evaluated using the functional traits of dung beetles: i) grouping species into functional groups, and ii) comparing the total and proportional abundance of each functional trait. In the first case, a dissimilarity analysis was performed to group species into functional groups using the Gower distance with the gower.dist function of StatMatch package (D’Orazio 2019). This measure computes the distances between pairs of variables over two data sets and combines those distances into a single value. In the second case, the total abundance of each functional trait was compared by the Kruskal–Wallis test using the posthoc.kruskal.nemenyi from the PMCMR package (Pohlert 2014). The proportional abundance of functional traits of each forest (FH and FC) was compared using the Friedman test (friedman.test) from the stats package (R Core Team 2018). Statistical analyses were performed using R software (R Core Team 2018).
Results
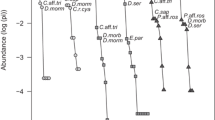
A total of 678 individuals belonging to 18 species and nine genera were collected (Table 1). The estimator of sampling coverage showed that sampling effort was enough to capture most of the species present in FH and FC forests (C. hat > 99%), which means that amount of pitfall was enough for the monitoring. The forest with hunting activities (FH) had 227 individuals distributed among seven genera and 12 species. The forest with cutting and thinning activities (FC) had 451 individuals distributed among nine genera and 16 species (Table 1). Shannon–Wiener index (J) and Simpson index (D) showed similar species diversity values in both forests (Shannon: FH = 0.72 and FC = 0.639 and Simpson: FH = 0.243 and FC = 0.239, Table 1). We did not find statistically significant differences by comparing these values through a chi-square test (X2). Dichotomius favi (Kohlmann and Solis 1997) and Sylvicanthon aequinoctialis (Harold 1868) were the most abundant species (n > 100) (Table 1). The similarity analysis showed close values with high equality between the species of FH and FC. The diversity profiles did not present any differences between forests in diversity measured by richness (q0) and abundance (q1 and q2). However, according to the upper confidence intervals, FC could maintain twice as many species (q0) as FH (Fig. 2). For example, FC showed a higher hypervolume compared to FH; both forests share the same species (ANOSIM: p < 0.01, Fig. 3A), and FH demonstrated a lower dissimilarity in the species composition with the Jaccard index (ANOVA: F = 9.83, p = 0.01, Fig. 3B). Finally, changes in both forests were probably for a higher species turnover (addition) than nestedness patterns (Fig. 3B).
Diversity profiles (and confidence intervals 95%) in two tropical moist forests (circles: FH, forest with hunting and triangles: FC, forest with cutting) of Chocó Biogeographic of Colombia. q0, species richness; q1 and q2, effective species numbers based on the relative abundance of rare and common species, and dominant species, respectively
a Changes in species composition based on a non-metric multidimensional model (NMDS). b Jaccard dissimilarity analysis of dung beetle assemblages in two tropical rain forests of the Chocó Biogeographic of Colombia. Circles: FH, forest with hunting and triangles: FC, forest with cutting. Dark grey bar: species turnover (βsim) and light grey bar: species nestedness (βnes). Anova and DGC posthoc comparisons. Different letters indicate statistical differences
Functional groups and trait diversity (paracoprids, telecoprids, endocoprids, small < 10 mm, medium 10 to 18 mm, large > 18 mm) identified five groups from 18 species associated with the tropical moist forest of Chocó Biogeographic of Colombia (Gower: cutline = 0.4) (Fig. 4A, B). FG1 - Deltochillum gibbosum (Fabricius 1775) and FG4 - Dichotomius satanas (Harold 1867) were composed by a single species (Fig. 4B). Three species constituted the FG2 and FG3, and the FG5 was compounded by half of the total species captured in this study (Fig. 4B). The results of the functional groups in FH showed an absence of several species and the loss of some functional traits (Fig. 4A). Mainly, the loss of rollers and large beetles was observed (D. gibbosum), implying the loss of FG1 (Fig. 4A). In addition, dwellers and medium beetles were also lost (Eurysternus foedus, Guérin-Méneville 1844) (Fig. 4A). The absence of two species (tunneller and small beetles) belonging to FG5 was observed in FC (Fig. 4B). Although we do not observe statistical differences in total abundances of functional traits between FH and FC forests (Table 2), there seems to be a tendency between them. FC has three times more roller beetles and almost twice as many small, medium, and tunneller beetles than the FH (Table S1). Also, there was a critical absence of large species in both forests (Table S1). We found twice as many tunneller beetles than roller species in FH (Fig. 5 A), while FC had almost the same proportion of both functional traits (Fig. 5B). Regarding body size, both forests had a higher proportional abundance of medium beetles compared with small and large species (Fig. 5).
Species composition and traits associated with the functional groups (FG) of dung beetle assemblages. a Forest with hunting (FH). b Forest with cutting (FC) of Chocó Biogeographic of Colombia. FG were built at the Gower distance of 0.4 (total distance = 0.8). Letters that compose the FG belong to the following functional traits: R: Rollers, T: Tunnellers, and D: Dwellers and S: Small, M: Medium, and L: Large. (*) Asterisks indicate the absence of a trait/functional species
Proportional abundance of the food relocation strategy and the body size of dung beetle communities. a Forest with hunting (FH). b Forest with cutting (FC) of the Chocó Biogeographic of Colombia. Black points represent extreme values. Non-parametric data: Friedman test and posthoc comparisons. Different letters indicate statistical differences
Discussion
Our study hypothesis was that the cutting and thinning of trees affect the diversity and functional structure of dung beetle communities more than the hunting activities. However, our results do not support the initial hypothesis because we observed a uniform tendency in dung beetles’ assemblages in both forests (FC-cutting and FH-hunting), which implies that both perturbations have a similar effect on the diversity and functional structure of dung beetle assemblages. Diversity estimators and profiles showed a similar diversity and a loss of functional traits and groups between forests, and beta diversity estimators showed a lower dissimilarity in the species composition within sites, observing a higher abundance and richness of dung beetles in FC and a higher abundance of tunneller, roller, and beetles of medium size in both forests. These results are similar to recent studies by Davies et al. (2020) and López-Bedoya et al. (2022), where taxonomic and functional diversity of dung beetle species are higher and could be recovered in secondary forests in comparison to open or highly perturbated habitats (e.g., pastures) in different regions around the world.
Disturbance pressures on dung beetles could explain the similar patterns in FH and FC. The decrease of mammals by hunting activities is mainly associated with a loss of medium-size mammals, which usually are the abundant species (Nichols et al. 2009; Feer and Boissier 2015; Guerra et al. 2019). Then, if the mammal populations are reduced by hunting, the food resources for dung beetles will decrease, unleashing an impact on their diversity (Culot et al. 2013). Also, a lower heterogeneity of available resources could imply a lower diversity and a decrease in the richness and abundance of dung beetle populations, seeming to be negatively correlated with this disturbance (Gardner et al. 2008; Da Silva and Hernández 2016; Gómez et al. 2018). Deforestation of the tree canopy by cutting activities could modify the entrance of light, temperature, and humidity, affecting the ecological role of dung beetles, which are susceptible to microclimatic changes because they affect their nests and food resource (Hanski and Cambefort 1991; Damborsky et al. 2015). For example, a lower humidity of the excrements and changes in soil conditions due to high evaporation and low humidity can reduce the availability of resources for the dung beetles (Holter and Scholtz 2007). Also, cutting activities changes critical factors in the air level, such as temperatures (high) and humidity (low), causing a limitation in the thermoregulation, a crucial process for dung beetles to fly and disperse (Verdú et al. 2006; Verdú and Lobo 2008; Dortel et al. 2013). Cutting activities cause tree coverage changes that affect the fauna species, which are the primary producers of dung beetle food supply (Bourg et al. 2016).
Our results showed a similar diversity pattern among FC and FH forests, despite our initial hypothesis that FH could have higher diversity and composition in dung beetle populations due to its more heterogeneity, niches, and richness in comparison with FC. Perhaps, the intermediate disturbance hypothesis and abundant food supply offer could explain the similar diversity between FH and FC forests (Correa et al. 2019). The intermediate disturbance hypothesis mentions that rare or frequent ecological disturbances could maximize an ecosystem's diversity (Connell 1978). Diversity could be highest when disturbances are intermediate in intensity or size and lower when perturbations are extreme. The secondary forest (FC) in Chocó in 2009 had not yet been intervened by an intense cutting of trees compared to the current annual deforestation rate of humid tropical forests (Achard et al. 2002; Global Forest Watch 2020). Then, intermediate disturbances could make it possible to maintain species richness and biodiversity levels similar to FH. Gómez-Cifuentes et al. (2017) observed high diversity of dung beetles in land with continuous anthropic activities (cutting and logging) compared to land with few anthropic events (pastures for livestock or old pine plantations).
Additionally, cutting activities in FC could be compensated through high offer resources to dung beetles because the FC is closer to the human population. An increase in resource supply over a long and constant time makes dung beetles adapt quickly to modified environments, explaining their high diversity in FC (Rodrigues et al. 2013; Correa et al. 2019). Humans are omnivorous, and this makes the excrement more attractive for dung beetles due to the high concentration of nitrogen and phosphorus, which are essential nutrients for immature adults to complete the development of their muscular system and for females to complete the maturation of their eggs (Hanski 1991; Gómez-Cifuentes et al. 2015). Also, an important part of the diet of dung beetles is made up of bacteria that are found in large quantities in the excrement of omnivores, which is essential for the metabolism of these insects (Filgueiras et al. 2009). Perhaps, for these reasons, FC has three times more roller beetles and almost twice as many small, medium, and tunneller beetles than FH.
The species composition evidenced a higher dominance in both forests, observing that D. favi represented 45% of FH captured species. In comparison, D. favi and S. aequinoctialis represented 65% of the individuals in FC. We suggest that the similarity in the composition between forests can be attributed to the proximity in space (< 2 km), which would be evidence of a strong influence of high spatial mobility in the region despite differences in the type of anthropic disturbance (Beiroz et al. 2018). In general, we observed a higher presence of tunnellers and small beetles in both forests, grouped in one functional group (FG5). In contrast, we observed two functional groups with one unique species and the absence of several species and traits in most FH functional groups, while FC was the opposite.
The disturbances on FH imply the absence of the unique roller and large species (D. gibbosum) and dweller and medium species (E. foedus). The lack of medium and large species had several adverse effects on the forest function because these species bury higher portions of organic matter in the soil compared to small species (Slade et al. 2007; Shahabuddin et al. 2008; Dangles et al. 2012). The absence of roller and dweller species probably can be explained by the higher presence of tunneller beetles and could be associated with a competition mechanism for overlapping niches (Hanski and Cambefort 1991; Simmons and Ridsdill-Smith 2011). Finally, the Phanaeini tribe's absence, medium, and large tunneller species, in both forests show their susceptibility to anthropogenic disturbance due to the lack of large mammal communities (Edmonds and Zídek 2010; Noriega et al. 2020).
Implications for dung beetles’ conservation
The anthropic pressure in FH with frequent hunting events could disappear large mammals and their faeces, affecting dung beetles by the absence of food resources. At the same time, cutting activities in FC could modify the vegetation cover structure, entrance of light, temperature, and humidity, affecting the ecological role of dung beetles. Although deforestation strongly impacts fauna and flora by destroying dung beetles' niches, habitats, and resources. The human communities and their domestic fauna near secondary forests could benefit from dung beetles' diversity in FC, which would provide a higher resource offer and explain the similarities between these two disturbance effects.
Conclusions
Hunting and cutting activities in the sampled forests of Chocó Biogeographic similarly influence dung beetles' diversity and functional structure. This study shows that, despite conserving the vegetation cover, if the mammals existing in a forest disappear, the diversity and structure of the assemblage of dung beetles would be as harmful as the effect of cutting and thinning trees. In this context, it becomes evident that it is necessary to conserve both the vegetation cover and the mammalian fauna to maintain taxonomic and functional diversity. In conclusion, cutting and hunting disturbances had similar impacts on dung beetle communities. However, it is important to continue evaluating the anthropogenic effect on nearby forests of unique regions like Chocó Biogeographic to establish conservation strategies for dung beetles, including undisturbed forests.
References
Achard F, Eva HD, Stibig H, Mayaux P, Gallego J, Richards T, Malingreau JP (2002) Determination of deforestation rates of the world’s humid tropical forests. Science 297:999–1002
Andresen E, Laurance SGW (2007) Possible indirect effects of mammal hunting on dung beetle assemblages in Panama. Biotropica 39:141–146
Arias-Buriticá JA, Delgado-Gómez P, González FA, Vaz-de-Mello FZ (2011) Nuevos registros de escarabajos coprófagos (Coleoptera: Scarabaeidae: Scarabaeinae) para el departamento de Chocó (Colombia). Acta Zool Mexic 27(3):875–878
Barbar F, Lambertucci SA (2018) The roles of leporid species that have been translocated: a review of their ecosystem effects as native and exotic species. Mammal Rev 48(4):245–260
Baselga A, Orme D, Villeger S, Bortoli J, Leprieur F (2018) betapart: Partitioning beta diversity into turnover and nestedness components. R package version 1.5.0. https://CRAN.R-project.org/package=betapart
Beiroz W, Sayer E, Slade SM, Audino L, Braga RF, Louzada J, Barlow J (2018) Spatial and temporal shifts in functional and taxonomic diversity of dung beetle in a human-modified tropical forest landscape. Ecol Indicat 95:518–526
Bourg A, Escobar F, MacGregor-Fors I, Moreno CE (2016) Got dung? Resource selection by dung beetles in neotropical forest fragments and cattle pastures. Neotrop Entomol 45:490–498
Braga RF, Korasaki V, Andresen E, Louzada J (2013) Dung beetle community and functions along a habitat-disturbance gradient in the Amazon: a rapid assessment of ecological functions associated to biodiversity. PLoS One 8:e5778
Camero E (2010) Los escarabajos del género Eurysternus Dalman, 1824 (Coleoptera: Scarabaeidae) de Colombia. Bol Soc Entomol Aragonesa 46:147–179
Cardinale BJ, Duffy JE, Gonzalez A, Hooper DU, Perrings C, Venali P, Narwani A, Mace GM, Tilman D, Wardle DA, Kinzig AP, Daily GC, Loreau M, Grace JB, Larigauderie A, Srivastava DS, Naeem S (2012) Biodiversity loss and its impact on humanity. Nature 486:59–67
Cardoso P, Barton PS, Birkhofer K, Chichorro F, Deacon C, Fartmann T et al (2020) Scientists’ warning to humanity on insect extinctions. Biol Conserv 242:108426
Chao A, Ma KH, Hsieh TC (2016) iNEXT (iNterpolation and EXTrapolation) Online: Software for Interpolation and Extrapolation of Species Diversity. Program and User’s Guide published
Colwell RK (2013) EstimateS, v 9.1: Statistical estimation of species richness and shared species from samples (software and user’s guide). http://viceroy.eeb.uconn.edu/EstimateS
Connell JH (1978) Diversity in tropical rain forests and coral reefs. Science 199(4335):1302–1310
Correa CM, Braga RF, Puker A, Korasaki V (2019) Patterns of taxonomic and functional diversity of dung beetles in a human-modified variegated landscape in Brazilian Cerrado. J Insect Conserv 23(1):89–99
Culot L, Bovy E, Vaz-de-Mello FZ, Guevara R, Galetti M (2013) Selective defaunation affects dung beetle communities in continuous Atlantic rainforest. Biol Conserv 163:79–89
D’Orazio M (2019) StatMatch: Statistical Matching or Data Fusion. R package version 1.3.0. https://cran.r-project.org/web/packages/StatMatch/StatMatch.pdf
Da Silva PG, Hernández MIM (2016) Spatial variation of dung beetle assemblages associated with forest structure in remnants of southern Brazilian Atlantic forest. Rev Bras Entomol 60:73–81
Damborsky MP, Bohle MA, Polesel MI, Porcel EA, Fontana JL (2015) Spatial and temporal variation of dung beetle assemblages in a fragmented landscape at eastern humid Chaco. Neotrop Entomol 44(1):30–39
Dangles O, Carpio C, Woodward G (2012) Size-dependent species removal impairs ecosystem functioning in a large-scale tropical field experiment. Ecology 93:2615–2625
Davies RW, Edwards DP, Edwards FA (2020) Secondary tropical forests recover dung beetle functional diversity and trait composition. Anim Conserv 23(5):617–627
Di Rienzo JA, Guzmán AW, Casanoves F (2002) A multiple-comparisons method based on the distribution of the root node distance of a binary tree. J Agricul Biol Environ Stat 7:129–142
Dortel E, Thuiller W, Lobo JM, Bohbot H, Lumaret JP, Jay-Robert P (2013) Potential effects of climate change on the distribution of Scarabaeidae dung beetles in Western Europe. J Insect Conserv 17:1059–1070
Doube B (1990) A functional classification for analysis of the structure of dung beetle assemblages. Ecol Entomol 15:371–383
Edmonds WD, Zídek J (2010) A taxonomic review of the neotropical genus Coprophanaeus Olsoufieff, 1924 (Coleoptera: Scarabaeidae, Scarabaeinae). Insecta Mundi 1–111
Feer F, Boissier O (2015) Variations in dung beetle assemblages across a gradient of hunting in a tropical forest. Ecol Indicat 57:164–170
Filgueiras B, Liberal C, Aguiar C, Medina CM, Iannuzzi L (2009) Attractivity of omnivore, carnivore and herbivore mammalian dung to Scarabaeinae (Coleóptera, Scarabaeidae) in a tropical atlantic rainforest remnant. Rev Bras Entomol 53:422–427
França F, Louzada J, Barlow J (2018) Selective logging effects on “brown world” faecal-detritus pathway in tropical forests: a case study from Amazonia using dung beetles. Forest Ecol Manag 410:136–143
García-Valencia C (2007) Atlas del Golfo de Urabá: una mirada al Caribe de Antioquia y Chocó. (S. de publicaciones especiales INVEMAR, Ed.). Santa Marta, Colombia: Instituto de investigaciones marinas - INVEMAR y Gobernación de Antioquia
Gardner TA, Hernández MIM, Barlow J, Peres CA (2008) Understanding the biodiversity consequences of habitat change: the value of secondary and plantation forests for neotropical dung beetles. J Appl Ecol 45:883–893
Gill BD (1991) Dung Beetles in Tropical American Forests. In Hanski y I. Cambefort Y (ed.), Dung Beetle Ecology. Princeton University Press, Princeton, NJ pp 211–229
Global Forest Watch (2020) Tree cover loss in [country/province name]. Accessed 19 Nov 2020 from: www.globalforestwatch.org
Gómez VCG, Verdú JR, Gómez-Cifuentes A, Vaz-de-Mello FZ, Zurita GA (2018) Influence of land use on the trophic niche overlap of dung beetles in the semideciduous Atlantic forest of Argentina. Insect Conserv Divers 11:554–564
Gómez-Cifuentes A, Gómez VG, Lozano AM, Zurita GA (2015) Estructura y composición de las comunidades de escarabajos estercoleros (Scarabaeidae: Scarabaeinae) en diferentes sistemas ganaderos del bosque atlántico de Argentina. Entomol Mexic 2:588–594
Gómez-Cifuentes A, Munevar A, Giménez VC, Gatti MG, Zurita GA (2017) Influence of land use on the taxonomic and functional diversity of dung beetles (Coleoptera: Scarabaeinae) in the southern Atlantic forest of Argentina. J Insect Conserv 21:147–156
González FA, Molano F, Medina CA (2009) Los subgéneros Calhyboma, Hybomidium y Telhyboma (Coleoptera: Scarabaeidae: Scarabaeinae: Deltochilum) en Colombia. Rev Colomb Entomol 35:253–274
González JJ, Etter A, Sarmiento A, Orrego S, Ramírez C, Cabrera E et al (2011) Análisis de tendencias y patrones espaciales de deforestación en Colombia. Bogotá: Instituto de Hidrología, meteorología y estudios ambientales (IDEAM)
Guerra CB, Zurita GA, Bellocq MI (2019) Livestock areas with canopy cover sustain dung beetle diversity in the humid subtropical Chocó forest. Insect Conserv Divers 12:296–308
Halffter G, Edmonds WD (1982) The nesting behavior of dung beetles (Scarabaeinae): an ecological and evolutive approach. Instituto de Ecología, México, D.F. México, p 177
Halffter G, Favila ME (1993) The Scarabaeinae (Insecta: Coleoptera) an animal group for analysing, inventorying and monitoring biodiversity in tropical rainforest and modified landscapes. Biol Internat 27:15–21
Hanski I (1991) The dung insect community. In: Hanski I, Camberfort Y (eds) Dung beetle ecology. Princeton, New Jersey, pp 5–21
Hanski I, Cambefort Y (1991) Dung beetle ecology. Princeton University Press, USA, p 514
Hernández J, Hurtado A, Ortiz R, Walschburger T (1992) Unidades biogeográficas de Colombia. En: Halffter G (ed) La Diversidad Biológica de Iberoamérica I. CYTED-B Programa iberoamericano de ciencia y tecnología para el desarrollo. Instituto de Ecología, A.C., Xalapa, México, pp 105–152
Hill M (1973) Diversity and evenness: a unifying notation and its consequences. Ecology 54:427–432
Holter P, Scholtz CH (2007) What do dung beetles eat? Ecol Entomol 32:690–697
Howden H, Young O (1981) Panamanian Scarabaeinae taxonomie distribution and habits. Contrib Am Entomol Inst 18:1–204
Hrdina A, Romporti D (2017) Evaluating global biodiversity hotspots – very rich and even more endangered. J Lands Ecol 10:108–115
IGAC (2004) Estudio general de suelos y zonificación de tierras. Departamento de Chocó
Jha S, Bawa KS (2006) Population growth, human development, and deforestation in biodiversity hotspots. Conserv Biol 20:906–912
Kenyon TM, Mayfield MM, Monteith GB, Menéndez R (2016) The effects of land use change on native dung beetle diversity and function in Australia’s wet tropics. Austral Ecol 41:797–808
Kohlmann B (1997) The Costa Rican species of Ateuchus (Coleoptera: Scarabaeidae). Biol Trop 44(45):177–192
Kohlmann B, Solis A (1997) El género Dichotomius (Coleoptera: Scarabaeidae) en Costa Rica. G Ital Entomol 8:343–382
Kohlmann B, Solis A (2001) El género Onthophagus (Coleoptera: Scarabaeidae) en Costa Rica. G Ital Entomol 49:159–261
Kohlmann B, Solis A (2006) El género Canthidium (Coleoptera: Scarabaeidae) en Norteamérica. G Ital Entomol 11:235–295
Laurance WF, Sayer J, Cassman KG (2014) Agricultural expansion and its impacts on tropical nature. Trends Ecol Evol 29:107–116
Lobo JM, Martin-Piera E, Veiga CM (1988) Las trampas pitfall con cebo, sus posibilidades en el estudio de las comunidades coprófagas de Scarabaeoidea (Col.) I: Características determinantes de su capacidad de captura. Revue D’écologie Et De Biologie Du Sol 25:77–100
López-Bedoya PA, Bohada-Murillo M, Ángel-Vallejo MC, Audino LD, Davis ALV, Gurr G, Noriega JA (2022) Primary forest loss and degradation reduces biodiversity and ecosystem functioning: a global meta-analysis using dung beetles as an indicator taxon. J Appl Ecol. https://doi.org/10.1111/1365-2664.14167
Louzada JNC, Carvalho e Silva PR (2009) Utilisation of introduced Brazilian pastures ecosystems by native dung beetles: Diversity patterns and resource use. Insect Conserv Divers 2:45–52
Manning P, Slade EM, Beynon SA, Lewis OT (2016) Functionally rich dung beetle assemblages are required to provide multiple ecosystem services. Agr Ecosyst Environ 218:87–94
Majeed W, Rana N, de Azevedo Koch EB, Nargis S (2020) Seasonality and climatic factors affect diversity and distribution of arthropods around wetlands. Pak J Zool 52(6):2135–2144
McGeoch MA, Van Rensburg BJ, Botes A (2002) The verification and application of bioindicators: a case study of dung beetles in a savanna ecosystem. J Appl Ecol 39:661–672
Mendiburu F (2017) agricolae: Statistical Procedures for Agricultural Research. R package version 1.2-8. https://CRAN.R-project.org/package=agricolae
Mittermeier RA, Turner WR, Larsen FW, Brooks TM, Gascon C (2011) Global biodiversity conservation: The critical role of hotspots. In: Zachos F, Habel J (eds) Biodiversity Hotspots. Springer, Berlin, pp 3–22
Moreno CE (2001) Métodos para medir la biodiversidad. M&T-Manuales y Tesis SEA, vol 1. Zaragoza, España, p 83
Moreno CE, Barragán F, Pineda E, Pavón NP (2011) Reanálisis de la diversidad alfa: alternativas para interpretar y comparar información sobre comunidades ecológicas. Revista Mexicana de Biodiversidad 82(4):1249-1261.
Mosquera-Andrade DH (2014) Motores de la deforestación del bosque húmedo tropical bh-t de la región noroccidental colombiana. Rev Inst Univ Tecnol Chocó 33(2):96–104
Myers N, Mittermeier R, Mittermeier C et al (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Neita JC, Escobar F (2012) The potential value of agroforestry to dung beetle diversity in the wet tropical forests of the Pacific lowlands of Colombia. Agroforest Syst 85:121–131
Neita JC, Pardo LC, Quinto D, Cuesta NG (2003) Los escarabajos copronecrófilos (Coleoptera: Scarabaeidae) en la parcela permanente de investigación en biodiversidad (PPIB) en Salero, Unión Panamericana, Chocó. En: Salero: Diversidad biológica de un bosque pluvial tropical (bp-T). Garcia F, Ramos YA, Palacios JC, Arroyo JE, Mena A, González M (eds) Universidad Tecnológica del Chocó, Instituto de Investigaciones Ambientales del Pacífico – IIAP, Unión Panamericana, Chocó, pp 79–90
Neita-Moreno JC (2011) Escarabajos (Coleoptera: Scarabaeoidea) del departamento del Chocó, Colombia. Rev Biodivers Neotrop 1:17–27
Nervo B, Caprio E, Celi L, Lonati M, Rolando A (2017) Ecological functions provided by dung beetles are interlinked across space and time: evidence from 15N isotope tracing. Ecology 98:433–446
Nichols E, Gardner TA, Peres CA, Spector S, The Scarabaeinae Research Network (2009) Co-declining mammals and dung beetles: an impending ecological cascade. Oikos 118:481–487
Nichols E, Larsen T, Spector S, Davis AL, Escobar F, Favila F, Vulinec F, The Scarabaeinae Research Network (2007) Global dung beetle response to tropical forest modification and fragmentation: a quantitative literature review and meta-analysis. Biol Conserv 137:1–19
Noriega JA, Camero E, Arias-Buriticá J, Pardo-Locarno LC, Montes JM, Acevedo AA, Esparza A, Murcia B, García H, Solis C (2015) Grado de cobertura del muestreo de escarabajos coprófagos (Coleoptera: Scarabaeidae: Scarabaeinae) en Colombia. Rev Biol Trop 63(1):97–125
Noriega JA, Fagua G (2009) Monitoreo de escarabajos coprófagos (Coleoptera: Scarabaeidae) en la región neotropical. En: Acosta A, Zapata AM, Fagua G (eds) Técnicas de campo en ambientes tropicales: Manual para el monitoreo en ecosistemas acuáticos y artrópodos terrestres. Unidad de Ecología y Sistemática–UNESIS, Pontificia Universidad Javeriana Bogotá. pp 165–188
Noriega JA, March-Salas M, Castillo S, García-Q H, Hortal J, Santos AM (2021) Human perturbations reduce dung beetle diversity and dung removal ecosystem function. Biotropica 53(3):753–766
Noriega JA, Realpe E, Fagua G (2007) Diversidad de escarabajos coprófagos (Coleoptera: Scarabaeidae) en un bosque de galería con tres estadios de alteración. Univers Scientiarum 12:51–63
Noriega JA, Zapata-Prisco C, García H, Hernández E, Hernández J, Martínez R et al (2020) Does ecotourism impact biodiversity? An assessment using dung beetles (Coleoptera: Scarabaeinae) as bioindicators in a tropical dry forest natural park. Ecol Indicat 117:106580
Otavo SE, Parrado-Rosselli A, Noriega JA (2013) Superfamilia Scarabaeoidea (Insecta: Coleoptera) como elemento bioindicador de perturbación antropogénica en un Parque Nacional amazónico. Rev Biol Trop 61:735–752
Pardo-Locarno LC (2007) Escarabajos coprófagos (Coleoptera-Scarabaeidae) de Lloró, departamento del Chocó, Colombia. Bol Científico Museo Historia Natural Univ Caldas 11:377–388
Petchey OL, O’Gorman EJ, Flynn DFB (2009) A functional guide to functional diversity measures. Pp 49–59. In: Biodiversity, ecosystem functioning, and human wellbeing: and ecological and economic perspective. Naeem S, Bunker DE, Hector A, Loreau M, Perrings C (Eds). Oxford University Press, Oxford, UK
Pohlert T (2014) The Pairwise Multiple Comparison of Mean Ranks Package (PMCMR). R package version 4.3. https://CRAN.R-project.org/package=PMCMR
R Core Team (2018) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL: https://www.R-project.org/
Ramzan U, Majeed W, Rana N, Nargis S (2021) Occurrence of different insect species with emphasis on their abundance and diversity in different habitats of Faisalabad, Pakistan. Int J Trop Insect Sci 41(2):1237–1244
Rodrigues MM, Uchôa MA, Ide S (2013) Dung beetle (Coleoptera: Scarabaeoidea) in three landscapes in Mato Grosso do Sul, Brazil. Braz J Biol 73:211–220
Rodríguez-Mahecha JV, Rueda-Almonacid JV, Gutiérrez TD (2008) Guía ilustrada de la fauna del Santuario de Vida Silvestre Los Besotes, Valledupar, Cesar, Colombia. Serie de guías tropicales de campo Nº 7 Conservación Internacional. Editorial Panamericana. Bogotá, Colombia
Schoolmeesters P (2020) Scarabs: Scarabs: World Scarabaeidae Database (version 2019-11-02). In: Species 2000 & ITIS Catalogue of Life, 2020-04-16 Beta (Roskov Y, Ower G, Orrell T, Nicolson D, Bailly N, Kirk PM, Bourgoin T, DeWalt RE, Decock W, Nieukerken E, Penev (eds.). Digital resource at www.catalogueoflife.org/col. Species 2000: Naturalis, Leiden, the Netherlands
Shahabuddin S, Manuwoto S, Hidayat P, Noerdjito WA, Schulze CH (2008) The role of coprophagous beetles on dung decomposition and enhancement of soil fertility: Effect of body size, species diversity and biomass. J Biol Indonesia 5:109–119
Simmons LW, Ridsdill-Smith TJ (2011) Reproductive competition and its impact on the evolution and ecology of dung beetles. In: Simmons LW, Ridsdill-Smith TJ (eds) Ecology and evolution of dung beetles. John Wiley & Sons, Ltd., West Sussex, UK, pp 1–20
Slade EM, Mann DJ, Lewis OT (2011) Biodiversity and ecosystem functions of tropical forest dung beetles under contrasting logging regimes. Biol Cons 144:166–174
Slade EM, Mann DJ, Villanueva JF, Lewis OT (2007) Experimental evidence for the effects of dung beetle functional group richness and composition on ecosystem function in a tropical forest. J Anim Ecol 76:1094–1104
Spector S (2006) Scarabaeine dung beetles (Coleoptera: Scarabaeidae: Scarabaeinae): an invertebrate focal taxon for biodiversity research and conservation. Coleopts Bull 60:71–83
Vaz-De-Mello FZ, Edmonds WD, Ocampo FC, Schoolmeesters P (2011) A multilingual key to the genera and subgenera of the subfamily Scarabaeinae of the new world (Coleoptera: Scarabaeidae). Zootaxa 2854:1–73
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York, USA, p 481
Verdú JR, Arellano L, Numa C (2006) Thermoregulation in endothermic dung beetles (Coleoptera: Scarabaeidae): Effect of body size and ecophysiological constraints in flight. J Insect Physiol 52:854–860
Verdú JR, Lobo J (2008) Ecophysiology of thermoregulation in endothermic dung beetles: Ecological and geographical implications. Insect Ecol Conserv 661:1–28
Acknowledgements
The authors are grateful to the Centre of Applied Ecology and Sustainability (CAPES) for the opportunity to carry out this research. Also, thanks to Paula Andrea Amaya for contributing to the collecting and analysis of the samples. To Javier Santos, David Morris and Megan McFarland for kindly checking the English version of the manuscript.
Funding
To ANID PIA/BASAL FB0002 for funding.
Author information
Authors and Affiliations
Contributions
JPCC and JAN contributed to the study conception and design. JPCC performed data collection and laboratory identification. JAN performed laboratory identification and trait measurements. AG and JAN performed data analyses. JPCC and JAN wrote the first draft of the manuscript, and all authors commented on previous versions. All authors read and approved the final version of the manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Correa-Cuadros, J.P., Gómez-Cifuentes, A. & Noriega, J.A. Comparative effect of forest cutting and mammal hunting on dung beetle assemblages in Chocó Biogeographic forests in Colombia. Int J Trop Insect Sci 42, 3045–3055 (2022). https://doi.org/10.1007/s42690-022-00839-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-022-00839-x